Impact of Quercetin against Salmonella Typhimurium Biofilm Formation on Food–Contact Surfaces and Molecular Mechanism Pattern
Abstract
:1. Introduction
2. Materials and Methods
2.1. Bacterial Strains and Culture Conditions
2.2. Preparation of Quercetin
2.3. Preparation of Samples (Food-Contact Surfaces)
2.4. Determination of Minimum Inhibitory Concentration (MIC)
2.5. Motility Assays
2.6. Biofilm Formation and Detachment
2.7. Field Emission Scanning Electron Microscopy (FE-SEM)
2.8. RNA Extraction, cDNA Synthesis, and Real-Time PCR (RT–PCR) Analysis
2.9. Statistical Analysis
3. Results
3.1. Determination of Minimum Inhibitory Concentration (MIC) and Sub-MIC
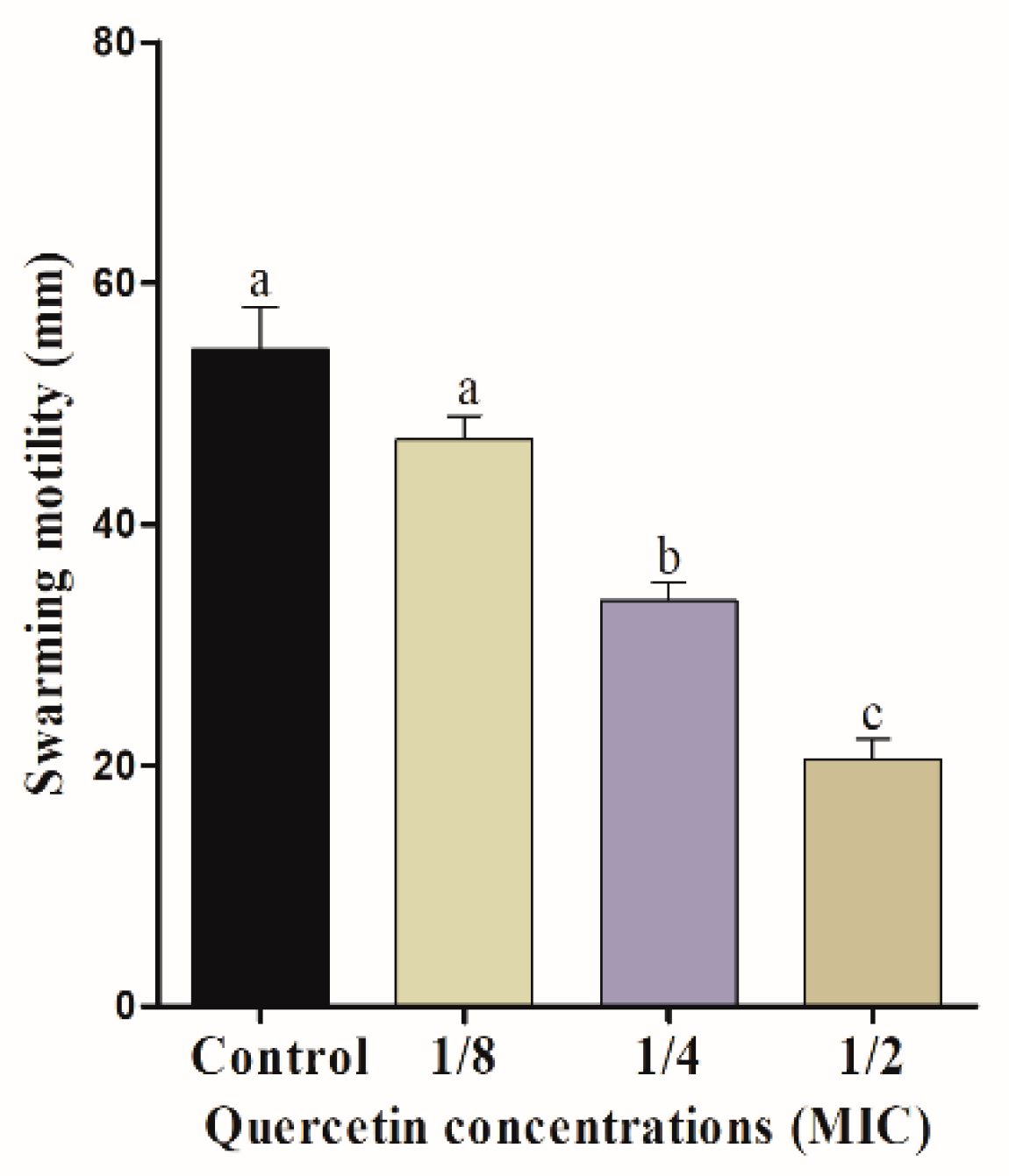
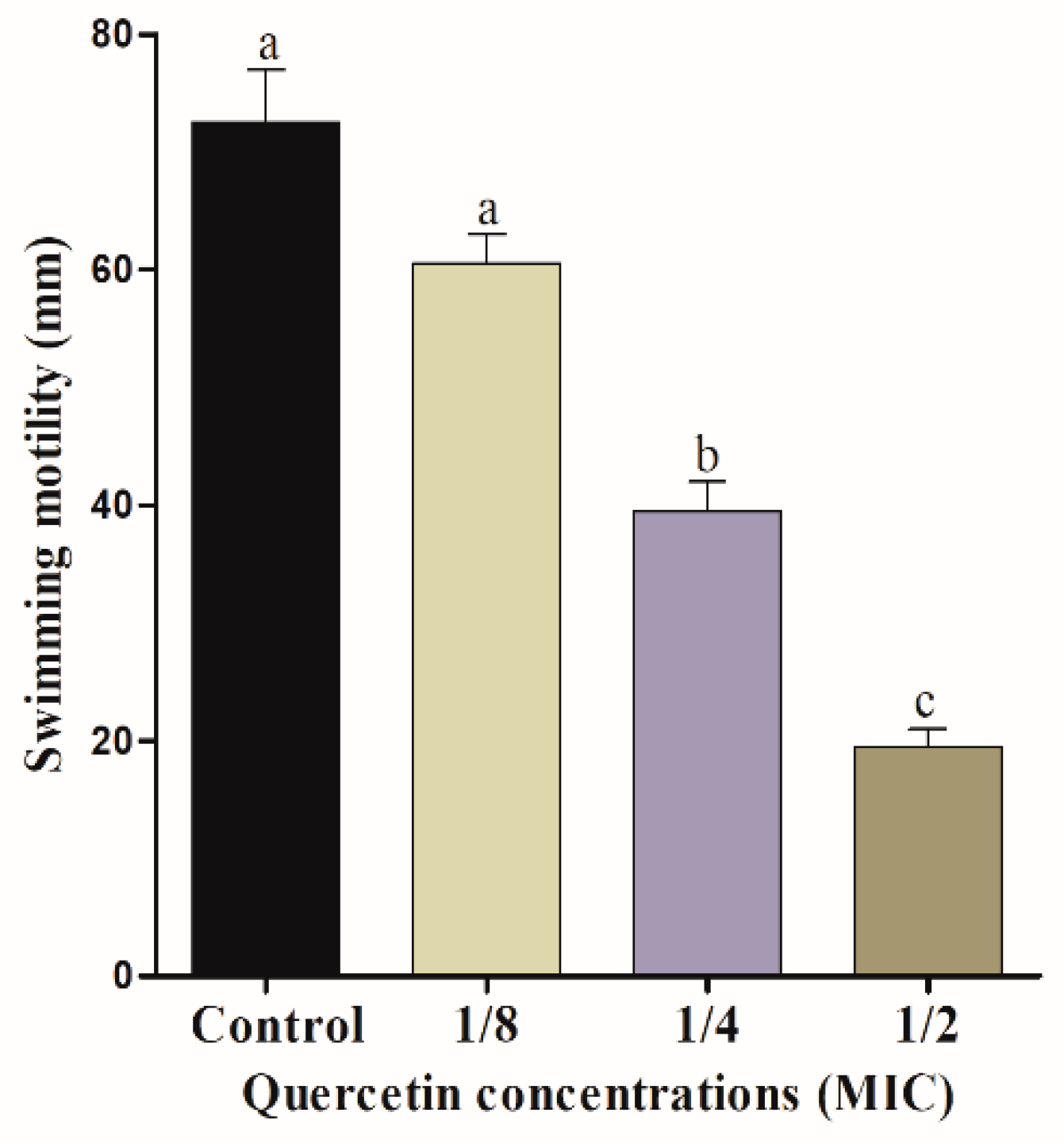
3.2. Motility Assays (Swimming and Swarming)
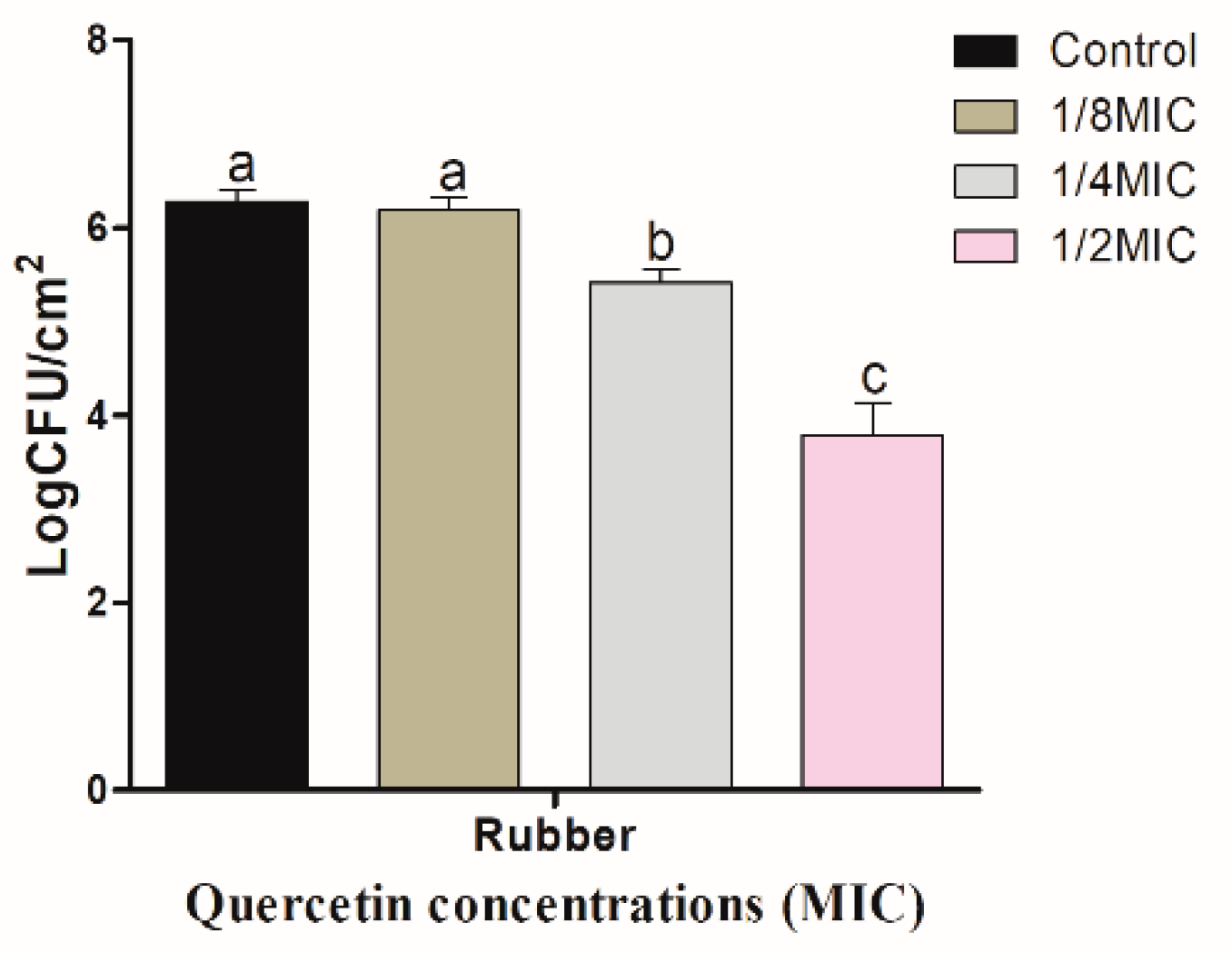
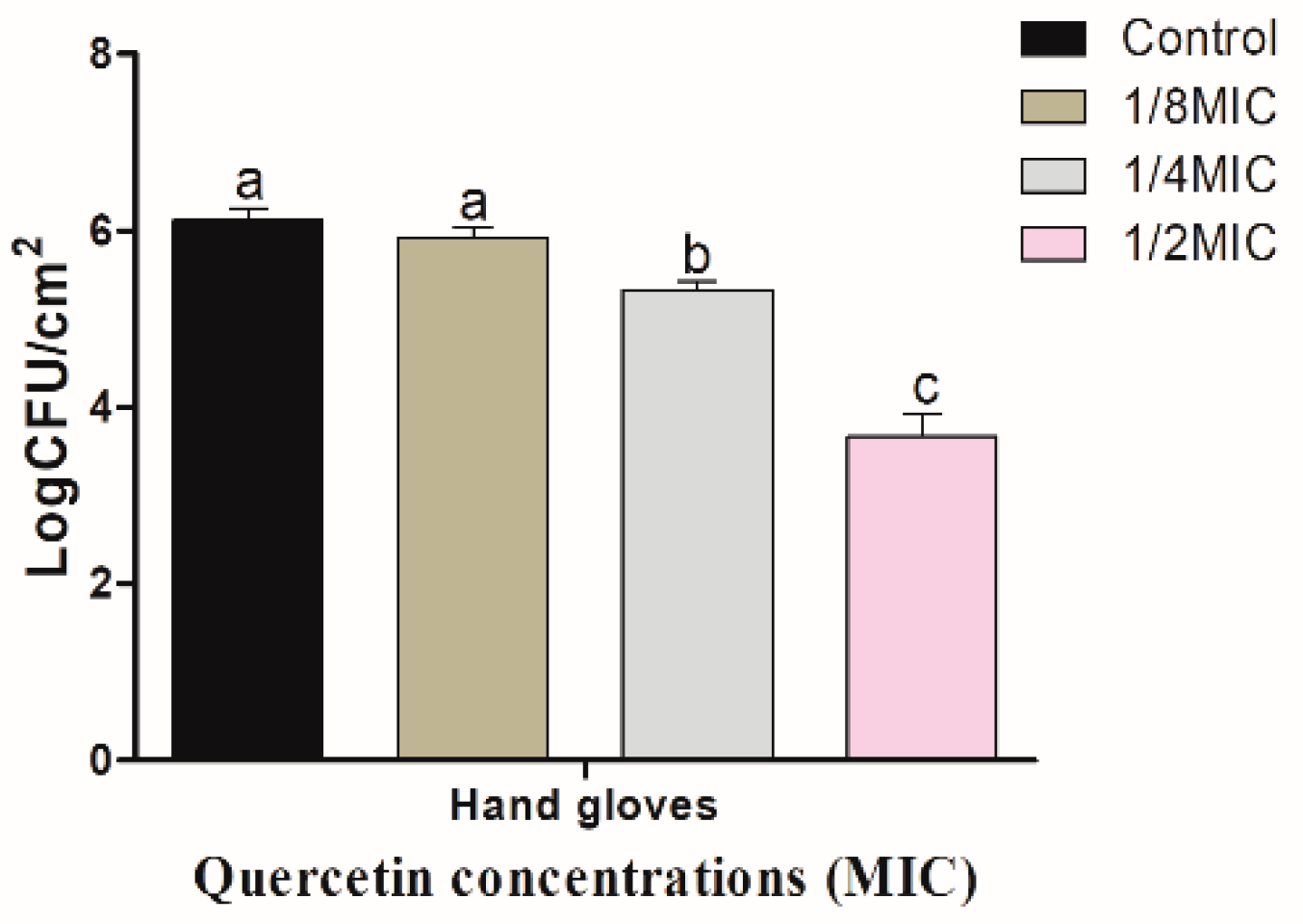
3.3. Inhibitory Effect of Quercetin against S. Typhimurium Biofilm on Food-Contact Surfaces (R and HG)
