Dynamic Mechanical Analysis as a Complementary Technique for Stickiness Determination in Model Whey Protein Powders
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Powder Composition
2.3. Surface Composition
2.4. Moisture Sorption Isotherms
2.5. Stickiness by Fluidization
2.6. Powder Equilibration
2.7. Differential Scanning Calorimetry
2.8. Dynamic Mechanical Analysis
2.9. Statistical Analysis
3. Results and Discussion
3.1. Bulk and Surface Composition
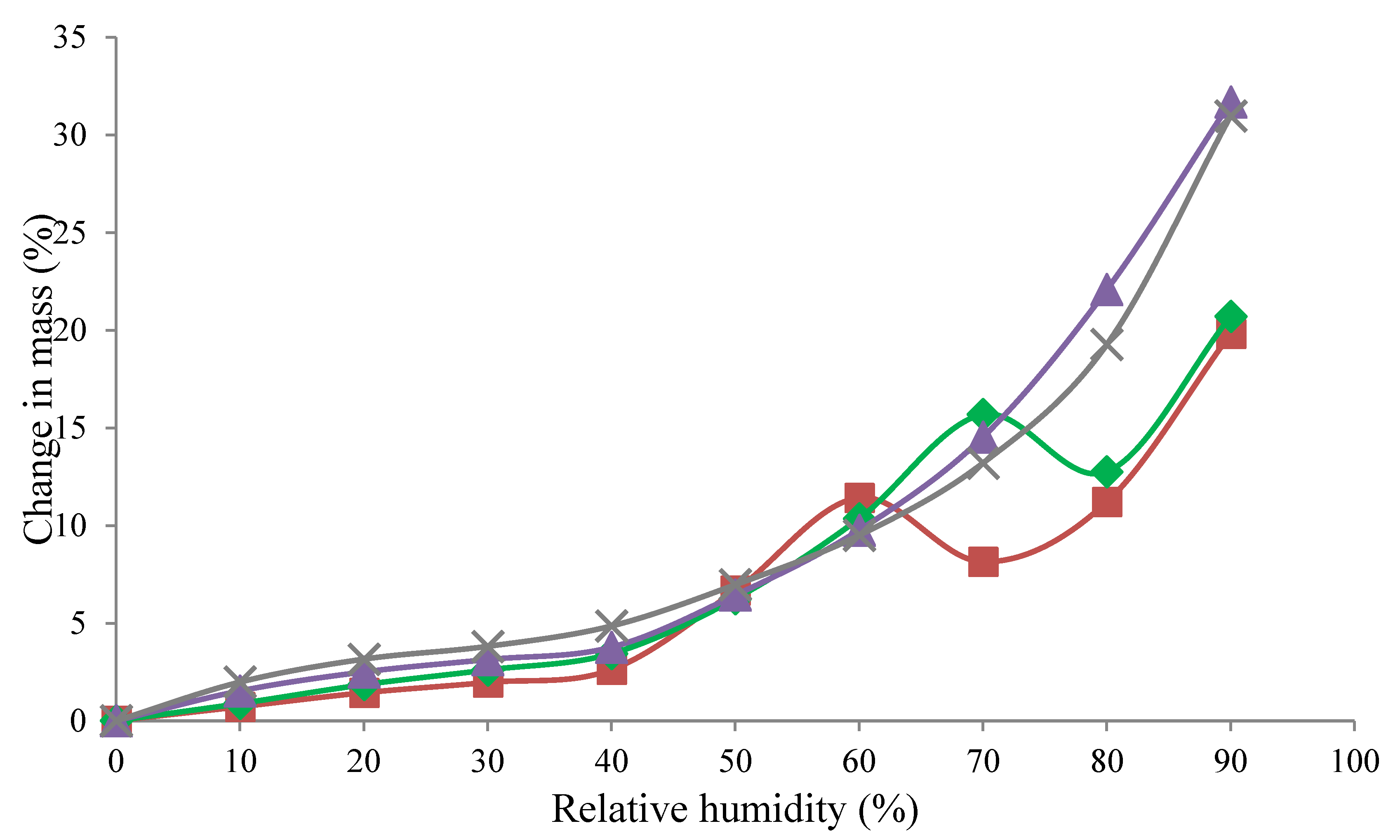
3.2. Moisture Sorption Isotherms
3.3. Glass Transition Temperature Determination
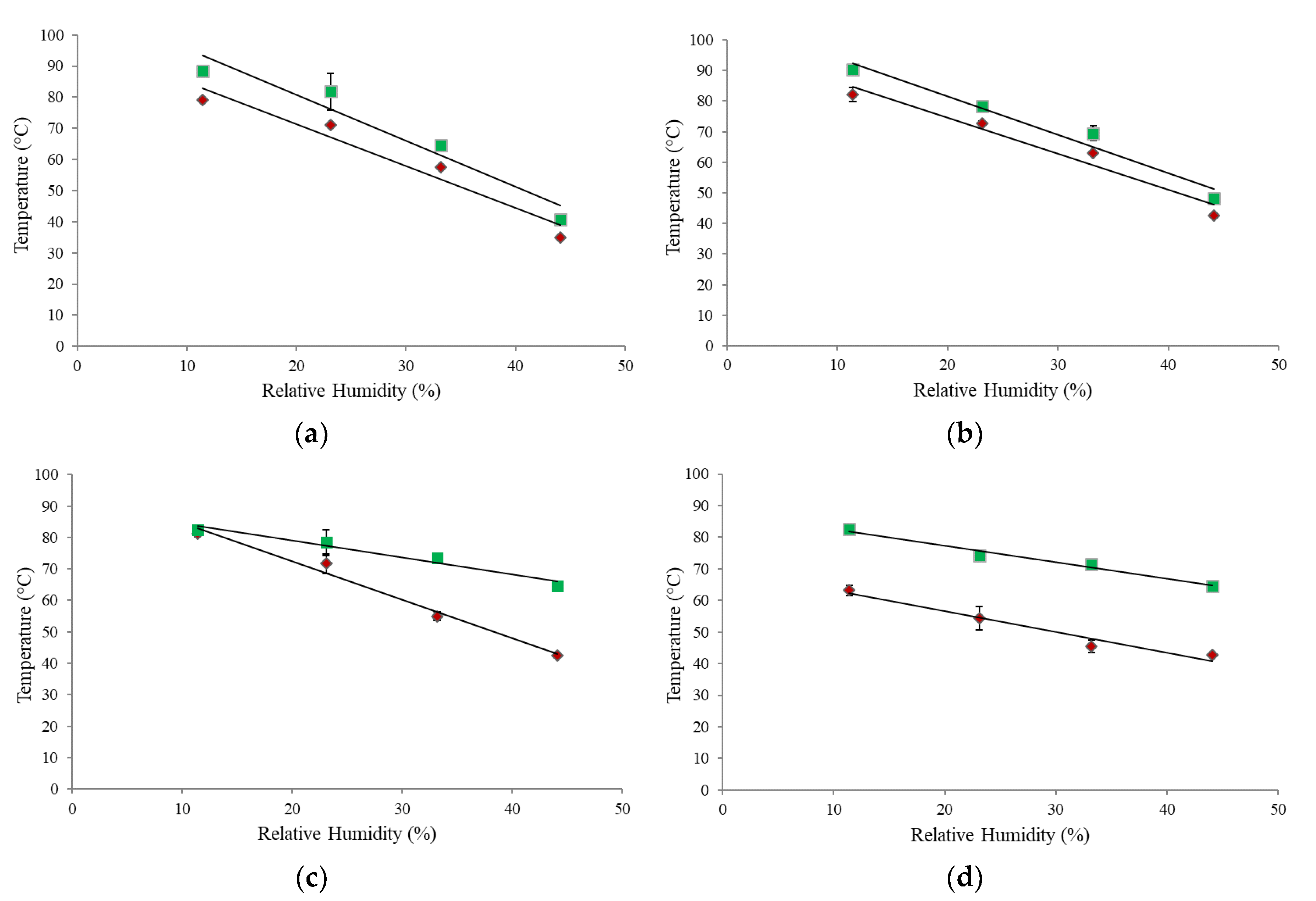
3.4. Powder Fluidization Analysis
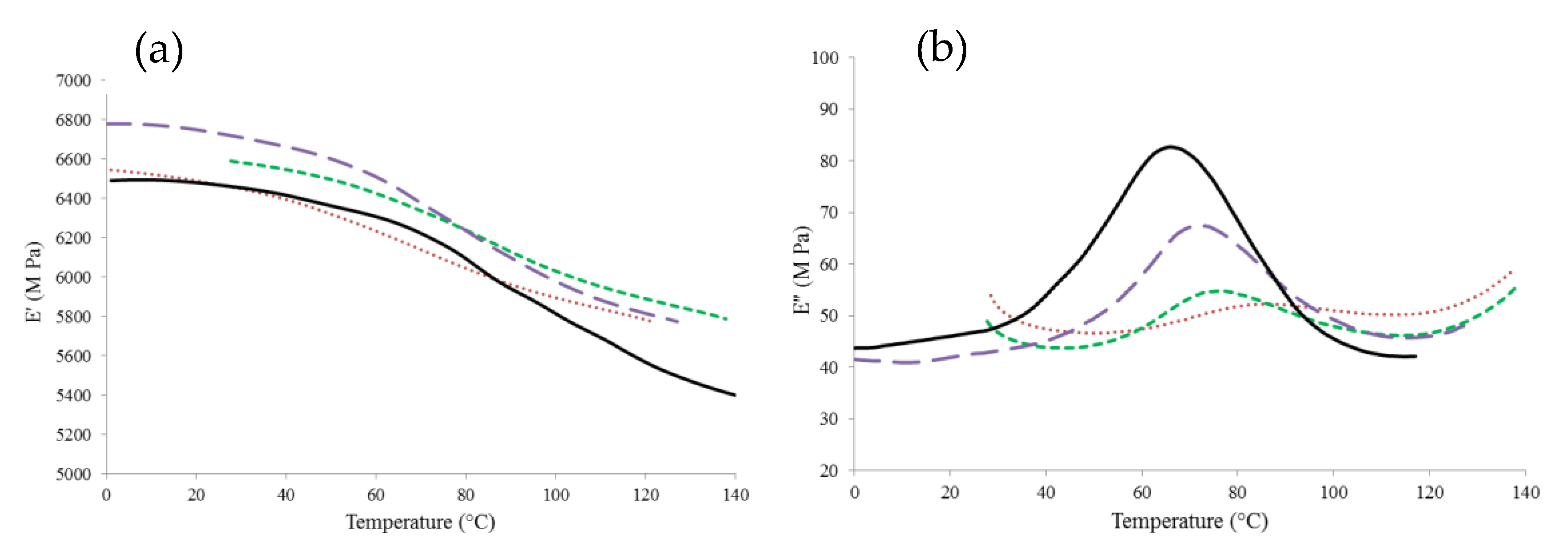
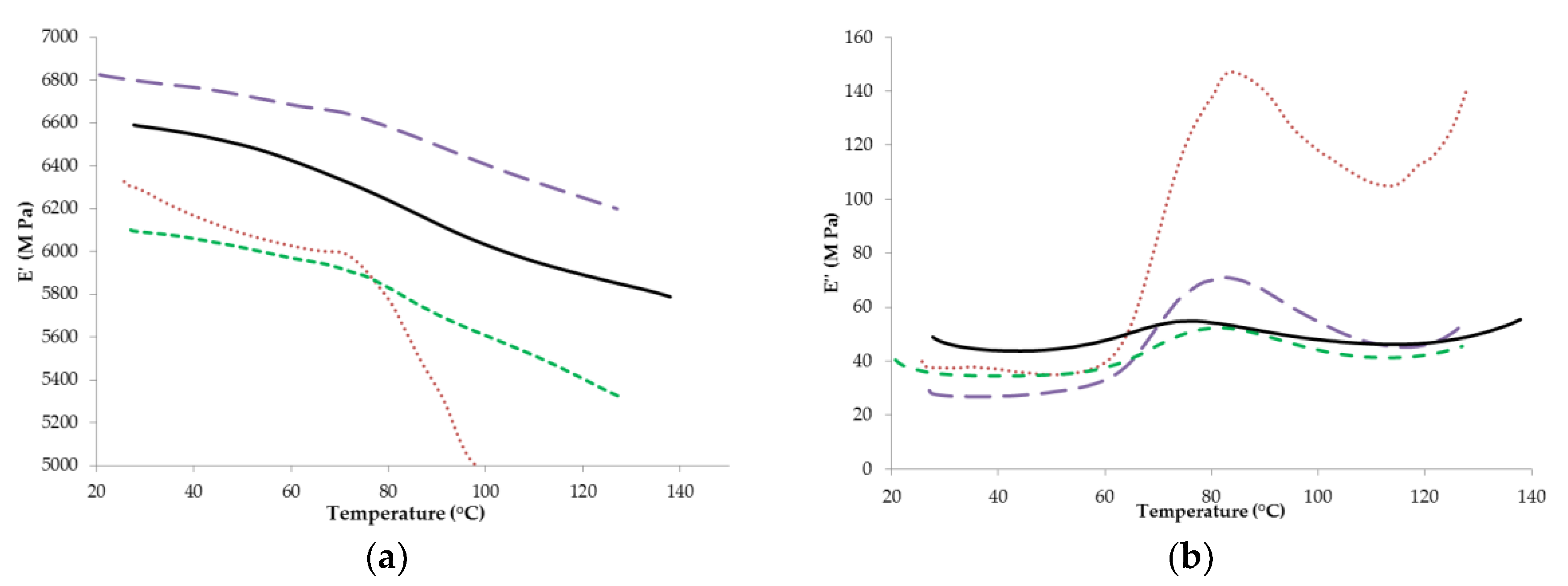
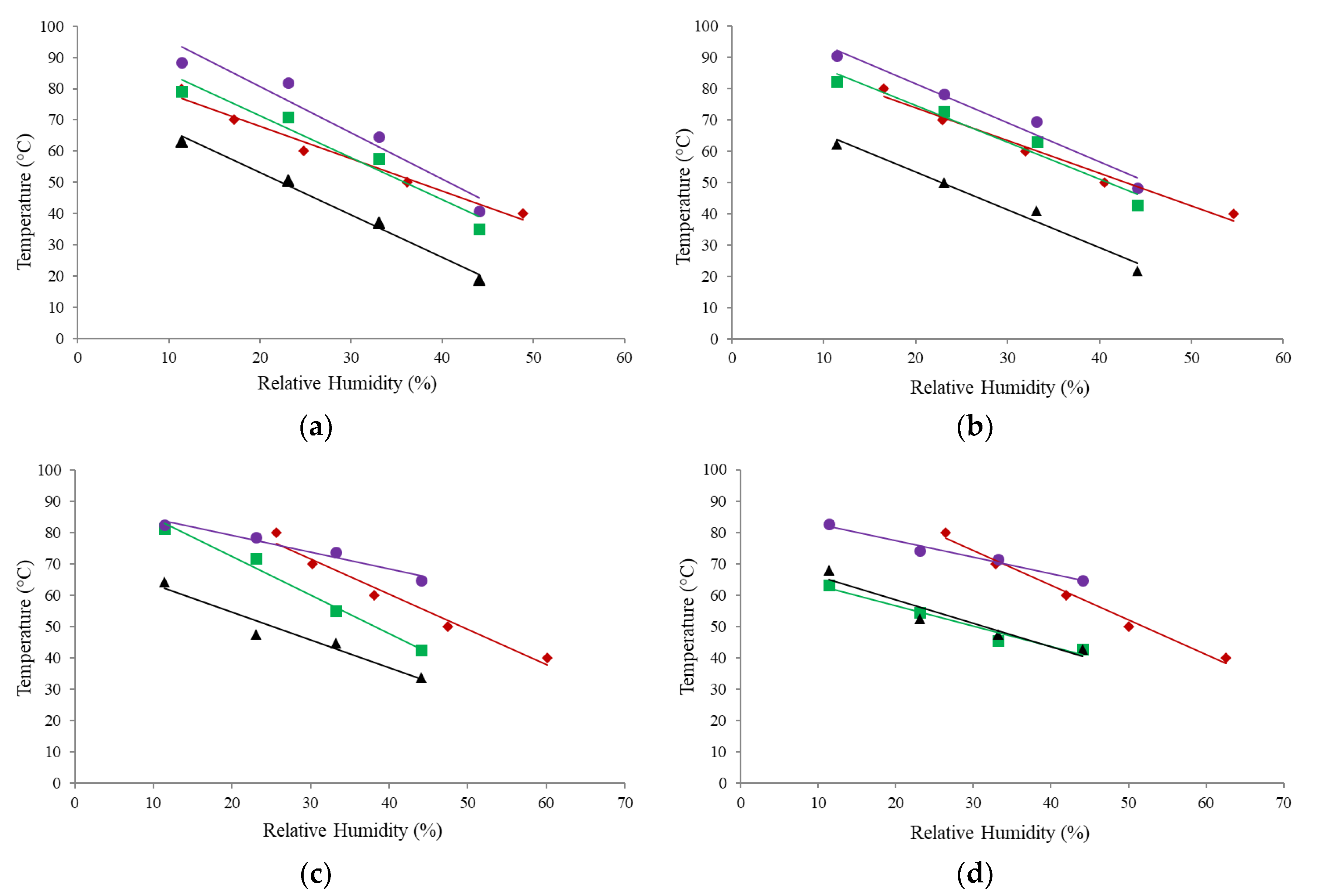
3.5. Dynamic Mechanical Analysis
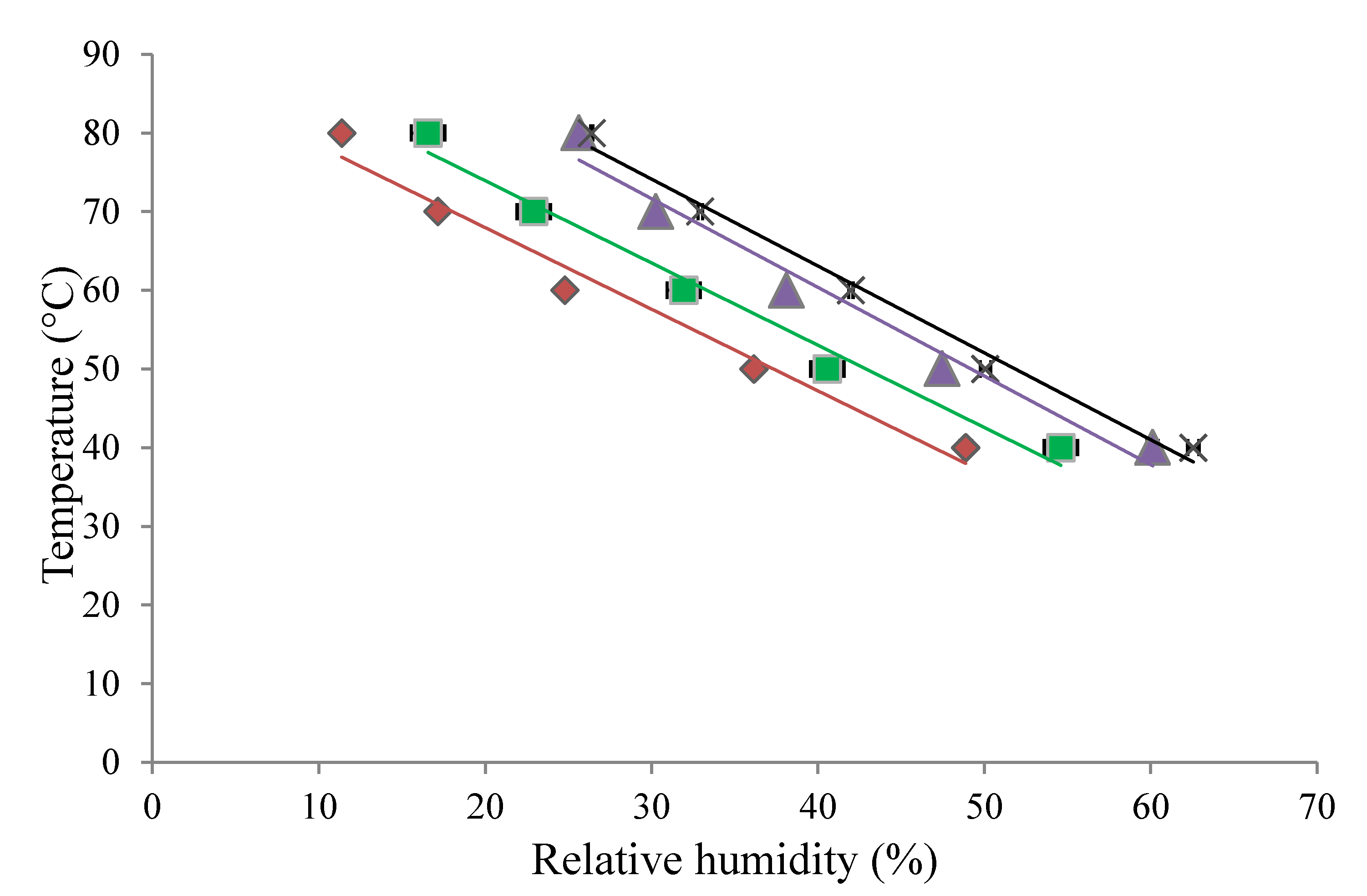
3.6. Comparison of α-Relaxation, Stickiness, and Glass Transition Curves
3.7. Comparison of T–Tg Results from Different Measurement Techniques
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Chuy, L.E.; Labuza, T.P. Caking and stickiness of dairy-based food powders as related to glass transition. J. Food Sci. 1994, 59, 43–46. [Google Scholar] [CrossRef]
- Paterson, A.H.J.; Bronlund, J.E.; Brooks, G.F. The blow test for measuring the stickiness of powders. In Proceedings of the AIChE 2001 Annual Meeting, Reno, NV, USA, 4–9 November 2001. [Google Scholar]
- Özkan, N.; Walisinghe, N.; Chen, X.D. Characterization of stickiness and cake formation in whole and skim milk powders. J. Food Eng. 2002, 55, 293–303. [Google Scholar] [CrossRef]
