Applying Computational Fluid Dynamics in the Development of Smart Ripening Rooms for Traditional Cheeses
Abstract
1. Introduction
2. Materials and Methods
2.1. Cheesemaking
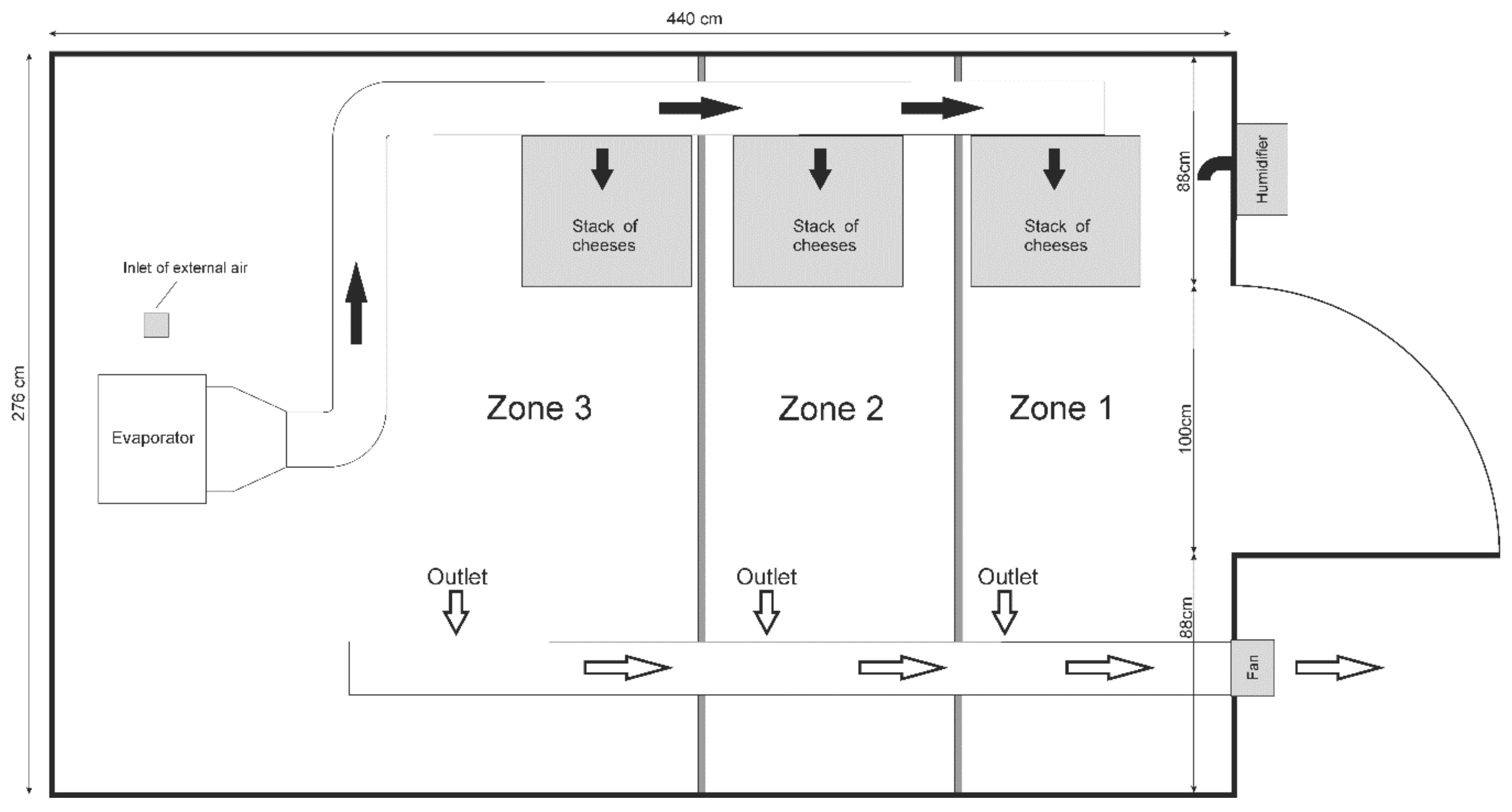
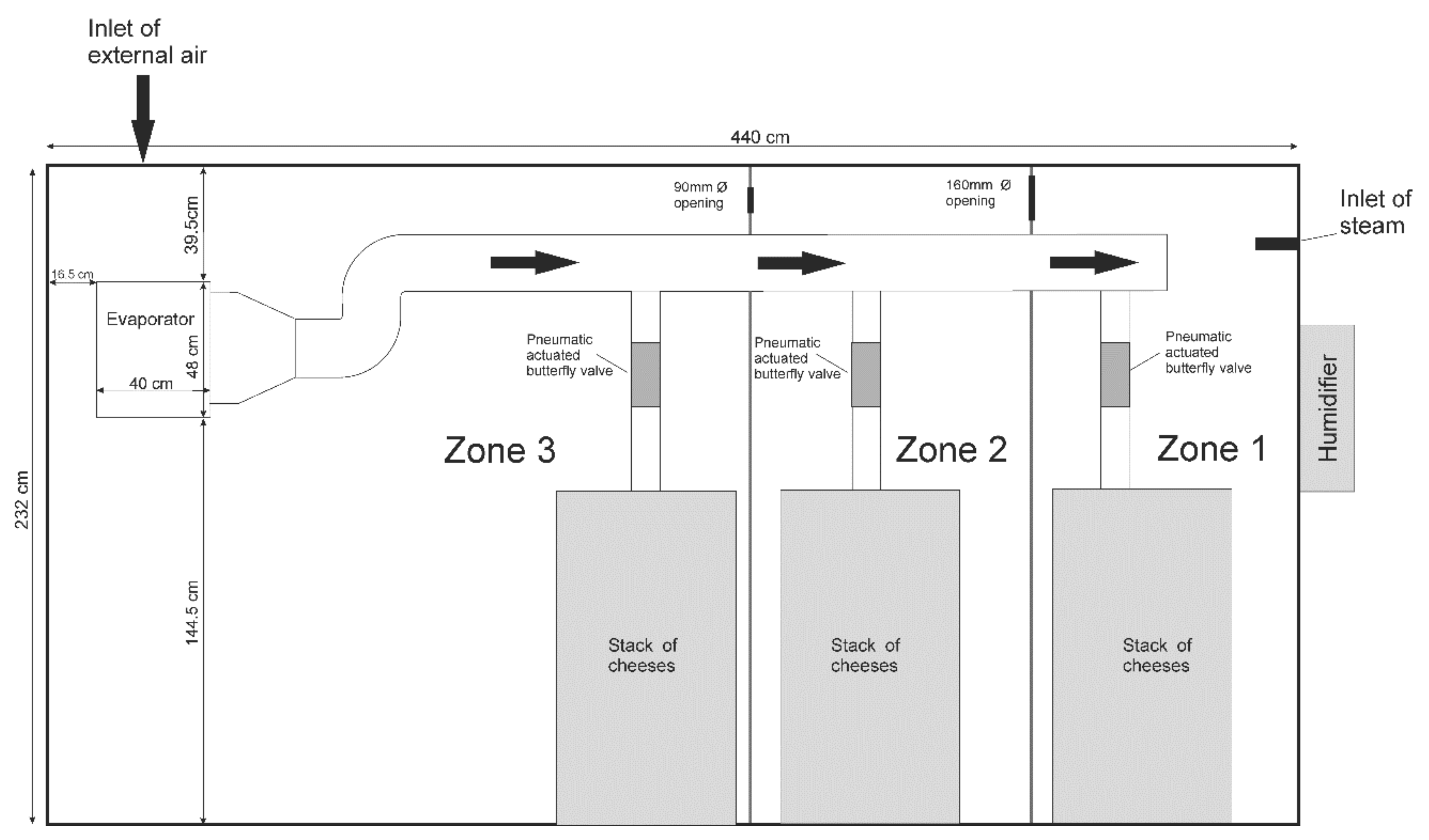
2.2. Instrumentation of the Ripening Chamber
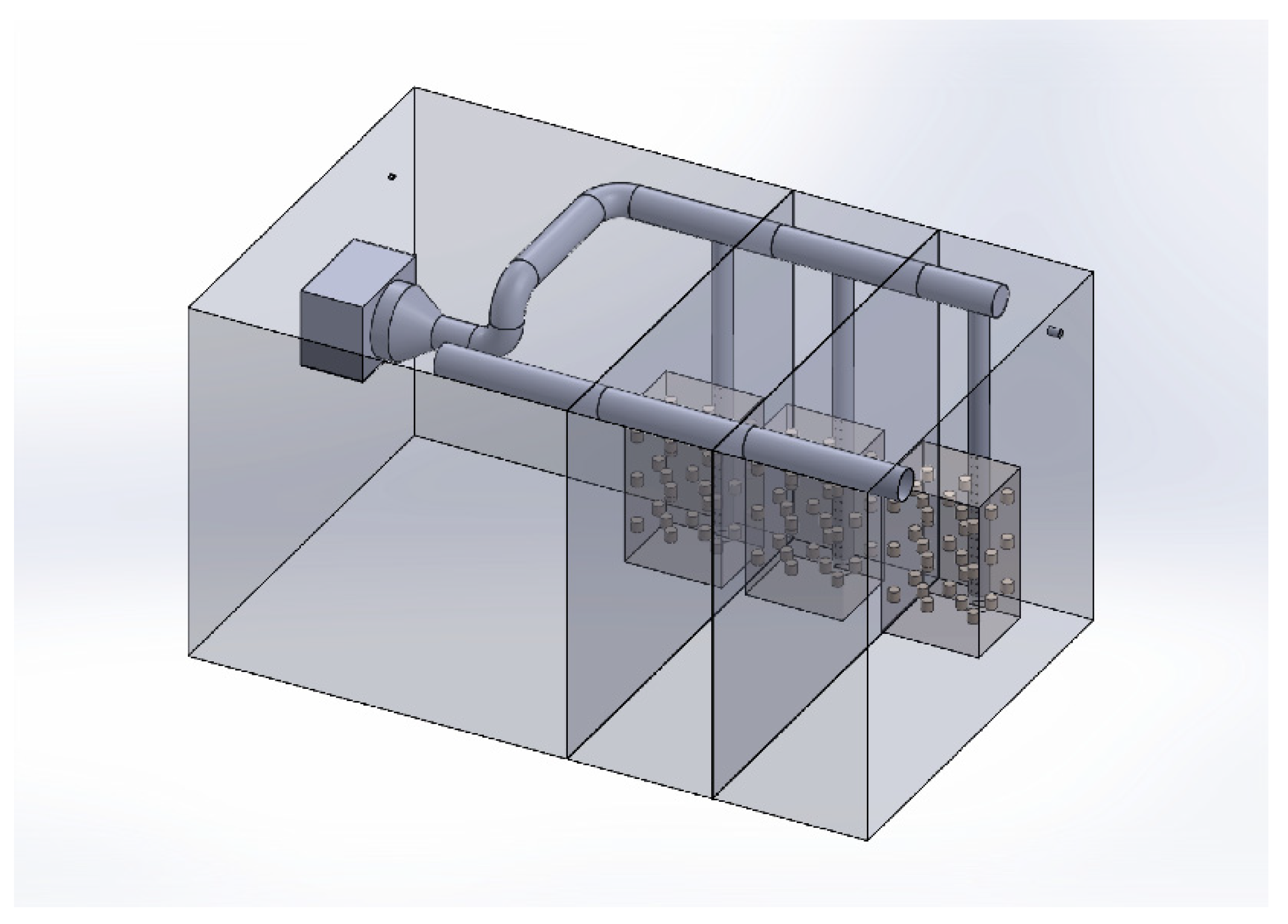
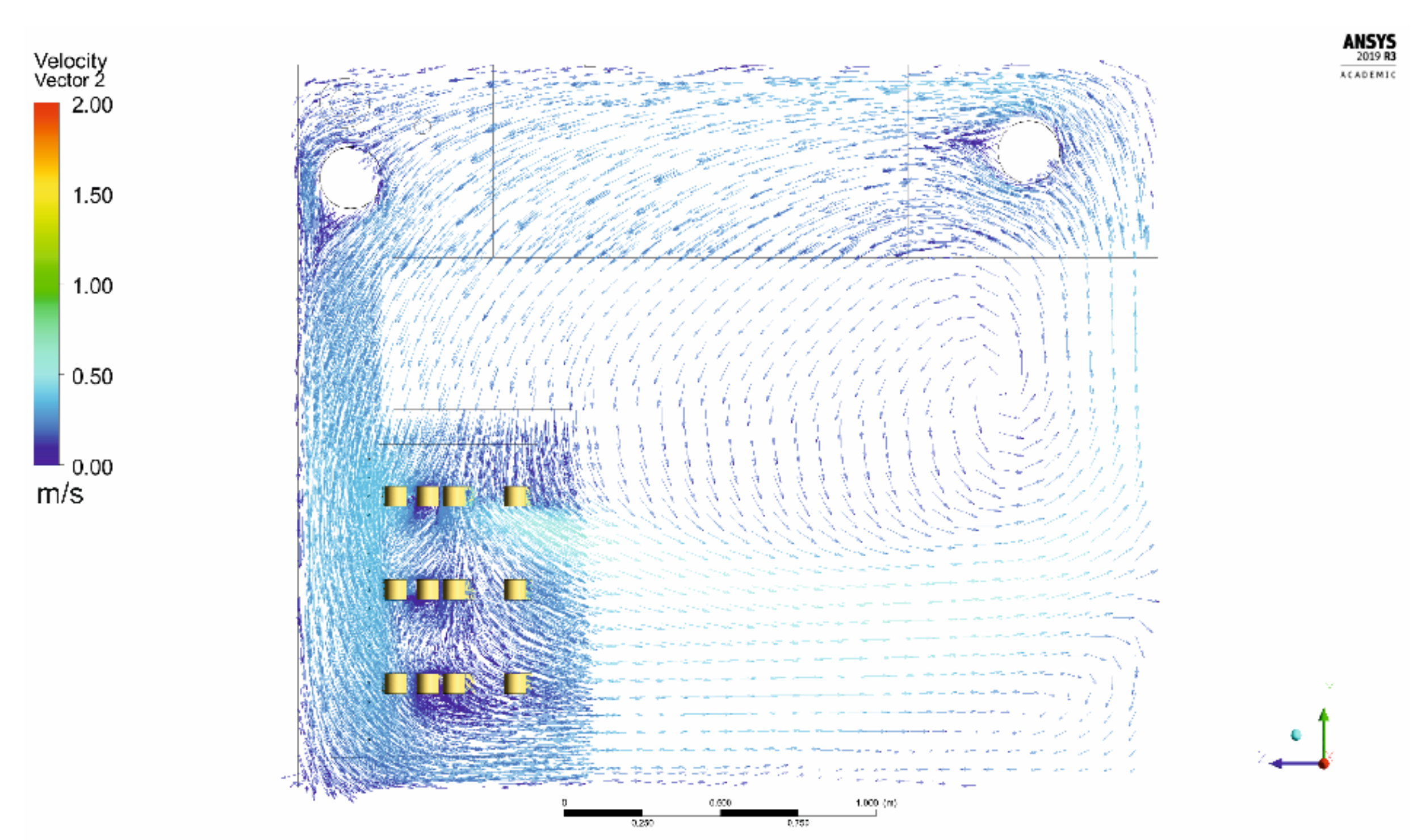
2.3. CFD Modelling
2.4. Physical and Chemical Analysis
2.5. Microbiological Analysis
2.6. Statistical Analysis
3. Results
Microbiological Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Sharma, A.K.; Bhandari, R.; Pinca-Bretotean, C.; Sharma, C.; Dhakad, S.K.; Mathur, A. A study of trends and industrial prospects of Industry 4.0. Mater. Today Proc. 2021, in press. [Google Scholar]
- Defraeye, T. Advanced computational modelling for drying processes—A review. Appl. Energy 2014, 131, 323–344. [Google Scholar] [CrossRef]
- Khan, F.A.; Fischer, C.; Straatman, A.G. Numerical model for non-equilibrium heat and mass exchange in conjugate fluid/solid/porous domains with application to evaporative cooling and drying. Int. J. Heat Mass Transf. 2015, 80, 513–528. [Google Scholar] [CrossRef]
- Mirade, P.-S.; Kondjoyan, A.; Daudin, J.-D. Three-dimensional CFD calculations for designing large food chillers. Comput. Electron. Agric. 2002, 34, 67–88. [Google Scholar] [CrossRef]
- Mirade, P.-S.; Picgirard, L. Improvement of ventilation homogeneity in an industrial batch-type carcass chiller by CFD investigation. Food Res. Int. 2006, 39, 871–881. [Google Scholar] [CrossRef]
- Defraeye, T.; Herremans, E.; Verboven, P.; Carmeliet, J.; Nicolai, B. Convective heat and mass exchange at surfaces of horticultural products: A microscale CFD modelling approach. Agric. For. Meteorol. 2012, 162–163, 71–84. [Google Scholar] [CrossRef]
- Le Page, J.-F.; Chevarin, C.; Kondjoyan, A.; Daudin, J.-D.; Mirade, P.-S. Development of an approximate empirical-CFD model estimating coupled heat and water transfers of stacked food products placed in airflow. J. Food Eng. 2009, 92, 208–216. [Google Scholar] [CrossRef]
