Process-Structure-Function in Association with the Main Bioactive of Black Rice Flour Sieving Fractions
Abstract
1. Introduction
2. Materials and Methods
2.1. Black Rice Sample
2.2. Proximate Chemical Composition
2.3. Phytochemicals Extraction
2.4. Phytochemicals Analysis
2.5. Proteins Extraction
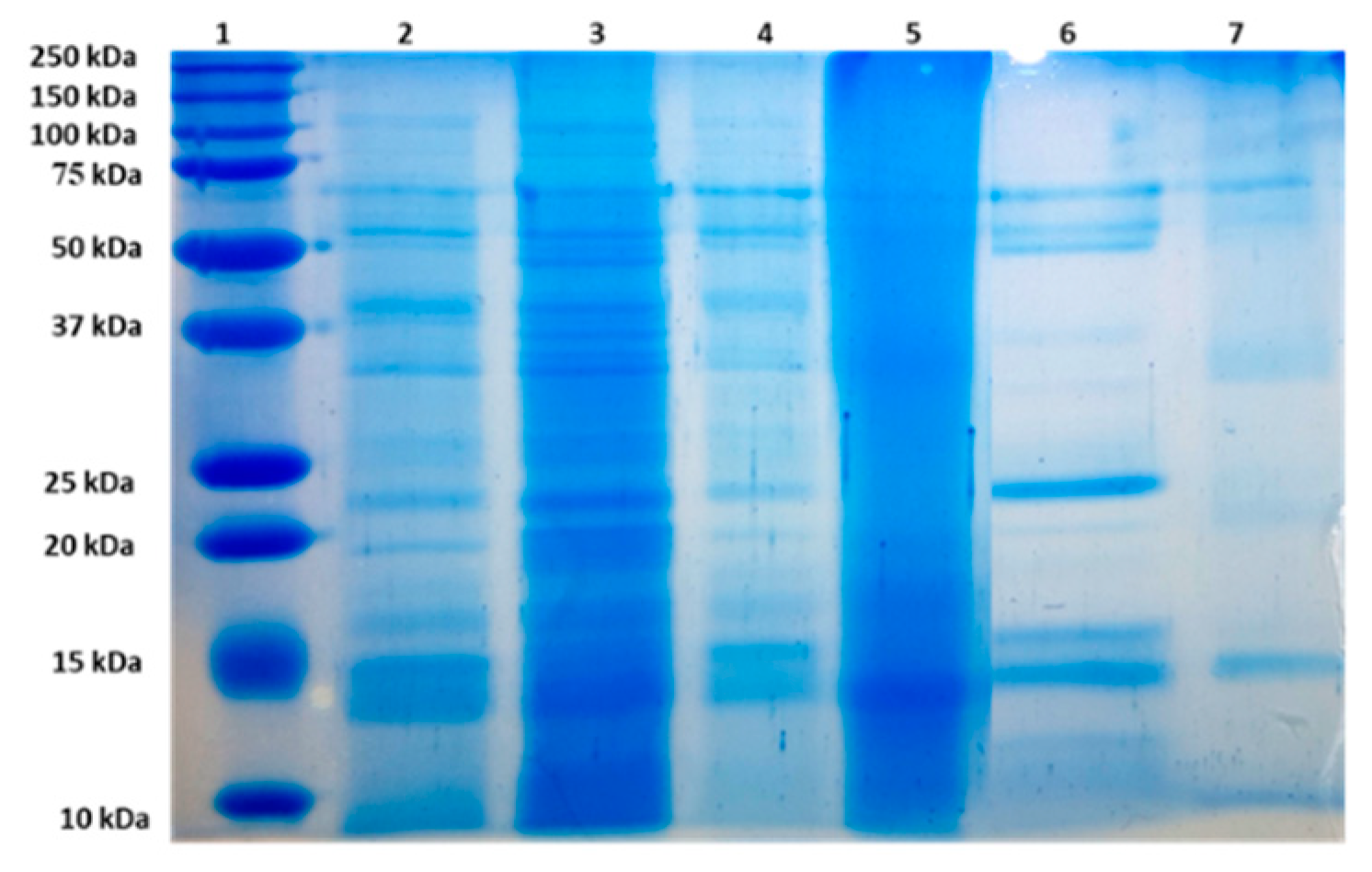
2.6. Black Rice Proteins Separation through SDS-PAGE
2.7. Heat Treatment
2.8. Kinetic Data Analysis
2.9. Fluorescence Spectroscopy Measurements
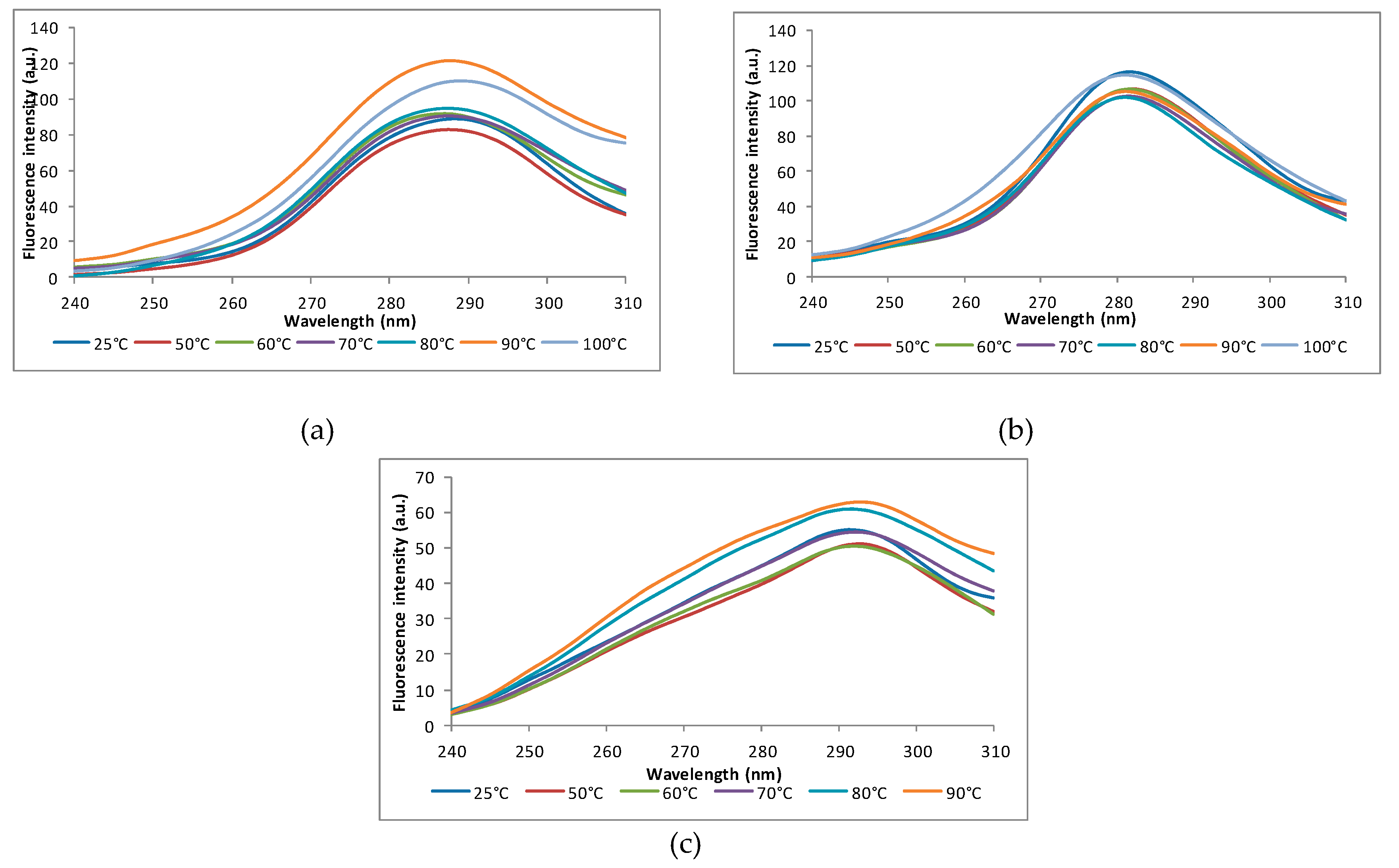
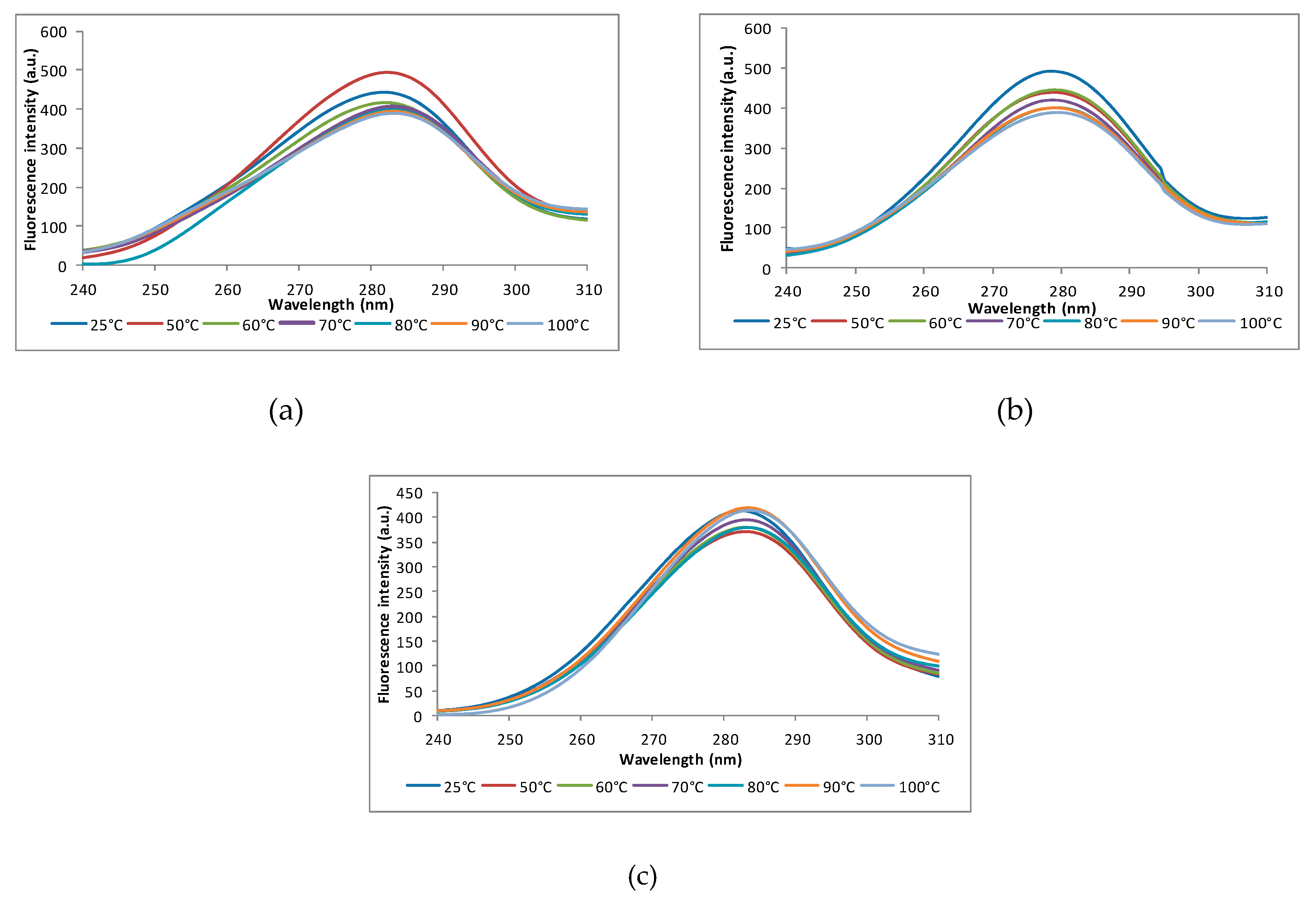
2.9.1. Intrinsic Fluorescence
2.9.2. Quenching Experiments
2.9.3. Synchronous Spectra
2.10. Statistical Analysis
3. Results and Discussion
3.1. Proximate Analysis of Chemical Composition of The Rice Milled Fractions
3.2. Phytochemical Content
3.3. Proteins Patterns of Black Rice
3.4. Kinetics of Phytochemical Thermal Degradation in Different Milled Fractions
3.5. The Effect of Temperature on the Black Rice Protein Fractions
3.5.1. Intrinsic Fluorescence
3.5.2. Synchronous Fluorescence Spectra
3.5.3. Quenching Experiments
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Okarter, O.; Liu, R.H. Health benefits of whole grain phytochemicals. Crit. Rev. Food Sci. Nutr. 2010, 50, 193–208. [Google Scholar] [CrossRef]
- Agboola, S.; Ng, D.; Mills, D. Characterisation and functional properties of Australian rice protein isolates. J. Cereal Sci. 2005, 41, 283–290. [Google Scholar] [CrossRef]
- Clampett, W.S.; Nguyen, V.N.; Tran, D.V. The Development and Use of Integrated Crop Management for Rice Production; International Rice Commission; FAO: Bangkok, Thailand, 2002. [Google Scholar]
- Prasad, K.; Prakash, P.; Prasad, K.K. Rice Based Functional Cookies for Celiac: Studies on Its Formulation; LAP Lambert Academic Publishing: Saarbrucken, Germany, 2010; p. 128. [Google Scholar]
