The Impact of Chitosan on the Chemical Composition of Wines Fermented with Schizosaccharomyces pombe and Saccharomyces cerevisiae
Abstract
1. Introduction
2. Experimental Section
2.1. Microorganisms
2.2. Vinification
2.3. Analytical Determinations
2.4. Statistical Analysis
3. Results and Discussion
3.1. Basic Chemical Parameters of the Wines
3.2. Volatile Compounds
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Jolly, N.P.; Varela, C.; Pretorius, I.S. Not your ordinary yeast: Non-Saccharomyces yeasts in wine production uncovered. FEMS Yeast Res. 2014, 14, 215–217. [Google Scholar] [CrossRef]
- Benito, S. The impacts of Schizosaccharomyces on winemaking. Appl. Microbiol. Biotechnol. 2019, 103, 4291–4312. [Google Scholar] [CrossRef]
- Vilela, A. Use of Nonconventional Yeasts for Modulating Wine Acidity. Fermentation 2019, 5, 27. [Google Scholar] [CrossRef]
- Liu, S.; Laaksonen, O.; Kortesniemi, M.; Kalpio, M.; Yang, B. Chemical composition of bilberry wine fermented with non-Saccharomyces yeasts (Torulaspora delbrueckii and Schizosaccharomyces pombe) and Saccharomyces cerevisiae in pure, sequential and mixed fermentations. Food Chem. 2018, 266, 262–274. [Google Scholar] [CrossRef]
- Liu, S.; Laaksonen, O.; Yang, B. Volatile composition of bilberry wines fermented with non-Saccharomyces and Saccharomyces yeasts in pure, sequential and simultaneous inoculations. Food Microbiol. 2019, 80, 25–39. [Google Scholar] [CrossRef] [PubMed]
- Minnaar, P.; Jolly, N.; Paulsen, V.; Du Plessis, H.; Van Der Rijst, M. Schizosaccharomyces pombe and Saccharomyces cerevisiae yeasts in sequential fermentations: Effect on phenolic acids of fermented Kei-apple (Dovyalis caffra L.) juice. Int. J. Food Microbiol. 2017, 257, 232–237. [Google Scholar] [CrossRef] [PubMed]
- Satora, P.; Semik-Szczurak, D.; Tarko, T.; Bułdys, A. Influence of Selected Saccharomyces and Schizosaccharomyces Strains and Their Mixed Cultures on Chemical Composition of Apple Wines. J. Food Sci. 2018, 83, 424–431. [Google Scholar] [CrossRef] [PubMed]
- Miljić, U.D.; Puškaš, V.; Vučurović, V.; Muzalevski, A. Fermentation Characteristics and Aromatic Profile of Plum Wines Produced with Indigenous Microbiota and Pure Cultures of Selected Yeast. J. Food Sci. 2017, 47, 1139–1450. [Google Scholar] [CrossRef] [PubMed]
- Domizio, P.; Lencioni, L.; Calamai, L.; Portaro, L.; Bisson, L.F. Evaluation of the Yeast Schizosaccharomyces japonicus for Use in Wine Production. Am. J. Enol. Vitic. 2018, 69, 266–277. [Google Scholar] [CrossRef]
- Benito, Á; Calderón, F.; Benito, S. Combined Use of S. pombe and L. thermotolerans in Winemaking. Beneficial Effects Determined through the Study of Wines’ Analytical Characteristics. Molecules 2016, 21, 1744. [Google Scholar] [CrossRef] [PubMed]
- Vilela, A. Lachancea thermotolerans, the Non-Saccharomyces yeast that reduces the volatile acidity of wines. Fermentation 2018, 4, 56. [Google Scholar] [CrossRef]
- Porter, T.J.; Divol, B.; Setati, M.E. Lachancea yeast species: Origin, biochemical characteristics and oenological significance. Food Res. Int. 2019, 119, 378–389. [Google Scholar] [CrossRef] [PubMed]
- Benito, S. Combined Use of Lachancea thermotolerans and Schizosaccharomyces pombe in Winemaking: A Review. Microorganisms 2020, 8, 655. [Google Scholar] [CrossRef]
