Green Innovation for Solid Post-Distillation Residues Valorization: Narrative Review of Circular Bio-Economy Solutions
Abstract
1. Introduction
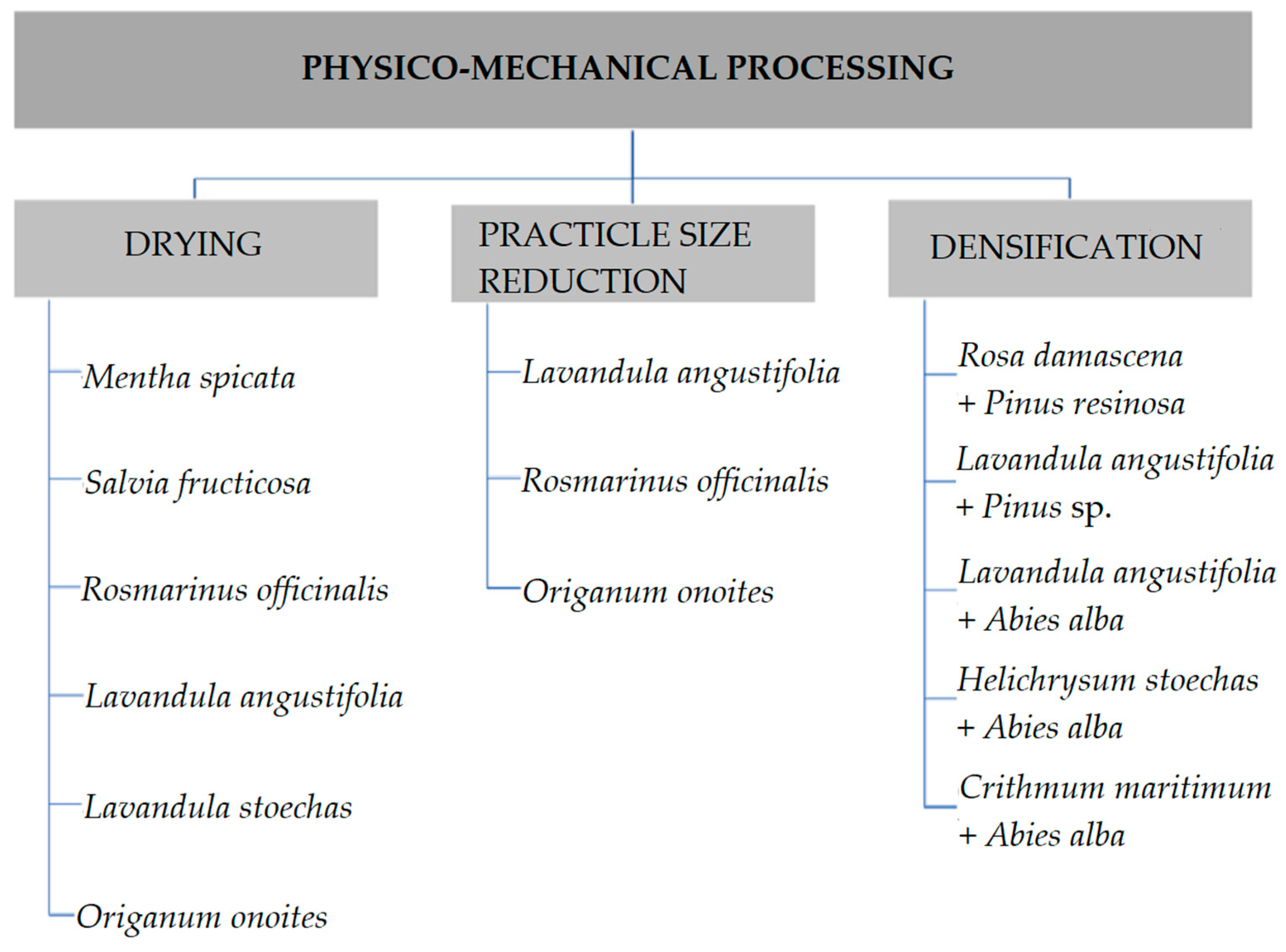
2. Physico-Mechanical Processing
2.1. Drying
2.2. Particle Size Reduction
2.3. Densification
3. Thermochemical Processing
3.1. Combustion
3.2. Pyrolysis
3.2.1. Biochar
3.2.2. Bio-Oil
3.2.3. Syngas
3.3. Hydrothermal Carbonization
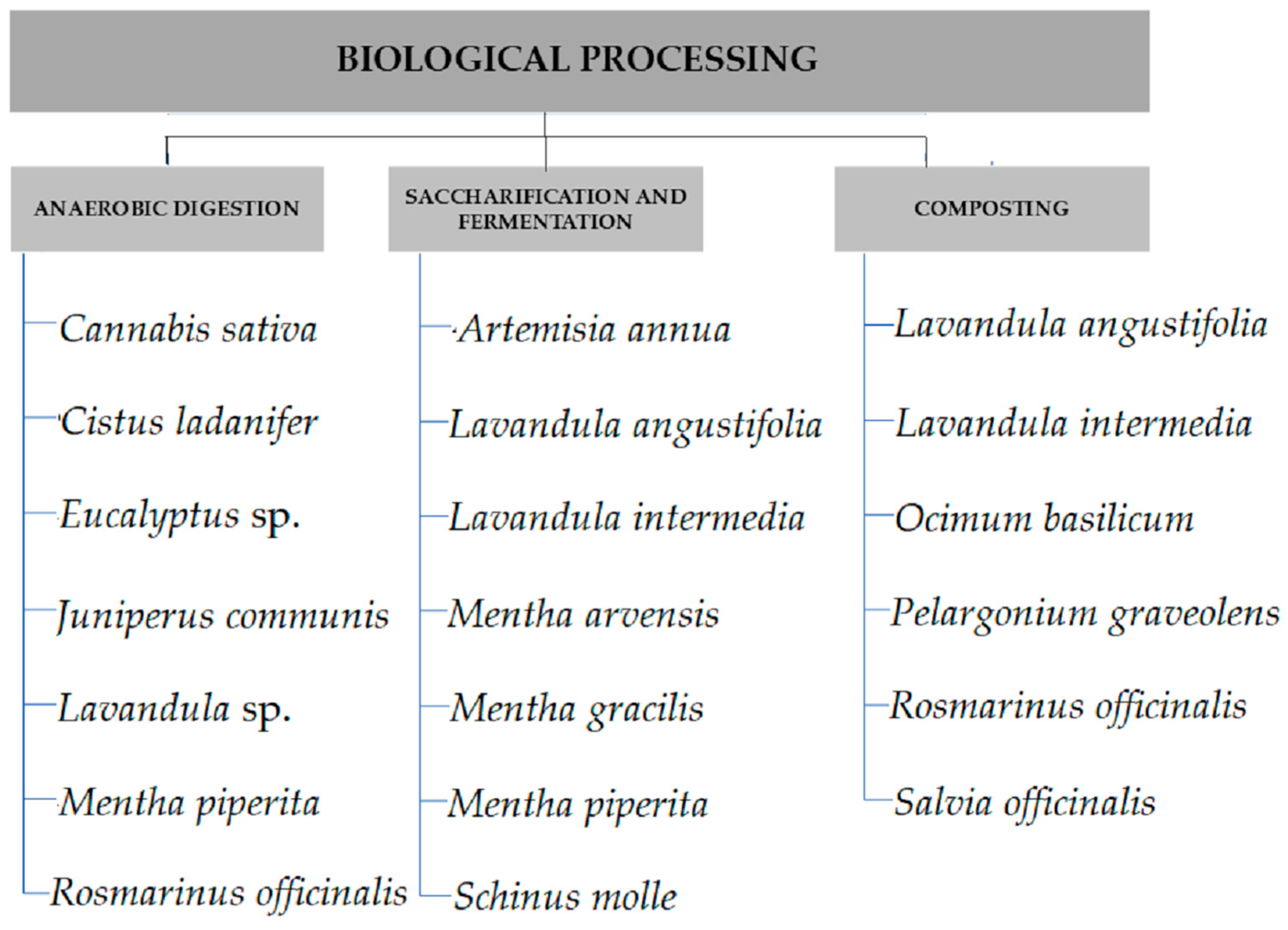
4. Biological Processing
4.1. Anaerobic Digestion
4.2. Saccharification and Fermentation
4.3. Composting
5. Chemical Processing
5.1. Extraction of Phenolics and Flavonoids
5.2. Extraction of Polysaccharides
5.3. Extraction of Other Bioactive Compounds
6. Practical Application
6.1. Energy Source
6.2. Soil Amendments
6.3. Growing Medium Production
6.4. Mulching
6.5. Building Material Production
6.6. Waste Water Treatment
6.7. Food and Pharmaceuticals
7. Disadvantages and Limitations
8. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Aćimović, M. Essential oils: Inhalation aromatherapy—A comprehensive review. J. Agron. Technol. Eng. Manag. 2021, 4, 547–557. [Google Scholar]
- Zhou, W.; Li, J.; Wang, X.; Liu, L.; Li, Y.; Song, R.; Zhang, M.; Li, X. Research progress on extraction, separation, and purification methods of plant essential oils. Separations 2023, 10, 596. [Google Scholar] [CrossRef]
- Machado, C.A.T.; Hodel, K.V.S.; Lepikson, H.A.; Machado, B.A.S. Distillation of essential oils: An innovative technological approach focused on productivity, quality and sustainability. PLoS ONE 2024, 19, e0299502. [Google Scholar] [CrossRef]
- Elguea-Culebras, G.O.; Bravo, E.M.; Sanchez-Vioque, R. Potential sources and methodologies for the recovery of phenolic compounds from distillation residues of Mediterranean aromatic plants. An approach to the valuation of by-products of the essential oil market—A review. Ind. Crops Prod. 2022, 175, 114261. [Google Scholar] [CrossRef]
- Erceg, T.; Šovljanski, O.; Tomić, A.; Aćimović, M.; Stupar, A.; Baloš, S. Comparison of the properties of pullulan-based active edible coatings implemented for improving sliced cheese shelf life. Polymers 2024, 16, 178. [Google Scholar] [CrossRef] [PubMed]
- Dessie, W.; Luo, X.; He, F.; Liao, Y.; Duns, G.J.; Qin, Z. Lignin valorization: A crucial step towards full utilization of biomass, zero waste and circular bioeconomy. Biocatal. Agric. Biotechnol. 2023, 51, 102777. [Google Scholar] [CrossRef]
- Mujtaba, M.; Fraceto, L.F.; Fazeli, M.; Mukherjee, S.; Savassa, S.M.; de Medeiros, G.A.; Pereira, A.E.S.; Mancini, S.D.; Lipponen, J.; Vilaplana, F. Lignocellulosic biomass from agricultural waste to the circular economy: A review with focus on biofuels, biocomposites and bioplastics. J. Clean. Prod. 2023, 402, 136815. [Google Scholar] [CrossRef]
- Lesage-Meessen, L.; Bou, M.; Sigoillot, J.C.; Faulds, C.B.; Lomascolo, A. Essential oils and distilled straw of lavender and lavandin: A review of current use and potential application in white biotechnology. Appl. Microbiol. Biotechnol. 2015, 99, 3375–3385. [Google Scholar] [CrossRef]
- Saha, A.; Basak, B.B. Scope of value addition and utilization of residual biomass from medicinal and aromatic plants. Ind. Crops Prod. 2020, 145, 111979. [Google Scholar] [CrossRef]
- Alexandri, M.; Christaki, S.; Gkatzionis, K.; Mourtzinos, I. Residual biomass from major aromatic and medicinal flora of the Mediterranean: Challenges towards sustainable integration into food systems within the circular bioeconomy. Trends Food Sci. Technol. 2023, 139, 104123. [Google Scholar] [CrossRef]
- Balkrishna, A.; Srivastava, S.; Srivastava, D.; Sharma, N.; Arya, V.; Gautam, A.K. Unleashing the potential of medicinal and aromatic plant wastes with particular consideration of vermicomposting: A comprehensive review of literature. J. Appl. Res. Med. Arom. Plants 2024, 39, 100527. [Google Scholar] [CrossRef]
- Khiri, S.; Ullah, N.; Boubal, Z.; Janati, W.; Amalich, S.; Lagaz, H.; Jacobsone, M.G.; Elmaaiden, E. Transforming essential oil extraction wastes: Sustainable valorization approaches for agricultural, industrial, and cosmetic application. Biomass Bioenergy 2025, 201, 108109. [Google Scholar] [CrossRef]
- Zakarya, A.Y.; Rasheed, D.M.; Farag, M.A. Valorization of aromatic plant distillation by-products (Solid biomass, wastewater, and aromatic water): Case study on the Lamiaceae family. Waste Biomass Valor. 2025. [Google Scholar] [CrossRef]
- Zotti, M.; Cecchi, G.; Canonica, L.; Di Piazza, S. A review of nature-based solutions for valorizing aromatic plants’ lignocellulosic waste through oyster mushroom cultivation. Sustainability 2025, 17, 4410. [Google Scholar] [CrossRef]
- Kolev, N.; Ivanova, M.; Balabanov, A.; Vlahova-Vangelova, D.; Kišova, A.; Vizzarri, F. Utilization of agro-industrial residues from the Rosa damascena Mill. oil industry: A literature review on biomass potential for food and feed ingredients. Processes 2025, 13, 1945. [Google Scholar] [CrossRef]
- Prakash, O.; Naik, M.; Katyar, R.; Naik, S.; Kumar, D.; Maji, D.; Shukla, A.; Nannaware, A.D.; Kalra, A.; Rout, P.K. Novel process for isolation of major bio-polymers from Mentha arvensis distilled biomass and saccharification of the isolated cellulose to glucose. Ind. Crops Prod. 2018, 119, 1–8. [Google Scholar] [CrossRef]
- Singh, M.; Pandey, N.; Negi, P.; Jyoti; Larroche, C.; Mishra, B.B. Solvothermal conversion of spent aromatic waste to ethyl glucosides. Chemosphere 2022, 292, 133428. [Google Scholar] [CrossRef] [PubMed]
- Tanger, P.; Field, J.L.; Jahn, C.E.; Defoort, M.W.; Leach, J.E. Biomass for thermochemical conversion: Targets and challenges. Front. Plant. Sci. 2013, 4, 218. [Google Scholar] [CrossRef]
