Cake Perception, Texture and Aroma Profile as Affected by Wheat Flour and Cocoa Replacement with Carob Flour
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Cake Preparation and Measurements
2.3. Sensory Test
2.4. Volatile Compounds Analysis
2.5. Statistical Analysis
3. Results
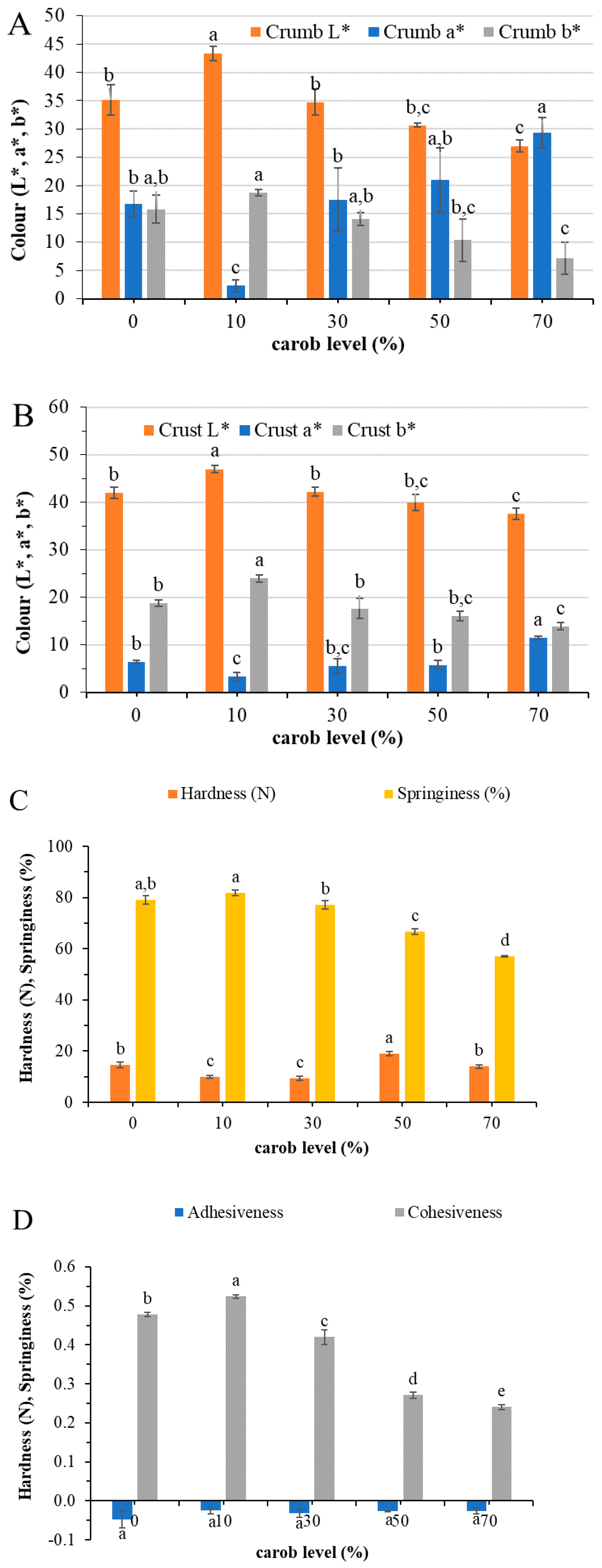
3.1. Effect of Carob Flour in the Physical Properties of Batter and Cake
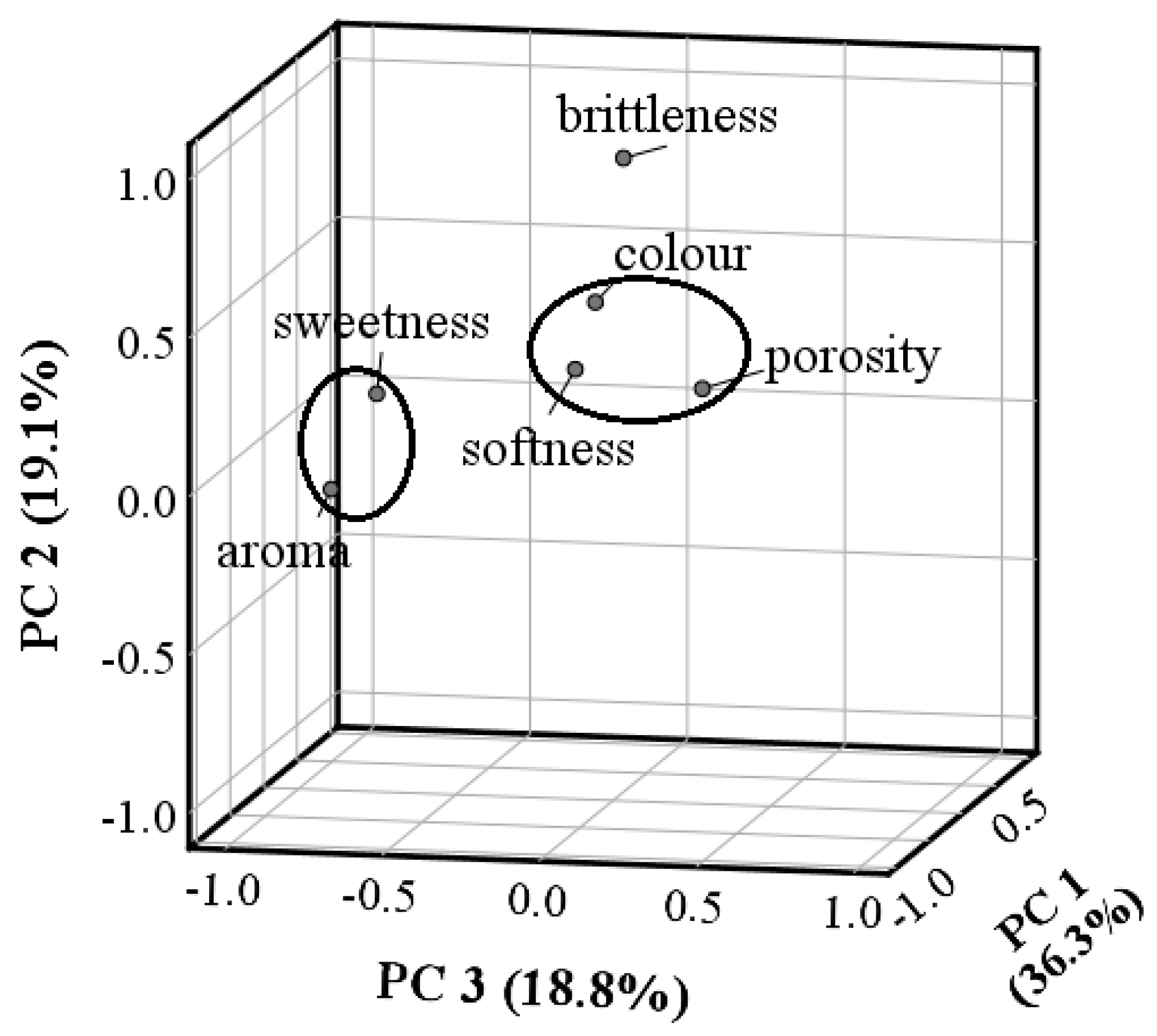
3.2. Effect of Carob Flour in the Sensory Analysis
3.3. GC-MS Analysis
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Al-Sayed, H.M.A.; Ahmed, A.R. Utilization of watermelon rinds and sharlyn melon peels as a natural source of dietary fiber and antioxidants in cake. Ann. Agric. Sci. 2013, 58, 83–95. [Google Scholar] [CrossRef]
- Campbell, L.; Euston, S.R.; Ahmed, M.A. Effect of addition of thermally modified cowpea protein on sensory acceptability and textural properties of wheat bread and sponge cake. Food Chem. 2016, 194, 1230–1237. [Google Scholar] [CrossRef] [PubMed]
- Gularte, M.A.; de la Hera, E.; Gómez, M.; Rosell, C.M. Effect of different fibers on batter and gluten-free layer cake properties. LWT-Food Sci. Technol. 2012, 48, 209–214. [Google Scholar] [CrossRef]
- Kim, J.H.; Lee, H.J.; Lee, H.-S.; Lim, E.-J.; Imm, J.-Y.; Suh, H.J. Physical and sensory characteristics of fibre-enriched sponge cakes made with Opuntia humifusa. LWT 2012, 47, 478–484. [Google Scholar] [CrossRef]
- Lebesi, D.M.; Tzia, C. Use of endoxylanase treated cereal brans for development of dietary fiber enriched cakes. Innov. Food Sci. Emerg. Technol. 2012, 13, 207–214. [Google Scholar] [CrossRef]
- Sudha, M.L.; Baskaran, V.; Leelavathi, K. Apple pomace as a source of dietary fiber and polyphenols and its effect on the rheological characteristics and cake making. Food Chem. 2007, 104, 686–692. [Google Scholar] [CrossRef]
- Yousif, A.K.; Alghzawi, H.M. Processing and characterization of carob powder. Food Chem. 2000, 69, 283–287. [Google Scholar] [CrossRef]
- Papaefstathiou, E.; Agapiou, A.; Giannopoulos, S.; Kokkinofta, R. Nutritional characterization of carobs and traditional carob products. Food Sci. Nutr. 2018, 6, 2151–2161. [Google Scholar] [CrossRef]
- Papagiannopoulos, M.; Wollseifen, H.R.; Mellenthin, A.; Haber, B.; Galensa, R. Identification and quantification of polyphenols in carob fruits (Ceratonia siliqua L.) and derived products by HPLC-UV-ESI/MSn. J. Agric. Food Chem. 2004, 52, 3784–3791. [Google Scholar] [CrossRef]
- Loullis, A.; Pinakoulaki, E. Carob as cocoa substitute: A review on composition, health benefits and food applications. Eur. Food Res. Technol. 2018, 244, 959–977. [Google Scholar] [CrossRef]
- McLeod, G.; Forcen, M. Analysis of volatile components derived from the carob bean Ceratonia siliqua. Phytochemistry 1992, 31, 3113–3119. [Google Scholar] [CrossRef]