- Mirade, P.-S. Computational fluid dynamics (CFD) modelling applied to the ripening of fermented food products: Basics and advances. Trends Food Sci. Technol. 2008, 19, 472–481. [Google Scholar] [CrossRef]
- Mirade, P.-S.; Daudin, J.-D. Computational fluid dynamics prediction and validation of gas circulation in a cheese-ripening room. Int. Dairy J. 2006, 16, 920–930. [Google Scholar] [CrossRef]
- Chanteloup, V.; Mirade, P.-S. Computational fluid dynamics (CFD) modelling of local mean age of air distribution in forced-ventilation food plants. J. Food Eng. 2009, 90, 90–103. [Google Scholar] [CrossRef]
- Mirade, P.-S.; Daudin, J.-D.; Ducept, F.; Trystram, G.; Clément, J. Characterization and CFD modelling of air temperature and velocity profiles in an industrial biscuit baking tunnel oven. Food Res. Int. 2004, 37, 1031–1039. [Google Scholar] [CrossRef]
- Ambaw, A.; Delele, M.A.; Defraeye, T.; Ho, Q.T.; Opara, L.U.; Nicolai, B.M.; Verboven, P. The use of CFD to characterize and design post-harvest storage facilities: Past, present and future. Comput. Electron. Agric. 2013, 93, 184–194. [Google Scholar] [CrossRef]
- Conceição, C.; Martins, P.; Alvarenga, N.; Dias, J.; Lamy, E.; Garrido, L.; Gomes, S.; Freitas, S.; Belo, A.; Brás, T.; et al. Cynara cardunculus: Use in Cheesemaking and Pharmaceutical Applications. In Technological Approaches for Novel Applications in Dairy Processing, 1st ed.; Koca, N., Ed.; InTechOpen: London, UK, 2018; pp. 73–107. [Google Scholar]
- Araújo-Rodrigues, H.; Tavaria, F.K.; dos Santos, M.T.P.G.; Alvarenga, N.; Pintado, M.M. A review on microbiological and technological aspects of Serpa PDO cheese: An ovine raw milk cheese. Int. Dairy J. 2020, 100, 104561. [Google Scholar] [CrossRef]
- Mirade, P.-S.; Rougier, T.; Daudin, J.-D.; Picque, D.; Corrieu, G. Effect of design of blowing duct on ventilation homogeneity around cheeses in a ripening chamber. J. Food Eng. 2006, 75, 59–70. [Google Scholar] [CrossRef]