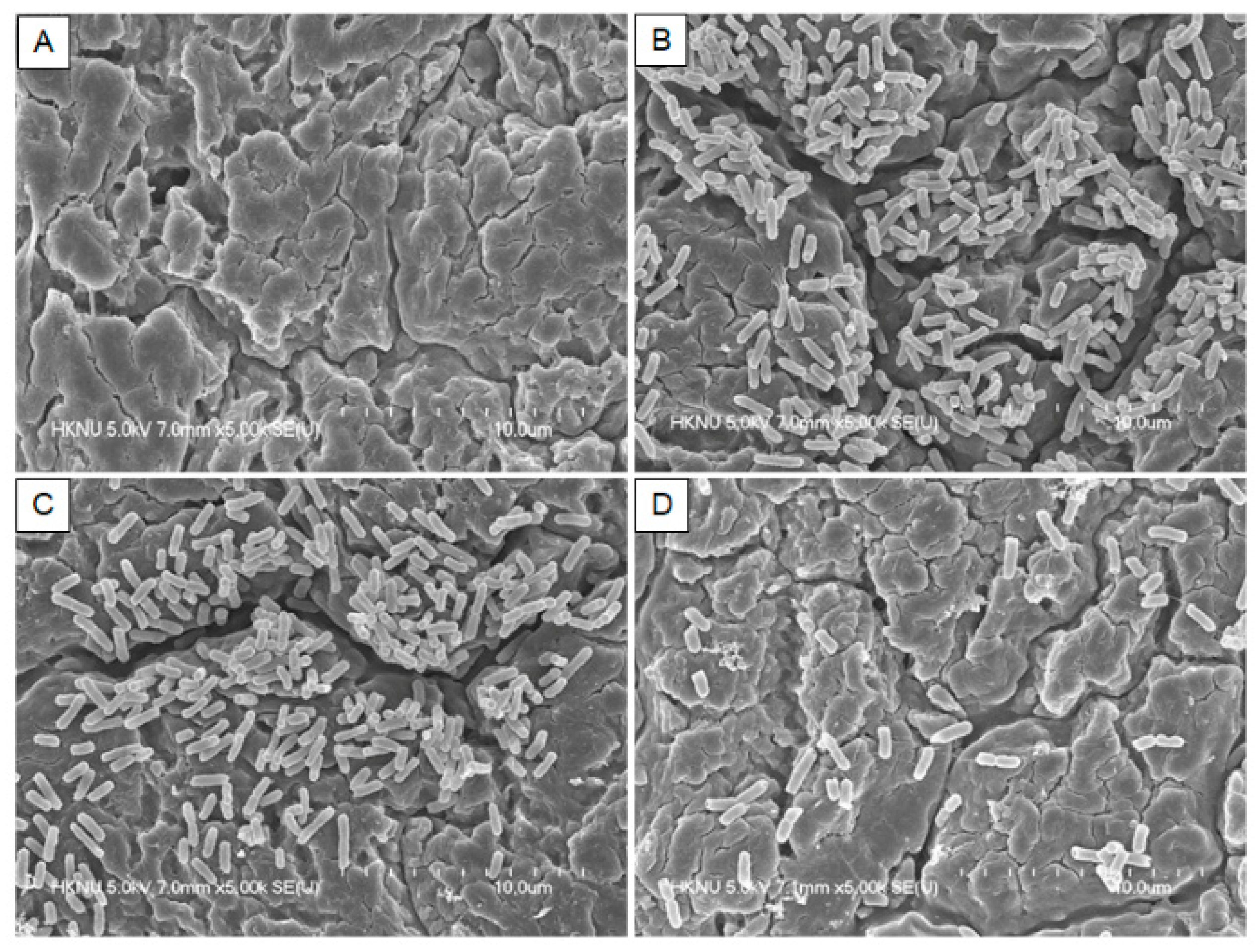
3.4. Field Emission Scanning Electron Microscopy (FE-SEM)
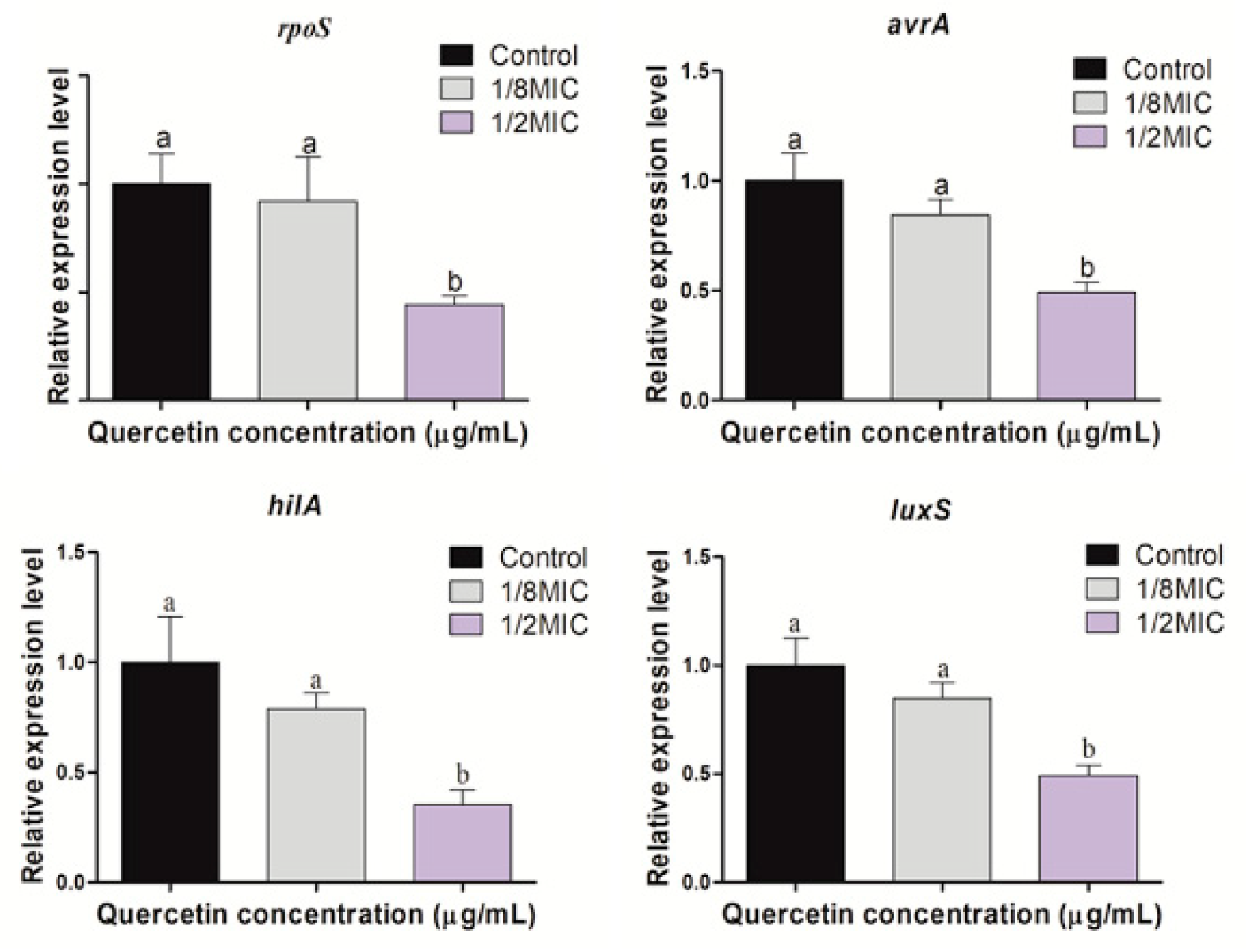
3.5. Virulence, Stress Response, and Quorum-Sensing Gene Expression
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Dantas, S.T.A.; Rossi, B.F.; Bonsaglia, E.C.R.; Castilho, I.G.; Hernandes, R.T.; Fernandes, A.; Rall, V.L.M. Cross-Contamination and Biofilm Formation by Salmonella enterica Serovar Enteritidis on Various Cutting Boards. Foodborne Pathog. Dis. 2018, 15, 81–85. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhao, X.H.; Zhao, F.H.; Wang, J.; Zhong, N.J. Biofilm formation and control strategies of foodborne pathogens: Food safety perspectives. RSC Adv. 2017, 7, 36670–36683. [Google Scholar] [CrossRef] [Green Version]
- CDC. 2016. Available online: https://www.cdc.gov/salmonella/index.html (accessed on 15 January 2021).