- Zhang, M.W.; Guo, B.J.; Peng, Z.M. Genetic effects on grain characteristics of indica black rice and their uses on indirect selections for some mineral element contents in grains. Genet. Resour. Crop Evol. 2005, 52, 1121–1128. [Google Scholar] [CrossRef]
- Liu, R.H. Potential synergy of phytochemicals in cancer prevention: Mechanism of action. J. Nutr. 2004, 134, 3479S–3485S. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.W.; Zhang, R.F.; Zhang, F.X.; Liu, R.H. Phenolic Profiles and Antioxidant Activity of Black Rice Bran of Different Commercially Available Varieties. J. Agric. Food Chem. 2010, 58, 7580–7587. [Google Scholar] [CrossRef] [PubMed]
- Liu, R.H. Whole grain phytochemicals and health. J. Cereal Sci. 2007, 46, 207–219. [Google Scholar] [CrossRef]
- Leardkamolkarn, V.; Thongthep, W.; Suttiarporn, P.; Kongkachuichai, R.; Wongpornchai, S.; Wanavijitr, A. Chemopreventive properties of the bran extracted from a newly-developed Thai rice: The Riceberry. Food Chem. 2011, 125, 978–985. [Google Scholar] [CrossRef]
- Chou, P.H.; Matsui, S.; Misaki, K.; Matsuda, T. Isolation and identification of xenobiotic aryl hydrocarbon receptor ligands in dyeing wastewater. Environ. Sci. Technol. 2007, 41, 652–657. [Google Scholar] [CrossRef] [PubMed]
- Djantou, E.B.; Mbofung, C.M.; Scher, J.; Desobry, S. A modelling approach to determine the effect of pre-treatment on the grinding ability of dried mangoes for powder production (Mangifera indica var kent). J. Food Eng. 2007, 80, 668–677. [Google Scholar] [CrossRef]
- Tiwari, B.K.; O’Donnell, C.P.; Muthukumarappan, K.; Cullen, P.J. Anthocyanin and color degradation in ozone treated blackberry juice. Innov. Food Sci. Emerg. Technol. 2009, 10, 70–75. [Google Scholar] [CrossRef]
- Wang, W.D.; Xu, S.Y. Degradation kinetics of anthocyanins in blackberry juice and concentrate. J. Food Eng. 2007, 82, 271–275. [Google Scholar] [CrossRef]
- Oancea, A.-M.; Aprodu, I.; Ghinea, O.I.; Ioniță, E.; Râpeanu, G.; Barbu, V.; Bahrim, G.; Stănciuc, N. A bottom-up approach for encapsulation of sour cherries anthocyanins by using b-lactoglobulin as matrices. J. Food Eng. 2017, 210, 83–90. [Google Scholar] [CrossRef]
