The Influence of Maltodextrin and Inulin on the Physico-Chemical Properties of Cranberry Juice Powders
Abstract
1. Introduction
2. Materials and Methods
2.1. Reagents
2.2. Material
2.3. Methods
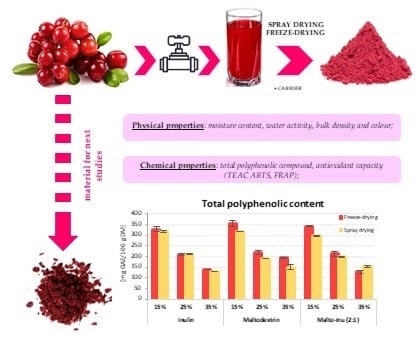
2.3.1. Drying Methods
2.3.2. Physical Properties
Moisture Content
Water Activity
Bulk Density
Color
2.3.3. Chemical Properties
Preparation of Extracts
Total Polyphenolic Compounds
Antioxidant Capacity
Statistical Analysis
3. Results and Discussion
3.1. Physical Properties
3.2. Chemical Properties
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hummer, K.E.; Lewers, K.S.; Bassil, N.V.; Vorsa, N.; Zalapa, J.E.; Iorizzo, M.; Tzanetakis, I. Cranberry Vulnerability Statement; GRIN: Munich, Germany, 2018. [Google Scholar]
- Borges, G.; Degeneve, A.; Mullen, W.; Crozier, A. Identification of flavonoid and phenolic antioxidants in black currants, blueberries, raspberries, red currants, and cranberries. J. Agric. Food Chem. 2009, 58, 3901–3909. [Google Scholar] [CrossRef]
- Sun, J.; Chu, Y.F.; Wu, X.; Liu, R.H. Antioxidant and antiproliferative activities of common fruits. J. Agric. Food Chem. 2002, 50, 7449–7454. [Google Scholar] [CrossRef]
- Blumberg, J.B.; Camesano, T.A.; Cassidy, A.; Kris-Etherton, P.; Howell, A.; Manach, C.; Vita, J.A. Cranberries and their bioactive constituents in human health. Adv. Nutr. Res. 2013, 4, 618–632. [Google Scholar] [CrossRef]
- Oszmiański, J.; Wojdyło, A.; Lachowicz, S.; Gorzelany, J.; Matłok, N. Comparison of bioactive potential of cranberry fruit and fruit-based products versus leaves. J. Funct. Foods 2016, 22, 232–242. [Google Scholar] [CrossRef]
- Michalska, A.; Wojdyło, A.; Honke, J.; Ciska, E.; Andlauer, W. Drying-induced physico-chemical changes in cranberry products. Food Chem. 2018, 240, 448–455. [Google Scholar] [CrossRef]
- Nowacka, M.; Wiktor, A.; Anuszewska, A.; Dadan, M.; Rybak, K.; Witrowa-Rajchert, D. The application of unconventional technologies as pulsed electric field, ultrasound and microwave-vacuum drying in the production of dried cranberry snacks. Ultrason. Sonochem. 2019, 56, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Oszmiański, J.; Kolniak-Ostek, J.; Lachowicz, S.; Gorzelany, J.; Matłok, N. Effect of dried powder preparation process on polyphenolic content and antioxidant capacity of cranberry (Vaccinium macrocarpon L.). Ind. Crops Prod. 2015, 77, 658–665. [Google Scholar] [CrossRef]
- Côté, J.; Caillet, S.; Doyon, G.J.; Dussault, D.; Sylvain, J.F.; Lacroix, M. Antimicrobial effect of cranberry juice and extracts. Food Control. 2011, 22, 1413–1418. [Google Scholar] [CrossRef]
- White, B.L.; Howard, L.R.; Prior, R.L. Impact of different stages of juice processing on the anthocyanin, flavonol, and procyanidin contents of cranberries. J. Agric. Food Chem. 2011, 59, 4692–4698. [Google Scholar] [CrossRef]
