Influence of Meat Spoilage Microbiota Initial Load on the Growth and Survival of Three Pathogens on a Naturally Fermented Sausage
Abstract
1. Introduction
2. Materials and Methods
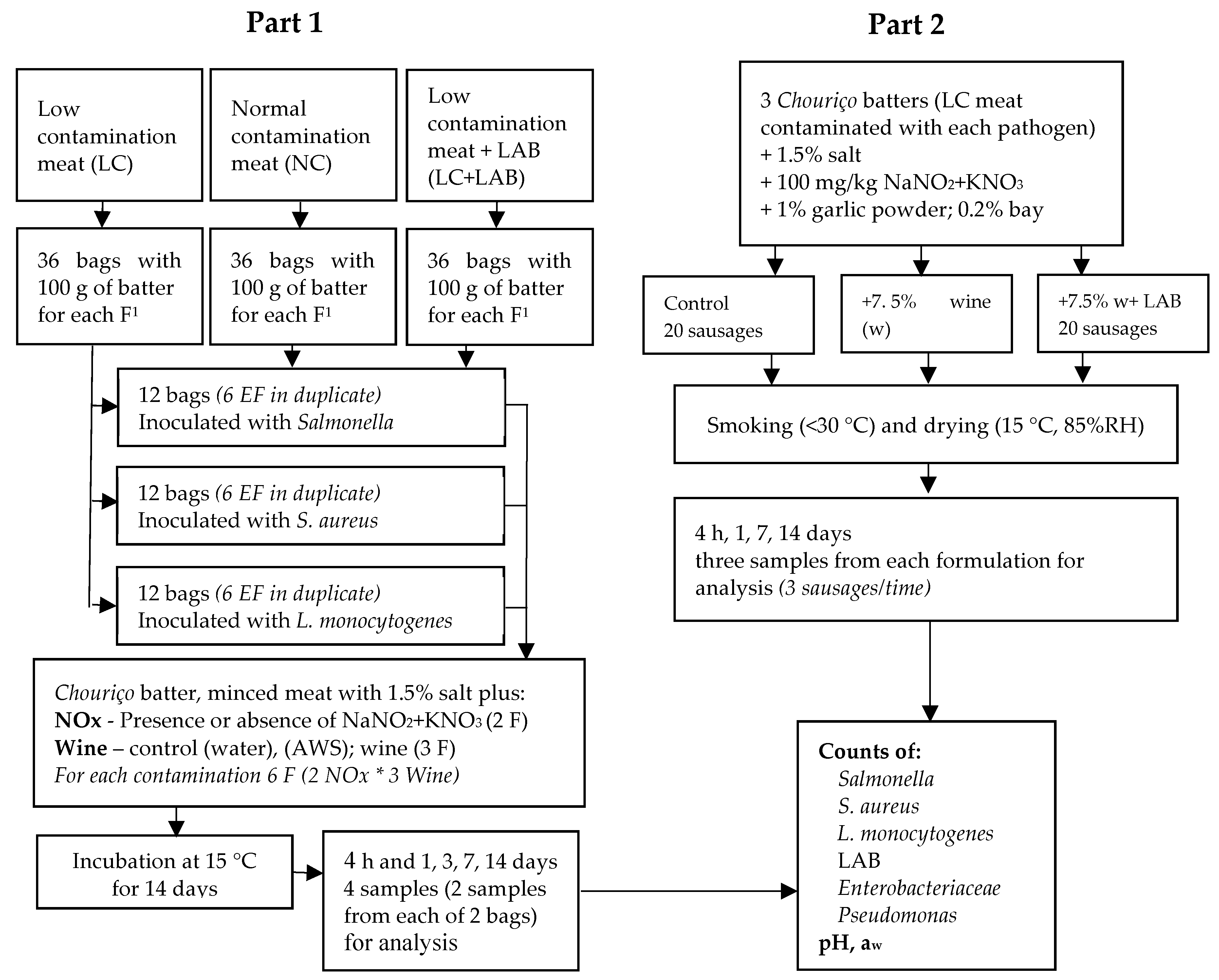
2.1. Experimental Design
2.2. Bacterial Strains and Preparation of Inoculum
2.3. Part 1. Experiments with a Chouriço Batter
2.4. Part 2. Experiment with a Naturally Fermented Sausage—Chouriço
2.5. Bacterial Enumeration
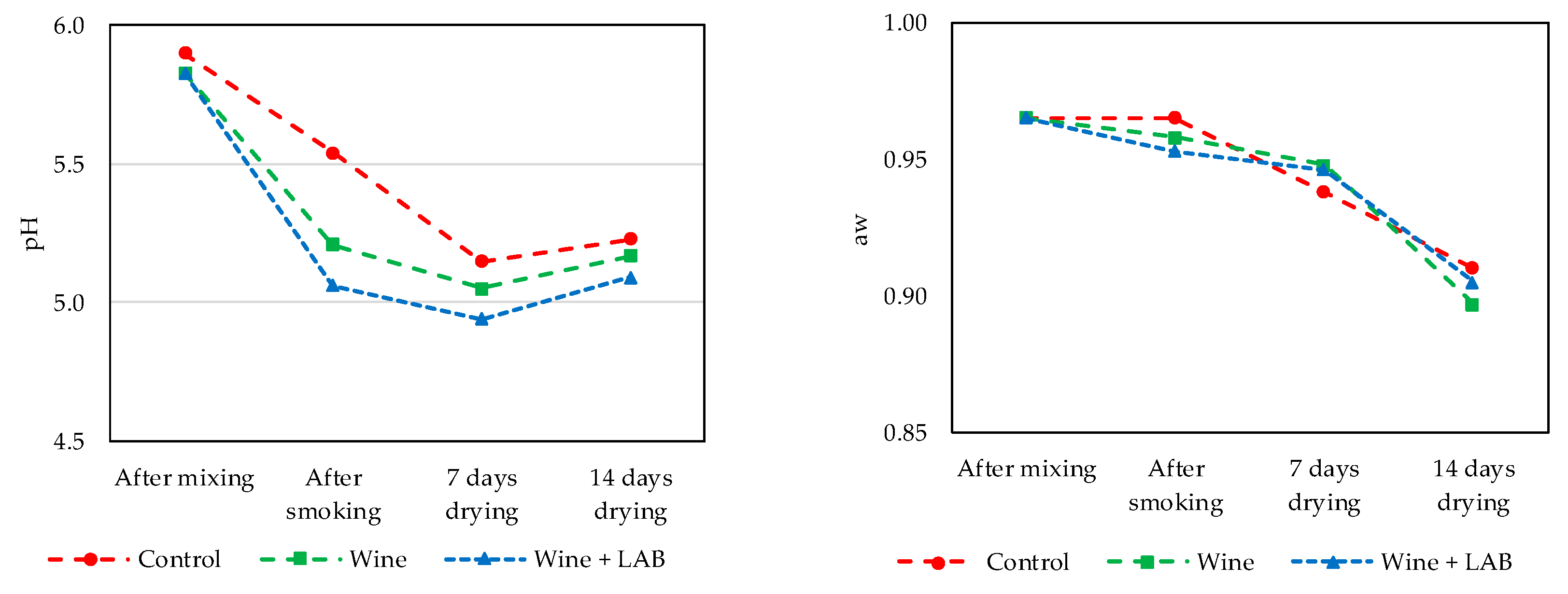
2.6. pH and Activity of Water
2.7. Data Analysis
3. Results
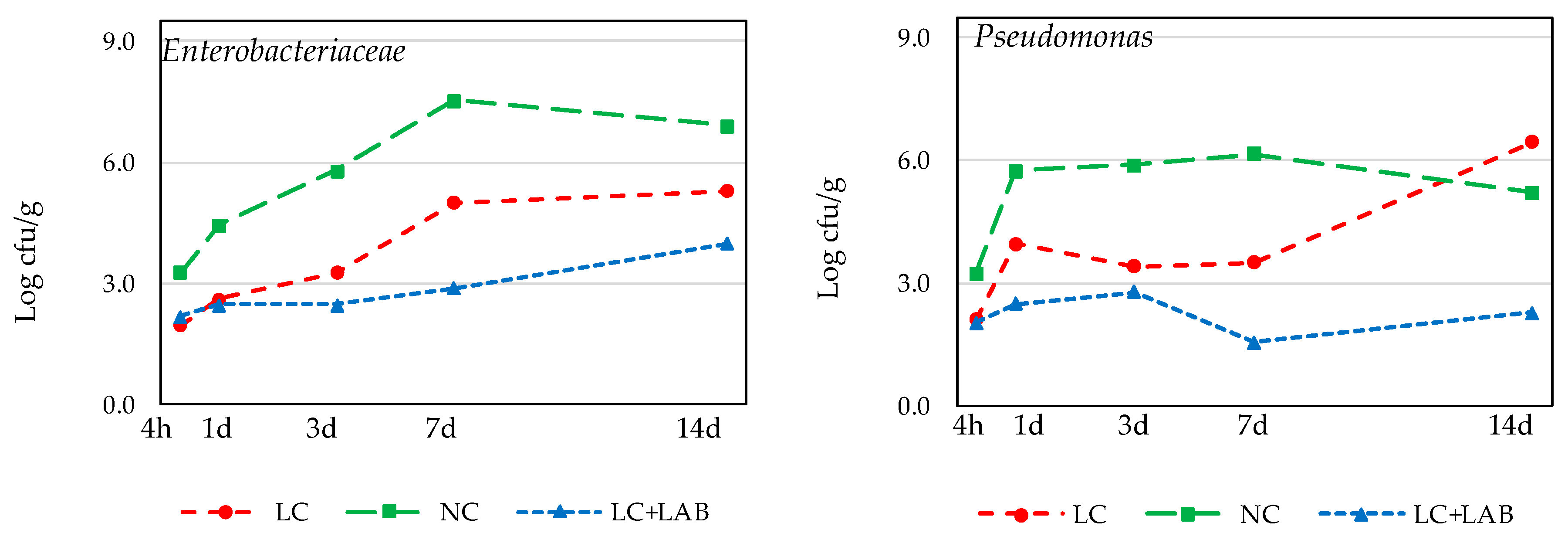
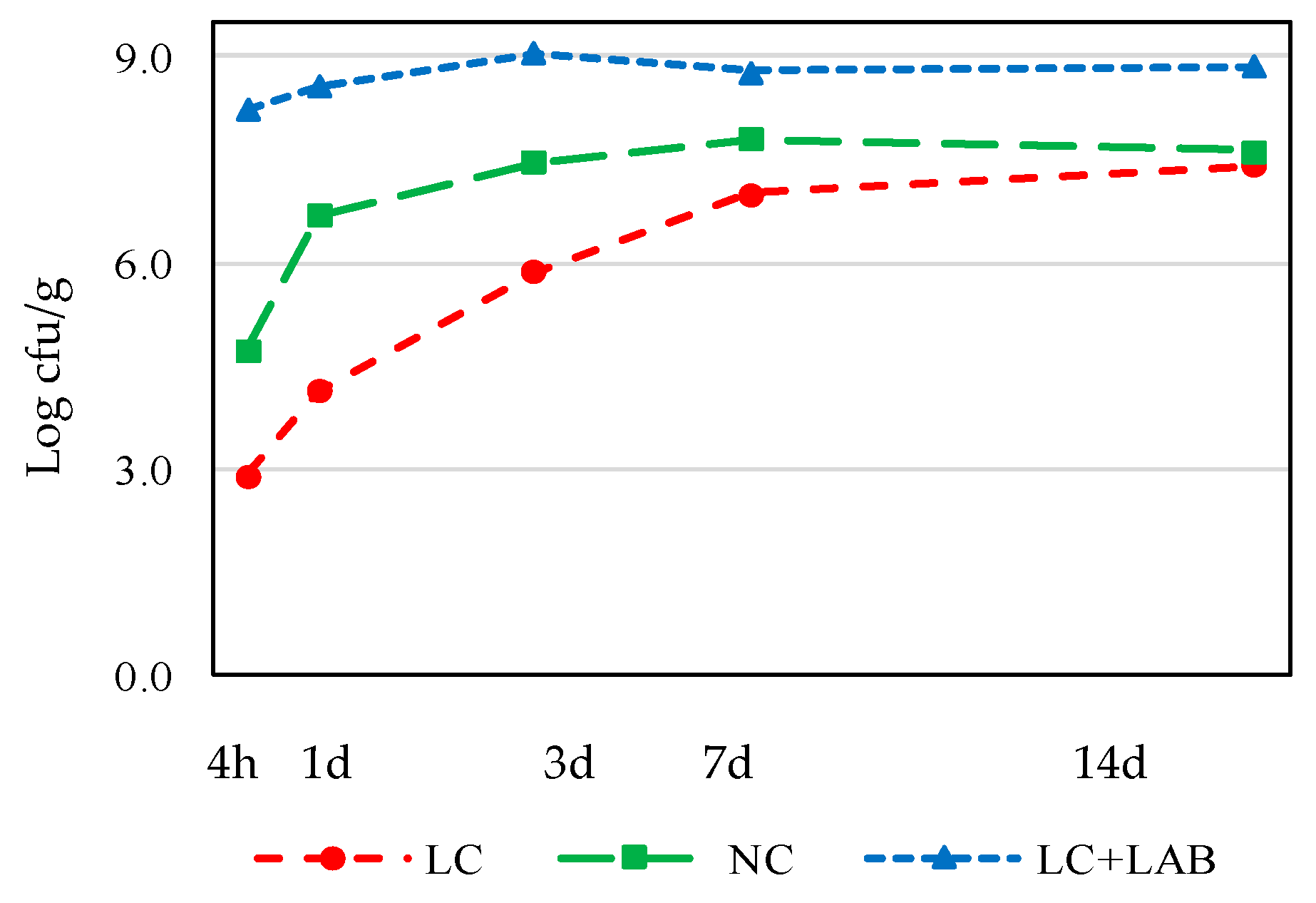
3.1. Naturally Fermented Sausage Batter
3.2. Part 2. Naturally Fermented Sausage—Chouriço
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- EFSA; ECDC. The European Union One Health 2018 Zoonoses Report. EFSA J. 2019, 17. [Google Scholar] [CrossRef]
- CDC. CDC 2015 Surveillance Report—FoodNet. 2017. Available online: https://www.cdc.gov/foodnet/pdfs/FoodNet-Annual-Report-2015-508c.pdf (accessed on 22 May 2020).
