Effect of Carrier Agents on the Physicochemical and Technofunctional Properties and Antioxidant Capacity of Freeze-Dried Pomegranate Juice (Punica granatum) Powder
Abstract
1. Introduction
2. Materials and Methods
2.1. Raw Material and Sample Preparation
2.2. Freeze-Drying Procedure
2.3. Yield, Water Activity and Physicochemical Attributes of PJP
2.3.1. Powder Yield Determination
2.3.2. Determination of Water Activity and Moisture Content
2.3.3. Colour Measurement
2.3.4. Determination of Total Soluble Solids (TSSs), Titratable Acidity (TA) and pH
2.4. Technofunctional Characterisation of PJP
2.4.1. Solubility
2.4.2. Hygroscopicity
2.4.3. Bulk Density
2.4.4. Water- and Oil-Holding Capacity Determination
2.4.5. Particle Size Distribution
2.4.6. Microstructure
2.5. Phenolic Contents and Antioxidant Capacity
2.5.1. Determination of Total Phenolic Content (TPC)
2.5.2. Total Anthocyanin Content
2.5.3. Radical-Scavenging Activity (RSA)
2.5.4. Ferric-Ion Reducing Antioxidant Power (FRAP)
2.6. Statistical Analysis
3. Results and Discussion
3.1. Quality Attributes of Pomegranate Juice
3.2. Yield, Moisture Content and Water Activity
3.3. Colour Attributes
3.4. Total Soluble Solids (TSSs) and Titratable Acidity (TA)
3.5. Technofunctional Properties
3.5.1. Solubility
3.5.2. Hygroscopicity
3.5.3. Bulk Density
3.5.4. Water- and Oil-Holding Capacity
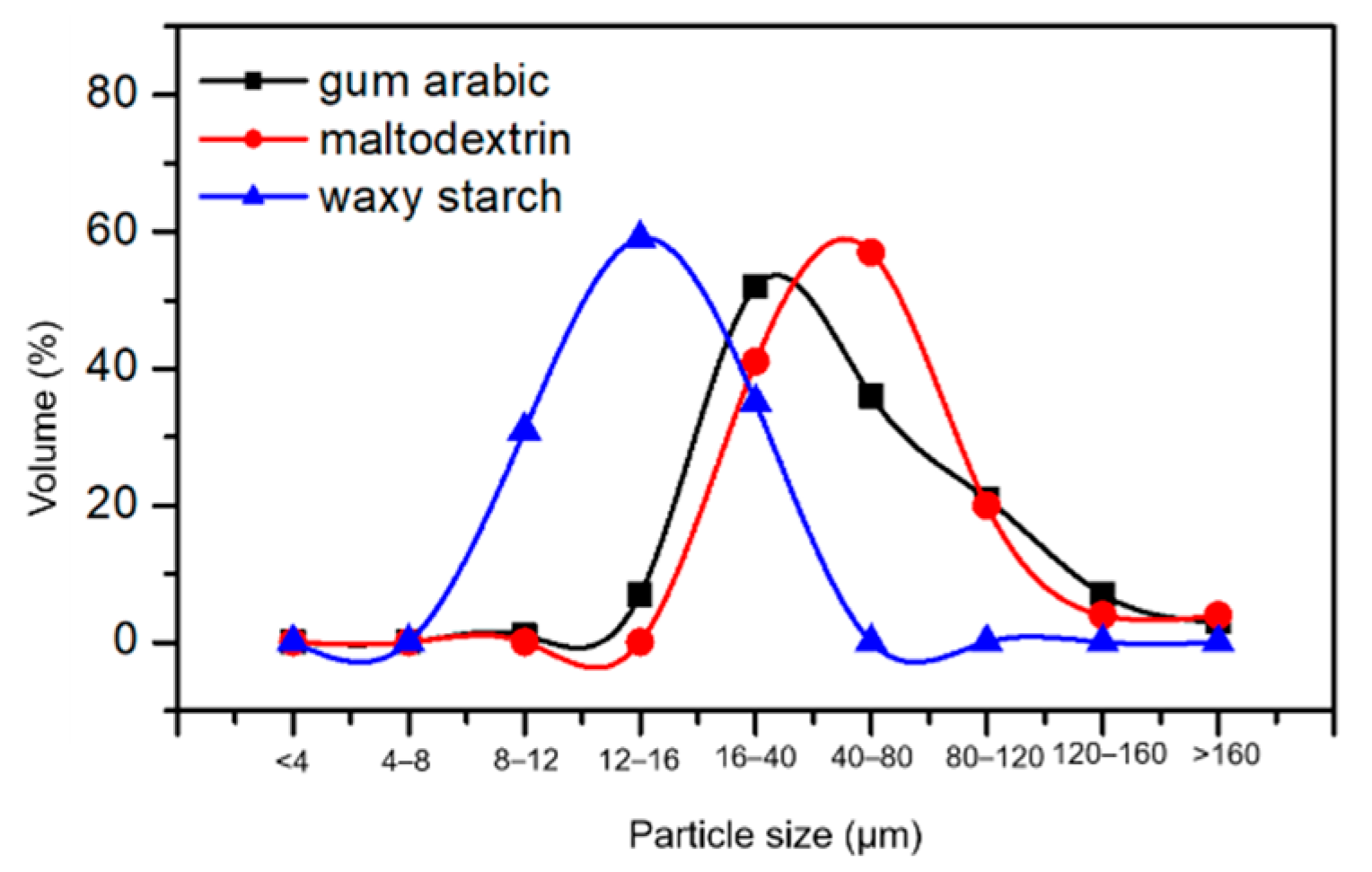
3.5.5. Particle Size Distribution
3.5.6. Microstructure
3.6. Total Phenolic and Anthocyanin Contents
3.7. DPPH Radical Scavenging Activity and FRAP Activity
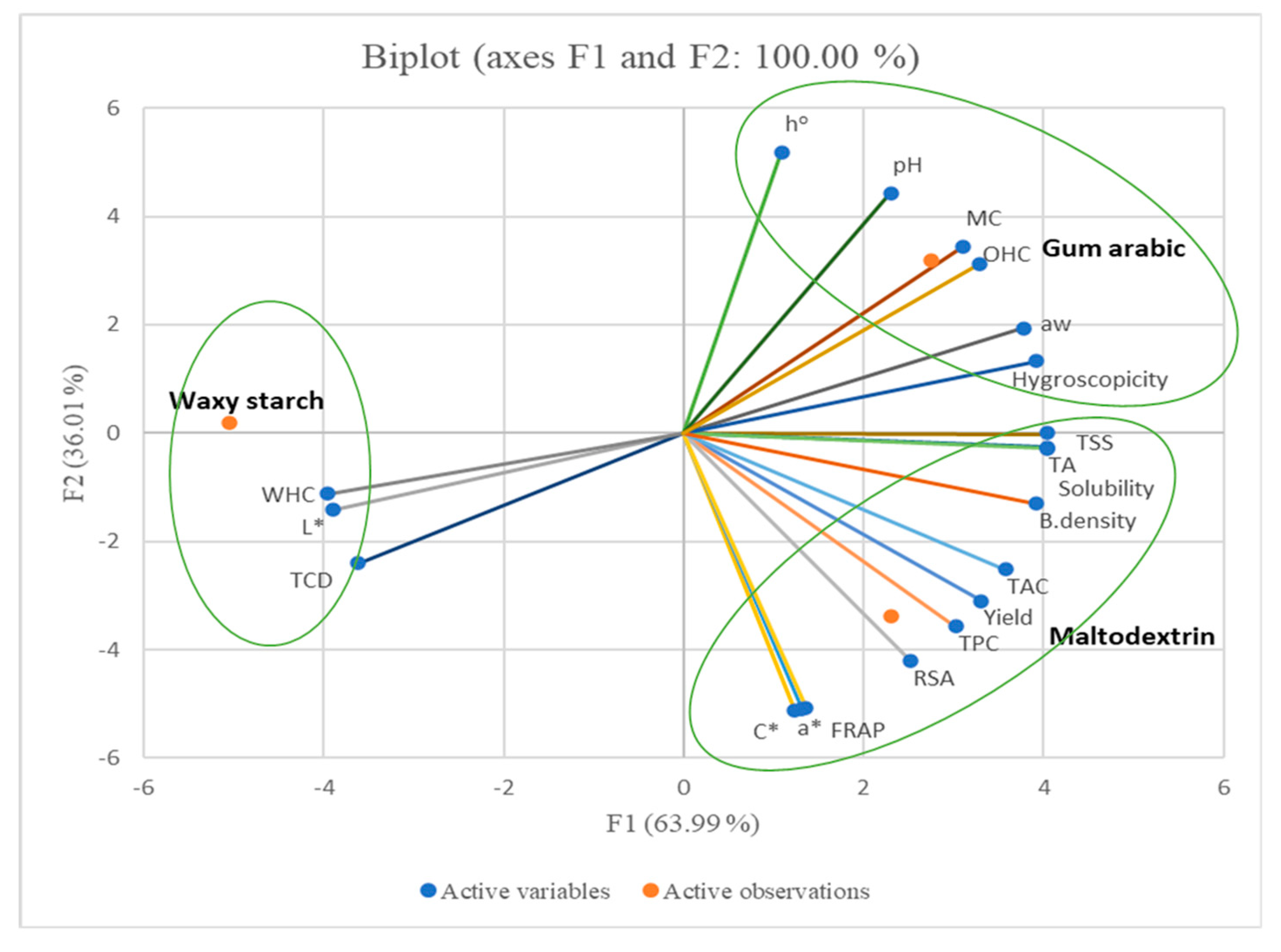
3.8. Principal Component Analysis
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| PJP | pomegranate juice powder |
| MC | moisture content |
| L* | lightness |
| a* | redness |
| C* | chroma |
| h° | hue angle |
| TCD | total colour difference |
| RSA | radical scavenging activity |
| FRAP | ferric reducing antioxidant power |
| TPC | total phenolic content |
| TAC | total anthocyanin content |
| TSS | total soluble solids |
| TA | titratable acidity |
| aw | water activity |
| WHC | water holding capacity |
| OHC | oil holding capacity |
| SEM | scanning electron microscopy |
| w.b. | wet basis |
| RH | relative humidity |
References
- Fawole, O.A.; Opara, U.L. Effects of maturity status on biochemical concentration, polyphenol composition and antioxidant capacity of pomegranate fruit arils (cv. Bhagwa). S. Afr. J. Bot. 2013, 85, 23–31. [Google Scholar] [CrossRef]
- Mphahlele, R.R.; Caleb, O.J.; Fawole, O.A.; Opara, U.L. Effects of different maturity stages and growing locations on changes in chemical, biochemical and aroma volatile composition of ‘Wonderful’ pomegranate juice. J. Sci. Food Agric. 2016, 96, 1002–1009. [Google Scholar] [CrossRef] [PubMed]
