Low-Cost Quantitation of Multiple Volatile Organic Compounds in Air Using Solid-Phase Microextraction
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. GC-MS Conditions
2.3. Selection of the Optimal SPME Fiber
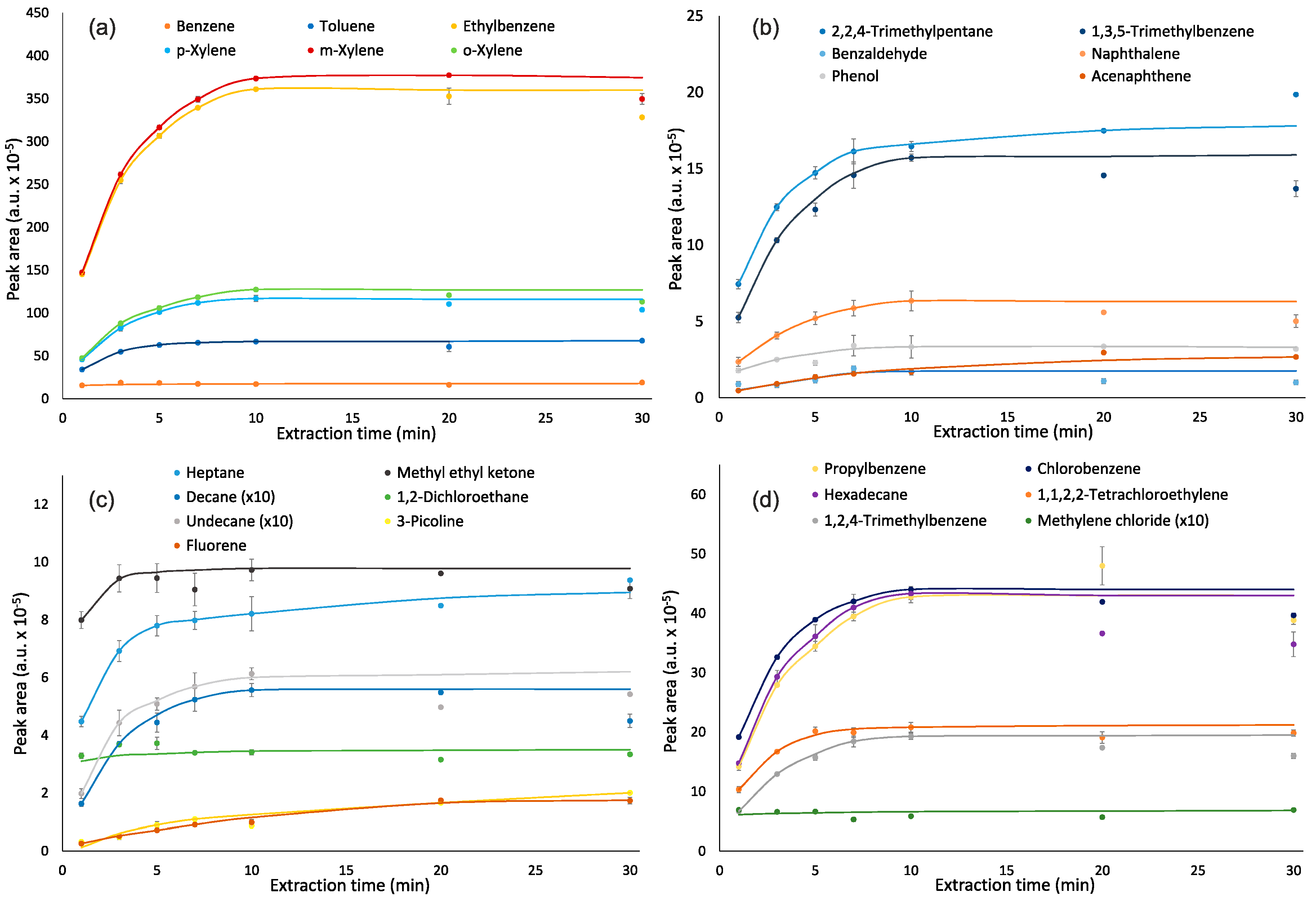
2.4. Effects of Extraction and Desorption Times
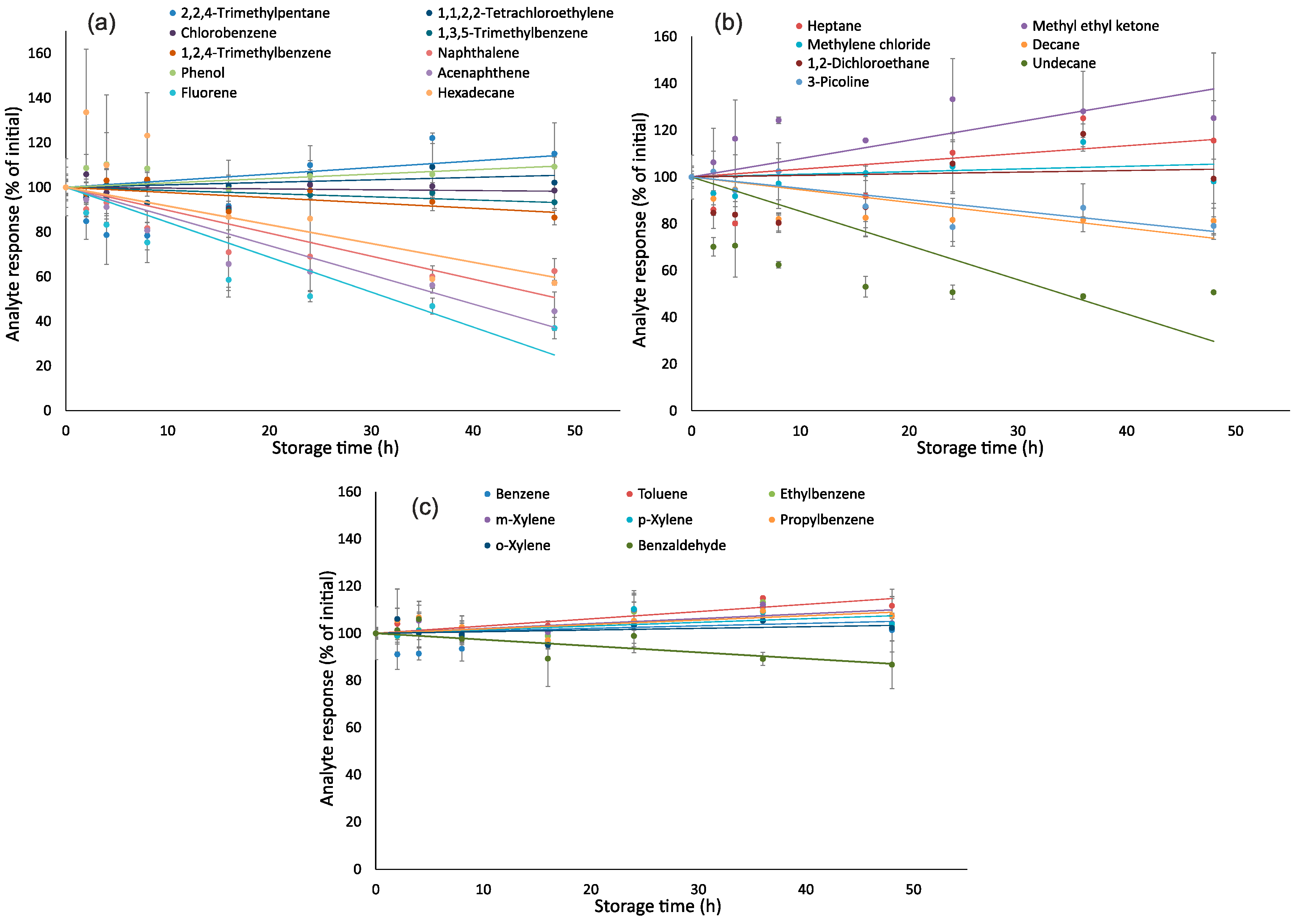
2.5. Effect of Storage Time
2.6. Estimation of the Method Accuracy
2.7. Air Sampling and Analysis
3. Results
3.1. Selection of the Optimal SPME Fiber
3.2. Effects of Extraction and Desorption Times
3.3. Effect of Storage Time
3.4. Estimation of the Method Accuracy
3.5. Air Sampling and Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- World Health Organization. Ambient Air Pollution: A Global Assessment of Exposure and Burden of Disease; WHO: Geneva, Switzerland, 2016. [Google Scholar]
- US EPA. Compendium Method TO-14A Determination of Volatile Organic Compounds (VOCs) in Ambient Air Using Specially Prepared Canisters with Subsequent Analysis by Gas Chromatography; United States Environmental Protection Agency: Cincinnati, OH, USA, 1999.
- US EPA. Compendium Method TO-17 Determination of Volatile Organic Compounds in Ambient Air Using Active Sampling Onto Sorbent Tubes; United States Environmental Protection Agency: Cincinnati, OH, USA, 1999.
