Monitoring of Remaining Thiophenic Compounds in Liquid Fuel Desulphurization Studies Using a Fast HPLC-UV Method
Abstract
1. Introduction
2. Materials and Methods
2.1. Instrumentation
2.2. Chemicals and Reagents
2.3. Preparation of Standard Solutions
2.4. Method Validation
2.5. Adsorption of DBT and 4,6-DMDBT
3. Results
3.1. HPLC Method
3.2. Calibration Curves
3.3. Application in Real Sample
3.4. Comparison with Other Methods
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- European Environment Agency. EMEP/EEA Air Pollutant Emission Inventory Guidebook 2019. Available online: http://www.eea.europa.eu (accessed on 1 February 2021).
- Bandosz, T.J. Chapter 5 Desulfurization on activated carbons. Interface Sci. Technol. 2006. [Google Scholar] [CrossRef]
- Link, D.D.; Baltrus, J.P.; Rothenberger, K.S.; Zandhuis, P.; Minus, D.K.; Striebich, R.C. Class-and structure-specific separation, analysis, and identification techniques for the characterization of the sulfur components of JP-8 aviation fuel. Energy Fuels 2003, 17, 1292–1302. [Google Scholar] [CrossRef]
- Directive (EU) 2016/802 of the European Parliament and of the Council of 11 May 2016 Relating to a Reduction in the Sulfur Content of Certain Liquid Fuels (Codification); L 132/58 21.5.2016; EU: Brussels, Belgium, 2016.
- Directive (EU) 2015/1513 of the European Parliament and of the Council of 9 September 2015 Amending Directive 98/70/EC Relating to the Quality of Petrol and Diesel Fuels and Amending Directive 2009/28/EC on the Promotion of the Use of Energy from Renewable Sources (Text with EEA Relevance); L 239/1 15.09.2015; EU: Brussels, Belgium, 2015.
- Iruretagoyena, D.; Montesano, R. Selective Sulfur Removal from Liquid Fuels Using Nanostructured Adsorbents, In Nanotechnology in Oil and Gas Industries: Principles and Applications; Saleh, T.A., Ed.; Springer International Publishing: Cham, Switzerland, 2018; pp. 133–150. [Google Scholar]
- Song, T.; Zhang, Z.; Chen, J.; Ring, Z.; Yang, H.; Zheng, Y. Effect of aromatics on deep hydrodesulfurization of dibenzothiophene and 4,6-dimethyldibenzothiopene over NiMo/Al2O3 catalyst. Energy Fuels 2006, 20, 2344–2349. [Google Scholar] [CrossRef]
- Rabarihoela-Rakotovao, V.; Brunet, S.; Perot, G.; Diehl, F. Effect of H2S partial pressure on the HDS of dibenzothiophene and 4,6-dimethyldibenzothiophene over sulfided NiMoP/Al2O3 and CoMoP/Al2O3 catalysts. Appl. Catal. A Gen. 2006, 306, 34–44. [Google Scholar] [CrossRef]
- Matsuzawa, S.; Tanaka, J.; Sato, S.; Ibusuki, T. Photocatalytic oxidation of dibenzothiophenes in acetonitrile using TiO2: Effect of hydrogen peroxide and ultrasound irradiation. J. Photochem. Photobiol. A Chem. 2002, 149, 183–189. [Google Scholar] [CrossRef]
- Tao, H.; Nakazato, T.; Sato, S. Energy-efficient ultra-deep desulfurization of kerosene based on selective photooxidation and adsorption. Fuel 2009, 88, 1961–1969. [Google Scholar] [CrossRef]
- Song, C. An overview of new approaches to deep desulfurization for ultra-clean gasoline, diesel fuel and jet fuel. Catal. Today 2003, 86, 211–263. [Google Scholar] [CrossRef]