- Holck, A.; Axelsson, L.; McLeod, A.; Rode, T.M.; Heir, E. Health and safety considerations of fermented sausages. J. Food Qual. 2017, 2017, 1–25. [Google Scholar] [CrossRef]
- Meloni, D. Presence of Listeria monocytogenes in mediterranean-style dry fermented sausages. Foods 2015, 4, 34–50. [Google Scholar] [CrossRef] [PubMed]
- Kadariya, J.; Smith, T.C.; Thapaliya, D. Staphylococcus aureus and staphylococcal food-borne disease: An ongoing challenge in public health. BioMed Res. Int. 2014, 2014, 827965. [Google Scholar] [CrossRef]
- Freilich, S.; Zarecki, R.; Eilam, O.; Segal, E.S.; Henry, C.S.; Kupiec, M.; Gophna, U.; Sharan, R.; Ruppin, E. Competitive and cooperative metabolic interactions in bacterial communities. Nat. Commun. 2011, 2, 589. [Google Scholar] [CrossRef]
- Simon-Cornu, M.; Billoir, E.; Bergis, H.; Beaufort, A.; Zuliani, V. Modeling microbial competition in food: Application to the behavior of Listeria monocytogenes and lactic acid flora in pork meat products. Food Microbiol. 2011, 28, 639–647. [Google Scholar] [CrossRef]
- Laranjo, M.; Elias, M.; Fraqueza, M. The use of starter cultures in traditional meat products. J. Food Qual. 2017, 2017, 1–18. [Google Scholar] [CrossRef]
- Talon, R.; Leroy, S. Diversity and safety hazards of bacteria involved in meat fermentations. Meat Sci. 2011, 89, 303–309. [Google Scholar] [CrossRef]
- Hospital, X.F.; Hierro, E.; Stringer, S.; Fernández, M. A study on the toxigenesis by Clostridium botulinum in nitrate and nitrite-reduced dry fermented sausages. Int. J. Food Microbiol. 2016, 218, 66–70. [Google Scholar] [CrossRef]
- Majou, D.; Christieans, S. Mechanisms of the bactericidal effects of nitrate and nitrite in cured meats. Meat Sci. 2018, 145, 273–284. [Google Scholar] [CrossRef]
- Patarata, L.; Martins, S.; Silva, J.A.; Fraqueza, M. Red wine and garlic as a possible alternative to minimize the use of nitrite for controlling clostridium sporogenes and salmonella in a cured sausage: Safety and sensory implications. Foods 2020, 9, 206. [Google Scholar] [CrossRef] [PubMed]
- IARC. Red Meat and Processed Meat; IARC Working Group on the Evaluation of Carcinogenic Risks to Humans International Agency for Research on Cancer: Geneva, Switzerland, 2018; Volume 114.
