Natural Sources of Selenium as Functional Food Products for Chemoprevention
Abstract
1. Introduction
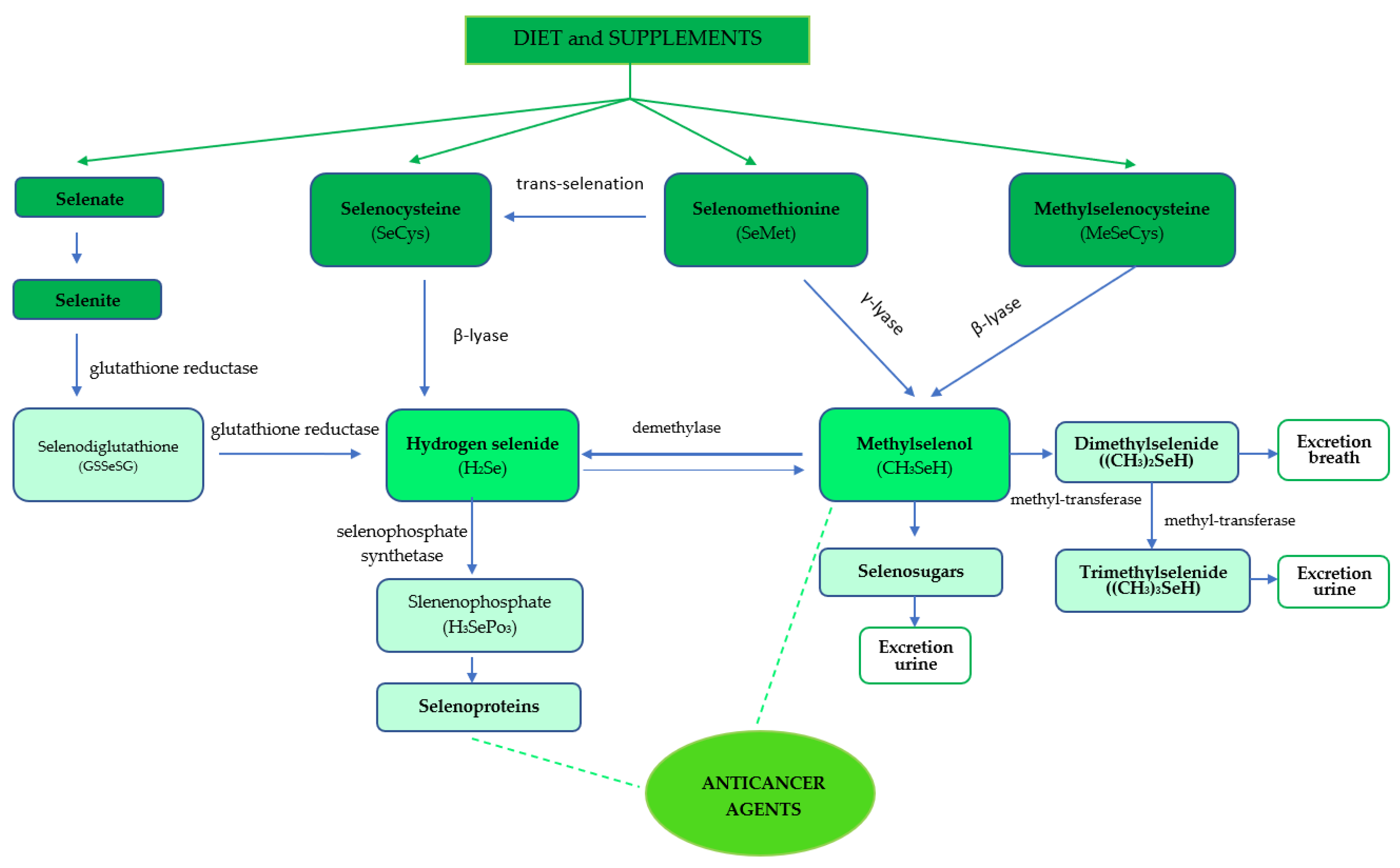
2. Dietary Sources of Selenium Compounds and Their Metabolism
Selenium Bioavailability
3. Functional Foods Rich in Selenium
3.1. Food Products Naturally Rich in Selenium Used in Cancer Prevention
3.2. Brazil Nuts
3.3. Brassica Species
3.4. Allium Species
3.5. Se-Enriched Yeast
3.6. Other Se-Enriched Food Products
4. Concluding Remarks—The Chemoprevention Effect of Se-Enriched Products
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Ferlay, J.; Colombet, M.; Soerjomataram, I.; Parkin, D.M.; Piñeros, M.; Znaor, A.; Bray, F. Cancer Statistics for the Year 2020: An Overview. Int. J. Cancer 2021, 149, 778–789. [Google Scholar] [CrossRef]
- Siegel, R.L.; Miller, K.D.; Fuchs, H.E.; Jemal, A. Cancer Statistics, 2022. CA A Cancer J. Clin. 2022, 72, 7–33. [Google Scholar] [CrossRef]
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA A Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef]
- Glade, M.J. Food, Nutrition, and the Prevention of Cancer: A Global Perspective. American Institute for Cancer Research/World Cancer Research Fund, American Institute for Cancer Research, 1997. Nutrition 1999, 15, 523–526. [Google Scholar] [CrossRef]
- Donaldson, M.S. Nutrition and Cancer: A Review of the Evidence for an Anti-Cancer Diet. Nutr. J. 2004, 3, 19. [Google Scholar] [CrossRef]
- Rataan, A.O.; Geary, S.M.; Zakharia, Y.; Rustum, Y.M.; Salem, A.K. Potential Role of Selenium in the Treatment of Cancer and Viral Infections. Int. J. Mol. Sci. 2022, 23, 2215. [Google Scholar] [CrossRef]
- Clark, L.C. Effects of Selenium Supplementation for Cancer Prevention in Patients With Carcinoma of the Skin: A Randomized Controlled Trial. JAMA 1996, 276, 1957. [Google Scholar] [CrossRef]
- Klein, E.A.; Thompson, I.M.; Lippman, S.M.; Goodman, P.J.; Albanes, D.; Taylor, P.R.; Coltman, C. SELECT: The Selenium and Vitamin E Cancer Prevention Trial. Urol. Oncol. Semin. Orig. Investig. 2003, 21, 59–65. [Google Scholar] [CrossRef] [PubMed]
- El-Bayoumy, K. The Negative Results of the SELECT Study Do Not Necessarily Discredit the Selenium-Cancer Prevention Hypothesis. Nutr. Cancer 2009, 61, 285–286. [Google Scholar] [CrossRef] [PubMed]
- Lippman, S.M.; Goodman, P.J.; Klein, E.A.; Parnes, H.L.; Thompson, I.M.; Kristal, A.R.; Santella, R.M.; Probstfield, J.L.; Moinpour, C.M.; Albanes, D.; et al. Designing the Selenium and Vitamin E Cancer Prevention Trial (SELECT). JNCI J. Natl. Cancer Inst. 2005, 97, 94–102. [Google Scholar] [CrossRef] [PubMed]
- Lippman, S.M.; Klein, E.A.; Goodman, P.J.; Lucia, M.S.; Thompson, I.M.; Ford, L.G.; Parnes, H.L.; Minasian, L.M.; Gaziano, J.M.; Hartline, J.A.; et al. Effect of Selenium and Vitamin E on Risk of Prostate Cancer and Other Cancers: The Selenium and Vitamin E Cancer Prevention Trial (SELECT). JAMA 2009, 301, 39. [Google Scholar] [CrossRef]
- Kristal, A.R.; Darke, A.K.; Morris, J.S.; Tangen, C.M.; Goodman, P.J.; Thompson, I.M.; Meyskens, F.L.; Goodman, G.E.; Minasian, L.M.; Parnes, H.L.; et al. Baseline Selenium Status and Effects of Selenium and Vitamin E Supplementation on Prostate Cancer Risk. JNCI J. Natl. Cancer Inst. 2014, 106, djt456. [Google Scholar] [CrossRef] [PubMed]
- Vinceti, M.; Filippini, T.; Del Giovane, C.; Dennert, G.; Zwahlen, M.; Brinkman, M.; Zeegers, M.P.; Horneber, M.; D’Amico, R.; Crespi, C.M. Selenium for Preventing Cancer. Cochrane Database Syst. Rev. 2018, 1, 1–233. [Google Scholar] [CrossRef] [PubMed]
- Rayman, M.P. Food-Chain Selenium and Human Health: Emphasis on Intake. Br. J. Nutr. 2008, 100, 254–268. [Google Scholar] [CrossRef] [PubMed]
- Combs, G.F. Selenium in Global Food Systems. Br. J. Nutr. 2001, 85, 517–547. [Google Scholar] [CrossRef]
- Rayman, M.P. The Importance of Selenium to Human Health. Lancet 2000, 356, 233–241. [Google Scholar] [CrossRef]
- Shreenath, A.P.; Ameer, M.A.; Dooley, J. Selenium Deficiency. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2022. [Google Scholar]
- EFSA Panel on Dietetic Products, Nutrition and Allergies (NDA). Scientific Opinion on Dietary Reference Values for Selenium. EFSA J. 2014, 12, 3846. [Google Scholar] [CrossRef]
- El-Bayoumy, K. The Protective Role of Selenium on Genetic Damage and on Cancer. Mutat. Res./Fundam. Mol. Mech. Mutagen. 2001, 475, 123–139. [Google Scholar] [CrossRef]
- Turck, D.; Bohn, T.; Castenmiller, J.; de Henauw, S.; Hirsch-Ernst, K.; Knutsen, H.K.; Maciuk, A.; Mangelsdorf, I.; McArdle, H.J.; et al.; EFSA Panel on Nutrition, Novel Foods and Food Allergens (NDA) Scientific Opinion on the Tolerable Upper Intake Level for Selenium. EFS2 2023, 21, 1–194. [Google Scholar] [CrossRef]
- Jaworska-Bieniek, K.; Lener, M.; Muszynska, M.; Serrano-Fernández, P.; Sukiennicki, G.; Durda, K.; Gromowski, T.; Gupta, S.; Kladny, J.; Wiechowska-Kozlowska, A.; et al. CHAPTER 22. Selenium and Cancer. In Food and Nutritional Components in Focus; Preedy, V.R., Ed.; Royal Society of Chemistry: Cambridge, UK, 2015; pp. 377–390. ISBN 978-1-84973-891-0. [Google Scholar]
- Collery, P. Strategies for the Development of Selenium-Based Anticancer Drugs. J. Trace Elem. Med. Biol. 2018, 50, 498–507. [Google Scholar] [CrossRef]
- Weekley, C.M.; Harris, H.H. Which Form Is That? The Importance of Selenium Speciation and Metabolism in the Prevention and Treatment of Disease. Chem. Soc. Rev. 2013, 42, 8870. [Google Scholar] [CrossRef]
- Hariharan, S.; Dharmaraj, S. Selenium and Selenoproteins: It’s Role in Regulation of Inflammation. Inflammopharmacol 2020, 28, 667–695. [Google Scholar] [CrossRef]
- Morris, D.L. CHAPTER 4. Chemistry and Activity of Selenium Dioxide and Selenite Ions. In Food and Nutritional Components in Focus; Preedy, V.R., Ed.; Royal Society of Chemistry: Cambridge, UK, 2015; pp. 65–80. ISBN 978-1-84973-891-0. [Google Scholar]
- Kotrebai, M.; Tyson, J.F.; Uden, P.C.; Birringer, M.; Block, E. Selenium Speciation in Enriched and Natural Samples by HPLC-ICP-MS and HPLC-ESI-MS with Perfluorinated Carboxylic Acid Ion-Pairing Agents. Analyst 2000, 125, 71–78. [Google Scholar] [CrossRef]
- Rayman, M.P.; Infante, H.G.; Sargent, M. Food-Chain Selenium and Human Health: Spotlight on Speciation. Br. J. Nutr. 2008, 100, 238–253. [Google Scholar] [CrossRef] [PubMed]
