Exome Sequencing to Identify Novel Variants Associated with Secondary Amenorrhea and Premature Ovarian Insufficiency (POI) in Saudi Women
Abstract
1. Introduction
2. Materials and Methods
2.1. Recruitment of POI Patients/Controls and Ethical Approval
2.2. POI Selection Criteria
2.3. Biochemical Profile and Hormonal Analysis
2.4. Genomic DNA Extraction and Spectrophotometry
2.5. Genomic Library Preparation and Whole-Exome Sequencing (WES)
2.6. Prediction of the Pathogenicity of Variants
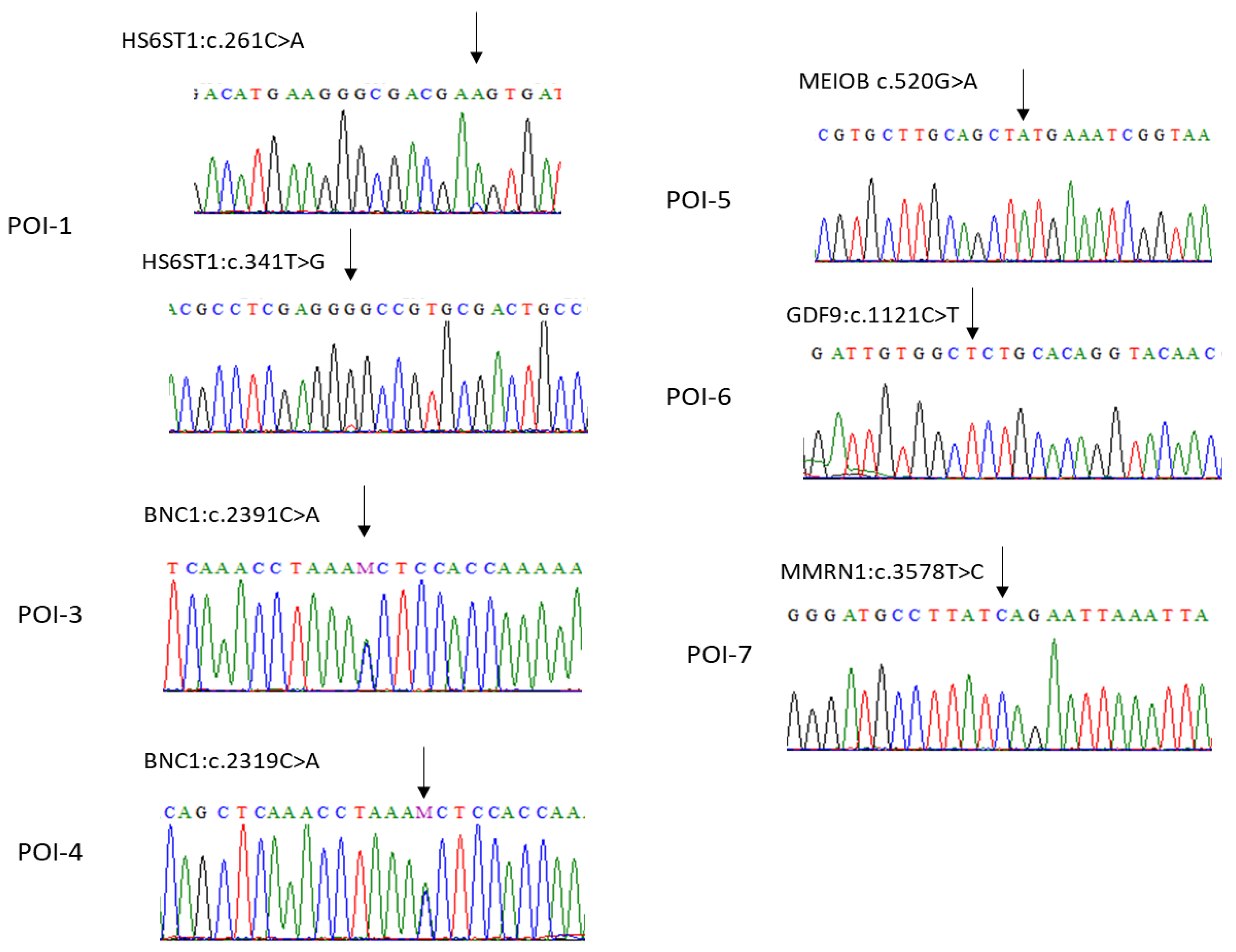
2.7. Design of Specific Primers and Validation of Variants through Sanger Sequencing
2.8. In Silico Analysis
3. Results
3.1. Clinical Characteristics and Hormonal Analysis Evaluation
3.2. POI-Associated Variants Identified in Patients
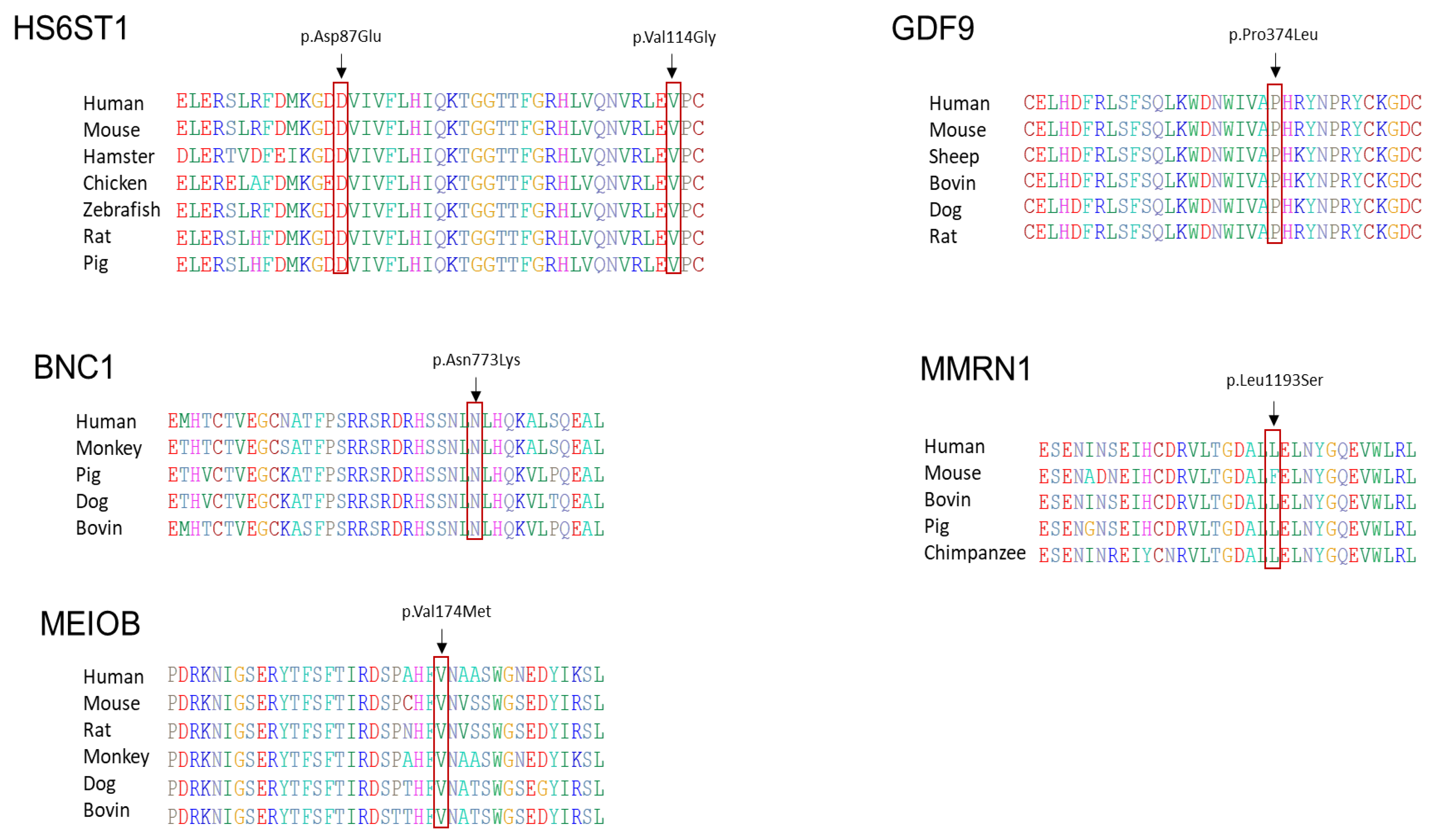
3.3. In Silico Analysis
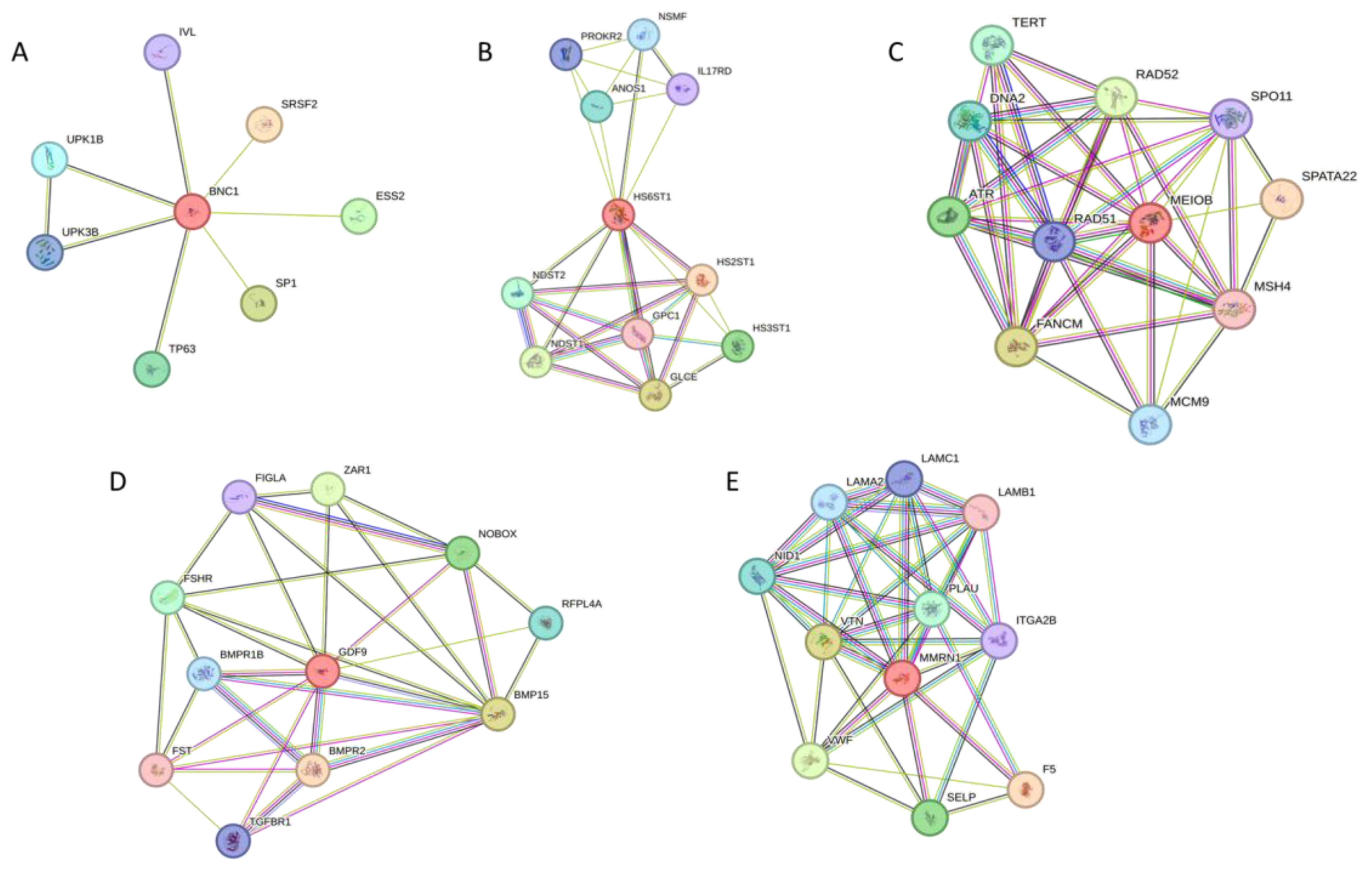
3.4. Protein Interaction Network of Candidate POI Genes
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Pellicer, N.; Cozzolino, M.; Diaz-García, C.; Galliano, D.; Cobo, A.; Pellicer, A.; Herraiz, S. Ovarian rescue in women with premature ovarian insufficiency: Facts and fiction. Reprod. Biomed. Online 2023, 46, 543–565. [Google Scholar] [CrossRef]
- Inhorn, M.C.; Patrizio, P. Infertility around the globe: New thinking on gender, reproductive technologies and global movements in the 21st century. Hum. Reprod. Update 2015, 21, 411–426. [Google Scholar] [CrossRef]
