Recent Applications and Newly Developed Strategies of Solid-Phase Microextraction in Contaminant Analysis: Through the Environment to Humans
Abstract
1. Introduction
2. The Beginning Step: Environmental Matrices
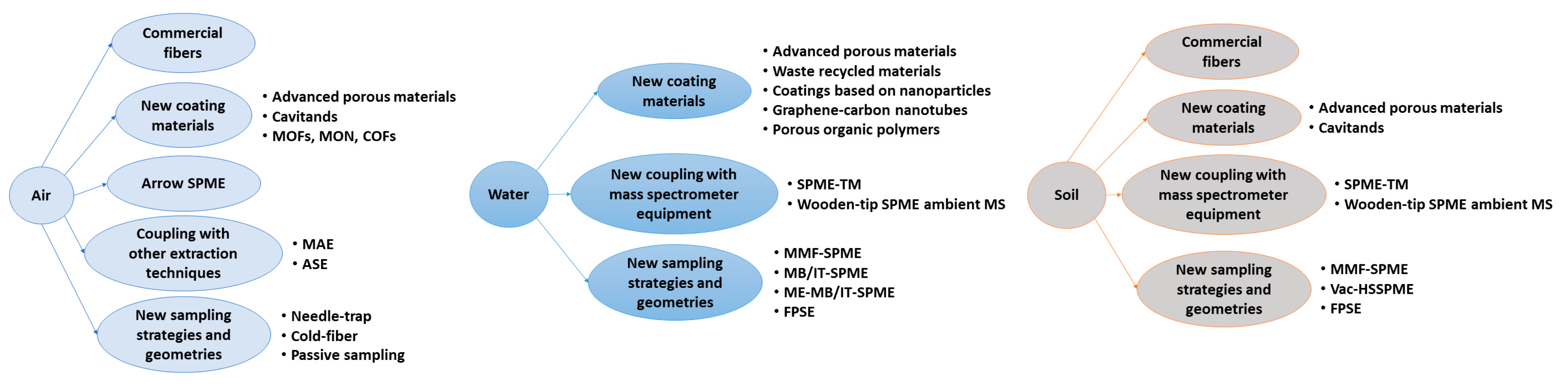
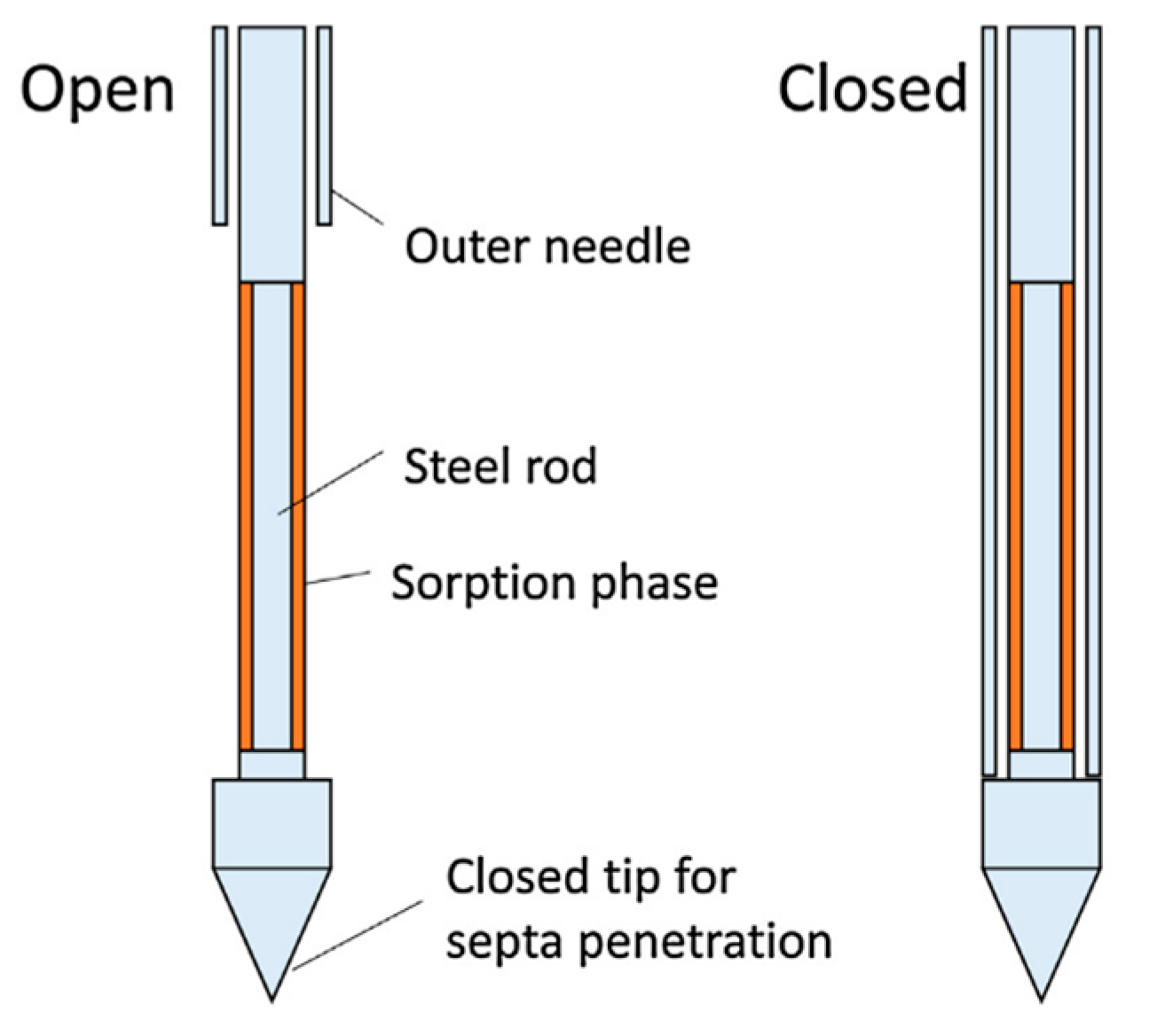
2.1. Air
2.2. Water
2.3. Soil
3. The Spread Step: Food Matrices and Drinking Water
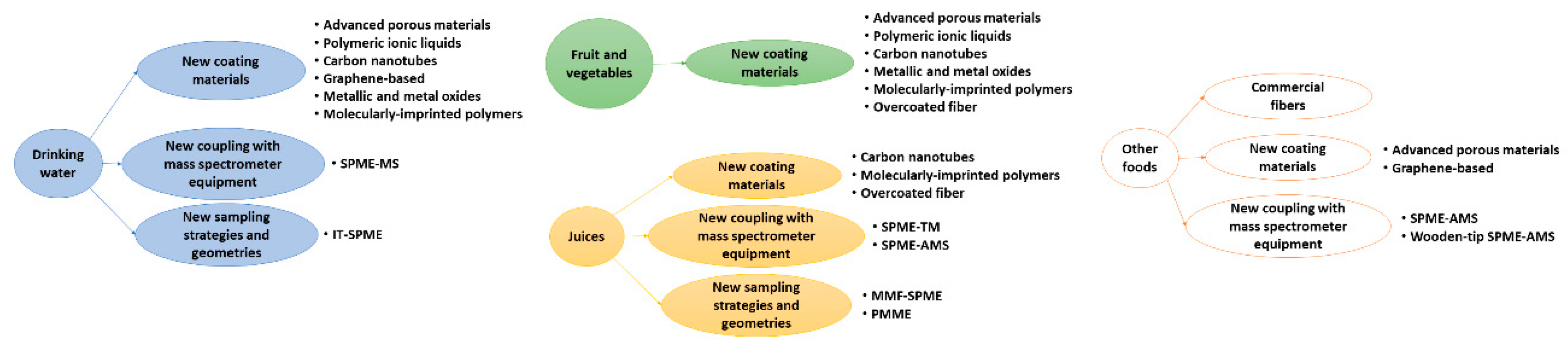
3.1. Drinking Water
3.2. Fruits and Vegetables
3.3. Juices
3.4. Other Foods
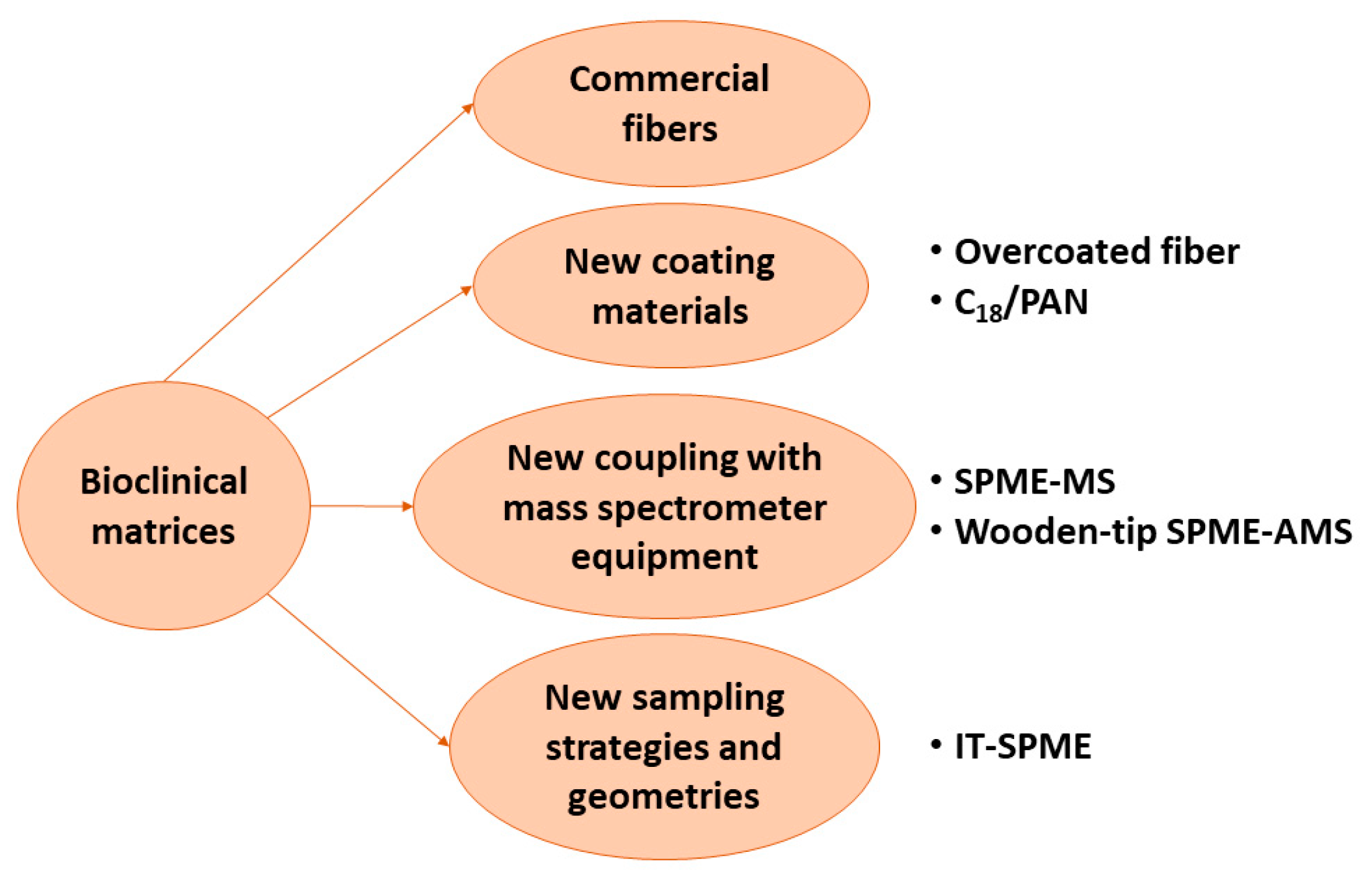
4. The Final Endpoint: Bio-Clinical Matrices
5. Concluding Remarks and Future Directions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| VOCs | volatile organic compounds |
| SVOCs | semivolatile organic compounds |
| DVB/CAR/PDMS | divinylbenzene/carboxen/polydimethylsiloxane |
| CAR/PDMS | carboxen/polydimethylsiloxane |
| PDMS | polydimethylsiloxane |
| PA | polyacrylate |
| PDMS/DVB | polydimethylsiloxane-divinylbenzene |
| PDMS/DVB/PDMS | polydimethylsiloxane-divinylbenzene-polydimethylsiloxane |
| PAHs | polycyclic aromatic hydrocarbons |
| THMs | trihalomethanes |
| PCBs | polychlorinated biphenyls |
| PFBHA | (pentafluorobenzyl)hydroxylamine |
| MtBSTFA | N-methyl-N-(tert.-butyldimethylsilyl) trifluoroacetamide |
| OPEs | organophosphate esters |
| BTEX | benzene, toluene, ethylbenzene and xylenes |
| PBDEs | polybrominated diphenyl ethers |
| PM | particulate matter |
| TWA | Time-Weighted Average sampling |
| MOF | metal-organic framework |
| COV | covalent organic frameworks |
| IL | Ionic liquids |
| PIL | polymeric ionic liquid |
| MMF-SPME | multiple monolithic fiber solid-phase microextraction |
| SPME-TM | Solid-Phase Micro Extraction-Transmission Mode |
| SPME-TM-DART/MS | Solid-Phase Micro Extraction-Transmission Mode Direct Analysis in Real-Time Mass Spectrometry |
References
- Carter, R.A.A.; West, N.; Heitz, A.; Joll, C.A. An analytical method for the analysis of trihalomethanes in ambient air using solid-phase microextraction gas chromatography-mass spectrometry: An application to indoor swimming pool complexes. Indoor Air 2019, 29, 499–509. [Google Scholar] [CrossRef] [PubMed]
- Saucedo-Lucero, J.O.; Revah, S. Monitoring key organic indoor pollutants and their elimination in a biotrickling biofilter. Environ. Sci. Pollut. Res. 2018, 25, 9806–9816. [Google Scholar] [CrossRef] [PubMed]
- Lucaire, V.; Schwartz, J.J.; Delhomme, O.; Ocampo-Torres, R.; Millet, M. A sensitive method using SPME pre-concentration for the quantification of aromatic amines in indoor air. Anal. Bioanal. Chem. 2018, 410, 1955–1963. [Google Scholar] [CrossRef] [PubMed]
- Ghislain, M.; Beigbeder, J.; Plaisance, H.; Desauziers, V. New sampling device for on-site measurement of SVOC gas-phase concentration at the emitting material surface. Anal. Bioanal. Chem. 2017, 409, 3199–3210. [Google Scholar] [CrossRef] [PubMed]
- Bourdin, D.; Desauziers, V. Development of SPME on-fiber derivatization for the sampling of formaldehyde and other carbonyl compounds in indoor air. Anal. Bioanal. Chem. 2014, 406, 317–328. [Google Scholar] [CrossRef]
- Helin, A.; Rönkkö, T.; Parshintsev, J.; Hartonen, K.; Schilling, B.; Läubli, T.; Riekkola, M.-L. Solid phase microextraction Arrow for the sampling of volatile amines in wastewater and atmosphere. J. Chromatogr. A 2015, 1426, 56–63. [Google Scholar] [CrossRef]
- Ruiz-Jimenez, J.; Zanca, N.; Lan, H.; Jussila, M.; Hartonen, K.; Riekkola, M.L. Aerial drone as a carrier for miniaturized air sampling systems. J. Chromatogr. A 2019, 1597, 202–208. [Google Scholar] [CrossRef]
- Celeiro, M.; Dagnac, T.; Llompart, M. Determination of priority and other hazardous substances in football fields of synthetic turf by gas chromatography-mass spectrometry: A health and environmental concern. Chemosphere 2018, 195, 201–211. [Google Scholar] [CrossRef]
- Baimatova, N.; Kenessov, B.; Koziel, J.A.; Carlsen, L.; Bektassov, M.; Demyanenko, O.P. Simple and accurate quantification of BTEX in ambient air by SPME and GC-MS. Talanta 2016, 154, 46–52. [Google Scholar] [CrossRef]
- Sayed Mohamed Zain, S.M.; Shaharudin, R.; Kamaluddin, M.A.; Daud, S.F. Determination of hydrogen cyanide in residential ambient air using SPME coupled with GC–MS. Atmos. Pollut. Res. 2017, 8, 678–685. [Google Scholar] [CrossRef]
- Van der Veen, I.; de Boer, J. Phosphorus flame retardants: Properties, production, environmental occurrence, toxicity and analysis. Chemosphere 2012, 88, 1119–1153. [Google Scholar] [CrossRef] [PubMed]
- Naccarato, A.; Elliani, R.; Sindona, G.; Tagarelli, A. Multivariate optimization of a microextraction by packed sorbent-programmed temperature vaporization-gas chromatography–tandem mass spectrometry method for organophosphate flame retardant analysis in environmental aqueous matrices. Anal. Bioanal. Chem. 2017, 409, 7105–7120. [Google Scholar] [CrossRef]
- Naccarato, A.; Tassone, A.; Moretti, S.; Elliani, R.; Sprovieri, F.; Pirrone, N.; Tagarelli, A. A green approach for organophosphate ester determination in airborne particulate matter: Microwave-assisted extraction using hydroalcoholic mixture coupled with solid-phase microextraction gas chromatography-tandem mass spectrometry. Talanta 2018, 189, 657–665. [Google Scholar] [CrossRef] [PubMed]
- Naccarato, A.; Gionfriddo, E.; Sindona, G.; Tagarelli, A. Simultaneous determination of benzothiazoles, benzotriazoles and benzosulfonamides by solid phase microextraction-gas chromatography-triple quadrupole mass spectrometry in environmental aqueous matrices and human urine. J. Chromatogr. A 2014, 1338, 164–173. [Google Scholar] [CrossRef] [PubMed]
- Raeppel, C.; Fabritius, M.; Nief, M.; Appenzeller, B.M.R.; Millet, M. Coupling ASE, sylilation and SPME-GC/MS for the analysis of current-used pesticides in atmosphere. Talanta 2014, 121, 24–29. [Google Scholar] [CrossRef] [PubMed]
- Gutiérrez-Serpa, A.; Pacheco-Fernández, I.; Pasán, J.; Pino, V. Metal–Organic Frameworks as Key Materials for Solid-Phase Microextraction Devices—A Review. Separations 2019, 6, 47. [Google Scholar] [CrossRef]
- Li, J.; Li, H.; Zhao, Y.; Wang, S.; Chen, X.; Zhao, R.S. A hollow microporous organic network as a fiber coating for solid-phase microextraction of short-chain chlorinated hydrocarbons. Microchim. Acta 2018, 185, 416. [Google Scholar] [CrossRef]
- Jia, Y.; Su, H.; Wang, Z.; Wong, Y.L.E.; Chen, X.; Wang, M.; Chan, T.W.D. Metal-Organic Framework@Microporous Organic Network as Adsorbent for Solid-Phase Microextraction. Anal. Chem. 2016, 88, 9364–9367. [Google Scholar] [CrossRef]
- Li, J.; Liu, Y.; Su, H.; Elaine Wong, Y.L.; Chen, X.; Dominic Chan, T.W.; Chen, Q. In situ hydrothermal growth of a zirconium-based porphyrinic metal-organic framework on stainless steel fibers for solid-phase microextraction of nitrated polycyclic aromatic hydrocarbons. Microchim. Acta 2017, 184, 3809–3815. [Google Scholar] [CrossRef]
- Zhang, S.; Yang, Q.; Li, Z.; Wang, W.; Wang, C.; Wang, Z. Covalent organic frameworks as a novel fiber coating for solid-phase microextraction of volatile benzene homologues. Anal. Bioanal. Chem. 2017, 409, 3429–3439. [Google Scholar] [CrossRef]
- Riboni, N.; Trzcinski, J.W.; Bianchi, F.; Massera, C.; Pinalli, R.; Sidisky, L.; Dalcanale, E.; Careri, M. Conformationally blocked quinoxaline cavitand as solid-phase microextraction coating for the selective detection of BTEX in air. Anal. Chim. Acta 2016, 905, 79–84. [Google Scholar] [CrossRef]
- Heidari, M.; Bahrami, A.; Ghiasvand, A.R.; Shahna, F.G.; Soltanian, A.R.; Rafieiemam, M. Application of graphene nanoplatelets silica composite, prepared by sol-gel technology, as a novel sorbent in two microextraction techniques. J. Sep. Sci. 2015, 38, 4225–4232. [Google Scholar] [CrossRef] [PubMed]
- Dias, C.M.; Menezes, H.C.; Cardeal, Z.L. Environmental and biological determination of acrolein using new cold fiber solid phase microextraction with gas chromatography mass spectrometry. Anal. Bioanal. Chem. 2017, 409, 2821–2828. [Google Scholar] [CrossRef] [PubMed]
- Kenessov, B.; Koziel, J.A.; Baimatova, N.; Demyanenko, O.P.; Derbissalin, M. Optimization of time-weighted average air sampling by solid-phase microextraction fibers using finite element analysis software. Molecules 2018, 23, 2736. [Google Scholar] [CrossRef] [PubMed]
- Tursumbayeva, M.; Koziel, J.A.; Maurer, D.L.; Kenessov, B.; Rice, S. Development of Time-Weighted Average Sampling of Odorous Volatile Organic Compounds in Air with Solid-Phase Microextraction Fiber Housed inside a GC Glass Liner: Proof of concept. Molecules 2019, 24, 406. [Google Scholar] [CrossRef]
- Carasek, E.; Morés, L.; Merib, J. Basic principles, recent trends and future directions of microextraction techniques for the analysis of aqueous environmental samples. Trends Environ. Anal. Chem. 2018, 19, e00060. [Google Scholar] [CrossRef]
- Gómez-Ríos, G.A.; Gionfriddo, E.; Poole, J.; Pawliszyn, J. Ultrafast Screening and Quantitation of Pesticides in Food and Environmental Matrices by Solid-Phase Microextraction-Transmission Mode (SPME-TM) and Direct Analysis in Real Time (DART). Anal. Chem. 2017, 89, 7240–7248. [Google Scholar] [CrossRef]
- Wang, X.; Li, X.; Li, Z.; Zhang, Y.; Bai, Y.; Liu, H. Online coupling of in-tube solid-phase microextraction with direct analysis in real time mass spectrometry for rapid determination of triazine herbicides in water using carbon-nanotubes-incorporated polymer monolith. Anal. Chem. 2014, 86, 4739–4747. [Google Scholar] [CrossRef]
- Deng, J.; Yang, Y.; Fang, L.; Lin, L.; Zhou, H.; Luan, T. Coupling solid-phase microextraction with ambient mass spectrometry using surface coated wooden-tip probe for rapid analysis of ultra trace perfluorinated compounds in complex samples. Anal. Chem. 2014, 86, 11159–11166. [Google Scholar] [CrossRef]
- Grandy, J.J.; Lashgari, M.; Vander Heide, H.; Poole, J.; Pawliszyn, J. Introducing a mechanically robust SPME sampler for the on-site sampling and extraction of a wide range of untargeted pollutants in environmental waters. Environ. Pollut. 2019, 252, 825–834. [Google Scholar] [CrossRef]
- Bu, Y.; Feng, J.; Sun, M.; Zhou, C.; Luo, C. Facile and efficient poly(ethylene terephthalate) fibers-in-tube for online solid-phase microextraction towards polycyclic aromatic hydrocarbons. Anal. Bioanal. Chem. 2016, 408, 4871–4882. [Google Scholar] [CrossRef] [PubMed]
- Fu, M.; Xing, H.; Chen, X.; Zhao, R.; Zhi, C.; Wu, C.L. Boron nitride nanotubes as novel sorbent for solid-phase microextraction of polycyclic aromatic hydrocarbons in environmental water samples. Anal. Bioanal. Chem. 2014, 406, 5751–5754. [Google Scholar] [CrossRef] [PubMed]