- Arrighi, W.; Hartman, T.; Ho, C.T. Carob bean aroma dependence on roasting conditions. Perfum. Flavor. 1997, 22, 31–41. [Google Scholar]
- Fadel, H.H.M.; Abdel Mageed, M.A.; Abdel Samad, A.K.M.E.; Lotfy, S.N. Cocoa substitute: Evaluation of sensory qualities and flavour stability. Eur. Food Res. Technol. 2006, 223, 125–131. [Google Scholar] [CrossRef]
- Boublenza, I.; Lazouni, H.A.; Ghaffari, L.; Ruiz, K.; Fabiano-Tixier, A.-S.; Chemat, F. Influence of roasting on sensory, antioxidant, aromas, and physicochemical properties of carob pod powder (Ceratonia siliqua L.). J. Food Qual. 2017, 2017, 4193672. [Google Scholar] [CrossRef]
- Rosa, C.S.; Tessele, K.; Prestes, R.C.; Silveira, M.; Franco, F. Effect of substituting of cocoa powder for carob flour in cakes made with soy and banana flours. Int. Food Res. J. 2015, 22, 2111–2118. [Google Scholar]
- Benković, M.; Radić, K.; Vitali Čepo, D.; Jaškūnas, E.; Janutis, L.; Morkunaite, M.; Srečec, S. Production of cocoa and carob-based drink powders by foam mat drying. J. Food Process Eng. 2018, 41, e12825. [Google Scholar] [CrossRef]
- Pawłowska, K.; Kuligowski, M.; Jasińska-Kuligowska, I.; Kidoń, M.; Siger, A.; Rudzińska, M.; Nowak, J. Effect of replacing cocoa powder by carob powder in the muffins on sensory and physicochemical properties. Plant Foods Hum. Nutr. 2018, 73, 196–202. [Google Scholar] [CrossRef] [PubMed]
- Berk, E.; Sumnu, G.; Sahin, S. Usage of carob bean flour in gluten free cakes. Chem. Eng. Trans. 2017, 57, 1909–1914. [Google Scholar] [CrossRef]
- Fidan, H.; Petkova, N.; Sapundzhieva, T.; Baeva, M.; Goranova, Z.; Slavov, A.; Krastev, L. Carob syrup and carob flour (Ceratonia siliqua L.) as functional ingredients in sponge cakes. Carpathian J. Food Sci. Technol. 2019, 11, 71–82. [Google Scholar]
- Román, L.; González, A.; Espina, T.; Gómez, M. Degree of roasting of carob flour affecting the properties of gluten-free cakes and cookies. J. Food Sci. Technol. 2017, 54, 2094–2103. [Google Scholar] [CrossRef]
- Paraskevopoulou, A.; Anagnostara, I.; Bezati, G.; Rizou, T.; Pavlidou, E.; Vouvoudi, E.; Kiosseoglou, V. Water extraction residue from maize milling by-product as a potential functional ingredient for the enrichment with fibre of cakes. LWT 2020, 129, 109604. [Google Scholar] [CrossRef]
- Chauvin, M.A.; Younce, F.; Ross, C.; Swanson, B. Standard scales for crispmess, crackliness and crunchiness in dry and wet foods: Relationship with acustical determinations. J. Texture Stud. 2008, 39, 345–368. [Google Scholar] [CrossRef]
- Paraskevopoulou, A.; Chrysanthou, A.; Koutidou, M. Characterisation of volatile compounds of lupin protein isolate-enriched wheat flour bread. Food Res. Int. 2012, 48, 568–577. [Google Scholar] [CrossRef]
