Sorghum Grain: From a Simple Cereal to Food Applications and Health Benefits
Abstract
1. Introduction
2. Research Methodology
3. Main Findings
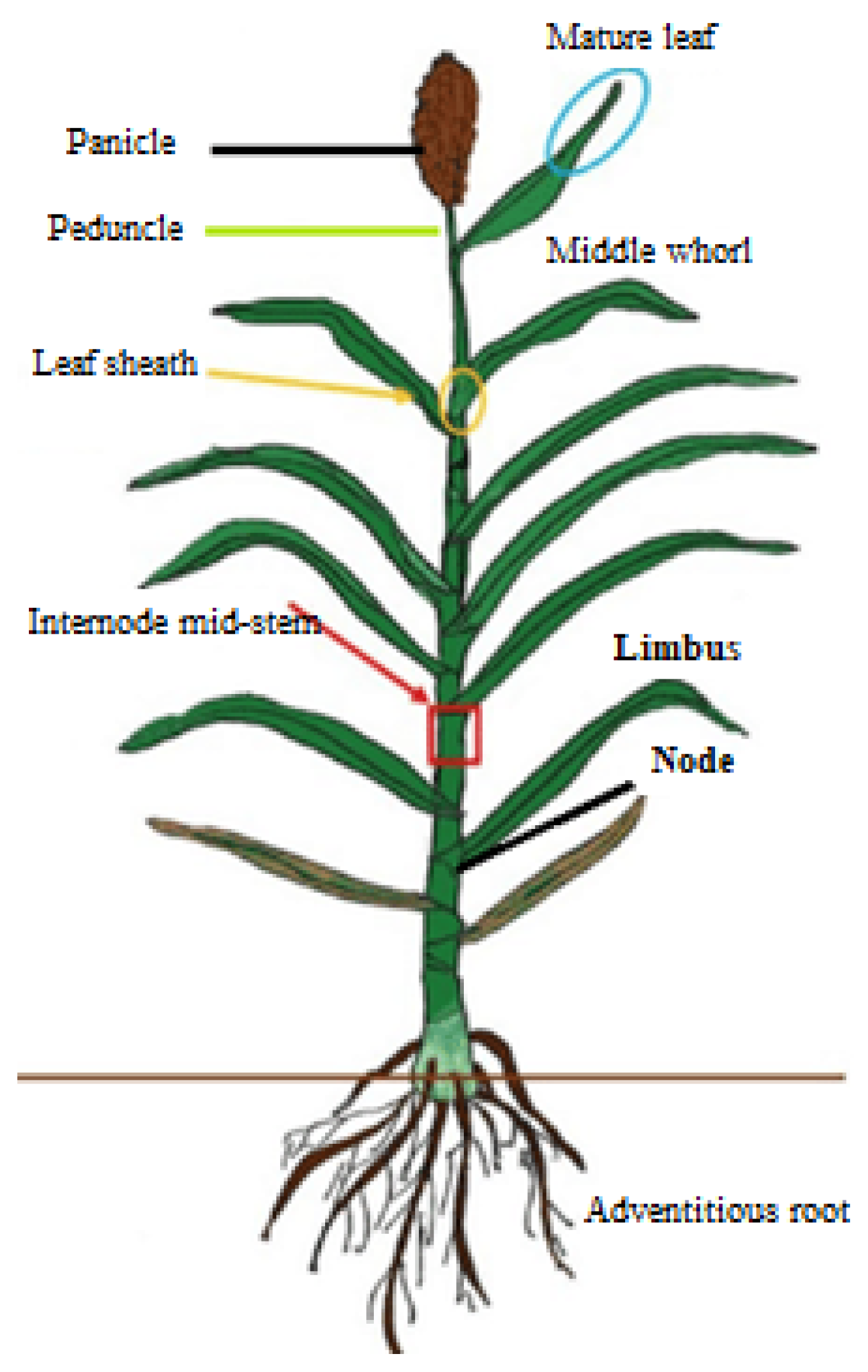
3.1. Botanical Description
3.2. Sorghum Composition
3.2.1. Sorghum Chemical and Nutritional Composition
3.2.2. Phytochemical Composition of Sorghum
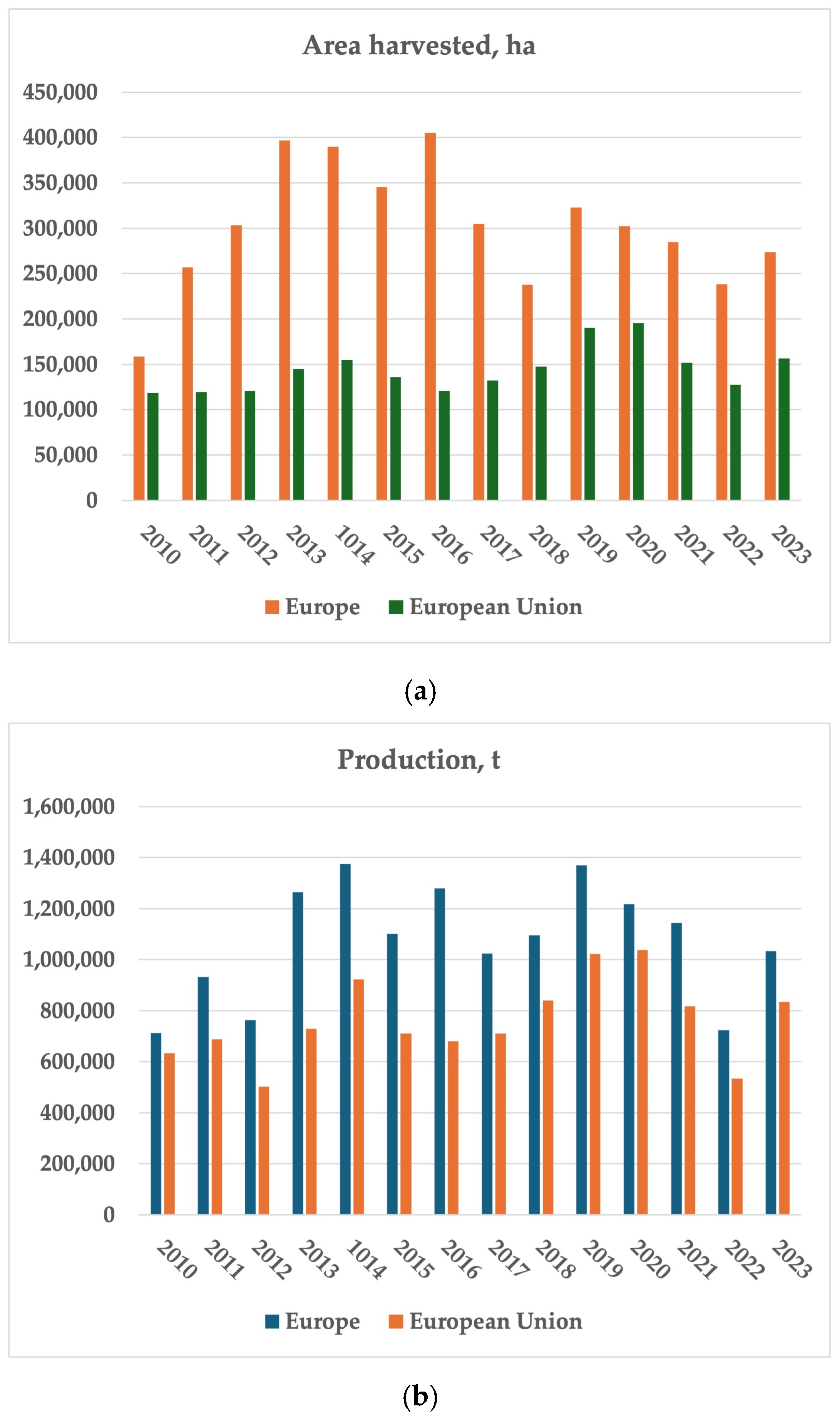
3.3. Global Production
3.4. Manufacturing Potential
3.4.1. Sorghum Valorization: Possibilities and Challenges
3.4.2. Sorghum Processing into Flour and Flour Derivates
3.4.3. Sorghum-Breakfast-Cereal
3.4.4. Sorghum Syrup
3.4.5. Sorghum in the Production of Nonalcoholic Fermented Beverages or Alcoholic Ones
3.4.6. Sorghum Snacks and Pops
3.4.7. Future Research Directions for Sorghum
3.5. Health Benefits
3.5.1. Antioxidant Activity
3.5.2. Anticancer Potential
3.5.3. Antidiabetic Activity
3.5.4. Anti-Obesity Effects
4. Conclusions and Outlook
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Winchell, F.; Stevens, C.J.; Murphy, C.; Champion, L.; Fuller, D.Q. Evidence for Sorghum Domestication in Fourth Millennium BC Eastern Sudan: Spikelet Morphology from Ceramic Impressions of the Butana Group. Curr. Anthropol. 2017, 58, 673–683. [Google Scholar] [CrossRef]
- Khalifa, M.; Eltahir, E.A.B. Assessment of Global Sorghum Production, Tolerance, and Climate Risk. Front. Sustain. Food Syst. 2023, 7, 1184373. [Google Scholar] [CrossRef]
- George, T.T.; Obilana, A.O.; Oyenihi, A.B.; Obilana, A.B.; Akamo, D.O.; Awika, J.M. Trends and Progress in Sorghum Research over Two Decades, and Implications for Global Food Security. S. Afr. J. Bot. 2022, 151, 960–969. [Google Scholar] [CrossRef]