- Kumar, B.; Mendoza-Martinez, C.; Ferenczi, T.; Nagy, G.; Koós, T.; Szamosi, Z. An experimental study to investigate the impact of solar drying on emission products of woody biomass in the torrefaction process. Energy Ecol. Environ. 2025, 10, 307–323. [Google Scholar] [CrossRef]
- Vidal, B.C., Jr.; Dien, B.S.; Ting, K.C.; Singh, V. Influence of feedstock particle size on lignocellulose conversion—A review. Appl. Biochem. Biotechnol. 2011, 164, 1405–1421. [Google Scholar] [CrossRef]
- Williams, C.L.; Westover, T.L.; Emerson, R.M.; Tumuluru, J.S.; Li, C. Sources of biomass feedstock variability and the potential impact on biofuels production. BioEnergy Res. 2016, 9, 1–14. [Google Scholar] [CrossRef]
- Christaki, S.; Bouloumpasi, E.; Lalidou, E.; Chatzopoulou, P.; Irakli, M. Bioactive profile of distilled solid by-products of rosemary, greek sage and spearmint as affected by distillation methods. Molecules 2022, 27, 9058. [Google Scholar] [CrossRef]
- Yılmaz Çinçin, R.G.; Öngen, A.; Ağdağ, O.N. Essential oil distillation residue as environmentally friendly feedstock in gasification: Effect of dry air flow rate and temperature on gasification performance. Biomass Conv. Bioref. 2025, 15, 11719–11733. [Google Scholar] [CrossRef]
- Vareltzis, P.; Fotiou, D.; Papatheologou, V.; Kyroglou, S.; Tsachouridou, E.; Goula, A.M. Optimized solid–liquid separation of phenolics from lavender waste and properties of the dried extracts. Separations 2024, 11, 67. [Google Scholar] [CrossRef]
- Psarrou, I.; Oreopoulou, A.; Tsimogiannis, D.; Oreopoulou, V. Extraction kinetics of phenolic antioxidants from the hydro distillation residues of rosemary and effect of pretreatment and extraction parameters. Molecules 2020, 25, 4520. [Google Scholar] [CrossRef]
- Elalami, D.; Carrère, H.; Abdelouahdi, K.; Oukarroum, A.; Dhiba, D.; Arji, M.; Barakat, A. Combination of dry milling and separation processes with anaerobic digestion of olive mill solid waste: Methane production and energy efficiency. Molecules 2018, 23, 3295. [Google Scholar] [CrossRef]
- Onur, M.E.; Ekinci, K.; Civan, M.; Bilgili, M.E.; Yurdakul, S. Quality properties and torrecaction characteristics of pellets: Rose oil distillation solid waste and red pine sawdust. Sustainability 2023, 15, 10971. [Google Scholar] [CrossRef]
- Cetinkaya, B.; Erkent, S.; Ekinci, K.; Civan, M.; Bilgili, M.E.; Yurdakul, S. Effect of torrefaction on fuel properties of biopellets. Heliyon 2024, 10, e23989. [Google Scholar] [CrossRef]
- Anestis, P.; Vasiliki, K. Utilization of aromatic plants residual biomass after distillation mixed with wood in solid biofuels production. Renew. Energy 2025, 248, 123198. [Google Scholar] [CrossRef]
- Kamperidou, V.; Terzopoulou, P. Co-pelletization of lavender waste and pine-wood for sustainable fuel pellet production. Forests 2025, 16, 1455. [Google Scholar] [CrossRef]
- Xin, S.; Huang, F.; Liu, X.; Mi, T.; Xu, Q. Torrefaction of herbal medicine wastes: Characterization of the physiocochemical properties and combustion behaviors. Bioresur. Technol. 2019, 287, 121408. [Google Scholar] [CrossRef]
- Marcelino, S.; Gaspar, P.D.; Paço, A. Sustainable waste management in the production of medicinal and aromatic plants—A systematic review. Sustainability 2023, 15, 13333. [Google Scholar] [CrossRef]
- Roig-Madrid, P.; Carmona-Cabello, M.; Perea-Moreno, A.-J.; Dorado, M.P.; David Munoz-Rodriguez, D. Thermochemical and elemental characterization of aromatic seed residues for solid biofuel applications in a circular economy context. Energy Nexus 2025, 20, 100594. [Google Scholar] [CrossRef]
- Vilas-Boas, A.C.M.; Tarelho, L.A.C.; Oliveira, H.S.M.; Silva, F.G.C.S.; Pio, D.T.; Matosa, M.A.A. Valorisation of residual biomass by pyrolysis: Influence of process conditions on products. Sustain. Energy Fuels 2024, 8, 379–396. [Google Scholar] [CrossRef]
- Gholizadeh, M.; Cristina Castro, C.; Fabrega, S.M.; Clarens, F. A review on thermochemical based biorefinery catalyst development progress. Sustain. Energy Fuels 2023, 7, 4758–4804. [Google Scholar] [CrossRef]
- Liu, W.J.; Yu, H.Q. Thermochemical conversion of lignocellulosic biomass into mass-producible fuels: Emerging technology progress and environmental sustainability evaluation. ACS Environ. Au 2021, 2, 98–114. [Google Scholar] [CrossRef] [PubMed]
- Durak, H. Comprehensive Assessment of thermochemical processes for sustainable waste management and resource recovery. Processes 2023, 11, 2092. [Google Scholar] [CrossRef]
- Berge, N.D.; Li, L.; Flora, J.R.; Ro, K.S. Assessing the environmental impact of energy production from hydrochar generated via hydrothermal carbonization of food wastes. Waste Manag. 2015, 43, 203–217. [Google Scholar] [CrossRef]
- Owsianiak, M.; Fozer, D.; Chrzanowski, Ł.; Renz, M.; Nowacki, B.; Ryberg, M. Evaluation of hydrothermal carbonization of biomass residues for bioenergy: A life-cycle based comparison against incumbent technologies. EFB Bioecon. J. 2024, 4, 100069. [Google Scholar] [CrossRef]
- Chakyrova, D.; Doseva, N. Analysis of the energy recovery posibilities of energy from lavender straws after a steam distillation process. In Proceedings of the International Scientific Conference of Communications, Information, Electronic and Energy Systems (CIEES 2020), Borovets, Bulgaria, 26–29 November 2020. [Google Scholar] [CrossRef]
- Alvaro, A.G.; Mediavilla, I.; Palomar, C.R.; Esteban, L.S.; Crespo, I.G. Energy valorization of solid residue from steam distillation of aromatic shrubs. Ind. Crops Prod. 2024, 222, 119485. [Google Scholar] [CrossRef]