- Yang, R.; Liu, Y.; Zhou, Z. Selenium and Selenoproteins, from Structure, Function to Food Resource and Nutrition. FSTR 2017, 23, 363–373. [Google Scholar] [CrossRef]
- Tóth, R.J.; Csapó, J. The Role of Selenium in Nutrition—A Review. Acta Univ. Sapientiae Aliment. 2018, 11, 128–144. [Google Scholar] [CrossRef]
- Hu, Y.; McIntosh, G.H.; Young, G.P. Selenium-Rich Foods: A Promising Approach to Colorectal Cancer Prevention. CPB 2012, 13, 165–172. [Google Scholar] [CrossRef]
- Weekley, C.; Aitken, J.; Finney, L.; Vogt, S.; Witting, P.; Harris, H. Selenium Metabolism in Cancer Cells: The Combined Application of XAS and XFM Techniques to the Problem of Selenium Speciation in Biological Systems. Nutrients 2013, 5, 1734–1756. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, A.P.; Gandin, V. Selenium Compounds as Therapeutic Agents in Cancer. Biochim. Et Biophys. Acta BBA-Gen. Subj. 2015, 1850, 1642–1660. [Google Scholar] [CrossRef]
- Fairweather-Tait, S.J.; Collings, R.; Hurst, R. Selenium Bioavailability: Current Knowledge and Future Research Requirements. Am. J. Clin. Nutr. 2010, 91, 1484S–1491S. [Google Scholar] [CrossRef] [PubMed]
- Young, V.R.; Nahapetian, A.; Janghorbani, M. Selenium Bioavailability with Reference to Human Nutrition. Am. J. Clin. Nutr. 1982, 35, 1076–1088. [Google Scholar] [CrossRef]
- Ip, C.; Hayes, C.; Budnick, R.M.; Ganther, H.E. Chemical Form of Selenium, Critical Metabolites, and Cancer Prevention. Cancer Res. 1991, 51, 595–600. [Google Scholar]
- Whanger, P.D. Selenocompounds in Plants and Animals and Their Biological Significance. J. Am. Coll. Nutr. 2002, 21, 223–232. [Google Scholar] [CrossRef]
- Combs, G.F.; Gray, W.P. Chemopreventive Agents. Pharmacol. Ther. 1998, 79, 179–192. [Google Scholar] [CrossRef] [PubMed]
- Carlson, B.A.; Xu, X.-M.; Kryukov, G.V.; Rao, M.; Berry, M.J.; Gladyshev, V.N.; Hatfield, D.L. Identification and Characterization of Phosphoseryl-TRNA [Ser]Sec Kinase. Proc. Natl. Acad. Sci. USA 2004, 101, 12848–12853. [Google Scholar] [CrossRef]
- Papp, L.V.; Lu, J.; Holmgren, A.; Khanna, K.K. From Selenium to Selenoproteins: Synthesis, Identity, and Their Role in Human Health. Antioxid. Redox Signal. 2007, 9, 775–806. [Google Scholar] [CrossRef]
- Brozmanová, J.; Mániková, D.; Vlčková, V.; Chovanec, M. Selenium: A Double-Edged Sword for Defense and Offence in Cancer. Arch. Toxicol. 2010, 84, 919–938. [Google Scholar] [CrossRef]
- Brown, K.; Arthur, J. Selenium, Selenoproteins and Human Health: A Review. Public Health Nutr. 2001, 4, 593–599. [Google Scholar] [CrossRef] [PubMed]
- Davis, C.D.; Tsuji, P.A.; Milner, J.A. Selenoproteins and Cancer Prevention. Annu. Rev. Nutr. 2012, 32, 73–95. [Google Scholar] [CrossRef]
- Suzuki, K.T.; Kurasaki, K.; Suzuki, N. Selenocysteine β-Lyase and Methylselenol Demethylase in the Metabolism of Se-Methylated Selenocompounds into Selenide. Biochim. Et Biophys. Acta BBA-Gen. Subj. 2007, 1770, 1053–1061. [Google Scholar] [CrossRef] [PubMed]
- Ohta, Y.; Suzuki, K.T. Methylation and Demethylation of Intermediates Selenide and Methylselenol in the Metabolism of Selenium. Toxicol. Appl. Pharmacol. 2008, 226, 169–177. [Google Scholar] [CrossRef] [PubMed]
- Tobe, R.; Mihara, H. Delivery of Selenium to Selenophosphate Synthetase for Selenoprotein Biosynthesis. Biochim. Et Biophys. Acta BBA-Gen. Subj. 2018, 1862, 2433–2440. [Google Scholar] [CrossRef] [PubMed]
- Okuno, T.; Motobayashi, S.; Ueno, H.; Nakamuro, K. Purification and Characterization of Mouse Hepatic Enzyme That Converts Selenomethionine to Methylselenol by Its α,γ-Elimination. BTER 2005, 106, 077–094. [Google Scholar] [CrossRef]
- Okuno, T.; Ueno, H.; Nakamuro, K. Cystathionine γ-Lyase Contributes to Selenomethionine Detoxification and Cytosolic Glutathione Peroxidase Biosynthesis in Mouse Liver. BTER 2006, 109, 155–172. [Google Scholar] [CrossRef]
- Malhotra, A.; Dhawan, D.K. CHAPTER 13. Selenium as a Modulator of Fourier Transform Infrared Spectra in Cancer. In Food and Nutritional Components in Focus; Preedy, V.R., Ed.; Royal Society of Chemistry: Cambridge, UK, 2015; pp. 240–257. ISBN 978-1-84973-891-0. [Google Scholar]
- Whanger, P.D. Selenium and Its Relationship to Cancer: An Update. Br. J. Nutr. 2004, 91, 11–28. [Google Scholar] [CrossRef] [PubMed]
- Dong, Y.; Lisk, D.; Block, E.; Ip, C. Characterization of the Biological Activity of Gamma-Glutamyl-Se-Methylselenocysteine: A Novel, Naturally Occurring Anticancer Agent from Garlic. Cancer Res. 2001, 61, 2923–2928. [Google Scholar]
- Kobayashi, Y.; Ogra, Y.; Ishiwata, K.; Takayama, H.; Aimi, N.; Suzuki, K.T. Selenosugars Are Key and Urinary Metabolites for Selenium Excretion within the Required to Low-Toxic Range. Proc. Natl. Acad. Sci. USA 2002, 99, 15932–15936. [Google Scholar] [CrossRef]
- Zeng, H.; Wu, M.; Botnen, J.H. Methylselenol, a Selenium Metabolite, Induces Cell Cycle Arrest in G1 Phase and Apoptosis via the Extracellular-Regulated Kinase 1/2 Pathway and Other Cancer Signaling Genes. J. Nutr. 2009, 139, 1613–1618. [Google Scholar] [CrossRef]
- Zeng, H.; Briske-Anderson, M.; Wu, M.; Moyer, M.P. Methylselenol, a Selenium Metabolite, Plays Common and Different Roles in Cancerous Colon HCT116 Cell and Noncancerous NCM460 Colon Cell Proliferation. Nutr. Cancer 2012, 64, 128–135. [Google Scholar] [CrossRef]
- Zeng, H.; Cheng, W.-H.; Johnson, L.K. Methylselenol, a Selenium Metabolite, Modulates P53 Pathway and Inhibits the Growth of Colon Cancer Xenografts in Balb/c Mice. J. Nutr. Biochem. 2013, 24, 776–780. [Google Scholar] [CrossRef]
- Zeng, H.; Briske-Anderson, M. Methylselenol, a Selenium Metabolite, Plays a Critical Role in Inhibiting Colon Cancer Cell Growth in Vitro and in Vivo. FASEB J. 2011, 25, 110–114. [Google Scholar] [CrossRef]
- Zeng, H.; Combs, G.F. Selenium as an Anticancer Nutrient: Roles in Cell Proliferation and Tumor Cell Invasion. J. Nutr. Biochem. 2008, 19, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Knox, S.J.; Husbeck, B. Methods for Treating a Neoplastic Disease in a Subject Using Inorganic Selenium-Containing Compounds. Patent Appl. Publ. within the TVPP-United States PAT: US8932649B2, 13 January 2015. Available online: https://Patents.Google.Com/Patent/US8932649B2/En (accessed on 8 March 2023).