- Gil, M.I.; Tomas-Barberan, F.A.; Hess-Pierse, B.; Holcroft, D.M.; Kader, A.A. Antioxidant activity of pomegranate juice and its relationship with phenolic composition and processing. J. Agric. Food Chem. 2000, 48, 4581–4589. [Google Scholar] [CrossRef] [PubMed]
- Pomegranate Association of South Africa (Pomasa) Pomegranate Industry Statistics. Paarl, South Africa, 2019. Available online: https://www.sapomegranate.co.za/statistics-and-information/pomegranate-industry-overview/ (accessed on 18 September 2020).
- Seeram, N.P.; Aviram, M.; Zhang, Y.; Henning, S.M.; Feng, L.; Dreher, M.; Heber, D. Comparison of antioxidant potency of commonly consumed polyphenol-rich beverages in the United States. J. Agric. Food Chem. 2008, 56, 1415–1422. [Google Scholar] [CrossRef] [PubMed]
- Cao, G.; Sofic, E.; Prior, R. Antioxidant capacity of tea and common vegetables. J. Agric. Food Chem. 1996, 44, 3426–3431. [Google Scholar] [CrossRef]
- Opara, U.L.; Al-Ani, M.R.; Al-Shuaibi, Y.S. Physico-chemical properties, vitamin C content and antimicrobial properties of pomegranate fruit (Punica granatum L.). Food Bioprocess Technol. 2009, 2, 315–321. [Google Scholar] [CrossRef]
- Malik, A.; Afaq, F.; Sarfaraz, S.; Adhami, V.M.; Syed, D.N.; Mukhtar, H. Pomegranate fruit juice for chemoprevention and chemotherapy of prostate cancer. Proc. Natl. Acad. Sci. USA 2005, 102, 14813–14818. [Google Scholar] [CrossRef] [PubMed]
- Caleb, O.J.; Opara, U.L.; Witthuhn, C.R. Modified atmosphere packaging of pomegranate fruit and arils: A review. Food Bioprocess Technol. 2012, 5, 15–30. [Google Scholar] [CrossRef]
- Kulkami, A.P.; Aradhya, S.M. Chemical changes and antioxidant activity in pomegranate arils during fruit development. Food Chem. 2005, 93, 319–324. [Google Scholar]
- Longtin, R. The pomegranate: Nature’s power fruit. J. Natl. Cancer Inst. 2003, 95, 346–348. [Google Scholar] [CrossRef]
- Różyło, R. Recent trends in methods used to obtain natural food colorants by freeze-drying. Trends Food Sci. Technol. 2020, 102, 39–50. [Google Scholar] [CrossRef]
- Sagar, V.R.; Kumar, P.S. Recent advances in drying and dehydration of fruits and vegetables: A review. J. Food Sci. Technol. 2010, 47, 15–26. [Google Scholar] [CrossRef] [PubMed]
- Du, J.; Ge, Z.Z.; Xu, Z.; Zou, B.; Zhang, Y.; Li, C.M. Comparison of the efficiency of five different drying carriers on the spray drying of persimmon pulp powders. Dry. Technol. 2014, 32, 1157–1166. [Google Scholar] [CrossRef]
- Patel, K.C.; Chen, X.D. Drying of aqueous lactose solutions in a single stream dryer. Food Bioprod. Process. 2008, 86, 185–197. [Google Scholar] [CrossRef]
- Sablani, S.S.; Shrestha, A.K.; Bhandari, B.R. A new method of producing date powder granules: Physicochemical characteristics of powder. J. Food Eng. 2008, 87, 416–421. [Google Scholar] [CrossRef]
