Abstract
Background/Objectives: The fluocinolone acetonide implant (FAI) is an intravitreal corticosteroid implant designed to have a therapeutic effect lasting up to 3 years. We performed a meta-analysis to investigate the efficacy and safety of the FAI (0.19 mg, releasing at 0.2 μg/day) in patients with non-infectious uveitis. Methods: The PubMed, EMBASE, and Cochrane Library databases were last searched on 6 September 2024. Studies comparing FAI with sham injections were investigated. The primary outcome was the recurrence of uveitis. Secondary outcomes included visual acuity, intraocular pressure (IOP), and occurrence of cataracts. Results: Significantly more patients in the FAI group experienced no uveitis recurrence for up to 36 months compared to the sham group, with a relatively lower number of recurrences. Systemic adjuvant therapy was similar between groups, while fewer patients required local rescue injections in the FAI group (95% confidence interval (CI): −2.91 to −1.70). Visual acuity changes and the proportion of eyes with ≥15 letters gain were not significantly different between the groups. More patients needed cataract surgery in the FAI group (95% CI: 0.68–1.96). No differences were observed in IOP change, final IOP, or treatment-requiring events related to an increased IOP. However, more subjects experienced events of IOP > 25 mmHg with the FAI (95% CI: 0.73 to 2.14). Conclusions: The 0.19 mg FAI was effective in preventing uveitis recurrence, and reduced the need for local injections. No significant impacts were noted in terms of systemic therapy, visual improvement, or most IOP-related complications.
1. Introduction
The fluocinolone acetonide implant (FAI) is designed to release submicrogram doses of corticosteroids into the vitreous cavity lasting up to 3 years [1,2]. This allows a theoretically smaller number of intravitreal injections, which can reduce the treatment burden for both patients and clinicians. The FAI is approved for the treatment of chronic diabetic macular edema (DME) that does not respond sufficiently to available therapies and for the prevention of relapse in chronic non-infectious uveitis (NIU) [1,2]. In NIU, current therapeutic strategies focus on the control of active inflammation and the prevention of recurrences [3]. Traditional therapeutic options include systemic corticosteroids and/or immunomodulatory agents, which can both induce systemic side effects. On the other hand, local treatments such as dexamethasone implants offer limited long-term efficacy, lasting up to six months [1]. Fluocinolone acetonide implants were therefore developed to provide a long-lasting local anti-inflammatory effect while avoiding systemic adverse events. Avoiding cumulative damage caused by recurrent episodes of inflammation is crucial to reduce irreversible damage and long-term complications [4].
The FAI that releases at 0.2 μg/day includes two implants: one containing 0.19 mg (Iluvien®; Alimera Sciences, Dublin, Ireland) and the other containing 0.18 mg (Yutiq®; Alimera Sciences Inc., Alpharetta, GA, USA) [3,5]. Both implants allow a steady-state concentration of 0.5 to 1.0 ng/mL to be reached within 6 to 9 months [1]. In contrast, the FAI containing 0.59 mg (Retisert®; Bausch and Lomb, Rochester, NY, USA) is designed to release at 0.6 μg/day over the first month, and then release a decreased dose of 0.03–0.04 μg/day over approximately 30 months [6]. The 0.19 mg FAI (Iluvien®) considered in this study is a nonbiodegradable cylindrical implant measuring 3.5 mm in length and 0.37 mm in diameter. It consists of a polyvinyl alcohol permeable membrane and an internal matrix containing fluocinolone acetonide [7]. The implant can be inserted into the vitreous cavity through the inbuilt 25-gauge needle of the device.
Besides the well-known risks of cataracts and glaucoma, there are other complications that can arise with FAIs. These include ocular hypotony, choroidal detachment, vitreous hemorrhage, retinal detachment, and implant migration to the anterior chamber [8,9], as previously reported with dexamethasone implants. The risk of implant migration is higher in eyes with previous vitrectomy, an open/defective lens capsule, and/or iris defects [10]. Persistent floaters can also cause discomfort as the implant remains permanently within the vitreous cavity. Another limitation of the use of FAI is its high cost. Although it was suggested to be cost-effective in NIU patients with unilateral disease, as it reduced the number of recurrences [11,12], this was not the case in patients with bilateral involvement [13].
A meta-analysis that compared various intravitreal agents for NIU reported that no specific regimen showed a significant superiority, whereas the 0.59 mg FAI was associated with a higher risk of cataract and intraocular pressure (IOP) rise [2]. All FAIs (including the 0.19 mg and 0.59 mg dosages) were associated with a lower risk of uveitis recurrence, while no significant differences were noted in terms of visual improvement compared to standard care [2]. Various real-world studies investigating the efficacy and safety of FAI in NIU-related macular edema (ME) noted significant anatomical improvements, but controversial results were reported regarding visual acuity outcomes [14,15,16,17,18,19,20].
Relatively fewer controlled studies and meta-analyses focus on NIU compared to DME [5,21]. Currently, two randomized controlled studies (RCTs) have investigated the efficacy and safety of FAI (0.2 μg/day) in NIU for up to 36 months [22,23]. They both reported significantly less uveitis recurrence and less use of local adjuvant injections, and no differences in the use of systemic adjuvant therapy or IOP-related events were noted [22,23]. Meanwhile, despite their similar design, discrepancies exist regarding the number of uveitis recurrences and outcomes associated with visual improvement [22,23]. Despite the limited number of available studies, we considered it meaningful to conduct a meta-analysis as the two RCTs showed different results in factors related to visual improvement. Accordingly, we investigated the efficacy and safety of FAI (0.19 mg) in patients with NIU through a literature search and meta-analysis.