- Kim, J.H.; Ma, X.; Zhou, A.; Song, C. Ultra-deep desulfurization and denitrogenation of diesel fuel by selective adsorption over three different adsorbents: A study on adsorptive selectivity and mechanism. Catal. Today 2006, 111, 74–83. [Google Scholar] [CrossRef]
- Seredych, M.; Bandosz, T.J. Template-derived mesoporous carbons with highly dispersed transition metals as media for the reactive adsorption of dibenzothiophene. Langmuir 2007, 23, 6033–6041. [Google Scholar] [CrossRef]
- Velu, S.; Watanabe, S.; Ma, X.; Song, C. Regenerable adsorbents for the adsorptive desulfurization of transportation fuels for fuel cell applications. ACS Div. Fuel Chem. Prepr. 2003, 48, 526–528. [Google Scholar]
- Yu, G.; Lu, S.; Chen, H.; Zhu, Z. Diesel fuel desulfurization with hydrogen peroxide promoted by formic acid and catalyzed by activated carbon. Carbon 2005, 43, 2285–2294. [Google Scholar] [CrossRef]
- Yang, Y.; Lu, H.; Ying, P.; Jiang, Z.; Li, C. Selective dibenzothiophene adsorption on modified activated carbons. Carbon 2007, 45, 3042–3044. [Google Scholar] [CrossRef]
- Deliyanni, E.; Seredych, M.; Bandosz, T.J. Interactions of 4,6-dimethyldibenzothiophene with the surface of activated carbons. Langmuir 2009, 25, 9302–9312. [Google Scholar] [CrossRef] [PubMed]
- Seredych, M.; Lison, J.; Jans, U.; Bandosz, T.J. Textural and chemical factors affecting adsorption capacity of activated carbon in highly efficient desulfurization of diesel fuel. Carbon 2009, 47, 2491–2500. [Google Scholar] [CrossRef]
- Jeon, H.J.; Ko, C.H.; Kim, S.H.; Kim, J.N. Removal of refractory sulfur compounds in diesel using activated carbon with controlled porosity. Energy Fuels 2009, 23, 2537–2543. [Google Scholar] [CrossRef]
- Seredych, M.; Bandosz, T.J. Adsorption of dibenzothiophenes on nanoporous carbons: Identification of specific adsorption sites governing capacity and selectivity. Energy Fuels 2010, 24, 3352–3360. [Google Scholar] [CrossRef]
- Jiang, Z.; Liu, Y.; Sun, X.; Tian, F.; Sun, F.; Liang, C.; You, W.; Han, C.; Li, C. Activated carbons chemically modified by concentrated H2SO4 for the adsorption of the pollutants from wastewater and the dibenzothiophene from fuel oils. Langmuir 2003, 19, 731–736. [Google Scholar] [CrossRef]
- Rekos, K.; Kampouraki, Z.C.; Panou, C.; Baspanelou, A.; Triantafyllidis, K.; Deliyanni, E. Adsorption of DBT and 4,6-DMDBTon nanoporous activated carbons: The role of surface chemistry and the solvent. Environ. Sci. Pollut. Res. 2020, 24. [Google Scholar] [CrossRef]
- Triantafyllidis, K.S.; Deliyanni, E.A. Desulfurization of diesel fuels: Adsorption of 4,6-DMDBT on different origin and surface chemistry nanoporous activated carbons. Chem. Eng. J. 2014, 236, 406–414. [Google Scholar] [CrossRef]
- Seredych, M.; Deliyanni, E.; Bandosz, T.J. Role of microporosity and surface chemistry in adsorption of 4,6-dimethyldibenzothiophene on polymer-derived activated carbons. Fuel 2010, 89, 1499–1507. [Google Scholar] [CrossRef]
- Ferreira, J.P.; Viveiros, R.; Lourenço, A.; Soares Da Silva, M.; Rosatella, A.; Casimiro, T.; Afonso, C.A.M. Integrated desulfurization of diesel by combination of metal-free oxidation and product removal by molecularly imprinted polymers. RSC Adv. 2014, 4, 54948–54952. [Google Scholar] [CrossRef]