- Jayaraman, K.S.; Das Gupta, D.K. Dehydration of fruits and vegetables-recent developments in principles and techniques. Dry. Technol. 1992, 10, 1–50. [Google Scholar] [CrossRef]
- Fazaeli, M.; Emam-Djomeh, Z.; Ashtari, A.K.; Omid, M. Effect of spray drying conditions and feed composition on the physical properties of black mulberry juice powder. Food Bioprod. Process. 2012, 90, 667–675. [Google Scholar] [CrossRef]
- Seerangurayar, T.; Manickavasagan, A.; Al-Ismaili, A.M.; Al-Mulla, Y.A. Effect of carrier agents on physicochemical properties of foam-mat freeze-dried date powder. Dry. Technol. 2018, 36, 1292–1303. [Google Scholar] [CrossRef]
- Dolinsky, A.; Maletskaya, K.; Snezhkin, Y. Fruit and vegetable powders production technology on the bases of spray and convective drying methods. Dry. Technol. 2000, 18, 747–758. [Google Scholar] [CrossRef]
- Adhikari, B.; Howes, T.; Bhandari, B.R.; Troung, V. Effect of addition of maltodextrin on drying kinetics and stickiness of sugar and acid-rich foods during convective drying: Experiments and modelling. J. Food Eng. 2004, 62, 53–68. [Google Scholar] [CrossRef]
- Osorio, C.; Forero, D.P.; Carriazo, J.G. Characterisation and performance assessment of guava (Psidium guajava L.) microencapsulates obtained by spray-drying. Food Res. Int. 2011, 44, 1174–1181. [Google Scholar] [CrossRef]
- Wang, W.; Zhou, W. Characterization of spray-dried soy sauce powders using maltodextrin as carrier. J. Food Eng. 2012, 109, 399–405. [Google Scholar] [CrossRef]
- Cano-Chauca, M.; Stringheta, P.C.; Ramos, A.M.; Cal-Vidal, J. Effect of the carriers on the microstructure of mango powder obtained by spray drying and its functional characterization. Innov. Food Sci. Emerg. Technol. 2005, 5, 420–428. [Google Scholar] [CrossRef]
- Silva, M.A.; Sobral, P.J.A.; Kieckbusch, T.G. State diagrams of freeze-dried camu-camu (Myrciaria dubia (HBK) Mc Vaugh) pulp with and without maltodextrin addition. J. Food Eng. 2006, 77, 426–432. [Google Scholar] [CrossRef]
- Phisut, N. Spray drying technique of fruit juice powder: Some factors influencing the properties of product. Int. Food Res. J. 2012, 4, 1297–1306. [Google Scholar]
- Etzbach, L.; Meinert, M.; Faber, T.; Klein, C.; Schieber, A.; Weber, F. Effects of carrier agents on powder properties, stability of carotenoids, and encapsulation efficiency of goldenberry (Physalis peruviana L.) powder produced by co-current spray drying. Curr. Res. Food Sci. 2020, 3, 73–81. [Google Scholar] [CrossRef]
- Yousefi, S.; Emam-Djomeh, Z.; Mousavi, S.M. Effect of carrier type and spray drying on the physicochemical properties of powdered and reconstituted pomegranate juice (Punica granatum L.). J. Food Sci. Technol. 2011, 48, 677–684. [Google Scholar] [CrossRef]