- Di Vita, G.; Blanc, S.; Mancuso, T.; Massaglia, S.; La Via, G.; D’Amico, M. Harmful Compounds and Willingness to Buy for Reduced-Additives Salami. Outlook Ital. Consum. Int. J. Environ. Res. Public Health 2019, 16, 2605. [Google Scholar] [CrossRef] [PubMed]
- Martins, N.; Ferreira, I.C.; Martins, N. Mountain food products: A broad spectrum of market potential to be exploited. Trends Food Sci. Technol. 2017, 67, 12–18. [Google Scholar] [CrossRef]
- Mortensen, A.; Aguilar, F.; Crebelli, R.; Di Domenico, A.; Dusemund, B.; Frutos, M.J.; Galtier, P.; Gott, D.; Gundert-Remy, U.; Lambre, C.; et al. Re-evaluation of potassium nitrite (E 249) and sodium nitrite (E 250) as food additives. EFSA J. 2017, 15. [Google Scholar] [CrossRef]
- Fraqueza, M.J.; Patarata, L. Fermented Meat Products: From the Technology to the Quality Control. In Fermented Food Products; Sankaranarayanan, A., Amaresan, N., Dhanasekaran, D., Eds.; CRC Press: Boca Raton, FL, USA, 2020; pp. 197–237. [Google Scholar]
- García-Díez, J.; Alheiro, J.; Pinto, A.L.; Soares, L.; Falco, V.; Fraqueza, M.J.; Patarata, L. The Impact of Essential Oils on Consumer Acceptance of Chouriço de vinho—A Dry-Cured Sausage Made from Wine-Marinated Meat—Assessed by the Hedonic Scale, JAR Intensity Scale and Consumers’ “Will to Consume and Purchase”. J. Food Process. Preserv. 2017, 41. [Google Scholar] [CrossRef]
- Coloretti, F.; Tabanelli, G.; Chiavari, C.; Lanciotti, R.; Grazia, L.; Gardini, F.; Montanari, C. Effect of wine addition on microbiological characteristics, volatile molecule profiles and biogenic amine contents in fermented sausages. Meat Sci. 2014, 96, 1395–1402. [Google Scholar] [CrossRef]
- Rason, J.; Laguet, A.; Berge, P.; Dufour, E.; Lebecque, A. Investigation of the physicochemical and sensory homogeneity of traditional French dry sausages. Meat Sci. 2007, 75, 359–370. [Google Scholar] [CrossRef]
- Cadavez, V.; Gonzales-Barron, U.; Pires, P.; Fernandes, E.; Pereira, A.P.; Gomes, A.; Araújo, J.P.; Lopes-Da-Silva, F.; Morales, P.; Fernandes, C.; et al. An assessment of the processing and physicochemical factors contributing to the microbial contamination of salpicão, a naturally-fermented Portuguese sausage. LWT Food Sci. Technol. 2016, 72, 107–116. [Google Scholar] [CrossRef]
- Spaziani, M.; Del Torre, M.; Stecchini, M.L. Changes of physicochemical, microbiological, and textural properties during ripening of Italian low-acid sausages. Proteolysis, sensory and volatile profiles. Meat Sci. 2009, 81, 77–85. [Google Scholar] [CrossRef]
- Díez, J.G.; Patarata, L. Behavior of Salmonella spp., Listeria monocytogenes, and Staphylococcus aureus in chouriço de vinho, a dry fermented sausage made from wine-marinated meat. J. Food Prot. 2013, 76, 588–594. [Google Scholar]