- Biney, M.A.; Gusiatin, M.Z.; Trakal, L.; Mosko, J.; Pohorelý, M. Utilization of co-substrates in municipal sewage sludge co-pyrolysis: Yields and characterization of biochar, bio-oil, and syngas, with economic feasibility analysis. J. Anal. Appl. Pyrolysis. 2025, 189, 107091. [Google Scholar] [CrossRef]
- Garcia-Nunez, J.A.; Pelaez-Samaniego, M.R.; Garcia-Perez, M.E.; Fonts, I.; Abrego, J.; Westerhof, R.J.M.; Garcia-Perez, M. Historical developments of pyrolysis reactors: A review. Energy Fuels 2017, 31, 5751–5775. [Google Scholar] [CrossRef]
- Safdari, M.S.; Amini, E.; Weise, D.R.; Fletcher, T.H. Heating rate and temperature effects on pyrolysis products from live wildland fuels. Fuel 2019, 242, 295–304. [Google Scholar] [CrossRef]
- Varkolu, M.; Gundekari, S.; Omvesh; Palla, V.C.S.; Kumar, P.; Bhattacharjee, S.; Vinodkumar, T. Recent advances in biochar production, characterization, and environmental applications. Catalysts 2025, 15, 243. [Google Scholar] [CrossRef]
- Saha, A.; Basak, B.B.; Banerjee, A. In-vitro antioxidant evaluation and production of biochar from distillation waste biomass of Mentha arvensis. J. Appl. Res. Med. Arom. Plants 2022, 31, 100428. [Google Scholar] [CrossRef]
- Yadav, V.; Khare, P.; Deshmukh, Y.; Shanker, K.; Nigam, N.; Karak, T. Performance of biochar derived from Cymbopogon winterianus waste at two temperatures on soil properties and growth of Bacopa manneri. Commun. Soil Sci. Plant Anal. 2018, 49, 2741–2764. [Google Scholar] [CrossRef]
- Sarkar, R.; Basak, B.B.; Banerjee, A.; Kumar, S. Recycling of distillation by-products of Ocimum sp. as potential source of antioxidants as well as feedstock for biochar. J. Mater. Cycles Waste Manag. 2023, 25, 2171–2186. [Google Scholar] [CrossRef]
- Dira, A.; Elmouwahidi, A.; Khouja, S.; Boufetacha, M.; Bailon-Garcia, E.; Barakat, A.; Tayibi, S.; Carrasco-Marin, F.; Gharibi, E. Feedstock type and pyrolysis temperature of rosemary wastes in fixed-bed reactor affect the characteristics and application potentials of the bio-char. J. Anal. Appl. Pyrolysis 2024, 182, 106697. [Google Scholar] [CrossRef]
- Blackwell, P.; Joseph, S.; Munroe, P.; Anawar, H.M.; Storer, P.; Gilkes, R.J.; Solaiman, Z.M. Influences of biochar and biochar-mineral complex on mycorrhizal colonisation and nutrition of wheat and sorghum. Pedosphere 2015, 25, 686–695. [Google Scholar] [CrossRef]
- Basak, B.B.; Saha, A.; Sarkar, B.; Kumar, B.P.; Gajbhiye, N.A.; Banerjee, A. Repurposing distillation waste biomass and low-value mineral resources through biochar-mineral-complex for sustainable production of high-value medicinal plants and soil quality improvement. Sci. Total Environ. 2021, 760, 143319. [Google Scholar] [CrossRef]
- Modak, S.; Katiyar, P.; Yadav, S.; Hans, A. Valorization of lignocellulosic waste obtained from essential oil industry for bio-oil production and dye-removal. Water Pract. Technol. 2023, 18, 2384. [Google Scholar] [CrossRef]
- Lachos-Perez, D.; Martins-Vieira, J.C.; Missau, J.; Anshu, K.; Siakpebru, O.K.; Thengane, S.K.; Morais, A.R.C.; Tanabe, E.H.; Bertuol, D.A. Review on biomass pyrolysis with a focus on bio-oil upgrading techniques. Analytica 2023, 4, 182–205. [Google Scholar] [CrossRef]
- Deshmukh, Y.; Yadav, V.; Nigam, N.; Yadav, A.; Khare, P. Quality of bio-oil by pyrolysis of distilled spent of Cymbopogon flexuous. J. Anal. Appl. Pyrolysis 2015, 115, 43–50. [Google Scholar] [CrossRef]
- Khosravani, H.; Meshksar, M.; Rahimpour, H.R.; Rahimpour, M.R. Introduction to syngas products and applications. In Advances in Synthesis Gas: Methods, Technologies and Applications; Rahimpour, M.R., Makarem, M.A., Meshksar, M., Eds.; Elsevier: Amsterdam, The Netherland, 2023; Volume 3, pp. 3–25. [Google Scholar] [CrossRef]
- Fiore, M.; Magi, V.; Viggiano, A. Internal combustion engines powered by syngas: A review. Appl. Energy 2020, 276, 115415. [Google Scholar] [CrossRef]
- Dhairiyasamy, R.; Dixit, S.; Varshney, D.; Gabiriel, D. Renewable syngas and biodiesel dual fuel applications for enhanced engine performance and emission control. Ind. Crops Prod. 2025, 225, 120509. [Google Scholar] [CrossRef]
- Wang, X.; Duo, J.; Jin, Z.; Yang, F.; Lai, T.; Collins, E. Effects of hydrothermal carbonization conditions on the characteristics of hydrochar and its application as a soil amendment: A review. Agronomy 2025, 15, 327. [Google Scholar] [CrossRef]
- Luo, Y.; Mi, T.; Huang, F.; Liu, Y.; Liu, Q.; Xin, S.; Liu, X. Hydrothermal carbonization of herbal medicine waste: Process parameters optimization, secondary char formation and its effect on hydrochar properties. J. Environ. Manag. 2025, 379, 124819. [Google Scholar] [CrossRef]
- Singh, M.; Padey, N.; Dwivedi, P.; Kumar, V.; Mishra, B.B. Production of xylose, levulinicacis, and lignin from spent aromatic biomass with a recyclable Bronsted acid synthesized from d-limonene as a renewable feedstock from citrus waste. Bioresour. Technol. 2019, 293, 122105. [Google Scholar] [CrossRef]
- Ischia, G.; Fiori, L. Hydrothermal carbonization of organic waste and biomass: A review on process, reactor, and plant modeling. Waste Biomass Valor. 2021, 12, 2797–2824. [Google Scholar] [CrossRef]
- Satira, A.; Paone, E.; Bressi, V.; Iannazzo, D.; Marra, F.; Calabrò, P.S.; Mauriello, F.; Espro, C. Hydrothermal carbonization as sustainable process for the complete upgrading of orange peel waste into value-added chemicals and bio-carbon materials. Appl. Sci. 2021, 11, 10983. [Google Scholar] [CrossRef]
- Li, X.; Baran, S.S.; Orange, F.; Bonjour, E.; Jame, P.; Verger-Dubois, G.; Mija, A.; Lomenech, C.; Kuzhir, P.; Hurel, C. Conversion of Lavandula straw into high-quality solid fuel: Effect of hydrothermal carbonization conditions on fuel characteristics. Bioenerg. Res. 2023, 16, 1156–1172. [Google Scholar] [CrossRef]
