The Impact of Germination on Sorghum Nutraceutical Properties
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals and Reagents
2.2. Plant Material and Germination Process
2.3. Plant Material Extraction
2.4. Total Phenolic Content Determination
2.5. Determination of Antioxidant Activity
2.6. Peptide Extraction
2.7. Angiotensin-Converting Enzyme (ACE) Inhibitory Activity
2.8. Statistical Analysis
3. Results
3.1. Total Phenolic Content and In Vitro Antioxidant Activities
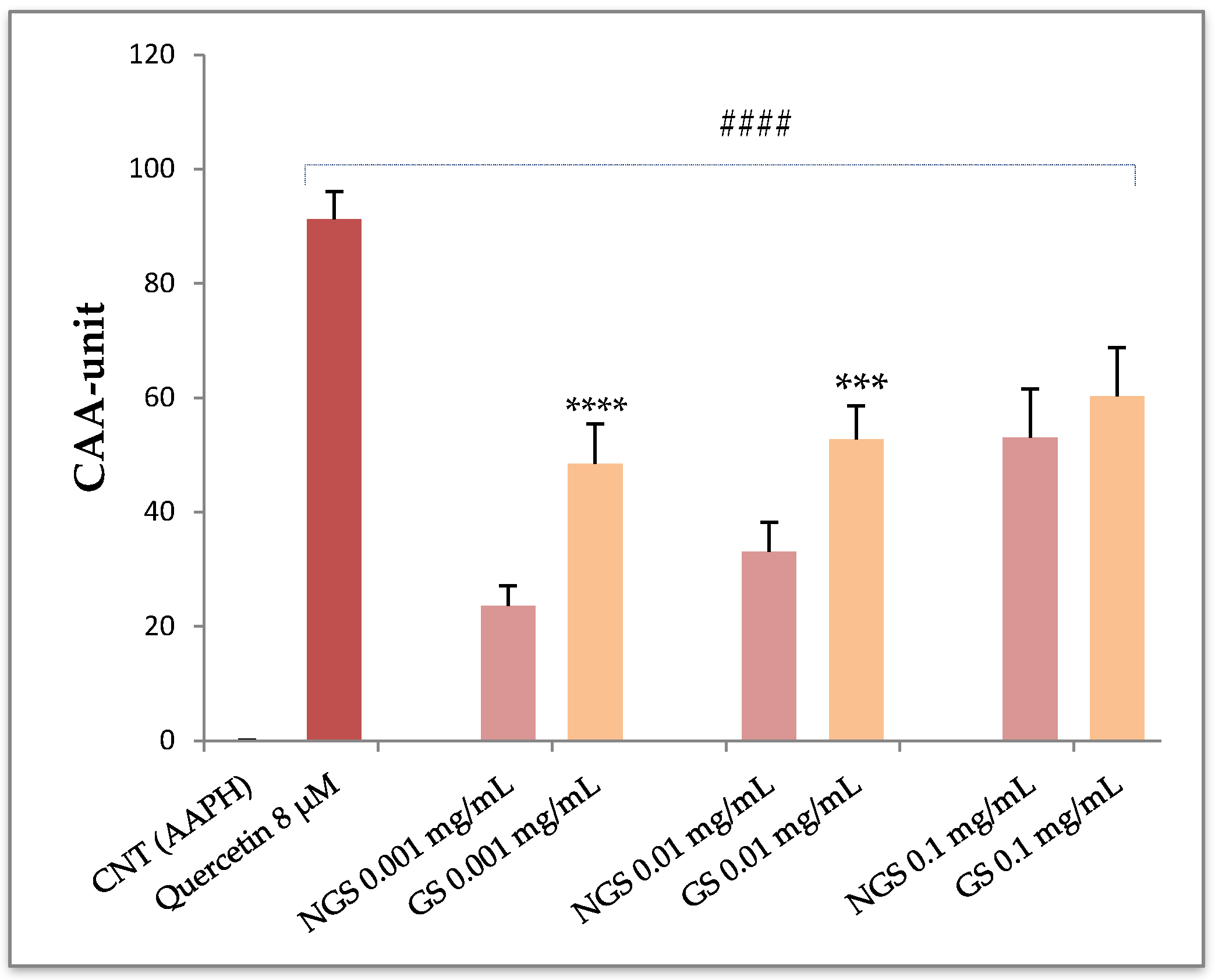
3.2. Cellular Antioxidant Activity in Red Blood Cells (CAA-RBC)
3.3. ACE Inhibitory Activity of Sorghum Peptides
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| AAPH | 2,2-azobis(2-amidinopropane) dihydrochloride |
| ACE | angiotensin-converting enzyme |
| CAA-RBC | cellular antioxidant activity in red blood cells |
| DCFH-DA | 2′,7′-dichlorofluorescein diacetate |
| DPPH | 2,2-diphenyl-1-picrylhydrazyl |
| FRAP | ferric reducing antioxidant power |
| GS | germinated sorghum |
| NGS | non-germinated sorghum |
| TPC | total phenolic content |
References
- FAOSTAT. Available online: http://www.fao.org/faostat/en/#compare (accessed on 28 June 2020).