- Gómez, M.; Manchón, L.; Oliete, B.; Ruiz, E.; Caballero, P.A. Adequacy of wholegrain non-wheat flours for layer cake elaboration. LWT-Food Sci. Technol. 2010, 43, 507–513. [Google Scholar] [CrossRef]
- Matsakidou, A.; Blekas, G.; Paraskevopoulou, A. Aroma and physical characteristics of cakes prepared by replacing margarine with extra virgin olive oil. LWT-Food Sci. Technol. 2010, 43, 949–957. [Google Scholar] [CrossRef]
- Rega, B.; Guerard, A.; Delarue, J.; Maire, M.; Giampaoli, P. On-line dynamic HS-SPME for monitoring endogenous aroma compounds released during the baking of a model cake. Food Chem. 2009, 112, 9–17. [Google Scholar] [CrossRef]
- Whitfield, F.B.; Mottram, D.S. Volatiles from interactions of Maillard reactions and lipids. Crit. Rev. Food Sci. Nutr. 1992, 31, 1–58. [Google Scholar] [CrossRef]
- Crafack, M.; Keul, H.; Eskildsen, C.E.; Petersen, M.A.; Saerens, S.; Blennow, A.; Skovmand-Larsen, M.; Swiegers, J.H.; Petersen, G.B.; Heimdal, H.; et al. Impact of starter cultures and fermentation techniques on the volatile aroma and sensory profile of chocolate. Food Res. Int. 2014, 63, 306–316. [Google Scholar] [CrossRef]
- Farag, M.A.; Maamoun, A.A.; Ehrlich, A.; Fahmy, S.; Wesjohann, L.A. Assessment of sensory metabolites distribution in 3 cactus Opuntia ficus-indica fruit cultivars using UV fingerprinting and GC/MS profiling techniques. LWT 2017, 80, 145–154. [Google Scholar] [CrossRef]
- Bonvehí, J.S. Investigation of aromatic compounds in roasted cocoa powder. Eur. Food Res. Technol. 2005, 221, 19–29. [Google Scholar] [CrossRef]
- Frauendorfer, F.; Schieberle, P. Identification of the key aroma compounds in cocoa powder based on molecular sensory correlations. J. Agric. Food Chem. 2006, 54, 5521–5529. [Google Scholar] [CrossRef] [PubMed]
- Cantalejo, M.J. Analysis of Volatile Components Derived from Raw and Roasted Earth-Almond (Cyperus esculentus L.). J. Agric. Food Chem. 1997, 45, 1853–1860. [Google Scholar] [CrossRef]
- Aprotosoaie, A.C.; Luca, S.V.; Miron, A. Flavor chemistry of cocoa and cocoa products—an overview. Compr. Rev. Food Sci. Food Saf. 2016, 15, 73–91. [Google Scholar] [CrossRef]
- Biner, B.; Gubbuk, H.; Karhan, M.; Aksu, M.; Pekmezci, M. Sugar profiles of the pods of cultivated and wild types of carob bean (Ceratonia siliqua L.) in Turkey. Food Chem. 2007, 100, 1453–1455. [Google Scholar] [CrossRef]
- Bailey, S.D.; Mitchell, D.G.; Bazinet, M.L.; Weurman, C. Studies on the volatile components of different varieties of cocoa beans. J. Food Sci. 1962, 27, 165–170. [Google Scholar] [CrossRef]
- The Pherobase: Database of Pheromones and Semiochemicals. Available online: https://www.pherobase.com/ (accessed on 30 October 2020).
- Flavornet. Available online: http://www.flavornet.org/ (accessed on 30 October 2020).
- The Good Scents Company Information System. Available online: http://www.thegoodscentscompany.com/ (accessed on 30 October 2020).