- Yin, L.; Hu, Q.; Mondal, S.; Xu, J.; Ouyang, G. Peanut shell-derived biochar materials for effective solid-phase microextraction of polycyclic aromatic hydrocarbons in environmental waters. Talanta 2019, 202, 90–95. [Google Scholar] [CrossRef] [PubMed]
- Sun, S.; Huang, L.; Xiao, H.; Shuai, Q.; Hu, S. In situ self-transformation metal into metal-organic framework membrane for solid-phase microextraction of polycyclic aromatic hydrocarbons. Talanta 2019, 202, 145–151. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.; Lei, Y.; Song, H. Exploration of metal-organic framework MOF-177 coated fibers for headspace solid-phase microextraction of polychlorinated biphenyls and polycyclic aromatic hydrocarbons. Talanta 2015, 144, 369–374. [Google Scholar] [CrossRef]
- Kong, J.; Zhu, F.; Huang, W.; He, H.; Hu, J.; Sun, C.; Xian, Q.; Yang, S. Sol–gel based metal-organic framework zeolite imidazolate framework-8 fibers for solid-phase microextraction of nitro polycyclic aromatic hydrocarbons and polycyclic aromatic hydrocarbons in water samples. J. Chromatogr. A 2019. [Google Scholar] [CrossRef]
- Hu, X.; Liu, C.; Li, J.; Luo, R.; Jiang, H.; Sun, X.; Shen, J.; Han, W.; Wang, L. Hollow mesoporous carbon spheres-based fiber coating for solid-phase microextraction of polycyclic aromatic hydrocarbons. J. Chromatogr. A 2017, 1520, 58–64. [Google Scholar] [CrossRef]
- Bagheri, H.; Soofi, G.; Javanmardi, H.; Karimi, M. A 3D nanoscale polyhedral oligomeric silsesquioxanes network for microextraction of polycyclic aromatic hydrocarbons. Microchim. Acta 2018, 185, 418. [Google Scholar] [CrossRef]
- Guo, M.; Song, W.; Wang, T.; Li, Y.; Wang, X.; Du, X. Phenyl-functionalization of titanium dioxide-nanosheets coating fabricated on a titanium wire for selective solid-phase microextraction of polycyclic aromatic hydrocarbons from environment water samples. Talanta 2015, 144, 998–1006. [Google Scholar] [CrossRef]
- Gutiérrez-Serpa, A.; Napolitano-Tabares, P.I.; Pino, V.; Jiménez-Moreno, F.; Jiménez-Abizanda, A.I. Silver nanoparticles supported onto a stainless steel wire for direct-immersion solid-phase microextraction of polycyclic aromatic hydrocarbons prior to their determination by GC-FID. Microchim. Acta 2018, 185, 341. [Google Scholar] [CrossRef]
- Wang, X.; Sheng, W.R.; Jiao, X.Y.; Zhao, R.S.; Wang, M.L.; Lin, J.M. Zinc(II)-based metal–organic nanotubes coating for high sensitive solid phase microextraction of nitro-polycyclic aromatic hydrocarbons. Talanta 2018, 186, 561–567. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.L.; Chen, X.F.; Wang, X.; Zhou, J.B.; Zhao, R.S. Sensitive determination of polychlorinated biphenyls in environmental water samples by headspace solid-phase microextraction with bamboo charcoal@iron oxide black fibers prior to gas chromatography with tandem mass spectrometry. J. Sep. Sci. 2014, 37, 1496–1502. [Google Scholar] [CrossRef] [PubMed]
- Sheng, W.R.; Chen, Y.; Wang, S.S.; Wang, X.L.; Wang, M.L.; Zhao, R.S. Cadmium(II)-based metal–organic nanotubes as solid-phase microextraction coating for ultratrace-level analysis of polychlorinated biphenyls in seawater samples. Anal. Bioanal. Chem. 2016, 408, 8289–8297. [Google Scholar] [CrossRef] [PubMed]
- Cheng, H.; Wang, F.; Bian, Y.; Ji, R.; Song, Y.; Jiang, X. Co- and self-activated synthesis of tailored multimodal porous carbons for solid-phase microextraction of chlorobenzenes and polychlorinated biphenyls. J. Chromatogr. A 2019, 1585, 1–9. [Google Scholar] [CrossRef]
- Li, Q.L.; Wang, X.; Liu, Y.L.; Chen, X.F.; Wang, M.L.; Zhao, R.S. Feasibility of metal-organic nanotubes [Cu3(μ3-O)(μ-OH)(triazolate)2]+-coated fibers for solid-phase microextraction of polychlorinated biphenyls in water samples. J. Chromatogr. A 2014, 1374, 58–65. [Google Scholar] [CrossRef]
- Ling, X.; Chen, Z. Immobilization of zeolitic imidazolate frameworks with assist of electrodeposited zinc oxide layer and application in online solid-phase microextraction of Sudan dyes. Talanta 2019, 192, 142–146. [Google Scholar] [CrossRef]
- Pei, M.; Zhu, X.; Huang, X. Mixed functional monomers-based monolithic adsorbent for the effective extraction of sulfonylurea herbicides in water and soil samples. J. Chromatogr. A 2018, 1531, 13–21. [Google Scholar] [CrossRef]
- Zhang, Z.; Mei, M.; Huang, Y.; Huang, X.; Huang, H.; Ding, Y. Facile preparation of a polydopamine-based monolith for multiple monolithic fiber solid-phase microextraction of triazine herbicides in environmental water samples. J. Sep. Sci. 2017, 40, 733–743. [Google Scholar] [CrossRef]
- Prieto, A.; Rodil, R.; Quintana, J.B.; Cela, R.; Möder, M.; Rodríguez, I. Evaluation of polyethersulfone performance for the microextraction of polar chlorinated herbicides from environmental water samples. Talanta 2014, 122, 264–271. [Google Scholar] [CrossRef]
- Bagheri, H.; Amanzadeh, H.; Yamini, Y.; Masoomi, M.Y.; Morsali, A.; Salar-Amoli, J.; Hassan, J. A nanocomposite prepared from a zinc-based metal-organic framework and polyethersulfone as a novel coating for the headspace solid-phase microextraction of organophosphorous pesticides. Microchim. Acta 2018, 185, 62. [Google Scholar] [CrossRef]
- Gutiérrez-Serpa, A.; Rocío-Bautista, P.; Pino, V.; Jiménez-Moreno, F.; Jiménez-Abizanda, A.I. Gold nanoparticles based solid-phase microextraction coatings for determining organochlorine pesticides in aqueous environmental samples. J. Sep. Sci. 2017, 40, 2009–2021. [Google Scholar] [CrossRef] [PubMed]
- Zheng, J.; Liang, Y.; Liu, S.; Jiang, R.; Zhu, F.; Wu, D.; Ouyang, G. Simple fabrication of solid phase microextraction fiber employing nitrogen-doped ordered mesoporous polymer by in situ polymerization. J. Chromatogr. A 2016, 1427, 22–28. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Mei, M.; Huang, X. Fabrication of boron-rich multiple monolithic fibers for the solid-phase microextraction of carbamate pesticide residues in complex samples. J. Sep. Sci. 2019, 42, 878–887. [Google Scholar] [CrossRef] [PubMed]
- Pang, J.; Liao, Y.; Huang, X.; Ye, Z.; Yuan, D. Metal-organic framework-monolith composite-based in-tube solid phase microextraction on-line coupled to high-performance liquid chromatography-fluorescence detection for the highly sensitive monitoring of fluoroquinolones in water and food samples. Talanta 2019, 199, 499–506. [Google Scholar] [CrossRef]
- Peng, J.; Liu, D.; Shi, T.; Tian, H.; Hui, X.; He, H. Molecularly imprinted polymers based stir bar sorptive extraction for determination of cefaclor and cefalexin in environmental water. Anal. Bioanal. Chem. 2017, 409, 4157–4166. [Google Scholar] [CrossRef]
- Barahona, F.; Albero, B.; Tadeo, J.L.; Martín-Esteban, A. Molecularly imprinted polymer-hollow fiber microextraction of hydrophilic fluoroquinolone antibiotics in environmental waters and urine samples. J. Chromatogr. A 2019, 1587, 42–49. [Google Scholar] [CrossRef]
- Kazemi, E.; Haji Shabani, A.M.; Dadfarnia, S. Application of graphene oxide-silica composite reinforced hollow fibers as a novel device for pseudo-stir bar solid phase microextraction of sulfadiazine in different matrices prior to its spectrophotometric determination. Food Chem. 2017, 221, 783–789. [Google Scholar] [CrossRef]
- Mei, M.; Huang, X.; Yu, J.; Yuan, D. Sensitive monitoring of trace nitrophenols in water samples using multiple monolithic fiber solid phase microextraction and liquid chromatographic analysis. Talanta 2015, 134, 89–97. [Google Scholar] [CrossRef]
- Li, Q.L.; Huang, F.; Wang, X.L.; Wang, X.; Zhao, R.S. Multiple-helix cobalt(II)-based metal-organic nanotubes on stainless steel fibers for solid-phase microextraction of chlorophenol and nitrophenols from water samples. Microchim. Acta 2017, 184, 1817–1825. [Google Scholar] [CrossRef]
- Gionfriddo, E.; Naccarato, A.; Sindona, G.; Tagarelli, A. Determination of hydrazine in drinking water: Development and multivariate optimization of a rapid and simple solid phase microextraction-gas chromatography-triple quadrupole mass spectrometry protocol. Anal. Chim. Acta 2014, 835, 37–45. [Google Scholar] [CrossRef]
- Li, L.; Guo, R.; Li, Y.; Guo, M.; Wang, X.; Du, X. In situ growth and phenyl functionalization of titania nanoparticles coating for solid-phase microextraction of ultraviolet filters in environmental water samples followed by high performance liquid chromatography-UV detection. Anal. Chim. Acta 2015, 867, 38–46. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Li, Y.; Liu, H.; Wang, X.; Du, X. Electrodeposition of gold nanoparticles onto an etched stainless steel wire followed by a self-assembled monolayer of octanedithiol as a fiber coating for selective solid-phase microextraction. J. Chromatogr. A 2014, 1372, 25–33. [Google Scholar] [CrossRef] [PubMed]
- Zhou, S.; Wang, H.; Jin, P.; Wang, Z.; Wang, X.; Du, X. Electrophoretic deposition strategy for the fabrication of highly stable functionalized silica nanoparticle coatings onto nickel-titanium alloy wires for selective solid-phase microextraction. J. Sep. Sci. 2017, 40, 4796–4804. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Zhang, M.; Yang, Y.; Wang, X.; Du, X. Electrochemical in situ fabrication of titanium dioxide-nanosheets on a titanium wire as a novel coating for selective solid-phase microextraction. J. Chromatogr. A 2014, 1358, 60–67. [Google Scholar] [CrossRef]
- Song, W.; Guo, M.; Zhang, Y.; Zhang, M.; Wang, X.; Du, X. Fabrication and application of zinc-zinc oxide nanosheets coating on an etched stainless steel wire as a selective solid-phase microextraction fiber. J. Chromatogr. A 2015, 1384, 28–36. [Google Scholar] [CrossRef]
- Bu, Y.; Feng, J.; Wang, X.; Tian, Y.; Sun, M.; Luo, C. In situ hydrothermal growth of polyaniline coating for in-tube solid-phase microextraction towards ultraviolet filters in environmental water samples. J. Chromatogr. A 2017, 1483, 48–55. [Google Scholar] [CrossRef]
- Wang, H.; Du, J.; Zhen, Q.; Zhang, R.; Wang, X.; Du, X. Selective solid-phase microextraction of ultraviolet filters in environmental water with oriented ZnO nanosheets coated nickel-titanium alloy fibers followed by high performance liquid chromatography with UV detection. Talanta 2019, 191, 193–201. [Google Scholar] [CrossRef]
- Mei, M.; Huang, X. Online analysis of five organic ultraviolet filters in environmental water samples using magnetism-enhanced monolith-based in-tube solid phase microextraction coupled with high-performance liquid chromatography. J. Chromatogr. A 2017, 1525, 1–9. [Google Scholar] [CrossRef]
