Chemical Compositions of the Volatile Oils and Antibacterial Screening of Solvent Extract from Downy Lavender
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Plant Material and Extraction
2.3. Bacterial Strains and Cultivation
2.4. GC-MS Analysis of L. pubescens
2.5. Antibacterial Screening
2.5.1. Disk Diffusion Method
2.5.2. Minimum Inhibitory Concentration
3. Results
3.1. GC-MS Analysis of L. pubescens
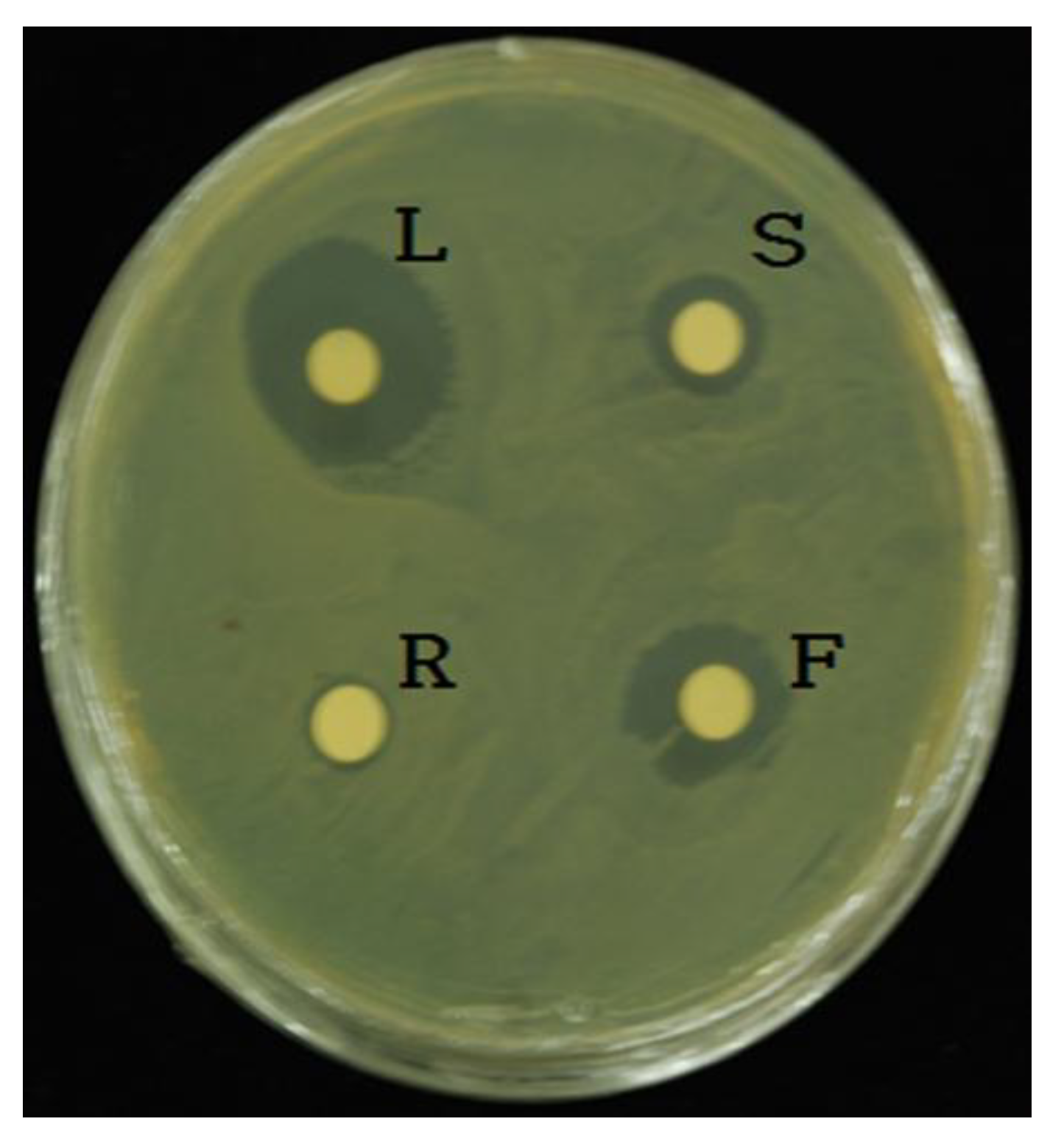
3.2. Antimicrobial Screening of L. pubescens
3.3. Minimum Inhibitory Concentration of L. pubescens
4. Discussion
5. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Lis-Balchin, M. Lavender: The Genus Lavandula, 1st ed.; CRC Press: London, UK, 2002; pp. 1–296. [Google Scholar]
- Bakkali, F.; Averbeck, S.; Averbeck, D.; Idaomar, M. Biological effects of essential oils—A review. Food Chem. Toxicol. 2008, 46, 446–475. [Google Scholar] [CrossRef] [PubMed]
- Djenane, D.; Aïder, M.; Yangüela, J.; Idir, L.; Gómez, D.; Roncalés, P. Antioxidant and antibacterial effects of Lavandula and Mentha essential oils in minced beef inoculated with E. coli O157: H7 and S. aureus during storage at abuse refrigeration temperature. Meat Sci. 2012, 92, 667–674. [Google Scholar] [CrossRef] [PubMed]
- An, M.; Haig, T.; Hatfield, P. On-site field sampling and analysis of fragrance from living Lavender (Lavandula angustifolia L.) flowers by solid-phase microextraction coupled to gas chromatography and ion-trap mass spectrometry. J. Chromatogr. 2001, 917, 245–250. [Google Scholar] [CrossRef]
- Bicchi, C.; Drigo, S.; Rubiolo, P. Influence of fibre coating in headspace solid-phase microextraction–gas chromatographic analysis of aromatic and medicinal plants. J. Chromatogr. 2000, 892, 469–485. [Google Scholar] [CrossRef]
- Kim, N.-S.; Lee, D.-S. Comparison of different extraction methods for the analysis of fragrances from Lavandula species by gas chromatography–mass spectrometry. J. Chromatogr. 2002, 982, 31–47. [Google Scholar] [CrossRef]
- Sarker, L.S.; Galata, M.; Demissie, Z.A.; Mahmoud, S.S. Molecular cloning and functional characterization of borneol dehydrogenase from the glandular trichomes of Lavandula x intermedia. Arch. Biochem. Biophys. 2012, 528, 163–170. [Google Scholar] [CrossRef]
- Swamy, M.K.; Akhtar, M.S.; Sinniah, U.R. Antimicrobial properties of plant essential oils against human pathogens and their mode of action: An updated review. Evid. Based Complement. Alternat. Med. 2016, 2016, 3012462. [Google Scholar] [CrossRef] [PubMed]