- Buttery, R.G.; Ling, L.C. Volatile flavor components of corn tortillas and related products. J. Agric. Food Chem. 1995, 43, 1878–1882. [Google Scholar] [CrossRef]
- Pino, J.A.; Mesa, J. Contribution of volatile compounds to mango (Mangifera indica L.) aroma. Flavour Fragr. J. 2006, 21, 207–213. [Google Scholar] [CrossRef]
- Van Gemert, L.J. Odour Thresholds: Compilations of Odour Threshold Values in Air, Water and Other Media, 2nd ed.; Oliemans Punter & Partners BV: Zeist, The Netherlands, 2011. [Google Scholar]
- Buttery, R.G.; Ling, L.C. Additional studies on flavor components of corn tortilla chips. J. Agric. Food Chem. 1998, 46, 2764–2769. [Google Scholar] [CrossRef]
- Giri, A.; Osako, K.; Okamoto, A.; Ohshima, T. Olfactometric characterization of aroma active compounds in fermented fish paste in comparison with fish sauce, fermented soy paste and sauce products. Food Res. Int. 2010, 43, 1027–1040. [Google Scholar] [CrossRef]
| Recipe | Wheat Flour (g) | Carob Flour (g) | Margarine (g) | Sugar (g) | Egg (g) | Cocoa (g) |
|---|---|---|---|---|---|---|
| 0 (control) | 60 | - | 75 | 75 | 75 | 15 |
| 10 | 67.5 | 7.5 | 75 | 75 | 75 | - |
| 30 | 52.5 | 22.5 | 75 | 75 | 75 | - |
| 50 | 37.5 | 37.5 | 75 | 75 | 75 | - |
| 70 | 22.5 | 52.5 | 75 | 75 | 75 | - |
| Carob Flour Level (%) | Cake Batter Density (g/cm3) | Cake Specific Volume (mL/g) | Cake Yield (%) |
|---|---|---|---|
| 0 | 0.83 ± 0.01 b | 2.22 ± 0.12 a | 8.14 ± 0.40 a |
| 10 | 0.78 ± 0.03 b | 2.01 ± 0.08 ab | 7.10 ±0.32 ab |
| 30 | 0.79 ± 0.01 b | 1.93 ± 0.00 abc | 7.37 ± 0.40 ab |
| 50 | 0.80 ± 0.03 b | 1.78 ± 0.10 bc | 7.45 ± 0.15 ab |
| 70 | 1.30 ± 0.03 a | 1.66 ± 0.02 c | 6.31 ± 0.12 b |
| Carob Flour Level (%) | Color | Porosity | Aroma | Sweetness | Softness | Brittleness | Overall Acceptability |
|---|---|---|---|---|---|---|---|
| 0 | 7.8 ± 3.3 b | 7.5 ± 2.9 a | 7.5 ± 3.1 a | 6.2 ± 3.0 b | 8.9 ± 1.8 ab | 8.8 ± 2.8 a | 8.9 ± 3.9 a |
| 10 | 3.0 ± 2.7 c | 7.9 ± 2.7 a | 6.3 ± 3.6 a | 7.0 ± 3.1 b | 10.4 ± 3.2 a | 7.1 ± 3.7 a | 9.1 ± 3.0 a |
| 30 | 7.9 ± 3.4 b | 9.2 ± 3.4 a | 7.4 ± 3.9 a | 8.4 ± 2.3 ab | 9.7 ± 2.7 ab | 7.2 ± 2.5 a | 10.4 ± 2.9 a |
| 50 | 10.5 ± 2.5 ab | 11.0 ± 2.5 a | 8.5 ± 3.9 a | 9.1 ± 3.0 ab | 6.9 ± 3.1 b | 5.7 ± 3.4 a | 9.7 ± 2.5 a |
| 70 | 12.6 ± 1.2 a | 11.1 ± 2.3 a | 7.9 ± 3.1 a | 10.8 ± 3.3 a | 6.8 ± 3.5 b | 8.7 ± 4.0 a | 8.5 ± 4.0 a |
| Component | Communalities | |||
|---|---|---|---|---|
| 1 | 2 | 3 | ||
| Color | 0.713 | 0.332 | −0.208 | 0.747 |
| Porosity | 0.877 | 0.042 | 0.098 | 0.741 |
| Aroma | −0.101 | −0.143 | −0.875 | 0.746 |
| Sweetness | 0.106 | 0.127 | −0.773 | 0.672 |
| Softness | −0.739 | 0.380 | 0.037 | 0.693 |
| Brittleness | −0.012 | 0.921 | 0.036 | 0.848 |
| Peak No a | Compound b | RI c | ID d | Odor Description e | OT f(mg/kg) | Concentration (OAV) | |||
|---|---|---|---|---|---|---|---|---|---|
| Carob | Cocoa | ||||||||
| Aldehydes (18) | |||||||||
| 1 | 3-Methyl-butanal | 911 | 1 | Malty, chocolate, cocoa | 0.0002 1 | 357.4 ± 42.8 | (1786.8) | 368.3 ± 77.1 | (1841.4) |
| 3 | Pentanal | 971 | 1 | Almond, pungent | 0.012 1 | 525.2 ± 91.8 | (43.8) | 520.9 ± 88.1 | (43.4) |
| 7 | Hexanal | 1069 | 1,2 | Grass, fatty | 0.0045 2 | 1453 ± 89.9 | (323.0) | 1400 ± 278.1 | (311.1) |
| 8 | (E)-2-Pentenal | 1122 | 1 | Apple, fruity, pungent | 0.31 3 | 199.1 ± 22.5 | (0.6) | − g | |
| 12 | Heptanal | 1178 | 1 | Fatty, rancid | 0.003 1 | 294.7 ± 37.9 * | (98.2) | 212.1 ± 21.8 | (70.7) |