- Stred, S.E.; Stubbart, J.R.; Argetsinger, L.S.; Smith, W.C.; Shafer, J.A.; Talamantes, F.; Carterr, C. Stimulation by growth hormone (GH) of GH receptor-associated tyrosine kinase activity. Endocrinology 1992, 130, 1626–1636. [Google Scholar] [CrossRef]
- Turturică, M.; Stanciuc, N.; Bahrim, G.; Rapeanu, R. Effect of thermal treatment on phenolic compounds from plum (Prunus domestica) extracts e a kinetic study. J. Food Eng. 2016, 171, 200–207. [Google Scholar] [CrossRef]
- Turturică, M.; Stănciuc, N.; Bahrim, G.; Râpeanu, G. Investigations on sweet cherry phenolics degradation during thermal treatment based on fluorescence spectroscopy and inactivation kinetics. Food Bioprocess Technol. 2016, 9, 1706–1715. [Google Scholar] [CrossRef]
- Bolea, C.; Turturică, M.; Stănciuc, N.; Vizireanu, C. Thermal degradation kinetics of bioactive compounds from black rice flour (Oryza sativa L.) extracts. J. Cereal Sci. 2016, 71, 160–166. [Google Scholar] [CrossRef]
- Ellepola, S.W.; Ma, C.-Y. Thermal properties of globulin from rice (Oryza sativa) seeds. Food Res. Int. 2006, 39, 257–264. [Google Scholar] [CrossRef]
- Dumitrașcu, L.; Stănciuc, N.; Bahrim, G.E.; Aprodu, I. Insights into the binding of ferulic acid to the thermally treated xanthine oxidase. Lumin. J. Biol. Chem. Lumin. 2016, 31, 1259–1266. [Google Scholar] [CrossRef]
- Verma, D.K.; Srivastav, P.P. Proximate Composition, Mineral Content and Fatty Acids Analyses of Aromatic and Non-Aromatic Indian Rice. Rice Sci. 2017, 24, 21–31. [Google Scholar] [CrossRef]
- Asaduzzaman, M.; Haque, M.E.; Rahman, J.; Hasan, S.K.; Ali, M.A.; Akter, M.S.; Ahmed, M. Comparisons of physiochemical, total phenol, flavonoid content and functional properties in six cultivars of aromatic rice in Bangladesh. Acad. J. 2013, 7, 198–203. [Google Scholar]