- Zuo, J.Y.; Paterson, A.H.J.; Bronlund, J.E.; Chatterjee, R. Using a particle-gun to measure initiation of stickiness of dairy powders. Int. Dairy J. 2007, 17, 268–273. [Google Scholar] [CrossRef]
- Hogan, S.; O’Callaghan, D.; Bloore, G. Application of fluidised bed stickiness apparatus to dairy powder production. Milchwissenschaft 2009, 64, 308–311. [Google Scholar]
- Silalai, N.; Roos, Y.H. Mechanical relaxation times as indicators of stickiness in skim milk–maltodextrin solids systems. J. Food Eng. 2011, 106, 306–317. [Google Scholar] [CrossRef]
- Lazar, M.; Brown, A.; Smith, G.; Wong, F.; Lindquist, F. Experimental production of tomato powder by spray drying. Food Technol. 1956, 10, 129–134. [Google Scholar]
- Hennigs, C.; Kockel, T.; Langrish, T. New measurements of the sticky behavior of skim milk powder. Dry. Technol. 2001, 19, 471–484. [Google Scholar] [CrossRef]
- Murti, R.A.; Paterson, A.H.J.; Pearce, D.L.; Bronlund, J.E. The influence of particle velocity on the stickiness of milk powder. Int. Dairy J. 2010, 20, 121–127. [Google Scholar] [CrossRef]
- Brooks, G.F. The Sticking and Crystallisation of Amorphous Lactose. Master’s Thesis, Massey University, Auckland, New Zeland, 2000. [Google Scholar]
- Couchman, P.; Karasz, F. A classical thermodynamic discussion of the effect of composition on glass-transition temperatures. Macromolecules 1978, 11, 117–119. [Google Scholar] [CrossRef]
- Jouppila, K.; Roos, Y. Glass transitions and crystallization in milk powders. J. Dairy Sci. 1994, 77, 2907–2915. [Google Scholar] [CrossRef]