- Roca-Domènech, G.; Cordero-Otero, R.; Rozès, N.; Cléroux, M.; Pernet, A.; De Orduña, R.M. Metabolism of Schizosaccharomyces pombe under reduced osmotic stress conditions afforded by fed-batch alcoholic fermentation of white grape must. Food Res. Int. 2018, 113, 401–406. [Google Scholar] [CrossRef]
- Scomparin, A.; Bureik, M. A convenient new method for reproducible fed-batch fermentation of fission yeast Schizosaccharomyces pombe. Biotechnol. Lett. 2020, 42, 937–943. [Google Scholar] [CrossRef] [PubMed]
- Domizio, P.; Liu, Y.; Bisson, L.; Barile, D. Cell Wall Polysaccharides Released during the Alcoholic Fermentation by Schizosaccharomyces Pombe and S. Japonicus: Quantification and Characterization. Food Microbiol. 2017, 61, 136–149. [Google Scholar] [CrossRef] [PubMed]
- Benito, Á.; Calderón, F.; Benito, S. Mixed alcoholic fermentation of Schizosaccharomyces pombe and Lachancea thermotolerans and its influence on mannose-containing polysaccharides wine Composition. AMB Express 2019, 9, 17. [Google Scholar] [CrossRef]
- Benito, S. The Management of Compounds that Influence Human Health in Modern Winemaking from an HACCP Point of View. Fermentation 2019, 5, 33. [Google Scholar] [CrossRef]
- Vilela, A. Non-Saccharomyces Yeasts and Organic Wines Fermentation: Implications on Human Health. Fermentation 2020, 6, 54. [Google Scholar] [CrossRef]
- Bornet, A.; Teissedre, P.-L. Chitosan, chitin-glucan and chitin effects on minerals (iron, lead, cadmium) and organic (ochratoxin A) contaminants in wines. Eur. Food Res. Technol. 2008, 226, 681–689. [Google Scholar] [CrossRef]
- Bağder Elmaci, S.; Gülgör, G.; Tokatli, M.; Erten, H.; İşci, A.; Özçelik, F. Effectiveness of chitosan against wine-related microorganisms. Antonie v. Leeuwenh. 2015, 107, 675–686. [Google Scholar] [CrossRef]
- Escudero-Abarca, B.I.; Aguilar-Uscanga, M.G.; Hayward-Jones, P.M.; Mendoza, P.; Gómez-Rivas, L.; Ramírez, M. Selective antimicrobial action of chitosan against spoilage yeasts in mixed culture fermentations. J. Ind. Microbiol. Biotechnol. 2004, 31, 16–22. [Google Scholar] [CrossRef] [PubMed]
- Petrova, B.; Cartwright, Z.M.; Edwards, C.G. Effectiveness of chitosan preparations against Brettanomyces bruxellensis grown in culture media and red wines. OENO ONE 2016, 50, 49. [Google Scholar] [CrossRef]
- Valera, M.J.; Sainz, F.; Mas, A.; Torija, M.J. Effect of chitosan and SO2 on viability of Acetobacter strains in wine. Int. J. Food Microbiol. 2017, 246, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Picariello, L.; Rinaldi, A.; Blaiotta, G.; Moio, L.; Pirozzi, P.; Gambuti, A. Effectiveness of chitosan as an alternative to sulfites in red wine production. Eur. Food Res. Technol. 2020, 246, 1795–1804. [Google Scholar] [CrossRef]
- Kanter, J.P.; Benito, S.; Brezina, S.; Beisert, B.; Fritsch, S.; Patz, C.D.; Rauhut, D. The impact of hybrid yeasts on the aroma profile of cool climate Riesling wines. Food Chem. X 2020, 5, 100072. [Google Scholar] [CrossRef] [PubMed]
- Mecca, D.; Benito, S.; Beisert, B.; Brezina, S.; Fritsch, S.; Semmler, H.; Rauhut, D. Influence of Nutrient Supplementation on Torulaspora Delbrueckii Wine Fermentation Aroma. Fermentation 2020, 6, 35. [Google Scholar] [CrossRef]
- Bergmeyer, H.U. From the Preface to the 1st Edition. In Methods of Enzymatic Analysis; Elsevier BV: Amsterdam, The Netherlands, 1974. [Google Scholar] [CrossRef]
- De Mendiburu, F. Package “Agricolae: Statistical Procedures for Agricultural Research”. 2020. Available online: https://cran.r-project.org/web/packages/agricolae/agricolae.pdf (accessed on 5 October 2020).