- Margaux Becker, V.; Silver, S.; Seufert, R.; Muensterer, O.J. The Association of Appendectomy, Adhesions, Tubal Pathology, and Female Infertility. JSLS 2019, 23, e2018.00099. [Google Scholar] [CrossRef]
- Nelson, L.M. Clinical practice. Primary ovarian insufficiency. N. Engl. J. Med. 2009, 360, 606–614. [Google Scholar] [CrossRef]
- Rudnicka, E.; Kruszewska, J.; Klicka, K.; Kowalczyk, J.; Grymowicz, M.; Skórska, J.; Pięta, W.; Smolarczyk, R. Premature ovarian insufficiency—Aetiopathology, epidemiology, and diagnostic evaluation. Prz. Menopauzalny 2018, 17, 105–108. [Google Scholar] [CrossRef]
- Chon, S.J.; Umair, Z.; Yoon, M.S. Premature Ovarian Insufficiency: Past, Present, and Future. Front. Cell Dev. Biol. 2021, 9, 672890. [Google Scholar] [CrossRef]
- Rebar, R.W. Premature ovarian failure. Obstet. Gynecol. 2009, 113, 1355–1363. [Google Scholar] [CrossRef]
- Torrealday, S.; Kodaman, P.; Pal, L. Premature Ovarian Insufficiency—An update on recent advances in understanding and management. F1000Research 2017, 6, 2069. [Google Scholar] [CrossRef]
- Chae-Kim, J.J.; Gavrilova-Jordan, L. Premature Ovarian Insufficiency: Procreative Management and Preventive Strategies. Biomedicines 2018, 7, 2. [Google Scholar] [CrossRef]
- Persani, L.; Rossetti, R.; Cacciatore, C.; Bonomi, M. Primary Ovarian Insufficiency: X chromosome defects and autoimmunity. J. Autoimmun. 2009, 33, 35–41. [Google Scholar] [CrossRef]
- Beke, A.; Piko, H.; Haltrich, I.; Csomor, J.; Matolcsy, A.; Fekete, G.; Rigo, J.; Karcagi, V. Molecular cytogenetic analysis of Xq critical regions in premature ovarian failure. Mol. Cytogenet. 2013, 6, 62. [Google Scholar] [CrossRef]