- Bhandari, B.R.; Senoussi, A.; Lebert, A. Spray drying of concentrated fruit juices. Dry. Technol. 1993, 11, 1081–1092. [Google Scholar] [CrossRef]
- Tonon, R.V.; Brabet, C.; Hubinger, M.D. Influence of process conditions on the physicochemical properties of açai (Euterpe oleraceae Mart.) powder produced by spray drying. J. Food Eng. 2008, 88, 411–418. [Google Scholar] [CrossRef]
- Caparino, O.A.; Tang, J.; Nindo, C.I.; Sablani, S.S.; Powers, J.R.; Fellman, J.K. Effect of drying methods on the physical properties and microstructures of mango (Philippine ‘Carabao’ var.) powder. J. Food Eng. 2012, 111, 135–148. [Google Scholar] [CrossRef]
- Oberoi, D.P.S.; Sogi, D.S. Effect of drying methods and maltodextrin concentration on pigment content of watermelon juice powder. J. Food Eng. 2015, 165, 172–178. [Google Scholar] [CrossRef]
- Kasprzyk, I.; Markowska, J.; Polak, E. Zamienniki tłuszczu w produktach spożywczych. Przem. Spoż. 2016, 70, 19–22. (In Polish) [Google Scholar]
- Fortuna, T.; Sobolewska, J. Maltodekstryny i ich wykorzystanie w przemyśle spożywczym. Żywn. Nauka Technol. Jakość 2000, 2, 100–109. (In Polish) [Google Scholar]
- Hashizume, C.; Kishimoto, Y.; Kanahori, S.; Yamamoto, T.; Okuma, K.; Yamamoto, K. Improvement effect of resistant maltodextrin in humans with metabolic syndrome by continuous administration. J. Nutr. Sci. Vitaminol. 2012, 58, 423–430. [Google Scholar] [CrossRef]
- Astina, J.; Sapwarobol, S. Resistant maltodextrin and metabolic syndrome: A review. J. Am. Coll. Nutr. 2019, 38, 380–385. [Google Scholar] [CrossRef]
- Ross, R.; Janssen, I.; Dawson, J.; Kungl, A.M.; Kuk, J.L.; Wong, S.L.; Nguyen-Duy, T.B.; Lee, S.; Kilpatrick, K.; Hudson, R. Exercise-induced reduction in obesity and insulin resistance in women: A randomized controlled trial. Obes. Res. 2004, 12, 789–798. [Google Scholar] [CrossRef]
- Nickerson, K.P.; McDonald, C. Crohn’s disease-associated adherent-invasive Escherichia Coli adhesion is enhanced by exposure to the ubiquitous dietary polysaccharide maltodextrin. PLoS ONE 2012, 7, e52132. [Google Scholar] [CrossRef]
- Nickerson, K.P.; Chanin, R.; McDonald, C. Deregulation of intestinal anti-microbial defense by the dietary additive, maltodextrin. Gut Microbes 2014, 6, 78–83. [Google Scholar] [CrossRef]
- Shoaib, M.; Shehzad, A.; Omar, M.; Rakha, A.; Raza, H.; Sharif, H.R.; Niazi, S. Inulin: Properties, health benefits and food applications. Carbohydr. Polym. 2016, 147, 444–454. [Google Scholar] [CrossRef] [PubMed]
- Marteau, P.; Seksik, P.; Lepage, P.; Doré, J. Cellular and physiological effects of probiotics and prebiotics. Mini Rev. Med. Chem. 2004, 4, 889–896. [Google Scholar] [CrossRef] [PubMed]
- Vogt, L.; Meyer, D.; Pullens, G.; Faas, M.; Smelt, M.; Venema, K.; De Vos, P. Immunological properties of inulin-type fructans. Crit. Rev. Food Sci. 2015, 55, 414–436. [Google Scholar] [CrossRef] [PubMed]