- Sinha, R.; Kiley, S.C.; Lu, J.X.; Thompson, H.J.; Moraes, R.; Jaken, S.; Medina, D. Effects of Methylselenocysteine on PKC Activity, Cdk2 Phosphorylation and Gadd Gene Expression in Synchronized Mouse Mammary Epithelial Tumor Cells. Cancer Lett. 1999, 146, 135–145. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Jiang, C.; Ganther, H.; Lü, J. Antimitogenic and Proapoptotic Activities of Methylseleninic Acid in Vascular Endothelial Cells and Associated Effects on PI3K-AKT, ERK, JNK and P38 MAPK Signaling. Cancer Res. 2001, 61, 7171–7178. [Google Scholar]
- Sanmartin, C.; Plano, D.; Palop, J. Selenium Compounds and Apoptotic Modulation: A New Perspective in Cancer Therapy. MRMC 2008, 8, 1020–1031. [Google Scholar] [CrossRef]
- Shini, S.; Sultan, A.; Bryden, W. Selenium Biochemistry and Bioavailability: Implications for Animal Agriculture. Agriculture 2015, 5, 1277–1288. [Google Scholar] [CrossRef]
- Thiry, C.; Ruttens, A.; De Temmerman, L.; Schneider, Y.-J.; Pussemier, L. Current Knowledge in Species-Related Bioavailability of Selenium in Food. Food Chem. 2012, 130, 767–784. [Google Scholar] [CrossRef]
- Ralston, N.V.C.; Raymond, L.J. Dietary Selenium’s Protective Effects against Methylmercury Toxicity. Toxicology 2010, 278, 112–123. [Google Scholar] [CrossRef]
- Reeves, P.G.; Leary, P.D.; Gregoire, B.R.; Finley, J.W.; Lindlauf, J.E.; Johnson, L.K. Selenium Bioavailability from Buckwheat Bran in Rats Fed a Modified AIN-93G Torula Yeast–Based Diet. J. Nutr. 2005, 135, 2627–2633. [Google Scholar] [CrossRef]
- Vagni, S.; Saccone, F.; Pinotti, L.; Baldi, A. Vitamin E Bioavailability: Past and Present Insights. FNS 2011, 2, 1088–1096. [Google Scholar] [CrossRef]
- Thomson, C.D. Assessment of Requirements for Selenium and Adequacy of Selenium Status: A Review. Eur. J. Clin. Nutr. 2004, 58, 391–402. [Google Scholar] [CrossRef]
- Krausova, G.; Kana, A.; Vecka, M.; Hyrslova, I.; Stankova, B.; Kantorova, V.; Mrvikova, I.; Huttl, M.; Malinska, H. In Vivo Bioavailability of Selenium in Selenium-Enriched Streptococcus Thermophilus and Enterococcus Faecium in CD IGS Rats. Antioxidants 2021, 10, 463. [Google Scholar] [CrossRef] [PubMed]
- Duttaroy, A.K. Regulation of Functional Foods in European Union. In Nutraceutical and Functional Food Regulations in the United States and around the World; Elsevier: Amsterdam, The Netherlands, 2019; pp. 267–276. ISBN 978-0-12-816467-9. [Google Scholar]
- Martirosyan, D.M.; Singh, J.A. New Definition for Functional Food by FFC: What Makes a New Definition Unique? Funct. Foods Health Dis. 2015, 5, 209–223. [Google Scholar] [CrossRef]
- Lagouri, V. Introductory Chapter: Functional Foods. In Functional Foods; Lagouri, V., Ed.; IntechOpen: Rijeka, Croatia, 2019; ISBN 978-1-83881-149-5. [Google Scholar]
- Hoadley, J.E. U.S. Regulation of Functional Foods. In Functional Foods; Elsevier: Amsterdam, The Netherlands, 2011; pp. 41–63. ISBN 978-1-84569-690-0. [Google Scholar]
- Adadi, P.; Barakova, N.V.; Muravyov, K.Y.; Krivoshapkina, E.F. Designing Selenium Functional Foods and Beverages: A Review. Food Res. Int. 2019, 120, 708–725. [Google Scholar] [CrossRef] [PubMed]
- Siró, I.; Kápolna, E.; Kápolna, B.; Lugasi, A. Functional Food. Product Development, Marketing and Consumer Acceptance—A Review. Appetite 2008, 51, 456–467. [Google Scholar] [CrossRef] [PubMed]
- Johnson, C.C.; Fordyce, F.M.; Rayman, M.P. Symposium on ‘Geographical and Geological Influences on Nutrition’ Factors Controlling the Distribution of Selenium in the Environment and Their Impact on Health and Nutrition: Conference on ‘Over- and Undernutrition: Challenges and Approaches’. Proc. Nutr. Soc. 2010, 69, 119–132. [Google Scholar] [CrossRef]
- Titov, A.F.; Kaznina, N.M.; Karapetyan, T.A.; Dorshakova, N.V.; Tarasova, V.N. Role of Selenium in Plants, Animals, and Humans. Biol. Bull Rev. 2022, 12, 189–200. [Google Scholar] [CrossRef]
- Parekh, P.P.; Khan, A.R.; Torres, M.A.; Kitto, M.E. Concentrations of Selenium, Barium, and Radium in Brazil Nuts. J. Food Compos. Anal. 2008, 21, 332–335. [Google Scholar] [CrossRef]
- Larsen, E.H.; Lobinski, R.; Burger-Meÿer, K.; Hansen, M.; Ruzik, R.; Mazurowska, L.; Rasmussen, P.H.; Sloth, J.J.; Scholten, O.; Kik, C. Uptake and Speciation of Selenium in Garlic Cultivated in Soil Amended with Symbiotic Fungi (Mycorrhiza) and Selenate. Anal. Bioanal. Chem. 2006, 385, 1098. [Google Scholar] [CrossRef]
- De Temmerman, L.; Waegeneers, N.; Thiry, C.; Du Laing, G.; Tack, F.; Ruttens, A. Selenium Content of Belgian Cultivated Soils and Its Uptake by Field Crops and Vegetables. Sci. Total Environ. 2014, 468–469, 77–82. [Google Scholar] [CrossRef]
- Pilarczyk, B.; Tomza-Marciniak, A.; Pilarczyk, R.; Kuba, J.; Hendzel, D.; Udała, J.; Tarasewicz, Z. Eggs as a Source of Selenium in the Human Diet. J. Food Compos. Anal. 2019, 78, 19–23. [Google Scholar] [CrossRef]
- Bratakos, M.S.; Zafiropoulos, T.F.; Siskos, P.A.; Ioannou, P.V. Selenium Losses on Cooking Greek Foods. Int. J. Food Sci. Technol. 2007, 23, 585–590. [Google Scholar] [CrossRef]
- Lu, X.; He, Z.; Lin, Z.; Zhu, Y.; Yuan, L.; Liu, Y.; Yin, X. Effects of Chinese Cooking Methods on the Content and Speciation of Selenium in Selenium Bio-Fortified Cereals and Soybeans. Nutrients 2018, 10, 317. [Google Scholar] [CrossRef] [PubMed]
- Thomson, C.D. Brazil Nuts (Bertholletia Excelsa). In Nuts and Seeds in Health and Disease Prevention; Elsevier: Amsterdam, The Netherlands, 2011; pp. 245–252. ISBN 978-0-12-375688-6. [Google Scholar]
- Lima, L.W.; Stonehouse, G.C.; Walters, C.; Mehdawi, A.F.E.; Fakra, S.C.; Pilon-Smits, E.A.H. Selenium Accumulation, Speciation and Localization in Brazil Nuts (Bertholletia Excelsa H.B.K.). Plants 2019, 8, 289. [Google Scholar] [CrossRef]
- Shahidi, F.; Tan, Z. Bioactives and Health Benefits of Brazil Nut. In Tree Nuts; Shahidi, F., Alasalvar, C., Eds.; Nutraceutical Science and Technology; CRC Press: Boca Raton, FL, USA, 2008; Volume 20084476, ISBN 978-0-8493-3735-2. [Google Scholar]