2. Materials and Methods
2.1. Search Methods
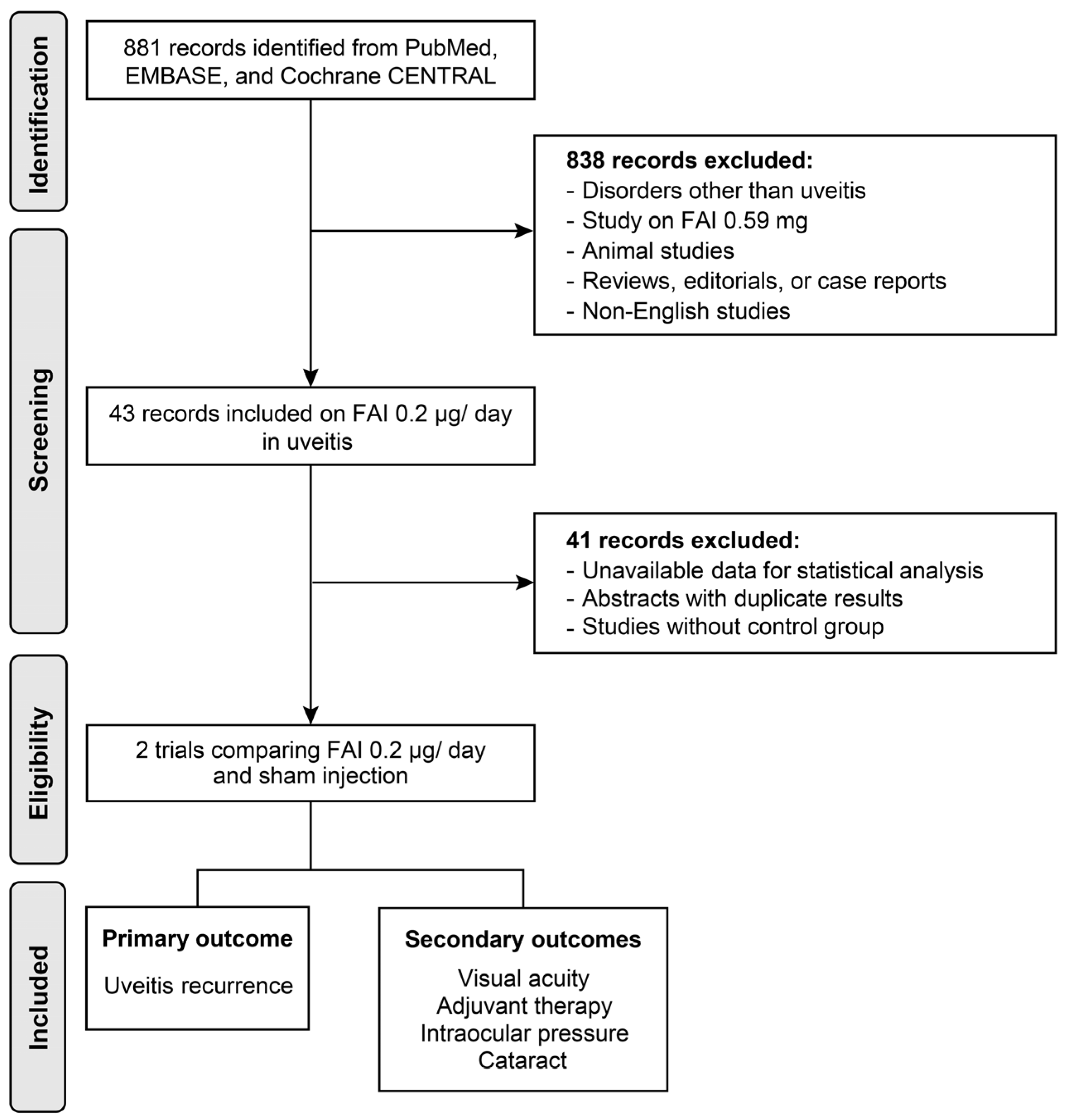
The PubMed, EMBASE, and Cochrane Library databases were last searched on 6 September 2024, using the following terms as principal keywords: ‘uveitis’, ‘inflammation’, ‘fluocinolone acetonide’, ‘intravitreal injection’, ‘implant’, ‘corticosteroid’, ‘steroid’, and ‘macular edema’ (see Supplementary File S1 for details). A total of 881 studies were identified in the preliminary search. Studies comparing FAI and sham injections were investigated for meta-analysis, and further exclusion was performed based on the following criteria: (1) studies involving disorders other than uveitis; (2) studies on 0.59 mg of FAI or other corticosteroid agents; (3) animal studies, case reports, or review articles; (4) duplicated articles (including congress abstracts that were lately published in articles); and (5) studies written in non-English languages (Figure 1). This review is registered in the International Prospective Register of Systematic Reviews (PROSPERO) under registration number CRD42025634703.
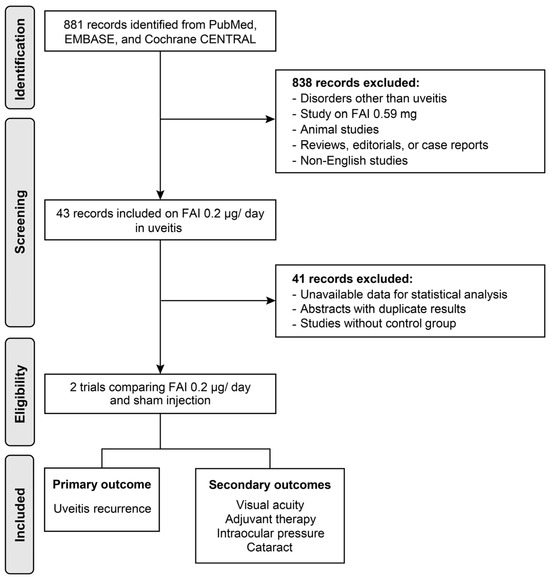
Figure 1.
Flow diagram of the study selection process. FAI: fluocinolone acetonide implant.
2.2. Assessment of Potential Bias
The risk of bias in included studies was assessed on 2 December 2024 using the study quality assessment tool developed by the National Heart, Lung, and Blood Institute (NHLBI, https://www.nhlbi.nih.gov/health-topics/study-quality-assessment-tools). Each criterion was assessed by two independent reviewers (Y.R.C. and S.T.) and then discussed for overall quality rate depending on the proportion of fulfilled criteria (poor: <50%, fair: 50–75%, and good: ≥75%).
2.3. Primary and Secondary Outcomes
The primary outcome was the recurrence of uveitis. The secondary outcomes were visual acuity measured by the Early Treatment Diabetic Retinopathy Study (ETDRS) letters, any adjuvant therapy, and complications including cataracts or increased IOP. Systemic adjuvant therapy included corticosteroid or immunosuppressive therapy, while local adjuvant therapy included intraocular or periocular corticosteroid injections.
2.4. Statistical Analysis
Data on the proportions, mean values, and standard deviations were obtained from the literature. The meta-analysis was conducted using RevMan 5.4, and heterogeneity was examined using I2 statistics. The fixed model was applied as few studies were included in this meta-analysis [24]. Squares indicate the mean difference estimates for continuous variables and odds ratio (OR) for categorical variables, and lines extending from the squares represent the associated 95% confidence intervals (CI) in the forest plots. CIs that do not intersect the vertical line at 0 for continuous variables and at 1 for categorical variables indicated statistical significance at p < 0.05.
3. Results
3.1. Results of the Literature Search
Among the records that investigated the FAI (0.19 mg) in uveitis, two RCTs comparing FAI and sham injections were identified and used for the meta-analysis [22,23]. The characteristics and brief results of these RCTs and other excluded studies without a control group are summarized in Table 1. Briefly, most of the included studies of retrospective non-comparative design revealed the effective control of inflammation and manageable IOP during the post-implantation period. In terms of visual acuity, some of these studies reported stable vision [25,26], whereas other case series reported visual improvement with FAI.

Table 1.
Summary of studies investigating the efficacy and safety of FAI.
3.2. Quality Assessment
The risk of bias, assessed using the NHLBI tool, is presented in Supplementary Table S1. Overall, the selected RCTs demonstrated good quality for inclusion in the meta-analysis.