- Silva, V.; Igrejas, G.; Falco, V.; Santos, T.P.; Torres, C.; Oliveira, A.M.; Pereira, J.E.; Amaral, J.S.; Poeta, P. Chemical composition, antioxidant and antimicrobial activity of phenolic compounds extracted from wine industry by-products. Food Control. 2018, 92, 516–522. [Google Scholar] [CrossRef]
- Augustin, J.-C.; Bergis, H.; Midelet-Bourdin, G.; Simon-Cornu, M.; Couvert, O.; Denis, C.; Huchet, V.; Lemonnier, S.; Pinon, A.; Vialette, M.; et al. Design of challenge testing experiments to assess the variability of Listeria monocytogenes growth in foods. Food Microbiol. 2011, 28, 746–754. [Google Scholar] [CrossRef]
- Pereira, J.; Silva, P.; Matos, T.; Patarata, L. Shelf Life Determination of Sliced Portuguese Traditional Blood Sausage- Morcela de Arroz de Monchique through Microbiological Challenge and Consumer Test. J. Food Sci. 2015, 80, M642–M648. [Google Scholar] [CrossRef]
- Linares, M.; Garrido, M.D.; Martins, C.; Patarata, L. Efficacies of Garlic and L. sakei in Wine-Based Marinades for Controlling Listeria monocytogenes and Salmonella spp. in Chouriço de Vinho, a Dry Sausage Made from Wine-Marinated Pork. J. Food Sci. 2013, 78. [Google Scholar] [CrossRef] [PubMed]
- Cristino, R.; Costa, E.; Cosme, F.; Jordão, A.M. General phenolic characterisation, individual anthocyanin and antioxidant capacity of matured red wines from two Portuguese Appellations of Origins. J. Sci. Food Agric. 2013, 93, 2486–2493. [Google Scholar] [CrossRef] [PubMed]
- Alfaiaa, A.; Alfaiab, C.M.; Patarata, L.; Fernandes, M.J.; Fernandes, M.H.; Eliasd, M.; Ribeiro, M.H.; Fraqueza, M.J. Binomial effects of high isostatic pressure and time on the microbiological, sensory characteristics and lipid composition stability of vacuum packed dry fermented sausages ‘chouriço’. Innov. Food Sci. Emerg. Technol. 2015, 32, 37–44. [Google Scholar] [CrossRef][Green Version]
- Fraqueza, M.J.; Patarata, L.; Lauková, A. Protective cultures and bacteriocins. In Fermented Meat Products. Health Aspects; Zdolec, N., Ed.; CRC Press Taylor & Francis Group: Boca Raton, FL, USA, 2017; pp. 228–269. [Google Scholar] [CrossRef]
- Silano, V.; Baviera, J.M.B.; Bolognesi, C.; Brüschweiler, B.J.; Chesson, A.; Cocconcelli, P.S.; Crebelli, R.; Gott, D.M.; Grob, K.; Lampi, E.; et al. Evaluation of the safety and efficacy of the organic acids lactic and acetic acids to reduce microbiological surface contamination on pork carcasses and pork cuts. EFSA J. 2018, 16. [Google Scholar] [CrossRef]
- Dickson, J.S.; Anderson, M.E. Microbiological decontamination of food animal carcasses by washing and sanitizing systems: A Review. J. Food Prot. 1992, 55, 133–140. [Google Scholar] [CrossRef]
- Duan, D.; Wang, H.; Xue, S.; Li, M.; Xu, X. Application of disinfectant sprays after chilling to reduce the initial microbial load and extend the shelf-life of chilled chicken carcasses. Food Control. 2017, 75, 70–77. [Google Scholar] [CrossRef]
- Reichel, J.; Kehrenberg, C.; Krischek, C. Inactivation of Yersinia enterocolitica and Brochothrix thermosphacta on pork by UV-C irradiation. Meat Sci. 2019, 158, 107909. [Google Scholar] [CrossRef]