- Ratnavathi, C.V.; Patil, J.V.; Chavan, U.D. Sorghum Biochemistry: An Industrial Perspective; Academic Press: Cambridge, MA, USA, 2016. [Google Scholar]
- Taleon, V.; Dykes, L.; Rooney, W.L.; Rooney, L.W. Effect of Genotype and Environment on Flavonoid Concentration and Profile of Black Sorghum Grains. J. Cereal Sci. 2012, 56, 470–475. [Google Scholar] [CrossRef]
- Lemlioglu-Austin, D. Sorghum: Obliging Alternative and Ancient Grain. Cereal Foods World 2014, 59, 12–20. [Google Scholar] [CrossRef]
- De Morais Cardoso, L.; Pinheiro, S.S.; Martino, H.S.D.; Pinheiro-Sant′Ana, H.M. Sorghum (Sorghum bicolor, L.): Nutrients, Bioactive Compounds, and Potential Impact on Human Health. Crit. Rev. Food Sci. Nutr. 2017, 57, 372–390. [Google Scholar] [CrossRef] [PubMed]
- Girard, A.L.; Awika, J.M. Sorghum Polyphenols and Other Bioactive Components as Functional and Health Promoting Food Ingredients. J. Cereal Sci. 2018, 84, 112–124. [Google Scholar] [CrossRef]
- Ochanda, S.O.; Akoth, O.C.; Mwasaru, M.A.; Kagwiria, O.J.; Mutiso, M.F. Effects of Malting and Fermentation Treatments on Group B-Vitamins of Red Sorghum, White Sorghum and Pearl Millets in Kenya. J. Appl. Biosci. 2010, 34, 2128–2134. [Google Scholar]
- Martino, H.S.D.; Tomaz, P.A.; Moraes, E.A.; Conceiçao, L.L.; Oliveira, D.S.; Queiroz, V.A.V.; Rodrigues, J.A.S.; Pirozi, M.R.; Pinheiro-Sant’Ana, H.M.; Ribeiro, M.R. Chemical characterization and size distribution of sorghum genotypes for human consumption. Rev. Inst. Adolfo Lutz 2012, 71, 337–344. [Google Scholar]
- Stefoska-Needham, A.; Beck, E.J.; Johnson, S.K.; Tapsell, L.C. Sorghum: An Underutilized Cereal Whole Grain with the Potential to Assist in the Prevention of Chronic Disease. Food Rev. Int. 2015, 31, 401–437. [Google Scholar] [CrossRef]
- Simnadis, T.G.; Tapsell, L.C.; Beck, E.J. Effect of Sorghum Consumption on Health Outcomes: A Systematic Review. Nutr. Rev. 2016, 74, 690–707. [Google Scholar] [CrossRef]
- Etuk, E.B.; Okeudo, N.J.; Esonu, B.O.; Udedibie, A.B.I. Antinutritional Factors in Sorghum: Chemistry, Mode of Action and Effects on Livestock and Poultry. Online J. Anim. Feed Res. 2012, 2, 113–119. [Google Scholar]
- Mohammed, N.A.; Ahmed, I.A.M.; Babiker, E.E. Nutritional Evaluation of Sorghum Flour (Sorghum bicolor, L. Moench) during Processing of Injera. World Acad. Sci. Eng. Technol. 2011, 51, 72–76. [Google Scholar]
- Osman, M.A. Effect of Different Processing Methods, on Nutrient Composition, Antinutrional Factors, and in Vitro Protein Digestibility of Dolichos Lablab Bean [Lablab purpuresus (L) Sweet]. Pak. J. Nutr. 2007, 6, 299–303. [Google Scholar] [CrossRef]
- Ogbonna, A.C.; Abuajah, C.I.; Ide, E.O.; Udofia, U.S. Effect of Malting Conditions on the Nutritional and Anti-Nutritional Factors of Sorghum Grist. Ann. Univ. Dunarea Jos Galati Fascicle VI Food Technol. 2012, 36, 64–72. [Google Scholar]