- Cardoso, B.R.; Duarte, G.B.S.; Reis, B.Z.; Cozzolino, S.M.F. Brazil Nuts: Nutritional Composition, Health Benefits and Safety Aspects. Food Res. Int. 2017, 100, 9–18. [Google Scholar] [CrossRef]
- Chang, J.C.; Gutenmann, W.H.; Reid, C.M.; Lisk, D.J. Selenium Content of Brazil Nuts from Two Geographic Locations in Brazil. Chemosphere 1995, 30, 801–802. [Google Scholar] [CrossRef] [PubMed]
- Broadley, M.R.; White, P.J.; Bryson, R.J.; Meacham, M.C.; Bowen, H.C.; Johnson, S.E.; Hawkesford, M.J.; McGrath, S.P.; Zhao, F.-J.; Breward, N.; et al. Biofortification of UK Food Crops with Selenium. Proc. Nutr. Soc. 2006, 65, 169–181. [Google Scholar] [CrossRef] [PubMed]
- Silva Junior, E.C.; Wadt, L.H.O.; Silva, K.E.; Lima, R.M.B.; Batista, K.D.; Guedes, M.C.; Carvalho, G.S.; Carvalho, T.S.; Reis, A.R.; Lopes, G.; et al. Natural Variation of Selenium in Brazil Nuts and Soils from the Amazon Region. Chemosphere 2017, 188, 650–658. [Google Scholar] [CrossRef] [PubMed]
- Thomson, C.D.; Chisholm, A.; McLachlan, S.K.; Campbell, J.M. Brazil Nuts: An Effective Way to Improve Selenium Status. Am. J. Clin. Nutr. 2008, 87, 379–384. [Google Scholar] [CrossRef]
- Ip, C.; Lisk, D.J. Bioactivity of Selenium from Brazil Nut for Cancer Prevention and Selenoenzyme Maintenance. Nutr. Cancer 1994, 21, 203–212. [Google Scholar] [CrossRef]
- Hu, Y.; McIntosh, G.H.; Le Leu, R.K.; Somashekar, R.; Meng, X.Q.; Gopalsamy, G.; Bambaca, L.; McKinnon, R.A.; Young, G.P. Supplementation with Brazil Nuts and Green Tea Extract Regulates Targeted Biomarkers Related to Colorectal Cancer Risk in Humans. Br. J. Nutr. 2016, 116, 1901–1911. [Google Scholar] [CrossRef] [PubMed]
- Godos, J.; Giampieri, F.; Micek, A.; Battino, M.; Forbes-Hernández, T.Y.; Quiles, J.L.; Paladino, N.; Falzone, L.; Grosso, G. Effect of Brazil Nuts on Selenium Status, Blood Lipids, and Biomarkers of Oxidative Stress and Inflammation: A Systematic Review and Meta-Analysis of Randomized Clinical Trials. Antioxidants 2022, 11, 403. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Liu, R.H.; Halim, L. Antioxidant and Antiproliferative Activities of Common Edible Nut Seeds. LWT-Food Sci. Technol. 2009, 42, 1–8. [Google Scholar] [CrossRef]
- Martens, I.B.G.; Cardoso, B.R.; Hare, D.J.; Niedzwiecki, M.M.; Lajolo, F.M.; Martens, A.; Cozzolino, S.M.F. Selenium Status in Preschool Children Receiving a Brazil Nut–Enriched Diet. Nutrition 2015, 31, 1339–1343. [Google Scholar] [CrossRef] [PubMed]
- McNeill, I.R.; Isoardi, K.Z. Barium Poisoning: An Uncommon Cause of Severe Hypokalemia. Toxicol. Commun. 2019, 3, 88–90. [Google Scholar] [CrossRef]
- Gustafson, P.F.; Stehney, A.F. Exposure Data for Radium Patients. In Environmental Research Division Annual Report; ANL-84-103 Part 11:98-180; Argonne National Laboratory: Argonne, IL, USA, 1985. [Google Scholar]
- Martínez-Hernandez, G.B.; Gomez, P.A.; Art´es, F.; Art´es-Hernandez, F. New Broccoli Varieties with Improved Health Benefits and Suitability for the Fresh–Cut and Fifth Range Industries: An Opportunity to Increase Its Consumption; Minglin, L., Ed.; Brassicaceae: Characterization, Functional Genomics and Health Benefits; Nova Science Publishers Inc.: New York, NY, USA, 2013; pp. 67–92. [Google Scholar]
- Kristal, A.R.; Lampe, J.W. Brassica Vegetables and Prostate Cancer Risk: A Review of the Epidemiological Evidence. Nutr. Cancer 2002, 42, 1–9. [Google Scholar] [CrossRef]
- White, P.J. Selenium Accumulation by Plants. Ann. Bot. 2015, 117, 217–235. [Google Scholar] [CrossRef]
- Wiesner-Reinhold, M.; Schreiner, M.; Baldermann, S.; Schwarz, D.; Hanschen, F.S.; Kipp, A.P.; Rowan, D.D.; Bentley-Hewitt, K.L.; McKenzie, M.J. Mechanisms of Selenium Enrichment and Measurement in Brassicaceous Vegetables, and Their Application to Human Health. Front. Plant Sci. 2017, 8, 1365. [Google Scholar] [CrossRef]
- Ávila, F.W.; Faquin, V.; Yang, Y.; Ramos, S.J.; Guilherme, L.R.G.; Thannhauser, T.W.; Li, L. Assessment of the Anticancer Compounds Se -Methylselenocysteine and Glucosinolates in Se-Biofortified Broccoli (Brassica Oleracea L. Var. Italica) Sprouts and Florets. J. Agric. Food Chem. 2013, 61, 6216–6223. [Google Scholar] [CrossRef]
- Finley, J.W.; Davis, C.D.; Feng, Y. Selenium from High Selenium Broccoli Protects Rats from Colon Cancer. J. Nutr. 2000, 130, 2384–2389. [Google Scholar] [CrossRef]
- Finley, J.W.; Ip, C.; Lisk, D.J.; Davis, C.D.; Hintze, K.J.; Whanger, P.D. Cancer-Protective Properties of High-Selenium Broccoli. J. Agric. Food Chem. 2001, 49, 2679–2683. [Google Scholar] [CrossRef]
- Davis, C.D.; Zeng, H.; Finley, J.W. Selenium-Enriched Broccoli Decreases Intestinal Tumorigenesis in Multiple Intestinal Neoplasia Mice. J. Nutr. 2002, 132, 307–309. [Google Scholar] [CrossRef] [PubMed]
- Zeng, H.; Davis, C.D. Effect of Selenium-Enriched Broccoli Diet on Differential Gene Expression in Min Mouse Liver. J. Nutr. Biochem. 2003, 14, 227–231. [Google Scholar] [CrossRef] [PubMed]
- Tsai, C.-F.; Ou, B.-R.; Liang, Y.-C.; Yeh, J.-Y. Growth Inhibition and Antioxidative Status Induced by Selenium-Enriched Broccoli Extract and Selenocompounds in DNA Mismatch Repair-Deficient Human Colon Cancer Cells. Food Chem. 2013, 139, 267–273. [Google Scholar] [CrossRef]
- Yamanoshita, O.; Ichihara, S.; Hama, H.; Ichihara, G.; Chiba, M.; Kamijima, M.; Takeda, I.; Nakajima, T. Chemopreventive Effect of Selenium-Enriched Japanese Radish Sprout against Breast Cancer Induced by 7,12-Dimethylbenz[a]Anthracene in Rats. Tohoku J. Exp. Med. 2007, 212, 191–198. [Google Scholar] [CrossRef]
- Zagrodzki, P.; Paśko, P.; Galanty, A.; Tyszka-Czochara, M.; Wietecha-Posłuszny, R.; Rubió, P.S.; Bartoń, H.; Prochownik, E.; Muszyńska, B.; Sułkowska-Ziaja, K.; et al. Does Selenium Fortification of Kale and Kohlrabi Sprouts Change Significantly Their Biochemical and Cytotoxic Properties? J. Trace Elem. Med. Biol. 2020, 59, 126466. [Google Scholar] [CrossRef]