- Roy, P.K.; Ha, A.J.; Mizan, M.F.R.; Hossain, M.I.; Ashrafudoulla, M.; Toushik, S.H.; Nahar, S.; Kim, Y.K.; Ha, S.-D. Effects of environmental conditions (temperature, pH, and glucose) on biofilm formation of Salmonella enterica serotype Kentucky and virulence gene expression. Poult. Sci. 2021, 100, 101209. [Google Scholar] [CrossRef]
- Nahar, S.; Mizan, M.F.R.; Ha, A.J.; Ha, S.-D. Advances and Future Prospects of Enzyme-Based Biofilm Prevention Approaches in the Food Industry. Compr. Rev. Food Sci. Food Saf. 2018, 17, 1484–1502. [Google Scholar] [CrossRef] [Green Version]
- Hossain, I.; Mizan, F.R.; Roy, P.K.; Nahar, S.; Toushik, S.H.; Ashrafudoulla, M.; Jahid, I.K.; Lee, J.; Ha, S.-D. Listeria monocytogenes biofilm inhibition on food contact surfaces by application of postbiotics from Lactobacillus curvatus B.67 and Lactobacillus plantarum M.2. Food Res. Int. 2021, 148, 110595. [Google Scholar] [CrossRef]
- Toushik, S.H.; Kim, K.; Ashrafudoulla, M.; Mizan, M.F.R.; Roy, P.K.; Nahar, S.; Kim, Y.; Ha, S.-D. Korean kimchi-derived lactic acid bacteria inhibit foodborne pathogenic biofilm growth on seafood and food processing surface materials. Food Control 2021, 129, 108276. [Google Scholar] [CrossRef]
- Hossain, M.I.; Kim, K.; Mizan, M.F.R.; Toushik, S.H.; Ashrafudoulla, M.; Roy, P.K.; Nahar, S.; Jahid, I.K.; Choi, C.; Park, S.H.; et al. Comprehensive molecular, probiotic, and quorum-sensing characterization of anti-listerial lactic acid bacteria, and application as bioprotective in a food (milk) model. J. Dairy Sci. 2021, 104, 6516–6534. [Google Scholar] [CrossRef] [PubMed]
- Suo, Y.J.; Huang, Y.Y.; Liu, Y.H.; Shi, C.L.; Shi, X.M. The Expression of Superoxide Dismutase (SOD) and a Putative ABC Transporter Permease Is Inversely Correlated during Biofilm Formation in Listeria monocytogenes 4b G. PLoS ONE 2012, 7, e48467. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, K.-H.; Lee, J.-Y.; Roy, P.K.; Mizan, M.F.R.; Hossain, M.I.; Park, S.H.; Ha, S.-D. Viability of Salmonella Typhimurium biofilms on major food-contact surfaces and eggshell treated during 35 days with and without water storage at room temperature. Poult. Sci. 2020, 99, 4558–4565. [Google Scholar] [CrossRef] [PubMed]
- Nahar, S.; Ha, A.J.-W.; Byun, K.-H.; Hossain, M.I.; Mizan, M.F.R.; Ha, S.-D. Efficacy of flavourzyme against Salmonella Typhimurium, Escherichia coli, and Pseudomonas aeruginosa biofilms on food-contact surfaces. Int. J. Food Microbiol. 2020, 336, 108897. [Google Scholar] [CrossRef]
- Nickerson, C.A.; Curtiss, R. Role of sigma factor RpoS in initial stages of Salmonella typhimurium infection. Infect. Immun. 1997, 65, 1814–1823. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, H.H.; Dong, Y.; Wang, G.Y.; Xu, X.L.; Zhou, G.H. Effect of growth media on gene expression levels in Salmonella Typhimurium biofilm formed on stainless steel surface. Food Control 2016, 59, 546–552. [Google Scholar] [CrossRef]
- Li, X.; Wu, B.; Chen, H.; Nan, K.H.; Jin, Y.Y.; Sun, L.; Wang, B. Recent developments in smart antibacterial surfaces to inhibit biofilm formation and bacterial infections. J. Mater. Chem. B 2018, 6, 4274–4292. [Google Scholar] [CrossRef] [PubMed]
- Gambino, M.; Cappitelli, F. Mini-review: Biofilm responses to oxidative stress. Biofouling 2016, 32, 167–178. [Google Scholar] [CrossRef] [PubMed]
- Skandamis, P.N.; Nychas, G.-J.E. Quorum Sensing in the Context of Food Microbiology. Appl. Environ. Microbiol. 2012, 78, 5473–5482. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Quecan, B.X.V.; Santos, J.T.C.; Rivera, M.L.C.; Hassimotto, N.M.A.; de Almeida, F.A.; Pinto, U. Effect of Quercetin Rich Onion Extracts on Bacterial Quorum Sensing. Front. Microbiol. 2019, 10, 867. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Martins, M.; Pinto, U.M.; Riedel, K.; Vanetti, M.C.D. Quorum Sensing and Spoilage Potential of Psychrotrophic Enterobacteriaceae Isolated from Milk. BioMed Res. Int. 2018, 2018, 2723157. [Google Scholar] [CrossRef] [Green Version]
- Martínez-Flórez, S.; González-Gallego, J.; Culebras, J.M.; Tuñón, M.J. Flavonoids: Properties and anti-oxidizing action. Nutr. Hosp. 2003, 17, 271–278. [Google Scholar]