| 15 | (E)-2-Hexenal | 1211 | 1,2 | Green, apple | 0.017 4 | 113 ± 17.1 * | (6.6) | 37.5 ± 6.1 | (2.2) |
| 22 | Octanal | 1282 | 1 | Citrus, green, oily | 0.007 4 | 295.8 ± 36.7 * | (422.6) | 217.9 ± 29.4 | (311.3) |
| 25 | (E)-2-Heptenal | 1314 | 1 | Green, fatty | 0.013 4 | 217.2 ± 19.0 | (16.7) | 229.0 ± 36.4 | (10.5) |
| 34 | Nonanal | 1386 | 1 | Tallowy, soapy | 0.001 1 | 358.4 ± 24.9 | (358.4) | 309.7 ± 17.6 | (309.7) |
| 40 | (E)-2-Octenal | 1418 | 1 | Fresh, leafy, fatty | 0.003 4 | 183.7 ± 8.8 * | (61.2) | 128.5 ± 7.9 | (42.8) |
| 51 | (E,E)-2,4-Heptadienal | 1484 | 1 | Nutty, fatty, hay, fishy | 0.0154 5 | 228.2 ± 25.0 | (14.8) | 214.8 ± 17.6 | (13.9) |
| 56 | Benzaldehyde | 1508 | 1 | Bitter almond | 0.35 1 | 620.3 ± 23.6 * | (1.8) | 1623.2 ± 182.7 | (4.6) |
| 58 | (E)-2-Nonenal | 1522 | 1 | Cucumber, fatty | 0.00008 1 | 114.9 ± 9.0 * | (1436.3) | 75.0 ± 10.6 | (937.5) |
| 68 | Phenylacetaldehyde | 1626 | 1 | Honey, green | 0.004 1 | 138.2 ± 13.9 * | (34.6) | 407.7 ± 20.4 | (101.9) |
| 77 | Dodecanal | 1701 | 1 | Lily, fat, citrus | 0.0016 3 | 60.5 ± 6.7 * | (37.8) | 37.9 ± 1.0 | (23.7) |
| 82 | (E,Z)-2,4-Decadienal | 1754 | 1 | Fried, fatty | 0.00007 1 | 69.1 ± 5.8c * | (987.1) | 49.3 ± 2.4 | (704.3) |
| 85 | (E,E)-2,4-Decadienal | 1794 | 1 | Fatty, fried, citrus | 0.00007 1 | 216.9 ± 4.9 * | (3098.6) | 173.1 ± 15.5 | (2472.9) |
| 113 | Vanillin | 2542 | 1,2 | Vanilla, chocolate | 0.058 1 | 5.3 ± 0.3 | (0.1) | 6.1 ± 2.8 | (0.1) |
| Subtotal | 5508.5 | (8728.9) | 5917.9 | (7202.1) | |||||
| Alcohols (17) | |||||||||
| 2 | Ethyl alcohol | 925 | 1 | Pleasant, fresh | 100 2 | 4351 ± 610.3 | (<0.1) | 3734.7 ± 381 | (<0.1) |
| 9 | 1-Butanol | 1142 | 1 | Fruity | 0.50 4 | 48.0 ± 3.4 | (0.1) | 42.6 ± 13.1 | (0.1) |
| 10 | 1-Penten-3-ol | 1159 | 1 | Butter, pungent | 0.40 4 | 73.4 ± 19.4 | (0.2) | 103.1 ± 27.2 | (0.3) |
| 17 | 1-Pentanol | 1249 | 1,2 | Floral, sweet, green | 0.1502 5 | 441.3 ± 89.2 | (2.9) | 568.8 ± 99.9 | (3.8) |
| 27 | (Z)-2-Penten-1-ol | 1321 | 1 | Fruity, green | 0.72 3 | 163.1 ± 24.3 * | (0.2) | 232.1 ± 28.0 | (0.3) |
| 31 | 1-Hexanol | 1353 | 1,2 | Green, fatty, leafy | 0.5 2 | 1602 ± 125.2 | (3.2) | 1800.8 ± 263.7 | (3.6) |
| 37 | 2-Butoxy-ethanol | 1394 | 1 | Earthy, nutty | 0.88 3 | 174.9 ± 8.0 * | (0.2) | 242.8 ± 17.5 | (0.3) |
| 45 | 1-Octen-3-ol | 1452 | 1 | Earthy, green | 0.0014 1 | 102.1 ± 14.5 * | (72.9) | 146.1 ± 26.5 | (104.4) |
| 47 | 1-Heptanol | 1456 | 1 | Green, fresh, nutty | 0.425 2 | 429.2 ± 53.7 * | (1.0) | 531.0 ± 31.7 | (1.2) |
| 53 | 2-Ethyl-1-hexanol | 1491 | 1 | Green, vegetable | 25.48 5 | 263.6 ± 23.3 | (0.9) | 263.2 ± 26.2 | (0.9) |
| 60 | 1-Octanol | 1557 | 1,2 | Herbal, fatty, green | 0.1258 5 | 269.8 ± 20.4 | (2.1) | 249.9 ± 7.5 | (2.0) |
| 67 | (E)-2-Octen-1-ol | 1612 | 1 | Green, citrus | 0.02 3 | 28.2 ± 1.4 | (1.4) | 40.6 ± 10.2 | (2.0) |
| 71 | 1-Nonanol | 1659 | 1 | Oily, green, floral | 0.0455 5 | 94.8 ± 10.1 | (2.1) | 90.6 ± 2.2 | (2.0) |
| 89 | Benzyl alcohol | 1866 | 1 | Sweet, fresh | 2.54 5 | 314.1 ± 12.7 | (0.1) | 316.9 ± 22.0 | (0.1) |
| 91 | Phenylethyl alcohol | 1897 | 1,2 | Floral | 0.57 5 | 100.3 ± 11.2 * | (0.2) | 314.3 ± 13.4 | (0.6) |
| 96 | 1-Dodecanol | 1965 | 1 | Sweet, soapy, waxy | 0.073 3 | 61.7 ± 16.0 * | (0.8) | 28.5 ± 1.3 | (0.4) |
| 104 | 2-Methoxy-4-vinylphenol | 2183 | 1 | Herbal | 0.003 1 | 17.5 ± 1.6 * | (5.8) | 11.0 ± 1.6 | (3.7) |
| Subtotal | 8534.8 | (92.7) | 8717.0 | (124.1) | |||||