- Król, S.; Zabiegała, B.; Namieśnik, J. Monitoring VOCs in atmospheric air II. Sample collection and preparation. Trends Anal. Chem. 2010, 29, 1101–1112. [Google Scholar] [CrossRef]
- Miller, L.; Xu, X. Multi-season, multi-year concentrations and correlations amongst the BTEX group of VOCs in an urbanized industrial city. Atmos. Environ. 2012, 61, 305–315. [Google Scholar] [CrossRef]
- Goodman, N.B.; Steinemann, A.; Wheeler, A.J.; Paevere, P.J.; Cheng, M.; Brown, S.K. Volatile organic compounds within indoor environments in Australia. Build. Environ. 2017, 122, 116–125. [Google Scholar] [CrossRef]
- Woolfenden, E. Monitoring VOCs in air using sorbent tubes followed by thermal desorption-capillary GC analysis: Summary of data and practical guidelines. J. Air Waste Manag. Assoc. 1997, 47, 20–36. [Google Scholar] [CrossRef]
- Pawliszyn, J. (Ed.) Applications of Solid Phase Microextraction. In RSC Chromatography Monographs; Royal Society of Chemistry: Cambridge, UK, 1999; ISBN 978-0-85404-525-9. [Google Scholar]
- Haberhauer-Troyer, C.; Rosenberg, E.; Grasserbauer, M. Evaluation of solid-phase microextraction for sampling of volatile organic sulfur compounds in air for subsequent gas chromatographic analysis with atomic emission detection. J. Chromatogr. A 1999, 848, 305–315. [Google Scholar] [CrossRef]
- Tuduri, L.; Desauziers, V.; Fanlo, J.L. Dynamic versus static sampling for the quantitative analysis of volatile organic compounds in air with polydimethylsiloxane-Carboxen solid-phase microextraction fibers. J. Chromatogr. A 2002, 963, 49–56. [Google Scholar] [CrossRef]
- Ouyang, G. 8-SPME and Environmental Analysis. In Handbook of Solid Phase Microextraction; Elsevier Inc.: Amsterdam, The Netherlands, 2012; pp. 251–290. [Google Scholar] [CrossRef]
- Tumbiolo, S.; Gal, J.-F.; Maria, P.-C.; Zerbinati, O. Determination of benzene, toluene, ethylbenzene and xylenes in air by solid phase micro-extraction/gas chromatography/mass spectrometry. Anal. Bioanal. Chem. 2004, 380, 824–830. [Google Scholar] [CrossRef] [PubMed]
- Prikryl, P.; Sevcik, J.G.K. Characterization of sorption mechanisms of solid-phase microextraction with volatile organic compounds in air samples using a linear solvation energy relationship approach. J. Chromatogr. A 2008, 1179, 24–32. [Google Scholar] [CrossRef]
- Yassaa, N.; Meklati, B.Y.; Cecinato, A. Analysis of volatile organic compounds in the ambient air of Algiers by gas chromatography with a b-cyclodextrin capillary column. J. Chromatogr. A 1999, 846, 287–293. [Google Scholar] [CrossRef]
- Tumbiolo, S.; Gal, J.-F.; Maria, P.; Zerbinati, O. SPME sampling of BTEX before GC/MS analysis: Examples of outdoor and indoor air quality measurements in public and private sites. Ann. Chim. 2005, 95, 757–766. [Google Scholar] [CrossRef]
- Koziel, J.A.; Pawliszyn, J. Air sampling and analysis of volatile organic compounds with solid phase microextraction. J. Air Waste Manag. Assoc. 2001, 51, 173–184. [Google Scholar] [CrossRef] [PubMed]
- Curran, K.; Underhill, M.; Gibson, L.T.; Strlic, M. The development of a SPME-GC/MS method for the analysis of VOC emissions from historic plastic and rubber materials. Microchem. J. 2016, 124, 909–918. [Google Scholar] [CrossRef]