- Ouadrhiri, F.E.; Saleh, E.A.M.; Husain, K.; Adachi, A.; Hmamou, A.; Hassan, I.; Moharam, M.M.; Lahkimi, A. Acid assisted-hydrothermal carbonization of solid waste from essential oils industry: Optimization using I-optimal experimental design and removal dye application. Arabian J. Chem. 2023, 16, 104872. [Google Scholar] [CrossRef]
- Sayara, T.; Sánchez, A. A review on anaerobic digestion of lignocellulosic wastes: Pretreatments and operational conditions. Appl. Sci. 2019, 9, 4655. [Google Scholar] [CrossRef]
- Tsegaye, B.; Balomajumder, C.; Roy, P. Microbial delignification and hydrolysis of lignocellulosic biomass to enhance biofuel production: An overview and future prospect. Bull. Natl. Res. Cent. 2019, 43, 51. [Google Scholar] [CrossRef]
- Lohri, C.R.; Diener, S.; Zabaleta, I.; Mertenat, A.; Zurbrügg, C. Treatment technologies for urban solid biowaste to create value products: A review with focus on low- and middle-income settings. Rev. Environ. Sci. Biotechnol. 2017, 16, 81–130. [Google Scholar] [CrossRef]
- Fu, Z.; Liu, F.; Li, P.; Zhang, Y.; Chen, J. Converting cellulose to biogas with anaerobic digestion at incubation temperatures of 5 °C difference in the effort for efficiency and low carbon emission. Ind. Crops Prod. 2025, 224, 120354. [Google Scholar] [CrossRef]
- Ni, J.Q. A review of household and industrial anaerobic digestion in Asia: Biogas development and safety incidents. Renew. Sust. Energ. Rev. 2024, 197, 114371. [Google Scholar] [CrossRef]
- Awadalla, O.A.; Atawy, W.A.; Bedaiwy, M.Y.; Ali, S.S.; Mahmoud, Y.A.G. Anaerobic digestion of lignocellulosic waste for enhanced methane production and biogas-digestate utilization. Ind. Crops Prod. 2023, 195, 116420. [Google Scholar] [CrossRef]
- Hjouji, K.; Haldhar, R.; Alobaid, A.A.; Taleb, M.; Rais, Z. Maximizing resource recovery: Anaerobic digestion of residual biomass from essential oil extraction in four aromatic and medicinal plants. Ind. Crops Prod. 2024, 216, 118820. [Google Scholar] [CrossRef]
- Kontodimos, I.; Papadelis, C.E.; Margaritis, N.; Grammelis, P. Valorization of medicinal plants residues through anaerobic digestion. Waste Biomass Valor. 2025, 16, 119–129. [Google Scholar] [CrossRef]
- Carmo-Calado, L.; Mota-Panizio, R.; Assis, A.C.; Nobre, C.; Alves, O.; Lourinho, G.; Brito, P. Pre-feasibility study of multy-product biorefinery for the production of essential oils and biomethane. C 2023, 9, 2. [Google Scholar] [CrossRef]
- Nhat, P.V.H.; Ngo, H.H.; Guo, W.S.; Chang, S.W.; Nguyen, D.D.; Nguyen, P.D.; Bui, X.T.; Zhang, X.B.; Guo, J.B. Can algae-based technologies be an affordable green process for biofuel production and wastewater remediation? Bioresour. Technol. 2018, 256, 491–501. [Google Scholar] [CrossRef] [PubMed]
- Gandam, P.K.; Chinta, M.L.; Pabbathi, N.P.P.; Baadhe, R.R.; Sharma, M.; Thakur, V.K.; Sharma, G.D.; Ranjitha, J.; Gupta, V.K. Second-generation bioethanol production from corncob—A comprehensive review on pretreatment and bioconversion strategies, including techno-economic and lifecycle perspective. Ind. Crops Prod. 2022, 186, 115245. [Google Scholar] [CrossRef]
- Brown, J.; Lindstrom, J.K.; Ghosh, A.; Rollag, S.A.; Brown, R.C. Production of sugars from lignocellulosic biomass via biochemical and thermochemical routes. Front. Energy Res. 2024, 12, 1347373. [Google Scholar] [CrossRef]
- Lesage-Meessen, L.; Bou, M.; Ginies, C.; Chevert, D.; Navarro, D.; Drula, E.; Bonnin, E.; del Rio, J.C.; Odinot, E.; Bisotto, A.; et al. Lavender- and lavandin-distilled straws: An untapped feedstock with great potential for the production of high-added value compounds and fungal enzymes. Biotechnol. Biofuels 2018, 11, 217. [Google Scholar] [CrossRef]
- Zheljazkov, V.D.; Stewart, C.N., Jr.; Joyce, B.; Baxter, H.; Cantrell, C.L.; Astatkie, T.; Jeliazkova, E.A.; Poovaiah, C.R. Dual utilization of medicinal and aromatic crops as bioenergy feedstocks. J. Agric. Food Chem. 2018, 66, 8744–8752. [Google Scholar] [CrossRef]
- Solis, J.L.; Davila, R.; Sandoval, C.; Guzman, D.; Guzman, H.; Alejo, L.; Kiros, Y. Ethanol production from Schinus molle essential oil extraction residues. Waste Biomass Valor. 2020, 11, 4053–4065. [Google Scholar] [CrossRef]
- Greff, B.; Szigeti, J.; Varga, Á.; Lakatos, E.; Sáhó, A.; Varga, L. Effect of bacterial inoculation on co-composting of lavender (Lavandula angustifolia Mill.) waste and cattle manure. 3 Biotech 2021, 11, 306. [Google Scholar] [CrossRef]
- González-Moreno, M.Á.; García Gracianteparaluceta, B.; Marcelino Sádaba, S.; Prieto Cobo, E.; Seco Meneses, A. Vermicomposting of lavender waste: A biological laboratory investigation. Agronomy 2022, 12, 2957. [Google Scholar] [CrossRef]
- Zaccardelli, M.; Roscigno, G.; Pane, C.; Celano, G.; Di Matteo, M.; Mainente, M.; Vuotto, A.; Mencherini, T.; Esposito, T.; Vitti, A.; et al. Essential oils and quality composts sourced by recycling vegetable residues from the aromatic plant supply chain. Ind. Crops Prod. 2021, 162, 113255. [Google Scholar] [CrossRef]
- Verma, R.K.; Verma, R.S.; Rahman, L.U.; Yadav, A.; Patra, D.D.; Kalra, A. Utilization of distillation waste-based vermicompost and other organic and inorganic fertilizers on improving production potential in geranium and soil health. Commun. Soil Sci. Plant Anal. 2014, 45, 141–152. [Google Scholar] [CrossRef]
- Khiri, S.; El-Mrabet, A.; Boubal, Z.; Ullah, N.; Ullah, A.; Janati, W.; Gueddari, L.E.; Chafik, A.; Lgaz, H.; Amalich, S.; et al. Valorization of residual biomass from essential oil extraction of Ammodaucus leucotrichus: A step towards sustainable cosmetic and pharmaceutical applications. Biomass Bioenergy 2025, 197, 107820. [Google Scholar] [CrossRef]