- Mwamahonje, A.; Mdindikasi, Z.; Mchau, D.; Mwenda, E.; Sanga, D.; Garcia-Oliveira, A.L.; Ojiewo, C.O. Advances in Sorghum Improvement for Climate Resilience in the Global Arid and Semi-Arid Tropics: A Review. Agronomy 2024, 14, 3025. [Google Scholar] [CrossRef]
- Stoicea, P.; Iorga, A.M.; David, L.; Bucur, C.G. Sorghum, the Crop of the Future under Climate Change Conditions. Sci. Pap. Ser. Manag. Econ. Eng. Agric. Rural Dev. 2025, 25, 789–803. [Google Scholar]
- Hossain, M.S.; Islam, M.N.; Rahman, M.M.; Mostofa, M.G.; Khan, M.A.R. Sorghum: A Prospective Crop for Climatic Vulnerability, Food and Nutritional Security. J. Agric. Food Res. 2022, 8, 100300. [Google Scholar] [CrossRef]
- Bakari, H.; Djomdi; Ruben, Z.F.; Roger, D.D.; Cedric, D.; Guillaume, P.; Pascal, D.; Philippe, M.; Gwendoline, C. Sorghum (Sorghum bicolor L. Moench) and Its Main Parts (By-Products) as Promising Sustainable Sources of Value-Added Ingredients. Waste Biomass Valor. 2023, 14, 1023–1044. [Google Scholar] [CrossRef]
- Rumler, R.; Bender, D.; Schönlechner, R. Sorghum and Its Potential for the Western Diet. J. Cereal Sci. 2022, 104, 103425. [Google Scholar] [CrossRef]
- Pontieri, P.; Troisi, J.; Calcagnile, M.; Bean, S.R.; Tilley, M.; Aramouni, F.; Boffa, A.; Pepe, G.; Campiglia, P.; Del Giudice, F.; et al. Chemical Composition, Fatty Acid and Mineral Content of Food-Grade White, Red and Black Sorghum Varieties Grown in the Mediterranean Environment. Foods 2022, 11, 436. [Google Scholar] [CrossRef] [PubMed]
- Haziman, M.L.; Ishaq, M.I.; Qonit, M.A.H.; Lestari, E.G.; Susilawati, P.N.; Widarsih, W.; Syukur, C.; Herawati, H.; Arief, R.; Santosa, B.; et al. Sorghum Starch Review: Structural Properties, Interactions with Proteins and Polyphenols, and Modification of Physicochemical Properties. Food Chem. 2025, 463, 139810. [Google Scholar] [CrossRef]