- Giarratana, F.; Muscolino, D.; Ziino, G.; Giuffrida, A.; Marotta, S.M.; Presti, V.L.; Chiofalo, V.; Panebianco, A. Activity of Tagetes minuta Linnaeus (Asteraceae) essential oil against L3 Anisakis larvae type 1. Asian Pac. J. Trop. Med. 2017, 10, 461–465. [Google Scholar] [CrossRef] [PubMed]
- Giarratana, F.; Muscolino, D.; Ziino, G.; Lo Presti, V.; Rao, R.; Chiofalo, V.; Giuffrida, A.; Panebianco, A. Activity of Catmint (Nepeta cataria) essential oil against Anisakis larvae. Trop. Biomed. 2017, 34, 22–31. [Google Scholar]
- Giarratana, F.; Muscolino, D.; Beninati, C.; Ziino, G.; Giuffrida, A.; Panebianco, A. Activity of R (+) limonene on the maximum growth rate of fish spoilage organisms and related effects on shelf-life prolongation of fresh gilthead sea bream fillets. Int. J. Food Microbiol. 2016, 237, 109–113. [Google Scholar] [CrossRef] [PubMed]
- Giarratana, F.; Muscolino, D.; Beninati, C.; Ziino, G.; Giuffrida, A.; Panebianco, A. Effects of thyme and rosemary essential oils on the microbiology and shelf life of Italian Mortadella. Fleischwirtschaft 2013, 93, 183–187. [Google Scholar]
- Burt, S. Essential oils: Their antibacterial properties and potential applications in foods—A review. Int. J. Food Microbiol. 2004, 94, 223–253. [Google Scholar] [CrossRef]
- Cavanagh, H.M.; Wilkinson, J.M. Lavender essential oil: A review. Aust. Infect. Control 2005, 10, 35–37. [Google Scholar] [CrossRef]
- De Castro, M.L.; Jimenez-Carmona, M.; Fernandez-Perez, V. Towards more rational techniques for the isolation of valuable essential oils from plants. TrAC Trends Anal. Chem. 1999, 18, 708–716. [Google Scholar] [CrossRef]
- Da Porto, C.; Decorti, D.; Kikic, I. Flavour compounds of Lavandula angustifolia L. to use in food manufacturing: Comparison of three different extraction methods. Food Chem. 2009, 112, 1072–1078. [Google Scholar] [CrossRef]
- Parkash, V.; Singh, H. Lavandula angustifolia L. (lavender): An important aromatic medicinal shrub and its in vitro micro-propagation for conservation. J. Agric. Technol. 2013, 9, 91–702. [Google Scholar]
- Kageyama, A.; Ueno, T.; Oshio, M.; Masuda, H.; Horiuchi, H.; Yokogoshi, H. Antidepressant-like effects of an aqueous extract of lavender (Lavandula angustifolia Mill.) in rats. Food Sci. Technol. Res 2012, 18, 473–479. [Google Scholar] [CrossRef]
- Koulivand, P.H.; Khaleghi Ghadiri, M.; Gorji, A. Lavender and the nervous system. Evid. Based Complement. Alternat. Med. 2013, 2013, 681304. [Google Scholar] [CrossRef] [PubMed]
- Kovatcheva, E.G.; Koleva, I.I.; Ilieva, M.; Pavlov, A.; Mincheva, M.; Konushlieva, M. Antioxidant activity of extracts from Lavandula vera MM cell cultures. Food Chem. 2001, 72, 295–300. [Google Scholar] [CrossRef]