- Du Plessis, H.; Hoff, J.; Hart, R.; Ndimba, B.; Jolly, N.; Du Toit, M. Characterisation of Non-Saccharomyces Yeasts Using Different Methodologies and Evaluation of their Compatibility with Malolactic Fermentation. S. Afr. J. Enol. Vitic. 2017, 38, 46–63. [Google Scholar] [CrossRef]
- Liu, S.; Laaksonen, O.; Yang, W.; Zhang, B.; Yang, B. Pyranoanthocyanins in bilberry (Vaccinium myrtillus L.) wines fermented with Schizosaccharomyces pombe and their evolution during aging. Food Chem. 2020, 305, 125438. [Google Scholar] [CrossRef]
- Colangelo, D.; Torchio, F.; De Faveri, D.M.; Lambri, M. The use of chitosan as alternative to bentonite for wine fining: Effects on heat-stability, proteins, organic acids, colour, and volatile compounds in an aromatic white wine. Food Chem. 2018, 264, 301–309. [Google Scholar] [CrossRef]
- Ruiz, J.; Kiene, F.; Belda, I.; Fracassetti, D.; Marquina, D.; Navascués, E.; Calderón, F.; Benito, A.; Rauhut, D.; Santos, A.; et al. Effects on varietal aromas during wine making: A review of the impact of varietal aromas on the flavor of wine. Appl. Microbiol. Biotechnol. 2019, 103, 7425–7450. [Google Scholar] [CrossRef] [PubMed]
- Song, Z.; Du, H.; Zhang, M.; Nie, Y.; Xu, Y. Schizosaccharomyces pombe Can Reduce Acetic Acid Produced by Baijiu Spontaneous Fermentation Microbiota. Microorganisms 2019, 7, 606. [Google Scholar] [CrossRef] [PubMed]
- Gamero, A.; Manzanares, P.; Querol, A.; Belloch, C. Monoterpene alcohols release and bioconversion by Saccharomyces species and hybrids. Int. J. Food Microbiol. 2011, 145, 92–97. [Google Scholar] [CrossRef]
- Stribny, J.; Gamero, A.; Pérez-Torrado, R.; Querol, A. Saccharomyces kudriavzevii and Saccharomyces uvarum differ from Saccharomyces cerevisiae during the production of aroma-active higher alcohols and acetate esters using their amino acidic precursors. Int. J. Food Microbiol. 2015, 205, 41–46. [Google Scholar] [CrossRef]