- Ojha, P.; Adhikari, R.; Karki, R.; Mishra, A.; Subedi, U.; Karki, T.B. Malting and Fermentation Effects on Antinutritional Components and Functional Characteristics of Sorghum Flour. Food Sci. Nutr. 2018, 6, 47–53. [Google Scholar] [CrossRef] [PubMed]
- Donkor, O.N.; Stojanovska, L.; Ginn, P.; Ashton, J.; Vasiljevic, T. Germinated Grains–Sources of Bioactive Compounds. Food Chem. 2012, 135, 950–959. [Google Scholar] [CrossRef] [PubMed]
- Mamilla, R.K.; Mishra, V.K. Effect of Germination on Antioxidant and ACE Inhibitory Activities of Legumes. LWT 2017, 75, 51–58. [Google Scholar] [CrossRef]
- Nwanguma, B.C.; Eze, M.O. Changes in the Concentrations of the Polyphenolic Constituents of Sorghum during Malting and Mashing. J. Sci. Food Agric. 1996, 70, 162–166. [Google Scholar] [CrossRef]
- Garzón, A.G.; Drago, S.R. Free α-Amino Acids, γ-Aminobutyric Acid (GABA), Phenolic Compounds and Their Relationships with Antioxidant Properties of Sorghum Malted in Different Conditions. J. Food Sci. Technol. 2018, 55, 3188–3198. [Google Scholar] [CrossRef]
- Singh, A.; Sharma, S.; Singh, B.; Kaur, G. In Vitro Nutrient Digestibility and Antioxidative Properties of Flour Prepared from Sorghum Germinated at Different Conditions. J. Food Sci. Technol. 2019, 56, 3077–3089. [Google Scholar] [CrossRef]
- Osuntogun′, B.A.; Adewusi, S.R.A.; Nwasike, C.C. Effect of Cultivar, Steeping, and Malting on Tannin, Total Polyphenol, and Cyanide Content of Nigerian Sorghum. Cereal Chem. 1989, 66, 87–89. [Google Scholar]
- Kayodé, A.P.P.; Hounhouigan, J.D.; Nout, M.J.R. Impact of Brewing Process Operations on Phytate, Phenolic Compounds and in Vitro Solubility of Iron and Zinc in Opaque Sorghum Beer. LWT Food Sci. Technol. 2007, 40, 834–841. [Google Scholar] [CrossRef]
- Khoddami, A.; Mohammadrezaei, M.; Roberts, T. Effects of Sorghum Malting on Colour, Major Classes of Phenolics and Individual Anthocyanins. Molecules 2017, 22, 1713. [Google Scholar] [CrossRef]
- Dicko, M.H.; Gruppen, H.; Traoré, A.S.; van Berkel, W.J.; Voragen, A.G. Evaluation of the Effect of Germination on Phenolic Compounds and Antioxidant Activities in Sorghum Varieties. J. Agric. Food Chem. 2005, 53, 2581–2588. [Google Scholar] [CrossRef]
- Gabriele, M.; Pucci, L. Diet Bioactive Compounds: Implications for Oxidative Stress and Inflammation in the Vascular System. Endocr. Metab. Immune Disord. Drug Targets. 2017, 17, 264–275. [Google Scholar] [CrossRef]
- Bamdad, F.; Dokhani, S.; Keramat, J.; Zareie, R. The Impact of Germination and in Vitro Digestion on the Formation of Angiotensin Converting Enzyme (ACE) Inhibitory Peptides from Lentil Proteins Compared to Whey Proteins. Heart Fail. 2009, 3, 4. [Google Scholar]
- López-Amorós, M.L.; Hernández, T.; Estrella, I. Effect of Germination on Legume Phenolic Compounds and Their Antioxidant Activity. J. Food Compos. Anal. 2006, 19, 277–283. [Google Scholar] [CrossRef]