- Luca, A.; Kjær, A.; Edelenbos, M. Volatile organic compounds as markers of quality changes during the storage of wild rocket. Food Chem. 2017, 232, 579–586. [Google Scholar] [CrossRef]
- Parshintsev, J.; Niina, K.; Hartonen, K.; Miguel, L.; Jussila, M.; Kajos, M.; Kulmala, M.; Riekkola, M. Field measurements of biogenic volatile organic compounds in the atmosphere by dynamic solid-phase microextraction and portable gas chromatography-mass spectrometry. Atmos. Environ. 2015, 115, 214–222. [Google Scholar]
- Zain, S.M.S.M.; Shaharudin, R.; Kamaluddin, M.A.; Daud, S.F. Determination of hydrogen cyanide in residential ambient air using SPME coupled with GC–MS. Atmos. Pollut. Res. 2017, 8, 678–685. [Google Scholar] [CrossRef]
- Mokbel, H.; Al Dine, E.J.; Elmoll, A.; Liaud, C.; Millet, M. Simultaneous analysis of organochlorine pesticides and polychlorinated biphenyls in air samples by using accelerated solvent extraction (ASE) and solid-phase micro-extraction (SPME) coupled to gas chromatography dual electron capture detection. Environ. Sci. Pollut. Res. 2016, 23, 8053–8063. [Google Scholar] [CrossRef]
- Naccarato, A.; Tassone, A.; Moretti, S.; Elliani, R.; Sprovieri, F.; Pirrone, N.; Tagarelli, A. A green approach for organophosphate ester determination in airborne particulate matter: Microwave-assisted extraction using hydroalcoholic mixture coupled with solid-phase microextraction gas chromatography-tandem mass spectrometry. Talanta 2018, 189, 657–665. [Google Scholar] [CrossRef]
- Hussam, A.; Alauddin, M.; Khan, A.H.; Chowdhury, D.; Bibi, H.; Bhattacharjee, M.; Sultana, S. Solid phase microextraction: Measurement of volatile organic compounds (VOCs) in Dhaka city air pollution. J. Environ. Sci. Health Part A 2002, 37, 1223–1239. [Google Scholar] [CrossRef]
- Lee, J.H.; Hwang, S.M.; Lee, D.W.; Heo, G.S. Determination of volatile organic compounds (VOCs) using tedlar bag/solid-phase microextraction/gas chromatography/mass spectrometry (SPME/GC/MS) in ambient and workplace air. Bull. Korean Chem. Soc. 2002, 23, 488–496. [Google Scholar]
- Baimatova, N.; Kenessov, B.; Koziel, J.A.; Carlsen, L.; Bektassov, M.; Demyanenko, O.P. Simple and accurate quantification of BTEX in ambient air by SPME and GC-MS. Talanta 2016, 154, 46–52. [Google Scholar] [CrossRef]
- Woolcock, P.J.; Koziel, J.A.; Johnston, P.A.; Brown, R.C.; Broer, K.M. Analysis of trace contaminants in hot gas streams using time-weighted average solid-phase microextraction: Pilot-scale validation. Fuel 2015, 153, 552–558. [Google Scholar] [CrossRef]
- Carlsen, L.; Baimatova, N.; Kenessov, B.; Kenessova, O. Assessment of the air quality of Almaty. Focussing on the traffic component. Int. J. Biol. Chem. 2013, 5, 49–69. [Google Scholar]
- Nicoara, S.; Tonidandel, L.; Traldi, P.; Watson, J.; Morgan, G.; Popa, O. Determining the levels of volatile organic pollutants in urban air using a gas chromatography-mass spectrometry method. J. Environ. Public Health 2009, 2009, 148527. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.C.; Chiu, M.Y. Volatile organic compounds (VOCs) in urban atmosphere of Hong Kong. Chemosphere 2002, 48, 375–382. [Google Scholar] [CrossRef]