- Lis-Balchin, M.; Hart, S. Studies on the mode of action of the essential oil of Lavender Lavandula angustifolia P. Miller. Phytother. Res. 1999, 13, 540–542. [Google Scholar] [CrossRef]
- Soltani, R.; Soheilipour, S.; Hajhashemi, V.; Asghari, G.; Bagheri, M.; Molavi, M. Evaluation of the effect of aromatherapy with lavender essential oil on post-tonsillectomy pain in pediatric patients: A randomized controlled trial. Int. J. Pediatr. Otorhinolaryngol. 2013, 77, 1579–1581. [Google Scholar] [CrossRef]
- Cavanagh, H.; Wilkinson, J. Biological activities of lavender essential oil. Phytother. Res. 2002, 16, 301–308. [Google Scholar] [CrossRef]
- Kashani, M.S.; Tavirani, M.R.; Talaei, S.A.; Salami, M. Aqueous extract of lavender (Lavandula angustifolia) improves the spatial performance of a rat model of Alzheimer’s disease. Neurosci. Bull. 2011, 27, 99–106. [Google Scholar] [CrossRef]
- Sienkiewicz, M.; Lysakowska, M.; Ciecwierz, J.; Denys, P.; Kowalczyk, E. Antibacterial activity of thyme and lavender essential oils. Med. Chem. 2011, 7, 674–689. [Google Scholar] [CrossRef]
- Park, C.H.; Yeo, H.J.; Baskar, T.B.; Park, Y.E.; Park, J.S.; Lee, S.Y.; Park, S.U. In Vitro Antioxidant and Antimicrobial Properties of Flower, Leaf, and Stem Extracts of Korean Mint. Antioxidants 2019, 8, 75. [Google Scholar] [CrossRef]
- Lim, S.S.; Jang, J.M.; Park, W.T.; Uddin, M.R.; Chae, S.C.; Kim, H.H.; Park, S.U. Chemical Composition of Essential Oils from Flower and Leaf of Korean Mint, Agastache rugosa. Asian J. Chem. 2013, 25, 4361–4363. [Google Scholar] [CrossRef]
- Abdullah Al-Dhabi, N.; Valan Arasu, M.; Rejiniemon, T.S. In vitro antibacterial, antifungal, antibiofilm, antioxidant, and anticancer properties of isosteviol isolated from endangered medicinal plant Pittosporum tetraspermum. Evid. Based Complement. Alternat. Med. 2015, 2015, 164261. [Google Scholar] [CrossRef]
- Barocelli, E.; Calcina, F.; Chiavarini, M.; Impicciatore, M.; Bruni, R.; Bianchi, A.; Ballabeni, V. Antinociceptive and gastroprotective effects of inhaled and orally administered Lavandula hybrida Reverchon “Grosso” essential oil. Life Sci. 2004, 76, 213–223. [Google Scholar] [CrossRef]
- Dadalioǧlu, I.; Evrendilek, G.A. Chemical compositions and antibacterial effects of essential oils of Turkish oregano (Origanum minutiflorum), bay laurel (Laurus nobilis), Spanish lavender (Lavandula stoechas L.), and fennel (Foeniculum vulgare) on common foodborne pathogens. J. Agric. Food Chem. 2004, 52, 8255–8260. [Google Scholar] [CrossRef]
- Karamanoli, K.; Vokou, D.; Menkissoglu, U.; Constantinidou, H.-I. Bacterial colonization of phyllosphere of Mediterranean aromatic plants. J. Chem. Ecol. 2000, 26, 2035–2048. [Google Scholar] [CrossRef]