- Ozmen, L.; Langrish, T. Comparison of glass transition temperature and sticky point temperature for skim milk powder. Dry. Technol. 2002, 20, 1177–1192. [Google Scholar] [CrossRef]
- Haque, M.K.; Roos, Y. Water plasticization and crystallization of lactose in spray-dried lactose/protein mixtures. J. Food Sci. 2004, 69, FEP23–FEP29. [Google Scholar] [CrossRef]
- Haque, M.K.; Roos, Y. Water sorption and plasticization behavior of spray-dried lactose/protein mixtures. J. Food Sci. 2004, 69, E384–E391. [Google Scholar] [CrossRef]
- Silalai, N.; Roos, Y.H. Roles of water and solids composition in the control of glass transition and stickiness of milk powders. J. Food Sci. 2010, 75, E285–E296. [Google Scholar] [CrossRef]
- Maidannyk, V.; Roos, Y. Water sorption, glass transition and “strength” of lactose-whey protein systems. Food Hydrocoll. 2017, 70, 76–87. [Google Scholar] [CrossRef]
- O’Donoghue, L.T.; Haque, M.K.; Kennedy, D.; Laffir, F.R.; Hogan, S.A.; O’Mahony, J.A.; Murphy, E.G. Influence of particle size on the physicochemical properties and stickiness of dairy powders. Int. Dairy J. 2019, 98, 54–63. [Google Scholar] [CrossRef]
- Roos, Y.; Karel, M. Plasticizing effect of water on thermal behavior and crystallization of amorphous food models. J. Food Sci. 1991, 56, 38–43. [Google Scholar] [CrossRef]
- Karel, M.; Anglea, S.; Buera, P.; Karmas, R.; Levi, G.; Roos, Y. Stability-related transitions of amorphous foods. Thermochim. Acta 1994, 246, 249–269. [Google Scholar] [CrossRef]