- Mirade, P.S. 28-CFD Modeling of indoor atmosphere and water exchanges during the cheese ripening process. In Computational Fluid Dynamics in Food Processing, 1st ed.; Sun, D.-W., Ed.; CRC Press: Boca Ratón, FL, USA, 2007; pp. 697–726. [Google Scholar]
- ISO 5534. Cheese and Processed Cheese: Determination of the Total Solids Content (Reference Method); International Organization for Standardization: Geneva, Switzerland, 2004. [Google Scholar]
- Alvarenga, N.; Canada, J.; Sousa, I. Effect of freezing on the rheological and chemical properties of raw ewe’s milk semisoft cheese. J. Dairy Res. 2011, 78, 80–87. [Google Scholar] [CrossRef] [PubMed]
- Dias, J.M.; Lage, P.; Alvarenga, N.; Garcia, J.; Borrega, J.; Santos, M.T.; Lampreia, C.; Coelho, L.; Pássaro, J.; Martins, J.; et al. Impact of environmental conditions on the ripening of Queijo de Évora PDO cheese. J. Food Sci. Technol. 2020, 1–11. [Google Scholar] [CrossRef]
- Dias, J.; Lage, P.; Garrido, A.; Machado, E.; Conceição, C.; Gomes, S.; Martins, A.; Paulino, A.; Duarte, M.F.; Alvarenga, N. Evaluation of gas holes in “Queijo de Nisa” PDO cheese using computer vision. J. Food Sci. Technol. 2021, 58, 1072–1080. [Google Scholar] [CrossRef]
- Nopens, I.; Foubert, I.; De Graef, V.; Van Laere, D.; Dewettinck, K.; Vanrolleghem, P. Automated image analysis tool for migration fat bloom evaluation of chocolate coated food products. LWT 2008, 41, 1884–1891. [Google Scholar] [CrossRef]
- Incropera, F.P.; DeWitt, D.P. Fundamentals of Heat and Mass Transfer, 5th ed.; John Wiley & Sons: New York, NY, USA, 2002; pp. 325–384. [Google Scholar]
- Leclercq-Perlat, M.-N.; Saint-Eve, A.; Le Jan, E.; Raynaud, S.; Morge, S.; Lefrileux, Y.; Picque, D. Physicochemical and sensory evolutions of the lactic goat cheese Picodon in relation to temperature and relative humidity used throughout ripening. J. Dairy Sci. 2009, 102, 5713–5725. [Google Scholar] [CrossRef]