- Ma, M.; Wang, H.; Zhen, Q.; Zhang, M.; Du, X. Development of nitrogen-enriched carbonaceous material coated titania nanotubes array as a fiber coating for solid-phase microextraction of ultraviolet filters in environmental water. Talanta 2017, 167, 118–125. [Google Scholar] [CrossRef]
- Wang, H.; Song, W.; Zhang, M.; Zhen, Q.; Guo, M.; Zhang, Y.; Du, X. Hydrothermally grown and self-assembled modified titanium and nickel oxide composite nanosheets on Nitinol-based fibers for efficient solid phase microextraction. J. Chromatogr. A 2016, 1468, 33–41. [Google Scholar] [CrossRef]
- Yang, Y.; Guo, M.; Zhang, Y.; Song, W.; Li, Y.; Wang, X.; Du, X. Self-assembly of alkyldithiols on a novel dendritic silver nanostructure electrodeposited on a stainless steel wire as a fiber coating for solid-phase microextraction. RSC Adv. 2015, 5, 71859–71867. [Google Scholar] [CrossRef]
- Li, Y.; Ma, M.; Zhang, M.; Yang, Y.; Wang, X.; Du, X. In situ anodic growth of rod-like TiO2 coating on a Ti wire as a selective solid-phase microextraction fiber. RSC Adv. 2014, 4, 53820–53827. [Google Scholar] [CrossRef]
- Zhang, Y.; Song, W.; Yang, Y.; Guo, M.; Wang, X.; Du, X. Self-assembly of mercaptoundecanol on cedar-like Au nanoparticle coated stainless steel fiber for selective solid-phase microextraction. Anal. Methods 2015, 7, 7680–7689. [Google Scholar] [CrossRef]
- Naccarato, A.; Gionfriddo, E.; Elliani, R.; Pawliszyn, J.; Sindona, G.; Tagarelli, A. Investigating the robustness and extraction performance of a matrix-compatible solid-phase microextraction coating in human urine and its application to assess 2–6-ring polycyclic aromatic hydrocarbons using GC–MS/MS. J. Sep. Sci. 2018, 41, 929–939. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Pan, G.; Yang, H.; Cai, Z.; Zhu, F.; Ouyang, G. Determination and elimination of hazardous pollutants by exploitation of a Prussian blue nanoparticles-graphene oxide composite. Anal. Chim. Acta 2019, 1054, 17–25. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Xie, L.; Hu, Q.; Yang, H.; Pan, G.; Zhu, F.; Yang, S.; Ouyang, G. A tri-metal centered metal-organic framework for solid-phase microextraction of environmental contaminants with enhanced extraction efficiency. Anal. Chim. Acta 2017, 987, 38–46. [Google Scholar] [CrossRef]
- Cen, J.; Wei, S.; Nan, H.; Xu, J.; Huang, Z.; Liu, S.; Hu, Q.; Yan, J.; Ouyang, G. Incorporation of carbon nanotubes into graphene for highly efficient solid-phase microextraction of benzene homologues. Microchem. J. 2018, 139, 203–209. [Google Scholar] [CrossRef]
- Liu, S.; Hu, Q.; Zheng, J.; Xie, L.; Wei, S.; Jiang, R.; Zhu, F.; Liu, Y.; Ouyang, G. Knitting aromatic polymers for efficient solid-phase microextraction of trace organic pollutants. J. Chromatogr. A 2016, 1450, 9–16. [Google Scholar] [CrossRef]
- Huang, Z.; Liu, S.; Xu, J.; Yin, L.; Zheng, J.; Zhou, N.; Ouyang, G. Porous organic polymers with different pore structures for sensitive solid-phase microextraction of environmental organic pollutants. Anal. Chim. Acta 2017, 989, 21–28. [Google Scholar] [CrossRef]
- Xie, X.; Wang, J.; Zheng, J.; Huang, J.; Ni, C.; Cheng, J.; Hao, Z.; Ouyang, G. Low-cost Scholl-coupling microporous polymer as an efficient solid-phase microextraction coating for the detection of light aromatic compounds. Anal. Chim. Acta 2018, 1029, 30–36. [Google Scholar] [CrossRef]
- Mei, M.; Huang, X.; Yuan, D. Multiple monolithic fiber solid-phase microextraction: A new extraction approach for aqueous samples. J. Chromatogr. A 2014, 1345, 29–36. [Google Scholar] [CrossRef] [PubMed]
- Mei, M.; Huang, X.; Liao, K.; Yuan, D. Sensitive monitoring of benzoylurea insecticides in water and juice samples treated with multiple monolithic fiber solid-phase microextraction and liquid chromatographic analysis. Anal. Chim. Acta 2015, 860, 29–36. [Google Scholar] [CrossRef] [PubMed]
- Pei, M.; Zhang, Z.; Huang, X.; Wu, Y. Fabrication of a polymeric ionic liquid-based adsorbent for multiple monolithic fiber solid-phase microextraction of endocrine disrupting chemicals in complicated samples. Talanta 2017, 165, 152–160. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Lu, M.; Li, H.; Bai, M.; Huang, X. Sensitive determination of perfluoroalkane sulfonamides in water and urine samples by multiple monolithic fiber solid-phase microextraction and liquid chromatography tandem mass spectrometry. Talanta 2019, 192, 24–31. [Google Scholar] [CrossRef] [PubMed]
- Mei, M.; Huang, X.; Luo, Q.; Yuan, D. Magnetism-Enhanced Monolith-Based In-Tube Solid Phase Microextraction. Anal. Chem. 2016, 88, 1900–1907. [Google Scholar] [CrossRef]
- Kabir, A.; Furton, K.G. Fabric Phase Sorptive Extractor (FPSE); US Patent and Trademark Office: Alexandria, VA, USA, 2014. [Google Scholar]
- Racamonde, I.; Rodil, R.; Quintana, J.B.; Sieira, B.J.; Kabir, A.; Furton, K.G.; Cela, R. Fabric phase sorptive extraction: A new sorptive microextraction technique for the determination of non-steroidal anti-inflammatory drugs from environmental water samples. Anal. Chim. Acta 2015, 865, 22–30. [Google Scholar] [CrossRef]
- Santana-Viera, S.; Guedes-Alonso, R.; Sosa-Ferrera, Z.; Santana-Rodríguez, J.J.; Kabir, A.; Furton, K.G. Optimization and application of fabric phase sorptive extraction coupled to ultra-high performance liquid chromatography tandem mass spectrometry for the determination of cytostatic drug residues in environmental waters. J. Chromatogr. A 2017, 1529, 39–49. [Google Scholar] [CrossRef]
- Roldán-Pijuán, M.; Lucena, R.; Cárdenas, S.; Valcárcel, M.; Kabir, A.; Furton, K.G. Stir fabric phase sorptive extraction for the determination of triazine herbicides in environmental waters by liquid chromatography. J. Chromatogr. A 2015, 1376, 35–45. [Google Scholar] [CrossRef]
- Lakade, S.S.; Borrull, F.; Furton, K.G.; Kabir, A.; Marcé, R.M.; Fontanals, N. Dynamic fabric phase sorptive extraction for a group of pharmaceuticals and personal care products from environmental waters. J. Chromatogr. A 2016, 1456, 19–26. [Google Scholar] [CrossRef]
- Kenessov, B.; Koziel, J.A.; Bakaikina, N.V.; Orazbayeva, D. Perspectives and challenges of on-site quantification of organic pollutants in soils using solid-phase microextraction. TrAC Trends Anal. Chem. 2016, 85, 111–122. [Google Scholar] [CrossRef]
- Bianchi, F.; Bedini, A.; Riboni, N.; Pinalli, R.; Gregori, A.; Sidisky, L.; Dalcanale, E.; Careri, M. Cavitand-based solid-phase microextraction coating for the selective detection of nitroaromatic explosives in air and soil. Anal. Chem. 2014, 86, 10646–10652. [Google Scholar] [CrossRef] [PubMed]
- Yao, W.; Fan, Z.; Zhang, S. Preparation of metal-organic framework UiO-66-incorporated polymer monolith for the extraction of trace levels of fungicides in environmental water and soil samples. J. Sep. Sci. 2019, 42, 2679–2686. [Google Scholar] [CrossRef] [PubMed]
- Gao, J.; Huang, C.; Lin, Y.; Tong, P.; Zhang, L. In situ solvothermal synthesis of metal-organic framework coated fiber for highly sensitive solid-phase microextraction of polycyclic aromatic hydrocarbons. J. Chromatogr. A 2016, 1436, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Zang, X.H.; Wang, J.T.; Wang, C.; Wu, Q.H.; Wang, Z. Porous carbon derived from aluminum-based metal organic framework as a fiber coating for the solid-phase microextraction of polycyclic aromatic hydrocarbons from water and soil. Microchim. Acta 2015, 182, 2353–2359. [Google Scholar] [CrossRef]
- Zhang, N.; Huang, C.; Tong, P.; Feng, Z.; Wu, X.; Zhang, L. Moisture stable Ni-Zn MOF/g-C 3 N 4 nanoflowers: A highly efficient adsorbent for solid-phase microextraction of PAHs. J. Chromatogr. A 2018, 1556, 37–46. [Google Scholar] [CrossRef]
- Zhang, N.; Huang, C.; Feng, Z.; Chen, H.; Tong, P.; Wu, X.; Zhang, L. Metal-organic framework-coated stainless steel fiber for solid-phase microextraction of polychlorinated biphenyls. J. Chromatogr. A 2018, 1570, 10–18. [Google Scholar] [CrossRef]
- Wu, Y.Y.; Yang, C.X.; Yan, X.P. Fabrication of metal-organic framework MIL-88B films on stainless steel fibers for solid-phase microextraction of polychlorinated biphenyls. J. Chromatogr. A 2014, 1334, 1–8. [Google Scholar] [CrossRef]
- Zhang, C.Y.; Yan, Z.G.; Zhou, Y.Y.; Wang, L.; Xie, Y.B.; Bai, L.P.; Zhou, H.Y.; Li, F.S. Embedment of Ag(I)-organic frameworks into silica gels for microextraction of polybrominated diphenyl ethers in soils. J. Chromatogr. A 2015, 1383, 18–24. [Google Scholar] [CrossRef]
- Bakaikina, N.V.; Kenessov, B.; Ul’yanovskii, N.V.; Kosyakov, D.S. Quantification of transformation products of rocket fuel unsymmetrical dimethylhydrazine in soils using SPME and GC-MS. Talanta 2018, 184, 332–337. [Google Scholar] [CrossRef]
- Peruga, A.; Beltrán, J.; López, F.; Hernández, F. Determination of methylisothiocyanate in soil and water by HS-SPME followed by GC-MS-MS with a triple quadrupole. Anal. Bioanal. Chem. 2014, 406, 5271–5282. [Google Scholar] [CrossRef]
- Yegemova, S.; Bakaikina, N.V.; Kenessov, B.; Koziel, J.A.; Nauryzbayev, M. Determination of 1-methyl-1H-1,2,4-triazole in soils contaminated by rocket fuel using solid-phase microextraction, isotope dilution and gas chromatography-mass spectrometry. Talanta 2015, 143, 226–233. [Google Scholar] [CrossRef] [PubMed]
- Škulcová, L.; Hale, S.E.; Hofman, J.; Bielská, L. Laboratory versus field soil aging: Impact on DDE bioavailability and sorption. Chemosphere 2017, 186, 235–242. [Google Scholar] [CrossRef] [PubMed]
- Shen, H.; Li, W.; Graham, S.E.; Starr, J.M. The role of soil and house dust physicochemical properties in determining the post ingestion bioaccessibility of sorbed polychlorinated biphenyls. Chemosphere 2019, 217, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Ma, L.; Yates, S.R. Dissolved organic matter and estrogen interactions regulate estrogen removal in the aqueous environment: A review. Sci. Total Environ. 2018, 640–641, 529–542. [Google Scholar] [CrossRef] [PubMed]
- Guo, M.; Gong, Z.; Allinson, G.; Tai, P.; Miao, R.; Li, X.; Jia, C.; Zhuang, J. Variations in the bioavailability of polycyclic aromatic hydrocarbons in industrial and agricultural soils after bioremediation. Chemosphere 2016, 144, 1513–1520. [Google Scholar] [CrossRef] [PubMed]
- Kumar, R.; Gaurav; Kabir, A.; Furton, K.G.; Malik, A.K. Development of a fabric phase sorptive extraction with high-performance liquid chromatography and ultraviolet detection method for the analysis of alkyl phenols in environmental samples. J. Sep. Sci. 2015, 38, 3228–3238. [Google Scholar] [CrossRef]