- Koehler, P.; Hartmann, G.; Wieser, H.; Rychlik, M. Changes of Folates, Dietary Fiber, and Proteins in Wheat as Affected by Germination. J. Agric. Food Chem. 2007, 55, 4678–4683. [Google Scholar] [CrossRef]
- Gabriele, M.; Parri, E.; Felicioli, A.; Sagona, S.; Pozzo, L.; Biondi, C.; Domenici, V.; Pucci, L. Phytochemical Composition and Antioxidant Activity of Tuscan Bee Pollen of Different Botanic Origins. Ital. J. Food Sci. 2015, 27, 248–259. [Google Scholar]
- Colosimo, R.; Gabriele, M.; Cifelli, M.; Longo, V.; Domenici, V.; Pucci, L. The Effect of Sourdough Fermentation on Triticum Dicoccum from Garfagnana: 1H NMR Characterization and Analysis of the Antioxidant Activity. Food Chem. 2020, 305, 125510. [Google Scholar] [CrossRef]
- Gabriele, M.; Frassinetti, S.; Caltavuturo, L.; Montero, L.; Dinelli, G.; Longo, V.; Di Gioia, D.; Pucci, L. Citrus Bergamia Powder: Antioxidant, Antimicrobial and Anti-Inflammatory Properties. J. Funct. Foods 2017, 31, 255–265. [Google Scholar] [CrossRef]
- Frassinetti, S.; Gabriele, M.; Caltavuturo, L.; Longo, V.; Pucci, L. Antimutagenic and Antioxidant Activity of a Selected Lectin-Free Common Bean (Phaseolus Vulgaris L.) in Two Cell-Based Models. Plant Foods Hum. Nutr. 2015, 70, 35–41. [Google Scholar] [CrossRef]
- Wolfe, K.L.; Liu, R.H. Cellular Antioxidant Activity (CAA) Assay for Assessing Antioxidants, Foods, and Dietary Supplements. J. Agric. Food Chem. 2007, 55, 8896–8907. [Google Scholar] [CrossRef] [PubMed]
- Vilcacundo, R.; Martínez-Villaluenga, C.; Miralles, B.; Hernández-Ledesma, B. Release of Multifunctional Peptides from Kiwicha (Amaranthus caudatus) Protein under in Vitro Gastrointestinal Digestion. J. Sci. Food Agric. 2019, 99, 1225–1232. [Google Scholar] [CrossRef] [PubMed]
- Lowry, O.H.; Rosebrough, N.J.; Farr, A.L.; Randall, R.J. Protein Measurement with the Folin Phenol Reagent. J. Biol. Chem. 1951, 193, 265–275. [Google Scholar] [PubMed]
- Siow, H.-L.; Gan, C.-Y. Extraction of Antioxidative and Antihypertensive Bioactive Peptides from Parkia Speciosa Seeds. Food Chem. 2013, 141, 3435–3442. [Google Scholar] [CrossRef]
- Gabriele, M.; Pucci, L.; Árvay, J.; Longo, V. Anti-Inflammatory and Antioxidant Effect of Fermented Whole Wheat on TNFα-Stimulated HT-29 and NF-ΚB Signaling Pathway Activation. J. Funct. Foods 2018, 45, 392–400. [Google Scholar] [CrossRef]
- Ljevar, A.; Ćurko, N.; Tomašević, M.; Radošević, K.; Gaurina Srček, V.; Kovačević Ganić, K. Phenolic Composition, Antioxidant Capacity and in Vitro Cytotoxicity Assessment of Fruit Wines. Food Technol. Biotechnol. 2016, 54, 145–155. [Google Scholar] [CrossRef]
- Caddeo, C.; Pucci, L.; Gabriele, M.; Carbone, C.; Fernàndez-Busquets, X.; Valenti, D.; Pons, R.; Vassallo, A.; Fadda, A.M.; Manconi, M. Stability, Biocompatibility and Antioxidant Activity of PEG-Modified Liposomes Containing Resveratrol. Int. J. Pharm. 2018, 538, 40–47. [Google Scholar] [CrossRef]