- Carvalho, G.P.; Santos, R.; Fino, A.; Ferreira, P.; Rodrigues, F.M.; Dias, J. Evolution during Three Ripening Stages of Évora Cheese. Foods 2020, 9, 1140. [Google Scholar] [CrossRef]
- Macedo, A.; Costa, M.; Malcata, F. Changes in the microflora of Serra cheese: Evolution throughout ripening time, lactation period and axial location. Int. Dairy J. 1996, 6, 79–94. [Google Scholar] [CrossRef]
- Alvarenga, N.; Silva, P.; Rodriguez Garcia, J.; Sousa, I. Estimation of Serpa cheese ripening time using multiple linear regression (MLR) considering rheological, physical and chemical data. J. Dairy Res. 2008, 75, 233–239. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Fox, P.F.; Guinee, T.P.; Cogan, T.M.; McSweeney, P.L.H. Chapter 6: Starter cultures. In Fundamentals of Cheese Science, 2nd ed.; Fox, P.F., Guinee, T.P., Cogan, T.M., McSweeney, P.L.H., Eds.; Springer: New York, NY, USA, 2017; pp. 121–183. [Google Scholar]
- Picque, D.; Guillemin, H.; Mirade, P.; Didienne, R.; Lavigne, R.; Perret, B.; Montel, M.; Corrieu, G. Effect of sequential ventilation on cheese ripening and energy consumption in pilot ripening rooms. Int. Dairy J. 2009, 19, 489–497. [Google Scholar] [CrossRef]
- Reis, P.; Malcata, F. Current state of Portuguese dairy products from ovine and caprine milks. Small Rumin. Res. 2011, 101, 122–133. [Google Scholar] [CrossRef]
- Guggisberg, D.; Schuetz, P.; Winkler, H.; Amrein, R.; Jakob, E.; Frohlich-Wyder, M.-T.; Irmler, S.; Bisig, W.; Jerjen, I.; Plamondon, M.; et al. Mechanism and control of the eye formation in cheese. Int. Dairy J. 2015, 47, 118–127. [Google Scholar] [CrossRef]
- Tabla, R.; Gómez, A.; Simancas, A.; Rebollo, J.; Molina, F.; Roa, I. Enterobacteriaceae species during manufacturing and ripening of semi-hard and soft raw ewe’s milk cheese: Gas production capacity. Small Rumin. Res. 2016, 145, 123–129. [Google Scholar] [CrossRef]