| Ketones (12) | |||||||||
| 4 | 1-Penten-3-one | 1015 | 1 | Pungent, spicy | 0.0013 3 | 147.9 ± 13.3 * | (113.8) | 106.1 ± 18.1 | (81.6) |
| 5 | 2,3-Pentanedione | 1051 | 1 | Caramel, buttery | 0.020 1 | 135.8 ± 19.1 * | (6.8) | 97.3 ± 13.8 | (4.9) |
| 11 | 2-Heptanone | 1175 | 1,2 | Fruity, green | 0.14 4 | 233.2 ± 32.5 * | (1.7) | 132 ± 6.4 | (0.9) |
| 20 | 3-Hydroxy-2-butanone | 1276 | 1,2 | Fatty, creamy | 0.8 3 | − | 38.6 ± 4.3 | (<0.1) | |
| 21 | 2-Octanone | 1278 | 1 | Earthy, herbal | 0.05 4 | 54.1 ± 5.2 | (1.1) | 40.5 ± 8.2 | (0.8) |
| 23 | 1-Hydroxy-2-propanone | 1292 | 1 | Caramel, sweet | 100 4 | 305.1 ± 36.6 | (<0.1) | − | |
| 28 | 6-Methyl-5-hepten-2-one | 1319 | 1 | Earthy, fruity | 0.068 5 | 112.5 ± 17.1 * | (1.7) | 59.9 ± 4.9 | (0.9) |
| 33 | 2-Nonanone | 1381 | 1 | Floral, fatty | 0.005 1 | 199.1 ± 5.9 * | (39.8) | 70.9 ± 7.8 | (14.2) |
| 52 | 2-Decanone | 1486 | 1 | Orange, floral, fatty | tr h | tr | |||
| 57 | 3,5-Octadien-2-one | 1512 | 1 | Fruity, fatty, mushroom | 0.15 1 | 419.8 ± 24.9 * | (2.8) | 681.9 ± 49.3 | (4.5) |
| 63 | 2-Undecanone | 1587 | 1 | Fruity, fresh | 0.007 1 | 35.3 ± 2.1 * | (5.0) | 25.9 ± 2.9 | (3.7) |
| 70 | Acetophenone | 1630 | 1 | Floral, almond | 0.065 4 | 17.7 ± 4.6 | (0.3) | 25.1 ± 4.1 | (0.4) |
| Subtotal | 1660.5 | (172.9) | 1278.2 | (112.0) | |||||
| Monocarbonic acids (16) | |||||||||
| 46 | Acetic acid | 1455 | 1,2 | Pungent, sour | 22 4 | 234.0 ± 56.9 | (<0.1) | tr | (<0.1) |
| 59 | Propanoic acid | 1539 | Rancid | 2 4 | 81.7 ± 13.6 * | (<0.1) | 18.7 ± 2.6 | (<0.1) | |
| 61 | 2-Methylpropanoic acid | 1564 | 1 | Rancid | 0.05 4 | 222.8 ± 29.0 | (4.5) | − | |
| 68 | Butanoic acid | 1624 | 1,2 | Sweaty, cheesy | 0.24 4 | 557.0 ± 96.8 | (2.3) | − | |
| 73 | 2-Methyl-butanoic acid | 1666 | 1 | Acidic, cheesy | 0.5 3 | 162.6 ± 14.2 * | (0.3) | 86.5 ± 4.2 | (0.2) |
| 79 | Pentanoic acid | 1736 | 1 | Sweaty, acidic | 3 3 | 154.1 ± 13.6 * | (0.1) | 11.3 ± 3.4 | (<0.1) |
| 86 | 4-Methyl-pentanoic acid | 1799 | 1 | Pungent, cheesy | 0.81 3 | 45.8 ± 4.5 | 0.1 | − | |
| 88 | Hexanoic acid | 1842 | 1,2 | Rancid, sweaty | 3 2 | 6067 ± 310.7 * | (2.0) | 968.3 ± 175.5 | (0.3) |
| 92 | 2-Ethylhexanoic acid | 1943 | 1 | Sweet, musty | 27 3 | 15.3 ± 2.1 * | (<0.1) | 10.2 ± 1.9 | (<0.1) |
| 93 | Heptanoic acid | 1948 | 1 | Rancid, sour | 0.64 3 | 57.9 ± 10.1 * | (0.1) | 28.3 ± 1.2 | (<0.1) |
| 101 | Octanoic acid | 2055 | 1,2 | Sweaty, fatty | 3 2 | 452 ± 261.8 | (0.2) | 55.2 ± 13.5 | (<0.1) |
| 102 | Nonanoic acid | 2162 | 1 | Green, fatty | 4.6 3 | 4.2 ± 0.3 | (<0.1) | 4.2 ± 0.3 | (<0.1) |
| 107 | Decanoic acid | 2269 | 1 | Fatty, rancid | 10 3 | 64.4 ± 9.9 * | (<0.1) | 15.2 ± 1.9 | (<0.1) |
| 109 | Benzoic acid | 2417 | 1 | Urine | 31.7 ± 2.7 * | 17.2 ± 6.7 | |||
| 110 | Dodecanoic acid | 2482 | 1 | Metallic, fatty | 6.8 ± 0.9 | − | |||
| 113 | Hexadecanoic acid | 2897 | 1 | Waxy | 24.5 ± 5.4 | 21.0 ± 4.1 | |||
| Subtotal | 8182.2 | (19.7) | 1236.1 | (2.2) | |||||
| Pyrazine/Pyrrole derivatives (19) | |||||||||
| 14 | Pyrazine | 1209 | 1 | Sweet, nutty | 75 1 | 54.2 ± 9.0 * | (<0.1) | 28.6 ± 9.3 | (<0.1) |
| 19 | Methylpyrazine | 1257 | 1 | Cocoa, green, pop corn | 60 3 | 70.1 ± 7.1 * | (<0.1) | 84.1 ± 8.5 | (<0.1) |
| 24 | 2,5-Dimethylpyrazine | 1311 | 1 | Chocolate, roasted nuts, earthy | 1.7 4 | 57.6 ± 14.3 * | (<0.1) | 100.4 ± 4.8 | (0.1) |