- Blasa, M.; Angelino, D.; Gennari, L.; Ninfali, P. The Cellular Antioxidant Activity in Red Blood Cells (CAA-RBC): A New Approach to Bioavailability and Synergy of Phytochemicals and Botanical Extracts. Food Chem. 2011, 125, 685–691. [Google Scholar] [CrossRef]
- Xu, M.; Rao, J.; Chen, B. Phenolic Compounds in Germinated Cereal and Pulse Seeds: Classification, Transformation, and Metabolic Process. Crit. Rev. Food Sci. Nutr. 2020, 60, 740–759. [Google Scholar] [CrossRef]
- Angelino, D.; Gennari, L.; Blasa, M.; Selvaggini, R.; Urbani, S.; Esposto, S.; Servili, M.; Ninfali, P. Chemical and Cellular Antioxidant Activity of Phytochemicals Purified from Olive Mill Waste Waters. J. Agric. Food Chem. 2011, 59, 2011–2018. [Google Scholar] [CrossRef]
- Awika, J.M.; Rose, D.J.; Simsek, S. Complementary Effects of Cereal and Pulse Polyphenols and Dietary Fiber on Chronic Inflammation and Gut Health. Food Funct. 2018, 9, 1389–1409. [Google Scholar] [CrossRef] [PubMed]
- Ragaee, S.; Abdel-Aal, E.-S.M.; Noaman, M. Antioxidant Activity and Nutrient Composition of Selected Cereals for Food Use. Food Chem. 2006, 98, 32–38. [Google Scholar] [CrossRef]
- Awika, J.M.; Rooney, L.W. Sorghum Phytochemicals and Their Potential Impact on Human Health. Phytochemistry 2004, 65, 1199–1221. [Google Scholar] [CrossRef] [PubMed]
- Burdette, A.; Garner, P.L.; Mayer, E.P.; Hargrove, J.L.; Hartle, D.K.; Greenspan, P. Anti-Inflammatory Activity of Select Sorghum (Sorghum bicolor) Brans. J. Med. Food 2010, 13, 879–887. [Google Scholar] [CrossRef] [PubMed]
- Moraes, É.A.; Natal, D.I.G.; Queiroz, V.A.V.; Schaffert, R.E.; Cecon, P.R.; de Paula, S.O.; dos Anjos Benjamim, L.; Ribeiro, S.M.R.; Martino, H.S.D. Sorghum Genotype May Reduce Low-Grade Inflammatory Response and Oxidative Stress and Maintains Jejunum Morphology of Rats Fed a Hyperlipidic Diet. Food Res. Int. 2012, 49, 553–559. [Google Scholar] [CrossRef]
- Kamath, V.G.; Chandrashekar, A.; Rajini, P.S. Antiradical Properties of Sorghum (Sorghum bicolor L. Moench) Flour Extracts. J. Cereal Sci. 2004, 40, 283–288. [Google Scholar] [CrossRef]
- Hagerman, A.E.; Riedl, K.M.; Jones, G.A.; Sovik, K.N.; Ritchard, N.T.; Hartzfeld, P.W.; Riechel, T.L. High Molecular Weight Plant Polyphenolics (Tannins) as Biological Antioxidants. J. Agric. Food Chem. 1998, 46, 1887–1892. [Google Scholar] [CrossRef]
- Awika, J.M. Behavior of 3-Deoxyanthocyanidins in the Presence of Phenolic Copigments. Food Res. Int. 2008, 41, 532–538. [Google Scholar] [CrossRef]
- Awika, J.M.; Rooney, L.W.; Waniska, R.D. Properties of 3-Deoxyanthocyanins from Sorghum. J. Agric. Food Chem. 2004, 52, 4388–4394. [Google Scholar] [CrossRef]
- Kamath, V.; Niketh, S.; Chandrashekar, A.; Rajini, P.S. Chymotryptic Hydrolysates of α-Kafirin, the Storage Protein of Sorghum (Sorghum Bicolor) Exhibited Angiotensin Converting Enzyme Inhibitory Activity. Food Chem. 2007, 100, 306–311. [Google Scholar] [CrossRef]