- Saikia, S.; Himjyoti, D.; Daizi, S.; Charu, L.M. Quality characterization and estimation of phytochemical content capacity of aromatic pigmented and non-pigmented rice varieties. Food Res. Int. 2012, 46, 334–340. [Google Scholar] [CrossRef]
- Sompong, R.; Ehn, S.; Martin, L.; Berghofer, E. Physicochemical and antioxidant properties of red and black rice varieties from Thailand, China and Sri Lanka. Food Chem. 2011, 124, 132–140. [Google Scholar] [CrossRef]
- Chagam, K.R.; Lalmuan, K.; Sundaramoorthy, H.; Nayoung, K. Effects of Polishing on Proximate Composition, Physico-Chemical Characteristics, Mineral Composition and Antioxidant Properties of Pigmented Rice. Rice Sci. 2017, 24, 241–252. [Google Scholar]
- Itani, T.; Tatemoto, H.; Okamoto, M.; Fujii, K.; Muto, N. A comparative study on antioxidative activity and polyphenol content of colored kernel rice. J. Jpn. Soc. Nutr. Food Sci. 2002, 49, 540–543. [Google Scholar] [CrossRef]
- Paiva, F.F.; Vanier, N.L.; Berrios, J.D.J.; Pinto, V.Z.; Wood, D.; Williams, T.; Pan, J.; Elias, M.C. Polishing and parboiling effect on the nutritional and technological properties of pigmented rice. Food Chem. 2016, 191, 105–112. [Google Scholar] [CrossRef] [PubMed]
- Murdifin, M.; Pakki, E.; Rahim, A.; Syaiful, S.A.; Evary, Y.M.; Bahar, M.A. Physicochemical Properties of Indonesian Pigmented Rice (Oryza sativa Linn.) Varieties from South Sulawesi. Asian J. Plant Sci. 2015, 14, 59–65. [Google Scholar] [CrossRef]
- Bolea, C.; Vizireanu, C. Polyphenolic content and antioxidant properties of black rice flour. The Annals of the University Dunarea de Jos of Galati Fascicle VI—Food Technology 2017, 41, 75–85. [Google Scholar]
- Gong, E.R.; Luo, S.J.; Li, T.; Liu, C.M.; Zhang, G.W.; Chen, J.; Zenga, Z.C.; Liu, R.H. Phytochemical profiles and antioxidant activity of brown rice varieties. Food Chem. 2017, 227, 432–443. [Google Scholar] [CrossRef]
- Van Der Borght, A.; Vandeputte, G.E.; Derycke, V.; Brijs, K.; Daenen, G.; Delcour, J.A. Extractability and chromatographic separation of rice endosperm proteins. J. Cereal Sci. 2006, 44, 68–74. [Google Scholar] [CrossRef]