- Śliżewska, K.; Nowak, A.; Barczyński, R.; Libudzisz, Z. Prebiotyki—definicja, właściwości i zastosowanie w przemyśle. Żywn. Nauka Technol. Jakość 2013, 20, 5–20. (In Polish) [Google Scholar]
- Martín, M.A.; Ramos, S.; Mateos, R.; Marais, J.P.J.; Bravo-Clemente, L.; Khoo, C.; Goya, L. Chemical characterization and chemo-protective activity of cranberry phenolic powders in a model cell culture. Response of the antioxidant defenses and regulation of signaling pathways. Food Res. Int. 2015, 71, 68–82. [Google Scholar] [CrossRef]
- Figiel, A. Drying kinetics and quality of beetroots dehydrated by combination of convective and vacuum-microwave methods. J. Food Eng. 2010, 98, 461–470. [Google Scholar] [CrossRef]
- Shahidi, F.; Naczk, M. Methods of analysis and quantification of phenolic compounds. In Food Phenolic: Sources, Chemistry, Effects and Applications; Shahidi, F., Naczk, M., Eds.; Technomic Publishing Company: Lancaster, PA, USA, 1995; pp. 287–293. [Google Scholar]
- Re, R.; Pellegrini, N.; Proteggente, A.; Pannala, A.; Yang, M.; Rice-Evans, C. Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radic. Biol. Med. 1999, 26, 1231–1237. [Google Scholar] [CrossRef]
- Benzie, I.F.F.; Strain, J.J. Ferric reducing/antioxidant power assay: Direct measure of total antioxidant activity of biological fluids and modified version for simultaneous measurement of total antioxidant power and ascorbic acid concentration. In Oxidants and Antioxidants Part A. Methods Enzymol. 1999, 299, 15–27. [Google Scholar]
- Michalska, A.; Lech, K. The effect of carrier quantity and drying method on the physical properties of apple juice powders. Beverages 2018, 4, 2. [Google Scholar] [CrossRef]
- Quek, S.Y.; Chok, N.K.; Swedlund, P. The physicochemical properties of spray-dried watermelon powders. Chem. Eng. Process. 2007, 46, 386–392. [Google Scholar] [CrossRef]
- Kha, T.C.; Nguyen, M.H.; Roach, P.D. Effects of spray drying conditions on the physicochemical and antioxidant properties of the Gac (Momordica cochinchinensis) fruit aril powder. J. Food Eng. 2010, 98, 385–392. [Google Scholar] [CrossRef]
- Phoungchandang, S.; Sertwasana, A. Spray-drying of ginger juice and physicochemical properties of ginger powders. Sci. Asia 2010, 36, 40–45. [Google Scholar] [CrossRef]
- Tontul, I.; Topuz, A. Spray-drying of fruit and vegetable juices: Effect of drying conditions on the product yield and physical properties. Trends Food Sci. Technol. 2017, 63, 91–102. [Google Scholar] [CrossRef]
- Souza, B.W.S.; Cerqueira, M.A.; Teixeira, J.A.; Vicente, A.A. The use of electric fields for edible coatings and films development and production: A review. Food Eng. Rev. 2010, 2, 244–255. [Google Scholar] [CrossRef]
- Phisut, N. Spray drying technique of fruit juice powder: Some factors influencing the properties of product. Int. Food Res. J. 2012, 19, 1297–1306. [Google Scholar]