3.3. Outcomes
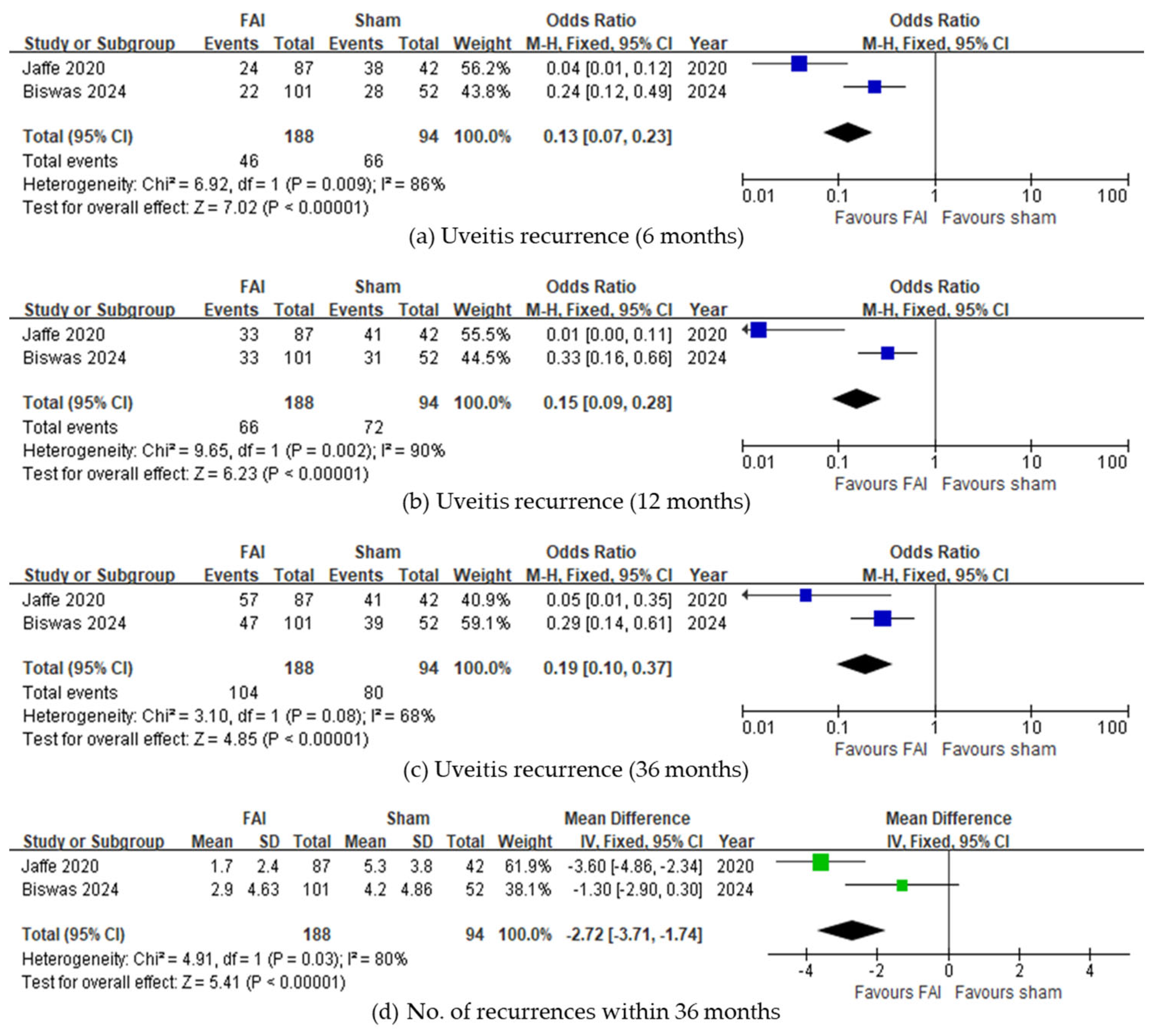
Regarding uveitis recurrence, fewer subjects experienced recurrence with the FAI compared to sham injections up to 6 months (95% CI 0.07–0.23, p < 0.001, Figure 2a), 12 months (95% CI 0.09–0.28, p < 0.001, Figure 2b), and 36 months (95% CI 0.10–0.37, p < 0.001, Figure 2c). Overall, fewer recurrences were observed in subjects with the FAI (95% CI −3.71 to −1.74, p < 0.001, Figure 2d). Regarding ME, both studies reported fewer eyes with ME at 36 months, although the meta-analysis was not conducted due to a lack of data.
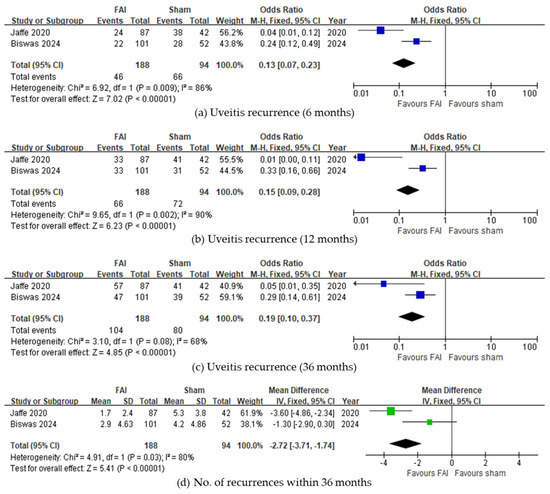
Figure 2.
Forest plots of the odds ratios and mean difference in uveitis recurrence-related outcomes. Proportion of subjects with uveitis recurrence for (a) 6, (b) 12, and (c) 36 months. (d) Number of uveitis recurrences within 36 months. The studies labeled as “Jaffe 2020” and “Biswas 2024” correspond to Ref. [22] and Ref. [23], respectively. The blue squares represent the effect estimate for dichotomous data, while the green squares represent the effect estimate for continuous data. CI: confidence interval; FAI: fluocinolone acetonide implant; SD: standard deviation.
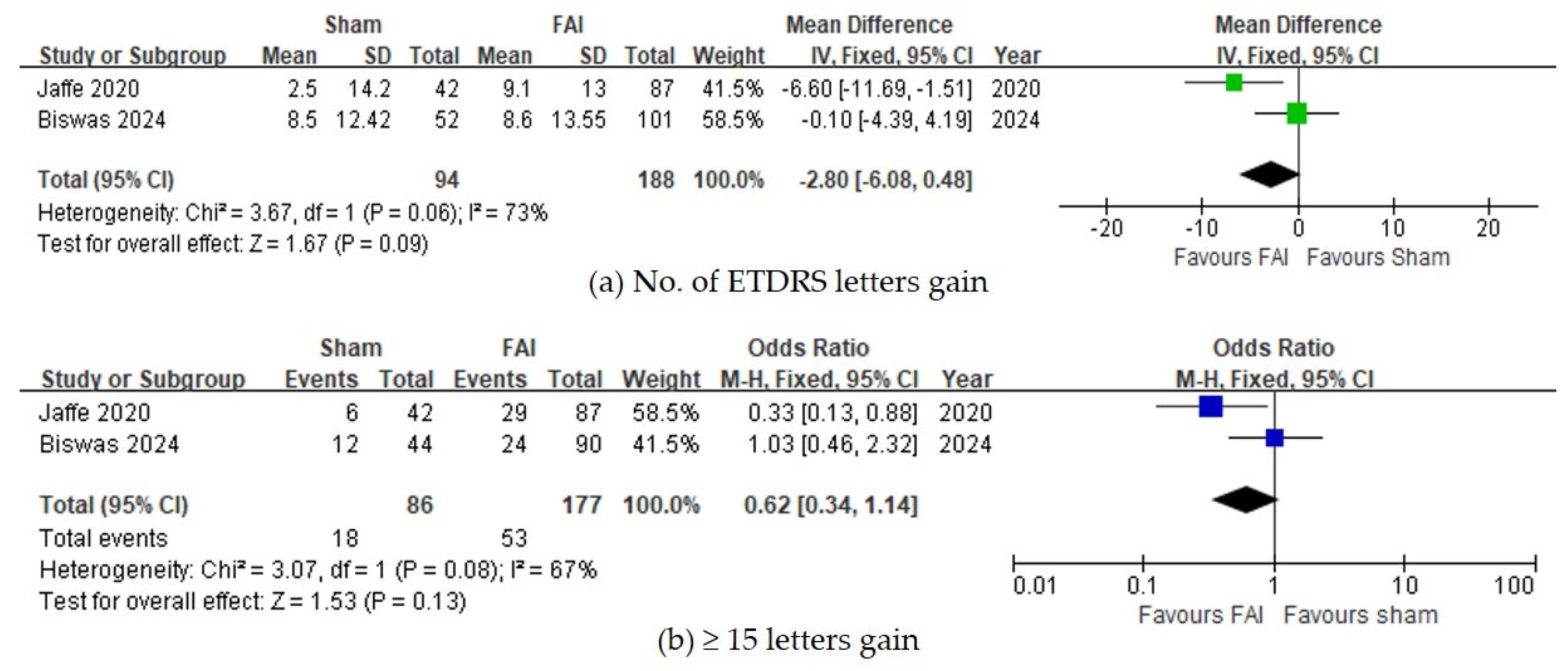
For visual acuity, no significant differences were observed between the FAI and sham for the number of gained ETDRS letters (95% CI −0.48 to 6.08, p = 0.09, Figure 3a) and for the proportion of subjects gaining ≥15 ETDRS letters (95% CI 0.87–2.95, p = 0.13, Figure 3b).