- Zhao, Q.; Selomulya, C.; Xiong, H.; Chen, D.X.; Ruan, X.; Wang, S.; Xie, J.; Peng, H.; Sun, W.; Zhou, Q. Comparison of functional and structural properties of native and industrial process-modified proteins from long-grain indica rice. J. Cereal Sci. 2012, 56, 568–575. [Google Scholar] [CrossRef]
- Ju, Z.Y.; Hettiarachchy, N.S.; Rath, N. Extraction, Denaturation and Hydrophobic Properties of Rice Flour Proteins. J. Food Sci. 2001, 66, 229–232. [Google Scholar] [CrossRef]
- Kawakatsu, T.; Yamamoto, M.P.; Hirose, S.; Yano, M.; Takaiwa, F. Characterization of a new rice glutelin gene GluD-1 expressed in the starchy endosperm. J. Exp. Bot. 2008, 59, 4233–4245. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Okita, T.W. Accumulation of Prolamines and Glutelins during Rice Seed Development: A Quantitative Evaluation. Plant Cell Physiol. 1993, 34, 385–390. [Google Scholar]
- Baccaro, S.; Bal, O.; Cemmi, A.; Di Sarcina, I. The Effect of Gamma Irradiation on Rice Protein Aqueous Solution. Radiat. Phys. Chem. 2018, 146, 1–4. [Google Scholar] [CrossRef]
- Romani, V.P.; Prentice, C.; Martins, V.G. Active and sustainable materials from rice starch, fish protein and oregano essential oil for food packaging. Ind. Crop. Prod. 2017, 97, 268–274. [Google Scholar] [CrossRef]
- Thakur, S.; Singh, P.K.; Das, A.; Rathour, R.; Variar, M.; Prashanthi, S.K.; Singh, A.K.; Singh, U.D.; Chand, D.; Singh, N.K.; et al. Extensive sequence variation in rice blast resistance gene Pi54 makes it broad spectrum in nature. Front Plant Sci. 2015, 6, 345. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Han, S.-W.; Chee, K.; Cho, S.-J. Nutritional quality of rice bran protein in comparison to animal and vegetable protein. Food Chem. 2015, 172, 766–769. [Google Scholar] [CrossRef]
- Shin, R.; Jez, J.M.; Basra, A.; Zhang, B.; Schachtman, D.P. 14-3-3 proteins fine-tune plant nutrient metabolism. FEBS Lett. 2011, 585, 143–147. [Google Scholar] [CrossRef] [PubMed]