- Stanisz, E.; Werner, J.; Matusiewicz, H. Task specific ionic liquid-coated PTFE tube for solid-phase microextraction prior to chemical and photo-induced mercury cold vapour generation. Microchem. J. 2014, 114, 229–237. [Google Scholar] [CrossRef]
- Psillakis, E. Vacuum-assisted headspace solid-phase microextraction: A tutorial review. Anal. Chim. Acta 2017, 986, 12–24. [Google Scholar] [CrossRef]
- Wu, M.; Wang, L.; Zhao, F.; Zeng, B. Ionic liquid polymer functionalized carbon nanotubes-coated polyaniline for the solid-phase microextraction of benzene derivatives. RSC Adv. 2015, 5, 99483–99490. [Google Scholar] [CrossRef]
- Sarafraz-Yazdi, A.; Rounaghi, G.; Vatani, H.; Razavipanah, I.; Amiri, A. Headspace solid phase microextraction of volatile aromatic hydrocarbons using a steel wire coated with an electrochemically prepared nanocomposite consisting of polypyrrole, carbon nanotubes, and titanium oxide. Microchim. Acta 2015, 182, 217–225. [Google Scholar] [CrossRef]
- Song, X.L.; Chen, Y.; Yuan, J.P.; Qin, Y.J.; Zhao, R.S.; Wang, X. Carbon nanotube composite microspheres as a highly efficient solid-phase microextraction coating for sensitive determination of phthalate acid esters in water samples. J. Chromatogr. A 2016, 1468, 17–22. [Google Scholar] [CrossRef]
- Di Pietro Roux, K.C.; Jasinski, É.F.; Merib, J.; Sartorelli, M.L.; Carasek, E. Application of a robust solid-phase microextraction fiber consisting of NiTi wires coated with polypyrrole for the determination of haloanisoles in water and wine. Anal. Methods 2016, 8, 5503–5510. [Google Scholar] [CrossRef]
- Huang, S.; Xu, J.; Tao, X.; Chen, X.; Zhu, F.; Wang, Y.; Jiang, R.; Ouyang, G. Fabrication of polyaniline/silver composite coating as a dual-functional platform for microextraction and matrix-free laser desorption/ionization. Talanta 2017, 172, 155–161. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Ma, R.; Bai, S.; Wang, C.; Wang, Z. A solid phase microextraction fiber coated with graphene-poly(ethylene glycol) composite for the extraction of volatile aromatic compounds from water samples. Talanta 2014, 119, 498–504. [Google Scholar] [CrossRef]
- Sarafraz-Yazdi, A.; Yekkebashi, A. Development of a poly(ethylene glycol)-graphene oxide sol-gel coating for solid-phase microextraction of aromatic amines in water samples with a gas chromatography-flame ionization detector method. New J. Chem. 2015, 39, 1287–1294. [Google Scholar] [CrossRef]
- De Toffoli, A.L.; Fumes, B.H.; Lanças, F.M. Packed in-tube solid phase microextraction with graphene oxide supported on aminopropyl silica: Determination of target triazines in water samples. J. Environ. Sci. Health Part B Pestic. Food Contam. Agric. Wastes 2018, 53, 434–440. [Google Scholar] [CrossRef] [PubMed]
- Tan, F.; Zhao, C.; Li, L.; Liu, M.; He, X.; Gao, J. Graphene oxide based in-tube solid-phase microextraction combined with liquid chromatography tandem mass spectrometry for the determination of triazine herbicides in water. J. Sep. Sci. 2015, 38, 2312–2319. [Google Scholar] [CrossRef]
- Li, S.; Lu, C.; Zhu, F.; Jiang, R.; Ouyang, G. Preparation of C18 composite solid-phase microextraction fiber and its application to the determination of organochlorine pesticides in water samples. Anal. Chim. Acta 2015, 873, 57–62. [Google Scholar] [CrossRef]
- Jiang, H.; Li, J.; Jiang, M.; Lu, R.; Shen, J.; Sun, X.; Han, W.; Wang, L. Ordered mesoporous carbon film as an effective solid-phase microextraction coating for determination of benzene series from aqueous media. Anal. Chim. Acta 2015, 888, 85–93. [Google Scholar] [CrossRef]
- Saraji, M.; Shahvar, A. Metal-organic aerogel as a coating for solid-phase microextraction. Anal. Chim. Acta 2017, 973, 51–58. [Google Scholar] [CrossRef]
- Bagheri, H.; Javanmardi, H.; Abbasi, A.; Banihashemi, S. A metal organic framework-polyaniline nanocomposite as a fiber coating for solid phase microextraction. J. Chromatogr. A 2016, 1431, 27–35. [Google Scholar] [CrossRef] [PubMed]
- Xie, L.; Liu, S.; Han, Z.; Jiang, R.; Zhu, F.; Xu, W.; Su, C.; Ouyang, G. Amine-functionalized MIL-53(Al)-coated stainless steel fiber for efficient solid-phase microextraction of synthetic musks and organochlorine pesticides in water samples. Anal. Bioanal. Chem. 2017, 409, 5239–5247. [Google Scholar] [CrossRef] [PubMed]
- Zang, X.; Zhang, X.; Chang, Q.; Li, S.; Wang, C.; Wang, Z. Metal–organic framework UiO-67-coated fiber for the solid-phase microextraction of nitrobenzene compounds from water. J. Sep. Sci. 2016, 39, 2770–2776. [Google Scholar] [CrossRef]
- Zang, X.; Zhang, G.; Chang, Q.; Zhang, X.; Wang, C.; Wang, Z. Metal organic framework MIL-101 coated fiber for headspace solid phase microextraction of volatile aromatic compounds. Anal. Methods 2015, 7, 918–923. [Google Scholar] [CrossRef]
- Zhang, J.; Zhang, W.; Bao, T.; Chen, Z. Polydopamine-based immobilization of zeolitic imidazolate framework-8 for in-tube solid-phase microextraction. J. Chromatogr. A 2015, 1388, 9–16. [Google Scholar] [CrossRef] [PubMed]
- Kang, H.; Mao, Y.; Wang, X.; Zhang, Y.; Wu, J.; Wang, H. Disposable ionic liquid-coated etched stainless steel fiber for headspace solid-phase microextraction of organophosphorus flame retardants from water samples. RSC Adv. 2015, 5, 41934–41940. [Google Scholar] [CrossRef]
- Pacheco-Fernández, I.; Najafi, A.; Pino, V.; Anderson, J.L.; Ayala, J.H.; Afonso, A.M. Utilization of highly robust and selective crosslinked polymeric ionic liquid-based sorbent coatings in direct-immersion solid-phase microextraction and high-performance liquid chromatography for determining polar organic pollutants in waters. Talanta 2016, 158, 125–133. [Google Scholar] [CrossRef]
- Pena-Pereira, F.; Marcinkowski, Ł.; Kloskowski, A.; Namieśnik, J. Ionogel fibres of bis(trifluoromethanesulfonyl)imide anion-based ionic liquids for the headspace solid-phase microextraction of chlorinated organic pollutants. Analyst 2015, 140, 7417–7422. [Google Scholar] [CrossRef]
- Zhang, C.; Anderson, J.L. Polymeric ionic liquid bucky gels as sorbent coatings for solid-phase microextraction. J. Chromatogr. A 2014, 1344, 15–22. [Google Scholar] [CrossRef]
- Yu, H.; Merib, J.; Anderson, J.L. Crosslinked polymeric ionic liquids as solid-phase microextraction sorbent coatings for high performance liquid chromatography. J. Chromatogr. A 2016, 1438, 10–21. [Google Scholar] [CrossRef]
- Feng, J.; Wang, X.; Tian, Y.; Luo, C.; Sun, M. Basalt fibers grafted with a poly(ionic liquids) coating for in-tube solid-phase microextraction. J. Sep. Sci. 2018, 41, 3267–3274. [Google Scholar] [CrossRef] [PubMed]
- Sun, M.; Feng, J.; Bu, Y.; Wang, X.; Duan, H.; Luo, C. Palladium-coated stainless-steel wire as a solid-phase microextraction fiber. J. Sep. Sci. 2015, 38, 1584–1590. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Feng, J.; Tian, Y.; Luo, C.; Sun, M. Co-Al bimetallic hydroxide nanocomposites coating for online in-tube solid-phase microextraction. J. Chromatogr. A 2018, 1550, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Feng, J.; Tian, Y.; Wang, X.; Luo, C.; Sun, M. Basalt fibers functionalized with gold nanoparticles for in-tube solid-phase microextraction. J. Sep. Sci. 2018, 41, 1149–1155. [Google Scholar] [CrossRef]
- Pena-Pereira, F.; Marcinkowski, Ł.; Kloskowski, A.; Namieśnik, J. Silica-based ionogels: Nanoconfined ionic liquid-rich fibers for headspace solid-phase microextraction coupled with gas chromatography-barrier discharge ionization detection. Anal. Chem. 2014, 86, 11640–11648. [Google Scholar] [CrossRef]
- Jiang, H.; Li, J.; Hu, X.; Shen, J.; Sun, X.; Han, W.; Wang, L. Ordered mesoporous silica film as a novel fiber coating for solid-phase microextraction. Talanta 2017, 174, 307–313. [Google Scholar] [CrossRef]
- Bagheri, H.; Akbarinejad, A.; Aghakhani, A. A highly thermal-resistant electrospun-based polyetherimide nanofibers coating for solid-phase microextraction Microextraction Techniques. Anal. Bioanal. Chem. 2014, 406, 2141–2149. [Google Scholar] [CrossRef]
- Bagheri, H.; Roostaie, A. Roles of inorganic oxide nanoparticles on extraction efficiency of electrospun polyethylene terephthalate nanocomposite as an unbreakable fiber coating. J. Chromatogr. A 2015, 1375, 8–16. [Google Scholar] [CrossRef]
- Sarafraz-Yazdi, A.; Yekkebashi, A. A non-ionic surfactant-mediated sol-gel coating for solid-phase microextraction of benzene, toluene, ethylbenzene and o-xylene in water samples using a gas chromatography-flame ionization detector. New J. Chem. 2014, 38, 4486–4493. [Google Scholar] [CrossRef]
- Saraji, M.; Mehrafza, N. A simple approach for the preparation of simazine molecularly imprinted nanofibers via self-polycondensation for selective solid-phase microextraction. Anal. Chim. Acta 2016, 936, 108–115. [Google Scholar] [CrossRef]
- Mirabelli, M.F.; Wolf, J.C.; Zenobi, R. Direct Coupling of Solid-Phase Microextraction with Mass Spectrometry: Sub-pg/g Sensitivity Achieved Using a Dielectric Barrier Discharge Ionization Source. Anal. Chem. 2016, 88, 7252–7258. [Google Scholar] [CrossRef] [PubMed]
- Feng, J.; Sun, M.; Li, L.; Wang, X.; Duan, H.; Luo, C. Multiwalled carbon nanotubes-doped polymeric ionic liquids coating for multiple headspace solid-phase microextraction. Talanta 2014, 123, 18–24. [Google Scholar] [CrossRef] [PubMed]
- Wu, M.; Wang, L.; Zeng, B.; Zhao, F. Ionic liquid polymer functionalized carbon nanotubes-doped poly(3,4-ethylenedioxythiophene) for highly efficient solid-phase microextraction of carbamate pesticides. J. Chromatogr. A 2016, 1444, 42–49. [Google Scholar] [CrossRef] [PubMed]
- Ai, Y.; Zhang, J.; Zhao, F.; Zeng, B. Hydrophobic coating of polyaniline-poly(propylene oxide) copolymer for direct immersion solid phase microextraction of carbamate pesticides. J. Chromatogr. A 2015, 1407, 52–57. [Google Scholar] [CrossRef] [PubMed]
- Cavaliere, B.; Monteleone, M.; Naccarato, A.; Sindona, G.; Tagarelli, A. A solid-phase microextraction-gas chromatographic approach combined with triple quadrupole mass spectrometry for the assay of carbamate pesticides in water samples. J. Chromatogr. A 2012, 1257, 149–157. [Google Scholar] [CrossRef] [PubMed]
- Saraji, M.; Jafari, M.T.; Mossaddegh, M. Carbon nanotubes@silicon dioxide nanohybrids coating for solid-phase microextraction of organophosphorus pesticides followed by gas chromatography-corona discharge ion mobility spectrometric detection. J. Chromatogr. A 2016, 1429, 30–39. [Google Scholar] [CrossRef]