- Rodrigues, A.C.; De Oliveira, B.D.; Da Silva, E.R.; Sacramento, N.T.B.; Bertoldi, M.C.; Pinto, U.M. Anti-quorum sensing activity of phenolic extract from Eugenia brasiliensis (Brazilian cherry). Food Sci. Technol. 2016, 36, 337–343. [Google Scholar] [CrossRef] [Green Version]
- Mutha, R.E.; Tatiya, A.U.; Surana, S.J. Flavonoids as natural phenolic compounds and their role in therapeutics: An overview. Future J. Pharm. Sci. 2021, 7, 25. [Google Scholar] [CrossRef]
- Cichon, N.; Saluk-Bijak, J.; Gorniak, L.; Przyslo, L.; Bijak, M. Flavonoids as a Natural Enhancer of Neuroplasticity—An Overview of the Mechanism of Neurorestorative Action. Antioxidants 2020, 9, 1035. [Google Scholar] [CrossRef] [PubMed]
- Panche, A.N.; Diwan, A.D.; Chandra, S.R. Flavonoids: An overview. J. Nutr. Sci. 2016, 5, e47. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ortega-Vidal, J.; Cobo, A.; Ortega-Morente, E.; Gálvez, A.; Alejo-Armijo, A.; Salido, S.; Altarejos, J. Antimicrobial and antioxidant activities of flavonoids isolated from wood of sweet cherry tree (Prunus avium L.). J. Wood Chem. Technol. 2021, 41, 104–117. [Google Scholar] [CrossRef]
- Osonga, F.J.; Akgul, A.; Miller, R.M.; Eshun, G.B.; Yazgan, I.; Akgul, A.; Sadik, O.A. Antimicrobial Activity of a New Class of Phosphorylated and Modified Flavonoids. ACS Omega 2019, 4, 12865–12871. [Google Scholar] [CrossRef] [Green Version]
- Vandeputte, O.M.; Kiendrebeogo, M.; Rasamiravaka, T.; Stévigny, C.; Duez, P.; Rajaonson, S.; Diallo, B.; Mol, A.; Baucher, M.; El Jaziri, M. The flavanone naringenin reduces the production of quorum sensing-controlled virulence factors in Pseudomonas aeruginosa PAO1. Microbiology 2011, 157, 2120–2132. [Google Scholar] [CrossRef] [Green Version]
- Paczkowski, J.E.; Mukherjee, S.; McCready, A.R.; Cong, J.-P.; Aquino, C.J.; Kim, H.; Henke, B.R.; Smith, C.D.; Bassler, B.L. Flavonoids Suppress Pseudomonas aeruginosa Virulence through Allosteric Inhibition of Quorum-sensing Receptors. J. Biol. Chem. 2017, 292, 4064–4076. [Google Scholar] [CrossRef] [Green Version]
- He, Z.Y.; Zhang, X.; Song, Z.C.; Li, L.; Chang, H.S.; Li, S.L.; Zhou, W. Quercetin inhibits virulence properties of Porphyromas gingivalis in periodontal disease. Sci. Rep. 2020, 10, 18313. [Google Scholar] [CrossRef]
- Sarangapani, S.; Jayachitra, A. Targeting biofilm inhibition using Quercetin—Interaction with bacterial cell membrane and ROS mediated biofilm control. Funct. Foods Health Dis. 2018, 8, 292–306. [Google Scholar] [CrossRef]
- Mu, Y.; Zeng, H.; Chen, W. Quercetin Inhibits Biofilm Formation by Decreasing the Production of EPS and Altering the Composition of EPS in Staphylococcus epidermidis. Front. Microbiol. 2021, 12, 1058. [Google Scholar] [CrossRef]
- Ong, K.S.; Mawang, C.I.; Daniel-Jambun, D.; Lim, Y.Y.; Lee, S.M. Current anti-biofilm strategies and potential of antioxidants in biofilm control. Expert Rev. Anti-Infect. Ther. 2018, 16, 855–864. [Google Scholar] [CrossRef]
- Amin, M.U.; Khurram, M.; Khattak, B.; Khan, J. Antibiotic additive and synergistic action of rutin, morin and quercetin against methicillin resistant Staphylococcus aureus. BMC Complement. Altern. Med. 2015, 15, 59. [Google Scholar] [CrossRef] [Green Version]
- Ouyang, J.; Sun, F.; Feng, W.; Sun, Y.; Qiu, X.; Xiong, L.; Liu, Y.; Chen, Y. Quercetin is an effective inhibitor of quorum sensing, biofilm formation and virulence factors in Pseudomonas aeruginosa. J. Appl. Microbiol. 2016, 120, 966–974. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, S.N.; Yao, J.Y.; Zhou, B.; Yang, J.X.; Chaudry, M.T.; Wang, M.; Xiao, F.L.; Li, Y.; Yin, W.Z. Bacteriostatic Effect of Quercetin as an Antibiotic Alternative In Vivo and Its Antibacterial Mechanism In Vitro. J. Food Prot. 2018, 81, 68–78. [Google Scholar] [CrossRef]
- Ashrafudoulla, M.; Mizan, M.F.R.; Ha, A.J.-W.; Park, S.H.; Ha, S.-D. Antibacterial and antibiofilm mechanism of eugenol against antibiotic resistance Vibrio parahaemolyticus. Food Microbiol. 2020, 91, 103500. [Google Scholar] [CrossRef] [PubMed]