- Slavov, A.; Ognyanov, M.; Vasileva, I. Pectic polysaccharides extracted from pot marigold (Calendula officinalis) industrial waste. Food Hydrocoll. 2020, 101, 105545. [Google Scholar] [CrossRef]
- Fiorito, D.; Tessaro, D.; Sangalli, F.; Nobbio, C.; Nebuloni, M.; Vezzini, M.; Brenna, E.; Parmeggiani, F. Valorization of the industrial hemp residue from essential oil production by recovery of cannabidiol and chemo-enzimatic conversion to cannabielsoin. Green Chem. 2024, 26, 5211. [Google Scholar] [CrossRef]
- Dudziec, P.; Warminski, K.; Stolarski, M.J. Industrial hemp solid hydro-distillation residue-properties and diversity. Ind. Crops Prod. 2025, 235, 121800. [Google Scholar] [CrossRef]
- Sarkar, R.; Basak, B.B.; Banerjee, A.; Mossaffa, E.; Bhowmik, A. Recycling of residual biomass of medicinal and aromatic plants: A valuable alternative towards bio-refinery. J. Appl. Res. Med. Arom. Plants 2025, 45, 100624. [Google Scholar] [CrossRef]
- Truzzi, E.; Chaouch, M.A.; Rossi, G.; Tagliazucchi, L.; Bertelli, D.; Benvenuti, S. Characterization and valorization of the agricultural waste obtained from Lavandula steam distillation for its reuse in the food and pharmaceutical fields. Molecules 2022, 27, 1613. [Google Scholar] [CrossRef]
- Poveda, J.; Vítores, D.; Sánchez-Gómez, T.; Santamaría, Ó.; Velasco, P.; Zunzunegui, I.; Rodríguez, V.M.; Herrero, B.; Martín-García, J. Use of by-products from the industrial distillation of lavandin (Lavandula × intermedia) essential oil as effective bioherbicides. J. Environ. Manag. 2025, 373, 123723. [Google Scholar] [CrossRef] [PubMed]
- Marovska, G.; Vasileva, I.; Petkova, N.; Ognyanov, M.; Gandova, V.; Stoyanova, A.; Merdzhanov, P.; Simitchiev, A.; Slavov, A. Lavender (Lavandula angustifolia Mill.) industrial by-products as a source of polysaccharides. Ind. Crops Prod. 2022, 188, 115678. [Google Scholar] [CrossRef]
- Pérez-Magarino, S.; Bueno-Herrera, M.; Asensio-S.-Manzanera, M.C. Characterization of bioactive phenolic compounds extracted from hydro-distillation by-products of Spanish Lamiaceae plants. Molecules 2024, 29, 5285. [Google Scholar] [CrossRef]
- Slavov, A.; Yantcheva, N.; Vasileva, I. Chamomile wastes (Matricaria chamomilla): New source of polysaccharides. Waste Biomass Valor. 2019, 10, 2583–2594. [Google Scholar] [CrossRef]
- Bouloumpasi, E.; Hatzikamari, M.; Christaki, S.; Lazaridou, A.; Chatzopoulou, P.; Biliaderis, C.G.; Irakli, M. Assessment of antioxidant and antibacterial potential of phenolic extracts from post-distillation solid residues of oregano, rosemary, sage, lemon balm, and spearmint. Processes 2024, 12, 140. [Google Scholar] [CrossRef]
- Gavarić, N.; Radovanović, K.; Kladar, N.; Hitl, M.; Srđenović Čonić, B.; Mijatović Jovin, V.; Samojlik, I. Can we use Melissa officinalis (lemon balm) postdistillation waste extracts in pharmacy? In vivo pharmacodynamic studies. S. Afr. J. Bot. 2024, 172, 396–406. [Google Scholar] [CrossRef]
- Skendi, A.; Bouloumpasi, E.; Chatzopoulou, P.; Biliaderis, C.C.; Irakli, M. Comparison of drying methods for the retention of phenolic antioxidants in post-distillation solid residues of aromatic plants. LWT 2023, 189, 115463. [Google Scholar] [CrossRef]
- Anmol; Aggarwal, G.; Shivani; Gupta, M.K.; Sharma, M.; Sharma, U. Natural deep euteric solvents-based concurent approach for qualitative and quantitative enhancement of Rosa damascena essential oil and recovery of phenolics from distilled rose petals. Sep. Purif. Technol. 2025, 354, 128699. [Google Scholar] [CrossRef]
- Nilofar; Dall’Acqua, S.; Sut, S.; Baskose, I.; Kargılı, U.; Orlando, G.; Zengin, G. Exploring the effects of post-distillation and post-supercritical CO2 extraction on chemical profile and biological activities of two Salvia species (S. chrysophylla and S. microstegia). Microchem. J. 2024, 198, 110183. [Google Scholar] [CrossRef]
- Sanchez-Vioque, R.; Izquierdo-Melero, M.E.; Quilez, M.; Herraiz-Penalver, D.; Santana-Meridas, O.; Jordan, M.J. Solid residues from distillarion of Salvia lavandulifolia Vahl. as a natural source of antioxidant compounds. J. Am. Oil Chem. Soc. 2018, 95, 1277–1284. [Google Scholar] [CrossRef]
- Gavarić, N.; Kladar, N.; Mišan, A.; Nikolić, A.; Samojlik, I.; Mimica-Dukić, N.; Božin, B. Postdistillation waste material of thyme (Thymus vulgaris L., Lamiaceae) as a potential source of biologically active compounds. Ind. Crops Prod. 2015, 74, 457–464. [Google Scholar] [CrossRef]
- Hostetler, G.L.; Riedl, K.M.; Schwartz, S.J. Effects of food formulation and thermal processing on flavones in celery and chamomile. Food Chem. 2013, 141, 1406–1411. [Google Scholar] [CrossRef]
- Tzima, K.; Brunton, N.P.; Rai, D.K. Qualitative and quantitative analysis of polyphenols in Lamiaceae plants—A review. Plants 2018, 7, 25. [Google Scholar] [CrossRef]
- Nouska, C.; Irakli, M.; Georgiou, M.; Lytou, A.E.; Skendi, A.; Bouloumpasi, E.; Chatzopoulou, P.; Biliaderis, C.G.; Lazaridou, A. Physicochemical characteristics, antioxidant properties, aroma profile, and sensory qualities of value-added wheat breads fortified with post-distillation solid wastes of aromatic plants. Foods 2023, 12, 4007. [Google Scholar] [CrossRef]
- Benalaya, I.; Alves, G.; Lopes, J.; Silva, L.R. A review of natural polysaccharides: Sources, characteristics, properties, food, and pharmaceutical applications. Int. J. Mol. Sci. 2024, 25, 1322. [Google Scholar] [CrossRef]