- Freitas, C.; Malcata, F.X. Microbiology and Biochemistry of Cheeses with Appélation d’Origine Protegée and Manufactured in the Iberian Peninsula from Ovine and Caprine Milks. J. Dairy Sci. 2000, 83, 584–602. [Google Scholar] [CrossRef]
- Santos, M.T.P.; Benito, M.J.; Córdoba, M.D.G.; Egas, C.; Merchán, A.V.; Galván, A.I.; Ruiz-Moyano, S. Bacterial communities in Serpa cheese by culture 608 dependent techniques, 16S rRNA gene sequencing and high-throughput 609 sequencing analysis. J. Food Sci. 2018, 83, 1333–1341. [Google Scholar] [CrossRef]
- Beresford, T.; Fitzsimons, N.; Brennan, N.; Cogan, T. Recent advances in cheese microbiology. Int. Dairy J. 2001, 11, 259–274. [Google Scholar] [CrossRef]
- Delgado, F.; González-Crespo, J.; Ladero, L.; Cava, R.; Ramírez, R. Free fatty acids and oxidative changes of a Spanish soft cheese (PDO ‘Torta del Casar’) during ripening. Int. J. Food Sci. Technol. 2009, 44, 1721–1728. [Google Scholar] [CrossRef]
- Del Pozo, B.F.; Gaya, P.; Medina, M.; Rodriguez-Marin, M.A.; Nuñez, M. Changes in the microflora of La Serena ewes’ milk cheese during ripening. J. Dairy Res. 1988, 55, 449–455. [Google Scholar] [CrossRef]
- Settanni, L.; Di Grigoli, A.; Tornambé, G.; Bellina, V.; Francesca, N.; Moschetti, G.; Bonanno, A. Persistence of wild Streptococcus thermophilus strains on wooden vat and during the manufacture of a traditional Caciocavallo type cheese. Int. J. Food Microbiol. 2012, 155, 73–81. [Google Scholar] [CrossRef] [PubMed]
- Kornacki, J.; Gurtler, J.; Stawick, B. 9. Enterobacteriaceae, Coliforms, and Escherichia coli as quality and safety indicators. In Compendium of Methods for the Microbiological Examination of Foods, 4th ed.; Salfinger, Y., Tortorello, M.L., Eds.; American Public Health Association: Washington, DC, USA, 2013; pp. 103–120. [Google Scholar]
- Tavaria, F.; Malcata, F. Microbiological characterization of Serra da Estrela cheese throughout its appellation d’origine protegee region. J. Food Protect. 1998, 61, 601–607. [Google Scholar] [CrossRef] [PubMed]