- Mizan, M.F.R.; Jahid, I.K.; Kim, M.; Lee, K.-H.; Kim, T.J.; Ha, S.-D. Variability in biofilm formation correlates with hydrophobicity and quorum sensing amongVibrio parahaemolyticusisolates from food contact surfaces and the distribution of the genes involved in biofilm formation. Biofouling 2016, 32, 497–509. [Google Scholar] [CrossRef]
- Byun, K.-H.; Han, S.H.; Yoon, J.-W.; Park, S.H.; Ha, S.-D. Efficacy of chlorine-based disinfectants (sodium hypochlorite and chlorine dioxide) on Salmonella Enteritidis planktonic cells, biofilms on food contact surfaces and chicken skin. Food Control 2021, 123, 107838. [Google Scholar] [CrossRef]
- Ashrafudoulla, M.; Na, K.W.; Byun, K.-H.; Kim, D.H.; Yoon, J.W.; Mizan, M.F.R.; Kang, I.; Ha, S.-D. Isolation and characterization of Salmonella spp. from food and food contact surfaces in a chicken processing factory. Poult. Sci. 2021, 100, 101234. [Google Scholar] [CrossRef]
- Roy, P.K.; Qamar, A.Y.; Fang, X.; Hassan, B.M.S.; Cho, J. Effects of cobalamin on meiotic resumption and developmental competence of growing porcine oocytes. Theriogenology 2020, 154, 24–30. [Google Scholar] [CrossRef]
- Roy, P.K.; Qamar, A.Y.; Fang, X.; Kim, G.; Bang, S.; De Zoysa, M.; Shin, S.T.; Cho, J. Chitosan nanoparticles enhance developmental competence of in vitro-matured porcine oocytes. Reprod. Domest. Anim. 2021, 56, 342–350. [Google Scholar] [CrossRef] [PubMed]
- Cho, J.; Kim, G.; Qamar, A.Y.; Fang, X.; Roy, P.K.; Tanga, B.M.; Bang, S.; Kim, J.K.; Galli, C.; Perota, A.; et al. Improved efficiencies in the generation of multigene-modified pigs by recloning and using sows as the recipient. Zygote 2022, 30, 103–110. [Google Scholar] [CrossRef] [PubMed]
- Roy, P.-K.; Qamar, A.-Y.; Tanga, B.-M.; Bang, S.; Seong, G.; Fang, X.; Kim, G.; Edirisinghe, S.-L.; De Zoysa, M.; Kang, D.-H.; et al. Modified Spirulina maxima Pectin Nanoparticles Improve the Developmental Competence of In Vitro Matured Porcine Oocytes. Animals 2021, 11, 2483. [Google Scholar] [CrossRef] [PubMed]
- Roy, P.K.; Qamar, A.Y.; Tanga, B.M.; Fang, X.; Kim, G.; Bang, S.; Cho, J. Enhancing Oocyte Competence With Milrinone as a Phosphodiesterase 3A Inhibitor to Improve the Development of Porcine Cloned Embryos. Front. Cell Dev. Biol. 2021, 9, 647616. [Google Scholar] [CrossRef] [PubMed]
- Kim, G.; Roy, P.K.; Fang, X.; Hassan, B.M.; Cho, J. Improved preimplantation development of porcine somatic cell nuclear transfer embryos by caffeine treatment. J. Vet. Sci. 2019, 20, e31. [Google Scholar] [CrossRef]
- Braga, P.C.; Sasso, M.D.; Sala, M.T. Sub-MIC concentrations of cefodizime interfere with various factors affecting bacterial virulence. J. Antimicrob. Chemother. 2000, 45, 15–25. [Google Scholar] [CrossRef] [PubMed]
- Ha, D.G.; Kuchma, S.L.; O’Toole, G.A. Plate-based assay for swimming motility in Pseudomonas aeruginosa. In Pseudomonas Methods and Protocols; Humana Press: New York, NY, USA, 2014; pp. 59–65. [Google Scholar]
- Murray, T.S.; Ledizet, M.; Kazmierczak, B.I. Swarming motility, secretion of type 3 effectors and biofilm formation phenotypes exhibited within a large cohort of Pseudomonas aeruginosa clinical isolates. J. Med. Microbiol. 2010, 59, 511–520. [Google Scholar] [CrossRef]
- Kim, Y.K.; Roy, P.K.; Ashrafudoulla; Nahar, S.; Toushik, S.H.; Hossain, M.I.; Mizan, M.F.R.; Park, S.H.; Ha, S.-D. Antibiofilm effects of quercetin against Salmonella enterica biofilm formation and virulence, stress response, and quorum-sensing gene expression. Food Control 2022, 137, 108964. [Google Scholar] [CrossRef]
- Vazquez-Armenta, F.J.; Bernal-Mercado, A.T.; Rodriguez, M.R.T.; Gonzalez-Aguilar, G.A.; Lopez-Zavala, A.A.; Martínez-Téllez, M.A.; Hernandez-Onate, M.A.; Ayala-Zavala, J.F. Quercetin reduces adhesion and inhibits biofilm development by Listeria monocytogenes by reducing the amount of extracellular proteins. Food Control 2018, 90, 266–273. [Google Scholar] [CrossRef]
- Stepanovic, S.; Cirkovic, I.; Ranin, L.; Svabic-Vlahovic, M. Biofilm formation by Salmonella spp. and Listeria monocytogenes on plastic surface. Lett. Appl. Microbiol. 2004, 38, 428–432. [Google Scholar] [CrossRef] [PubMed]