- Baah, R.O.; Duodu, K.G.; Harasym, J.; Emmambux, M.N. Nutritional and Functional Properties of Decorticated and Microwave Heat Moisture Treated White Sorghum Meal with Added Non-Tannin and Tannin Phenolic Extract. Food Chem. 2025, 475, 143261. [Google Scholar] [CrossRef]
- Jannasch, A.; Wang, Y.-J.; Lee, S.-O.; Liyanage, R.; Alraawi, Z.; Thallapuranam, S.; Rooney, W.L. Polyphenol-Induced Protein Structural Modifications in Sorghum on Pasting Properties. LWT 2024, 210, 116881. [Google Scholar] [CrossRef]
- Xu, L.; Song, X.; Yao, D.; Wang, C.; Yao, X.; Li, Z. Dynamic Migration of Phenolics in Microwaved Combined Cooked Sorghum: Focus on the Polyphenols Interact with Starch/Protein. Food Chem. X 2025, 27, 102342. [Google Scholar] [CrossRef]
- Yang, C.; Lv, P.; Han, H.; Xiao, X.; Xu, C.; Gilbert, R.G.; Li, E. Investigation of Changes in the Fine Structure of Sorghum Starch in the Baijiu Fermentation Process. Food Hydrocoll. 2024, 152, 109905. [Google Scholar] [CrossRef]
- Zhang, K.; Ke, F.; Zhou, H.; Wang, J.; Ma, Z.; Zhang, F.; Wang, Y.; Zhang, Z.; Lu, F.; Duan, Y.; et al. The Correlation of Starch Composition, Physicochemical and Structural Properties of Different Sorghum Grains. Front. Plant Sci. 2025, 16. [Google Scholar] [CrossRef]
- Kang, X.; Gao, W.; Cheng, Y.; Yu, B.; Cui, B.; Abd El-Aty, A.M. Investigating Structural and Property Modifications in Starch from Waxy, Stick, and H37 Sorghum Varieties: Advancing Starch Structure Understanding and Applications. Ind. Crops Prod. 2023, 203, 117239. [Google Scholar] [CrossRef]