- Muzaffar, K.; Kumar, P. Parameter optimization for spray drying of tamarind pulp using response surface methodology. Powder Technol. 2015, 279, 179–184. [Google Scholar] [CrossRef]
- Pathare, P.B.; Opara, U.L.; Al-Said, F.A.J. Colour measurement and analysis in fresh and processed foods: A review. Food Bioproc. Technol. 2013, 6, 36–60. [Google Scholar] [CrossRef]
- Ferrari, C.C.; Germer, S.P.M.; Aguirre, J.M. Effects of spray drying conditions on the physicochemical properties of blackberry powder. Dry. Technol. 2011, 30, 154–163. [Google Scholar] [CrossRef]
- Largo Avila, E.; Cortes Rodríguez, M.; Velásquez, C.; José, H. Influence of maltodextrin and spray drying process conditions on sugarcane juice powder quality. Rev. Fac. Nac. Agron. Medellin 2015, 68, 7509–7520. [Google Scholar] [CrossRef]
- Goula, A.M.; Adamopoulos, K.G. Effect of maltodextrin addition during spray drying of tomato pulp in dehumidified air: II. Powder properties. Dry. Technol. 2008, 26, 726–737. [Google Scholar] [CrossRef]
- Jalal, H.; Pal, M.A.; Ahmad, S.R.; Rather, M.; Andrabi, M.; Hamdani, S. Physico-chemical and functional properties of pomegranate peel and seed powder. J. Pharm. Innov. 2018, 7, 1127–1131. [Google Scholar]
- Ferrari, C.C.; Germer, S.P.M.; Alvim, I.D.; Vissotto, F.Z.; de Aguirre, J.M. Influence of carrier agents on the physicochemical properties of blackberry powder produced by spray drying. J. Food Sci. Technol. 2012, 47, 1237–1245. [Google Scholar] [CrossRef]
- Wrolstad, R.E. Colour and Pigment Analyses in Fruit Products; Agricultural Experiment Station, Oregon State University Station: Corvallis, OR, USA, 1993; Volume 624. [Google Scholar]
- Benzie, I.F.F.; Strain, J.J. The ferric reducing ability of plasma (FRAP) as a measure of “antioxidant power”. The FRAP assay. Anal. Biochem. 1996, 239, 70–76. [Google Scholar] [CrossRef]
- Fawole, O.A.; Opara, U.L. Physico-mechanical, phyto-chemical, and free radical scavenging properties and volatile compounds in eight pomegranate cultivars and classification by principal component and cluster analyses. Br. Food J. 2013, 116, 544. [Google Scholar] [CrossRef]
- Fawole, O.A.; Opara, U.L. Changes in physical properties, chemical and elemental composition and antioxidant capacity of pomegranate (cv. Ruby) fruit at five maturity stages. Sci. Hortic. 2013, 150, 37–46. [Google Scholar] [CrossRef]
- Quek, S.Y.; Chok, N.K.; Swedlund, P. The physicochemical properties of spray-dried watermelon powder. Chem. Eng. Process. 2007, 46, 386–392. [Google Scholar] [CrossRef]