- Tang, J.; Li, X.; Xu, D.; Liu, G.; Zhang, X.; Xiong, X.; Yang, X.; Qin, X.; Deng, Y.; Hou, C.; et al. Physicochemical, functional, and antioxidant properties of pectic polysaccharides extracted from three bast fibrous plants. Life 2025, 15, 1618. [Google Scholar] [CrossRef]
- Navarrete, A.; Herrero, M.; Martín, A.; Cocero, M.J.; Ibáñez, E. Valorization of solid wastes from essential oil industry. J. Food Eng. 2011, 104, 196–201. [Google Scholar] [CrossRef]
- Almeida, H.H.; Crugeira, P.J.; Santamaria-Echart, A.; Amaral, J.S.; Finimundy, T.C.; Barros, L.; Rodrigues, A.E.; Barreiro, M.F. Circular valorisation of essential oil post-distillation by-products for enhanced xanthan gum bioproduction and antimicrobial treatments against Staphylococcus aureus. Waste Biomass Valor. 2025. [Google Scholar] [CrossRef]
- Agustina, S.E.; Kholifah, L.S. Prospect of Essential Oil Industrial Waste as Energy Resources for Essential Oil Production Process. In Proceedings of the 4th International Conference on Agricultural Technology, Engineering, and Environmental Sciences (ICATES-2022), Banda Aceh, Indonesia, 9–10 August 2022. [Google Scholar] [CrossRef]
- Jensch, C.; Strube, J. Proposal of a new green process for waste valorization and cascade utilization of essential oil plants. Sustainability 2022, 14, 3227. [Google Scholar] [CrossRef]
- Yadav, D.; Yadav, A.; Singh, M.; Khare, P. Cultivation of aromatic plant for nature-based sustainable solutions for the management of degraded/marginal lands: Techno-economics and carbon dynamic. Carbon Res. 2023, 2, 27. [Google Scholar] [CrossRef]
- Karagianni, A.-G.; Paraschou, A.; Matsi, T. A preliminary evaluation of the use of solid residues from the distillation of medicinal and aromatic plants as fertilizers in mediterranean soils. Agronomy 2025, 15, 1903. [Google Scholar] [CrossRef]
- Malahi, S.E.; Irahoui, L.; Mokhtari, W.; Ennami, M.M.; Taimourya, H.; Zim, J.; Zayani, A.; Zakri, B.; Dhassi, K.; Mokhtari, M.; et al. Exploring the synergistic effect of rose distillation waste and biostimulant beside other organic amendments on Rosa damascena seedlings’ growth. Int. J. Rec. Org. Waste Agric. 2024, 13, 132416. [Google Scholar]
- Chrysargyris, A.; Louka, S.; Petropoilos, S.A.; Tzortzakis, N. Soilless cultivation of Portulaca oleracea using medicinal and aromatic plant residues for partial peat replacement. Horticulturae 2023, 9, 474. [Google Scholar] [CrossRef]
- Chrysargyris, A.; Tzortzakis, N. Residues from medicinal and aromatic plants after fistillation can be used in replace some peat in the growing media for Viola × wittrockiana production. Agronomy 2024, 14, 187. [Google Scholar] [CrossRef]
- Angelova, G.; Brazkova, M.; Stefanova, P.; Blazheva, D.; Vladev, V.; Petkova, N.; Slavov, A.; Denev, P.; Karashanova, D.; Zaharieva, R.; et al. Waste rose flower and lavender straw biomass—An innovative lignocellulose feedstock for mycelium bio-materials development using newly isolated Ganoderma resinaceum GA1M. J. Fungi 2021, 7, 866. [Google Scholar] [CrossRef] [PubMed]
- Singh, A.; Singh, K.; Singh, V. Sustainability of organic mulch (distillation waste) and herbicides for weed management in perenial aromatic grasses. Trop. Pest Manag. 1991, 37, 162–165. [Google Scholar] [CrossRef]
- Singh, A.; Singh, M.; Singh, S. Effective utilization of distillation waste as organic mulch for weed management in the aromatic grass, Citronella java. Int. J. Pest Manag. 2001, 47, 253–257. [Google Scholar] [CrossRef]
- Ratiarisoa, R.V.; Magniont, C.; Ginestet, S.; Oms, C.; Escadeillas, G. Assessment of distilled lavender stalks as bioaggregate for building materials: Hygrothermal properties, mechanical performance and chemical interactions with mineral pozzolanic binder. Constr. Build. Mater. 2016, 124, 801–816. [Google Scholar] [CrossRef]
- Strzałkowski, J.; Kampragkou, P.; Stefanidou, M.; Markowska-Szczupak, A.; Horszczaruk, E.; Głowacka, A. Lavender and black pine waste as additives enhancing selected mechanical and hygrothermal properties of cement mortars. Materials 2024, 17, 5475. [Google Scholar] [CrossRef]
- Feisther, V.A.; Scherer Filho, J.; Hackbarth, F.V.; Mayer, D.A.; de Souza, A.A.U.; de Souza, S.M.A.G.U. Raw leaves and leaf residues from the extraction of essential oils as biosorbents for metal removal. J. Environ. Chem. Eng. 2019, 7, 103047. [Google Scholar] [CrossRef]
- Ivanova, L.; Vassileva, P.; Detcheva, A.; Koleva, V.; Avramova, I. Plant-based biosorbents for copper(II) removal: A comparative study of biomass and essential oil residues. Appl. Sci. 2025, 15, 7695. [Google Scholar] [CrossRef]
- Hanif, A.; Bhatti, H.N.; Hanif, M.A. Removal and recovery of Cu(II) and Zn(II) using immobilized Mentha arvensis distillation waste biomass. Ecol. Eng. 2009, 35, 1427–1434. [Google Scholar] [CrossRef]
- Iftikhar, A.R.; Bhatti, H.N.; Hanif, M.A.; Nadeem, R. Kinetic and thermodynamic aspects of Cu(II) and Cr(III) removal from aqueous solutions using rose waste biomass. J. Hazard. Mater. 2009, 161, 941–947. [Google Scholar] [CrossRef]
- Riaz, B.; Ansari, T.M.; Hanif, M.A.; Riaz, S.; Khan, M.A.; Riaz, M.; Jilani, M.I. Utilization of extensively available environmental waste Mentha spicata for uptake of Pb(II) from aqueous solutions. Asian J. Chem. 2013, 25, 4551–4555. [Google Scholar] [CrossRef]
- Tanasa, A.; Puitel, A.C.; Zaharia, C.; Suteu, D. Sorption of reactive dyes from aqueous media using the lavender waste as biosorbent. Desalination Water Treat. 2021, 236, 348–358. [Google Scholar] [CrossRef]
- Tadesse, A.W.; Huang, M.; Zhou, T. Biochar for wastewater treatment: Preparation, modification, characterization, and its applications. Molecules 2025, 30, 4288. [Google Scholar] [CrossRef] [PubMed]