- Pereira-Dias, S.; Potes, M.; Marinho, A.; Malfeito-Ferreira, M.; Loureiro, V. Characterisation of yeast flora isolated from an artisanal Portuguese ewes’ cheese. Int. J. Food Microbiol. 2000, 60, 55–63. [Google Scholar] [CrossRef]
- Santos, M.T.G.; Benito, M.J.; Córdoba, M.G.; Alvarenga, N.; Herrera, S.R.-M.S. Yeast community in traditional Portuguese Serpa cheese by culture-dependent and -independent DNA approaches. Int. J. Food Microbiol. 2017, 262, 63–70. [Google Scholar] [CrossRef]
- Dahl, S.; Tavaria, F.; Malcata, F.X. Relationships between flavour and microbiological profiles in Serra da Estrela cheese throughout ripening. Int. Dairy J. 2000, 10, 255–262. [Google Scholar] [CrossRef]
- Dias, J.; Pires, M.C.; Carvalho, G.; Ferreira, P.; Santos, R.; Mondragão-Rodrigues, F.; Teixeira, N.; Alvarenga, N. Capítulo 2—Descrição das queijarias nas regiões de Serpa, Évora e Azeitão. In Aplicação da Mecânica dos Fluídos Computacional e Monitorização Remota em Queijarias Tradicionais; Instituto Politécnico de Beja: Beja, Portugal, 2020; pp. 39–57. [Google Scholar]
- Pajonk, A. La solution flux variables alternants pour homogénéiser l’ambiance. Rev. Laitière Française 2001, 614, 20–22. [Google Scholar]
- Malcata, F.X.; Gomes, A.M.P.; da Costa, M.L. “Probiotic” goat cheese: Effect of ripening temperature and relative humidity on proteolysis and lipolysis. J. Dairy Sci. 1995, 78 (Suppl. 1), 99. [Google Scholar]
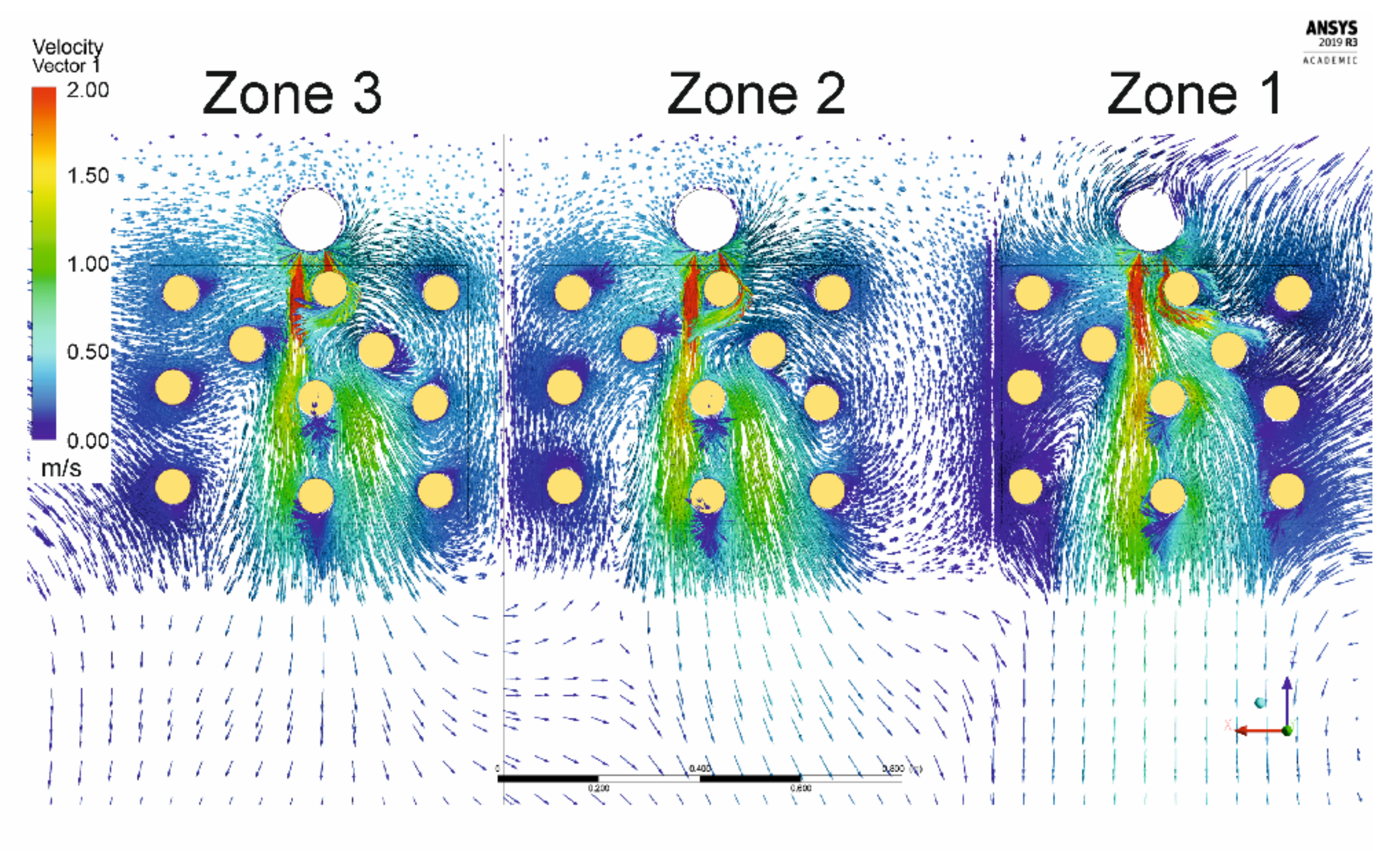
| . | Zone 1 | Zone 2 | Zone 3 |
|---|---|---|---|
| Temperature (°C) | 14.6 a (1.4) | 13.6 b (0.4) | 12.3 c (0.7) |
| Humidity (%) | 82.9 b (3.1) | 86.8 a (4.1) | 80.8 b (2.7) |
| Air velocity (m/s) | 0.14 a (0.14) | 0.20 a (0.15) | 0.20 a (0.19) |
| Parameter | 0 Days | After 35 Days of Ripening | ||
|---|---|---|---|---|
| Zone 1 | Zone 2 | Zone 3 | ||
| Moisture (%) | 58.9 (1.1) a | 24.7 (1.4) bc | 25.5 (1.3) b | 23.9 (1.4) c |
| aw (-) | 0.95 (0.02) a | 0.82 (0.01) c | 0.84 (0.02) b | 0.83 (0.01) bc |