- Kumar, A.A. Botany, Taxonomy and Breeding. In The Sorghum Genome; Rakshit, S., Wang, Y.-H., Eds.; Springer International Publishing: Cham, Switzerland, 2016; pp. 27–45. ISBN 978-3-319-47789-3. [Google Scholar]
- Taxon: Sorghum Bicolor. Available online: https://acir.aphis.usda.gov/s/cird-taxon/a0ut0000000rAYjAAM/sorghum-bicolor (accessed on 6 November 2025).
- Rooney, L.W.; Rooney, W.L.; Saldivar, S.O.S. Sorghum. Ref. Modul. Food Sci. 2016. [Google Scholar] [CrossRef]
- Khoddami, A.; Messina, V.; Vadabalija Venkata, K.; Farahnaky, A.; Blanchard, C.L.; Roberts, T.H. Sorghum in Foods: Functionality and Potential in Innovative Products. Crit. Rev. Food Sci. Nutr. 2023, 63, 1170–1186. [Google Scholar] [CrossRef]
- Xue, W.; Yang, S.; Liu, X.; Qian, M.; Wang, H.; Yang, H.; Liu, X.; Shen, Y.; Li, J.; Sun, Z. Enhanced Sweet Sorghum Growth and Soil Quality in Coastal Saline–Alkali Soils Through Organic Acid-Containing Bio-Based Materials and Microbial Synergy. Agronomy 2025, 15, 56. [Google Scholar] [CrossRef]
- The Sorghum Plant. Available online: https://sorghumtrust.co.za/the-sorghum-plant/ (accessed on 6 November 2025).
- Sorghum Morphology and Anatomy. Available online: https://evolution.earthathome.org/grasses/andropogoneae/sorghum-morphology/ (accessed on 6 November 2025).
- Mawouma, S.; Condurache, N.N.; Turturică, M.; Constantin, O.E.; Croitoru, C.; Rapeanu, G. Chemical Composition and Antioxidant Profile of Sorghum (Sorghumbicolor (L.) Moench) and Pearl Millet (Pennisetumglaucum (L.) R.Br.) Grains Cultivated in the Far-North Region of Cameroon. Foods 2022, 11, 2026. [Google Scholar] [CrossRef] [PubMed]
- Food Search|USDA FoodData Central. Available online: https://fdc.nal.usda.gov/food-search?query=sorghum (accessed on 6 November 2025).