- Hanamanthagouda, M.S.; Kakkalameli, S.B.; Naik, P.M.; Nagella, P.; Seetharamareddy, H.R.; Murthy, H.N. Essential oils of Lavandula bipinnata and their antimicrobial activities. Food Chem. 2010, 118, 836–839. [Google Scholar] [CrossRef]
- Jianu, C.; Pop, G.; TGruia, A.; Horhat, F.G. Chemical composition and antimicrobial activity of essential oils of lavender (Lavandula angustifolia) and lavandin (Lavandula × intermedia) grown in Western Romania. Int. J. Agric. Biol. 2013, 15, 772–776. [Google Scholar]
- Al-Badani, R.N.; da Silva, J.K.R.; Mansi, I.; Muharam, B.A.; Setzer, W.N.; Awadh Ali, N.A. Chemical composition and biological activity of Lavandula pubescens essential oil from Yemen. J. Essent. Oil. Bear. Plants 2017, 20, 509–515. [Google Scholar] [CrossRef]
- Thaker, A.; ANJARIA, J. Antimicrobial and infected wound healing response of some traditional drugs. Indian J. Pharmacol. 1986, 18, 171–174. [Google Scholar]
- Costa, P.; Gonçalves, S.; Valentão, P.; Andrade, P.B.; Almeida, C.; Nogueira, J.M.; Romano, A. Metabolic profile and biological activities of Lavandula pedunculata subsp. lusitanica (Chaytor) Franco: Studies on the essential oil and polar extracts. Food Chem. 2013, 141, 2501–2506. [Google Scholar] [CrossRef]
- Zuzarte, M.; Gonçalves, M.; Cruz, M.; Cavaleiro, C.; Canhoto, J.; Vaz, S.; Pinto, E.; Salgueiro, L. Lavandula luisieri essential oil as a source of antifungal drugs. Food Chem. 2012, 135, 1505–1510. [Google Scholar] [CrossRef]
- Sienkiewicz, M.; Głowacka, A.; Kowalczyk, E.; Wiktorowska-Owczarek, A.; Jóźwiak-Bębenista, M.; Łysakowska, M. The biological activities of cinnamon, geranium and lavender essential oils. Molecules 2014, 19, 20929–20940. [Google Scholar] [CrossRef]
- Cherrat, L.; Espina, L.; Bakkali, M.; Pagán, R.; Laglaoui, A. Chemical composition, antioxidant and antimicrobial properties of Mentha pulegium, Lavandula stoechas and Satureja calamintha Scheele essential oils and an evaluation of their bactericidal effect in combined processes. Innov. Food Sci. Emerg. Technol. 2014, 22, 221–229. [Google Scholar] [CrossRef]
- Hui, L.; He, L.; Huan, L.; XiaoLan, L.; AiGuo, Z. Chemical composition of lavender essential oil and its antioxidant activity and inhibition against rhinitis-related bacteria. Afr. J. Microbiol. Res. 2010, 4, 309–313. [Google Scholar]
- Hossain, S.; Heo, H.; De Silva, B.; Wimalasena, S.; Pathirana, H.; Heo, G.-J. Antibacterial activity of essential oil from lavender (Lavandula angustifolia) against pet turtle-borne pathogenic bacteria. Lab. Anim. Res. 2017, 33, 195–201. [Google Scholar] [CrossRef]