- Abdulah, R.; Faried, A.; Kobayashi, K.; Yamazaki, C.; Suradji, E.W.; Ito, K.; Suzuki, K.; Murakami, M.; Kuwano, H.; Koyama, H. Selenium Enrichment of Broccoli Sprout Extract Increases Chemosensitivity and Apoptosis of LNCaP Prostate Cancer Cells. BMC Cancer 2009, 9, 414. [Google Scholar] [CrossRef]
- Ramirez, D.; Abellán-Victorio, A.; Beretta, V.; Camargo, A.; Moreno, D.A. Functional Ingredients From Brassicaceae Species: Overview and Perspectives. Int. J. Mol. Sci. 2020, 21, 1998. [Google Scholar] [CrossRef] [PubMed]
- Bastaki, S.M.A.; Ojha, S.; Kalasz, H.; Adeghate, E. Chemical Constituents and Medicinal Properties of Allium Species. Mol. Cell. Biochem. 2021, 476, 4301–4321. [Google Scholar] [CrossRef] [PubMed]
- Dalhat, M.H.; Adefolake, F.A.; Musa, M. Nutritional Composition and Phytochemical Analysis of Aqueous Extract of Allium Cepa (Onion) and Allium Sativum (Garlic). AFSJ 2018, 3, 1–9. [Google Scholar] [CrossRef]
- Galeone, C.; Pelucchi, C.; Levi, F.; Negri, E.; Franceschi, S.; Talamini, R.; Giacosa, A.; La Vecchia, C. Onion and Garlic Use and Human Cancer. Am. J. Clin. Nutr. 2006, 84, 1027–1032. [Google Scholar] [CrossRef]
- De Greef, D.; Barton, E.M.; Sandberg, E.N.; Croley, C.R.; Pumarol, J.; Wong, T.L.; Das, N.; Bishayee, A. Anticancer Potential of Garlic and Its Bioactive Constituents: A Systematic and Comprehensive Review. Semin. Cancer Biol. 2021, 73, 219–264. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Zhuang, W.; Hu, W.; Liu, G.; Wu, T.; Wu, X. Consumption of Large Amounts of Allium Vegetables Reduces Risk for Gastric Cancer in a Meta-Analysis. Gastroenterology 2011, 141, 80–89. [Google Scholar] [CrossRef] [PubMed]
- Tavares, L.; Santos, L.; Zapata Noreña, C.P. Bioactive Compounds of Garlic: A Comprehensive Review of Encapsulation Technologies, Characterization of the Encapsulated Garlic Compounds and Their Industrial Applicability. Trends Food Sci. Technol. 2021, 114, 232–244. [Google Scholar] [CrossRef]
- Bianchini, F.; Vainio, H. Allium Vegetables and Organosulfur Compounds: Do They Help Prevent Cancer? Environ. Health Perspect. 2001, 109, 893–902. [Google Scholar] [CrossRef] [PubMed]
- Lee, W.S.; Yi, S.M.; Yun, J.W.; Jung, J.H.; Kim, D.H.; Kim, H.J.; Chang, S.-H.; Kim, G.; Ryu, C.H.; Shin, S.C.; et al. Polyphenols Isolated from Allium Cepa L. Induces Apoptosis by Induction of P53 and Suppression of Bcl-2 through Inhibiting PI3K/Akt Signaling Pathway in AGS Human Cancer Cells. J. Cancer Prev. 2014, 19, 14–22. [Google Scholar] [CrossRef] [PubMed]
- González-Morales, S.; Pérez-Labrada, F.; García-Enciso, E.; Leija-Martínez, P.; Medrano-Macías, J.; Dávila-Rangel, I.; Juárez-Maldonado, A.; Rivas-Martínez, E.; Benavides-Mendoza, A. Selenium and Sulfur to Produce Allium Functional Crops. Molecules 2017, 22, 558. [Google Scholar] [CrossRef]
- Arnault, I.; Auger, J. Seleno-Compounds in Garlic and Onion. J. Chromatogr. A 2006, 1112, 23–30. [Google Scholar] [CrossRef]
- Jiang, C.; Jiang, W.; Ip, C.; Ganther, H.; Lu, J. Selenium-Induced Inhibition of Angiogenesis in Mammary Cancer at Chemopreventive Levels of Intake. Mol. Carcinog. 1999, 26, 213–225. [Google Scholar] [CrossRef]
- Ip, C.; Lisk, D.J.; Stoewsand, G.S. Mammary Cancer Prevention by Regular Garlic and Selenium-enriched Garlic. Nutr. Cancer 1992, 17, 279–286. [Google Scholar] [CrossRef]
- Ip, C.; Lisk, D.J. Enrichment of Selenium in Allium Vegetables for Cancer Prevention. Carcinogenesis 1994, 15, 1881–1885. [Google Scholar] [CrossRef] [PubMed]
- Ip, C.; Lisk, D.J. Efficacy of Cancer Prevention by High-Selenium Garlic Is Primarily Dependent on the Action of Selenium. Carcinogenesis 1995, 16, 2649–2652. [Google Scholar] [CrossRef] [PubMed]
- Ip, C.; Birringer, M.; Block, E.; Kotrebai, M.; Tyson, J.F.; Uden, P.C.; Lisk, D.J. Chemical Speciation Influences Comparative Activity of Selenium-Enriched Garlic and Yeast in Mammary Cancer Prevention. J. Agric. Food Chem. 2000, 48, 2062–2070. [Google Scholar] [CrossRef]
- Pyrzynska, K. Selenium Speciation in Enriched Vegetables. Food Chem. 2009, 114, 1183–1191. [Google Scholar] [CrossRef]
- Wrobel, K.; Kannamkumarath, S.S.; Wrobel, K.; Caruso, J.A. Hydrolysis of Proteins with Methanesulfonic Acid for Improved HPLC-ICP-MS Determination of Seleno-Methionine in Yeast and Nuts. Anal. Bioanal. Chem. 2003, 375, 133–138. [Google Scholar] [CrossRef] [PubMed]
- Dumont, E.; Ogra, Y.; Vanhaecke, F.; Suzuki, K.T.; Cornelis, R. Liquid Chromatography–Mass Spectrometry (LC–MS): A Powerful Combination for Selenium Speciation in Garlic (Allium Sativum). Anal. Bioanal. Chem. 2006, 384, 1196. [Google Scholar] [CrossRef] [PubMed]
- Dumont, E.; Vanhaecke, F.; Cornelis, R. Selenium Speciation from Food Source to Metabolites: A Critical Review. Anal. Bioanal. Chem. 2006, 385, 1304–1323. [Google Scholar] [CrossRef]
- Gao, X.; Kong, L. Treatment of Cancer with Selenium Nanoparticles, Patent Appl. Publ. within the TVPP-United States. PAT: US2011262564, 18 June 2010. Available online: https://Europepmc.Org/Article/PAT/US2011262564#abstract (accessed on 8 March 2023).
- Ip, C.; Lisk, D.J.; Thompson, H.J. Selenium-Enriched Garlic Inhibits the Early Stage but Not the Latestage of Mammary Carcinogenesis. Carcinogenesis 1996, 17, 1979–1982. [Google Scholar] [CrossRef]
- Block, E.; Birringer, M.; Jiang, W.; Nakahodo, T.; Thompson, H.J.; Toscano, P.J.; Uzar, H.; Zhang, X.; Zhu, Z. Allium Chemistry: Synthesis, Natural Occurrence, Biological Activity, and Chemistry of Se -Alk(En)Ylselenocysteines and Their γ-Glutamyl Derivatives and Oxidation Products. J. Agric. Food Chem. 2001, 49, 458–470. [Google Scholar] [CrossRef]
- Thomson, M.; Ali, M. Garlic [Allium Sativum]: A Review of Its Potential Use as an Anti-Cancer Agent. CCDT 2003, 3, 67–81. [Google Scholar] [CrossRef]
- Rayman, M.P. The Use of High-Selenium Yeast to Raise Selenium Status: How Does It Measure Up? Br. J. Nutr. 2004, 92, 557–573. [Google Scholar] [CrossRef] [PubMed]
- Esmaeili, S.; Khosravi-Darani, K. Selenium-Enriched Yeast: As Selenium Source for Nutritional Purpose. CNF 2014, 10, 49–56. [Google Scholar] [CrossRef]