- Boonyai, P.; Bhandari, B.; Howes, T. Stickiness measurement techniques for food powders: A review. Powder Technol. 2014, 145, 34–46. [Google Scholar] [CrossRef]
- Murti, R.A.; Paterson, A.H.J.; Pearce, D.L.; Bronlund, J.E. Stickiness of skim milk powder using the particle gun technique. Int. Dairy J. 2009, 19, 137–141. [Google Scholar] [CrossRef]
- Silalai, N.; Roos, Y.H. Coupling of dielectric and mechanical relaxations with glass transition and stickiness of milk solids. J. Food Eng. 2011, 104, 445–454. [Google Scholar] [CrossRef]
- Fan, F.; Roos, Y.H. Structural relaxations of amorphous lactose and lactose-whey protein mixtures. J. Food Eng. 2016, 173, 106–115. [Google Scholar] [CrossRef]
- Fan, F.; Roos, Y.H. Structural strength and crystallization of amorphous lactose in food model solids at various water activities. Inn. Food Sci. Emerg. Technol. 2017, 40, 27–34. [Google Scholar] [CrossRef]
- IDF. Determination of Fat Content—Rose Gottlieb Reference Method. IDF Standard 9C; International Dairy Federation: Brussels, Belgium, 1987. [Google Scholar]
- Faldt, P.; Bergenstahl, B.; Carlsson, G. The surface coverage of fat on food powders analyzed by ESCA (electron spectroscopy for chemical analysis). Food Struct. 1993, 12, 10. [Google Scholar]
- Murphy, E.G.; Roos, Y.H.; Hogan, S.A.; Maher, P.G.; Flynn, C.G.; Fenelon, M.A. Physical stability of infant milk formula made with selectively hydrolysed whey proteins. Int. Dairy J. 2015, 40, 39–46. [Google Scholar] [CrossRef]
- Nijdam, J.; Langrish, T. The effect of surface composition on the functional properties of milk powders. J. Food Eng. 2006, 77, 919–925. [Google Scholar] [CrossRef]