- Kim, M.S.; Lee, K.W.; Park, E.J. Antimicrobial Activity of Lavander and Rosemary Essential Oil Nanoemulsions. Korean J. Food Cook. Sci. 2017, 33, 256–263. [Google Scholar] [CrossRef]
- Moon, T.; Wilkinson, J.; Cavanagh, H. Antibacterial activity of essential oils, hydrosols and plant extracts from Australian grown Lavandula spp. Int. J. Aromather. 2006, 16, 9–14. [Google Scholar] [CrossRef]
- Akerele, O. Nature’s medicinal bounty: Don’t throw it away. World Health Forum 1993, 14, 390–395. [Google Scholar] [PubMed]
- Ekor, M. The growing use of herbal medicines: Issues relating to adverse reactions and challenges in monitoring safety. Front. Pharmacol. 2014, 4, 177. [Google Scholar] [CrossRef] [PubMed]

| Volatile Compound | Peak Area (%) | ||||
|---|---|---|---|---|---|
| RT 1 | Leaf | Stem | Flower | Root | |
| p-Xylene | 5.22 | ND 2 | ND | 0.15 | 2.46 |
| d-Limonene | 6.8 | ND | ND | 0.08 | 2 |
| γ-Terpinene | 7.37 | 1.64 | 11.09 | 2.39 | 2.28 |
| α-Terpinene | 7.49 | ND | ND | 0.33 | 4.22 |
| 1,3,5,5-Tetramethyl-1,3-cyclohexadiene | 7.6 | ND | ND | 2.95 | ND |
| m-Cymene | 7.67 | 2.1 | 2.89 | 1.31 | 49 |
| 2,5,5-Trimethyl-1,3,6-heptatriene | 7.81 | 0.76 | 1.83 | 2.75 | ND |
| 4-Terpinyl acetate | 8.19 | ND | 1.34 | 16.99 | 3.73 |
| Terpinolene | 8.67 | 3.31 | 10.17 | ND | 1.9 |
| p-Cymenene | 8.74 | 18.15 | 7.66 | 11.66 | 1.99 |
| trans-3-Caren-2-ol | 9.11 | 0.76 | 2.51 | 1.12 | 0.33 |
| (−)-4-Terpineol | 10.13 | ND | ND | ND | 3.05 |
| Neo-allo-ocimene | 9.32 | 21.91 | 15.68 | 8.55 | 0.36 |
| α-Terpinene | 10.35 | ND | ND | 0.54 | 1.57 |
| Isothymol methyl ether | 11.06 | 2.8 | 13.35 | ND | 0.35 |
| Carvacrol | 11.97 | 0.78 | 2.54 | 0.27 | 0.41 |
| α-Cubebene | 12.58 | ND | 0.65 | ND | 0.43 |
| α-Ylangene | 12.9 | ND | 0.38 | ND | 0.41 |
| α-Copaene | 12.96 | ND | ND | ND | 1.54 |
| (−)-β-Elemene | 13.15 | 0.16 | ND | ND | 0.66 |
| Alloaromadendrene | 13.22 | 2.45 | ND | ND | 0.16 |
| (+)-Ledene | 13.41 | ND | ND | ND | 0.14 |
| Longifolene | 13.46 | ND | 1.04 | 2.2 | ND |
| Caryophyllene | 13.58 | 2.45 | 9.97 | 0.23 | 0.91 |
| β-Ylangene | 13.67 | 0.13 | 1.46 | ND | ND |
| α-Bergamotene | 13.7 | ND | ND | 0.36 | 1 |
| Aromandendrene | 13.82 | ND | 0.11 | ND | 0.07 |
| Caryophyllene oxide | 13.94 | ND | 0.33 | ND | 0.19 |
| Acoradien | 14.02 | 0.07 | 0.08 | 0.07 | 0.38 |
| δ-Cadinene | 14.23 | ND | ND | ND | 0.88 |
| Cyclosativene | 14.25 | ND | ND | ND | 0.81 |
| α-Muurolene | 14.3 | 0.17 | ND | ND | 0.86 |
| β-Copaene | 14.35 | ND | ND | ND | 0.61 |
| β-Eudesmene | 14.45 | ND | 0.58 | ND | 1.21 |
| α-Gurjunene | 14.53 | ND | ND | ND | 3.3 |
