1. Introduction
“Amaretti” cookies are a renowned Italian pastry, appreciated for their bitter-sweet taste and almond flavor. The amaretti recipe is quite simple, since they are obtained by grinding and mixing together sweet and bitter almonds (or alternatively using almond extract), sucrose and egg white. The cookies are characterized by a delicate almond flavor, a crispy crust, with low water activity (a
w), and a softer inner part, which retains most of the moisture [
1]. Therefore, according to the definition by Labuza and Hyman [
2], amaretti can be considered a macromolecular multi-domain system. Regions at different water activities in multi-domain foods cause the whole system to be in a non-equilibrium state. This results in moisture migration from the higher a
w to the lower a
w region. Shelf life limiting factors of amaretti cookies are the progressive firming, due mostly to moisture loss and sucrose recrystallization, during storage [
3]. As a consequence, texture may move from rubbery to grainy. Sucrose recrystallization is related to moisture migration between components, and is accompanied by an increase in a
w values [
4], with the potential exposure to microbial spoilage (mold growth), as a
w approaches 0.8. Packaging solutions [
3,
5] and addition of fortifiers [
6] have been studied to extend the shelf life of almond pastries. No contributions have been reported on the impact of different almond varieties on the shelf life and acceptability of this type of product. Almonds are characterized by a high lipid and protein contents. Lipids may act as moisture barriers to moisture migration in a multi-domain system, largely depending on their composition, the pore size in the matrix of the food domain, crystals or lipid interferences in the system. Fat acts as a lubricant and contributes to the plasticity of the cookie dough, imparts desirable eating qualities and contributes to texture and flavor of the product. Proteins can interact with water, from loose interactions to structural water entrapped in proteins structures, which could be unavailable for chemical reactions [
7]. Starting from the premise that differences between varieties could affect the final product structure and composition and thus the shelf life limiting phenomena, the aim of the present paper was to assess the differences in amaretti cookies prepared using different almond varieties. Moreover, surface cracks, such as those found in this type of product, can play a role in chemical physical changes and stability. In this regard, we have developed a workflow for image analysis, based on the use of free software for image pre-processing, and a MATLAB
® (MathWorks, Inc., Natick, MA, USA) code for automation of operations. The procedure allows to have a description of the frequencies of the dimensional classes of the surfaces affected by breakages. We will illustrate the math together with the results, while the MATLAB
® code will be made available on Github website (
https://github.com/AntonioDEM/powerlawlog_project). Instrumental texture, chemical-physical measures and sensory characteristics were assessed to monitor the evolution of shelf life parameters during shelf life. Variables were subjected to PCA to identify patterns in data based on the correlation between features.
2. Materials and Methods
2.1. Almond Origin and Processing
Almonds were collected from the almond germplasm field collection of the Agricultural Research Agency of Sardinia (AGRIS), located in Sardinia (Italy). The soil is sandy-clay (42% sand), pH 7.4, the average annual precipitations (5-year average) were 475.4 mm The plants of the studied varieties, grafted onto GF677 rootstock, are planted at a 6 m × 6 m distance, trained to a goblet shape at 80 cm trunk height. Winter and green pruning are provided annually, and irrigation is supplied using two self-compensating drip rows buried at 30 cm depth. In each dripline emitters (4 l/h) are spaced 40 cm, and about 3.500 m3/ha of irrigation water are provided to the plants each year. Harvest took place from the end of August until the first days of September 2018. The fruits were manually collected from the ground at full ripening. Hulling and shelling operations were conducted mechanically, then the almonds were peeled by dipping the fruits in boiling water until skin removal was achievable by hand. After that, moisture was rebalanced for all varieties to reach values below 6% by air drying in a desiccator (36 °C). Four varieties, of which two (Texas, TX; Tuono, TU) are widely diffused commercial varieties and two (Arrubia, AR; Cossu, CO) are local varieties were evaluated for their attitude to industrial transformation. Peeled almond kernels have been used for both making cookies and chemical analyses.