- Shrestha, A.K.; Howes, T.; Adhikari, B.P.; Wood, B.J.; Bhandari, B.R. Effect of protein concentration on the surface composition, water sorption and glass transition temperature of spray-dried skim milk powders. Food Chem. 2007, 104, 1436–1444. [Google Scholar] [CrossRef]
- Kim, E.H.-J.; Chen, X.D.; Pearce, D. Surface composition of industrial spray-dried milk powders. 2. Effects of spray drying conditions on the surface composition. J. Food Eng. 2009, 94, 169–181. [Google Scholar] [CrossRef]
- Foerster, M.; Gengenbach, T.; Woo, M.W.; Selomulya, C. The impact of atomization on the surface composition of spray-dried milk droplets. Colloids Surf. B Biointerfaces 2016, 140, 460–471. [Google Scholar] [CrossRef]
- Maidannyk, V.; McSweeney, D.J.; Hogan, S.A.; Miao, S.; Montgomery, S.; Auty, M.A.; McCarthy, N.A. Water sorption and hydration in spray-dried milk protein powders: Selected physicochemical properties. Food Chem. 2020, 304, 125418. [Google Scholar] [CrossRef]
- Berlin, E.; Anderson, B.A.; Pallansch, M.J. Comparison of water vapor sorption by milk powder components. J. Dairy Sci. 1968, 51, 1912–1915. [Google Scholar] [CrossRef]
- Foster, K.D.; Bronlund, J.E.; Paterson, A.H.J. The prediction of moisture sorption isotherms for dairy powders. Int. Dairy J. 2005, 15, 411–418. [Google Scholar] [CrossRef]
- Hogan, S.; O’Callaghan, D. Influence of milk proteins on the development of lactose-induced stickiness in dairy powders. Int. Dairy J. 2010, 20, 212–221. [Google Scholar] [CrossRef]