- EFSA. Growth of spoilage bacteria during storage and transport of meat. EFSA J. 2016, 14, 4523. [Google Scholar] [CrossRef]
- Møller, C.; Ilg, Y.; Aabo, S.; Christensen, B.; Dalgaard, P.; Hansen, T.B. Effect of natural microbiota on growth of Salmonella spp. in fresh pork—A predictive microbiology approach. Food Microbiol. 2013, 34, 284–295. [Google Scholar]
- Gonzales-Barron, U.; Cadavez, V.; Pereira, A.P.; Gomes, A.; Araújo, J.P.; Saavedra, M.; Estevinho, L.; Butler, F.; Pires, P.; Dias, T. Relating physicochemical and microbiological safety indicators during processing of linguiça, a Portuguese traditional dry-fermented sausage. Food Res. Int. 2015, 78, 50–61. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Thakur, M.; Asrani, R.K.; Patial, V. Listeria monocytogenes: A Food-Borne Pathogen. Foodborne Dis. 2018, 55, 157–192. [Google Scholar]
- Baka, A.; Papavergou, E.; Pragalaki, T.; Bloukas, J.G.; Kotzekidou, P. Effect of selected autochthonous starter cultures on processing and quality characteristics of Greek fermented sausages. LWT Food Sci. Technol. 2011, 44, 54–61. [Google Scholar] [CrossRef]
- Fraqueza, M.J.; Borges, A.F.; Patarata, L. Strategies to reduce the formation of carcinogenic chemicals in dry cured meat products. Food Control Biosecur. 2018, 16, 295–342. [Google Scholar]
- Mataragas, M.; Alessandria, V.; Rantsiou, K.; Cocolin, L. Evaluation of the Listeria monocytogenes inactivation during post-process storage of fermented sausages: A basis for the development of a decision support tool. Food Control. 2015, 50, 568–573. [Google Scholar] [CrossRef]
- Christieans, S.; Picgirard, L.; Parafita, E.; Lebert, A.; Gregori, T. Impact of reducing nitrate/nitrite levels on the behavior of Salmonella Typhimurium and Listeria monocytogenes in French dry fermented sausages. Meat Sci. 2018, 137, 160–167. [Google Scholar] [CrossRef]
- Hospital, X.F.; Hierro, E.; Fernández, M. Effect of reducing nitrate and nitrite added to dry fermented sausages on the survival of Salmonella Typhimurium. Food Res. Int. 2014, 62, 410–415. [Google Scholar] [CrossRef]
- Hospital, X.F.; Hierro, E.; Fernández, M. Survival of Listeria innocua in dry fermented sausages and changes in the typical microbiota and volatile profile as affected by the concentration of nitrate and nitrite. Int. J. Food Microbiol. 2012, 153, 395–401. [Google Scholar] [CrossRef]
| Salmonella | S. aureus | L. monocytogenes |
|---|---|---|
| CECT 4155 | ATCC 25923 | NCTC 7973 |
| MPI-B-S07 1 (Chouriço batter) | EDS-B-LM05 1 (Chouriço batter) | EDS-ChM-SA01 1 (Chouriço at the middle of the drying process) |
| EDS-E-S26 1 (Environment of meat products preparation) | MPI-E-LM18 1 (Environment of meat products preparation) | EDS-E-SA07 1 (Environment of meat products preparation) |