2.2. Almond Composition
2.2.1. Moisture Content
For each variety, 35 whole peeled almonds (about 50 g) were sampled. Almonds were ground and sifted with stainless steel sieves 18 (1 mm pore size). Crushing lasted less than 30 s, in order to avoid sample heating and agglomeration. Five g of wet almond flour were weighed with an analytical balance (E42S-B, Gibertini, Novate Milanese (MI), Italy) to determine. Then, almond flour was desiccated at 105 °C until constant weight. After cooling the samples in a vacuum dryer for 15 min, dry weight was recorded and moisture content calculated.
2.2.2. Ether Extract
For crude fat content determination, a Buchi B-811 extractor (Buchi, Essen, Germany) was used, performing a Soxhlet type automatic extraction. About 4 g of dried flour were introduced into the extraction thimble, closed at the end with a cotton pad. 120 mL of petroleum ether 40–60 °C RPE (Carlo Erba Reagents, Cornaredo (MI), Italy) were poured in the system and the standard Soxhlet program was set up with 13 extraction cycles for each station and 5 min of rinsing (without drying). At the end of the extraction, the flour was placed in oven at 105 °C for one night, in order to remove the solvent. Then the samples were weighed and the fat content calculated.
2.2.3. Fatty Acid Profile
Fatty acids methyl esters (FAME) were prepared by dissolving 0.5 g of oil in 6 mL of n-hexane, then adding 0.25 mL of KOH 2N in methanol. After 10 s of vigorous shaking, samples were centrifuged at 3000 rpm for 10 min and the supernatant collected. FAME analysis was done with an Agilent 7890A Gas Chromatograph (Agilent, Palo Alto, CA, USA), equipped with a flame ionization detector (FID). Separation was carried out with a, SP-2380 Capillary GC Column (Supelco, Bellefonte, PA, USA; 60 m × 0.25 mm i.d.; 0.2 µm film thickness) using Helium as the carrier gas, at a flow rate of 1.2 mL/min. The GC oven temperature program began with the oven held at 185 °C for 17 min, then increased to 220 °C at 4 °C/min, maintained at 220 °C for 8 min, then to 230 °C at 2 °C/min, for 6 min. The total run time was 45 min. Detector temperature was set at 300 °C, H2 flow at 30 mL/min, air flow at 400 mL/min, make up gas (N2) flow at 25 mL/min. Sample Injection (1 μL) was made in Split mode (50:1) at 60 mL/min. FAME were identified comparing retention times with those of authentic standards (Sigma Aldrich, Saint Louis, MO, USA) and expressed as area units (%) in respect to the total total ion current (TIC) area.
2.2.4. Protein Content
Proteins content was assessed on defatted samples according to the bicinchoninic acid assay (BCA) method using a total protein colorimetric assay Kit (Bio-Rad Protein Assay Kit, Bio-Rad, Hercules, CA, USA).
2.2.5. Amygdalin Content
Amygdalin extraction was performed adding 5 mL of MeOH (Ultra Gradient HPLC Grade JT Baker, Kansas city, NJ, USA) to 0.4 g of ground almonds in 20 mL flasks. The mixture was stirred in water bath at 30 °C for 16 h. Subsequently, the mixture was centrifuged (with a Heraeus Megafuge 40R centrifuge, Thermo Scientific, Waltham, MA, USA) at 4500 rpm for 15 min; the supernatant obtained was then filtered with nylon filters (OlimPeak0.45 µm, 25 mm Ø, Teknokroma, Barcelona, Spain) [
8]. Finally, 1000 µL of sample were taken and transferred to vials for HPLC analysis of TU, CO, and TX varieties almond flour, while for AR (which had supposedly a higher amygdalin content) the sample was diluted in MeOH with a ratio of 1:2 before HPLC analysis. Analytical HPLC grade standard of amygdalin was purchased from Sigma Aldrich and used to construct a 7-point calibration curve. The initial concentration of 33.85 ppm was chosen after preliminary analysis. For each point of the line, three standard dilutions were prepared, each injected in triplicate. An Alliance HPLC instrument (Waters, Milford, MA, USA) equipped with an e2695 XC separation module, PDA 2998 detector and Gemini 3U C18 110A column (150 × 4.60 mm 3 µm, Phenomenex, Torrance, CA, USA) was used. Starting from an original mixture of CH
3CN 20:80 [
8,
9] with a column temperature of 42 °C, the following parameters were set: injection volume: 20 µL, run time: 5 min for standards and 6 min for samples, wavelength (λ): 218 nm, mobile phase: isocratic with unique mixture 21% CH
3CN (acetonitrile RS, Carlo Erba Reagents for HPLC-isocratic grade) and 79% ultrapure H
2O, mobile phase flow: 1.00 mL/min. At sample setting end, the column system was flushed with MeOH, gradually lowering the flow (from 1 to 0 mL/min).