- Jafari, M.T.; Saraji, M.; Sherafatmand, H. Polypyrrole/montmorillonite nanocomposite as a new solid phase microextraction fiber combined with gas chromatography-corona discharge ion mobility spectrometry for the simultaneous determination of diazinon and fenthion organophosphorus pesticides. Anal. Chim. Acta 2014, 814, 69–78. [Google Scholar] [CrossRef]
- Kamalabadi, M.; Mohammadi, A.; Alizadeh, N. Polypyrrole nanowire as an excellent solid phase microextraction fiber for bisphenol A analysis in food samples followed by ion mobility spectrometry. Talanta 2016, 156–157, 147–153. [Google Scholar] [CrossRef]
- Li, J.W.; Wang, Y.L.; Yan, S.; Li, X.J.; Pan, S.Y. Molecularly imprinted calixarene fiber for solid-phase microextraction of four organophosphorous pesticides in fruits. Food Chem. 2016, 192, 260–267. [Google Scholar] [CrossRef]
- Liang, W.; Wang, J.; Zang, X.; Dong, W.; Wang, C.; Wang, Z. Barley husk carbon as the fiber coating for the solid-phase microextraction of twelve pesticides in vegetables prior to gas chromatography–mass spectrometric detection. J. Chromatogr. A 2017, 1491, 9–15. [Google Scholar] [CrossRef]
- Wu, M.; Chen, G.; Liu, P.; Zhou, W.; Jia, Q. Polydopamine-based immobilization of a hydrazone covalent organic framework for headspace solid-phase microextraction of pyrethroids in vegetables and fruits. J. Chromatogr. A 2016, 1456, 34–41. [Google Scholar] [CrossRef] [PubMed]
- Wu, M.; Chen, G.; Ma, J.; Liu, P.; Jia, Q. Fabrication of cross-linked hydrazone covalent organic frameworks by click chemistry and application to solid phase microextraction. Talanta 2016, 161, 350–358. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Yang, Q.; Wang, W.; Wang, C.; Wang, Z. Covalent Bonding of Metal-Organic Framework-5/Graphene Oxide Hybrid Composite to Stainless Steel Fiber for Solid-Phase Microextraction of Triazole Fungicides from Fruit and Vegetable Samples. J. Agric. Food Chem. 2016, 64, 2792–2801. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Yang, Q.; Li, Z.; Wang, W.; Wang, C.; Wang, Z. Zeolitic imidazole framework templated synthesis of nanoporous carbon as a novel fiber coating for solid-phase microextraction. Analyst 2016, 141, 1127–1135. [Google Scholar] [CrossRef] [PubMed]
- Tian, M.; Cheng, R.; Ye, J.; Liu, X.; Jia, Q. Preparation and evaluation of ionic liquid-calixarene solid-phase microextraction fibres for the determination of triazines in fruit and vegetable samples. Food Chem. 2014, 145, 28–33. [Google Scholar] [CrossRef]
- Gionfriddo, E.; Naccarato, A.; Sindona, G.; Tagarelli, A. A reliable solid phase microextraction-gas chromatography-triple quadrupole mass spectrometry method for the assay of selenomethionine and selenomethylselenocysteine in aqueous extracts: Difference between selenized and not-enriched selenium potatoes. Anal. Chim. Acta 2012, 747, 58–66. [Google Scholar] [CrossRef]
- Hou, X.; Guo, Y.; Liang, X.; Wang, X.; Wang, L.; Wang, L.; Liu, X. Bis(trifluoromethanesulfonyl)imide-based ionic liquids grafted on graphene oxide-coated solid-phase microextraction fiber for extraction and enrichment of polycyclic aromatic hydrocarbons in potatoes and phthalate esters in food-wrap. Talanta 2016, 153, 392–400. [Google Scholar] [CrossRef]
- Saraji, M.; Jafari, M.T.; Mossaddegh, M. Halloysite nanotubes-titanium dioxide as a solid-phase microextraction coating combined with negative corona discharge-ion mobility spectrometry for the determination of parathion. Anal. Chim. Acta 2016, 926, 55–62. [Google Scholar] [CrossRef]
- Zhang, N.; Gao, J.; Huang, C.; Liu, W.; Tong, P.; Zhang, L. In situ hydrothermal growth of ZnO/g-C3N4 nanoflowers coated solid-phase microextraction fibers coupled with GC-MS for determination of pesticides residues. Anal. Chim. Acta 2016, 934, 122–131. [Google Scholar] [CrossRef]
- Souza Silva, É.A.; Pawliszyn, J. Optimization of fiber coating structure enables direct immersion solid phase microextraction and high-throughput determination of complex samples. Anal. Chem. 2012, 84, 6933–6938. [Google Scholar] [CrossRef]
- De Grazia, S.; Gionfriddo, E.; Pawliszyn, J. A new and efficient Solid Phase Microextraction approach for analysis of high fat content food samples using a matrix-compatible coating. Talanta 2017, 167, 754–760. [Google Scholar] [CrossRef] [PubMed]
- Naccarato, A.; Pawliszyn, J. Matrix compatible solid phase microextraction coating, a greener approach to sample preparation in vegetable matrices. Food Chem. 2016, 206, 67–73. [Google Scholar] [CrossRef] [PubMed]
- Souza-Silva, É.A.; Pawliszyn, J. Direct immersion solid-phase microextraction with matrix-compatible fiber coating for multiresidue pesticide analysis of grapes by gas chromatography-time-of-flight mass spectrometry (DI-SPME-GC-ToFMS). J. Agric. Food Chem. 2015, 63, 4464–4477. [Google Scholar] [CrossRef] [PubMed]
- Brereton, R.G. Chemometrics: Data Analysis for the Laboratory and Chemical Plant; John Wiley & Sons, Ltd.: Chichester, UK, 2003; ISBN 0471489778. [Google Scholar]
- Naccarato, A.; Furia, E.; Sindona, G.; Tagarelli, A. Multivariate class modeling techniques applied to multielement analysis for the verification of the geographical origin of chili pepper. Food Chem. 2016, 206, 217–222. [Google Scholar] [CrossRef] [PubMed]
- Di Donna, L.; Mazzotti, F.; Naccarato, A.; Salerno, R.; Tagarelli, A.; Taverna, D.; Sindona, G. Secondary metabolites of Olea europaea leaves as markers for the discrimination of cultivars and cultivation zones by multivariate analysis. Food Chem. 2010, 121, 492–496. [Google Scholar] [CrossRef]
- Abdulra’uf, L.B.; Tan, G.H. Chemometric approach to the optimization of HS-SPME/GC-MS for the determination of multiclass pesticide residues in fruits and vegetables. Food Chem. 2015, 177, 267–273. [Google Scholar] [CrossRef]
- Wang, X.; Li, X.; Bai, Y.; Liu, H. Just dip it: Online coupling of “dip-it” polymer monolith microextraction with plasma assisted laser desorption ionization mass spectrometry. Chem. Commun. 2015, 51, 4615–4618. [Google Scholar] [CrossRef]
- Gómez-Ríos, G.A.; Vasiljevic, T.; Gionfriddo, E.; Yu, M.; Pawliszyn, J. Towards on-site analysis of complex matrices by solid-phase microextraction-transmission mode coupled to a portable mass spectrometer: Via direct analysis in real time. Analyst 2017, 142, 2928–2935. [Google Scholar] [CrossRef]
- Asiabi, H.; Yamini, Y.; Seidi, S.; Esrafili, A.; Rezaei, F. Electroplating of nanostructured polyaniline-polypyrrole composite coating in a stainless-steel tube for on-line in-tube solid phase microextraction. J. Chromatogr. A 2015, 1397, 19–26. [Google Scholar] [CrossRef]
- de Souza Freitas, L.A.; Vieira, A.C.; Mendonça, J.A.F.R.; Figueiredo, E.C. Molecularly imprinted fibers with renewable surface for solid-phase microextraction of triazoles from grape juice samples followed by gas chromatography mass spectrometry analysis. Analyst 2014, 139, 626–632. [Google Scholar] [CrossRef]
- Wu, M.; Chen, G.; Liu, P.; Zhou, W.; Jia, Q. Preparation of porous aromatic framework/ionic liquid hybrid composite coated solid-phase microextraction fibers and their application in the determination of organochlorine pesticides combined with GC-ECD detection. Analyst 2016, 141, 243–250. [Google Scholar] [CrossRef] [PubMed]
- Pelit, F.O.; Pelit, L.; Dizdaş, T.N.; Aftafa, C.; Ertaş, H.; Yalçinkaya, E.E.; Türkmen, H.; Ertaş, F.N. A novel polythiophene - ionic liquid modified clay composite solid phase microextraction fiber: Preparation, characterization and application to pesticide analysis. Anal. Chim. Acta 2015, 859, 37–45. [Google Scholar] [CrossRef] [PubMed]
- Viñas, P.; Pastor-Belda, M.; Torres, A.; Campillo, N.; Hernández-Córdoba, M. Use of oleic-acid functionalized nanoparticles for the magnetic solid-phase microextraction of alkylphenols in fruit juices using liquid chromatography-tandem mass spectrometry. Talanta 2016, 151, 217–223. [Google Scholar] [CrossRef] [PubMed]
- Mirabelli, M.F.; Gionfriddo, E.; Pawliszyn, J.; Zenobi, R. A quantitative approach for pesticide analysis in grape juice by direct interfacing of a matrix compatible SPME phase to dielectric barrier discharge ionization-mass spectrometry. Analyst 2018, 143, 891–899. [Google Scholar] [CrossRef]
- Lan, H.; Pan, D.; Sun, Y.; Guo, Y.; Wu, Z. Thin metal organic frameworks coatings by cathodic electrodeposition for solid-phase microextraction and analysis of trace exogenous estrogens in milk. Anal. Chim. Acta 2016, 937, 53–60. [Google Scholar] [CrossRef]
- Merib, J.; Nardini, G.; Carasek, E. Use of Doehlert design in the optimization of extraction conditions in the determination of organochlorine pesticides in bovine milk samples by HS-SPME. Anal. Methods 2014, 6, 3254–3260. [Google Scholar] [CrossRef]
- Lin, W.; Wei, S.; Jiang, R.; Zhu, F.; Ouyang, G. Calibration of the complex matrix effects on the sampling of polycyclic aromatic hydrocarbons in milk samples using solid phase microextraction. Anal. Chim. Acta 2016, 933, 117–123. [Google Scholar] [CrossRef]
- Lv, F.; Gan, N.; Cao, Y.; Zhou, Y.; Zuo, R.; Dong, Y. A molybdenum disulfide/reduced graphene oxide fiber coating coupled with gas chromatography–mass spectrometry for the saponification-headspace solid-phase microextraction of polychlorinated biphenyls in food. J. Chromatogr. A 2017, 1525, 42–50. [Google Scholar] [CrossRef]
- Joshi, M.D.; Ho, T.D.; Cole, W.T.S.; Anderson, J.L. Determination of polychlorinated biphenyls in ocean water and bovine milk using crosslinked polymeric ionic liquid sorbent coatings by solid-phase microextraction. Talanta 2014, 118, 172–179. [Google Scholar] [CrossRef]
- Pang, J.; Yuan, D.; Huang, X. On-line combining monolith-based in-tube solid phase microextraction and high-performance liquid chromatography- fluorescence detection for the sensitive monitoring of polycyclic aromatic hydrocarbons in complex samples. J. Chromatogr. A 2018, 1571, 29–37. [Google Scholar] [CrossRef]
- Roszkowska, A.; Yu, M.; Bessonneau, V.; Bragg, L.; Servos, M.; Pawliszyn, J. Metabolome Profiling of Fish Muscle Tissue Exposed to Benzo[a]pyrene Using in Vivo Solid-Phase Microextraction. Environ. Sci. Technol. Lett. 2018, 5, 431–435. [Google Scholar] [CrossRef]
- Huang, S.; Xu, J.; Wu, J.; Hong, H.; Chen, G.; Jiang, R.; Zhu, F.; Liu, Y.; Ouyang, G. Rapid detection of five anesthetics in tilapias by in vivo solid phase microextraction coupling with gas chromatography-mass spectrometry. Talanta 2017, 168, 263–268. [Google Scholar] [CrossRef] [PubMed]