- Rawat, A.P.; Kumar, V.; Singh, D.P. A combined effect of adsorption and reduction potential of biochar derived from Mentha plant waste on removal of methylene blue dye from aqueous solution. Separation Sci. Technol. 2019, 55, 907–921. [Google Scholar] [CrossRef]
- Solomakou, N.; Fotiou, D.; Tsachouridou, E.; Goula, A.M. Valorization of waste from lavender distillation through optimized encapsulation processes. Foods 2025, 14, 2684. [Google Scholar] [CrossRef] [PubMed]
- Ahamefule, C.S.; Osilo, C.; Ahamefule, B.C.; Madueke, S.N.; Moneke, A.N. Simultaneous production of biofuel from agricultural wastes and bioremediation of the waste substrates: A review. Curr. Res. Microb. Sci. 2024, 7, 100305. [Google Scholar] [CrossRef]
- Yameen, M.Z.; Naqvi, S.R.; Juchelková, D.; Khan, M.N.A. Harnessing the power of functionalized biochar: Progress, challenges, and future perspectives in energy, water treatment, and environmental sustainability. Biochar 2024, 6, 25. [Google Scholar] [CrossRef]
- Motola, V.; Scarlat, N.; Buffi, M.; Hurtig, O.; Rejtharova, J.; Georgakaki, A.; Mountraki, A.; Letout, S.; Salvucci, R.; Rózsai, M.; et al. Clean Energy Technology Observatory: Bioenergy in the European Union—2024 Status Report on Technology Development, Trends, Value Chains and Markets; Publications Office of the European Union: Luxembourg, 2024. [CrossRef]
- Alcocer-García, H.; Sánchez-Ramírez, E.; García-García, E.; Ramírez-Márquez, C.; Ponce-Ortega, J.M. Unlocking the potential of biomass resources: A review on sustainable process design and intensification. Resources 2025, 14, 143. [Google Scholar] [CrossRef]
- Ahmed, I.; Zia, M.A.; Afzal, H.; Ahmed, S.; Ahmad, M.; Akram, Z.; Sher, F.; Iqbal, H.M.N. Socio-economic and environmental impacts of biomass valorisation: A strategic drive for sustainable bioeconomy. Sustainability 2021, 13, 4200. [Google Scholar] [CrossRef]
- Yazdani, M.; Chatterjee, P.; Montero-Simo, M.J.; Araque-Padilla, R.A. An Integrated multi-attribute model for evaluation of sustainable mobile phone. Sustainability 2019, 11, 3704. [Google Scholar] [CrossRef]
- Gavrilas, S.; Rat, M.; Munteanu, F.-D. Biowaste valorisation and its possible perspectives within sustainable food chain development. Processes 2025, 13, 2085. [Google Scholar] [CrossRef]
- Roszkowska, S.; Szubska-Włodarczyk, N. What are the barriers to agricultural biomass market development? The case of Poland. Environ. Syst. Decis. 2022, 42, 75–84. [Google Scholar] [CrossRef]

| Process | Typical Conditions | Main Products & Yields (Typical) | Energy Parameters | Reference |
|---|---|---|---|---|
| Combustion | >800 °C | Solid fuel for direct heat | HHV 15–20 MJ/kg HHV 13.55–20.31 MJ/kg | [32,33] |
| Biochar production | Dominantly as in pyrolysis | Biochar (carbon-rich char) 25–50% | HHV 25–34 MJ/kg | [34] |
| Bio-oil production | Fast pyrolysis | Bio-oil 60–75% | HHV 15–32 MJ/kg | [35] |
| Syngas production (via gasification) | 700–1000 °C | CO, H2, CH4 | Syngas for heat/electricity (energy content process dependent) | [32] |
| Oxygen Requirement | Community | Time Scale | Main Product | Reference | |
|---|---|---|---|---|---|
| Anaerobic digestion | Anaerobic | Anaerobic bacteria | Weeks to months | Biogas (CH4 and CO2), digestate | [65] |
| Saccharification and fermentation | Anaerobic—fermentation, limited oxygen—saccharification (for enzymatic activity) | Yeast (Saccharomyces), bacteria | Hours to days | Ethanol, organic acids, or other biochemicals | [66] |
| Composting | Aerobic | Aerobic bacteria and fungi, earthworms | Weeks to months | Compost (humus-like material) | [67] |
| Plant Species | Plant Part | Essential Oil Distillation Technique | Method for Recovery of Bioactive Compounds from Post-Distillation Waste | Extraction Solvent | Obtained Bioactive Compounds | Reference |
|---|---|---|---|---|---|---|
| Ammodaucus leucotrichus | Leaves and stems | HD, SD | Microwave-assisted extraction | Water | Flavonoids | [84] |
| Calendula officinalis | Flowers | HD | Pretreatment (ethanol, filtration, drying) | HCl | Polysaccharides | [85] |
| Cannabis sativa | Apical parts | SD | Dynamic maceration, Soxhlet extraction | Methanol, ethanol, n-heptane, chloroform | Cannabidiols | [86] |
| Cannabis sativa | Apical parts | HD | Maceration | Water | Cellulose, hemicellulose, lignin | [87] |
| Cymbopogon flexuosus | Leaves | HD | Pretreatment (NaOH, ethanol, filtration, drying) | HCl | Cellulose, hemicellulose, lignin | [88] |
| Ultrasound-assisted extraction | hexane, chloroform, ethyl acetate, acetone, methanol, water | Phenolics, flavonoids | ||||
| Cymbopogon martini | Leaves | HD | Pretreatment (NaOH, ethanol, filtration, drying) | HCl | Cellulose, hemicellulose, lignin | [88] |
| Ultrasound-assisted extraction | hexane, chloroform, ethyl acetate, acetone, methanol, water | Phenolics, flavonoids | ||||
| Cymbopogon winterianus | Leaves | HD | Pretreatment (NaOH, ethanol, filtration, drying) | HCl | Cellulose, hemicellulose, lignin | [88] |
| Ultrasound-assisted extraction | hexane, chloroform, ethyl acetate, acetone, methanol, water | Phenolics, flavonoids | ||||
| Lavandula × intermedia | Aerial parts | SD | Dynamic maceration | Ethanol, methanol, water, ethyl acetate | Phenolics, flavonoids | [89] |
| Lavandula × intermedia | Aerial parts | SD | Ultrasound-assisted extraction | Methanol | Phospholipids, Phenolics | [90] |
| Lavandula angustifolia | Aerial parts | SD | Dynamic maceration | Ethanol, methanol, water, ethyl acetate | Phenolics, flavonoids | [89] |
| Lavandula angustifolia | Stems and flowers | SD | Stirring-assisted extraction | HCl | Polysaccharides | [91] |