2.3. Amaretti Cookie Quality Characteristics
Two batches of “amaretti” cookies were produced in a local bakery. The following recipe was adopted, expressed as g per 1000 g of dough: 397 g of sucrose, 341 g of sweet almonds, 190 g of egg white (pasteurized), 68 g of bitter almonds, 4 g of citrus aroma. Sweet and bitter almonds together with sucrose (commercially available as granulated sugar and derived from sugar beet, with a particle size under 700 mm) were grinded in a bakery grinding machine. Egg white was then gradually added to the mixture. The ingredients were mixed for 15 min. After formation amaretti were cooked at 160 °C for 40 min in a static oven (Real Forni Srl, Verona, Italy). Baking was followed by cooling at room temperature, then cookies (disc of 35 mm Ø, 30 mm high and weighing 25 gr) were packed in card trays with topping of shrinkable polyolefin film (19 μm). The packaged samples were stored at 23 °C and 65% RH, in the dark, until analyses.
2.3.1. Image Analysis of Crackings Surfaces
Images of “amaretti” cookies were captured using a VersaDoc 4000MP system (Bio-Rad), obtaining images in 24-bit color, at a resolution of 400 dots per inch, i.e., 1 pixel = (60)2 μm
2. For each variety, 30 cookies were analyzed. Image processing was performed using ImageJ 1.51K (Rasband, W.S., ImageJ, National Institutes of Health, Bethesda, MD, USA). The images were pre-treated by conversion into grayscale images, then converted into 8-bit black and white binary images. To optimize the image analysis, Otsu thresholding method was applied. Due to convexity of samples, a planar portion of the surface was selected, capturing a 5 × 5 cm circle from each cookie, taken from the center of the image, as shown in
Figure 1.
The binary images were analyzed for particles number and sizes, using ImageJ particle analysis routine. The output of the analysis was the number of “cracking areas” (CKA) and their plain size (area, in mm2). Data for the areas and their frequency were used to compute the cumulative distribution function (CDF), the complementary cumulative distribution function (CCDF), and probability distribution function (PDF), fitting the entire CCDF distribution to a log-normal PDF, and then using the power-law PDF for the tail of the distribution. All the procedure was carried out using MATLAB® (MathWorks, Inc., Natick, MA, USA). For the convenience of the reader, the math used will be detailed along with the results hereinafter in the article.
2.3.2. Color Measurements
The color was measured on 30 cookies per variety, taking three measurements from each cookie, one from the center and two from the external parts, using a CM-700d spectrophotometer (Konica Minolta, Osaka, Japan), using Standard Illuminant D65/10°. Prior to measurements, the Instrument was calibrated against the white tile. CIE L*a*b* color space coordinates, lightness (L*), color in the red/green field (a*) and color in the blue/yellow field (b*), were computed. The differences in lightness (ΔL′), Chroma (ΔC′), Hue (ΔH) and Hue angle (Δh) were calculated and used to elaborate the ΔE00, the Euclidean distance between colors, as recommended by CIE (2001), using the following formula:
For the detailed explanation of computed parameters, see [
10]
The corresponding ΔE76 values were used to estimate the range of perceived difference between samples of close chroma [
11]):
0 < Δ76 < 1-the difference is unnoticeable
1 < Δ76 < 2-the difference is only noticed by an experienced observer
2 < Δ76 < 3.5-the difference is also noticed by an unexperienced observer
3.5 < Δ76 < 5-the difference is clearly noticeable
5 < Δ76-gives the impression that these are two different color
2.3.3. Water Activity (aw) and Moisture Determination
a
w and moisture were determined in triplicate on six ground amaretti cookies from the same batch, at 1, 7, 15, 30, 60 days of storage. The analyses were performed on whole cookies, due to the difficulty in separating the inner and outer part. Moisture content of amaretti cookies was determined putting the grinded samples (1.5 gr.) in a ventilated oven at 105 °C until constant weight. a
w determinations were performed using an AQUALAB instrument (Series 3, Decagon, Pullman, WA, USA), calibrated in the range 0.1–0.95 with solutions of LiCl, NaCl and KCl of known activity [
12].