- Navarro-Alarcon, M.; Cabrera-Vique, C. Selenium in Food and the Human Body: A Review. Sci. Total Environ. 2008, 400, 115–141. [Google Scholar] [CrossRef]
- Yin, H.; Fan, G.; Gu, Z. Optimization of Culture Parameters of Selenium-Enriched Yeast (Saccharomyces Cerevisiae) by Response Surface Methodology (RSM). LWT-Food Sci. Technol. 2010, 43, 666–669. [Google Scholar] [CrossRef]
- European Food Safety Authority (EFSA). EFSA Panel on Dietetic Products, Nutrition, and Allergies (NDA) Scientific Opinion on Dietary Reference Values for Fats, Including Saturated Fatty Acids, Polyunsaturated Fatty Acids, Monounsaturated Fatty Acids, Trans Fatty Acids, and Cholesterol. EFS2 2010, 8, 1–107. [Google Scholar] [CrossRef]
- European Food Safety Authority (EFSA). Selenium-enriched Yeast as Source for Selenium Added for Nutritional Purposes in Foods for Particular Nutritional Uses and Foods (Including Food Supplements) for the General Population-Scientific Opinion of the Panel on Food Additives, Flavourings, Processing Aids and Materials in Contact with Food. EFS2 2008, 6, 1–42. [Google Scholar] [CrossRef]
- Kieliszek, M. Selenium–Fascinating Microelement, Properties and Sources in Food. Molecules 2019, 24, 1298. [Google Scholar] [CrossRef] [PubMed]
- Yu Yu, S.; Zhu, Y.J.; Li, W.G. Protective Role of Selenium against Hepatitis B Virus and Primary Liver Cancer in Qidong. Biol Trace Elem. Res. 1997, 56, 117–124. [Google Scholar] [CrossRef]
- Spallholz, J.E. Selenomethionine and Methioninase: Selenium Free Radical Anticancer Activity. In Methionine Dependence of Cancer and Aging; Hoffman, R.M., Ed.; Methods in Molecular Biology; Springer: New York, NY, USA, 2019; Volume 1866, pp. 199–210. ISBN 978-1-4939-8795-5. [Google Scholar]
- Algotar, A.M.; Stratton, M.S.; Ahmann, F.R.; Ranger-Moore, J.; Nagle, R.B.; Thompson, P.A.; Slate, E.; Hsu, C.H.; Dalkin, B.L.; Sindhwani, P.; et al. Phase 3 Clinical Trial Investigating the Effect of Selenium Supplementation in Men at High-Risk for Prostate Cancer. Prostate 2013, 73, 328–335. [Google Scholar] [CrossRef]
- El-Bayoumy, K.; Richie, J.P.; Boyiri, T.; Komninou, D.; Prokopczyk, B.; Trushin, N.; Kleinman, W.; Cox, J.; Pittman, B.; Colosimo, S. Influence of Selenium-Enriched Yeast Supplementation on Biomarkers of Oxidative Damage and Hormone Status in Healthy Adult Males: A Clinical Pilot Study. Cancer Epidemiol. Biomark. Prev. 2002, 11, 1459–1465. [Google Scholar]
- Duffield-Lillico, A.J.; Reid, M.E.; Turnbull, B.W.; Combs, G.F.; Slate, E.H.; Fischbach, L.A.; Marshall, J.R.; Clark, L.C. Baseline Characteristics and the Effect of Selenium Supplementation on Cancer Incidence in a Randomized Clinical Trial: A Summary Report of the Nutritional Prevention of Cancer Trial. Cancer Epidemiol. Biomark. Prev. 2002, 11, 630–639. [Google Scholar]
- Combs, G.F.; Clark, L.C.; Turnbull, B.W. Reduction of Cancer Risk with an Oral Supplement of Selenium. Biomed. Env. Sci 1997, 10, 227–234. [Google Scholar]
- Hu, Y.; McIntosh, G.H.; Le Leu, R.K.; Young, G.P. Selenium-Enriched Milk Proteins and Selenium Yeast Affect Selenoprotein Activity and Expression Differently in Mouse Colon. Br. J. Nutr. 2010, 104, 17–23. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.; McIntosh, G.H.; Le Leu, R.K.; Upton, J.M.; Woodman, R.J.; Young, G.P. The Influence of Selenium-Enriched Milk Proteins and Selenium Yeast on Plasma Selenium Levels and Rectal Selenoprotein Gene Expression in Human Subjects. Br. J. Nutr. 2011, 106, 572–582. [Google Scholar] [CrossRef] [PubMed]
- Shang, D.; Li, Y.; Wang, C.; Wang, X.; Yu, Z.; Fu, X. A Novel Polysaccharide from Se-Enriched Ganoderma Lucidum Induces Apoptosis of Human Breast Cancer Cells. Oncol. Rep. 2011, 25, 267–272. [Google Scholar] [CrossRef]
- Sun, X.; Zhong, Y.; Luo, H.; Yang, Y. Selenium-Containing Polysaccharide-Protein Complex in Se-Enriched Ulva Fasciata Induces Mitochondria-Mediated Apoptosis in A549 Human Lung Cancer Cells. Mar. Drugs 2017, 15, 215. [Google Scholar] [CrossRef]
- Cheng, L.; Chen, L.; Yang, Q.; Wang, Y.; Wei, X. Antitumor Activity of Se-Containing Tea Polysaccharides against Sarcoma 180 and Comparison with Regular Tea Polysaccharides and Se-Yeast. Int. J. Biol. Macromol. 2018, 120, 853–858. [Google Scholar] [CrossRef]
- Luo, L.; Ran, R.; Yao, J.; Zhang, F.; Xing, M.; Jin, M.; Wang, L.; Zhang, T. Se-Enriched Cordyceps Militaris Inhibits Cell Proliferation, Induces Cell Apoptosis, And Causes G2/M Phase Arrest In Human Non-Small Cell Lung Cancer Cells. OTT 2019, 12, 8751–8763. [Google Scholar] [CrossRef]
- Lemire, M.; Philibert, A.; Fillion, M.; Passos, C.J.S.; Guimarães, J.R.D.; Barbosa, F.; Mergler, D. No Evidence of Selenosis from a Selenium-Rich Diet in the Brazilian Amazon. Environ. Int. 2012, 40, 128–136. [Google Scholar] [CrossRef]
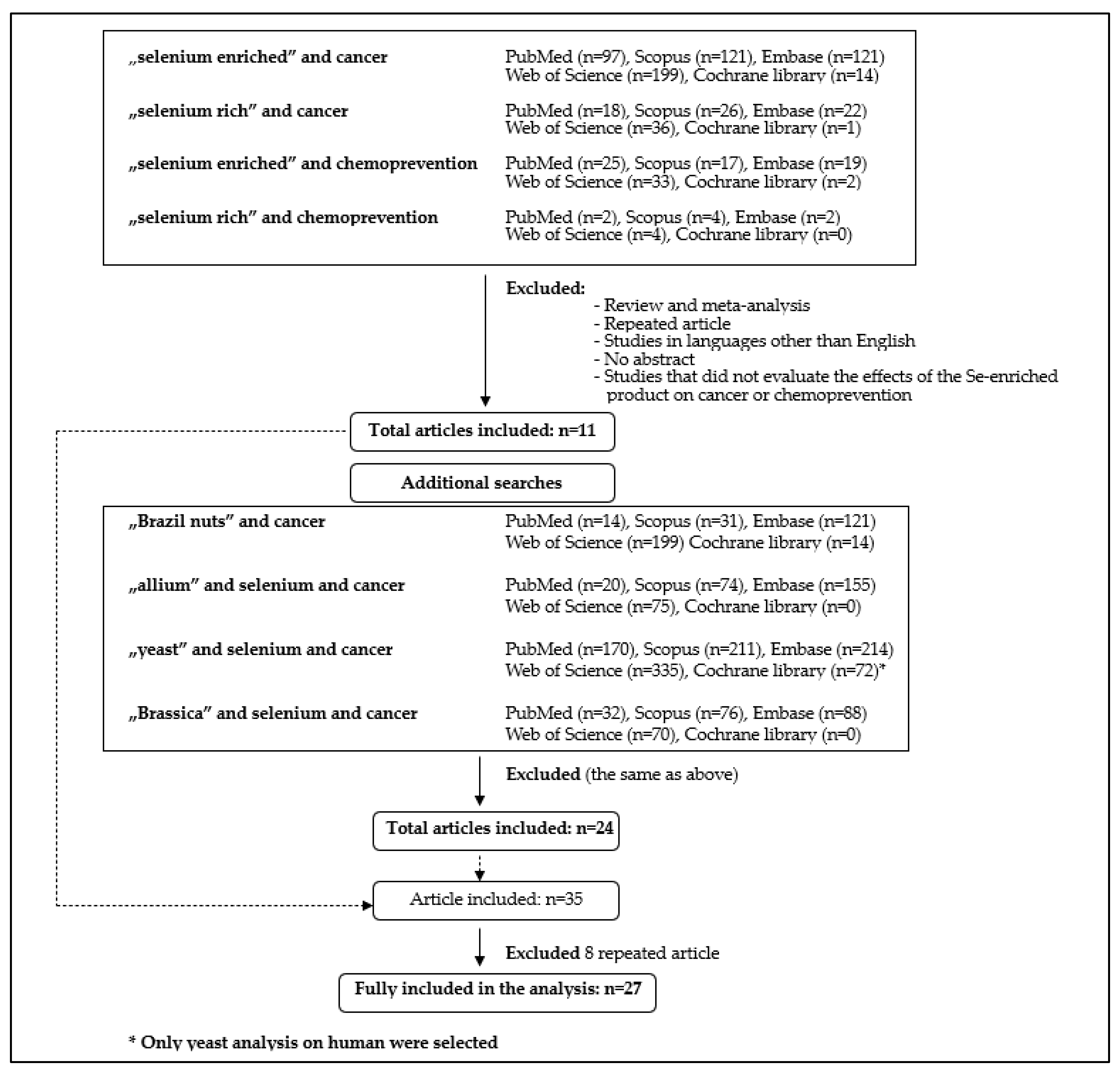
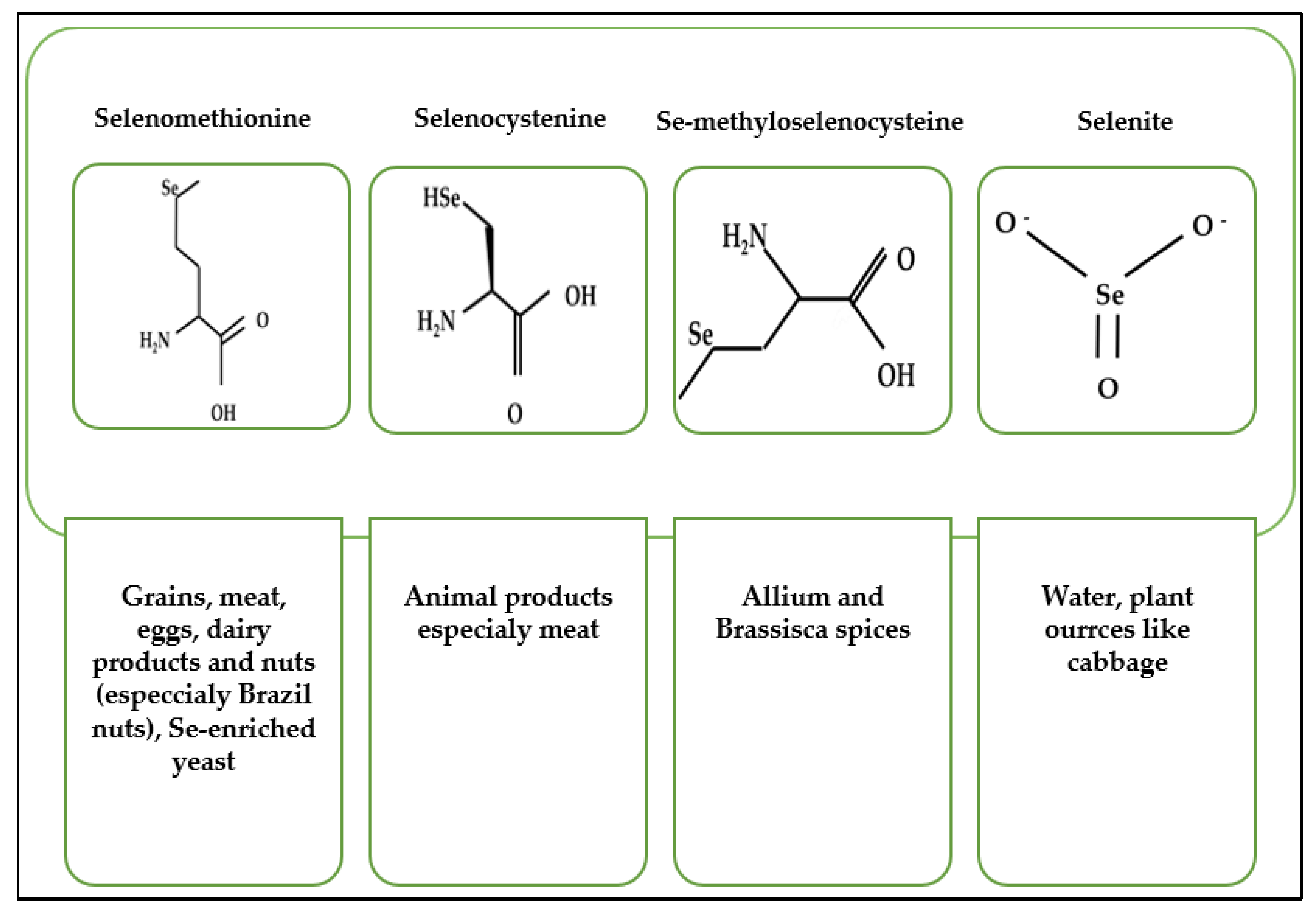
| Food Source | Average Content µg/g | Reference |
|---|---|---|
| Brazil nuts | 2−20 | Parekh P. P. et al., 2008 [76] |
| Garlic | 0.15 | Larsen E. H. et al., 2006 [77] |
| Broccoli | 0.13 * | De Temmerman et al., 2014 [78] |
| Brussels sprouts | 0.25 * | De Temmerman et al., 2014 [78] |
| Fish | 0.4−4.3 | Rayman M. P.et al., 2008 [27] |
| Meats (mussels) | 0.3 | Rayman M. P. et al., 2008 [27] |
| Yolk from egg | 0.12−0.42 | Pilarczyk B. et al., 2019 [79] |
| Form, Sources and Dose of Se | Period | Effect | Experimental Model | Reference |
|---|---|---|---|---|
| Two preparations of processed Brazil nuts Experiment 1: Final dietary Se levels 2 and 3 µg/g (from Brazil nuts content 16 µg Se/g. Experiment 2: Final dietary Se levels 1 and 2 µg/g (from Brazil nuts 30 µg Se/g) (dominant form of Se: MeSeCys) vs. Selenite—dietary Se content 1 and 2 µg/g | 2 weeks and 6 months (2 weeks before administration of 7,12-dimethylbenz(a)anthracene and 6 months after administration) | Mammary cancer: protection increased Se retention in the mammary gland, plasma, liver, and kidney Se in Brazil nuts and selenite are similarly bioactive | Pathogen-free female Sprague-Dawley rats | Ip C. et al., 1994 [90] |