- Delvaux Júnior, N.A.; de Queiroz, M.E.L.R.; Neves, A.A.; Oliveira, A.F.; da Silva, M.R.F.; Faroni, L.R.A.; Heleno, F.F. Headspace solid phase microextraction-gas chromatography for the determination of trihalomethanes in fish. Microchem. J. 2017, 133, 539–544. [Google Scholar] [CrossRef]
- Hashemi-Moghaddam, H.; Ahmadifard, M. Novel molecularly imprinted solid-phase microextraction fiber coupled with gas chromatography for analysis of furan. Talanta 2016, 150, 148–154. [Google Scholar] [CrossRef] [PubMed]
- Lin, S.; Gan, N.; Qiao, L.; Zhang, J.; Cao, Y.; Chen, Y. Magnetic metal-organic frameworks coated stir bar sorptive extraction coupled with GC-MS for determination of polychlorinated biphenyls in fish samples. Talanta 2015, 144, 1139–1145. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Wang, J.; Zhang, S.; Cui, P.; Wang, C.; Wang, Z. A novel Schiff base network-1 nanocomposite coated fiber for solid-phase microextraction of phenols from honey samples. Talanta 2016, 161, 22–30. [Google Scholar] [CrossRef]
- Al-Alam, J.; Fajloun, Z.; Chbani, A.; Millet, M. A multiresidue method for the analysis of 90 pesticides, 16 PAHs, and 22 PCBs in honey using QuEChERS–SPME. Anal. Bioanal. Chem. 2017, 409, 5157–5169. [Google Scholar] [CrossRef]
- Yang, M.; Wu, X.; Xi, X.; Zhang, P.; Yang, X.; Lu, R.; Zhou, W.; Zhang, S.; Gao, H.; Li, J. Using β-cyclodextrin/attapulgite-immobilized ionic liquid as sorbent in dispersive solid-phase microextraction to detect the benzoylurea insecticide contents of honey and tea beverages. Food Chem. 2016, 197, 1064–1072. [Google Scholar] [CrossRef]
- Liang, W.; Wang, J.; Zang, X.; Wang, C.; Wang, Z. A porous carbon derived from amino-functionalized material of Institut Lavoisier as a solid-phase microextraction fiber coating for the extraction of phthalate esters from tea. J. Sep. Sci. 2016, 39, 1331–1338. [Google Scholar] [CrossRef]
- Cheng, X.; Yan, H.; Wang, X.; Sun, N.; Qiao, X. Vortex-assisted magnetic dispersive solid-phase microextraction for rapid screening and recognition of dicofol residues in tea products. Food Chem. 2014, 162, 104–109. [Google Scholar] [CrossRef]
- Ng, N.T.; Sanagi, M.M.; Wan Ibrahim, W.N.; Wan Ibrahim, W.A. Agarose-chitosan-C18 film micro-solid phase extraction combined with high performance liquid chromatography for the determination of phenanthrene and pyrene in chrysanthemum tea samples. Food Chem. 2017, 222, 28–34. [Google Scholar] [CrossRef] [PubMed]
- Cagliero, C.; Nan, H.; Bicchi, C.; Anderson, J.L. Matrix-compatible sorbent coatings based on structurally tuned polymeric ionic liquids for the determination of acrylamide in brewed coffee and coffee powder using solid-phase microextraction. J. Chromatogr. A 2016, 1459, 17–23. [Google Scholar] [CrossRef] [PubMed]
- Ho, T.D.; Toledo, B.R.; Hantao, L.W.; Anderson, J.L. Chemical immobilization of crosslinked polymeric ionic liquids on nitinol wires produces highly robust sorbent coatings for solid-phase microextraction. Anal. Chim. Acta 2014, 843, 18–26. [Google Scholar] [CrossRef] [PubMed]
- Amanzadeh, H.; Yamini, Y.; Moradi, M.; Asl, Y.A. Determination of phthalate esters in drinking water and edible vegetable oil samples by headspace solid phase microextraction using graphene/polyvinylchloride nanocomposite coated fiber coupled to gas chromatography-flame ionization detector. J. Chromatogr. A 2016, 1465, 38–46. [Google Scholar] [CrossRef]
- Wang, Y.; Sun, Y.; Xu, B.; Li, X.; Jin, R.; Zhang, H.; Song, D. Magnetic ionic liquid-based dispersive liquid-liquid microextraction for the determination of triazine herbicides in vegetable oils by liquid chromatography. J. Chromatogr. A 2014, 1373, 9–16. [Google Scholar] [CrossRef]
- Wu, L.; Song, Y.; Hu, M.; Xu, X.; Zhang, H.; Yu, A.; Ma, Q.; Wang, Z. Determination of sulfonamides in butter samples by ionic liquid magnetic bar liquid-phase microextraction high-performance liquid chromatography. Anal. Bioanal. Chem. 2015, 407, 569–580. [Google Scholar] [CrossRef]
- Matin, A.A.; Biparva, P.; Gheshlaghi, M. Gas chromatographic determination of polycyclic aromatic hydrocarbons in water and smoked rice samples after solid-phase microextraction using multiwalled carbon nanotube loaded hollow fiber. J. Chromatogr. A 2014, 1374, 50–57. [Google Scholar] [CrossRef]
- Chen, D.; Huang, Y.Q.; He, X.M.; Shi, Z.G.; Feng, Y.Q. Coupling carbon nanotube film microextraction with desorption corona beam ionization for rapid analysis of Sudan dyes (I-IV) and Rhodamine B in chilli oil. Analyst 2015, 140, 1731–1738. [Google Scholar] [CrossRef]
- Li, X.; Li, Z.; Wang, X.; Nie, H.; Zhang, Y.; Bai, Y.; Liu, H. Monolith dip-it: A bifunctional device for improving the sensitivity of direct analysis in real time mass spectrometry. Analyst 2016, 141, 4947–4952. [Google Scholar] [CrossRef]
- Zhang, L.; Gionfriddo, E.; Acquaro, V.; Pawliszyn, J. Direct immersion solid-phase microextraction analysis of multi-class contaminants in edible seaweeds by gas chromatography-mass spectrometry. Anal. Chim. Acta 2018, 1031, 83–97. [Google Scholar] [CrossRef]
- Jian, J.M.; Guo, Y.; Zeng, L.; Liang-Ying, L.; Lu, X.; Wang, F.; Zeng, E.Y. Global distribution of perfluorochemicals (PFCs) in potential human exposure source–A review. Environ. Int. 2017, 108, 51–62. [Google Scholar] [CrossRef] [PubMed]
- Monteleone, M.; Naccarato, A.; Sindona, G.; Tagarelli, A. A rapid and sensitive assay of perfluorocarboxylic acids in aqueous matrices by headspace solid phase microextraction-gas chromatography-triple quadrupole mass spectrometry. J. Chromatogr. A 2012, 1251, 160–168. [Google Scholar] [CrossRef] [PubMed]
- Ouyang, G.; Oakes, K.D.; Bragg, L.; Wang, S.; Liu, H.; Cui, S.; Servos, M.R.; Dixon, D.G.; Pawliszyn, J. Sampling-rate calibration for rapid and nonlethal monitoring of organic contaminants in fish muscle by solid-phase microextraction. Environ. Sci. Technol. 2011, 45, 7792–7798. [Google Scholar] [CrossRef] [PubMed]
- Ouyang, G.; Vuckovic, D.; Pawliszyn, J. Nondestructive sampling of living systems using in vivo solid-phase microextraction. Chem. Rev. 2011, 111, 2784–2814. [Google Scholar] [CrossRef]
- Wang, H.; Sun, W.; Zhang, J.; Yang, X.; Lin, T.; Ding, L. Desorption corona beam ionization source for mass spectrometry. Analyst 2010, 135, 688–695. [Google Scholar] [CrossRef]
- World Health Organization Regional Office for Europe. Human Biomonitoring: Facts and Figures; WHO: Geneva, Switzerland, 2015. [Google Scholar]
- Centers for Disease Control and Prevention—National Biomonitoring Program. Available online: https://www.cdc.gov/biomonitoring/ (accessed on 1 November 2019).
- Souza-Silva, É.A.; Reyes-Garcés, N.; Gómez-Ríos, G.A.; Boyaci, E.; Bojko, B.; Pawliszyn, J. A critical review of the state of the art of solid-phase microextraction of complex matrices III. Bioanalytical and clinical applications. TrAC Trends Anal. Chem. 2015, 71, 249–264. [Google Scholar] [CrossRef]
- Naccarato, A.; Gionfriddo, E.; Elliani, R.; Sindona, G.; Tagarelli, A. A fast and simple solid phase microextraction coupled with gas chromatography-triple quadrupole mass spectrometry method for the assay of urinary markers of glutaric acidemias. J. Chromatogr. A 2014, 1372, 253–259. [Google Scholar] [CrossRef]
- Naccarato, A.; Elliani, R.; Cavaliere, B.; Sindona, G.; Tagarelli, A. Development of a fast and simple gas chromatographic protocol based on the combined use of alkyl chloroformate and solid phase microextraction for the assay of polyamines in human urine. J. Chromatogr. A 2018, 1549, 1–13. [Google Scholar] [CrossRef]
- Yang, B.C.; Fang, S.F.; Wan, X.J.; Luo, Y.; Zhou, J.Y.; Li, Y.; Li, Y.J.; Wang, F.; Huang, O.P. Quantification of monohydroxylated polycyclic aromatic hydrocarbons in human urine samples using solid-phase microextraction coupled with glass-capillary nanoelectrospray ionization mass spectrometry. Anal. Chim. Acta 2017, 973, 68–74. [Google Scholar] [CrossRef]
- Antonucci, A.; Vitali, M.; Avino, P.; Manigrasso, M.; Protano, C. Sensitive multiresidue method by HS-SPME/GC-MS for 10 volatile organic compounds in urine matrix: A new tool for biomonitoring studies on children. Anal. Bioanal. Chem. 2016, 408, 5789–5800. [Google Scholar] [CrossRef]
- Lamani, X.; Horst, S.; Zimmermann, T.; Schmidt, T.C. Determination of aromatic amines in human urine using comprehensive multi-dimensional gas chromatography mass spectrometry (GCxGC-qMS). Anal. Bioanal. Chem. 2015, 407, 241–252. [Google Scholar] [CrossRef] [PubMed]
- Song, H.N.; Kim, C.H.; Lee, W.Y.; Cho, S.H. Simultaneous determination of volatile organic compounds with a wide range of polarities in urine by headspace solid-phase microextraction coupled to gas chromatography/mass spectrometry. Rapid Commun. Mass Spectrom. 2017, 31, 613–622. [Google Scholar] [CrossRef] [PubMed]
- Koureas, M.; Karagkouni, F.; Rakitskii, V.; Hadjichristodoulou, C.; Tsatsakis, A.; Tsakalof, A. Serum levels of organochlorine pesticides in the general population of Thessaly, Greece, determined by HS-SPME GC-MS method. Environ. Res. 2016, 148, 318–321. [Google Scholar] [CrossRef] [PubMed]
- Sajid, M.; Basheer, C.; Daud, M.; Alsharaa, A. Evaluation of layered double hydroxide/graphene hybrid as a sorbent in membrane-protected stir-bar supported micro-solid-phase extraction for determination of organochlorine pesticides in urine samples. J. Chromatogr. A 2017, 1489, 1–8. [Google Scholar] [CrossRef]
- Wang, S.L.; Xu, H. Inorganic–organic hybrid coating material for the online in-tube solid-phase microextraction of monohydroxy polycyclic aromatic hydrocarbons in urine. J. Sep. Sci. 2016, 39, 4610–4620. [Google Scholar] [CrossRef]
- Hardy, E.M.; Duca, R.C.; Salquebre, G.; Appenzeller, B.M.R. Multi-residue analysis of organic pollutants in hair and urine for matrices comparison. Forensic Sci. Int. 2015, 249, 6–19. [Google Scholar] [CrossRef]
- Yamamoto, Y.; Ishizaki, A.; Kataoka, H. Biomonitoring method for the determination of polycyclic aromatic hydrocarbons in hair by online in-tube solid-phase microextraction coupled with high performance liquid chromatography and fluorescence detection. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2015, 1000, 187–191. [Google Scholar] [CrossRef]
- Tzatzarakis, M.N.; Barbounis, E.G.; Kavvalakis, M.P.; Vakonaki, E.; Renieri, E.; Vardavas, A.I.; Tsatsakis, A.M. Rapid method for the simultaneous determination of DDTs and PCBs in hair of children by headspace solid phase microextraction and gas chromatography-mass spectrometry (HSSPME/GC-MS). Drug Test. Anal. 2014, 6, 85–92. [Google Scholar] [CrossRef]