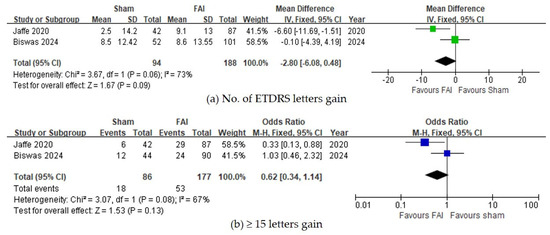
Figure 3.
Forest plots of the mean differences in visual acuity. (a) Change of best-corrected visual acuity by ETDRS letters over a 36-month period. (b) Proportion of subjects with ≥15 ETDRS letter gains over a 36-month period. The studies labeled as “Jaffe 2020” and “Biswas 2024” correspond to Ref. [22] and Ref. [23], respectively. The blue squares represent the effect estimate for dichotomous data, while the green squares represent the effect estimate for continuous data. CI: confidence interval; ETDRS: Early Treatment Diabetic Retinopathy Study; FAI: fluocinolone acetonide implant; SD: standard deviation.
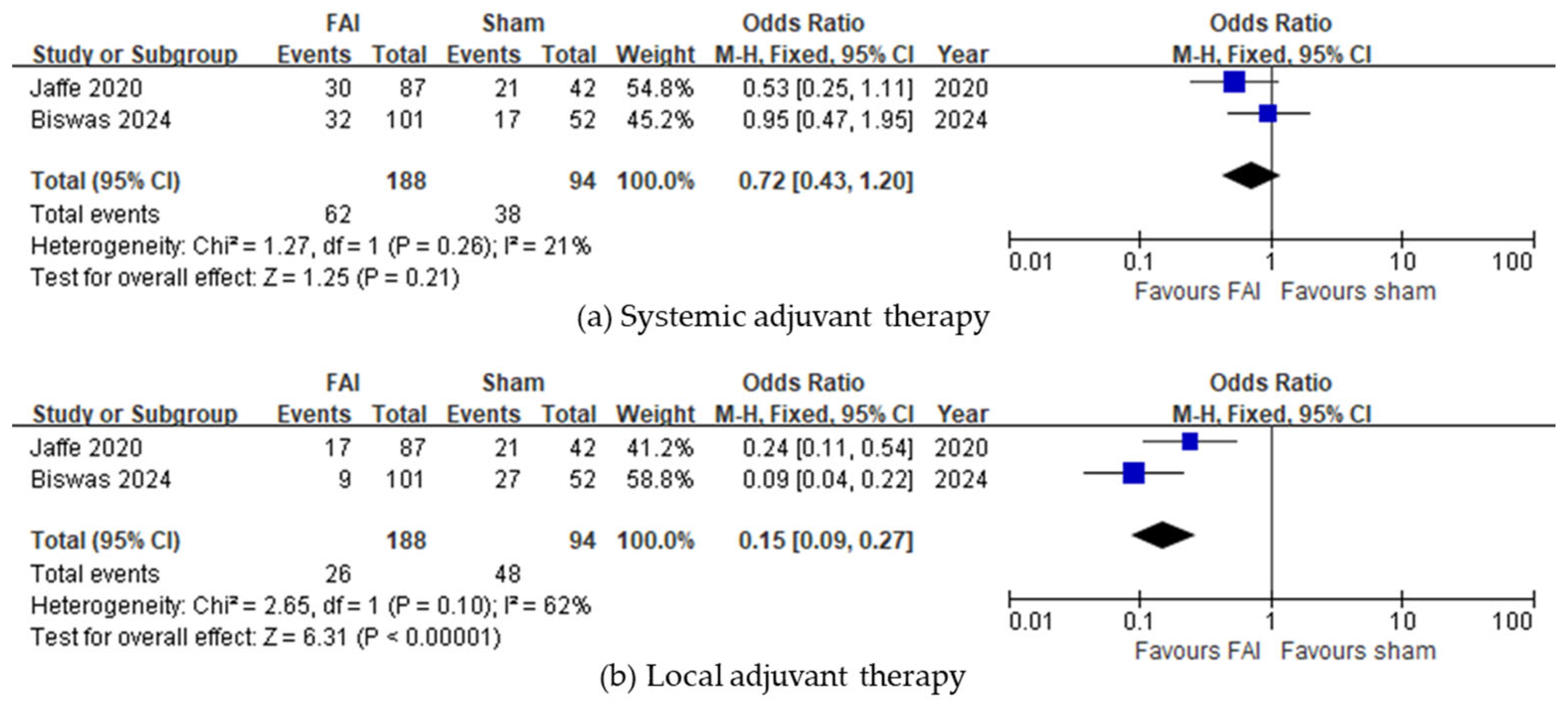
Regarding the need for any adjuvant therapy, the FAI was associated with a reduced need for local injections (95% CI 0.09–0.27, p < 0.001, Figure 4b), whereas no difference was observed regarding systemic treatment (95% CI 0.43–1.20, p = 0.21, Figure 4a).
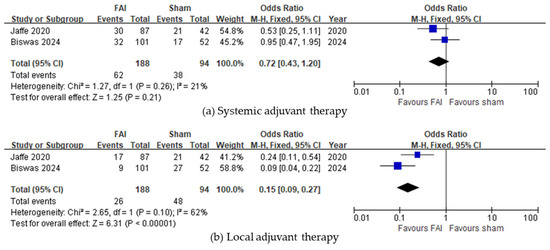
Figure 4.
Forest plots of the mean differences in adjuvant therapy. Proportion of subjects receiving adjuvant therapy in the form of (a) systemic treatment or (b) local injections over a 36-month period. The studies labeled as “Jaffe 2020” and “Biswas 2024” correspond to Ref. [22] and Ref. [23], respectively. The blue squares represent the effect estimate for dichotomous data. CI: confidence interval; FAI: fluocinolone acetonide implant.