| 26 | 2,6-Dimethylpyrazine | 1317 | 1 | Chocolate, roasted nuts, fried potato | 1.5 4 | 99.2 ± 3.3 * | (0.1) | 136.0 ± 18.3 | (0.1) |
| 29 | 2,3-Dimethylpyrazine | 1335 | 1 | Nutty, cocoa, coffee | 2.5 4 | 38.5 ± 10.9 * | (<0.1) | 224.5 ± 20.3 | (0.1) |
| 32 | 2-Ethyl-6-methylpyrazine | 1373 | 1 | Nutty, sweet, roasted | 0.04 3 | 36.0 ± 2.6 * | (0.9) | 72.8 ± 6.1 | (1.8) |
| 35 | 2-Ethyl-3-methylpyrazine | 1391 | 1 | Potato, nutty, roasted, cereal | 0.13 3 | 54.9 ± 7.8 | (0.4) | − | |
| 36 | Trimethylpyrazine | 1391 | 1 | Nutty, baked potato, roasted cocoa, burnt | 0.023 4 | − | 351.4 ± 31.3 | (15.3) | |
| 38 | 2-Methoxy-3-methyl pyrazine | 1398 | 1 | Chocolate, nutty, earthy, roasted | 0.004 3 | 170.3 ± 12.7 | (42.6) | 165.5 ± 13.3 | (41.4) |
| 41 | 3-Ethyl-2,5-dimethylpyrazine | 1433 | 1 | Cocoa, coffee, roasted, nutty | 0.0004 1 | 25.8 ± 4.6 * | (64.5) | 87.5 ± 10.4 | (218.8) |
| 42 | 2,5-Diethylpyrazine | 1444 | 1 | Cocoa, roasted, nutty | 0.02 3 | − | 9.9 ± 1.3 | (0.5) | |
| 44 | 2-Ethyl-3,5-dimethyl-pyrazine | 1448 | 1 | Cocoa, nutty, roasted, woody | 0.001 3 | − | 100.5 ± 10.6 | (100.5) | |
| 48 | Tetramethylpyrazine | 1462 | 1 | Nutty, musty, chocolate | 1 3 | tr | (<0.1) | 370.1 ± 18.8 | (0.4) |
| 50 | 3,5-Diethyl-2-methylpyrazine | 1481 | 1 | Nutty, meaty vegetable | − | 14.5 ± 6.0 | |||
| 55 | 2,3,5-Trimethyl-6-ethylpyrazine | 1502 | 1 | − | 141.9 ± 12.1 | ||||
| 64 | 1-Ethyl-1H-pyrrole-2-carboxaldehyde | 1589 | 1 | Roasted, burnt, smoky | 57.3 ± 1.1 | − | |||
| 74 | 2-Acetyl-6-methylpyrazine | 1675 | 1 | Roasted coffee, cocoa | 3 3 | − | 17.2 ± 1.6 | (<0.1) | |
| 9 | 2-Acetylpyrrole | 1957 | 1 | Earthy, hazelnut | 58.6 3 | 1531 ± 57.7 * | (<0.1) | 467 ± 44.1 | (<0.1) |
| 107 | 2-Formyl pyrrole | 2006 | 1 | Musty, beefy, coffee | 85.5 ± 8.6 * | 56.8 ± 8.6 | |||
| Subtotal | 2280.4 | (108.5) | 2414.2 | (378.8) | |||||
| Furan/Pyran derivatives (11) | |||||||||
| 16 | 2-Pentyl furan | 1226 | 1 | Green, earthy, beany | 0.0065 | 212.2 ± 17.4 * | (35.4) | 116 ± 27.2 | (19.3) |
| 18 | Dihydro-2-methyl-3(2H)-furanone | 1257 | 1 | Sweet, caramel, nutty | − | 128.4 ± 20.7 * | − | 64.6 ± 2.7 | − |
| 49 | Furfural | 1464 | 1 | Sweet, bready, almond, caramel, fruity | 3 2 | 4865 ± 346.2 * | (1.6) | 503.2 ± 14.7 | (0.2) |
| 54 | 2-Acetylfuran | 1499 | 1 | Balsamic, cinnamon, sweet, cocoa | 10 1 | 630.4 ± 28.4 * | (0.1) | 65.7 ± 3.2 | (<0.1) |
| 62 | 5-Methyl-furfural | 1565 | 1 | Raw potato, sweet, grass, almond | 0.5 3 | 116.4 ± 19.6 * | (0.2) | 241.5 ± 44.0 | (0.5) |
| 72 | 2-Furanmethanol | 1663 | 1 | Burnt sugar, bread, coffee | 2 4 | 1213 ± 15.6 * | (0.6) | 358.4 ± 7.7 | (0.2) |
| 78 | 5-Methyl-2-furanmethanol | 1725 | 1 | Sweet, caramel | 90.6 ± 2.6 * | 22.5 ± 2.5 | |||
| 95 | Maltol | 1948 | 1 | Caramel | 2.5 4 | 62.9 ± 9.6 * | (<0.1) | 38.8 ± 4.0 | (<0.1) |
| 107 | 2,3-dihydro-3.5-dihydroxy-6-methyl-4H-pyran-4-one | 2249 | 1 | Caramel, roasted | − | 20.3 ± 5.0 | |||
| 109 | 3,5-Dihydroxy-2-methyl-4H-pyran-4-one (5-hydroxymaltol) | 2282 | 1 | Caramel, roasted | 22.6 ± 3.3 | − | |||
| 112 | 5-Hydroxymethylfurfural | 2494 | 1 | Fatty, buttery, caramel | 1000 3 | 43.9 ± 12.2 * | (<0.1) | 4.8 ± 0.5 | (<0.1) |
| Subtotal | 7385.2 | (37.9) | 1435.8 | (20.2) | |||||
| Lactones (10) | |||||||||
| 39 | 5-Methyl-2(3H)furanone (α-Angelica lactone) | 1403 | 1 | Coconut, nutty, herbal, sweet | 90.8 ± 1.3 | − | |||
| 66 | Dihydro-2(3H)-furanone (Butyrolactone) | 1605 | 1 | Caramel, buttery | 1 4 | 75.5 ± 5.4 | (0.1) | 67.5 ± 0.7 | (0.1) |