- Khan, A.; Szulejko, J.E.; Kim, K.H.; Brown, R.J.C. Airborne volatile aromatic hydrocarbons at an urban monitoring station in Korea from 2013 to 2015. J. Environ. Manag. 2018, 209, 525–538. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Li, H.; Zhang, X.; Wang, L.; Xu, L.; Wang, X.; Yu, Y.; Zhang, Y.; Cao, G. Pollution characteristics and health risk assessment of benzene homologues in ambient air in the northeastern urban area of Beijing, China. J. Environ. Sci. 2014, 26, 214–223. [Google Scholar] [CrossRef]
- Zhang, Y.; Mu, Y. Atmospheric BTEX and carbonyls during summer seasons of 2008–2010 in Beijing. Atmos. Environ. 2012, 59, 186–191. [Google Scholar] [CrossRef]
- Bigazzi, A.Y.; Figliozzi, M.A.; Luo, W.; Pankow, J.F. Breath Biomarkers to Measure Uptake of Volatile Organic Compounds by Bicyclists. Environ. Sci. Technol. 2016, 50, 5357–5363. [Google Scholar] [CrossRef]
- Pawliszyn, J. Handbook of Solid Phase Microextraction; Elsevier Inc.: Amsterdam, The Netherlands, 2012; ISBN 9780124160170. [Google Scholar]
- Kenessov, B.; Derbissalin, M.; Koziel, J.A.; Kosyakov, D.S. Modeling solid-phase microextraction of volatile organic compounds by porous coatings using finite element analysis. Anal. Chim. Acta 2019, 1076, 73–81. [Google Scholar] [CrossRef]
- Lion, L.W. Sorption and Transport of Polynuclear Aromatic Hydrocarbons in Low-Carbon Aquifer Materials; AFESC: Ithaca, NY, USA, 1988. [Google Scholar]
| Sampling Principle | SPME Fiber, Extraction Time | Instrument | Analytes | LOD (µg m−3) | Ref. | |
|---|---|---|---|---|---|---|
| SPME from open air | 75-µm Car/PDMS, 100-µm PDMS, 20 min | GC-AED | Car/PDMS | PDMS | [9] | |
| Methanethiol | 0.04–0.06 | 4 | ||||
| Dimethyl sulfide | 0.003–0.004 | 2 | ||||
| Isopropanethiol | 0.005–0.007 | 2 | ||||
| Isobutanethiol | 0.003–0.004 | 0.7 | ||||
| SPME from static or moving air | 75-µm Car/PDMS, 1 min | GC-FID | Methanol | 2–5 | [10] | |
| Acetone | ||||||
| Dichloromethane | ||||||
| Methyl ethyl ketone | ||||||
| Ethyl acetate | ||||||
| Dichloroethane | ||||||
| Methyl isobutyl ketone | ||||||
| Toluene | ||||||
| Butyl acetate | ||||||
| Ethylbenzene | ||||||
| p-Xylene | ||||||
| SPME from fan-blown air | 65-µm PDMS/DVB, 2 h | GC-MS | Δ3-Carene | n/a | [19] | |
| α-Pinene | ||||||
| Limonene | ||||||
| Pinonaldehyde | ||||||
| Pinonic acid | ||||||
| Dimethylamine + ethylamine | ||||||
| Air purging via bubbler impinger with KOH solution, HS SPME | 75-µm Car/PDMS, | GC-MS | HCN | 0.16 | [20] | |
| 5 min | ||||||
| Sampling on XAD-2 resin, accelerated solvent extraction with ACN, dilution with water, DI SPME | 100-µm PDMS, | GC-Dual ECD | 22 PCBs | 2 × 10−5–4.9 × 10−3 | [21] | |
| 40 min | 19 OCPs | |||||
| Sampling of PM10 on quartz fiber filter, microwave extraction with ethanol-water mixture, dilution with water and DI SPME | 50/30-µm DVB/Car/PDMS, 5 min | GC-MS/MS | Tripropyl phosphate | 20 | [22] | |
| Tri-n-butyl phosphate | 40 | |||||
| Tris(2-chloroethyl) phosphate | 70 | |||||
| Tris(1-chloro-2-propyl) phosphate | 42 | |||||
| Tris(1,3-dichloro-2-propyl) phosphate | 138 | |||||
| Triphenyl phosphate | 51 | |||||
| Tricresyl phosphate | 60 | |||||
| SPME from open air | 100-µm PDMS, 30-45 min | GC-MS | BTEX | 1–100 | [23] | |
| propylbenzene | ||||||
| 1,3,5-trimethylbenzene butyl benzene alkanes (C5, C10-C27) | ||||||
| Sampling to Tedlar bags, SPME | 75-µm Car/PDMS, | GC-MS | 36 VOCs | 0.01–0.93 | [24] | |
| 15 min | ||||||
| Sampling into 20-mL vials, SPME | 100-µm PDMS, 3 min | GC-MS | Benzene | 5 | [25] | |