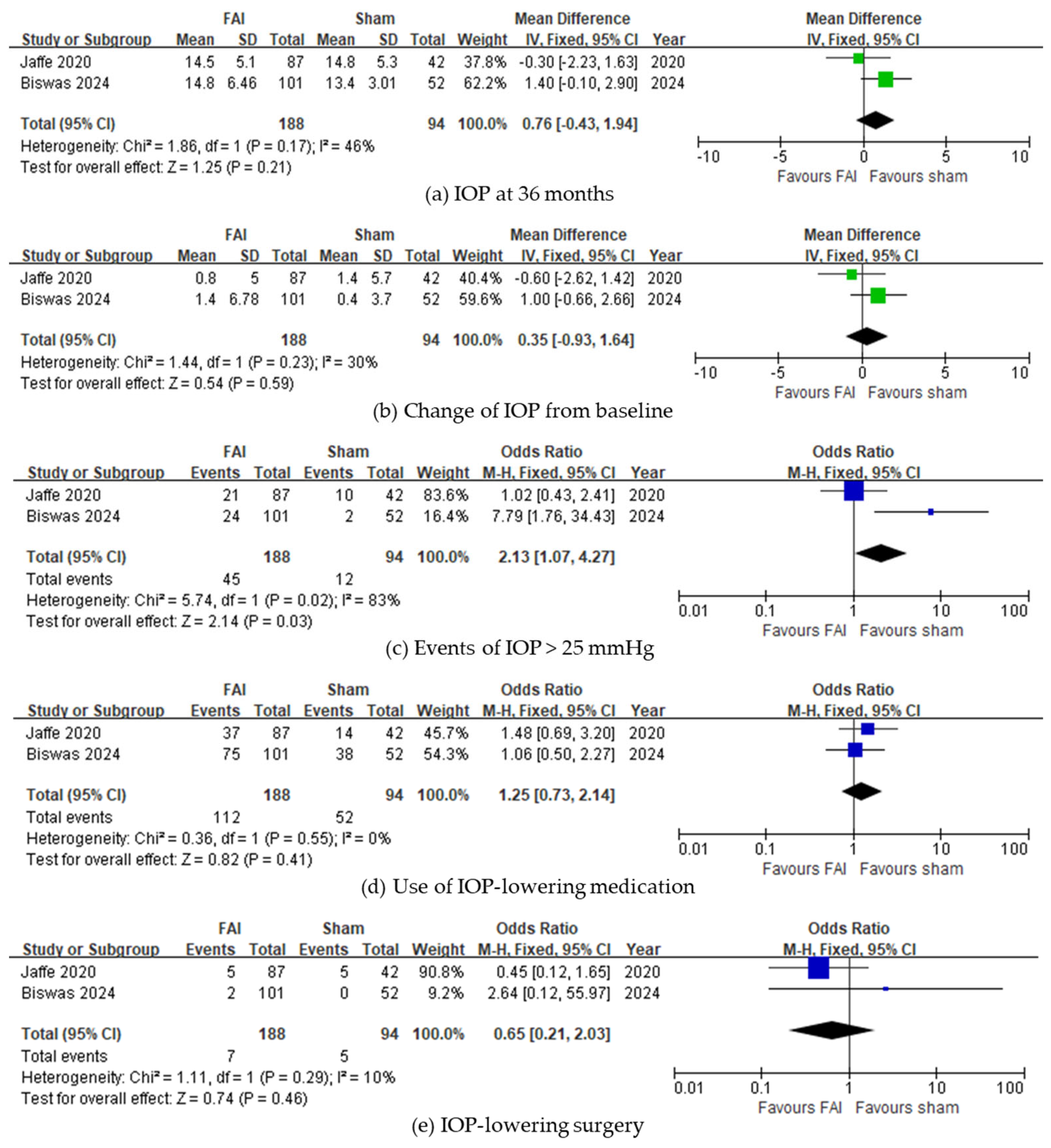
In IOP-related events, subjects with the FAI experienced more events of IOP > 25 mmHg (95% CI 0.73–2.14, p = 0.03, Figure 5c). However, no differences were noted in the final IOP at 36 months (95% CI −0.43 to 1.94, p = 0.21, Figure 5a) and change of IOP from baseline (95% CI −0.93 to 1.64, p = 0.59, Figure 5b). The use of IOP-lowering treatments was not significantly different between subjects treated with the FAI and those treated with sham injections (IOP-lowering medications: 95% CI 0.73–2.14, p = 0.41, Figure 5d; IOP-lowering surgery: 95% CI 0.21–2.03, p = 0.46, Figure 5e).
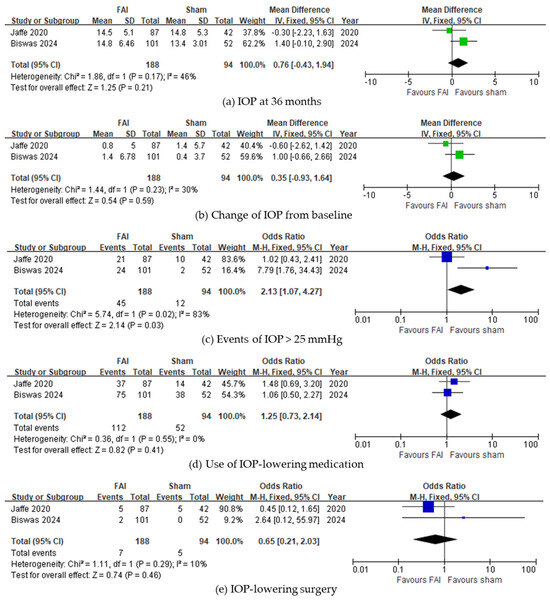
Figure 5.
Forest plots of the mean differences in IOP-related events. (a) IOP at month 36 (mmHg). (b) Change of IOP from baseline over a 36-month period. (c–e) Proportion of subjects (c) with IOP events of >25 mmHg, (d) requiring IOP-lowering medication, and (e) requiring IOP-lowering surgery up to month 36. The studies labeled as “Jaffe 2020” and “Biswas 2024” correspond to Ref. [22] and Ref. [23], respectively. The blue squares represent the effect estimate for dichotomous data, while the green squares represent the effect estimate for continuous data. CI: confidence interval; FAI: fluocinolone acetonide implant; IOP: intraocular pressure; SD: standard deviation.
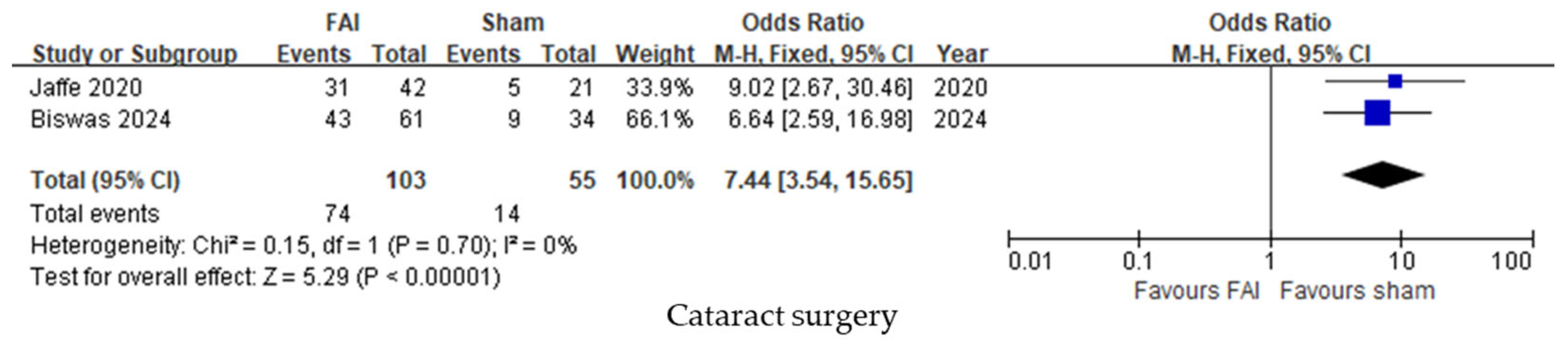
Cataract surgery was performed more frequently in subjects treated with the FAI compared to sham injections (95% CI 3.54–15.65, p < 0.001, Figure 6).