- Jocelyne, R.E.; Béhiblo, K.; Ernest, A.K. Comparative Study of Nutritional Value of Wheat, Maize, Sorghum, Millet, and Fonio: Some Cereals Commonly Consumed in Côte d’Ivoire. Eur. Sci. J. 2020, 16, 118. [Google Scholar] [CrossRef]
- Taylor, J.R.; Duodu, K.G. Effects of Processing Sorghum and Millets on Their Phenolic Phytochemicals and the Implications of This to the Health-Enhancing Properties of Sorghum and Millet Food and Beverage Products. J. Sci. Food Agric. 2015, 95, 225–237. [Google Scholar] [CrossRef]
- Dadnia, M.R.; Mojaddam, M.; Ayaran, A. Role of Copper on Physiological Parameters and Salt Tolerance in Sweet Sorghum (Sorghum bicolor L.) Cultivars. J. Crop Nutr. Sci. 2018, 4, 47–59. [Google Scholar]
- Moraes, É.A.; da Marineli, R.S.; Lenquiste, S.A.; Queiroz, V.A.V.; Camargo, R.L.; Borck, P.C.; Carneiro, E.M.; Maróstica Júnior, M.R. Whole Sorghum Flour Improves Glucose Tolerance, Insulin Resistance and Preserved Pancreatic Islets Function in Obesity Diet-Induced Rats. J. Funct. Food. 2018, 45, 530–540. [Google Scholar] [CrossRef]
- Pontieri, P.; Del Giudice, L. Sorghum: A Novel and Healthy Food. Int. J. Food Sci. 2016, 33–42. [Google Scholar] [CrossRef]
- Dykes, L.; Rooney, L.W. Sorghum and Millet Phenols and Antioxidants. J. Cereal Sci. 2006, 44, 236–251. [Google Scholar] [CrossRef]
- Dykes, L.; Peterson, G.C.; Rooney, W.L.; Rooney, L.W. Flavonoid Composition of Lemon-Yellow Sorghum Genotypes. Food Chem. 2011, 128, 173–179. [Google Scholar] [CrossRef]
- Davis, H.; Xiaoyu, S.; Yanting, S.; Jingwen, X.; Donghai, W.; Scott, S.; Fadi, A.; Weiqung, W. Phenotypic Diversity of Colored Phytochemicals in Sorghum Accessions with Various Pericarp Pigments. In Polyphenols in Plants; Academic Press: Cambridge, MA, USA, 2019; pp. 123–131. [Google Scholar]
- Shen, Y.; Su, X.; Rhodes, D.; Herald, T.; Xu, J.; Chen, X.; Smith, S.; Wang, W. The Pigments of Sorghum Pericarp Are Associated with the Contents of Cartenoids and Pro-Vitamin A. Int. J. Food Nutrit. Sci. 2017, 6, 48–56. [Google Scholar]
- Xu, J.; Wang, W.; Zhao, Y. Phenolic Compounds in Whole Grain Sorghum and Their Health Benefits. Foods 2021, 10, 1921. [Google Scholar] [CrossRef]
- Wallace, T.C. Anthocyanins in Cardiovascular Disease. Adv. Nutr. 2011, 2, 1–7. [Google Scholar] [CrossRef]
- Thilakarathna, R.C.N.; Madhusankha, G.D.M.P.; Navaratne, S.B. Potential Food Applications of Sorghum (Sorghum bicolor) and Rapid Screening Methods of Nutritional Traits by Spectroscopic Platforms. J. Food Sci. 2022, 87, 36–51. [Google Scholar] [CrossRef]
- FAOSTAT. Available online: https://www.fao.org/faostat/en/#data/QCL (accessed on 7 November 2025).
- Afify, A.E.-M.M.R.; El-Beltagi, H.S.; El-Salam, S.M.A.; Omran, A.A. Protein Solubility, Digestibility and Fractionation after Germination of Sorghum Varieties. PLoS ONE 2012, 7, e31154. [Google Scholar] [CrossRef]
- Palacios, C.E.; Nagai, A.; Torres, P.; Rodrigues, J.A.; Salatino, A. Contents of Tannins of Cultivars of Sorghum Cultivated in Brazil, as Determined by Four Quantification Methods. Food Chem. 2021, 337, 127970. [Google Scholar] [CrossRef]
- Wang, L.; Duan, W.; Zhou, S.; Qian, H.; Zhang, H.; Qi, X. Effects of Extrusion Conditions on the Extrusion Responses and the Quality of Brown Rice Pasta. Food Chem. 2016, 204, 320–325. [Google Scholar] [CrossRef] [PubMed]