2.3.4. Texture Measures
Texture evolution over time was determined with a texture analyzer (TA.XT Plus, Stable Microsystems, Surrey, UK) equipped with a 25 kg load cell and Texture Expert Exceed software, version 2.64a. Analyses were performed at 1, 7, 15, 30, 60 days of storage on six amaretti cookies per batch and time. A puncture test was carried out with a 5 mm diameter cylinder probe (mod. P/5). Samples were placed in the confectionery holder, supplied with a 6mm diameter top and bottom hole (punch test). This holder allows complete penetration of the probe into the sample avoiding sample displacement at the same time. Samples were placed centrally on the holder and secured on the heavy duty platform before the test. The sample was punctured right through. The following test parameters were set: pretest, test and post-test speeds were, 2, 1 and 5 mm/s, respectively. During the test the probe was lowered 20 mm, and after the test it returned to its start position. Three main parameters were computed: Hardness of the upper crust, as the maximum force (N) reached during puncturing; the work of deformation (WOD), as the area under the curve (N mm) between the reaching of the maximum force mm and the complete probe penetration; the thickness of the upper crust, as the distance (mm) between the starting point of the test and the onset of maximum force.
2.3.5. Sensory Analyses
Panel Training
The panel was trained in accordance with the ISO standards [
13,
14]. The panel was made up of nine expert judges, five females and four males, aged between 35 and 50, with previous experience in the sensory profiling of fruits.
Sensory Profile
The sensory descriptive technique was applied to the amaretti cookies [
15]. Amaretti produced with the four almonds varieties (TX, TU, AR and CO) were analyzed at 1, 7, 30 and 60 days of storage. The samples, previously acclimatized in a thermostatically controlled oven (20 °C), placed in containers of odorless material, marked with a three-digit random number [
16] were presented to the judges in a randomized and balanced order [
17], in a tray containing also a cracker and a glass of water, as palate cleansers between samples. Tests were performed in tasting booths [
18]. Judges evaluated the intensity of 18 amaretti’s attributes on a 10 cm unstructured scale, from 0 (low perception) to 10 (high perception), for each attribute identified. Three attributes belong to the visual and tactile characteristics (color, roughness and tactile-hardness), 4 to the olfactory (amaretto, citrus fruits, sweets, odor, and caramel), 2 to the taste (salty and bitter), 4 to the aromatic (amaretto, citrus, bitter and off-flavor almond) and 5 to the texture (hardness, friability, humidity, adhesiveness and solubility). Two experimental replicates were performed for each control point. To measure the analytical reliability of the panel’s response, two samples of amaretti cookies were replicated for each shelf-life sampling points. The acquisition of sensory data was carried out using a specific computerized application [
19].
Consumer Testing
An acceptability test was performed [
15]. The acceptability test was carried out by 60 consumers, 30 women and 30 men, most of them recruited on the basis of interest and willingness. They were regular consumers of the product, aged between 32 and 60 years, not trained in the sensory analysis. Consumers were asked to give a score to the following attributes: flavor, taste, texture, appearance and overall acceptability. A nine-point structured hedonic scale ranging from 1 (extremely disliked it) to 9 (extremely liked it) was used, and sample was considered acceptable when scoring above 5 (neither like nor dislike).