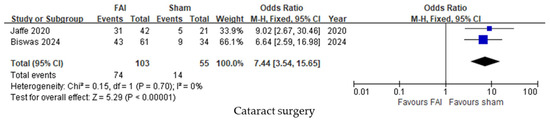
Figure 6.
Forest plots of the mean differences in cataract surgery up to month 36. The studies labeled as “Jaffe 2020” and “Biswas 2024” correspond to Ref. [22] and Ref. [23], respectively. The blue squares represent the effect estimate for dichotomous data. CI: confidence interval; FAI: fluocinolone acetonide implant.
4. Discussion
This study conducted a meta-analysis on the efficacy and safety of the 0.19 mg FAI (releasing at 0.2 μg/day) using two RCTs of similar design [22,23]. In terms of NIU recurrence, the FAI was obviously beneficial for the prevention of NIU recurrence, both in the proportion of subjects that experienced recurrence and in the mean number of recurrences up to 3 years. The efficacy of the FAI in preventing NIU recurrence was evident and has also been reported in non-comparative real-world studies [26,27], including those reported in this paper. The reduced recurrence was also noted in studies involving specific ocular inflammatory disorders, such as Vogt–Koyanagi–Harada disease [25].
Regarding visual improvement, some discrepancies existed between both RCTs, as one study reported the benefits of FAIs whereas the other study did not. This meta-analysis revealed that no significant benefits were noted with the FAI in terms of visual gain, both in the final visual gain by ETDRS letters and the proportion of patients gaining ≥15 letters. This might be due to the different proportion of eyes undergoing cataract surgery between the included RCTs, as it significantly improves the long-term visual acuity in eyes with ocular inflammatory diseases [35]. It is also to note that the study by Biswas et al. [23], which reported no differences in visual outcomes between the FAI and sham groups, included patients with a shorter duration of uveitis compared to those involved in the study by Jaffe et al. [22] (approximately 3.3 vs. 7.1 years). Visual impairment is expected to be more frequent in patients with NIU and longer disease duration as irreversible damage is more likely to occur, especially in NIU cases affecting the posterior segment with macular involvement [36]. Another potential explanation for the absence of a difference in visual improvement between the FAI and sham groups could be the definition of visual outcomes. The two RCTs included in this meta-analysis evaluated the proportion of patients achieving a gain of ≥15 letters over a period of up to three years, whereas most retrospective case series reported smaller degrees of visual improvement [16,18,34]. Moreover, most real-world non-comparative studies reported visual improvement for up to 2 years [14,16,17]; thus, further investigations are needed to confirm the long-term efficacy of FAI in terms of visual outcomes.
Studies evaluating the control of inflammation with detailed scores reported that anterior chamber cells and vitreous haze were improved with the FAI [31,34]. One study that investigated the efficacy of FAI in birdshot retinochoroidopathy also revealed that vascular leakage was improved on fluorescein angiography along with functional improvements on electroretinograms [29]. Another indicator of effective inflammation control is the need for any adjuvant therapy. This meta-analysis showed that local treatment was less needed in FAI-treated patients, whereas no change was noted in the use of systemic adjuvant therapy. This suggests that the FAI is beneficial in lightening the burden of treatment for clinicians and patients by reducing the need for local injections [30]. Some real-world studies reported that systemic adjunctive therapy could be decreased or even discontinued in FAI-treated eyes [17,31]. We should, however, consider the relationship between NIU and various systemic autoimmune disorders that may still require systemic treatments.
The efficacy of the FAI on ME was not investigated in this meta-analysis due to the unsuitability of reported results in the included RCTs. Overall, only a few eyes with ME were treated with the FAI in both trials. However, various real-world studies reported an improved central macular thickness with stable/improved vision [20]. Cai et al. [28] reported that NIU-related ME recurred in 16% of cases treated with FAI, with a mean time-to-recurrence of 37 months. In the study involving birdshot retinochoroiditis, FAI was able to resolve ME in all patients by 6 months [19]. Comparative studies are therefore needed to clarify the exact efficacy of the FAI in NIU patients with ME.
Among the non-controlled small-sized studies published in the literature, one investigated the differences between responders and non-responders to the FAI, suggesting that older age, higher need for previous injections of dexamethasone implant, and defects in the external limiting membrane and ellipsoid zone were more frequent in non-responders [33]. The present meta-analysis could not investigate any predictive factors of FAI efficacy. Thus, further studies are needed to identify patients that would benefit most from the implant.
In terms of safety, most variables associated with IOP were not significantly different between the 0.19 mg FAI and sham injections. One study comparing the IOP change with the dexamethasone implant and subsequent FAI reported that a significant increase was noted with dexamethasone, but not with FAI [32]. Most real-world studies also reported a non-significant IOP change or change manageable with standard therapy after the FAI [19,20,26,34]. Unlike the FAI using a higher dose (0.59 mg) that needs more glaucoma surgery than dexamethasone implants [37], IOP changes seemed to be controllable with the FAI containing 0.19 mg.
Cataract, which is a well-known complication of corticosteroids, was inevitable with the use of FAI [25,30]. Although cataract is a surgically treatable ocular condition, uveitic eyes are more prone to experience postoperative complications such as ME and epiretinal membranes [38,39]. Therefore, given this higher risk of postoperative complications [38,39], the decision to inject FAI must be made carefully and is usually not the first choice in young phakic patients.
The main limitations of this meta-analysis are the small number of included studies and its restriction to publications written in English. Although we included two RCTs with similar designs, some discrepancies were noted with regard to the duration of uveitis, patients’ age at enrollment, and the proportion of patients under systemic treatment at baseline. In the present work, analysis was limited to three time points (months 6, 12, and 36) due to the design of the included studies. More frequent follow-up intervals might reveal different outcomes regarding uveitis recurrences, which represents a potential bias that should also be acknowledged. The strength of this study lies in its comprehensive analysis of outcomes with reported discrepancies. Since most studies in the literature consist of non-controlled case series, we aimed to discuss these case series alongside the interpretations derived from the current meta-analysis.
5. Conclusions
In conclusion, the 0.19 mg FAI (0.2 μg/day) was effective in preventing NIU recurrence; however, its impact on visual acuity remained unclear, as no significant differences were observed between the FAI and the sham treatment. Additionally, the FAI did not appear to reduce the need for adjuvant systemic therapy, as systemic treatment usage was similar across groups. Nonetheless, IOP-related events could be controlled without significant surgical intervention, though cataract progression was observed in phakic eyes.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/biomedicines13020248/s1, File S1: Details of the literature search in the PubMed, EMBASE, and Cochrane Library databases; Table S1: Quality assessment of controlled intervention studies.
Author Contributions
Conceptualization, Y.-R.C. and S.T.; methodology, Y.-R.C., B.B. and S.T.; investigation, S.Y., Y.-R.C. and S.T.; data curation, S.Y. and Y.-R.C.; writing—original draft preparation, S.Y., Y.-R.C. and S.T.; writing—review and editing, Y.-R.C., J.H.S., B.B. and S.T.; supervision, J.H.S. and B.B. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Ethical review and approval were waived for this study due to the nature of the meta-analysis studies.