- Palavecino, P.M.; Curti, M.I.; Bustos, M.C.; Penci, M.C.; Ribotta, P.D. Sorghum Pasta and Noodles: Technological and Nutritional Aspects. Plant Foods Hum. Nutr. 2020, 75, 326–336. [Google Scholar] [CrossRef] [PubMed]
- Rashwan, A.K.; Yones, H.A.; Karim, N.; Taha, E.M.; Chen, W. Potential Processing Technologies for Developing Sorghum-Based Food Products: An Update and Comprehensive Review. Trends Food Sci. Technol. 2021, 110, 168–182. [Google Scholar] [CrossRef]
- Chen, S.; Wang, L.; Ni, D.; Lin, L.; Wang, H.; Xu, Y. Characterization of Aroma Compounds in Cooked Sorghum Using Comprehensive Two-Dimensional Gas Chromatography-Time-of-Flight Mass Spectrometry and Gas Chromatography-Olfactometry-Mass Spectrometry. Molecules 2021, 26, 4796. [Google Scholar] [CrossRef]
- Ratnavathi, C. Sorghum Processing and Utilization. In Sorghum Biochemistry: An Industrial Perspective; Academic Press: Cambridge, MA, USA, 2016; ISBN 978-0-12-803182-7. [Google Scholar]
- Adebo, O. African Sorghum-Based Fermented Foods: Past, Current and Future Prospects. Nutrients 2020, 12, 1111. [Google Scholar] [CrossRef]
- Mazumdar, S.D.; Poshadri, A.; Srinivasa Rao, P.; Ravinder Reddy, C.; Reddy, B.V.S. Innovative Use of Sweet Sorghum Juice in the Beverage Industry. Int. Food Res. J. 2012, 19, 1361–1366. [Google Scholar]
- Nimbkar, N.; Kolekar, N.M.; Akade, J.H.; Rajvanshi, A.K. Syrup Production from Sweet Sorghum. Natl. Agric. Res. Inst. 2006, 1–10. [Google Scholar]
- da Silva, L.A.; de São José, V.P.B.; Rodrigues, L.A.; do Prado, P.V.C.; Toledo, R.C.L.; de Barros, F.A.R.; de Souza, A.M.; Antoniassi, R.; de Carvalho, C.W.P.; Queiroz, V.A.V.; et al. Effects of a Sorghum Beverage with Lacticaseibacillus paracasei on Body Composition, Lipid Profiles, and Intestinal Health in Overweight and Obese Adults: A Randomized Single-Blind Pilot Study. Foods 2024, 13, 3128. [Google Scholar] [CrossRef]
- Ajiboye, T.O.; Iliasu, G.A.; Adeleye, A.O.; Abdussalam, F.A.; Akinpelu, S.A.; Ogunbode, S.M.; Jimoh, S.O.; Oloyede, O.B. Nutritional and Antioxidant Dispositions of Sorghum/Millet-Based Beverages Indigenous to Nigeria. Food Sci. Nutr. 2014, 2, 597–604. [Google Scholar] [CrossRef]
- Nge, S.T.M.; Ballo, A. Development of Functional Beverages Based on Sorghum (Sorghum bicolor) Sap Through Controlled Fermentation. J. Pendidik. IPA Indones. 2025, 11, 793–801. [Google Scholar] [CrossRef]
- Sajjadi Alhashem, S.H.; Ehsani, M.R.; Akhondzadeh Basti, A.; Sharifan, A. Functional, Nutritional, and Sensorial Evaluation of Sorghum-Based Beverages Produced by Single- and Two-Stage Acid, α-Amylase Enzyme, and Germination Treatments. Food Sci. Nutr. 2024, 12, 8129–8136. [Google Scholar] [CrossRef]
- Bwamu, K.E.; Nkirote, K.C.; Kahiu, N.E.; Edgar, K.D. Optimization of Fermentation and Malting Process of Sorghum Beverage and Effects on Nutritional Quality. Afr. J. Food Sci. 2022, 16, 252–260. [Google Scholar] [CrossRef]
- Adebo, O.A.; Gabriela Medina-Meza, I. Impact of Fermentation on the Phenolic Compounds and Antioxidant Activity of Whole Cereal Grains: A Mini Review. Molecules 2020, 25, 927. [Google Scholar] [CrossRef] [PubMed]
- Balasubramanian, S.; Patil, R. Modern Processing Technologies for Sorghum Products. Process. Food Ind. 2011, 13–18. Available online: https://www.researchgate.net/publication/278686120_Modern_processing_technologies_for_sorghum_products (accessed on 6 November 2025).