- Roudil, L.; Russo, P.; Berbegal, C.; Albertin, W.; Spano, G.; Capozzi, V. Non-Saccharomyces Commercial Starter Cultures: Scientific Trends, Recent Patents and Innovation in the Wine Sector. Recent Pat. Food Nutr. Agric. 2019, 10, 1. [Google Scholar] [CrossRef]
- Gamero, A.; Quintilla, R.; Groenewald, M.; Alkema, W.; Boekhout, T.; Hazelwood, L. High-throughput screening of a large collection of non-conventional yeasts reveals their potential for aroma formation in food fermentation. Food Microbiol. 2016, 60, 147–159. [Google Scholar] [CrossRef] [PubMed]
- Berbegal, C.; Fragasso, M.; Russo, P.; Bimbo, F.; Grieco, F.; Spano, G.; Capozzi, V. Climate changes and food quality: The potential of microbial activities as mitigating strategies in the wine sector. Fermentation 2019, 5, 85. [Google Scholar] [CrossRef]
- Filipe-Ribeiro, L.; Cosme, F.; Nunes, F.M. Reducing the negative sensory impact of volatile phenols in red wine with different chitosans: Effect of structure on efficiency. Food Chem. 2018, 242, 591–600. [Google Scholar] [CrossRef]

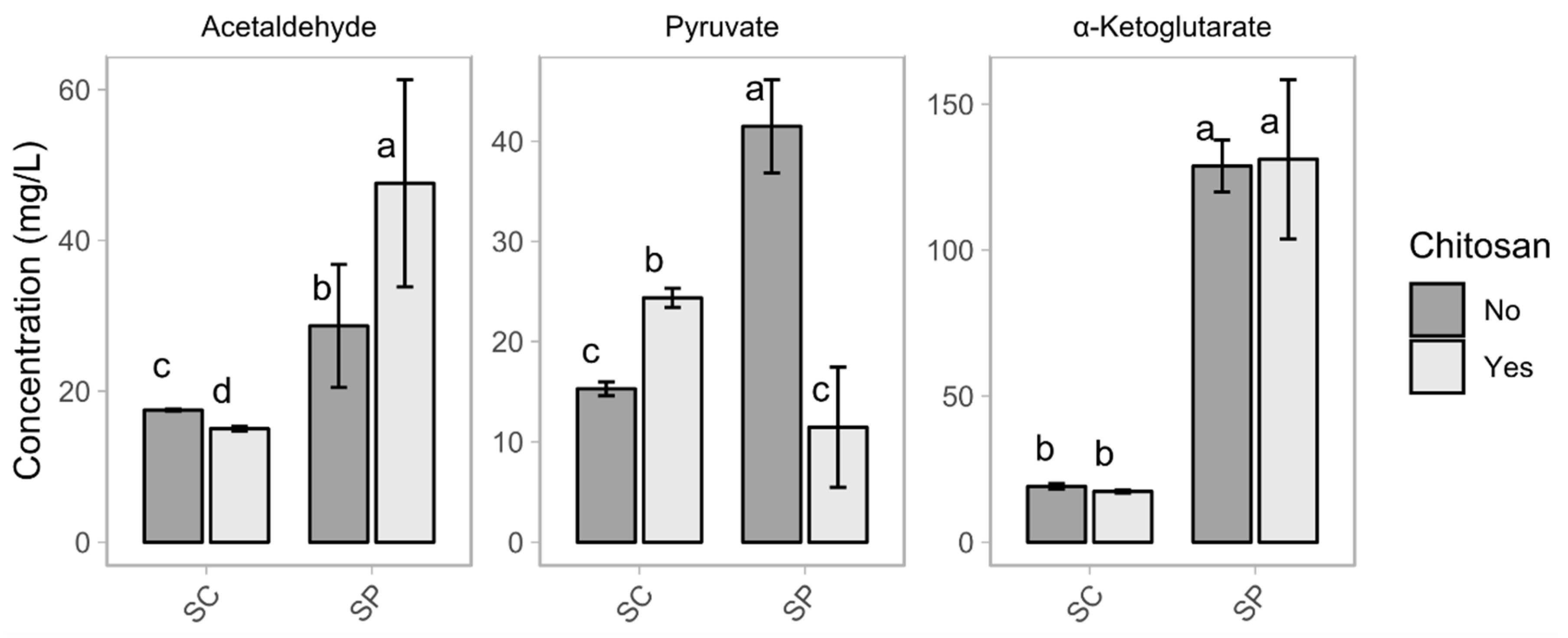

| Parameter | SC | SC_ch | SP | SP_ch |
|---|---|---|---|---|
| Ethanol [% v/v] | 11.26 ± 0.13 b | 11.08 ± 0.01 b | 11.52 ± 0.06 a | 11.52 ± 0.09 a |
| Residual Sugars [g L−1] | <2 | <2 | <2 | <2 |
| Glucose [g L−1] | n.q. | n.q. | n.q. | n.q. |
| Fructose [g L−1] | n.q. | n.q. | n.q. | n.q. |
| Glycerol [g L−1] | 7.20 ± 0.03 c | 8.48 ± 0.14 a | 7.74 ± 0.17 b | 8.54 ± 0.41 a |
| Tartaric acid [g L−1] | 1.24 ±0.02 a | 1.23 ±0.02 a | 1.10 ± 0.01 b | 1.28 ± 0.06 a |
| L-Malic acid [g L−1] | 4.75 ± 0.01 a | 4.65 ± 0.03 b | n.q. | n.q. |
| Shikimic acid [mg L−1] | 8.32 ± 0.09 b | 7.84 ± 0.19 b | 8.27 ± 0.05 b | 9.40 ± 0.63 a |
| L-Lactic acid [g L−1] | 0.18 ± 0.01 b | 0.20 ± 0.00 a,b | 0.21 ± 0.00 a | 0.20 ± 0.02 a,b |