| pH (-) | 6.58 (0.11) a | 5.32 (0.15) b | 5.30 (0.13) b | 5.44 (0.21) b |
| Hardness (N) | 5.84 (1.0) c | 93.48 (18.07) a | 57.70 (16.41) b | 76.82 (21.6) ab |
| Adhesiveness (-N.s) | 0.96 (0.43) b | 18.26 (4.51) a | 16.68 (3.70) a | 15.89 (5.57) a |
| G′1Hz (kPa) | 41.0 (7.3) b | 2 693.1 (408.4) a | 2 299.7 (530.8) a | 2 823.3 (796.0) a |
| G″1Hz (kPa) | 10.0 (1.9) b | 569.7 (84.0) a | 505.0 (109.0) a | 617.4 (151.5) a |
| Yrind (-) | 207 (10) b | 223 (3) a | 205 (10) bc | 196 (6) c |
| Area of holes (%) | 0.45 (0.58) b | 3.78 (2.76) ab | 3.08 (1.24) b | 5.84 (1.90) a |
| Area (mm2/hole) | 3.28 (2.72) a | 4.33 (1.78) a | 3.96 (1.29) a | 4.43 (1.27) a |
| Circularity (-) | 0.42 (0.11) a | 0.36 (0.06) a | 0.32 (0.05) a | 0.35 (0.03) a |
| Feret (mm) | 2.84 (1.61) a | 3.11 (0.47) a | 3.17 (0.53) a | 2.82 (0.34) a |
| mFeret (mm) | 1.86 (0.78) a | 1.79 (0.27) a | 1.86 (0.32) a | 1.7 (0.19) a |
| Group (log10 cfu/g) | 0 Days | After 35 Days of Ripening | ||
|---|---|---|---|---|
| Zone 1 | Zone 2 | Zone 3 | ||
| Total mesophilic bacteria | 6.17 (0.08) b | 8.89 (0.45) a | 8.64 (0.31) a | 8.94 (0.14) a |
| LAB | 4.82 (0.19) b | 8.65 (0.36) a | 8.43 (0.31) a | 8.62 (0.09) a |
| Leuconostoc | 2.32 (0.47) b | 4.25 (3.21) ab | 6.11 (0.16) a | 6.82 (0.93) a |
| Streptococcus | 3.42 (0.39) b | 6.40 (0.72) a | 6.15 (0.27) a | 6.83 (0.50) a |
| Enterobacteriaceae | 4.25 (0.31) a | 2.39 (2.15) b | 4.26 (0.50) a | 4.44 (0.07) a |
| Yeasts | 2.46 (0.51) a | 2.34 (1.20) a | 1.75 (0.66) a | 1.38 (0.09) a |
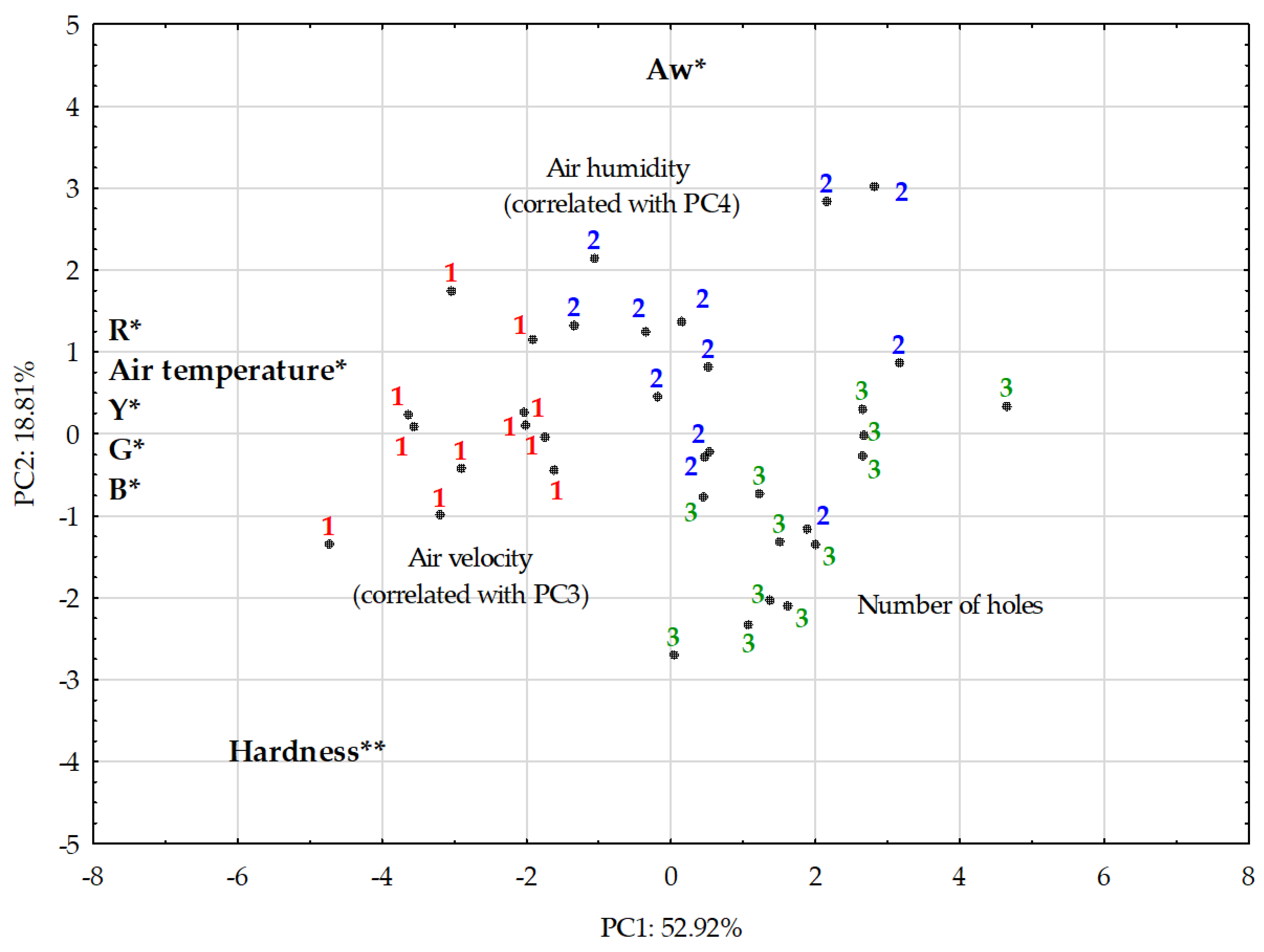
| Temperature | Humidity | Velocity | |
|---|---|---|---|
| aw | −0.51 | 0.56 * | −0.48 |
| pH | 0.46 | 0.00 | 0.79 ** |
| Moisture (%) | −0.15 | 0.37 | −0.45 |
| Rrind | 0.71 ** | −0.21 | −0.11 |
| Grind | 0.74 ** | −0.21 | 0.00 |
| Brind | 0.74 ** | −0.15 | −0.01 |
| Yrind | 0.73 ** | −0.2 | −0.03 |
| Hardness (N) | 0.60 * | −0.53 | 0.54 |
| Adhesiveness (-N.s) | 0.03 | −0.18 | −0.33 |
| G′1Hz (kPa) | 0.23 | −0.34 | 0.23 |