| Toluene | 2 | |||||
| Ethylbenzene | 2 | |||||
| o-Xylene | 2 | |||||
| Sampling into 20-mL vials, SPME | 65-µm PDMS/DVB, | GC-MS | 25 VOCs | 0.01–6.9 | This study | |
| 10 min | ||||||
| No. | Retention Time (min) | Group No. | Analyte | Quantification Ion m z−1 (amu (dwell)) | Confirmation Ions m z−1 amu (dwell)) | Group Start Time (min) |
|---|---|---|---|---|---|---|
| 1 | 5.1 | 1 | 2,2,4-Trimethylpentane | 57 (100) | 56, 41 (50) | 0 |
| 2 | 5.5 | n-Heptane | 43 (50) | 41 (50) | ||
| 3 | 9.9 | Methyl ethyl ketone | 43 (50) | 72 (50) | ||
| 4 | 10.4 | 2 | Methylene chloride | 49 (100) | 84 (50) | 10.2 |
| 5 | 10.8 | Benzene | 78 (100) | |||
| 6 | 11.8 | n-Decane | 142 (100) | |||
| 7 | 12.6 | 3 | 1,1,2,2-Tetrachloroethylene | 166 (50) | 164 (50) | 12.2 |
| 8 | 13.1 | Toluene | 91 (100) | |||
| 9 | 13.5 | 1,2-Dichloroethane | 62 (100) | 64 (50) | ||
| 10 | 13.8 | n-Undecane | 156 (100) | |||
| 11 | 14.9 | Ethylbenzene | 106 (100) | |||
| 12 | 15.0 | m-Xylene | ||||
| 13 | 15.1 | p-Xylene | ||||
| 14 | 15.7 | 4 | Propylbenzene | 105 (100) | 15.5 | |
| 15 | 15.9 | o-Xylene | 106 (50) | |||
| 16 | 16.5 | Chlorobenzene | 112 (100) | 77 (50) | ||
| 17 | 17.0 | 1,3,5-Trimethylbenzene | 105 (100) | |||
| 18 | 17.7 | 1,2,4-Trimethylbenzene | ||||
| 19 | 18.1 | 3-Picoline | 93 (100) | 66 (50) | ||
| 20 | 23.4 | 5 | Benzaldehyde | 77 (100) | 22.0 | |
| 21 | 24.8 | n-Hexadecane | 57 (100) | 43 (100) | ||
| 22 | 27.9 | Naphthalene | 128 (100) | |||
| 23 | 31.0 | 6 | Phenol | 94 (100) | 30.0 | |
| 24 | 33.7 | Acenaphthene | 153 (100) | |||
| 25 | 36.8 | Fluorene | 166 (100) |
| Analyte | Limit of Detection (µg m−3) | |||
|---|---|---|---|---|
| Car/PDMS | PDMS | DVB/Car/PDMS | PDMS/DVB | |
| 2,2,4-Trimethylpentane | 3 | 8 | 0.7 | 1.8 |
| n-Heptane | 50 | 9 | 8 | 7 |
| Methyl ethyl ketone | 15 | 7 | 0.8 | 1.9 |
| Methylene chloride | 1.8 | 9 | 1.1 | 0.6 |
| Benzene | 0.6 | 6 | 0.5 | 1.2 |
| n-Decane | 3 | 3 | 1.2 | 1.5 |
| 1,1,2,2-Tetrachloroethylene | 0.04 | 0.2 | 0.04 | 0.04 |
| Toluene | 0.5 | 4 | 1.6 | 1.2 |
| 1,2-Dichloroethane | 0.2 | 1.2 | 0.8 | 1.7 |
| n-Undecane | 2 | 1.0 | 0.8 | 1.0 |
| Ethylbenzene | 0.010 | 0.10 | 0.2 | 0.03 |
| m-Xylene | 0.2 | 0.2 | 0.6 | 0.2 |
| p-Xylene | 0.10 | 0.2 | 0.2 | 0.2 |
| Propylbenzene | 0.3 | 0.3 | 0.2 | 0.10 |
| o-Xylene | 0.04 | 0.10 | 0.10 | 0.04 |
| Chlorobenzene | 0.04 | 0.10 | 0.05 | 0.04 |
| 1,3,5-Trimethylbenzene | 0.3 | 0.10 | 0.10 | 0.10 |
| 1,2,4-Trimethylbenzene | 0.2 | 0.04 | 0.04 | 0.10 |
| 3-Picoline | 0.5 | 0.2 | 0.10 | 0.010 |
| Benzaldehyde | 0.10 | 0.10 | 0.4 | 0.10 |
| n-Hexadecane | - | 5 | 10 | 5 |
| Naphthalene | 0.2 | 0.10 | 0.10 | 0.04 |
| Phenol | 0.8 | 0.10 | 0.10 | 0.10 |
| Acenaphthene | 0.3 | 0.10 | 0.2 | 0.10 |
| Fluorene | 5 | 0.10 | 0.6 | 0.10 |
| Number of analytes with a limit of detection ≤ | ||||
| 1 µg m−3 | 17 | 16 | 20 | 17 |
| 2 µg m−3 | 19 | 17 | 23 | 23 |
| 5 µg m−3 | 22 | 20 | 23 | 24 |
| 10 µg m−3 | 22 | 25 | 25 | 25 |
| Analyte | Relative Standard Deviation of a Slope (%) | |||
|---|---|---|---|---|
| Car/PDMS | PDMS | DVB/Car/PDMS | PDMS/DVB | |
| 2,2,4-Trimethylpentane | 0.50 | 2.7 | 1.7 | 2.7 |
| n-Heptane | 44 | 11 | 4.5 | 5.2 |
| Methyl ethyl ketone | 42 | 14 | 7.8 | 25 |
| Methylene chloride | 95 | 200 | 5.3 | 4.2 |
| Benzene | 4.4 | 9.4 | 3.2 | 1.1 |
| n-Decane | 3.5 | 5.6 | 0.50 | 5.9 |