- Belmouloud, R.; Bourekoua, H.; Chemache, L.; Mitrus, M.; Benatallah, L.; Różyło, R.; Wójtowicz, A. Application of Fermented Wheat, Acorns, and Sorghum in Processing of Couscous: Effect on Culinary Quality, Pasting Properties, and Microstructure. Appl. Sci. 2025, 15, 7418. [Google Scholar] [CrossRef]
- Bantilan, M.C.S.; Deb, U.K.; Gowda, C.L.L.; Reddy, B.V.S.; Obilana, A.B.; Evenson, R.E. Future Directions for Food Security and Diversity Partnership and Research Strategy for Sorghum. In Sorghum Genetic Enhancement: Research Process, Dissemination and Impacts; International Crops Research Institute for the Semi-Arid Tropics (ICRISAT): Patancheru, India, 2004; pp. 263–268. ISBN 978-92-9066-470-3. [Google Scholar]
- Shen, S.; Huang, R.; Li, C.; Wu, W.; Chen, H.; Shi, J.; Chen, S.; Ye, X. Phenolic Compositions and Antioxidant Activities Differ Significantly among Sorghum Grains with Different Applications. Molecules 2018, 23, 1203. [Google Scholar] [CrossRef]
- Hong, S.; Pangloli, P.; Perumal, R.; Cox, S.; Noronha, L.E.; Dia, V.P.; Smolensky, D. A Comparative Study on Phenolic Content, Antioxidant Activity and Anti-Inflammatory Capacity of Aqueous and Ethanolic Extracts of Sorghum in Lipopolysaccharide-Induced RAW 264.7 Macrophages. Antioxidants 2020, 9, 1297. [Google Scholar] [CrossRef]
- Ham, D.Y.; Seo, J.W.; Choi, H.J.; Park, J.; Kim, N.Y.; Kim, M.J.; Yu, C.Y.; Seong, E.S. Evaluation of Anti-Inflammatory Activity in Methanolic Seed Extracts of International Sorghum bicolor L. Resources. Agronomy 2024, 14, 997. [Google Scholar] [CrossRef]
- Reyna-Reyna, L.Y.; Montaño-Leyva, B.; Valencia, D.; Cinco-Moroyoqui, F.J.; González-Vega, R.I.; Bernal-Mercado, A.T.; Ballesteros-Monrreal, M.G.; Mendez-Encinas, M.A.; Del-Toro-Sánchez, C.L. Antioxidant, Antibacterial, Anti-Inflammatory, and Antiproliferative Activity of Sorghum Lignin (Sorghum bicolor) Treated with Ultrasonic Pulses. Metabolites 2023, 13, 394. [Google Scholar] [CrossRef] [PubMed]
- Seo, J.; Lee, J.; Ghimire, B.; Kim, M.; Yu, C.; Seong, E. Characterization of Antioxidant and Anti-Inflammatory Activities in the Grains of Collected Sorghum bicolor Varieties. J. Appl. Biol. Chem. 2023, 66, 227–235. [Google Scholar] [CrossRef]
- Espitia-Hernández, P.; Esparza-González, S.C.; Rodríguez-Herrera, R.; Dávila-Medina, M.D.; Chávez-González, M.L.; Ruelas-Chacón, X.; Ascacio-Valdés, J.A.; Flores-Naveda, A.; Sepúlveda, L. Evaluation of Antimicrobial and Antiproliferative Activities of Sorghum Extracts Obtained by Green Extraction Technologies and Solid-State Fermentation. Food Bioprocess. Technol. 2025. [Google Scholar] [CrossRef]
- Chen, X.; Shen, J.; Xu, J.; Herald, T.; Smolensky, D.; Perumal, R.; Wang, W. Sorghum Phenolic Compounds Are Associated with Cell Growth Inhibition through Cell Cycle Arrest and Apoptosis in Human Hepatocarcinoma and Colorectal Adenocarcinoma Cells. Foods 2021, 10, 993. [Google Scholar] [CrossRef]
- Msimango, L.D.; Ogwuegbu, M.C.; Mthiyane, D.M.N.; Onwudiwe, D.C. Biogenic α-Fe2O3 Nanoparticles from Sorghum Bicolor Leaf Extracts and Assessment of the Anticancer and Antioxidant Properties. Discov. Nano 2025, 20, 113. [Google Scholar] [CrossRef]
- Petchimuthu, P.; Kunjiappan, S.; Pandian, S.R.K.; Sankaranarayanan, M.; Sundar, K. Sorghum Grain-Derived Kafirin Nanoparticles for Effective Delivery of Corosolic Acid into Breast Cancer Cells for Potential Treatment of Breast Cancer. J. Clust. Sci. 2025, 36, 81. [Google Scholar] [CrossRef]