- Lakowicz, J.R. Principles of Fluorescence Spectroscopy, 3rd ed.; Plenum Press: New York, NY, USA, 2006. [Google Scholar]
- Mensi, A.; Choiset, Y.; Rabesona, H.; Haertlé, T.; Borel, P.; Chobert, J.-M. Interactions of β-lactoglobulin variants A and B with vitamin A. Competitive binding of retinoids and carotenoids. J. Agric. Food Chem. 2013, 61, 4114–4119. [Google Scholar] [CrossRef] [PubMed]
- Tang, C.H.; Ma, C.Y. Effect of high pressure treatment on aggregation and structural properties of soy protein isolate. LWT Food Sci. Technol. 2009, 42, 606–611. [Google Scholar] [CrossRef]
- Zhao, Q.; Xiong, H.; Selomulya, C.; Chen, X.D.; Zhong, H.; Wang, S.; Sun, W.; Zhou, Q. Enzymatic hydrolysis of rice dreg protein: Effects of enzyme type on the functional properties and antioxidant activities of recovered proteins. Food Chem. 2012, 134, 1360–1367. [Google Scholar] [CrossRef] [PubMed]
- Vekshin, N.L. Separation of the tyrosine and tryptophan components of fluorescence using synchronous scanning method. Biofizika 1996, 41, 1176–1179. [Google Scholar]
| Compounds | Temperature ᵒC | F1 | F2 | F3 | F5 | F6 | F7 |
|---|---|---|---|---|---|---|---|
| k∙10−2 (1/min) | |||||||
| TAC | 60 | 17.43 ± 1.01 a | 10.89 ± 1.70 b | 3.75 ± 1.11 a | 4.01 ± 0.91 c | 5.61 ± 1.26 a | 3.01 ± 1.23 b |
| 70 | 18.42 ± 1.15 a | 9.51 ± 1.72 b | 4.26 ± 0.10 a | 13.35 ± 1.53 b | 5.12 ± 1.21 a | 2.99 ± 0.82 ab | |
| 80 | 18.51 ± 1.14 a | 9.76 ± 2.81 b | 4.72 ± 1.21 a | 13.28 ± 1.62 ab | 5.08 ± 1.42 a | 3.38 ± 1.25 ab | |
| 90 | 20.38 ± 1.28 a | 12.45 ± 1.71 b | 5.59 ± 0.10 a | 13.67 ± 1.89 ab | 5.34 ± 1.12 a | 3.96 ± 1.23 ab | |
| 100 | 20.42 ± 1.22 a | 17.11 ± 2.20 a | 5.55 ± 0.41 a | 15.03 ± 1.24 a | 5.61 ± 1.10 a | 5.15 ± 1.21 a | |
| Ea (kJ/Mol) | 4.31 ± 0.98 | 4.69 ± 0.93 | 10.77 ± 1.53 | 10.18 ± 2.89 | 11.99 ± 1.81 | 13.79 ± 0.20 | |