- Sinde, E.; Carballo, J. Attachment of Salmonella spp. and Listeria monocytogenes to stainless steel, rubber and polytetrafluorethylene: The influence of free energy and the effect of commercial sanitizers. Food Microbiol. 2000, 17, 439–447. [Google Scholar] [CrossRef]
- Gopu, V.; Meena, C.K.; Shetty, P.H. Quercetin Influences Quorum Sensing in Food Borne Bacteria: In-Vitro and In-Silico Evidence. PLoS ONE 2015, 10, e0134684. [Google Scholar] [CrossRef] [Green Version]
- El-Khawagah, A.R.M.; Kandiel, M.M.M.; Samir, H. Effect of Quercetin Supplementation in Extender on Sperm Kinematics, Extracellular Enzymes Release, and Oxidative Stress of Egyptian Buffalo Bulls Frozen–Thawed Semen. Front. Veter- Sci. 2020, 7, 604460. [Google Scholar] [CrossRef] [PubMed]
- Rojas-Lema, S.; Torres-Giner, S.; Quiles-Carrillo, L.; Gomez-Caturla, J.; Garcia-Garcia, D.; Balart, R. On the Use of Phenolic Compounds Present in Citrus Fruits and Grapes as Natural Antioxidants for Thermo-Compressed Bio-Based High-Density Polyethylene Films. Antioxidants 2021, 10, 14. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.-P.; Li, L.; Shen, J.-Z.; Yang, F.-J.; Wu, Y.-N. Quinolone-resistance in Salmonella is associated with decreased mRNA expression of virulence genes invA and avrA, growth and intracellular invasion and survival. Vet. Microbiol. 2009, 133, 328–334. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.J.; Brisbin, J.; Yu, H.; Wang, Q.; Yin, F.G.; Zhang, Y.G.; Sabour, P.; Sharif, S.; Gong, J. Selected Lactic Acid-Producing Bacterial Isolates with the Capacity to Reduce Salmonella Translocation and Virulence Gene Expression in Chickens. PLoS ONE 2014, 9, e93022. [Google Scholar] [CrossRef]
- Bajaj, V.; Hwang, C.; Lee, C.A. hilA is a novel ompR/toxR family member that activates the expression of Salmonella typhimurium invasion genes. Mol. Microbiol. 1995, 18, 715–727. [Google Scholar] [CrossRef]
- Liu, X.Y.; Lu, R.; Xia, Y.L.; Wu, S.P.; Sun, J. Eukaryotic signaling pathways targeted by Salmonella effector protein AvrA in intestinal infection in vivo. BMC Microbiol. 2010, 10, 326. [Google Scholar] [CrossRef] [Green Version]
- Aswathanarayan, J.B.; Vittal, R.R. Inhibition of biofilm formation and quorum sensing mediated phenotypes by berberine in Pseudomonas aeruginosa and Salmonella typhimurium. RSC Adv. 2018, 8, 36133–36141. [Google Scholar] [CrossRef] [Green Version]
- Waters, C.M.; Bassler, B.L. QUORUM SENSING: Cell-to-Cell Communication in Bacteria. Annu. Rev. Cell Dev. Biol. 2005, 21, 319–346. [Google Scholar] [CrossRef] [Green Version]
- Gonzalez, J.E.; Keshavan, N.D. Messing with Bacterial Quorum Sensing. Microbiol. Mol. Biol. Rev. 2006, 70, 859–875. [Google Scholar] [CrossRef] [Green Version]
| Target Primers | Sequence (5′-3′) | Product Size (bp) |
|---|---|---|
| 16S rRNA | F: CAGAAGAAGCACCGGCTAAC R: GACTCAAGCCTGCCAGTTTC | 167 |
| rpoS | F: GAATCTGACGAACACGCTCA R: CCACGCAAGATGACGATATG | 171 |
| avrA | F: GAGCTGCTTTGGTCCTCAAC R: AATGGAAGGCGTTGAATCTG | 173 |
| hilA | F: ATTAAGGCGACAGAGCTGGA R: GCAGAAATGGGCGAAAGTAA | 134 |
| LuxS | F: CGGGTTGCAAAAACGATGA R: GTTGAGGTGGTCGCGCATA | 150 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Roy, P.K.; Song, M.G.; Park, S.Y. Impact of Quercetin against Salmonella Typhimurium Biofilm Formation on Food–Contact Surfaces and Molecular Mechanism Pattern. Foods 2022, 11, 977. https://doi.org/10.3390/foods11070977
Roy PK, Song MG, Park SY. Impact of Quercetin against Salmonella Typhimurium Biofilm Formation on Food–Contact Surfaces and Molecular Mechanism Pattern. Foods. 2022; 11(7):977. https://doi.org/10.3390/foods11070977
Chicago/Turabian StyleRoy, Pantu Kumar, Min Gyu Song, and Shin Young Park. 2022. "Impact of Quercetin against Salmonella Typhimurium Biofilm Formation on Food–Contact Surfaces and Molecular Mechanism Pattern" Foods 11, no. 7: 977. https://doi.org/10.3390/foods11070977
APA StyleRoy, P. K., Song, M. G., & Park, S. Y. (2022). Impact of Quercetin against Salmonella Typhimurium Biofilm Formation on Food–Contact Surfaces and Molecular Mechanism Pattern. Foods, 11(7), 977. https://doi.org/10.3390/foods11070977