| β-Bisabolene | 14.6 | 0.96 | 3.52 | 0.72 | ND |
| γ-Cadinene | 14.74 | ND | ND | ND | 0.48 |
| (−)-Calamenene | 14.85 | ND | ND | ND | 6.33 |
| β-Guaiene | 15.03 | 0.11 | ND | ND | 0.42 |
| α-Calacorene | 15.11 | 0.11 | ND | ND | 0.48 |
| Jolkinol D | 20.22 | 0.16 | ND | ND | ND |
| 1,3,5,7-Cyclooctatetraene | 5.57 | 0.85 | ND | 0.37 | ND |
| (3E)-2,7-Dimethyl-3-octen-5-yen | 6.05 | ND | 0.14 | 0.18 | 2.35 |
| 5,6-Dimethylene-1-cyclooctene | 6.62 | 0.48 | ND | 0.02 | ND |
| Benzaldehyde | 6.79 | 1.22 | ND | 0.12 | ND |
| 1,2,3,4,5-Pentamethylcyclopentadiene | 6.88 | 0.63 | ND | 0.85 | ND |
| 2,5-Dimethyl-3-methylene-5-heptadiene | 7.15 | ND | 1.04 | 2.42 | ND |
| α-Cumene hydroperoxide | 7.71 | 1.45 | 2.05 | 0.13 | ND |
| 6-Methyl-5-(1-methylethylidene)-6,8-nonadien-2-one | 7.72 | 8.25 | 2.66 | 6.47 | ND |
| (3Z)-2,7-Dimethyl-3-octen-5-yne | 8.04 | 0.55 | ND | ND | ND |
| 1,3-Dimethyl-1,5-cyclooctadiene | 8.41 | 0.31 | ND | 19.76 | ND |
| 1-Methyl6-isopropylidene | 8.6 | 6.18 | 2.51 | 14.94 | ND |
| 4-(1,5-Dihydro-2,4,3-benzodioxaborepin-3-yl) benzoic acid | 9.04 | 0.36 | ND | ND | ND |
| 1-Amino-2-(4-chlorobenzoyl)-6,7,8,9-tetrahydro-5-methylthieno[2,3-c]isoquinoline | 9.62 | 0.65 | 2.87 | ND | 0.61 |
| Naphthalene | 10.31 | 0.39 | ND | ND | ND |
| 6-Isopropenyl-3-(methoxymethoxy)-3-methyl-1-cyclohexene | 10.34 | 0.62 | ND | ND | ND |
| Benzothiazole | 10.98 | 0.38 | ND | 1.42 | ND |
| 3,5,7-Trimethoxy-2-(4-methoxyphenyl)-4H-chromen-4-one | 12.09 | 0.35 | 0.83 | ND | 0.18 |
| δ-Valerolactone | 14.06 | 0.44 | ND | 0.09 | 0.24 |
| 2,6-Di-tert-butylbenzoquinone | 14.13 | 0.27 | ND | ND | 0.38 |
| 4,4,5,6-Tetramethyl-1,3-oxazinane-2-thione | 16.23 | 3.06 | ND | ND | ND |
| 2-(3-Acetoxy-4,4,10,13,14-pentamethyl-2,3,4,5,6,7,10,11,12,13,14,15,16,17-tetradecahydro-1H-cyclopenta[a]phenanthren-17-yl)-propanoic acid | 16.34 | 0.26 | ND | ND | 0.09 |
| 2,6-Di-tert-butylhydroquinone | 16.74 | 0.65 | ND | ND | ND |
| 3,5-Bis(tert-butyl)-4-hydroxy-propiophenon | 17.79 | 0.21 | ND | ND | ND |
| (6-tert-Butyl-1,1-dimethyl-2,3-dihydro-1H-inden-4-yl)acetic acid | 17.88 | 0.5 | ND | ND | ND |
| 2,4,6-Tri-tert-butylphenol | 18.2 | 5.18 | ND | ND | ND |
| 1-(3,5-Di-tert-butyl-4-hydroxyphenyl)propan-1-one | 18.95 | 0.52 | ND | ND | ND |
| 3,3,3′,3′,5,5,5′,5′-Octamethyl-1,1′-bi(1-cyclohexen-1yl) | 19.23 | 0.29 | 0.2 | ND | ND |
| 3-Deoxyestradiol | 19.49 | 0.14 | ND | ND | 0.15 |
| 1-Nitrosopyrrolidine | 20.74 | 1.48 | ND | ND | ND |
| 3-Hydroxyspirost-8-en-11-one | 28.36 | ND | ND | ND | 0.1 |
| Total identified (%) | 94.65 | 99.48 | 99.44 | 99.02 | |