| TPC | 60 | 2.37 ± 0.24 a | 1.88 ± 0.12 d | 1.88 ± 0.10 c | 0.89 ± 0.31 c | 0.92 ± 0.11 a | 0.82 ± 0.20 c |
| 70 | 2.53 ± 0.25 a | 2.23 ± 0.20 c | 2.07 ± 0.22 bc | 1.15 ± 0.31 b | 1.19 ± 0.12 a | 0.92 ± 0.11 c | |
| 80 | 2.97 ± 0.37 a | 2.59 ± 0.21 c | 2.12 ± 0.92 ab | 1.22 ± 0.16 b | 1.24 ± 0.31 a | 1.31 ± 0.11 b | |
| 90 | 3.23 ± 0.27 a | 2.58 ± 0.63 a | 2.11 ± 0.52 ab | 1.31 ± 0.23 ab | 1.35 ± 0.22 a | 1.45 ± 0.10 b | |
| 100 | 3.23 ± 0.17 a | 2.71 ± 0.10 b | 2.32 ± 0.20 a | 1.58 ± 0.11 a | 1.63 ± 0.31 a | 1.63 ± 0.11 a | |
| Ea (kJ/Mol) | 7.05 ± 1.33 | 12.58 ± 0.76 | 4.57 ± 1.68 | 13.16 ± 1.86 | 13.18 ± 1.99 | 18.77 ± 2.35 | |
| TFC | 60 | 4.83 ± 0.40 b | 3.24 ± 0.30 d | 2.67 ± 0.21 a | 1.10 ± 0.41 d | 2.56 ± 0.11 bc | 2.25 ± 0.20 a |
| 70 | 4.92 ± 0.41 b | 3.38 ± 0.63 d | 2.69 ± 0.28 a | 1.77 ± 0.51 c | 2.54 ± 0.23 c | 2.23 ± 0.29 a | |
| 80 | 5.02 ± 0.04 b | 3.77 ± 0.30 c | 2.58 ± 0.10 b | 1.95 ± 0.10 bc | 2.71 ± 0.20 ab | 2.18 ± 0.12 a | |
| 90 | 5.24 ± 0.50 b | 4.28 ± 0.40 b | 2.71 ± 0.24 a | 2.23 ± 0.23 b | 2.97 ± 0.21 ab | 2.14 ± 0.12 a | |
| 100 | 5.31 ± 0.50 a | 4.88 ± 0.40 a | 2.78 ± 0.62 a | 2.83 ± 0.52 a | 3.01 ± 0.32 a | 2.11 ± 0.22 a | |
| Ea (kJ/Mol) | 1.99 ± 0.70 | 10.78 ± 1.25 | 0.90 ± 1.51 | 21.93 ± 3.43 | 3.84 ± 2.21 | 1.98 ± 0.87 | |
| DPPH | 60 | 1.33 ± 0.11 a | 1.28 ± 0.21 c | 1.30 ± 0.10 b | 0.78 ± 0.21 a | 0.69 ± 0.43 b | 0.94 ± 0.12 bc |
| 70 | 1.40 ± 0.10 a | 1.51 ± 0.10 bc | 1.42 ± 0.31 a | 0.73 ± 0.22 a | 0.76 ± 0.32 b | 0.92 ± 0.63 c | |
| 80 | 1.49 ± 0.31 a | 1.51 ± 0.12 bc | 1.48 ± 0.21 a | 0.78 ± 0.33 a | 0.76 ± 0.31 b | 1.01 ± 1.10 bc | |
| 90 | 2.14 ± 0.20 a | 1.65 ± 0.20 a | 1.49 ± 0.10 a | 0.86 ± 0.2 a | 1.01 ± 0.61 a | 1.10 ± 1.00 b | |
| 100 | 2.18 ± 0.32 a | 1.84 ± 0.10 b | 2.18 ± 0.31 a | 1.22 ± 0.52 a | 1.26 ± 0.51a | 1.63 ± 1.10 a | |
| Ea (kJ/Mol) | 16.61 ± 1.98 | 8.27 ± 1.30 | 9.39 ± 1.87 | 9.82 ± 1.23 | 16.61 ± 3.98 | 13.80 ± 1.49 | |
| Temperature °C | KSV (10−3 L/mol) | ||
|---|---|---|---|
| Albumins | Globulins | Glutenins | |