- Yue, D.; Lei, J.; Zhou, L.; Du, X.; Guo, Z.; Li, J. Oxidative desulfurization of fuels at room temperature using ordered meso/macroporous H3PW12O40/SiO2 catalyst with high specific surface areas. Arab. J. Chem. 2020, 13, 2649–2658. [Google Scholar] [CrossRef]
- Yazu, K.; Sato, S.; Sugimoto, Y.; Matsumura, A.; Saito, I. Tungstophosphoric acid-catalyzed oxidative desulfurization of naphtha with hydrogen peroxide in naphtha/acetic acid biphasic system. J. Jpn. Pet. Inst. 2007, 50, 329–334. [Google Scholar] [CrossRef][Green Version]
- Sharma, R.; Singh, J.; Verma, N. A novel spectrophotometric method for simultaneous estimation of dibenzothiophene and 2-hydroxybiphenyl in their mixed spectrum and its application in screening of specific biodesulfurizing microbes. 3 Biotech 2020, 10, 1–9. [Google Scholar] [CrossRef]
- Gratz, S.R.; Ciolino, L.A.; Mohrhaus, A.S.; Gamble, B.M.; Gracie, J.M.; Jackson, D.S.; Roetting, J.P.; McCauley, H.A.; Heitkemper, D.T.; Fricke, F.L.; et al. Screening and determination of polycyclic aromatic hydrocarbons in seafoods using QuEChERS-based extraction and high-performance liquid chromatography with fluorescence detection. J. AOAC Int. 2011, 94, 1601–1616. [Google Scholar] [CrossRef]
- Saravanabhavan, G.; Helferty, A.; Hodson, P.V.; Brown, R.S. A multi-dimensional high performance liquid chromatographic method for fingerprinting polycyclic aromatic hydrocarbons and their alkyl-homologs in the heavy gas oil fraction of Alaskan North Slope crude. J. Chromatogr. A 2007, 1156, 124–133. [Google Scholar] [CrossRef]
- Santos de Freitas, C.; Valencia-Dávila, J.A.; Lopes Pereira, R.C.; Torres Abrantes, L.; Kato, L.; Gontijo Vaz, B. Isolation and characterization of sulfur-containing compounds by positive-ion electrospray ionization and online HPLC/Atmospheric pressure chemical ionization coupled to Orbitrap mass spectrometry. Fuel 2021, 289. [Google Scholar] [CrossRef]
- Mawad, A.M.M.; Hassanein, M.; Aldaby, E.S.; Yousef, N. Desulphurisation kinetics of thiophenic compound by sulphur oxidizing Klebsiella oxytoca SOB-1. J. Appl. Microbiol. 2020. [Google Scholar] [CrossRef] [PubMed]
- Mezcua, M.; Fernández-Alba, A.R.; Rodríguez, A.; Boltes, K.; Leton, P.; García-Calvo, E. Chromatographic methods applied in the monitoring of biodesulfurization processes—State of the art. Talanta 2007, 73, 103–114. [Google Scholar] [CrossRef]
- Che, Y.; Ma, W.; Ren, Y.; Chen, C.; Zhang, X.; Zhao, J.; Zang, L. Photooxidation of dibenzothiophene and 4,6-dimethyldibenzothiophene sensitized by N-methylquinolinium tetrafluoborate: Mechanism and intermediates investigation. J. Phys. Chem. B 2005, 109, 8270–8276. [Google Scholar] [CrossRef]
- Decision 2002/657/EC of the European Communities Commission of 12 August 2002 Implementing Council Directive 96/23/EC Concerning the Performance of Analytical Methods and the Interpretation of Results; Off. J. Eur. Communities 2002. L 221/8, 17.08.2002; EU: Brussels, Belgium, 2002.