- Daza, L.D.; Fujita, A.; Fávaro-Trindade, C.S.; Rodrigues-Ract, J.N.; Granato, D.; Genovese, M.I. Effect of spray drying conditions on the physical properties of Cagaita (Eugenia dysenterica DC.) fruit extracts. Food Bioprod. Process. 2016, 97, 20–29. [Google Scholar] [CrossRef]
- Can Karaca, A.; Guzel, O.; Ak, M.M. Effects of processing conditions and formulation on spray drying of sour cherry juice concentrate. J. Sci. Food Agric. 2016, 96, 449–455. [Google Scholar] [CrossRef]
- Schuck, P. Dairy powders. In Handbook of Food Powders; Smithers, G.W., Augustin, M.A., Eds.; John Wiley Sons, Inc.: New York, NY, USA; The Institute of Food Technologists: Chicago, IL, USA, 2013; pp. 437–464. [Google Scholar]
- Bhusari, S.N.; Muzaffar, K.; Kumar, P. Effect of carrier agents on physical and microstructural properties of spray dried tamarind pulp powder. Powder Technol. 2014, 266, 354–364. [Google Scholar] [CrossRef]
- Papadakis, S.; Gardeli, C.; Tzia, C. Spray drying of raisin juice concentrate. Dry. Technol. 2006, 24, 173–180. [Google Scholar] [CrossRef]
- Goula, A.M.; Adamopoulos, K.G. Effect of Maltodextrin Addition during Spray Drying of Tomato Pulp in Dehumidified Air: II. Powder Properties. Dry. Technol. 2008, 26, 726–737. [Google Scholar] [CrossRef]
- Moghaddam, A.D.; Pero, M.; Askari, G.R. Optimizing spray drying conditions of sour cherry juice based on physicochemical properties, using response surface methodology (RSM). J. Food Sci. Technol. 2017, 54, 174–184. [Google Scholar] [CrossRef] [PubMed]
- Mishra, P.; Mishra, S.; Mahanta, C.L. Effect of maltodextrin concentration and inlet temperature during spray drying on physicochemical and antioxidant properties of amla (Emblica officinalis) juice powder. Food Bioprod. Process. 2014, 92, 252–258. [Google Scholar] [CrossRef]
- Daza, L.D.; Fujita, A.; Granato, D.; Silvia Fávaro-Trindade, C.; Inés Genovese, M. Functional properties of encapsulated Cagaita (Eugenia dysenterica DC.) fruit extract. Food Biosci. 2017, 18, 15–21. [Google Scholar] [CrossRef]
- Tonon, R.V.; Brabet, C.; Pallet, D.; Brat, P.; Hubinger, M.D. Physicochemical and morphological characterisation of açai (Euterpe oleraceae Mart.) powder produced with different carrier agents. Int. J. Food Sci. Technol. 2009, 44, 1950–1958. [Google Scholar] [CrossRef]
- Knorr, D. Technology aspects related to microorganisms in functional foods. Trends Food Sci. Technol. 1998, 9, 295–306. [Google Scholar] [CrossRef]
- Gouin, S. Microencapsulation: Industrial appraisal of existing technologies and trends. Trends Food Sci. Technol. 2004, 15, 330–347. [Google Scholar] [CrossRef]
- Michalska, A.; Wojdyło, A.; Łysiak, G.P.; Figiel, A. Chemical composition and antioxidant properties of powders obtained from different plum juice formulations. Int. J. Mol. Sci. 2017, 18, 176. [Google Scholar] [CrossRef]