| 1,1,2,2-Tetrachloroethylene | 2.6 | 1.2 | 0.40 | 3.1 |
| Toluene | 18 | 3.4 | 5.8 | 6.4 |
| 1,2-Dichloroethane | 2.1 | 9.0 | 10 | 20 |
| n-Undecane | 5.9 | 5.7 | 1.8 | 4.7 |
| Ethylbenzene | 0.50 | 2.7 | 4.8 | 4.4 |
| m-Xylene | 3.8 | 1.7 | 9.6 | 4.6 |
| p-Xylene | 1.2 | 6.8 | 7.5 | 15 |
| Propylbenzene | 4.0 | 2.4 | 9.6 | 2.2 |
| o-Xylene | 0.60 | 1.3 | 7.5 | 3.4 |
| Chlorobenzene | 3.8 | 1.3 | 1.3 | 1.3 |
| 1,3,5-Trimethylbenzene | 0.30 | 0.80 | 2.2 | 3.2 |
| 1,2,4-Trimethylbenzene | 7.6 | 3.5 | 6.1 | 3.8 |
| 3-Picoline | 5.1 | 6.5 | 15 | 6.2 |
| Benzaldehyde | 8.0 | 3.2 | 79 | 3.7 |
| n-Hexadecane | - | 4.5 | 32 | 8.3 |
| Naphthalene | 5.7 | 1.8 | 20 | 2.2 |
| Phenol | 15 | 1.4 | 24 | 2.5 |
| Acenaphthene | 4.2 | 5.3 | 4.3 | 2.5 |
| Fluorene | 5.5 | 1.5 | 4.0 | 3.5 |
| Number of analytes with a relative standard deviation of a slope ≤ | ||||
| 1% | 4 | 1 | 2 | 0 |
| 2% | 5 | 8 | 5 | 2 |
| 5% | 13 | 15 | 11 | 17 |
| 10% | 19 | 22 | 20 | 22 |
| Analyte | Cair (µg m−3) | Csp (µg m−3) | Cmeas (µg m−3) | Recovery (%) |
|---|---|---|---|---|
| 2,2,4-Trimethylpentane | 22.3 | 100 | 124 | 102 |
| n-Heptane | 55.0 | 100 | 158 | 103 |
| Methyl ethyl ketone | 88.4 | 10 | 93.6 | 51 |
| Methylene chloride | 20.7 | 10 | 29.6 | 89 |
| Benzene | 80.2 | 100 | 183 | 103 |
| n-Decane | 17.7 | 100 | 117 | 100 |
| 1,1,2,2-Tetrachloroethylene | 0.58 | 10 | 10.6 | 100 |
| Toluene | 25.7 | 100 | 128 | 102 |
| 1,2-Dichloroethane | n/d | 10 | 9.3 | 93 |
| n-Undecane | 50.2 | 100 | 146 | 96 |
| Ethylbenzene | 1.36 | 10 | 11.4 | 101 |
| m-Xylene | 1.74 | 10 | 11.9 | 102 |
| p-Xylene | 1.92 | 10 | 12.0 | 100 |
| Propylbenzene | 1.17 | 10 | 11.2 | 100 |
| o-Xylene | 1.57 | 10 | 11.7 | 101 |
| Chlorobenzene | 0.60 | 10 | 10.6 | 100 |
| 1,3,5-Trimethylbenzene | 1.82 | 10 | 11.6 | 97 |
| 1,2,4-Trimethylbenzene | 4.33 | 10 | 14.2 | 98 |
| 3-Picoline | 8.63 | 10 | 17.2 | 86 |
| Benzaldehyde | 0.83 | 10 | 10.4 | 96 |
| n-Hexadecane | 132 | 100 | 213 | 81 |
| Naphthalene | 3.80 | 10 | 13.6 | 98 |
| Phenol | 3.52 | 10 | 13.5 | 100 |
| Acenaphthene | 1.61 | 10 | 12.1 | 105 |
| Fluorene | 4.64 | 10 | 14.7 | 101 |
| Sampling Date >> | Concentration ± Standard Deviation (µg m−3) | Outliers | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Saturday, 30 March | Tuesday, 2 April | Thursday, 4 April | |||||||||
| Analyte | 8 AM | 8 PM | Mean | 8 AM | 8 PM | Mean | 8 AM | 8 PM | Mean | Number | % |
| 2,2,4-Trimethylpentane | 18 ± 2 | 20 ± 2 | 19 ± 2 | 19 ± 3 | 15 ± 2 | 17 ± 2 | 34 ± 6 | 22 ± 2 | 28 ± 4 | 6 | 5.7 |
| n-Heptane | 46 ± 5 | 53 ± 5 | 50 ± 5 | 46 ± 3 | 38 ± 5 | 42 ± 4 | 89 ± 9 | 60 ± 6 | 74 ± 8 | 5 | 4.6 |
| Methyl ethyl ketone | n/d | n/d | n/d | n/d | n/d | n/d | n/d | n/d | n/d | n/a | n/a |
| Methylene chloride | 25 ± 7 | 25 ± 5 | 25 ± 6 | 10 ± 1 | 8 ± 2 | 9 ± 2 | 45 ± 7 | 53 ± 13 | 49 ± 10 | 14 | 13 |
| Benzene | 40 ± 5 | 56 ± 4 | 48 ± 4 | 37 ± 3 | 46 ± 9 | 41 ± 6 | 48 ± 4 | 56 ± 7 | 52 ± 6 | 6 | 5.6 |
| n-Decane | 11 ± 3 | 13 ± 2 | 12 ± 2 | 4.8 ± 1.2 | 8.4 ± 1.0 | 6.6 ± 1.1 | 30 ± 6 | 28 ± 4 | 29 ± 5 | 6 | 5.6 |
| 1,1,2,2-Tetrachloroethylene | 0.14 ± 0.05 | 0.18 ± 0.03 | 0.16 ± 0.04 | 0.08 ± 0.01 | 0.12 ± 0.07 | 0.10 ± 0.04 | 1.3 ± 0.6 | 1.2 ± 0.2 | 1.2 ± 0.4 | 9 | 8.3 |
| Toluene | 34 ± 3 | 50 ± 3 | 42 ± 3 | 28 ± 1 | 21 ± 6 | 25 ± 3 | 81 ± 15 | 60 ± 6 | 70 ± 11 | 6 | 5.6 |