- Seeram, N.P.; Adams, L.S.; Henning, S.M.; Niu, Y.; Zhang, Y.; Nair, M.G.; Heber, D. In vitro anti-proliferative, apoptotic and antioxidant activities of punicalagin, ellagic acid and a total pomegranate tannin extract are enhanced in combination with other polyphenols as found in pomegranate juice. J. Nutr. Biochem. 2005, 16, 360–367. [Google Scholar] [CrossRef]
- Bourdoux, S.; Li, D.; Rajkovic, A.; Devlieghere, F.; Uyttendaele, M. Performance of drying technologies to ensure microbial safety of dried fruits and vegetables. Compr. Rev. Food Sci. 2016, 15, 1056–1066. [Google Scholar] [CrossRef]
- Laroche, C.; Fine, F.; Gervais, P. Water activity affects heat resistance of microorganisms in food powders. Int. J. Food Microbiol. 2005, 97, 307–315. [Google Scholar] [CrossRef] [PubMed]
- Daza, L.D.; Fujita, A.; Fávaro-Trindade, C.S.; Rodrigues-Ract, J.N.; Granato, D.; Genovese, M.I. Effect of spray drying conditions on the physical properties of Cagaita (Eugenia dysenterica DC.) fruit extracts. Food Bioprod. Process. 2016, 97, 20–29. [Google Scholar] [CrossRef]
- Mosquera, L.H.; Moraga, G.; Martınez-Navarrete, N. Critical water activity and critical water content of freeze-dried strawberry powder as affected by maltodextrin and arabic gum. Food Res. Int. 2011, 47, 201–206. [Google Scholar] [CrossRef]
- da Silva Delabona, P.; Pirota, R.D.P.B.; Codima, C.A.; Tremacoldi, C.R.; Rodrigues, A.; Farinas, C.S. Effect of initial moisture content on two Amazon rainforest Aspergillus strains cultivated on agro-industrial residues: Biomass-degrading enzymes production and characterization. Ind. Crop. Prod. 2013, 42, 236–242. [Google Scholar] [CrossRef]
- Hammami, C.; René, F. Determination of freeze-drying process variables for strawberries. J. Food Eng. 1997, 32, 133–154. [Google Scholar] [CrossRef]
- Goula, A.M.; Adamopoulos, K.G. A new technique for spray drying orange juice concentrate. Innov. Food Sci. Emerg. Technol. 2010, 11, 342–351. [Google Scholar] [CrossRef]
- Comunian, T.A.; Monterrey-Quintero, E.S.; Thomazini, M.; Balieiro, J.C.D.C.; Piccone, P.; Pittia, P.; Favaro-Trindade, C.S. Assessment of production efficiency, physicochemical properties and storage stability of spray-dried chlorophyllide, a natural food colourant, using gum Arabic, maltodextrin and soy protein isolate-based carrier systems. Int. J. Food Sci. Technol. 2011, 46, 1259–1265. [Google Scholar] [CrossRef]
- Kha, T.C.; Nguyen, M.H.; Roach, P.D. Effects of spray drying conditions on the physicochemical and antioxidant properties of the Gac (Momordica cochinchinensis) fruit aril powder. J. Food Eng. 2010, 98, 385–392. [Google Scholar] [CrossRef]
- Horuz, E.; Altan, A.; Maskan, M. Spray drying and process optimization of unclarified pomegranate (Punica granatum) juice. Dry. Technol. 2012, 30, 787–798. [Google Scholar] [CrossRef]
- Rahman, S.; Lamb, J. Air drying behaviour of fresh and osmotically dehydrated pineapple. J. Food Process. Eng. 1991, 14, 163–171. [Google Scholar] [CrossRef]