- Park, J.H.; Lee, S.H.; Chung, I.-M.; Park, Y. Sorghum Extract Exerts an Anti-Diabetic Effect by Improving Insulin Sensitivity via PPAR-γ in Mice Fed a High-Fat Diet. Nutr. Res. Pract. 2012, 6, 322–327. [Google Scholar] [CrossRef]
- Senevirathne, I.G.N.H.; Abeysekera, W.K.S.M.; Abeysekera, W.P.K.M.; Jayanath, N.Y.; Galbada Arachchige, S.P.; Wijewardana, D.C.M.S.I. Antiamylase, Antiglucosidase, and Antiglycation Properties of Millets and Sorghum from Sri Lanka. Evid. -Based Complement. Altern. Med. 2021, 2021, 5834915. [Google Scholar] [CrossRef] [PubMed]
- Indrianingsih, A.W.; Khasanah, Y.; Noviana, E.; Rahayu, E.; Hastuti, H.P.; Ni’maturrohmah, D.; Suryani, A.E.; Darsih, C.; Windarsih, A.; Handayani, S. The Effect of Soaking of White and Red Varieties of Sorghum bicolor Flour on Its Antioxidant, Antidiabetic and Physicochemical Properties. Food Humanit. 2023, 1, 1531–1538. [Google Scholar] [CrossRef]
- Ofosu, F.K.; Elahi, F.; Daliri, E.B.-M.; Tyagi, A.; Chen, X.Q.; Chelliah, R.; Kim, J.-H.; Han, S.-I.; Oh, D.-H. UHPLC-ESI-QTOF-MS/MS Characterization, Antioxidant and Antidiabetic Properties of Sorghum Grains. Food Chem. 2021, 337, 127788. [Google Scholar] [CrossRef] [PubMed]
- Arunraj, K.; Gokhale, J.S. Enhancing the Antidiabetic and Antioxidant Potential of Yellow Sorghum via Lactic Acid Bacteria Fermentation. J. Cereal Sci. 2025, 126, 104262. [Google Scholar] [CrossRef]
- Ofosu, F.K.; Elahi, F.; Daliri, E.B.-M.; Han, S.-I.; Oh, D.-H. Impact of Thermal Treatment and Fermentation by Lactic Acid Bacteria on Sorghum Metabolite Changes, Their Antioxidant and Antidiabetic Activities. Food Biosci. 2022, 45, 101502. [Google Scholar] [CrossRef]
- Lee, H.-S.; Santana, Á.L.; Peterson, J.; Yucel, U.; Perumal, R.; De Leon, J.; Lee, S.-H.; Smolensky, D. Anti-Adipogenic Activity of High-Phenolic Sorghum Brans in Pre-Adipocytes. Nutrients 2022, 14, 1493. [Google Scholar] [CrossRef] [PubMed]
- Aluko, O.O.; Ajayi, O.B. Investigation of Antilipidemic Efficacy of Condensed Tannins from Three Varieties of Sorghum Bicolor Seeds on High-Fat Diet-Induced Obese Rats. Comp. Clin. Pathol. 2023, 32, 847–858. [Google Scholar] [CrossRef]



| Components | Sorghum | Wheat | Maize | Millet |
|---|---|---|---|---|
| Water, g | 12.40 | 11.52 | 12.51 | 11.31 |
| Protein, g | 10.60 | 13.24 | 8.15 | 11.29 |
| Total lipids, g | 3.46 | 2.15 | 5.09 | 4.58 |
| Carbohydrates, g | 72.10 | 77.50 | 72.89 | 69.89 |
| Total dietary fibers, g | 6.70 | 3.06 | 6.69 | 3.89 |
| Ash, g | 1.43 | 1.41 | 1.70 | 2.16 |
| Calcium (Ca), mg | 13.00 | 19.02 | 9.60 | 6.99 |
| Iron (Fe), mg | 3.36 | 2.25 | 2.92 | 15.29 |
| Magnesium (Mg), mg | 165.00 | 120.75 | 120.12 | 155.23 |
| Phosphorus (P), mg | 289.00 | 240.60 | 265.16 | 250.08 |
| Potassium (K), mg | 363.00 | 111.29 | 270.12 | 264.74 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Andronoiu, D.-G.; Nistor, O.-V. Sorghum Grain: From a Simple Cereal to Food Applications and Health Benefits. Processes 2025, 13, 3958. https://doi.org/10.3390/pr13123958
Andronoiu D-G, Nistor O-V. Sorghum Grain: From a Simple Cereal to Food Applications and Health Benefits. Processes. 2025; 13(12):3958. https://doi.org/10.3390/pr13123958
Chicago/Turabian StyleAndronoiu, Doina-Georgeta, and Oana-Viorela Nistor. 2025. "Sorghum Grain: From a Simple Cereal to Food Applications and Health Benefits" Processes 13, no. 12: 3958. https://doi.org/10.3390/pr13123958
APA StyleAndronoiu, D.-G., & Nistor, O.-V. (2025). Sorghum Grain: From a Simple Cereal to Food Applications and Health Benefits. Processes, 13(12), 3958. https://doi.org/10.3390/pr13123958