- Man, L.; Lekovich, J.; Rosenwaks, Z.; Gerhardt, J. Fragile X-Associated Diminished Ovarian Reserve and Primary Ovarian Insufficiency from Molecular Mechanisms to Clinical Manifestations. Front. Mol. Neurosci. 2017, 10, 290. [Google Scholar] [CrossRef]
- Qin, Y.; Jiao, X.; Simpson, J.L.; Chen, Z.J. Genetics of primary ovarian insufficiency: New developments and opportunities. Hum. Reprod. Update 2015, 21, 787–808. [Google Scholar] [CrossRef]
- Tucker, E.J.; Grover, S.R.; Bachelot, A.; Touraine, P.; Sinclair, A.H. Premature Ovarian Insufficiency: New Perspectives on Genetic Cause and Phenotypic Spectrum. Endocr. Rev. 2016, 37, 609–635. [Google Scholar] [CrossRef]
- Jiao, X.; Ke, H.; Qin, Y.; Chen, Z.J. Molecular Genetics of Premature Ovarian Insufficiency. Trends Endocrinol. Metab. 2018, 29, 795–807. [Google Scholar] [CrossRef]
- Ke, H.; Tang, S.; Guo, T.; Hou, D.; Jiao, X.; Li, S.; Luo, W.; Xu, B.; Zhao, S.; Li, G.; et al. Landscape of pathogenic mutations in premature ovarian insufficiency. Nat. Med. 2023, 29, 483–492. [Google Scholar] [CrossRef]
- Liu, H.; Wei, X.; Sha, Y.; Liu, W.; Gao, H.; Lin, J.; Li, Y.; Tang, Y.; Wang, Y.; Wang, Y.; et al. Whole-exome sequencing in patients with premature ovarian insufficiency: Early detection and early intervention. J. Ovarian Res. 2020, 13, 114. [Google Scholar] [CrossRef]