Informed Consent Statement
Not applicable.
Data Availability Statement
The original contributions presented in this study are included in the article/supplementary material. Further inquiries can be directed to the corresponding author.
Acknowledgments
The authors are grateful to Yujin Kwon, medical librarian of the Medical Information and Media Center, Ajou University School of Medicine, for her assistance with the systematic literature search. This study was partially presented as a poster at the 132nd Annual Meeting of the Korean Ophthalmological Society in Seoul, Republic of Korea, 29 November–1 December 2024.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Mushtaq, Y.; Mushtaq, M.M.; Gatzioufas, Z.; Ripa, M.; Motta, L.; Panos, G.D. Intravitreal fluocinolone acetonide implant (ILUVIEN®) for the treatment of retinal conditions. a review of clinical studies. Drug. Des. Devel. Ther. 2023, 17, 961–975. [Google Scholar] [CrossRef] [PubMed]
- Liao, W.; Zhong, Z.; Su, G.; Feng, X.; Yang, P. Comparative Efficacy and Safety of Advanced Intravitreal Therapeutic Agents for Noninfectious Uveitis: A Systematic Review and Network Meta-Analysis. Front. Pharmacol. 2022, 13, 749312. [Google Scholar] [CrossRef] [PubMed]
- Pleyer, U.; Pavesio, C.; Miserocchi, E.; Heinz, C.; Devonport, H.; Llorenç, V.; Burke, T.; Nogueira, V.; Kodjikian, L.; Bodaghi, B. Fluocinolone acetonide 0.2 µg/day intravitreal implant in non-infectious uveitis affecting the posterior segment: EU expert user panel consensus-based clinical recommendations. J. Ophthalmic Inflamm. Infect. 2024, 14, 22. [Google Scholar] [CrossRef]
- Bodaghi, B.; Nguyen, Q.D.; Jaffe, G.; Khoramnia, R.; Pavesio, C. Preventing relapse in non-infectious uveitis affecting the posterior segment of the eye-evaluating the 0.2 μg/day fluocinolone acetonide intravitreal implant (ILUVIEN®). J. Ophthalmic Inflamm. Infect. 2020, 10, 32. [Google Scholar] [CrossRef] [PubMed]
- Fallico, M.; Maugeri, A.; Lotery, A.; Longo, A.; Bonfiglio, V.; Russo, A.; Avitabile, T.; Furino, C.; Cennamo, G.; Barchitta, M.; et al. Fluocinolone acetonide vitreous insert for chronic diabetic macular oedema: A systematic review with meta-analysis of real-world experience. Sci. Rep. 2021, 11, 4800. [Google Scholar] [CrossRef]
- Ghasemi Falavarjani, K. Implantable posterior segment drug delivery devices; novel alternatives to currently available treatments. J. Ophthalmic Vis. Res. 2009, 4, 191–193. [Google Scholar]
- Campochiaro, P.A.; Brown, D.M.; Pearson, A.; Chen, S.; Boyer, D.; Ruiz-Moreno, J.; Garretson, B.; Gupta, A.; Hariprasad, S.M.; Bailey, C.; et al. Sustained delivery fluocinolone acetonide vitreous inserts provide benefit for at least 3 years in patients with diabetic macular edema. Ophthalmology 2012, 119, 2125–2132. [Google Scholar] [CrossRef]
- Jomaa, E.; Schweitzer, C.; Delyfer, M.N. Elevated intraocular pressure associated with anterior chamber migration of fluocinolone acetonide implant 1 year after administration. JAMA Ophthalmol. 2022, 140, e223780. [Google Scholar] [CrossRef]
- Alzaabi, M.; Taguri, A.H.; Elbarky, A. Anterior migration of intravitreal fluocinolone acetonide (Iluvien®) implant in a pseudophakic eye with intact posterior capsule. Am. J. Ophthalmol. Case Rep. 2020, 20, 100922. [Google Scholar] [CrossRef]
- Rahimy, E.; Khurana, R.N. Anterior segment migration of dexamethasone implant: Risk factors, complications, and management. Curr. Opin. Ophthalmol. 2017, 28, 246–251. [Google Scholar] [CrossRef]
- Mao, X.; Dai, Z.; Yang, J.; Wu, Y.; Xie, F.; Lu, Y.; Yu, J.; Chang, F.; Lu, Y. Cost-effectiveness analysis of fluocinolone acetonide intravitreal (FAI) implant for chronic noninfectious uveitis affecting the posterior segment of the eye (NIU-PS) in China. Ophthalmol. Ther. 2024, 13, 1757–1772. [Google Scholar] [CrossRef] [PubMed]
- Pouwels, X.; Petersohn, S.; Carrera, V.H.; Denniston, A.K.; Chalker, A.; Raatz, H.; Armstrong, N.; Shah, D.; Witlox, W.; Worthy, G.; et al. Fluocinolone acetonide intravitreal implant for treating recurrent non-infectious uveitis: An evidence review group perspective of a NICE Single Technology Appraisal. Pharmacoeconomics 2020, 38, 431–441. [Google Scholar] [CrossRef] [PubMed]
- Sugar, E.A.; Holbrook, J.T.; Kempen, J.H.; Burke, A.E.; Drye, L.T.; Thorne, J.E.; Louis, T.A.; Jabs, D.A.; Altaweel, M.M.; Frick, K.D. Cost-effectiveness of fluocinolone acetonide implant versus systemic therapy for noninfectious intermediate, posterior, and panuveitis. Ophthalmology 2014, 121, 1855–1862. [Google Scholar] [CrossRef] [PubMed]
- Jabbour, M.; Kodjikian, L.; Bourdin, A.; Rougier, M.B.; Serrar, Y.; Weber, M.; Massé, H.; Mazhar, D.; Perez-Roustit, S.; Chiquet, C.; et al. Efficacity and Safety of the Fluocinolone Acetonide Implant in Uveitic Macular Edema: A Real-Life Study from the French Uveitis Network. J. Pers. Med. 2024, 14, 245. [Google Scholar] [CrossRef]
- Pockar, S.; Leal, I.; Chhabra, R.; Jones, N.P.; Steeples, L.R. Intravitreal Fluocinolone 0.19mg Implant in the Management of Chronic Non-Infectious Uveitis: 12-Month Outcomes from a Single Tertiary Centre. Ocul. Immunol. Inflamm. 2023, 31, 1572–1578. [Google Scholar] [CrossRef]
- Kessler, L.J.; Auffarth, G.U.; Khoramnia, R. Functional and Morphological Responses to Fluocinolone Acetonide 0.19 mg in Noninfectious Uveitic Macular Edema Evaluated as the Area-Under-the-Curve. J. Ocul. Pharmacol. Ther. 2023, 39, 449–455. [Google Scholar] [CrossRef]
- Studsgaard, A.; Clemmensen, K.; Nielsen, M.S. Intravitreal fluocinolone acetonide 0.19 mg (Iluvien®) for the treatment of uveitic macular edema: 2-year follow-up of 20 patients. Graefes Arch. Clin. Exp. Ophthalmol. 2022, 260, 1633–1639. [Google Scholar] [CrossRef]