| G′1Hz (kPa) | 0.01 | 0.12 | 0.07 |
| Number of holes | −0.57 * | −0.35 | 0.10 |
| Percentage of holes (%) | −0.59 * | −0.29 | 0.02 |
| Area per hole (mm2) | −0.21 | 0.20 | −0.18 |
| Perimeter per hole (mm) | −0.07 | 0.28 | 0.14 |
| Feret (mm) | 0.06 | 0.47 | −0.22 |
| mFeret (mm) | 0.09 | 0.55 * | −0.15 |
| Circularity (-) | −0.14 | 0.29 | −0.62 * |
| Total mesophiles (log10 cfu/g) | 0.18 | −0.42 | −0.19 |
| Enterobacteriaceae (log10 cfu/g) | 0.02 | 0.24 | 0.47 |
| LAB (log10 cfu/g) | 0.14 | −0.20 | −0.25 |
| Leuconostoc (log10 cfu/g) | −0.44 | 0.08 | −0.17 |
| Streptococcus (log10 cfu/g) | −0.17 | −0.30 | −0.31 |
| Yeasts (log10 cfu/g) | 0.09 | −0.09 | −0.31 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alvarenga, N.; Martins, J.; Caeiro, J.; Garcia, J.; Pássaro, J.; Coelho, L.; Santos, M.T.; Lampreia, C.; Martins, A.; Dias, J. Applying Computational Fluid Dynamics in the Development of Smart Ripening Rooms for Traditional Cheeses. Foods 2021, 10, 1716. https://doi.org/10.3390/foods10081716
Alvarenga N, Martins J, Caeiro J, Garcia J, Pássaro J, Coelho L, Santos MT, Lampreia C, Martins A, Dias J. Applying Computational Fluid Dynamics in the Development of Smart Ripening Rooms for Traditional Cheeses. Foods. 2021; 10(8):1716. https://doi.org/10.3390/foods10081716
Chicago/Turabian StyleAlvarenga, Nuno, João Martins, José Caeiro, João Garcia, João Pássaro, Luis Coelho, Maria Teresa Santos, Célia Lampreia, António Martins, and João Dias. 2021. "Applying Computational Fluid Dynamics in the Development of Smart Ripening Rooms for Traditional Cheeses" Foods 10, no. 8: 1716. https://doi.org/10.3390/foods10081716
APA StyleAlvarenga, N., Martins, J., Caeiro, J., Garcia, J., Pássaro, J., Coelho, L., Santos, M. T., Lampreia, C., Martins, A., & Dias, J. (2021). Applying Computational Fluid Dynamics in the Development of Smart Ripening Rooms for Traditional Cheeses. Foods, 10(8), 1716. https://doi.org/10.3390/foods10081716