- Downton, G.E.; Flores-Luna, J.L.; King, C.J. Mechanism of stickiness in hygroscopic, amorphous powders. Ind. Eng. Chem. Fund. 1982, 21, 447–451. [Google Scholar] [CrossRef]
- Menard, K.P. Dynamic Mechanical Analysis. In Encyclopedia of Polymer Science and Technology; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2002; pp. 1–33. [Google Scholar]
- Kalichevsky, M.; Jaroszkiewicz, E.; Ablett, S.; Blanshard, J.; Lillford, P. The glass transition of amylopectin measured by DSC, DMTA and NMR. Carbohydr. Polym. 1992, 18, 77–88. [Google Scholar] [CrossRef]
- Rahman, M.S.; Al-Marhubi, I.M.; Al-Mahrouqi, A. Measurement of glass transition temperature by mechanical (DMTA), thermal (DSC and MDSC), water diffusion and density methods: A comparison study. Chem. Phys. Lett. 2007, 440, 372–377. [Google Scholar] [CrossRef]
- Kararli, T.T.; Hurlbut, J.B.; Needham, T.E. Glass-rubber transitions of cellulosic polymers by dynamic mechanical analysis. J. Pharm. Sci. 1990, 79, 845–848. [Google Scholar] [CrossRef]
- Hallberg, L.; Chinachoti, P. Dynamic mechanical analysis for glass transitions in long shelf-life bread. J. Food Sci. 1992, 57, 1201–1229. [Google Scholar] [CrossRef]
- Siebenmorgen, T.; Yang, W.; Sun, Z. Glass transition temperature of rice kernels determined by dynamic mechanical thermal analysis. Trans. ASAE 2004, 47, 835. [Google Scholar] [CrossRef]
- Bengoechea, C.; Arrachid, A.; Guerrero, A.; Hill, S.E.; Mitchell, J.R. Relationship between the glass transition temperature and the melt flow behavior for gluten, casein and soya. J. Cereal Sci. 2007, 45, 275–284. [Google Scholar] [CrossRef]