| Acetic acid [g L−1] | 0.35 ± 0.00 a | 0.35 ± 0.01 a | 0.18 ± 0.02 c | 0.28 ± 0.02 b |
| Citric acid [g L−1] | 0.16 ± 0.01 a | 0.16 ± 0.01 a | 0.17 ± 0.02 a | 0.16 ± 0.03 a |
| pH | 3.26 ± 0.02 b | 3.22 ± 0.02 b | 3.89 ± 0.01 a | 3.89 ± 0.02 a |
| Volatile Compound | SC | SC_ch | SP | SP_ch |
|---|---|---|---|---|
| Acetate Esters | ||||
| Ethyl Acetate [mg L−1] | 169.8 ± 9.6 b | 187.0 ± 1.2 a | 163.6 ± 5.3 b | 117.2 ± 5.5 c |
| Isoamyl acetate [µg L−1] | 7813.4 ± 374.3 b | 11,301.5 ± 1197.0 a | 6350.6 ± 286.3 b | 3217.3 ± 1643.5 c |
| 2-Methyl butyl acetate [µg L−1] | 261.1 ± 10.7 a | 280.3 ± 9.1 a | 254.0 ± 9.6 a | 182.5 ± 43.4 b |
| Hexyl acetate [µg L−1] | 88.1 ± 4.9 a | 98.5 ± 10.4 a | 31.0 ± 2.4 b | n.q. |
| 2-Phenyl ethyl acetate [µg L−1] | 330.5 ± 4.0 b | 453.4 ± 36.4 a | 123.1 ± 11.6 c | 66.6 ± 30.3 d |
| Ʃ Acetates [µg L−1] | 178,293.1 | 199,133.7 | 170,358.7 | 15,186.4 |
| Ethyl Esters | ||||
| Ethyl propionate [µg L−1] | 170.1 ± 4.5 b | 187.4 ± 4.5 a | 93.6 ± 12.1 d | 128.9 ± 5.1 c |
| Ethyl butyrate [µg L−1] | 552.6 ± 12.1 a | 562.1 ± 53.7 a | 657.8 ± 16.6 a | 377.7 ± 102.5 b |
| Ethyl isobutyrate [µg L−1] | 39.7 ± 3.4 a | 41.4 ± 3.2 a | 20.3 ± 1.6 c | 30.1 ± 5.9 b |
| Ethyl hexanoate [µg L−1] | 907.1 ± 16.0 a,b | 1006.2 ± 103.1 a | 777.0 ± 49.7 b,c | 637.7 ± 64.7 c |
| Ethyl octanoate [µg L−1] | 1354.4 ± 26.6 a | 1293.8 ± 48.9 a | 917.6 ± 68.4 b | 749.2 ± 196.5 b |
| Ethyl decanoate [µg L−1] | 464.1 ± 4.7 a | 430.6 ± 28.6 a | 281.3 ± 26.0 b | 162.8 ± 59.5 c |
| Ʃ Ethyl esters [µg L−1] | 3488.0 | 3521.5 | 2747.6 | 2086.4 |
| Higher Alcohols | ||||
| Isobutanol [mg L−1] | 28.8 ± 0.5 b | 32.0 ± 1.3 a | 20.6 ± 1.2 d | 24.0 ± 2.5 c |
| Isoamyl alcohol [mg L−1] | 243.7 ± 7.8 a | 255.1 ± 7.4 a | 126.2 ± 5,7 b | 93.3 ± 7.5 c |
| Active amyl alcohol [mg L−1] | 22.4 ± 0.2 a,b | 26.3 ± 0.4 a | 19.3 ± 2.2 b | 21.6 ± 2.3 b |