| 1,2-Dichloroethane | n/d | n/d | n/d | n/d | n/d | n/d | n/d | n/d | n/d | n/a | n/a |
| n-Undecane | 22 ± 6 | 27 ± 3 | 25 ± 4 | 13 ± 2 | 14 ± 2 | 13 ± 2 | 36 ± 7 | 22 ± 4 | 29 ± 5 | 13 | 12 |
| Ethylbenzene | 0.49 ± 0.13 | 0.68 ± 0.05 | 0.58 ± 0.09 | 0.43 ± 0.02 | 0.35 ± 0.03 | 0.39 ± 0.03 | 0.94 ± 0.14 | 0.84 ± 0.08 | 0.89 ± 0.11 | n/d | n/d |
| m-Xylene | 0.55 ± 0.14 | 0.90 ± 0.05 | 0.73 ± 0.09 | 0.67 ± 0.03 | 0.47 ± 0.04 | 0.57 ± 0.04 | 1.3 ± 0.2 | 0.99 ± 0.09 | 1.1 ± 0.2 | n/d | n/d |
| p-Xylene | 0.81 ± 0.16 | 1.26 ± 0.09 | 1.04 ± 0.13 | 0.95 ± 0.04 | 0.72 ± 0.07 | 0.83 ± 0.05 | 1.9 ± 0.2 | 1.82 ± 0.12 | 1.9 ± 0.2 | 1 | 1.0 |
| Propylbenzene | 0.28 ± 0.12 | 0.11 ± 0.02 | 0.20 ± 0.07 | 0.18 ± 0.01 | 0.06 ± 0.02 | 0.12 ± 0.02 | 0.22 ± 0.13 | 0.07 ± 0.02 | 0.14 ± 0.08 | 11 | 10 |
| o-Xylene | 0.55 ± 0.14 | 0.59 ± 0.04 | 0.57 ± 0.09 | 0.45 ± 0.13 | 0.30 ± 0.02 | 0.37 ± 0.08 | 0.70 ± 0.16 | 0.61 ± 0.04 | 0.65 ± 0.10 | 2 | 1.9 |
| Chlorobenzene | 1.1 ± 0.4 | 1.49 ± 0.09 | 1.3 ± 0.2 | 1.1 ± 0.3 | 0.76 ± 0.06 | 0.9 ± 0.2 | 1.5 ± 0.2 | 1.25 ± 0.07 | 1.38 ± 0.18 | 2 | 1.9 |
| 1,3,5-Trimethylbenzene | 2.0 ± 0.6 | 1.22 ± 0.08 | 1.6 ± 0.4 | 2.1 ± 0.2 | 0.79 ± 0.05 | 1.45 ± 0.13 | 3.1 ± 1.0 | 1.09 ± 0.05 | 2.1 ± 0.5 | 3 | 2.8 |
| 1,2,4-Trimethylbenzene | 1.3 ± 0.5 | 2.97 ± 0.14 | 2.2 ± 0.3 | 1.3 ± 0.4 | 1.57 ± 0.11 | 1.4 ± 0.3 | 2.0 ± 0.8 | 3.2 ± 0.7 | 2.6 ± 0.7 | 5 | 4.6 |
| 3-Picoline | 8.1 ± 2.6 | 0.9 ± 0.2 | 4.5 ± 1.4 | 7.0 ± 1.9 | 0.39 ± 0.08 | 3.7 ± 1.0 | 12.5 ± 5.3 | 1.4 ± 1.1 | 7.0 ± 3.2 | 9 | 8.3 |
| Benzaldehyde | 1.3 ± 0.7 | 1.8 ± 0.3 | 1.5 ± 0.5 | 0.7 ± 0.4 | 1.4 ± 0.2 | 1.1 ± 0.3 | 0.6 ± 0.4 | 1.4 ± 0.5 | 1.0 ± 0.4 | 8 | 7.4 |
| n-Hexadecane | 123 ± 16 | 44 ± 6 | 84 ± 11 | 90 ± 10 | 50 ± 9 | 71 ± 10 | 85 ± 14 | 47 ± 4 | 66 ± 9 | 19 | 18 |
| Naphthalene | 1.8 ± 0.6 | 1.8 ± 0.2 | 1.8 ± 0.4 | 1.3 ± 0.4 | 2.4 ± 0.6 | 1.9 ± 0.5 | 1.5 ± 0.3 | 2.3 ± 0.4 | 1.9 ± 0.3 | 4 | 3.7 |
| Phenol | 2.1 ± 0.6 | 2.9 ± 0.5 | 2.5 ± 0.5 | 2.4 ± 0.6 | 3.9 ± 0.9 | 3.1 ± 0.8 | 3.3 ± 1.1 | 3.3 ± 1.2 | 3.3 ± 1.2 | 10 | 9.3 |
| Acenaphthene | 2.24 ± 0.13 | 0.13 ± 0.04 | 1.19 ± 0.09 | 2.6 ± 0.8 | 0.3 ± 0.03 | 1.4 ± 0.4 | 2.6 ± 0.8 | 0.29 ± 0.10 | 1.5 ± 0.5 | 12 | 12 |
| Fluorene | 0.4 ± 0.2 | 0.59 ± 0.01 | 0.48 ± 0.11 | 0.8 ± 0.2 | 0.57 ± 0.12 | 0.7 ± 0.2 | 0.6 ± 0.3 | 0.5 ± 0.3 | 0.5 ± 0.3 | 7 | 7.2 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ibragimova, O.P.; Baimatova, N.; Kenessov, B. Low-Cost Quantitation of Multiple Volatile Organic Compounds in Air Using Solid-Phase Microextraction. Separations 2019, 6, 51. https://doi.org/10.3390/separations6040051
Ibragimova OP, Baimatova N, Kenessov B. Low-Cost Quantitation of Multiple Volatile Organic Compounds in Air Using Solid-Phase Microextraction. Separations. 2019; 6(4):51. https://doi.org/10.3390/separations6040051
Chicago/Turabian StyleIbragimova, Olga P., Nassiba Baimatova, and Bulat Kenessov. 2019. "Low-Cost Quantitation of Multiple Volatile Organic Compounds in Air Using Solid-Phase Microextraction" Separations 6, no. 4: 51. https://doi.org/10.3390/separations6040051
APA StyleIbragimova, O. P., Baimatova, N., & Kenessov, B. (2019). Low-Cost Quantitation of Multiple Volatile Organic Compounds in Air Using Solid-Phase Microextraction. Separations, 6(4), 51. https://doi.org/10.3390/separations6040051