2.4. Statistical Analyses
Chemical, physical chemical, texture, and sensory data were subjected to analysis of variance (ANOVA) and Tukey test (n = 5, p ≤ 0.05) as the post hoc test, using the Statgraphics Centurion software package (version 16.1.11, StatPoint Technologies Inc., Warrenton, VA, USA). Image analysis data were processed using MATLAB® routines, using Kolmogorov-Smirnov statistics to evaluate the goodness of fit of CKA on log-normal and power-law distributions. The panel’s judgments, in terms of reproducibility and discriminant ability, was monitored through Three-way ANOVA model (judge, sample and replicates effect) with interaction. The hedonic scores collected from the consumer test were examined using ANOVA and Tukey tests (p ≤ 0.05), with consumers (random effect) and products as the main effects. The differences between samples were analyzed by principal component analysis (PCA) of the correlation matrix of selected variables. The component loadings were calculated as simple correlations (using Pearson’s r) between the components (i.e., the component scores) and the original variables.
4. Discussion
The present work was aimed to address the question if almond varieties have a significant effect on the quality features of derived products. Almonds are rich in proteins and lipids, which can affect the texture, flavor and physical chemical characteristics of the final product. The compositional analyses showed no differences in the protein content between varieties, except for TX, that showed the lower value. More marked differences were found in the fat fraction, with TX and CO samples having significantly higher crude fat content and MUFA in the fatty acids profile, compared to the other varieties. Cookies, being intermediate moisture food rich in sucrose, undergo to physical chemical changes during the shelf life. Water loss from the product to the headspace is due to moisture gradients, boosted by increase in a
w related to sucrose crystallization. Such changes affect the texture and finally the shelf life and acceptance. Amaretti cookies are characterized by presenting surface breaks, which can potentially represent ways out of the water. This aspect have been investigated by using a specifically developed tool for particles size distribution analyses. The best-fitting procedure was carried out, following the methodology reported in [
28], originally conceived for the analysis of gas-cell size distribution in wheat dough. Since there are no differences in the shape parameter, σ*, the distribution of the CKA follows the same shape among samples. On the other hand, μ*, the median of the log-normal distribution, and the geometric mean of the untransformed data, is significantly higher in CO samples. Since no differences were found in the tail of the distributions, the major differences between samples are in the small to medium size CKA. TU and AR samples presented the same μ* value, while TX presents the lower values. CKA could act as preferential ways for moisture exchanges in amaretti cookies, so that differences in the parameter μ* of the CKA, and the total area affected by cracking, could give an account of the differences between samples, regarding water loss and water migration between components, during the shelf life. Color was also affected by the variety used. Color develops during cooking, due to Maillard reactions and caramelization. Maillard reactions takes place in several steps, involving reducing sugars and amino acids, proteins, and/or other nitrogen-containing compounds, when they are heated together, while caramelization refers to complex group of reactions that occur due to direct heating of carbohydrates at higher temperatures, in particular sucrose and reducing sugars [
29]. In cookies, color development is strictly related to the browning reaction, which occurs especially on the surface, were a
w levels decrease 0.4–0.7 during cooking and temperature surpasses 105–120 °C. Our results showed that the almond variety has an impact on color development, with the major differences found between AR and CO varieties. The differences observed between cookies coming from different varieties can be related to differences in amino acids composition, proteins and sugar content and type [
30], and the presence of pigments. The importance of browning development during baking is not only related to sensorial aspects such as color formation but also on flavor generation [
31,
32], both affecting quality and acceptance. Regarding the texture features, the major differences were found in the intermediate time of analyses. One variety in particular performed worst. From 15 days and to 30 days CO cookies had significantly higher hardness values. At 15 days CO samples showed significantly higher values of WOD, in respect to other samples. At the same time of analysis, the same variety showed the lower moisture level. Descriptive analysis showed differences in few descriptors, particularly tactile, in fresh samples and at 30 days of storage. The sensory characteristics “tactile-hardness”, “odor of amaretto” and “hardness” measured in the mouth, significantly changed. Hardness, both tactile and during chewing, reached the higher intensity in TX. At 60 days, the sample with the highest value of both hardness and friability was the one produced with the CO variety. Amygdalin has a great sensory impact when raw almonds are eaten, while its effect has been attenuated in the processed product, since other ingredients and the use of bitter almonds in the recipe surely had a flattening effect on its olfactory impact.