| Hexanol [µg L−1] | n.q. | n.q. | n.q. | n.q. |
| 2-Phenyl−ethanol [mg L−1] | 18.6 ± 0.9 b | 22.0 ± 0.5 a | 7.9 ± 0.4 c | 6.6 ± 0.4 d |
| Ʃ Higher alcohols [mg L−1] | 313.5 | 335.4 | 174 | 145.5 |
| Fatty Acids | ||||
| Isovaleric acid [µg L−1] | 2526.2 ± 103.2 a | 2620.7 ± 53.0 a | 2040.2 ± 7.4 b | 1768.6 ± 18.3 c |
| Hexanoic acid [mg L−1] | 12.9 ± 0.7 a | 12.9 ± 0.4 a | 12.1 ± 0.6 a | 10.6 ± 0.5 b |
| Octanoic acid [mg L−1] | 10.8 ± 0.5 a | 11.0 ± 0.5 a | 7.9 ± 0.6 b | 6.3 ± 1.0 b |
| Decanoic acid [mg L−1] | 3.1 ± 0.1 a | 3.0 ± 0.2 a | 2.3 ± 0.2 b | 1.8 ± 0.4 c |
| Ʃ Fatty acids [mg L−1] | 29.3 | 29.5 | 24.3 | 20.5 |
| Monoterpenoids and C13-norisoprenoids | ||||
| Linalool oxide-1 [µg L−1] | 5.4 ± 0.5 b | 5.9 ± 0.6 b | 6.0 ± 0.3 b | 7.2 ± 0.2 a |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Scansani, S.; Rauhut, D.; Brezina, S.; Semmler, H.; Benito, S. The Impact of Chitosan on the Chemical Composition of Wines Fermented with Schizosaccharomyces pombe and Saccharomyces cerevisiae. Foods 2020, 9, 1423. https://doi.org/10.3390/foods9101423
Scansani S, Rauhut D, Brezina S, Semmler H, Benito S. The Impact of Chitosan on the Chemical Composition of Wines Fermented with Schizosaccharomyces pombe and Saccharomyces cerevisiae. Foods. 2020; 9(10):1423. https://doi.org/10.3390/foods9101423
Chicago/Turabian StyleScansani, Stefano, Doris Rauhut, Silvia Brezina, Heike Semmler, and Santiago Benito. 2020. "The Impact of Chitosan on the Chemical Composition of Wines Fermented with Schizosaccharomyces pombe and Saccharomyces cerevisiae" Foods 9, no. 10: 1423. https://doi.org/10.3390/foods9101423
APA StyleScansani, S., Rauhut, D., Brezina, S., Semmler, H., & Benito, S. (2020). The Impact of Chitosan on the Chemical Composition of Wines Fermented with Schizosaccharomyces pombe and Saccharomyces cerevisiae. Foods, 9(10), 1423. https://doi.org/10.3390/foods9101423