- Vidović, S.S.; Vladić, J.Z.; Vaštag, Ž.G.; Zeković, Z.P.; Popović, L.M. Maltodextrin as a carrier of health benefit compounds in Satureja montana dry powder extract obtained by spray drying technique. Powder Technol. 2014, 258, 209–215. [Google Scholar] [CrossRef]
| Drying Method | Carrier | Concentration of Carrier | MC | aw | ρb |
|---|---|---|---|---|---|
| (%) | (%) | (-) | (g∙cm−3) | ||
| (-) | Inulin | (-) | 3.44 ± 0.01 bc | 0.15 ± 0.005 h | 0.72 ± 0.01 abc |
| Maltodextrin | 6.43 ± 0.03 de | 0.29 ± 0.002 k | 0.68 ± 0.14 abc | ||
| Maltodextrin–Inulin (2:1) | 5.90 ± 0.00 d | 0.25 ± 0.002 i | 0.89 ± 0.04 c | ||
| FD | Inulin | 15% | 4.34 ± 0.02 c | 0.06 ± 0.006 de | 0.47 ± 0.03 ab |
| 25% | 3.07 ± 0.02 bc | 0.03 ± 0.001 ab | 0.45 ± 0.01 ab | ||
| 35% | 2.50 ± 0.02 b | 0.02 ± 0.001 a | 0.56 ± 0.01 abc | ||
| Maltodextrin | 15% | 7.78 ± 0.12 f | 0.30 ± 0.001 l | 0.73 ± 0.02 abc | |
| 25% | 7.23 ± 0.04 ef | 0.40 ± 0.001 n | 0.72 ± 0.02 abc | ||
| 35% | 6.81 ± 0.01 def | 0.36 ± 0.001 l | 0.71 ± 0.00 abc | ||
| Maltodextrin–Inulin (2:1) | 15% | 7.86 ± 0.53 f | 0.29 ± 0.001 jk | 0.65 ± 0.01 abc | |
| 25% | 6.87 ± 1.29 def | 0.28 ± 0.001 j | 0.78 ± 0.02 bc | ||
| 35% | 6.28 ± 0.01 de | 0.39 ± 0.003 m | 0.76 ± 0.00 bc | ||
| SD | Inulin | 15% | 1.10 ± 0.04 a | 0.10 ± 0.001 g | 0.48 ± 0.00 ab |
| 25% | 0.75 ± 0.01 a | 0.06 ± 0.001 e | 0.60 ± 0.00 abc | ||
| 35% | 1.01 ± 0.10 a | 0.06 ± 0.001 e | 0.67 ± 0.02 abc | ||
| Maltodextrin | 15% | 0.95 ± 0.04 a | 0.08 ± 0.002 f | 0.62 ± 0.01 abc | |
| 25% | 0.58 ± 0.10 a | 0.05 ± 0.001 cd | 0.62 ± 0.02 abc | ||
| 35% | 0.73 ± 0.07 a | 0.04 ± 0.000 b | 0.50 ± 0.02 abc | ||
| Maltodextrin–Inulin (2:1) | 15% | 0.85 ± 0.05 a | 0.08 ± 0.003 f | 0.61 ± 0.01 abc | |
| 25% | 0.62 ± 0.01 a | 0.05 ± 0.002 cd | 0.61 ± 0.03 ab | ||
| 35% | 0.32 ± 0.01 a | 0.05 ± 0.002 c | 0.39 ± 0.01 a |
| Drying Method | Carrier | Concentration of Carrier | Color | ||
|---|---|---|---|---|---|
| (%) | L* | a* | b* | ||
| FD | Inulin | 15% | 62.33 ± 0.18 g | 35.92 ± 0.47 cd | 5.89 ± 0.17 c |
| 25% | 67.24 ± 0.05 j | 32.68 ± 0.14 b | 4.13 ± 0.02 b | ||
| 35% | 70.22 ± 0.20 l | 29.08 ± 0.49 a | 3.42 ± 0.07 a | ||
| Maltodextrin | 15% | 51.05 ± 0.40 a | 43.37 ± 0.42 kl | 11.13 ± 0.40 j | |
| 25% | 57.88 ± 0.55 c | 39.07 ± 0.31 hi | 8.54 ± 0.20 g | ||
| 35% | 60.26 ± 0.55 ef | 37.67 ± 0.94 fg | 7.41 ± 0.39 e | ||
| Maltodextrin–Inulin (2:1) | 15% | 54.57 ± 0.42 b | 40.31 ± 0.58 j | 9.33 ± 0.27 h | |
| 25% | 59.19 ± 0.30 d | 38.17 ± 0.50 gh | 8.22 ± 0.24 fg | ||
| 35% | 61.64 ± 0.10 g | 36.41 ± 0.07 de | 7.28 ± 0.04 e | ||
| SD | Inulin | 15% | 59.99 ± 0.12 ef | 44.29 ± 0.27 l | 11.5 ± 0.11 j |
| 25% | 64.16 ± 0.27 h | 40.85 ± 0.21 k | 9.64 ± 0.06 h | ||
| 35% | 65.19 ± 0.34 i | 39.64 ± 0.57 jk | 9.30 ± 0.11 h | ||
| Maltodextrin | 15% | 60.68 ± 0.24 f | 42.28 ± 0.08 k | 10.28 ± 0.10 i | |