- Flores-Ramírez, R.; Ortiz-Pérez, M.D.; Batres-Esquivel, L.; Castillo, C.G.; Ilizaliturri-Hernández, C.A.; Díaz-Barriga, F. Rapid analysis of persistent organic pollutants by solid phase microextraction in serum samples. Talanta 2014, 123, 169–178. [Google Scholar] [CrossRef]
- Zhang, J.; Li, W.; Zhu, W.; Qin, P.; Lu, M.; Zhang, X.; Miao, Y.; Cai, Z. Mesoporous graphitic carbon nitride@NiCo 2 O 4 nanocomposite as a solid phase microextraction coating for sensitive determination of environmental pollutants in human serum samples. Chem. Commun. 2019, 55, 10019–10022. [Google Scholar] [CrossRef]
- Meyer-Monath, M.; Beaumont, J.; Morel, I.; Rouget, F.; Tack, K.; Lestremau, F. Analysis of BTEX and chlorinated solvents in meconium by headspace-solid-phase microextraction gas chromatography coupled with mass spectrometry. Anal. Bioanal. Chem. 2014, 406, 4481–4490. [Google Scholar] [CrossRef] [PubMed]
- de la Guardia, M.; Garrigues, S. Handbook of Green Analytical Chemistry; John Wiley & Sons, Ltd.: Chichester, UK, 2012; ISBN 9780470972014. [Google Scholar]


| Analytes | Matrix | SPME Approach and Coating Material | Instrumentation | Calibration Range | [Ref] Publishing Year |
|---|---|---|---|---|---|
| THMs | Ambient air | Fiber—DVB/CAR/PDMS | GC-MS | 2–5000 µg/m3 | [1]—2019 |
| VOCs | Indoor air | fiber—CAR/PDMS | GC-MS, GC-FID | - | [2]—2018 |
| aliphatic and aromatic amines | Indoor air | Fiber—DVB/CAR/PDMS | GC-MS | 10–50,000 ng/m3 | [3]—2018 |
| OPEs | Indoor air | fiber—PDMS | GC-MS | 0.05–500 mg/m3 | [4]—2017 |
| Formaldehyde, acetaldehyde, acetone, hexanal | Indoor air | fiber—PDMS/DVB | GC-MS, GC-FID | - | [5]—2014 |
| VOCs | Ambient air | SPME Arrow—Carbon WR, Carbon WR/PDMS, PDMS/DVB, lab-made functionalized mesoporous silica material | GC-MS | - | [7]—2019 |
| PAHs, phthalates, adipates, vulcanisation additives and antioxidants | Ambient air | fiber—PDMS/DVB | GC-MS | 0.01–2 µg/L | [8]—2018 |
| BTEX | Ambient air | fiber—PDMS | GC-MS | 2–200 mg/m3 | [9]—2016 |
| HCN | Ambient air | fiber—CAR/PDMS | GC-MS | 5–500 µg/L | [10]—2017 |
| OPEs | PM10 | fiber—DVB/CAR/PDMS | GC-MS/MS | 0.1–10 ng/mL | [13]—2018 |
| pesticides of different chemical classes | Ambient air and PM | fiber—PA | GC-MS | 2–2000 ng of each pesticide | [15]—2014 |
| Short-chain chlorinated paraffin | water, sediment, organisms, and PM | fiber—hollow microporous organic network (H-MON) | GC-MS | 0.05–10 ng/mL | [17]—2018 |
| PAHs | environmental water, PM2.5, and smoked meat | fiber—MOF@microporous organic network (MON) hybrid materials | GC-MS/MS | 0.1–500 ng/L | [18]—2016 |
| nitrated polycyclic aromatic hydrocarbons | environmental water, PM2.5, and soil | fiber—porphyrinic zirconium MOF | GC-MS | 0.4–400 ng/L | [19]—2017 |
| benzene homologues | indoor air | fiber—Covalent organic frameworks (COF) | GC-MS | 0.10–20 ng/L | [20]—2017 |
| BTEX | ambient air | fiber—functionalized tetraquinoxaline cavitand | GC-MS | 3.5–470 ng/m3 | [21]—2016 |
| acrolein | ambient air | cold fiber—PDMS | GC-MS | - | [23]—2017 |
| Acetic acid | indoor air | fiber—Car/PDMS | GC-MS | - | [25]—2019 |
| pesticides | river water | SPME-TM | DART-MS/MS DART-orbitrap | 0.10–100 ng/mL | [27]—2017 |
| triazine herbicides obtained | lake water | IT-SPME | DART-MS/MS | 0.02−0.46 ng/mL | [28]—2014 |
| perfluorinated compounds (PFCs) | lake and river water | wooden-tip SPME ambient MS | LC-MS/MS orbitrap | 0.5−100 ng/L | [29]—2014 |
| PAHs | rain and river water | IT-SPME—PEEK tube was packed with polyester fibers | LC- diode array detector | 0.03–80 μg/L | [31]—2016 |
| PAHs | lake and river water | fiber—boron nitride nanotube | GC-MS/MS | 1–1000 ng/L | [40]—2014 |
| PAHs | river water | fiber—peanut shell-derived biochar materials | GC-MS | 10–2000 ng/L | [41]—2019 |
| PAHs | lake water | fiber—MOF | GC-MS | 0.01–10 μg/L | [32]—2019 |
| PCBs | lake and river water | fiber—MOF | GC-MS | 1–50 ng/L | [33]—2015 |
| PAHs | lake and river water | fiber—zeolite imidazolate MOF | GC-MS | 10–20,000 ng/L | [34]—2019 |
| PAHs | lake and river water | fiber—hollow mesoporous carbon spheres | GC-MS | 5–2000 ng/L | [35]—2017 |
| PAHs | river water | fiber—titanium dioxide-nanosheets | HPLC-UV | 0.05–300 mg/L | [37]—2015 |
| PAHs | volcanic area water | fiber—silver nanoparticles | GC-FID | 5–300 μg/L | [38]—2018 |
| nitro-polycyclic aromatic hydrocarbons (NPAHs) | lake and snow water | fiber—metal–organic nanotubes | GC-MS/MS | 10–1000 ng/L | [39]—2018 |
| PCBs | river water | fiber—bamboo charcoal | GC-MS/MS | 0.2–1000 ng/L | [42]—2014 |
| PCBs | seawater | fiber—metal–organic nanotubes | GC-MS/MS | 10–5000 pg/L | [43]—2016 |
| PCBs | pond and lake water | fiber—multimodal porous carbons (MPCs) | GC-ECD | 10–1000 ng/L | [44]—2019 |
| PCBs | pond, river, underground and lake water | fiber—metal–organic nanotubes | GC-MS/MS | 0.1–500 ng/L | [45]—2014 |
| Sudan dyes | lake water | PEEK tube—zeolitic imidazolate frameworks | HPLC-UV | 0.02–20 ng/mL | [46]—2019 |
| sulfonylurea herbicides (SUHs) | lake, river and well water | MMF-SPME—mixed functional monomers | HPLC-DAD | 0.1–200 μg/L | [47]—2018 |
| triazine herbicides | lake and river water | MMF-SPME—polydopamine-based monolith | HPLC-DAD | 0.1–200 μg/L | [48]—2016 |
| chlorinated herbicides | river water | Fiber—polyethersulfone | GC-MS | 0.05–5 ng/mL | [49]—2014 |
| organophosphorous pesticides | well water | fiber—metal-organic framework/polyethersulfone nanocomposite (TMU-4/PES) | GC-NPD | 0.015–50 μg/L | [50]—2018 |
| organochlorine pesticides | well and pond water | fiber—gold nanoparticles | GC-ECD | 0.56–10 μg/L | [51]—2016 |
| organochlorine pesticides | river and pond water | fiber—nitrogen-doped ordered mesoporous polymer (NOMP) | GC–MS | 9−1500 ng/L | [52]—2016 |
| carbamate pesticides | river water | MMF-SPME—boron-rich coating | HPLC-DAD | 0.057–0.96 μg/L | [53]—2019 |
| fluoroquinolones | river water | IT-SPME—MOF-monolith composite | HPLC-FLD | 0.001–5.0 μg/L | [54]—2019 |
| cefaclor and cefalexin | lake water | stir bar—molecular imprinted polymers and magnetic carbon nanotubes | HPLC-UV | 15–320 ng/mL | [55]—2017 |
| sulfadiazine | well water | stir bar—graphene oxide-silica composite reinforced | UV-vis | 5–150 μg/L | [57]—2017 |
| nitrophenols | lake and river water | MMF-SPME—AMED coating | HPLC-DAD | 0.5–200 μg/L | [58]—2015 |
| chlorophenol and nitrophenols | river and spring water | fiber—cobalt (II)-based metal-organic nanotubes (Co-MONTs) | GC-MS | 0.5–1000 ng/L | [59]—2017 |
| ultraviolet filters | river and rain water | fiber—phenyl functionalization of titania nanoparticles | HPLC-UV | 0.005–25 μg/L | [61]—2015 |
| ultraviolet filters | river and rain water | fiber—gold nanoparticles | HPLC-UV | 0.10–400 μg/L | [70]—2014 |
| ultraviolet filters | river and rain water | fiber—silica nanoparticle | HPLC-UV | 0.05–300 μg/L | [71]—2017 |
| ultraviolet filters | river water | IT-SPME—polyaniline coating | HPLC-DAD | 0.06–100 μg/L | [62]—2017 |
| ultraviolet filters | river water | fiber—oriented ZnO nanosheets | HPLC-UV | 0.05–500 ng/mL | [63]—2019 |
| ultraviolet filters | river water | fiber—nitrogen-enriched carbonaceous material | HPLC-UV | 0.2–200 μg/L | [65]—2017 |
| ultraviolet filters | river water | fiber—in situ fabricated rod-like TiO2 coating | HPLC-UV | 0.05–200 μg/L | [68]—2014 |
| BTEX, organochlorine pesticides | sea, pond and river water | fiber—Prussian blue nanoparticles-doped graphene oxide | GC-MS | 1–1000 ng/L, 2–2000 ng/L | [75]—2019 |
| BTEX | pond and river water | fiber—tri-metal centered metal-organic frameworks (tM-MOFs) | GC-MS | 5–2000 ng/L | [76]—2017 |
| BTEX | lake and river water | fiber—graphene-carbon nanotubes composite | GC-MS | 5–5000 ng/L | [77]—2018 |
| BTEX | river water | fiber—porous organic polymers | GC-MS | 2–500 ng/L | [79]—2017 |
| BTEX and PAHs | pond and river water | fiber—microporous polymer | GC-MS | 1–20,000 ng/L | [80]—2018 |
| benzoylurea insecticides | river water | MMF-SPME—poly(methacrylic acid-co-ethylene dimethacrylate) | HPLC-DAD | 0.10–200 μg/L | [82]—2015 |
| endocrine disrupting | lake and river water | MMF-SPME—polymeric ionic liquid-based adsorbent | HPLC-UV | 0.10–200 μg/L | [83]—2017 |
| perfluoroalkane sulfonamides | lake and river water | MMF-SPME—polymeric material with boron | HPLC-MS/MS | 0.0025–30.0 μg/L | [84]—2019 |
| estrogens | lake and river water | magnetism-enhanced monolith-based in-tube IT-SPME—polymeric monolith with modified Fe3O4 nanoparticles | HPLC-DAD | 0.5–200 μg/L | [85]—2016 |
| non-steroidal anti-inflammatory drugs | river water | fabric phase sorptive extraction—poly(dime-thyldiphenylsiloxane); poly(tetrahydrofuran); | GC-MS | 3–20,000 ng/L | [87]—2015 |
| triazine herbicides | river water | Stir fabric phase sorptive extraction | UPLC-DAD | 0. 26–1.50 μg/L | [89]—2015 |
| nitroaromatic Explosives | soil | fiber—quinoxaline-bridged cavitand | GC-MS | 120−1200 ng/Kg | [92]—2014 |
| PAHs | soil | fiber—nanoporous carbon derived from an aluminum- based MOF | GC-MS | 0.1–12 μg/L | [95]—2015 |
| PCBs | soil | fiber—different MOF networks | GC-MS | 0.01–600 ng/L | [97]—2018 |
| PCBs | soil | fiber—MOF MIL-88B | GC-MS | 5–200 ng/L | [98]—2014 |
| polybrominated diphenyl ethers | soil | fiber—Ag(I)-organic frameworks | GC-μECD | 0.1–500 ng/g | [99]—2015 |
| dimethylhydrazine | soil | fiber—Car/PDMS | GC-MS | 0.5–2.5 mg/Kg | [100]—2018 |
| 1-methyl-1H-1,2,4-triazole | soil | fiber—PDMS/DVB | GC-MS | - | [102]—2015 |
| DDE | soil | fiber—PDMS | GC-MS | - | [103]—2017 |
| PCBs | soil | fiber—PDMS | GC-MS | - | [104]—2019 |
| PAHs | soil | fiber—PDMS | HPLC-UV | - | [106]—2016 |
| mercury | soil | ionic liquid coated PTFE tube | CV AAS | 0.5–60 ng/mL | [108]—2014 |
| Analytes | Matrix | SPME Approach and Coating Material | Instrumentation | Calibration Range | [Ref] Publishing Year |
|---|---|---|---|---|---|
| benzene derivatives | tap water | fiber—PANI-PIL/MWCNTs | GC-FID | 0.05–250 μg/L | [110]—2015 |
| BTEX | tap water, mineral water | fiber—polypyrrole/CNT/TiO2 | GC-FID | 0.03–500 μg/L | [111]—2015 |
| aromatic amines | tap water | fiber—poly(ethylene glycol)–graphene oxide | GC-FID | 1–2000 ng/L | [116]—2015 |
| triazines | mineral water | IT-SPME—graphene oxide | HPLC-MS/MS | 0.2–4 μg/L | [117]—2018 |
| triazines | tap water | IT-SPME—GO | LC-MS/MS | 5–500 ng/L | [118]—2015 |
| PAHs | tap water | IT-SPME—zeolitic imidazolate framework (ZIF)—ZIF-8 | HPLC-FLD | 0.01–5 μg/L | [126]—2015 |