- Karatas, S.; Esin, A.A. A laboratory scraped surface drying chamber for spray drying of tomato paste. Lebensm. Wiss. Technol. 1990, 23, 354–357. [Google Scholar]
- Bhandari, B.R.; Datta, N.; Crooks, R.; Howes, T.; Rigby, S. A Semi-empirical Approach to optimize the quantity of drying aids required to spray dry sugar rich foods. Dry. Technol. 1997, 15, 2509–2525. [Google Scholar] [CrossRef]
- Shrestha, A.K.; Ua-Arak, T.; Adhikari, B.P.; Howes, T.; Bhandari, B.R. Glass transition behavior of spray dried orange juice powder measured by differential scanning calorimetry (DSC) and thermal mechanical compression test (TMCT). Int. J. Food Prop. 2007, 10, 661–673. [Google Scholar] [CrossRef]
- Abadio, F.D.B.; Domingues, A.M.; Borges, S.V.; Oliveira, V.M. Physical properties of powdered pineapple (Ananas comosus) juice- Effect of malt dextrin concentration and atomization speed. J. Food Eng. 2004, 64, 285–287. [Google Scholar] [CrossRef]
- De Oliveira, M.A.; Maia, G.A.; De Figueiredo, R.W.; De Souza, A.C.R.; De Brito, E.S.; De Azeredo, H.M.C. Addition of cashew tree gum to maltodextrin-based carriers for spray drying of cashew apple juice. J. Food Sci. Technol. 2009, 44, 641–645. [Google Scholar] [CrossRef]
- Mishra, S.; Rai, T. Morphology and functional properties of corn, potato and tapioca starches. Food Hydrocoll. 2006, 20, 557–566. [Google Scholar] [CrossRef]
- Tonon, R.V.; Baroni, A.F.; Brabet, C.; Gibert, O.; Pallet, D.; Hubinger, M.D. Water sorption and glass transition temperature of spray dried açai (Euterpe oleracea Mart.) juice. J. Food Eng. 2009, 94, 215–221. [Google Scholar] [CrossRef]
- Gabas, A.L.; Telis, V.R.N.; Sobral, P.J.A.; Telis-Romero, J. Effect of maltodextrin and arabic gum in water vapour sorption thermodynamic properties of vacuum dried pineapple pulp powder. J. Food Eng. 2007, 82, 246–252. [Google Scholar] [CrossRef]
- Ahmed, M.; Akter, M.S.; Eun, J.B. Impact of alpha-amylase and maltodextrin on physicochemical, functional and antioxidant capacity of spray dried purple sweet potato flour. J. Sci. Food Agric. 2010, 90, 494–502. [Google Scholar] [CrossRef]
- Barbosa-Canovas, G.V.; Juliano, P. Physical and chemical properties of food powders. In Encapsulated and Powdered Foods; Onwulata, C., Ed.; Taylor & Francis: Boca Raton, FL, USA, 2005; pp. 39–71. [Google Scholar]
- Chegini, G.R.; Ghobadian, B. Effect of spray-drying conditions on physical properties of orange juice powder. Dry. Technol. 2005, 23, 656–668. [Google Scholar] [CrossRef]
- Zhang, L.H.; Li, S.F. Effects of micronization on properties of Chaenomeles sinensis (Thouin) Koehne fruit powder. Innov. Food Sci. Emerg. Technol. 2009, 10, 633–637. [Google Scholar] [CrossRef]