| Two Brazil nuts Average 53 µg/g Se per day (possible range: 20–84 µg Se) Dominant form of Se: SeMet | 14 weeks | Increased plasma Se concentration and enhanced GPx activity | 59 New Zealand adults | Thomson C. D. et al., 2008 [89] |
| Brazil nuts and green extract, alone and in combination Six Brazil nuts Average 48 µg/g Se per day | 6 weeks | Colorectal cancer: regulated genes associated with selenoproteins, WNT signalling (β-catenin), inflammation (NF-κB), and methylation The combination of Brazil nuts and green extract does not provide additional effects compared with either agent alone | 61 adults aged 52–75 years | Hu Y. et al., 2016 [91] |
| Form, Sources and Dose of Se | Period | Effect | Experimental Model | Reference |
|---|---|---|---|---|
| Se-enriched broccoli 2 μg Se/g diet as high Se broccoli (selenised broccoli—Se concentration 500 µg/g) | 3 weeks | Colon cancer: decreases aberrant crypts and aberrant crypt foci | Fisher F-344 inbred rats, weanling males | Finley J. W. et al., 2000 [102] |
| Se-fertilised broccoli or broccoli florets First experiment: 3 μg of Se/g of diet Second experiment: 2 µg Se/g of diet | 22 weeks | Mammary cancer: significantly reduces the number of tumours Colon cancer: significantly fewer aberrant colon crypts | Pathogen-free female Sprague–Dawley rats | Finley J. W. et al., 2001 [103] |
| Se-enriched broccoli sprout (dominant form of Se: MeSeCys) 2.1 mg Se/kg diet or 0.11 mg Se/kg (control group) | 10 weeks | Intestinal cancer: small tumours and a smaller total tumour burden | Heterozygotic male Min mice | Davis C. D. et al., 2002 [104] |
| Se-enriched broccoli (dominant form of Se: MeSeCys) 2.1 mg Se/kg diet or 0.11 mg Se/kg (control diet) | 10 weeks | Intestinal cancer: activates specific pro-apoptotic genes linked to tumour | Heterozygotic male Min mice | Zeng H. et al., 2003 [105] |
| Se-enriched Japanese radish sprouts (dominant form of Se: MeSeCys) 8.8 µg of Se/in diet or under 1 µg of Se/g in a control diet | 13 weeks | Mammary cancer: significantly lowers the incidence of tumours in the Se-enriched sprout-added test diet group than in the basal diet group | Virgin female Sprague–Dawley strain rats | Yamanoshita O. et al., 2007 [107] |
| Se-enriched broccoli sprouts (dominant form of Se: MeSeCys) 24.2 μM | 72 h | Prostate cancer: inhibits cell proliferation, decreases prostate-specific antigen secretion, and induces apoptosis | In vitro model | Abdulah et al., 2009 [109] |
| Se-enriched broccoli extract 1.08 × 10−4 M Se and 2.50 × 10−7 M Se | 2 × 48 h | Colon cancer: inhibits the growth of HCT116 and HCT116+Chr.3 human colon cancer cells | In vitro model | Tsai C. F. et al., 2013 [106] |
| Se-fortified kale and kohlrabi sprouts (dominant form of Se: SeMet) 0.07−0.17 mg of Se/g dried weight | 24 h | Human metastatic cancer: cytotoxic effect on SW480, SW620, HepG2, SiHa cell lines (at ≥1 mg/mL sprouts extract) | In vitro model | Zagrodzki P. et al., 2020 [108] |
| Form, Sources and Dose of Se | Period | Effect | Experimental Model | Reference |
|---|---|---|---|---|
| Se-enriched garlic 150 µg/g Se (concentration in diet 3 µg/g Se) vs. Regular garlic 0.06 µg/g Se (concentration in diet 0.0012 µg/g Se) vs. Selenite 3 µg/g Se (in diet 3 µg/g Se) vs. Control group (dominant form of Se in Se-enriched garlic: MeSeCys) | 26 weeks | Mammary cancer: inhibited total tumour yield and tissue Se levels were lower in animals ingesting the Se-enriched garlic than selenite | Pathogen-free female Sprague–Dawley rats | Ip C. et al., 1992 [122] |
| Experiment I - Control (0.1 µg/g Se) - 0.85% regular garlic (0.1 µg/g Se) - 1.7% regular garlic (0.1 µg/g Se) 0.85% high Se garlic (1 µg/g Se) - 1.7% high Se garlic (2 µg/g Se) Experiment II - Control (0.1 µg/g Se) - 3.5% regular onion (0.1 µg/g Se) - 7% regular onion (0.1 µg/g Se) 3.5% high Se onion (1 µg/g Se) - 7% high Se onion (2 µg/g Se) (dominant form of Se in Se-enriched garlic: MeSeCys) | 8 months and 2 weeks | Mammary cancer: consumption does not cause excessive Se accumulation in tissues | Female Sprague–Dawley rats | Ip C. et al., 1994 [123] |
| Se-enriched garlic 112 µg/g and 1355 µg/g in dry weight (final concentration in diet 2 µg/g Se) vs. Control group 0.1 µg/g Se in diet (dominant form of Se in Se-enriched garlic: MeSeCys) | 3 weeks and 22 weeks | Mammary cancer: tumour reduction was due to the effect of Se not the effect of garlic | Pathogen-free female Sprague–Dawley rats | Ip C. et al., 1995 [124] |
| Se-enriched garlic diet concentration 3 µg/g Se vs. Sodium selenite diet concentration 3 µg/g Se vs. Control group 0.01 µg/g Se in diet (dominant form of Se in Se-enriched garlic: MeSeCys) | 7 weeks | Mammary cancer: inhibited total tumour yield, as well as the proliferation, survival and matrix degradation of endothelial cells critical for angiogenesis | Female Sprague–Dawley rats | Jiang C. et al., 1999 [121] |
| Se-enriched garlic (296 µg/g Se) and Se-enriched yeast (1922 µg/g Se) (final concentration in diet 1, 2 or 3 µg/g Se) vs. Control group 0.1 µg/g Se in diet (dominant form of Se in Se-enriched garlic: γ-glutamyl-MeSeCys) | 6 weeks | Mammary cancer: decreased morbidity and mortality Se-garlic was significantly more effective in suppressing the development of premalignant lesions and adenocarcinomas than Se-yeast despite Se-enriched yeast having a higher total tissue Se content | Pathogen-free female Sprague–Dawley rats | Ip C. et al., 2000 [125] |
| Form, Sources and Dose of Se | Period | Effect | Experimental Model | Reference |
|---|---|---|---|---|
| High-Se brewer’s yeast tablet (200 µg Se/day) vs. Placebo | 4.5 ± 2.8 years | Skin cancer: Se supplementation does not protect against the development of basal or squamous cell carcinomas but may reduce total cancer, lung, colorectal, and prostate cancer incidence, as well as lung cancer mortality | 1312 patients with a history of basal cell or symptoms of cell carcinomas, randomly assigned to the Se-treatment group (n = 653) and placebo group (n = 659); Mean age 63 years | Clark L. C. et al., 1996 [7] and Combs G. F. et al., 1997 [146] |