| 25% | 65.28 ± 0.47 i | 39.33 ± 0.18 hi | 8.14 ± 0.09 fg | ||
| 35% | 68.15 ± 0.18 k | 37.45 ± 0.11 efg | 6.70 ± 0.02 d | ||
| Maltodextrin–Inulin (2:1) | 15% | 59.91 ± 0.32 de | 42.59 ± 0.40 k | 10.57 ± 0.15 i | |
| 25% | 66.60 ± 0.30 j | 36.70 ± 1.58 def | 7.95 ± 0.41 f | ||
| 35% | 70.40 ± 0.07 l | 34.81 ± 0.40 c | 6.55 ± 0.10 d | ||
| Drying Method | Carrier | Concentration of Carrier | TPC | TEAC ABTS | FRAP |
|---|---|---|---|---|---|
| (%) | (mg GAE/100g DM) | (mmol Trolox/100g DM) | |||
| FD | Inulin | 15% | 328.24 ± 9.71f | 1.78 ± 0.09 d | 1.67 ± 0.10 c |
| 25% | 208.36 ± 4.62 cd | 1.09 ± 0.10 cd | 1.10 ± 0.10 b | ||
| 35% | 137.66 ± 0.86 ab | 0.77 ± 0.05 a | 0.80 ± 0.06 a | ||
| Maltodextrin | 15% | 354.09 ± 14.90 g | 1.71 ± 0.04 d | 1.77 ± 0.06 c | |
| 25% | 219.81 ± 9.24 d | 1.16 ± 0.05 cd | 1.07 ± 0.09 b | ||
| 35% | 192.78 ± 2.34 c | 1.05 ± 0.07 bc | 0.95 ± 0.07ab | ||
| Maltodextrin–Inulin (2:1) | 15% | 341.33 ± 3.03 fg | 1.71 ± 0.16 d | 1.65 ± 0.12 c | |
| 25% | 211.79 ± 13.02 cd | 1.10 ± 0.03 cd | 1.07 ± 0.12 b | ||
| 35% | 127.76 ± 7.20 a | 0.77 ± 0.05 a | 0.79 ± 0.04 a | ||
| SD | Inulin | 15% | 316.47 ± 3.95 ef | 1.75 ± 0.03 d | 1.69 ± 0.04 c |
| 25% | 209.72 ± 2.74 cd | 1.30 ± 0.03 d | 1.11 ± 0.05 b | ||
| 35% | 129.26 ± 1.61 ab | 0.80 ± 0.02 a | 0.82 ± 0.05 a | ||
| Maltodextrin | 15% | 316.48 ± 1.09 ef | 1.70 ± 0.09 d | 1.64 ± 0.06 c | |
| 25% | 190.91 ± 0.33 c | 1.12 ± 0.08 cd | 1.13 ± 0.07 b | ||
| 35% | 150.34 ± 10.86 ab | 0.93 ± 0.07 abc | 0.92 ± 0.02 ab | ||
| Maltodextrin–Inulin (2:1) | 15% | 295.86 ± 2.53 e | 1.80 ± 0.06 d | 1.64 ± 0.03 c | |
| 25% | 198.77 ± 1.57 cd | 1.18 ± 0.00 cd | 1.09 ± 0.04 b | ||
| 35% | 155.01 ± 4.92 b | 0.82 ± 0.06 ab | 0.81 ± 0.05 a | ||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Michalska-Ciechanowska, A.; Majerska, J.; Brzezowska, J.; Wojdyło, A.; Figiel, A. The Influence of Maltodextrin and Inulin on the Physico-Chemical Properties of Cranberry Juice Powders. ChemEngineering 2020, 4, 12. https://doi.org/10.3390/chemengineering4010012
Michalska-Ciechanowska A, Majerska J, Brzezowska J, Wojdyło A, Figiel A. The Influence of Maltodextrin and Inulin on the Physico-Chemical Properties of Cranberry Juice Powders. ChemEngineering. 2020; 4(1):12. https://doi.org/10.3390/chemengineering4010012
Chicago/Turabian StyleMichalska-Ciechanowska, Anna, Joanna Majerska, Jessica Brzezowska, Aneta Wojdyło, and Adam Figiel. 2020. "The Influence of Maltodextrin and Inulin on the Physico-Chemical Properties of Cranberry Juice Powders" ChemEngineering 4, no. 1: 12. https://doi.org/10.3390/chemengineering4010012
APA StyleMichalska-Ciechanowska, A., Majerska, J., Brzezowska, J., Wojdyło, A., & Figiel, A. (2020). The Influence of Maltodextrin and Inulin on the Physico-Chemical Properties of Cranberry Juice Powders. ChemEngineering, 4(1), 12. https://doi.org/10.3390/chemengineering4010012