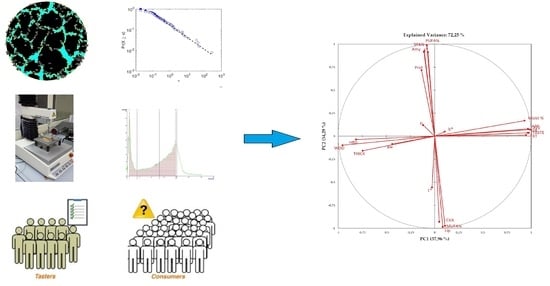
In an attempt to unscramble the complex interactions between food components, textural features, moisture and a
w changes, sensory characteristics and acceptance, a dimensionality reduction approach was used, performing a PCA over data. The first two principal components (PC1 and PC2) explained 72.25% of the variability of the data (
Figure 7 and
Figure 8).
Variables are distributed in the PCA plan based on their relative contribution to the principal components. Variables taken into account were: water activity (aw), moisture content (Moist%), total cracked areas (mm2) (CKA), the median of the log-normal distribution of cracking surfaces (μ), CIEL*a*b* color coordinates (L*, a* and b*), instrumental hardness (HRD), crust thickness (THICK), and work of deformation (WOD), sensory scores for appearance (APP), taste (TASTE), flavor (FLV), texture, and overall acceptability (OA), amygdalin content (Amy), crude fat content (Lip), Protein content (Prot), percentage of SFA (SFA%), MUFA % (MUFA %), PUFA (PUFA%). Tactile sensory parameters correlated well with instrumental texture, so we omitted these parameters in the PCA matrix. The PC1 is correlated mostly and positively with Moist % (0.93), APP (0.97), TASTE (0.9748), FLV (0.9862), TXT (0.9463), OA (0.9637), while negatively correlated with HRD (−0.8141), THICK (−0.7456), WOD (−0.9546). Samples with higher sensory scores and “softer” texture plot in the positive part of PC1. On the contrary, samples with lower sensory scores and “harder” texture plot in the negative part of PC1. PC1 (37.96%) can be interpreted in terms of “acceptability as related to instrumental texture”. PC2 is correlated positively mostly with a* (0.9136), Amy (0.9167), SFA% (91.98) and PUFA% (986) and negatively with CKA (−0.9299), Lip (−0.9738) and MUFA% (98.39). Samples with higher a*, Amy, SFA% and PUFA%, tend to plot in the positive part of PC2, while samples with higher CKA, Lip and MUFA% should plot in the negative part. This second PC could be interpreted in terms of “redness, cracking surfaces and composition”.
“High quality”, intended to maintain high sensorial standards during the shelf life, lasts up to 30 days. TX and TU samples showed close position along the PC1, for all the time of analyses. AR clustered in the positive part of PC1 until day 7, showing sensory acceptance similar to TX and TU. For most of the shelf life, CO samples were characterized by lower sensory scores, higher instrumental hardness, thicker crust and more viscous texture (higher WOD), showing larger Cracked Area, and lower moisture. Samples formed clearly distinguishable clusters along the PC2. Overall, our data showed a marked influence of variety in the physical chemical characteristics of derived products. The correlation matrix showed that Protein content was correlated positively (0.7318) with the parameter
µ, the median of the cracking surfaces distribution, which is correlated with a third PC (0.9809), although it’s interpretation in relation to physical chemical evolution and sensory acceptance results problematic. Crude fat content was positively correlated with the total area interested by cracking CKA (0.8636). The sensory acceptance, as clearly showed in the loading plot (
Figure 7) is inversely correlated with the magnitude of texture features, namely HRD, WOD, THICK, and a
w, and positively correlated with Moist%. Sensory acceptances were higher at 7 days for all the samples, and similar between samples until day 30, were CO samples showed the lower values. Sixty days was selected as the end of the shelf life for all samples.