- Hong, J.; Zhang, S.Y. Effect of ultra-fine pulverization by wet processing on particle structure and physical properties of soybean dietary fiber. J. China Agric. Univ. 2005, 10, 90–94. (In Chinese) [Google Scholar]
- Chau, C.F.; Wang, Y.T.; Wen, Y.L. Different micronization methods significantly improve the functionality of carrot insoluble fibre. Food Chem. 2007, 100, 1402–1408. [Google Scholar] [CrossRef]
- Tonon, R.V.; Freitas, S.S.; Hubinger, M.D. Spray drying of açai (Euterpe oleraceae Mart.) juice: Effect of inlet air temperature and type of carrier agent. J. Food Process. Preserv. 2011, 35, 691–700. [Google Scholar] [CrossRef]
- Syamaladevi, R.M.; Insan, S.K.; Dhawan, S.; Andrews, P.; Sablani, S.S. Physicochemical properties of encapsulated red raspberry (Rubus idaeus) powder: Influence of high-pressure homogenization. Dry. Technol. 2012, 30, 484–493. [Google Scholar] [CrossRef]
- Kurozawa, L.E.; Morassi, A.G.; Vanzo, A.A.; Park, K.J.; Hubinger, M.D. Influence of spray drying conditions on physicochemical properties of chicken meat powder. Dry. Technol. 2009, 27, 1248–1257. [Google Scholar] [CrossRef]
- Leonel, M. Ana´lise da forma e tamanho de graˆnulos de amidos de diferentes fontes botânicas. Ciênc. Tecnol. 2007, 23, 579–588. [Google Scholar] [CrossRef]
- Loksuwan, J. Characteristics of microencapsulated b-carotene formed by spray drying with modified tapioca starch, native tapioca starch and maltodextrin. Food Hydrocoll. 2007, 21, 928–935. [Google Scholar] [CrossRef]
- Tonon, R.V.; Brabet, C.; Hubinger, M.D. Anthocyanin stability and antioxidant activity of spray dried ac¸ai (Euterpe oleracea Mart.) juice powder produced with different carrier agents. Int. Food Res. J. 2010, 43, 907–914. [Google Scholar] [CrossRef]
- Lim, K.; Ma, M.; Dolan, K.D. Effects of spray drying on antioxidant capacity and anthocyanidin content of blueberry by-products. J. Food Sci. 2011, 76, 156–164. [Google Scholar] [CrossRef]
- Mishra, P.; Mishra, S.; Mahanta, C.L. Effect of maltodextrin concentration and inlet temperature during spray drying on physicochemical and antioxidant properties of amla (Emblica officinalis) juice powder. Food Bioprod. Process. 2014, 92, 252–258. [Google Scholar] [CrossRef]



| Quality Attributes | Values (Means ± SE) |
|---|---|
| TSS (°Brix) | 15.3 ± 0.07 |
| TA (% citric acid) | 1.35 ± 0.01 |
| pH | 3.10 ± 0.01 |
| TPC (mg GAE/100 mL) | 22.6 ± 2.82 |
| TAC (mg C3gE/100 mL) | 7.4 ± 1.95 |
| L* | 23.0 ± 2.21 |
| a* | 15.4 ± 1.37 |
| C* | 18.6 ± 1.44 |
| h° | 34.5 ± 0.94 |
| Carrier | Yield % | MC % | aw | L* | a* | C* | h° | TCD |
|---|---|---|---|---|---|---|---|---|
| Maltodextrin | 46.6 ± 0.04 a | 0.7 ± 0.02 b | 0.31 ± 0.00 b | 69.0 ± 1.42 b | 29.3 ± 0.49 a | 29.3 ± 0.48 a | 0.6 ± 0.19 c | 49.2 ± 1.39 b |