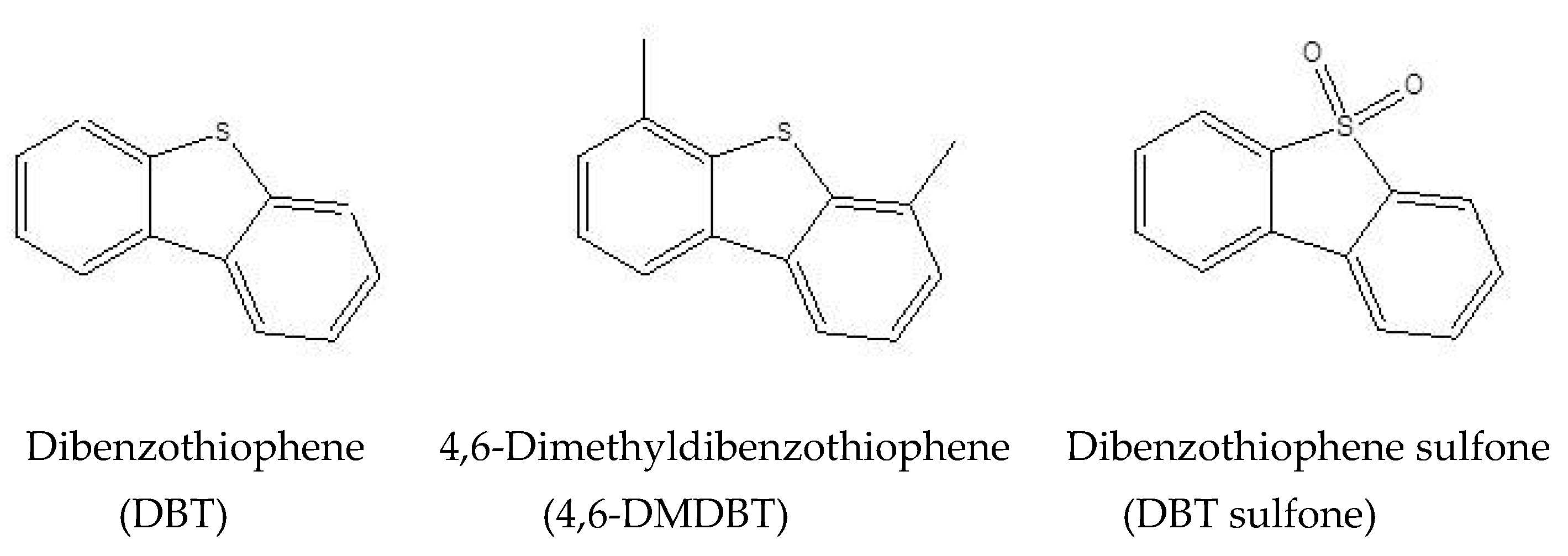

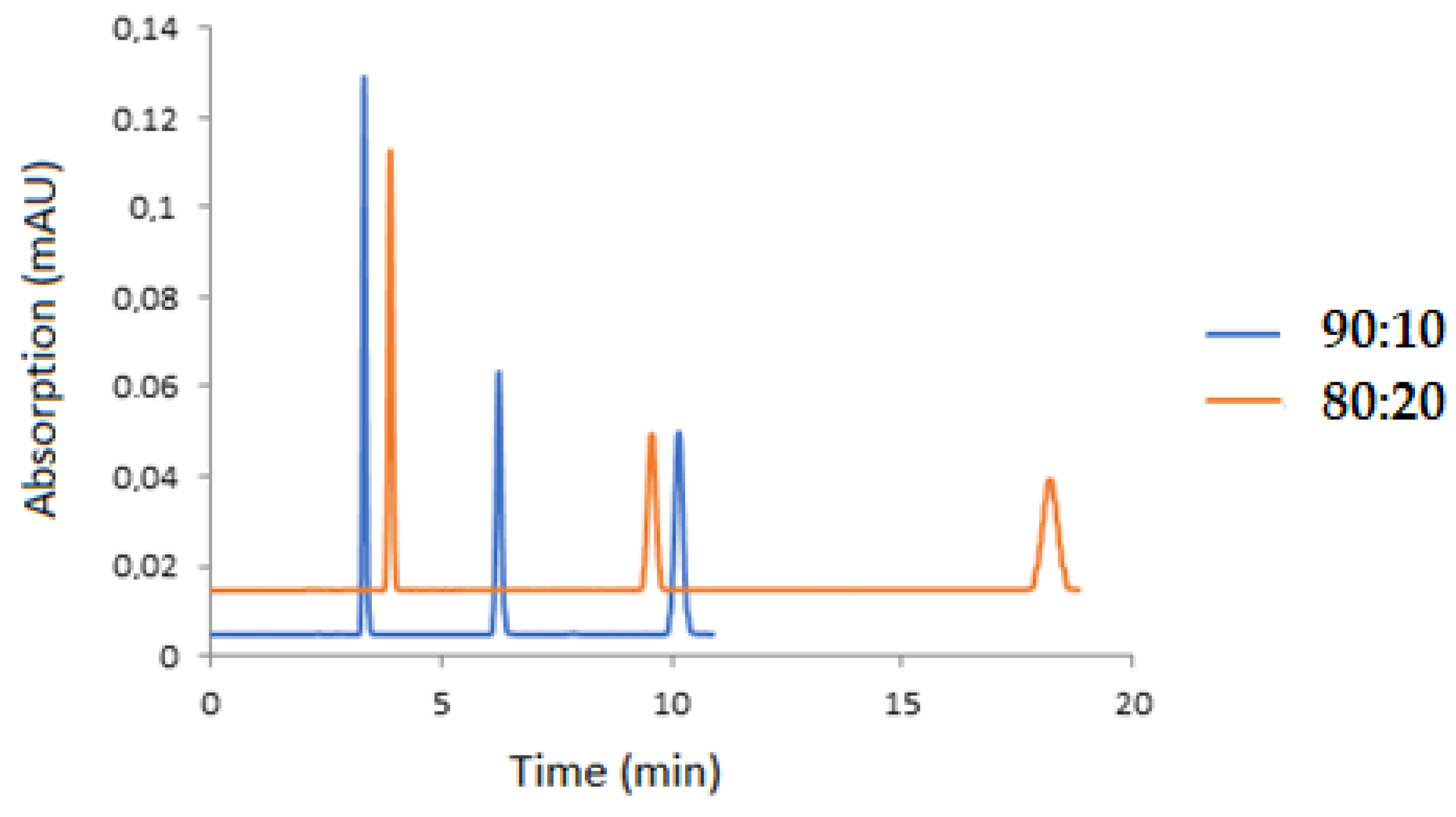
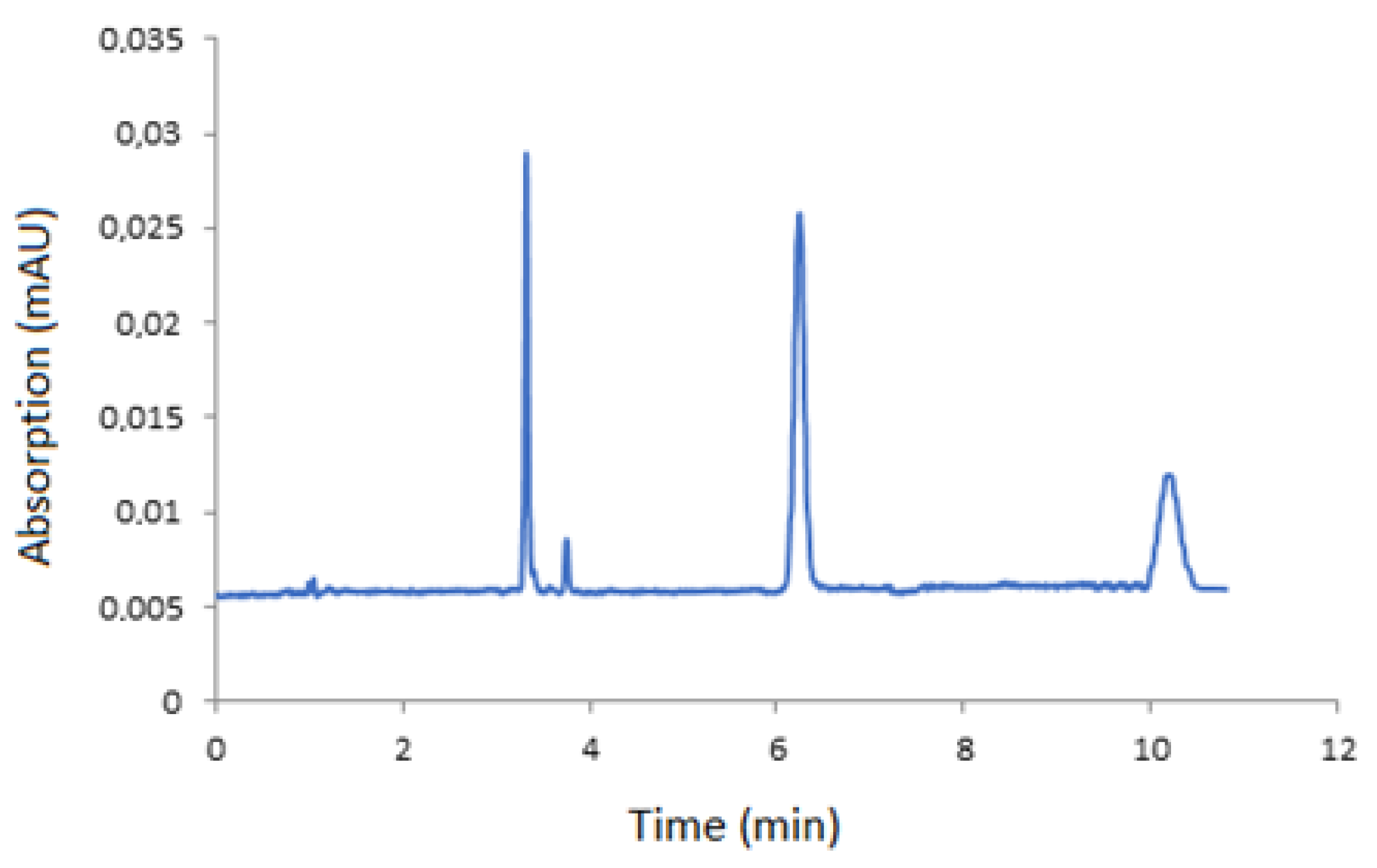
| Analyte | Slope | Intercept | Correlation Coefficient (R) |
|---|---|---|---|
| DBT | 0.0136 | 0.009 | 0.9925 |
| 4,6-DMDBT | 0.0135 | 0.0019 | 0.9953 |
| DBT Sulfone | 0.0670 | −0.0414 | 0.9993 |
| Analyte | Added (ng/μL) | Found ± SD (ng/μL) | R% | RSD% |
|---|---|---|---|---|
| 0.5 | 0.46 ± 0.04 | 92.0 | 8.7 | |
| DBT | 2 | 1.94 ± 0.20 | 97.0 | 10.3 |
| 10 | 9.8 ± 1.1 | 98.0 | 11.2 | |
| 0.5 | 0.47 ± 0.05 | 94.0 | 10.6 | |
| 4,6-DMDBT | 2 | 2.0 ± 0.2 | 100.0 | 10.0 |
| 10 | 10.2 ± 0.87 | 102.0 | 8.5 | |
| 0.5 | 0.52 ± 0.06 | 104.0 | 11.5 | |
| DBT Sulfone | 2 | 1.88 ± 1.3 | 94.0 | 6.9 |
| 10 | 9.8 ± 1.1 | 98.0 | 11.2 |