| Lavandula sp. | Aerial parts | HD | Orbital shaker extraction | Methanol | Phenolics, flavonoids | [92] |
| Matricaria chamomilla | Flowers | SD, HD | Pretreatment (ethanol, filtration, drying) | HCl | Polysaccharides | [93] |
| Melissa officinalis | Aerial parts | SD | Ultrasound-assisted extraction | Ethanol | Phenolics, flavonoids | [94] |
| Melissa officinalis | Leaves | HD | Maceration | Ethanol | Phenolics, flavonoids | [95] |
| Mentha arvensis | Leaves | HD | Pretreatment (NaOH, ethanol, filtration, drying) | HCl | Cellulose, hemicellulose, lignin | [88] |
| Ultrasound-assisted extraction | hexane, chloroform, ethyl acetate, acetone, methanol, water | Phenolics, flavonoids | ||||
| Mentha arvensis | Aerial parts | HD | Soxhlet extraction | Methanol, ethanol, acetone, water | Phenolics, flavonoids | [46] |
| Mentha spicata | Aerial parts | SD | Ultrasound-assisted extraction | Ethanol | Phenolics, flavonoids | [94] |
| Ocimum basilicum | Leaves | HD | Pretreatment (NaOH, ethanol, filtration, drying) | HCl | Cellulose, hemicellulose, lignin | [88] |
| Ultrasound-assisted extraction | hexane, chloroform, ethyl acetate, acetone, methanol, water | Phenolics, flavonoids | ||||
| Ocimum basilicum | Leaves | HD | Ultrasound-assisted extraction | Hexane, dichloromethane, acetone, ethyl acetate, methanol, water | Phenolics, flavonoids | [48] |
| Ocimum sanctum | Leaves | HD | Pretreatment (NaOH, ethanol, filtration, drying) | HCl | Cellulose, hemicellulose, lignin | [88] |
| Ultrasound-assisted extraction | hexane, chloroform, ethyl acetate, acetone, methanol, water | Phenolics, flavonoids | ||||
| Ocimum sanctum | Leaves | HD | Ultrasound-assisted extraction | Hexane, dichloromethane, acetone, ethyl acetate, methanol, water | Phenolics, flavonoids | [48] |
| Origanum vulgare | Aerial parts | SD | Ultrasound-assisted extraction | Ethanol | Phenolics, flavonoids | [94] |
| Origanum vulgare | Aerial parts | SD | Ultrasound-assisted extraction | Methanol | Phenolics | [96] |
| Rosa damascena | Petals | HD | Ultrasound-assisted extraction | Methanol | Phenolics | [97] |
| Rosmarinus officinalis | Aerial parts | SD | Ultrasound-assisted extraction | Ethanol | Phenolics, flavonoids | [94] |
| Rosmarinus officinalis | Aerial parts | SD | Ultrasound-assisted extraction | Methanol | Phenolics | [96] |
| Rosmarinus officinalis | Aerial parts | HD | Orbital shaker extraction | Methanol | Phenolics, flavonoids | [92] |
| Salvia chrysophylla | Aerial parts | HD | Maceration | Methanol | Phenolics, flavonoids | [98] |
| Salvia fructicosa | Aerial parts | SD | Ultrasound-assisted extraction | Ethanol | Phenolics, flavonoids | [94] |
| Salvia fructicosa | Aerial parts | SD | Ultrasound-assisted extraction | Methanol | Phenolics | [96] |
| Salvia lavandulifolia | Aerial parts | HD | Soxhlet | Ethanol | Phenolics, flavonoids | [99] |
| Salvia lavandulifolia | Aerial parts | HD | Orbital shaker extraction | Methanol | Phenolics, flavonoids | [92] |
| Salvia microstegia | Aerial parts | HD | Maceration | Methanol | Phenolics, flavonoids | [98] |
| Saturejathymbra | Aerial parts | SD | Ultrasound-assisted extraction | Methanol | Phenolics | [96] |
| Thymus mastichina | Aerial parts | HD | Orbital shaker extraction | Methanol | Phenolics, flavonoids | [92] |
| Thymus vulgaris | Leaves | HD | Maceration | Ethanol | Phenolics, flavonoids | [100] |
| Process | TLR Range | Description | Reference |
|---|---|---|---|
| Drying | 9 | Commercially mature | – |
| Particle size reduction (grinding/milling) | 9 | Commercially mature; Standard industrial pretreatment for biomass | – |
| Densification (pellets/briquettes) | 9 | Commercially used for biomass solid fuels | – |
| Combustion | 9 | Fully mature | – |
| Pyrolysis | 7–9 | Mature for wood, but variable for novel biomass resources | [35] |
| Biochar production (via pyrolysis) | 7–9 | Commercially mature; Plants are growing globally | [130] |
| Bio-oil production (via pyrolysis) | 6–8 | Pilot and demo scale for biomass bio-oil | [35] |
| Syngas production (gasification) | 7–8 | Commercial pilots | [35] |
| Hydrothermal carbonization (HTC) | 6–9 | Commercial; Technology spreading to wet biomass | [130] |
| Anaerobic digestion | 9 | Commercially mature | [131] |
| Saccharification + fermentation | 5–8 | For lignocellulosic substrates in pilot to early commercial stages | [132] |
| Composting | 9 | Commercially mature | – |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Aćimović, M.; Leovac Maćerak, A.; Pavlić, B.; Sikora, V.; Zeremski, T.; Erceg, T.; Djatkov, D. Green Innovation for Solid Post-Distillation Residues Valorization: Narrative Review of Circular Bio-Economy Solutions. Processes 2026, 14, 244. https://doi.org/10.3390/pr14020244
Aćimović M, Leovac Maćerak A, Pavlić B, Sikora V, Zeremski T, Erceg T, Djatkov D. Green Innovation for Solid Post-Distillation Residues Valorization: Narrative Review of Circular Bio-Economy Solutions. Processes. 2026; 14(2):244. https://doi.org/10.3390/pr14020244
Chicago/Turabian StyleAćimović, Milica, Anita Leovac Maćerak, Branimir Pavlić, Vladimir Sikora, Tijana Zeremski, Tamara Erceg, and Djordje Djatkov. 2026. "Green Innovation for Solid Post-Distillation Residues Valorization: Narrative Review of Circular Bio-Economy Solutions" Processes 14, no. 2: 244. https://doi.org/10.3390/pr14020244
APA StyleAćimović, M., Leovac Maćerak, A., Pavlić, B., Sikora, V., Zeremski, T., Erceg, T., & Djatkov, D. (2026). Green Innovation for Solid Post-Distillation Residues Valorization: Narrative Review of Circular Bio-Economy Solutions. Processes, 14(2), 244. https://doi.org/10.3390/pr14020244