- Schuck, P.; Dolivet, A.; Méjean, S.; Jeantet, R. Relative humidity of outlet air: The key parameter to optimize moisture content and water activity of dairy powders. Dairy Sci. Technol. 2008, 88, 45–52. [Google Scholar] [CrossRef]
| Sample | Protein (% w/w) | Fat (% w/w) | Lactose * (% w/w) | Ash (% w/w) | Free Moisture (% w/w) ** | Particle Size D4,3 *** (µm) |
|---|---|---|---|---|---|---|
| WPC 20 | 19.3 ± 0.02 a | 1.31 ± 0.04 a | 66.2 | 7.34 ± 0.01 a | 5.90 ± 0.01 a | 106 |
| WPC 35 | 35.7 ± 0.20 b | 3.19 ± 0.06 b | 48.6 | 6.36 ± 0.00 b | 6.11 ± 0.08 b | 118 |
| WPC 50 | 53.4 ± 0.15 c | 4.15 ± 0.02 c | 32.8 | 5.59 ± 0.24 c | 4.07 ± 0.14 c | 118 |
| WPC 65 | 69.1 ± 0.38 d | 5.33 ± 0.03 d | 17.5 | 4.23 ± 0.00 d | 3.90 ± 0.05 d | 115 |
| Sample | Protein (%) | Lactose (%) | Fat (%) |
|---|---|---|---|
| WPC 20 | 46.8 ± 0.50 a | 47.8 ± 0.11 a | 4.07 ± 0.38 a |
| WPC 35 | 48.9 ± 5.48 a,b | 33.8 ± 0.82 b | 16.7 ± 6.39 a,b |
| WPC 50 | 52.5 ± 1.49 a,b | 23.5 ± 2.29 c | 23.9 ± 3.70 b |
| WPC 65 | 59.2 ± 4.98 b | 13.6 ± 2.75 d | 27.2 ± 7.76 b |
| Sample | 0.11 aw | 0.23 aw | 0.33 aw | 0.44 aw |
|---|---|---|---|---|
| WPC 20 | 63.2 ± 0.56 a | 50.6 ± 0.00 a | 37.1 ± 0.25 a | 18.6 ± 0.07 a |
| WPC 35 | 62.2 ± 0.01 b | 49.9 ± 0.17 b | 40.9 ± 0.60 b | 21.6 ± 0.01 b |
| WPC 50 | 64.4 ± 0.13 c | 47.6 ± 0.30 c | 44.8 ± 0.26 c | 33.7 ± 0.42 c |
| WPC 65 | 67.8 ± 0.23 d | 52.4 ± 0.10 d | 47.3 ± 0.23 d | 42.7 ± 0.03 d |
| Method | Sample | T–Tg at Midpoint (°C) | T–Tg at 15% RH (°C) |