| Strains | Zone of Inhibition(mm) from L. pubescens Extracts | |||||
|---|---|---|---|---|---|---|
| MeOH 1 | EtOH 2 | DE 3 | EA 4 | Hexane | Streptomycin | |
| Escherichia coli (KF 918342) | NA 5 | NA | NA | 21.3 ± 0.6 | NA | 27.6 ± 0.6 |
| Staphylococcus haemolyticus | NA | NA | NA | 24.0 ± 0.0 | NA | 26.3 ± 0.6 |
| Aeromonas hydrophila | NA | NA | NA | 21.0 ± 1.0 | NA | 27.0 ± 0.0 |
| Escherichia coli (ATCC 35150) | NA | NA | NA | 20.6 ± 1.1 | NA | 28.3 ± 0.6 |
| Cronobacter sakazakii | NA | NA | NA | 22.3 ± 0.6 | NA | 25.6 ± 0.6 |
| Aeromonas salmonicida | NA | NA | NA | 19.6 ± 1.5 | NA | 27.0 ± 1.0 |
| Strains | Zone of Inhibition(mm) from L. pubescens Extracts | |||
|---|---|---|---|---|
| Flower | Leaf | Stem | Root | |
| Escherichia coli (KF 918342) | 17.6 ± 0.6 | 22.3 ± 0.6 | 15.3 ± 0.6 | NA 1 |
| staphylococcus haemolyticus | 17.6 ± 0.6 | 23.0 ± 1.0 | 19.0 ± 1.0 | NA |
| Aeromonas hydrophila | 14.3 ± 1.5 | 16.0 ± 0.0 | 13.7 ± 0.6 | NA |
| Escherichia coli (ATCC 35150) | 15.3 ± 0.6 | 18.0 ± 1.0 | 13.0 ± 0.0 | NA |
| Cronobacter sakazakii | 13.7 ± 0.6 | 21.3 ± 1.1 | 13.6 ± 0.6 | NA |
| Aeromonas salmonicida | 14.7 ± 0.6 | 20.3 ± 0.6 | 12.3 ± 0.6 | NA |
| Strains | Minimum Inhibitory Concentration (MIC) | |
|---|---|---|
| Compound (µL) | Standard (µg) | |
| Escherichia coli (KF 918342) | 6.25 | 25 |
| Staphylococcus haemolyticus | 12.5 | 50 |
| Aeromonas hydrophila | 12.5 | 50 |
| Escherichia coli (ATCC 35150) | 6.25 | 25 |
| Cronobacter sakazakii | 25 | 100 |
| Aeromonas salmonicida | 12.5 | 50 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Park, C.H.; Park, Y.E.; Yeo, H.J.; Chun, S.W.; Baskar, T.B.; Lim, S.S.; Park, S.U. Chemical Compositions of the Volatile Oils and Antibacterial Screening of Solvent Extract from Downy Lavender. Foods 2019, 8, 132. https://doi.org/10.3390/foods8040132
Park CH, Park YE, Yeo HJ, Chun SW, Baskar TB, Lim SS, Park SU. Chemical Compositions of the Volatile Oils and Antibacterial Screening of Solvent Extract from Downy Lavender. Foods. 2019; 8(4):132. https://doi.org/10.3390/foods8040132
Chicago/Turabian StylePark, Chang Ha, Ye Eun Park, Hyeon Ji Yeo, Se Won Chun, Thanislas Bastin Baskar, Soon Sung Lim, and Sang Un Park. 2019. "Chemical Compositions of the Volatile Oils and Antibacterial Screening of Solvent Extract from Downy Lavender" Foods 8, no. 4: 132. https://doi.org/10.3390/foods8040132
APA StylePark, C. H., Park, Y. E., Yeo, H. J., Chun, S. W., Baskar, T. B., Lim, S. S., & Park, S. U. (2019). Chemical Compositions of the Volatile Oils and Antibacterial Screening of Solvent Extract from Downy Lavender. Foods, 8(4), 132. https://doi.org/10.3390/foods8040132