| phenols, multiclass insecticides | tap water | fiber—PIL-1-vinyl-3-(10-hydroxydecyl) imidazolium chloride [VC10OHIM][Cl] | HPLC-UV | 1–500 μg/L | [131]—2016 |
| phthalates | bottle water | IT-SPME—1-dodecyl-3-vinylimidazolium bromide | HPLC-DAD | 0.03–12 μg/L | [132]—2018 |
| PAHs | tap water | IT-SPME—Co-Al bimetallic hydroxide nanocomposites | HPLC-DAD | 0.003–15 μg/L | [134]—2018 |
| PAHs | tap water | IT-SPME—gold nanoparticles | HPLC-DAD | 0.01–20 μg/L | [135]—2018 |
| VOCs | tap water, mineral water | fiber—hybrid silica-based material with IL 1-methyl-3-butylimidazolium bis(trifluoromethylsulfonyl)imide ([C4MIM][TFSI]) | GC—barrier ionization discharge (GC-BID) | 0.025–75 µg/L | [136]—2014 |
| chlorophenols | tap water | fiber—Ordered mesoporous silica (OMS) | GC-FID | 0.2–200 µg/L | [137]—2017 |
| PAHs | tap water | fiber—polyetherimide | GC-MS | 0.005–1.2 μg/L | [138]—2014 |
| BTEX | tap water | fiber—polyethylene terephthalate nanocomposites | GC-MS | 0.01–1 μg/L | [139]—2015 |
| BTEX | tap water, mineral water | fiber—PDMS-TX100 | GC-FID | 0.004–200 μg/L | [140]—2014 |
| multiclass pesticides | tap water | fiber—PDMS/DVB | MS—dielectric barrier discharge ionization (DBDI) | 0.01–30 μg/L | [142]—2016 |
| parabens | tap water, milk, juice | in-tube—polyaniline–polypyrrole composite | HPLC-UV | 0.07–50 μg/L | [171]—2015 |
| furan | tap water, canned tuna | fiber—MIP with pyrrole as template | GC-MS | 0.5–100 μg/L | [186]—2016 |
| benzoylurea insecticides | orange juice, grape juice | fiber—poly(methacrylic acid-co-ethylene dimethacrylate) | HPLC-DAD | 0.5–200 μg/L | [82]—2015 |
| multiclass pesticides | fruit juices | fiber—polythiophene-ionic liquid-Montmorillonite (PTh-IL-Mmt) | GC-ECD | 0.5–10 μg/L | [174]—2015 |
| multiclass pesticides | grape juice | fiber—PDMS/DVB/PDMS | DBDI-MS | 0.5–100 μg/L | [176]—2018 |
| alkylphenols | fruit juices | magnetic dispersive—CoFe2O4/oleic acid | LC-DAD-MS/MS | 16–200 μg/L | [175]—2016 |
| triazine herbicides | orange juice | in tube—poly(MAA-EDMA-SWNT) | DART-MS | 0.1–50 μg/L | [28]—2014 |
| triazoles | grape juice | fiber—MIP | GC-MS | 100–2000 μg/L | [172]—2014 |
| multiclass pesticides | grape juice, orange juice, cow milk | mesh—HLB/PAN | DART/MS | 0.1–100 μg/L | [27]—2017 |
| multiclass pesticides | grape juice, milk | mesh—HLB/PAN | DART/MS | 5–500 μg/L | [170]—2017 |
| organochlorine pesticides | juice, milk | fiber—PAF/IL | GC-ECD | 1–500 μg/L | [173]—2016 |
| bisphenol A | milk | fiber—(Et3NH)2Zn3(BDC)4 (E-MOF-5) | HPLC-SPD | 1–200 μg/L | [177]—2016 |
| PAHs | milk | home-made PDMS fiber | GC-MS | 0.1–5 μg/L | [179]—2016 |
| PCBs | milk | fiber—MoS2/RGO | GC-MS | 0.25–100 μg/L | [180]—2017 |
| PCBs | milk | fiber—PIL 1-vinylbenzyl-3-hexadecylimidazolium bis[(trifluoromethyl)sulfonyl]imide [VBHDIM] [NTf2] | GC-MS | 2.5–100 ng/L | [181]—2014 |
| perfluorinated compounds (PFCs) | milk | surface coated wooden-tip probe—n-octadecyldimethyl [3-(trimethoxysilyl)propyl]ammonium chloride | Orbitrap MS | 0.5–100 ng/L | [29]—2014 |
| 2-naphthol | pomelo and orange | fiber—MWCNTs-PILs | GC-FID | 0.5–5000 μg/Kg | [143]—2014 |
| carbamate pesticides | apple, lettuce | fiber—IL/CNT | GC-FID | 0.05–250 μg/Kg | [144]—2016 |
| organophosphorus pesticides | pear, grape, eggplant | fiber—CNTs@SiO2 | GC–corona discharge ion mobility spectrometric detection | 0.5–15 μg/Kg | [147]—2016 |
| organophosphorus pesticides | cucumber, lettuce, apple, tap water | fiber—polypyrrole/montmorillonite nanocomposites | GC-CD-IMS | 0.05–10 μg/Kg | [148]—2014 |
| BPA | canned beans, canned corn, canned peas | fiber—polypyrrole nanowire | ion mobility spectrometry | 10–150 μg/Kg | [149]—2016 |
| organophosphorous pesticides | fruits | fiber—molecularly imprinted polymer (MIP) with calixarene as template | GC-NPD | 0.2–1000 μg/Kg | [150]—2016 |
| multiclass pesticides | vegetables | fiber—Barley husk carbon | GC-MS | 0.2–75 μg/Kg | [151]—2017 |
| pyrethroids | vegetables, fruits | fiber—COF-PDA | GC-ECD | 1–1000 μg/Kg | [152]—2016 |
| organochlorine pesticides | cucumber | fiber—Covalent organic frameworks (COF) | GC-ECD | 0.008–800 ng/kg | [153]—2016 |
| triazole fungicides | vegetable, fruit | fiber—MOF-5/GO hybrid composite | GC-µECD | 0.17–500 μg/Kg | [154]—2016 |
| Organochlorine pesticides | vegetables | fiber—ZIF-67 | GC-µECD | 0.30–50 μg/Kg | [155]—2016 |
| triazines | fruits, vegetables | fiber—ionic liquid (IL)-calixarene | GC-FID | 25–5000 μg/Kg | [156]—2014 |
| PAHs | potatoes | fiber—Bis(trifluoromethanesulfonyl)imide-based ionic liquids grafted on graphene oxide | GC-FID | 0.05–50 μg/Kg | [158]—2016 |
| parathion | apple, strawberry, celery | fiber—Halloysite nanotubes-titanium dioxide (HNTs-TiO2) | negative corona discharge-ion mobility spectrometer | 0.1–25 μg/Kg | [159]—2016 |
| multiclass pesticides | cucumber, pear, green tea | fiber—C3N4 (ZnO/g-C3N4) | GC-MS | 3–5000 ng/Kg | [160]—2016 |
| multiclass pesticides and contaminants | avocado puree | fiber—PDMS/DVB/PDMS | GCxGC-ToF/MS | - | [162]—2017 |
| multiclass pesticides and contaminants | spinach, tomatoes, carrots | fiber—PDMS/DVB/PDMS | GC-MS | - | [163]—2016 |
| multiclass pesticides | grapes | fiber—PDMS-modified PDMS/DVB | GC-ToFMS | 1–1000 μg/Kg | [164]—2015 |
| multiclass pesticide | fruits, vegetables | fiber—PDMS | GC-MS | 1–500 μg/Kg | [168]—2015 |
| Benzobenzo[a]pyrene | living fish | fiber—mixed-mode/PAN | LC-MS | - | [183]—2018 |
| anesthetics | living fish | fiber—custom-made PDMS | GC-MS | 10–5000 μg/Kg | [184]—2017 |
| trihalomethanes | fish | fiber—DVB-CAR-PDMS | GC-ECD | 0.35–8 μg/Kg | [185]—2017 |
| PCBs | fish | SBSE—magnetic metal-organic frameworks—Fe3O4-MOF-5(Fe) | GC-MS | 0.01–500 μg/Kg | [187]—2015 |
| sudan dyes (I, II, III and IV), Rhodamine B | chili oil, chili powder | film—carbon nanotube | desorption corona beam ionization (DCBI) | 100–20,000 μg/Kg | [200]—2015 |
| Sudan dyes | chili powder | glass capillary—poly(BMA–EDMA–MAA) | DART-SVP-ToF MS | 20–2000 μg/Kg | [201]—2016 |
| PAHs, PCBs and pesticides | edible seaweeds | fiber—PDMS/DVB/PDMS | GC-MS | 5–2000 μg/Kg | [202]—2018 |
| triazine herbicides | vegetable oils | magnetic dispersive—1-hexyl-3-methylimidazolium tetrachloroferrate ([C6mim] [FeCl4] | HPLC-UV | 5–1000 μg/L | [197]—2014 |
| phthalates | drinking water, edible vegetable oil | fiber—graphene/polyvinylchloride | GC-FID | 0.3–100 μg/L | [196]—2016 |
| sulfonamides | butter | magnetic bar—1-octyl-3-methylimidazolium hexafluorophosphate ([C8MIM][PF6-] | HPLC-UV | 6–300 μg/Kg | [198]—2015 |
| PAHs | smoked rice | hollow fiber—MWCNTs | GC-FID | 0.02–1000 μg/Kg | [199]—2014 |
| phenols | honey | fiber—SNW-1 | GC-MS | 0.1–100 μg/Kg | [188]—2016 |
| organochlorine pesticides, PAHs, PCBs | honey | fiber—PDMS | GC-MS/MS | 10–3000 μg/Kg | [189]—2017 |
| phenols | honey | fiber—COF-SNW-1 | GC-MS | 0.1–100 μg/Kg | [188]—2016 |
| benzoylurea | honey, tea | dispersive ionic liquid (IL)-modified β-cyclodextrin/attapulgite (β-CD/ATP) | HPLC-DAD | 5–500 μg/L | [190]—2016 |
| phthalates | tea | fiber—C-NH2-MIL-125 | GC-MS | 0.05–30 μg/L | [191]—2016 |
| dicofol residues | tea | magnetic dispersive—magnetic molecular imprinted microspheres with DDT as template | GC-ECD | 0.2–160 μg/L | [192]—2014 |
| PAHs | tea | film—agarose-chitosan-C18 | HPLC-UV | 1–500 μg/L | [193]—2017 |
| acrylamide | brewed coffee and coffee powder | fiber—nine crosslinked PIL based coatings | GC-MS | 0.5–200 μg/Kg | [194]—2016 |
| phthalates | coffee | fiber—crosslinked PIL-based nitinol | GC-MS | - | [195]—2014 |
| Analytes | Matrix | SPME Approach and Coating Material | Instrumentation | Calibration Range | [Ref] Publishing Year |
|---|---|---|---|---|---|
| PAHs | urine | fiber—PDMS/DVB/PDMS | GC-MS/MS | 0.05–100 μg/L | [74]—2018 |
| OH-PAHs | urine | fiber—C18/PAN | nanoESI-MS/MS | 0.1–5 μg/L | [213]—2017 |
| endocrine disrupting chemicals (EDCs) | urine | multiple monolithic fiber—1-trimethyl-(4-vinylbenzyl) aminium chloride as monomer | HPLC-DAD | 0.1–200 μg/L | [83]—2017 |
| VOCs | urine | fiber—CAR/PDMS | GC-MS | 2.9–1500 μg/L | [214]—2016 |
| aromatic amines | urine | fiber—PDMS/DVB | GCxGC-qMS | 1–500 ng/L | [215]—2015 |
| VOCs | urine | fiber—CAR/PDMS | GC-MS | 2.5–100 μg/L | [216]—2017 |
| organochlorine pesticides | urine | fiber—PDMS | GC–MS | 0.5–20 μg/L | [217]—2016 |
| benzothiazoles, benzotriazoles, benzosulfonamides | urine | fiber—PA | GC-MS/MS | 1–100 μg/L | [14]—2014 |
| organochlorine pesticides | urine | stir bar—double hydroxide/graphene (LDH-G) | GC-MS | 1–200 μg/L | [218]—2017 |
| monohydroxy-PAHs | urine | in tube—inorganic–organic hybrid nanocomposite (zinc oxide/polypyrrole) | LC-MS/MS | 0.2–100 μg/L | [219]—2016 |
| multiclass pesticides, PCBs | hair | fiber—PDMS–DVB | GC–MS/MS | 0.002–10 μg/Kg | [220]—2015 |
| PAHs | hair | in tube—CP-Sil 19CB (14% cyanopropyl phenyl methylsilicone) | HPLC-FLD | 0.02–1 μg/Kg | [221]—2015 |
| DDTs, PCBs | hair | fiber—PDMS/DVB | GC-MS | 2.5–50 ng/Kg | [222]—2014 |
| perfluorinated compounds (PFCs) | blood | surface coated wooden-tip probe—n-octadecyldimethyl[3-(trimethoxysilyl)propyl]ammonium chloride | Orbitrap MS | 0.5–100 ng/L | [29]—2014 |
| PCBs, PAHs | Serum | fiber—mesoporous graphitic carbon nitride@NiCo2O4 nanocomposite | GC-FID | 0.002–100 μg/L | [224]—2019 |
| organochlorine pesticides, PCBs | Serum | fiber—PDMS | GC-MS | 3–100 μg/L | [223]—2014 |
| BTEX, chlorinated solvents | meconium | fiber—CAR/PDMS | GC-MS | 0.08–9 (ng) | [225]—2014 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Naccarato, A.; Tagarelli, A. Recent Applications and Newly Developed Strategies of Solid-Phase Microextraction in Contaminant Analysis: Through the Environment to Humans. Separations 2019, 6, 54. https://doi.org/10.3390/separations6040054
Naccarato A, Tagarelli A. Recent Applications and Newly Developed Strategies of Solid-Phase Microextraction in Contaminant Analysis: Through the Environment to Humans. Separations. 2019; 6(4):54. https://doi.org/10.3390/separations6040054
Chicago/Turabian StyleNaccarato, Attilio, and Antonio Tagarelli. 2019. "Recent Applications and Newly Developed Strategies of Solid-Phase Microextraction in Contaminant Analysis: Through the Environment to Humans" Separations 6, no. 4: 54. https://doi.org/10.3390/separations6040054
APA StyleNaccarato, A., & Tagarelli, A. (2019). Recent Applications and Newly Developed Strategies of Solid-Phase Microextraction in Contaminant Analysis: Through the Environment to Humans. Separations, 6(4), 54. https://doi.org/10.3390/separations6040054