- Almatrafi, A.M.; Hibshi, A.M.; Basit, S. Association of Homozygous PROP1 Mutation in a Saudi Family with Combined Pituitary Hormone Deficiency. Medicina 2023, 59, 474. [Google Scholar] [CrossRef]
- Almatrafi, A.; Umair, M.; Eldardear, A.; Al-Luqmani, M.; Hashmi, J.A.; Albalawi, A.M.; Alfadhel, M.; Ramzan, K.; Basit, S. A homozygous missense variant in the homeobox domain of the NKX6-2 results in progressive spastic ataxia type 8 associated with lower limb weakness and neurological manifestations. J. Gene Med. 2020, 22, e3196. [Google Scholar] [CrossRef]
- Teumer, J.; Tseng, H.; Green, H. The human basonuclin gene. Gene 1997, 188, 1–7. [Google Scholar] [CrossRef]
- Bülow, H.E.; Hobert, O. The molecular diversity of glycosaminoglycans shapes animal development. Annu. Rev. Cell Dev. Biol. 2006, 22, 375–407. [Google Scholar] [CrossRef]
- Tornberg, J.; Sykiotis, G.P.; Keefe, K.; Plummer, L.; Hoang, X.; Hall, J.E.; Quinton, R.; Seminara, S.B.; Hughes, V.; Van Vliet, G.; et al. Heparan sulfate 6-O-sulfotransferase 1, a gene involved in extracellular sugar modifications, is mutated in patients with idiopathic hypogonadotrophic hypogonadism. Proc. Natl. Acad. Sci. USA 2011, 108, 11524–11529. [Google Scholar] [CrossRef]
- Seminara, S.B.; Hayes, F.J.; Crowley, W.F., Jr. Gonadotropin-releasing hormone deficiency in the human (idiopathic hypogonadotropic hypogonadism and Kallmann’s syndrome): Pathophysiological and genetic considerations. Endocr. Rev. 1998, 19, 521–539. [Google Scholar] [CrossRef]