- Szewińska, J.; Simińska, J.; Bielawski, W. The Roles of Cysteine Proteases and Phytocystatins in Development and Germination of Cereal Seeds. J. Plant Physiol. 2016, 207, 10–21. [Google Scholar] [CrossRef]
- Martinez, M.; Gómez-Cabellos, S.; Giménez, M.J.; Barro, F.; Diaz, I.; Diaz-Mendoza, M. Plant Proteases: From Key Enzymes in Germination to Allies for Fighting Human Gluten-Related Disorders. Front. Plant Sci. 2019, 10, 721. [Google Scholar] [CrossRef]
- Fritz, M.; Vecchi, B.; Rinaldi, G.; Añón, M.C. Amaranth Seed Protein Hydrolysates Have in Vivo and in Vitro Antihypertensive Activity. Food Chem. 2011, 126, 878–884. [Google Scholar] [CrossRef]
- Domínguez, F.; González, M.; Cejudo, F.J. A Germination-Related Gene Encoding a Serine Carboxypeptidase Is Expressed during the Differentiation of the Vascular Tissue in Wheat Grains and Seedlings. Planta 2002, 215, 727–734. [Google Scholar] [CrossRef]
- Aluko, R.E. Structure and Function of Plant Protein-Derived Antihypertensive Peptides. Curr. Opin. Food Sci. 2015, 4, 44–50. [Google Scholar] [CrossRef]
- Isaacson, C. The Change of the Staple Diet of Black South Africans from Sorghum to Maize (Corn) Is the Cause of the Epidemic of Squamous Carcinoma of the Oesophagus. Med. Hypotheses 2005, 64, 658–660. [Google Scholar] [CrossRef]
- Cisse, F.; Erickson, D.P.; Hayes, A.M.; Opekun, A.R.; Nichols, B.L.; Hamaker, B.R. Traditional Malian Solid Foods Made from Sorghum and Millet Have Markedly Slower Gastric Emptying than Rice, Potato, or Pasta. Nutrients 2018, 10, 124. [Google Scholar] [CrossRef]
- Filippo, C.D.; Cavalieri, D.; Paola, M.D.; Ramazzotti, M.; Poullet, J.B.; Massart, S.; Collini, S.; Pieraccini, G.; Lionetti, P. Impact of Diet in Shaping Gut Microbiota Revealed by a Comparative Study in Children from Europe and Rural Africa. Proc. Natl. Acad. Sci. USA 2010, 107, 14691–14696. [Google Scholar] [CrossRef]
| Total Phenolic Content (mg GAE/g DW) | DPPH EC50 (mg/mL) | FRAP (µM Fe2+) | |
|---|---|---|---|
| Germinated | 6.38 ± 0.37 | 0.51 ± 0.01 | 2666 ± 23 |
| Non-germinated | 6.62 ± 0.28 | 0.29 ± 0.05 *** | 3636.2 ± 124.68 **** |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Arouna, N.; Gabriele, M.; Pucci, L. The Impact of Germination on Sorghum Nutraceutical Properties. Foods 2020, 9, 1218. https://doi.org/10.3390/foods9091218
Arouna N, Gabriele M, Pucci L. The Impact of Germination on Sorghum Nutraceutical Properties. Foods. 2020; 9(9):1218. https://doi.org/10.3390/foods9091218
Chicago/Turabian StyleArouna, Nafiou, Morena Gabriele, and Laura Pucci. 2020. "The Impact of Germination on Sorghum Nutraceutical Properties" Foods 9, no. 9: 1218. https://doi.org/10.3390/foods9091218
APA StyleArouna, N., Gabriele, M., & Pucci, L. (2020). The Impact of Germination on Sorghum Nutraceutical Properties. Foods, 9(9), 1218. https://doi.org/10.3390/foods9091218