| Gum arabic | 40.6 ± 0.12 b | 1.8 ± 0.02 a | 0.49 ± 0.01 a | 64.6 ± 0.39 c | 16.3 ± 0.24 c | 16.7 ± 0.23 c | 11.9 ± 0.37 a | 42.2 ± 0.40 c |
| Waxy starch | 35.4 ± 0.30 c | 0.2 ± 0.02 c | 0.05 ± 0.00 c | 78.7 ± 1.20 a | 18.6 ± 0.29 b | 18.6 ± 0.29 b | 4.0 ± 0.31 b | 56.5 ± 1.19 a |
| Carrier | TSSs (°Brix) | TA (% Citric Acid) |
|---|---|---|
| Maltodextrin | 10.3 ± 0.17 a | 0.24 ± 0.04 a |
| Gum arabic | 10.4 ± 0.21 a | 0.24 ± 0.02 a |
| Waxy starch | 8.6 ± 0.20 b | 0.18 ± 0.01 b |
| Carrier | Solubility % | Hygroscopicity % | Bulk Density (g cm−3) | WHC (g/g) | OHC (g/g) |
|---|---|---|---|---|---|
| Maltodextrin | 96.5 ± 0.09 a | 10.2 ± 0.04 b | 0.77 ± 0.01 a | 0.67 ± 0.03 b | 1.64 ± 0.01 b |
| Gum arabic | 96.1 ± 0.46 a | 12.6 ± 0.01 a | 0.74 ± 0.02 a | 0.25 ± 0.01 c | 1.96 ± 0.01 a |
| Waxy starch | 35.4 ± 0.09 b | 4.7 ± 0.07 c | 0.64 ± 0.02 b | 1.84 ± 0.01 a | 1.45 ± 0.01 c |
| Loadings | F1 | F2 |
|---|---|---|
| L* | −0.965 | −0.263 |
| a* | 0.306 | −0.952 |
| C* | 0.324 | −0.946 |
| h° | 0.270 | 0.963 |
| TCD | −0.895 | −0.446 |
| MC | 0.769 | 0.640 |
| aw | 0.933 | 0.359 |
| TSS | 1.000 | 0.000 |
| TA | 0.999 | −0.050 |
| pH | 0.570 | 0.822 |
| Yield | 0.818 | −0.576 |
| TPC | 0.748 | −0.664 |
| RSA | 0.624 | −0.781 |
| FRAP | 0.336 | −0.942 |
| TAC | 0.884 | −0.468 |
| Solubility | 0.999 | −0.054 |
| Hygroscopicity | 0.969 | 0.247 |
| Bulk density | 0.970 | −0.243 |
| WHC | -0.978 | −0.208 |
| OHC | 0.815 | 0.580 |
| Scores | ||
| Maltodextrin | 2.309 | −3.377 |
| Gum arabic | 2.744 | 3.188 |
| Waxy starch | −5.053 | 0.189 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Adetoro, A.O.; Opara, U.L.; Fawole, O.A. Effect of Carrier Agents on the Physicochemical and Technofunctional Properties and Antioxidant Capacity of Freeze-Dried Pomegranate Juice (Punica granatum) Powder. Foods 2020, 9, 1388. https://doi.org/10.3390/foods9101388
Adetoro AO, Opara UL, Fawole OA. Effect of Carrier Agents on the Physicochemical and Technofunctional Properties and Antioxidant Capacity of Freeze-Dried Pomegranate Juice (Punica granatum) Powder. Foods. 2020; 9(10):1388. https://doi.org/10.3390/foods9101388
Chicago/Turabian StyleAdetoro, Adegoke Olusesan, Umezuruike Linus Opara, and Olaniyi Amos Fawole. 2020. "Effect of Carrier Agents on the Physicochemical and Technofunctional Properties and Antioxidant Capacity of Freeze-Dried Pomegranate Juice (Punica granatum) Powder" Foods 9, no. 10: 1388. https://doi.org/10.3390/foods9101388
APA StyleAdetoro, A. O., Opara, U. L., & Fawole, O. A. (2020). Effect of Carrier Agents on the Physicochemical and Technofunctional Properties and Antioxidant Capacity of Freeze-Dried Pomegranate Juice (Punica granatum) Powder. Foods, 9(10), 1388. https://doi.org/10.3390/foods9101388