|---|---|---|---|
| Fluidization, Tf | WPC 20 | 18.1 | 13.2 |
| WPC 35 | 23.1 | 19.9 | |
| WPC 50 | 22.7 | 29.5 | |
| WPC 65 | 18.0 | 28.4 | |
| DMA, Tα onset | WPC 20 | 18.3 | 18.1 |
| WPC 35 | 21.5 | 21.1 | |
| WPC 50 | 15.1 | 19.5 | |
| WPC 65 | −1.14 | −2.35 | |
| DMA, Tα peak | WPC 20 | 26.6 | 28.1 |
| WPC 35 | 27.9 | 28.4 | |
| WPC 50 | 27.1 | 22.7 | |
| WPC 65 | 20.6 | 17.7 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
O’Donoghue, L.T.; Haque, M.K.; Hogan, S.A.; Laffir, F.R.; O’Mahony, J.A.; Murphy, E.G. Dynamic Mechanical Analysis as a Complementary Technique for Stickiness Determination in Model Whey Protein Powders. Foods 2020, 9, 1295. https://doi.org/10.3390/foods9091295
O’Donoghue LT, Haque MK, Hogan SA, Laffir FR, O’Mahony JA, Murphy EG. Dynamic Mechanical Analysis as a Complementary Technique for Stickiness Determination in Model Whey Protein Powders. Foods. 2020; 9(9):1295. https://doi.org/10.3390/foods9091295
Chicago/Turabian StyleO’Donoghue, Laura T., Md. Kamrul Haque, Sean A. Hogan, Fathima R. Laffir, James A. O’Mahony, and Eoin G. Murphy. 2020. "Dynamic Mechanical Analysis as a Complementary Technique for Stickiness Determination in Model Whey Protein Powders" Foods 9, no. 9: 1295. https://doi.org/10.3390/foods9091295
APA StyleO’Donoghue, L. T., Haque, M. K., Hogan, S. A., Laffir, F. R., O’Mahony, J. A., & Murphy, E. G. (2020). Dynamic Mechanical Analysis as a Complementary Technique for Stickiness Determination in Model Whey Protein Powders. Foods, 9(9), 1295. https://doi.org/10.3390/foods9091295