| 83 | 5-Ethyldihydro-2(3H)-furanone (γ-Hexalactone) | 1679 | 1 | Sweet, herbal, coconut | 0.05 3 | 70.3 ± 6.0 * | (1.4) | 46.5 ± 2.7 | (0.9) |
| 80 | 2(5H)-Furanone(γ-Crotonolactone) | 1738 | 1 | Buttery | 16.9 ± 2.0 * | − | 5.7 ± 0.7 | − | |
| 75 | Tetrahydro-6-methyl-2H-pyran-2-one (δ-Hexalactone) | 1764 | 1 | Creamy, fruity, coconut | 32.5 ± 0.4 | − | |||
| 90 | 5-Butyldihydro-2(3H) furanone (γ-Octalactone) | 1890 | 1 | Sweet, creamy, dairy, coconut | 0.007 4 | 26.8 ± 4.3 * | (3.8) | 11.5 ± 1.7 | (1.6) |
| 98 | Dihydro-5-pentyl-2(3H)-furanone (γ-Nonalactone) | 2000 | 1 | Coconut, sweet, creamy, fatty | 0.0097 3 | 33.5 ± 3.3 * | (3.5) | 15.4 ± 0.7 | (1.6) |
| 101 | 5-Acetyldihydro-2(3H)furanone (Solerone) | 2047 | 1 | Wine-like, fruity | 24.7 ± 0.2 | − | |||
| 104 | Tetrahydro-6-pentyl-2H-pyran-2-one (δ-Decalactone) | 2162 | 1 | Sweet, coconut, creamy, peach | 0.031 3 | 127.5 ± 7.2 * | (4.1) | 67.0 ± 4.0 | (2.2) |
| 106 | 5,6-Dihydro-6-pentyl-2H-pyran-2-one (δ-Decenolactone) | 2200 | 1 | Coconut, creamy, peach, herbal | 31.1 ± 2.6 | 15.5 ± 6.7 | |||
| Subtotal | 529.6 | (13.2) | 229.1 | (6.4) | |||||
| Esters (6) | |||||||||
| 30 | Ethyl 2-hydroxypropanoate | 1342 | 1 | Sweet, fruity, buttery | 50 3 | 35.1 ± 2.7 | (<0.1) | 44.2 ± 8.6 | (0.0) |
| 65 | Methyl benzoate | 1602 | 1 | Dry plum, sweet, herbal | 0.073 3 | 45.8 ± 3.8 | (0.6) | − | |
| 81 | Methyl phenylacetate | 1749 | 1 | Honey, floral, fruity | − | 5.7 ± 1.3 | |||
| 84 | Ethyl phenylacetate | 1775 | 1 | Floral, honey, cocoa | − | 6.2 ± 1.5 | |||
| 87 | 2-Phenylethyl acetate | 1801 | 1,2 | Floral, rose, honey | 0.25 5 | − | 43.1 ± 2.8 | (0.2) | |
| 100 | Isopropyl myristate | 2032 | 1 | Fatty | 33.4 ± 12.0 * | 10.5 ± 2.0 | |||
| Subtotal | 114.3 | (0.6) | 109.7 | (2.1) | |||||
| Sulfur compounds (2) | |||||||||
| 6 | Dimethyl disulfide | 1058 | 1 | Rotten, cabbage | 0.012 4 | 127.2 ± 5.2 | (10.6) | − | |
| 43 | 3-(Methylthio)-propanal (Methional) | 1448 | 1 | Cooked potatoes | 0.0002 1 | 35.8 ± 2.0 | (179.0) | − | |
| Subtotal | 163.0 | (189.6) | |||||||
| Terpenes (2) | |||||||||
| 13 | D-Limonene | 1182 | 1,2 | Citrus | 0.3 4 | 207.8 ± 27.7 | (0.7) | 184 ± 16.1 | (0.6) |
| 76 | α-Terpineol | 1686 | 1 | Fruity, floral | 0.28 3 | 76.1 ± 7.2 | (0.3) | 62.9 ± 6.3 | (0.2) |
| Subtotal | 283.9 | (1.0) | 246.9 | (0.8) | |||||
| Total | 34585.2 | (9365.2) | 21585.0 | (7848.6) | |||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Papageorgiou, M.; Paraskevopoulou, A.; Pantazi, F.; Skendi, A. Cake Perception, Texture and Aroma Profile as Affected by Wheat Flour and Cocoa Replacement with Carob Flour. Foods 2020, 9, 1586. https://doi.org/10.3390/foods9111586
Papageorgiou M, Paraskevopoulou A, Pantazi F, Skendi A. Cake Perception, Texture and Aroma Profile as Affected by Wheat Flour and Cocoa Replacement with Carob Flour. Foods. 2020; 9(11):1586. https://doi.org/10.3390/foods9111586
Chicago/Turabian StylePapageorgiou, Maria, Adamantini Paraskevopoulou, Foteini Pantazi, and Adriana Skendi. 2020. "Cake Perception, Texture and Aroma Profile as Affected by Wheat Flour and Cocoa Replacement with Carob Flour" Foods 9, no. 11: 1586. https://doi.org/10.3390/foods9111586
APA StylePapageorgiou, M., Paraskevopoulou, A., Pantazi, F., & Skendi, A. (2020). Cake Perception, Texture and Aroma Profile as Affected by Wheat Flour and Cocoa Replacement with Carob Flour. Foods, 9(11), 1586. https://doi.org/10.3390/foods9111586