- Battista, M.; Starace, V.; Cicinelli, M.V.; Capone, L.; Marchese, A.; Modorati, G.; Bandello, F.; Miserocchi, E. Efficacy of 0.19 mg Fluocinolone Acetonide Implant in Non-infectious Posterior Uveitis Evaluated as Area Under the Curve. Ophthalmol. Ther. 2022, 11, 215–224. [Google Scholar] [CrossRef]
- Hikal, M.; Celik, N.; Auffarth, G.U.; Kessler, L.J.; Mayer, C.S.; Khoramnia, R. Intravitreal 0.19 mg Fluocinolone Acetonide Implant in Non-Infectious Uveitis. J. Clin. Med. 2021, 10, 3966. [Google Scholar] [CrossRef]
- Weber, L.F.; Marx, S.; Auffarth, G.U.; Scheuerle, A.F.; Tandogan, T.; Mayer, C.; Khoramnia, R. Injectable 0.19-mg fluocinolone acetonide intravitreal implant for the treatment of non-infectious uveitic macular edema. J. Ophthalmic Inflamm. Infect. 2019, 9, 3. [Google Scholar] [CrossRef]
- Gao, L.; Zhao, X.; Jiao, L.; Tang, L. Intravitreal corticosteroids for diabetic macular edema: A network meta-analysis of randomized controlled trials. Eye Vis. 2021, 8, 35. [Google Scholar] [CrossRef] [PubMed]
- Jaffe, G.J.; Pavesio, C.E. Effect of a Fluocinolone Acetonide Insert on Recurrence Rates in Noninfectious Intermediate, Posterior, or Panuveitis: Three-Year Results. Ophthalmology 2020, 127, 1395–1404. [Google Scholar] [CrossRef] [PubMed]
- Biswas, J.; Tyagi, M.; Agarwal, M. The 0.2-μg/day Fluocinolone Acetonide Intravitreal Implant in Chronic Noninfectious Posterior Uveitis: A 3-year Randomized Trial in India. Ophthalmol. Sci. 2024, 4, 100403. [Google Scholar] [CrossRef]
- Dettori, J.R.; Norvell, D.C.; Chapman, J.R. Fixed-Effect vs Random-Effects Models for Meta-Analysis: 3 Points to Consider. Glob. Spine J. 2022, 12, 1624–1626. [Google Scholar] [CrossRef] [PubMed]
- Heo, J.W.; Cho, B.J.; Goldstein, D.A.; Sepah, Y.J.; Do, D.V.; Nguyen, Q.D. Fluocinolone Acetonide Implant for Vogt-Koyanagi-Harada Disease: Three-Year Outcomes of Efficacy and Safety. Retina 2016, 36, 2124–2131. [Google Scholar] [CrossRef]
- Buhl, L.; Thurau, S.; Kern, C. Fluocinolone acetonide 0.19-mg implant for the treatment of noninfectious uveitis with involvement of the posterior segment: A real-world study. Graefes Arch. Clin. Exp. Ophthalmol. 2023, 261, 1101–1108. [Google Scholar] [CrossRef]
- Jaffe, G.J.; Lin, P.; Keenan, R.T.; Ashton, P.; Skalak, C.; Stinnett, S.S. Injectable Fluocinolone Acetonide Long-Acting Implant for Noninfectious Intermediate Uveitis, Posterior Uveitis, and Panuveitis: Two-Year Results. Ophthalmology 2016, 123, 1940–1948. [Google Scholar] [CrossRef]
- Cai, C.X.; Skalak, C.; Keenan, R.T.; Grewal, D.S.; Jaffe, G.J. Time to disease recurrence in noninfectious uveitis following long-acting injectable fluocinolone acetonide implant. Graefes Arch. Clin. Exp. Ophthalmol. 2020, 258, 1023–1030. [Google Scholar] [CrossRef]
- Ajamil-Rodanes, S.; Testi, I.; Luis, J.; Robson, A.G.; Westcott, M.; Pavesio, C. Evaluation of fluocinolone acetonide 0.19 mg intravitreal implant in the management of birdshot retinochoroiditis. Br. J. Ophthalmol. 2022, 106, 234–240. [Google Scholar] [CrossRef]
- Pavesio, C.; Heinz, C. Non-infectious uveitis affecting the posterior segment treated with fluocinolone acetonide intravitreal implant: 3-year fellow eye analysis. Eye 2022, 36, 1231–1237. [Google Scholar] [CrossRef]
- Moll-Udina, A.; Hernanz, I.; Sainz-de-la-Maza, M.; Pelegrín, L.; Coelho-Borges, A.I.; Pazos, M.; Adán, A.; Llorenç, V. Intravitreal fluocinolone acetonide 0.19 mg (ILUVIEN®) in patients with non-infectious uveitis: Real-world effectiveness and safety outcomes at 12 months. Int. Ophthalmol. 2023, 43, 4181–4195. [Google Scholar] [CrossRef] [PubMed]
- Kriegel, M.; Heiligenhaus, A.; Heinz, C. Comparing the Efficacy of Intravitreal Dexamethasone and Time-displaced Fluocinolone Acetonide on Central Retinal Thickness in Patients with Uveitis. Ocul. Immunol. Inflamm. 2023, 31, 168–174. [Google Scholar] [CrossRef] [PubMed]
- Abu Arif, J.; Knecht, V.A.; Rübsam, A.; Lussac, V.; Jami, Z.; Pohlmann, D.; Müller, B.; Pleyer, U. Fluocinolone Acetonide Implant for Uveitis: Dissecting Responder and Non-Responder Outcomes at a Tertiary Center. Biomedicines 2024, 12, 1106. [Google Scholar] [CrossRef] [PubMed]
- Kozak, I.; Gurbaxani, A.; Pandova, M. The effectiveness of the 0.19 mg fluocinolone acetonide implant in treating non-infectious posterior uveitis: A real-world experience. J. Ophthalmic Inflamm. Infect. 2024, 14, 24. [Google Scholar] [CrossRef]
- Gangaputra, S.; Newcomb, C.; Armour, R.; Choi, D.; Ying, G.S.; Groth, S.; Begum, H.; Fitzgerald, T.; Artornsombudh, P.; Daniel, E.; et al. Long-term visual acuity outcomes following cataract surgery in eyes with ocular inflammatory disease. Br. J. Ophthalmol. 2024, 108, 380–385. [Google Scholar] [CrossRef]
- Amer, R.; Alsughayyar, W.; Almeida, D. Pattern and causes of visual loss in Behçet’s uveitis: Short-term and long-term outcomes. Graefes Arch. Clin. Exp. Ophthalmol. 2017, 255, 1423–1432. [Google Scholar] [CrossRef]
- José-Vieira, R.; Ferreira, A.; Menéres, P.; Sousa-Pinto, B.; Figueira, L. Efficacy and safety of intravitreal and periocular injection of corticosteroids in noninfectious uveitis: A systematic review. Surv. Ophthalmol. 2022, 67, 991–1013. [Google Scholar] [CrossRef]
- Gangaputra, S.; Newcomb, C.; Ying, G.S.; Groth, S.; Fitzgerald, T.D.; Artornsombudh, P.; Kothari, S.; Pujari, S.S.; Jabs, D.A.; Levy-Clarke, G.A.; et al. Incidence and Remission of Post-Surgical Cystoid Macular Edema Following Cataract Surgery in Eyes With Intraocular Inflammation. Am. J. Ophthalmol. 2024, 267, 182–191. [Google Scholar] [CrossRef]
- Bajraktari, G.; Jukić, T.; Kalauz, M.; Oroz, M.; Radolović Bertetić, A.; Vukojević, N. Early and Late Complications after Cataract Surgery in Patients with Uveitis. Medicina 2023, 59, 1877. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).