| Phase | NOx | Wine | Contamination | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Absent (n = 36) | Present (n = 36) | p | Water (n = 24) | AWS (n = 24) | Wine (n = 24) | p | LC (n = 24) | NC (n = 24) | LC + LAB (n = 24) | p | |
| Salmonella | |||||||||||
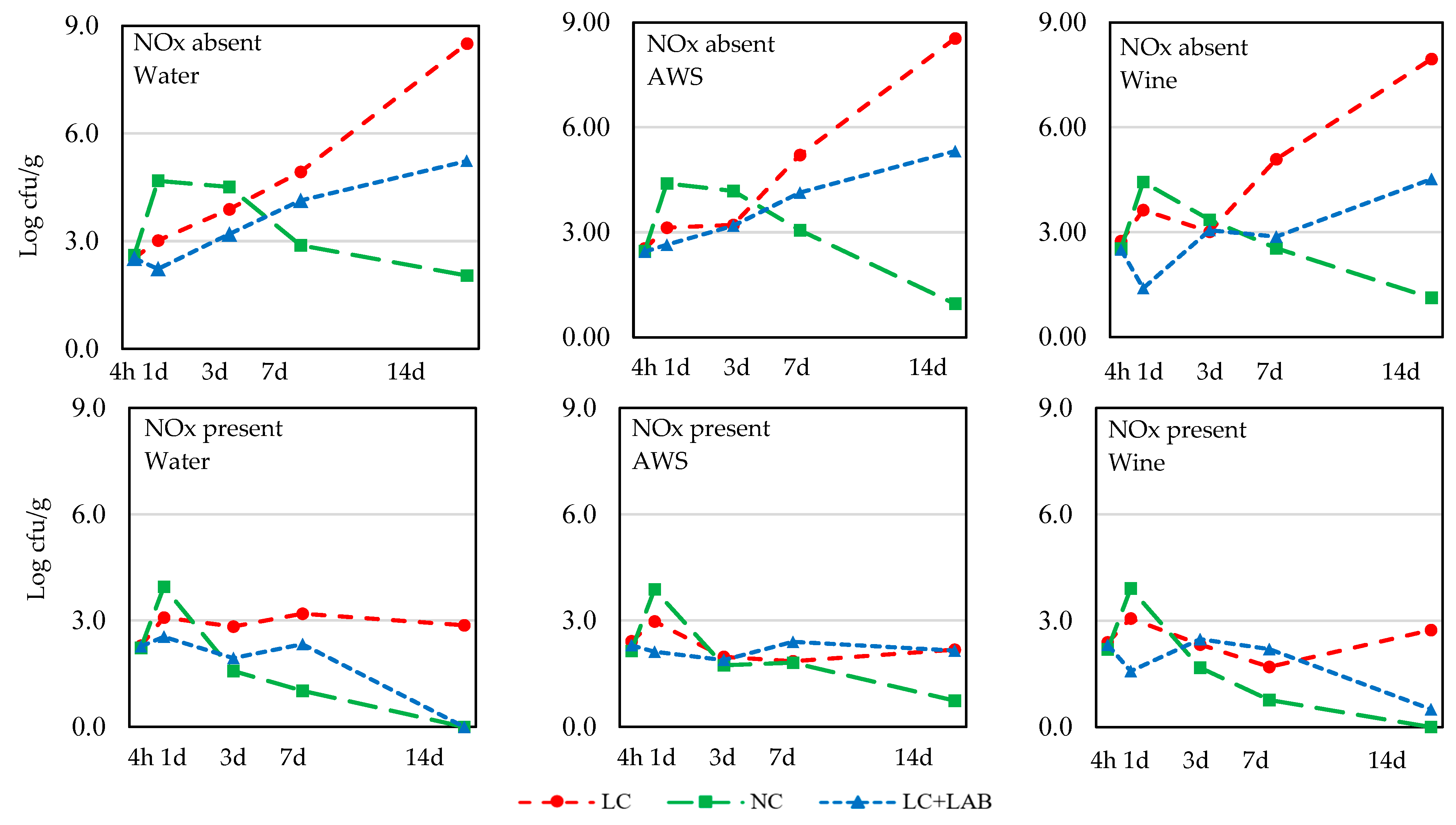
| 4 h | 2.54 ± 0.23 | 2.28 ± 0.37 | 0.001 | 2.41 ± 0.41 | 2.38 ± 0.25 | 2.44 ± 0.32 | ns | 2.48 ± 0.43 | 2.36 ± 0.30 | 2.39 ± 0.24 | ns |
| 1 d | 3.28 ± 1.19 | 3.01 ± 0.96 | ns | 3.25 ± 0.88 | 3.19 ± 0.95 | 3.00 ± 1.38 | ns | 3.15 ± 0.30 b | 4.21 ± 0.41 c | 2.08 ± 1.01 a | <0.001 |
| 3 d | 3.51 ± 1.16 | 2.05 ± 1.27 | <0.001 | 2.99 ± 1.41 | 2.70 ± 1.41 | 2.65 ± 1.46 | ns | 2.87 ± 1.89 | 2.84 ± 1.97 | 2.62 ± 0.93 | ns |
| 7 d | 3.87 ± 1.31 | 1.92 ± 1.32 | <0.001 | 3.08 ± 1.65 | 3.08 ± 1.59 | 2.52 ± 1.67 | ns | 3.67 ± 1.67 b | 2.01 ± 1.80 a | 3.01 ± 0.89 ab | 0.007 |
| 14 d | 4.91 ± 3.08 | 1.24 ± 1.40 | <0.001 | 3.11 ± 3.21 | 3.31 ± 3.01 | 2.80 ± 2.30 | ns | 5.46 ± 3.04 b | 0.80 ± 1.49 a | 2.95 ± 2.27 a | <0.001 |
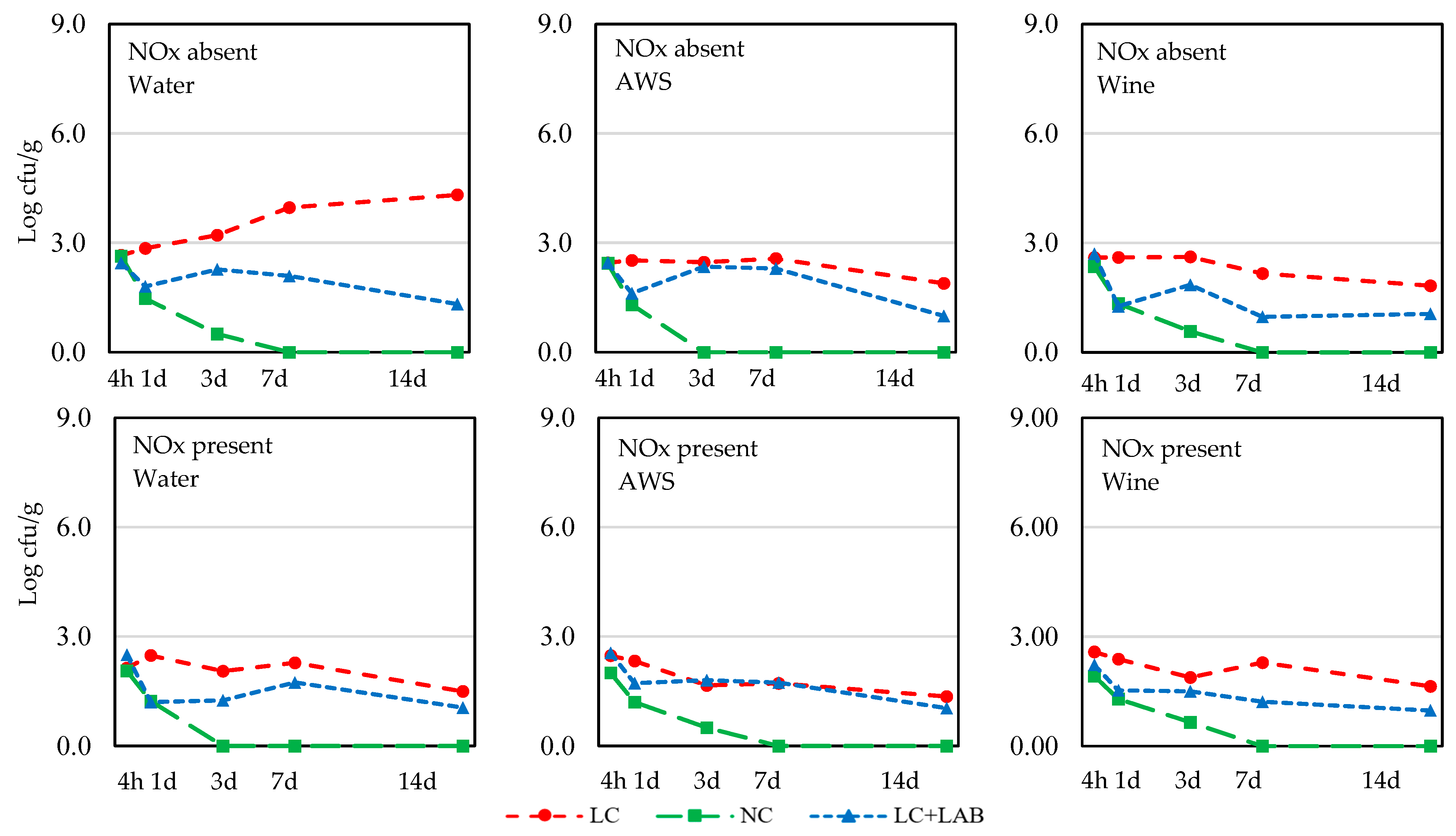
| S. aureus | |||||||||||
| 4 h | 2.53 ± 0.27 | 2.27 ± 0.53 | 0.010 | 2.41 ± 0.60 | 2.39 ± 0.34 | 2.39 ± 0.34 | ns | 2.48 ± 0.59 | 2.23 ± 0.30 | 2.48 ± 0.32 | ns |
| 1 d | 1.86 ± 0.86 | 1.71 ± 0.75 | ns | 1.84 ± 0.90 | 1.78 ± 0.71 | 1.73 ± 0.82 | ns | 2.58 ± 0.29 b | 1.30 ± 0.83 a | 1.52 ± 0.58 a | <0.001 |
| 3 d | 1.76 ± 1.21 | 1.25 ± 0.95 | 0.017 | 1.55 ± 1.24 | 1.46 ± 1.05 | 1.51 ± 1.07 | ns | 2.31 ± 0.64 b | 0.37 ± 0.85 a | 1.84 ± 0.72 b | <0.001 |
| 7 d | 1.56 ± 1.39 | 1.22 ± 1.05 | ns | 1.68 ± 1.43 | 1.38 ± 1.19 | 1.11 ± 1.03 | ns | 2.49 ± 0.95 c | <DLa | 1.67 ± 0.66 b | <0.001 |
| 14 d | 1.27 ± 1.65 | 0.84 ± 0.90 | ns | 1.36 ± 1.54 | 0.88 ± 1.27 | 0.92 ± 1.18 | ns | 2.09 ± 1.60 b | <DLa | 1.07 ± 0.82 a | <0.001 |
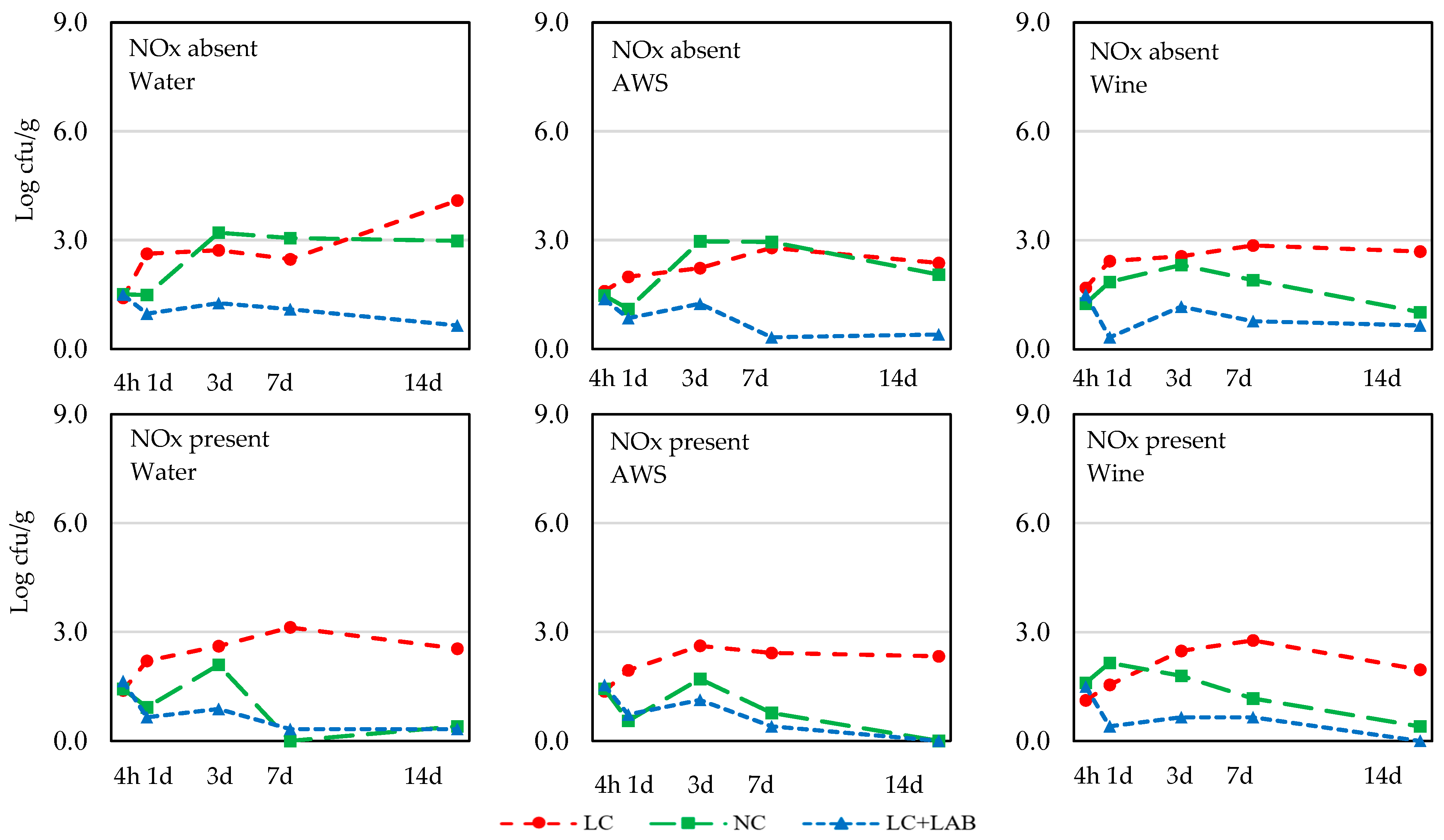
| L. monocytogenes | |||||||||||
| 4 h | 1.48 ± 0.39 | 1.44 ± 0.56 | ns | 1.48 ± 0.56 | 1.47 ± 0.40 | 1.44 ± 0.37 | ns | 1.43 ± 0.68 | 1.45 ± 0.29 | 1.51 ± 0.22 | ns |
| 1 d | 1.51 ± 0.99 | 1.23 ± 0.99 | ns | 1.48 ± 0.99 | 1.19 ± 1.00 | 1.45 ± 1.01 | ns | 2.12 ± 0.59 c | 1.34 ± 1.02 b | 0.65 ± 0.74 a | <0.001 |
| 3 d | 2.19 ± 0.95 | 1.77 ± 0.88 | 0.059 | 2.13 ± 1.04 | 1.98 ± 0.88 | 1.83 ± 0.89 | ns | 2.54 ± 0.44 b | 2.35 ± 0.71 b | 1.06 ± 0.79 a | <0.001 |
| 7 d | 2.02 ± 1.27 | 1.29 ± 1.24 | 0.017 | 1.68 ± 1.48 | 1.61 ± 1.28 | 1.69 ± 1.16 | ns | 2.74 ± 0.73 c | 1.64 ± 1.31 b | 0.59 ± 0.73 a | <0.001 |
| 14 d | 1.88 ± 1.40 | 0.88 ± 1.67 | 0.003 | 1.83 ± 1.57 | 1.19 ± 1.25 | 1.12 ± 1.21 | ns | 2.66 ± 0.87 b | 1.14 ± 1.34 a | 0.34 ± 0.60 a | <0.001 |
| Microorganism Phase of Processing | Control (n = 3) | Wine (n = 3) | Wine + LAB (n = 3) | p | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Salmonella | ||||||||||
| After mixing (4 h) | 2.50 | ± | 0.35 | 2.32 | ± | 0.28 | 2.55 | ± | 0.13 | ns |
| After smoking (1 day) | 3.04 | ± | 0.19 | 1.01 | ± | 1.76 | <LD | 0.078 | ||