| Analyte | Added (ng/μL) | Found ± SD (ng/μL) | R% | RSD% |
|---|---|---|---|---|
| 0.5 | 0.53 ± 0.06 | 106.0 | 11.3 | |
| DBT | 2 | 2.08 ± 0.25 | 104.0 | 12.0 |
| 10 | 9.49 ± 0.84 | 94.9 | 8.8 | |
| 0.5 | 0.5 ± 0.06 | 100.0 | 12.0 | |
| 4,6-DMDBT | 2 | 2.16 ± 0.3 | 108.0 | 13.9 |
| 10 | 9.79 ± 0.7 | 97.9 | 7.2 | |
| 0.5 | 0.49 ± 0.05 | 98.0 | 10.2 | |
| DBT Sulfone | 2 | 1.92 ± 0.15 | 96.0 | 7.8 |
| 10 | 9.4 ± 0.8 | 94.0 | 8.5 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kapsali, V.; Triantafyllidis, K.; Deliyanni, E.; Samanidou, V. Monitoring of Remaining Thiophenic Compounds in Liquid Fuel Desulphurization Studies Using a Fast HPLC-UV Method. Separations 2021, 8, 48. https://doi.org/10.3390/separations8040048
Kapsali V, Triantafyllidis K, Deliyanni E, Samanidou V. Monitoring of Remaining Thiophenic Compounds in Liquid Fuel Desulphurization Studies Using a Fast HPLC-UV Method. Separations. 2021; 8(4):48. https://doi.org/10.3390/separations8040048
Chicago/Turabian StyleKapsali, Vasiliki, Konstantinos Triantafyllidis, Eleni Deliyanni, and Victoria Samanidou. 2021. "Monitoring of Remaining Thiophenic Compounds in Liquid Fuel Desulphurization Studies Using a Fast HPLC-UV Method" Separations 8, no. 4: 48. https://doi.org/10.3390/separations8040048
APA StyleKapsali, V., Triantafyllidis, K., Deliyanni, E., & Samanidou, V. (2021). Monitoring of Remaining Thiophenic Compounds in Liquid Fuel Desulphurization Studies Using a Fast HPLC-UV Method. Separations, 8(4), 48. https://doi.org/10.3390/separations8040048