| 25 | 9.40 ± 0.55 b | 18.07 ± 1.79 ab | 13.60 ± 0.94 a |
| 50 | 11.72 ± 0.47 ab | 15.57 ± 0.54 ab | 8.55 ± 0.68 a |
| 60 | 15.11 ± 3.32 a | 19.41 ± 0.06 a | 12.47 ± 0.04 a |
| 70 | 13.79 ± 1.11 ab | 12.51 ± 0.19 b | 13.25 ± 0.46 a |
| 80 | 14.3 0± 1.84 ab | 12.22 ± 1.96 b | 10.96 ± 0.67 a |
| 90 | 13.75 ± 2.84 ab | 14.93 ± 0.86 ab | 13.79 ± 1.77 a |
| 100 | 13.52 ± 0.80 ab | 19.63 ± 2.70 a | 14.15 ± 1.67 a |
| Temperature °C | KSV (10−3 L/mol) | ||
|---|---|---|---|
| Albumins | Globulins | Glutenins | |
| 25 | 4.92 ± 0.87 a | 7.03 ± 1.17 a | 1.37 ±0.42 b |
| 50 | 5.04 ± 0.79 a | 6.56 ± 0.21 a | 2.37 ± 0.11 ab |
| 60 | 5.65 ± 1.01 a | 5.10 ± 0.95 a | 1.99 ± 0.51 b |
| 70 | 5.79 ± 1.41 a | 6.65 ± 1.80 a | 1.29 ± 0.65 b |
| 80 | 4.70 ± 0.57 a | 4.53 ± 1.50 a | 1.81 ± 0.32 b |
| 90 | 6.41 ± 1.00 a | 5.43 ± 2.26 a | 4.38 ± 1.15 a |
| 100 | 6.47 ± 0.53 a | 6.21 ± 1.10 a | 3.12 ± 0.91 ab |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bolea, C.A.; Grigore-Gurgu, L.; Aprodu, I.; Vizireanu, C.; Stănciuc, N. Process-Structure-Function in Association with the Main Bioactive of Black Rice Flour Sieving Fractions. Foods 2019, 8, 131. https://doi.org/10.3390/foods8040131
Bolea CA, Grigore-Gurgu L, Aprodu I, Vizireanu C, Stănciuc N. Process-Structure-Function in Association with the Main Bioactive of Black Rice Flour Sieving Fractions. Foods. 2019; 8(4):131. https://doi.org/10.3390/foods8040131
Chicago/Turabian StyleBolea, Carmen Alina, Leontina Grigore-Gurgu, Iuliana Aprodu, Camelia Vizireanu, and Nicoleta Stănciuc. 2019. "Process-Structure-Function in Association with the Main Bioactive of Black Rice Flour Sieving Fractions" Foods 8, no. 4: 131. https://doi.org/10.3390/foods8040131
APA StyleBolea, C. A., Grigore-Gurgu, L., Aprodu, I., Vizireanu, C., & Stănciuc, N. (2019). Process-Structure-Function in Association with the Main Bioactive of Black Rice Flour Sieving Fractions. Foods, 8(4), 131. https://doi.org/10.3390/foods8040131