| 7 days drying | 1.87 | ± | 1.63 | 0.82 | ± | 1.43 | <LD | ns | ||
| 14 days drying | <LD | <LD | <LD | - | ||||||
| S. aureus | ||||||||||
| After mixing (4 h) | 2.20 | ± | 0.17 | 2.16 | ± | 0.28 | 2.47 | ± | 0.40 | ns |
| After smoking (1 day) | 2.59 | ± | 0.26 | 1.43 | ± | 1.25 | <LD | 0.054 | ||
| 7 days drying | 0.67 | ± | 1.15 | 1.33 | ± | 1.15 | <LD | ns | ||
| 14 days drying | <LD | <LD | <LD | - | ||||||
| L. monocytogenes | ||||||||||
| After mixing (4 h) | 2.20 | ± | 0.17 | 2.46 | ± | 0.45 | 2.26 | ± | 0.24 | ns |
| After smoking (1 day) | 3.10 | ± | 0.44 b | 1.59 | ± | 1.38 ab | <LD | a | 0.034 | |
| 7 days drying | 2.65 | ± | 0.16 b | 1.43 | ± | 1.25 ab | <LD | a | 0.034 | |
| 14 days drying | 2.16 | ± | 0.28 | 0.93 | ± | 1.60 | <LD | ns | ||
| LAB | ||||||||||
| After mixing (4 h) | 2.22 | ± | 0.07 ab | 2.05 | ± | 0.10 a | 6.14 | ± | 0.53 b | 0.027 |
| After smoking (1 day) | 5.67 | ± | 0.00 ab | 4.83 | ± | 0.01 a | 7.73 | ± | 0.03 b | 0.024 |
| 7 days drying | 6.55 | ± | 0.01 a | 7.39 | ± | 0.01 b | 7.10 | ± | 0.04 ab | 0.027 |
| 14 days drying | 7.10 | ± | 0.04 ab | 6.87 | ± | 0.04 a | 7.48 | ± | 0.1 b | 0.044 |
| Enterobacteriaceae | ||||||||||
| After mixing (4 h) | 2.28 | ± | 0.11 a | 1.22 | ± | 0.24 ab | 0.73 | ± | 0.63 b | 0.048 |
| After smoking (1 day) | 1.77 | ± | 0.11 a | 0.57 | ± | 0.51 ab | <LD | b | 0.034 | |
| 7 days drying | 1.45 | ± | 0.26 | <LD | <LD | 0.022 Ψ | ||||
| 14 days drying | <LD | <LD | <LD | - | ||||||
| Pseudomonas | ||||||||||
| After mixing (4 h) | 2.74 | ± | 0.13 a | 2.40 | ± | 0.20 ab | 2.19 | ± | 0.20 b | 0.044 |
| After smoking (1 day) | 2.72 | ± | 0.26 a | 0.57 | ± | 0.98 ab | <LD | b | 0.035 | |
| 7 days drying | 1.33 | ± | 0.15 | <LD | <LD | ns | ||||
| 14 days drying | <LD | <LD | <LD | - | ||||||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Patarata, L.; Novais, M.; Fraqueza, M.J.; Silva, J.A. Influence of Meat Spoilage Microbiota Initial Load on the Growth and Survival of Three Pathogens on a Naturally Fermented Sausage. Foods 2020, 9, 676. https://doi.org/10.3390/foods9050676
Patarata L, Novais M, Fraqueza MJ, Silva JA. Influence of Meat Spoilage Microbiota Initial Load on the Growth and Survival of Three Pathogens on a Naturally Fermented Sausage. Foods. 2020; 9(5):676. https://doi.org/10.3390/foods9050676
Chicago/Turabian StylePatarata, Luis, Margarida Novais, Maria João Fraqueza, and José António Silva. 2020. "Influence of Meat Spoilage Microbiota Initial Load on the Growth and Survival of Three Pathogens on a Naturally Fermented Sausage" Foods 9, no. 5: 676. https://doi.org/10.3390/foods9050676
APA StylePatarata, L., Novais, M., Fraqueza, M. J., & Silva, J. A. (2020). Influence of Meat Spoilage Microbiota Initial Load on the Growth and Survival of Three Pathogens on a Naturally Fermented Sausage. Foods, 9(5), 676. https://doi.org/10.3390/foods9050676