| Selenised yeast tablets 200 µg Se/day vs. Placebo | 4 years | Primary liver cancer: no primary liver cancer was observed in 113 people supplemented with Se during the 4-year study, while 7 of the placebo group were diagnosed with primary liver cancer | 226 participants randomly assigned to the study group (n = 113) and placebo group (n = 113); aged 21−63 years | Yu S. Y. et al., 1997 [141] |
| Se-enriched yeast 247 µg Se/day vs. Placebo | 9 months + 3 months placebo after supplementation | Prostate cancer: increased blood glutathione levels and significantly decreased prostate-specific antigen levels | 36 healthy adults randomly assigned to the study group (n = 17) and placebo group (n = 19); aged 19−43 years | El-Bayoumy K. et al., 2002 [144] |
| High-Se yeast (200 or 400 µg Se/day) vs. Placebo | patients in the United States—5 years, patients in New Zealand no more than 3 years | Prostate cancer: no significant differences in the time to prostate cancer diagnosis between placebo and study groups | 699 men at high risk for prostate cancer randomly assigned to 200 µg Se/day (n = 234), 400 µg Se/day (n = 233) or placebo group (n = 232); aged < 80 years | Algotar A. M. et al., 2013 [143] |
| Form, Sources and Dose of Se | Period | Effect | Experimental Model | Reference |
|---|---|---|---|---|
| Se-enriched milk proteins vs. Se-enriched yeast (dominant form of Se: SeMet) Four groups: - milk protein control diet (0.068 µg/g Se) - dairy-Se diet (0.5 µg/g Se) - dairy-Se diet (1 µg/g Se) - milk protein control + yeast-Se diet (1 µg/g Se) | 4 weeks | Colorectal cancer: Se-enriched milk regulates colonic GPx-2 and selenoprotein P mRNA expression | Male C57BL/6J mice | Hu Y. et al., 2010 [147] |
| Se-enriched milk protein (150 μg/d) vs. Se-enriched yeast (150 μg/d) (dominant form of Se: SeMet) | 6 weeks | Colorectal cancer: selenoprotein gene expression (selenoproteins P, GPx-1, GPx-2) was regulated by dietary Se independent of plasma Se levels, and GPx activity Se-enriched milk had a more sustained effect than Se-enriched yeast | 23 healthy volunteers randomly assigned to consume Se-enriched milk (n = 12) or Se-enriched yeast (n = 11); aged 52−79 years | Hu Y. et al., 2011 [148] |
| Se-enriched Ganoderma lucidum 0.045 to 0.36 μM SeGLP-2B-1 | 24, 48 or 72 h | Breast cancer: inhibited the growth of breast cancer cells in a time- and dose-dependent manner and increased caspase-9 and caspase-3 activity | MCF-7 human breast cancer cells | Shang D. et al., 2011 [149] |
| Se-enriched Ulva fasciata A549 cells were treated with 3, 4, 5 and 6 µg/mL | 72 h | Lung cancer: induced apoptosis (sub-G1 phase cells, upregulation of p53, and activation of caspase-3 in lung cancer cells) | A549 human lung cancer cells | Sun X et al., 2017 [150] |
| Se-containing tea polysaccharides (Se-TPS) from Se-enriched tea vs. Se-enriched yeast (Se 89 µg/g) Six groups: - control - Se-yeast 100 mg/kg - TPS 100 mg/kg - Se-TPS 50 mg/kg - Se-TPS 100 mg/kg - Se-TPS 200 mg/kg | 13 days | Murine sarcoma (S-180): Se-TPS significantly inhibited the proliferation of S-180 cells in a dose-dependent manner; in vivo, Se-TPS inhibited tumour growth in a dose-dependent manner | In vitro model and Kunming mice | Cheng L. et al., 2018 [151] |
| Se-enriched Cordyceps militaris NCI-H292 cells were treated with 0, 4, 8 and 12 mg/mL and A549 cells were treated with 0, 12.5, 25 and 50 mg/mL | 24 h | Lung cancer: cell poliferation, apoptosis in non-small cell lung cancer | Human lung cancer cell lines NCI-H292 and A549 | Luo L. et al., 2019 [152] |
| Products | Potential Chemopreventive Effect | Availability on the Market | Recommended Daily Dose * | Comments/References |
|---|---|---|---|---|
| Brazil nuts | Mammary and colorectal cancer | Easily accessible | 4 nuts 1 | - The Se content depends on the origin of the nuts - Excess may lead to toxic effects [89,94,153] |
| Se-enriched broccoli | Mammary, colon, intestinal, prostate, human metastatic cancer | Not yet available for sale Only dried broccoli powder with unspecified Se content is available | Probably Se-enriched broccoli powder 0.15−1 g (depending on the enrichment process) | - The Se content depends on the enrichment process - No human clinical trials with Se-enriched broccoli |
| Se-enriched garlic | Mammary cancer | Not yet available for sale Only powdered garlic with unspecified Se content is available | Probably Se-enriched broccoli powder 0.15−1 g (depending on the enrichment process) | - The Se content depends on the enrichment process - No human clinical trials with Se-enrich garlic |
| Se-enriched yeast | Skin cancer, primary liver cancer | Easily accessible | Around 0.5 g (depending on the enrichment process) | - No significant effect on prostate cancer in a clinical human study |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dobrzyńska, M.; Drzymała-Czyż, S.; Woźniak, D.; Drzymała, S.; Przysławski, J. Natural Sources of Selenium as Functional Food Products for Chemoprevention. Foods 2023, 12, 1247. https://doi.org/10.3390/foods12061247
Dobrzyńska M, Drzymała-Czyż S, Woźniak D, Drzymała S, Przysławski J. Natural Sources of Selenium as Functional Food Products for Chemoprevention. Foods. 2023; 12(6):1247. https://doi.org/10.3390/foods12061247
Chicago/Turabian StyleDobrzyńska, Małgorzata, Sławomira Drzymała-Czyż, Dagmara Woźniak, Sylwia Drzymała, and Juliusz Przysławski. 2023. "Natural Sources of Selenium as Functional Food Products for Chemoprevention" Foods 12, no. 6: 1247. https://doi.org/10.3390/foods12061247
APA StyleDobrzyńska, M., Drzymała-Czyż, S., Woźniak, D., Drzymała, S., & Przysławski, J. (2023). Natural Sources of Selenium as Functional Food Products for Chemoprevention. Foods, 12(6), 1247. https://doi.org/10.3390/foods12061247