- Baronchelli, S.; Villa, N.; Redaelli, S.; Lissoni, S.; Saccheri, F.; Panzeri, E.; Conconi, D.; Bentivegna, A.; Crosti, F.; Sala, E.; et al. Investigating the role of X chromosome breakpoints in premature ovarian failure. Mol. Cytogenet. 2012, 5, 32. [Google Scholar] [CrossRef]
- Jeimy, S.B.; Fuller, N.; Tasneem, S.; Segers, K.; Stafford, A.R.; Weitz, J.I.; Camire, R.M.; Nicolaes, G.A.; Hayward, C.P. Multimerin 1 binds factor V and activated factor V with high affinity and inhibits thrombin generation. Thromb. Haemost. 2008, 100, 1058–1067. [Google Scholar] [CrossRef]
- Tian, Q.; Kopf, G.S.; Brown, R.S.; Tseng, H. Function of basonuclin in increasing transcription of the ribosomal RNA genes during mouse oogenesis. Development 2001, 128, 407–416. [Google Scholar] [CrossRef]
- Zhang, D.; Liu, Y.; Zhang, Z.; Lv, P.; Liu, Y.; Li, J.; Wu, Y.; Zhang, R.; Huang, Y.; Xu, G.; et al. Basonuclin 1 deficiency is a cause of primary ovarian insufficiency. Hum. Mol. Genet. 2018, 27, 3787–3800. [Google Scholar] [CrossRef]
- Souquet, B.; Abby, E.; Hervé, R.; Finsterbusch, F.; Tourpin, S.; Le Bouffant, R.; Duquenne, C.; Messiaen, S.; Martini, E.; Bernardino-Sgherri, J.; et al. MEIOB targets single-strand DNA and is necessary for meiotic recombination. PLoS Genet. 2013, 9, e1003784. [Google Scholar] [CrossRef]
- Luo, M.; Yang, F.; Leu, N.A.; Landaiche, J.; Handel, M.A.; Benavente, R.; La Salle, S.; Wang, P.J. MEIOB exhibits single-stranded DNA-binding and exonuclease activities and is essential for meiotic recombination. Nat. Commun. 2013, 4, 2788. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Liu, L.; Tan, C.; Meng, G.; Meng, L.; Nie, H.; Du, J.; Lu, G.X.; Lin, G.; He, W.B.; et al. Novel MEIOB variants cause primary ovarian insufficiency and non-obstructive azoospermia. Front. Genet. 2022, 13, 936264. [Google Scholar] [CrossRef] [PubMed]
- Caburet, S.; Todeschini, A.L.; Petrillo, C.; Martini, E.; Farran, N.D.; Legois, B.; Livera, G.; Younis, J.S.; Shalev, S.; Veitia, R.A. A truncating MEIOB mutation responsible for familial primary ovarian insufficiency abolishes its interaction with its partner SPATA22 and their recruitment to DNA double-strand breaks. eBioMedicine 2019, 42, 524–531. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Greenberg, R.A.; Schonbrunn, E.; Wang, P.J. Meiosis-specific proteins MEIOB and SPATA22 cooperatively associate with the single-stranded DNA-binding replication protein A complex and DNA double-strand breaks. Biol. Reprod. 2017, 96, 1096–1104. [Google Scholar] [CrossRef] [PubMed]
- Dong, J.; Albertini, D.F.; Nishimori, K.; Kumar, T.R.; Lu, N.; Matzuk, M.M. Growth differentiation factor-9 is required during early ovarian folliculogenesis. Nature 1996, 383, 531–535. [Google Scholar] [CrossRef] [PubMed]
- Yan, C.; Wang, P.; DeMayo, J.; DeMayo, F.J.; Elvin, J.A.; Carino, C.; Prasad, S.V.; Skinner, S.S.; Dunbar, B.S.; Dube, J.L.; et al. Synergistic roles of bone morphogenetic protein 15 and growth differentiation factor 9 in ovarian function. Mol. Endocrinol. 2001, 15, 854–866. [Google Scholar] [CrossRef] [PubMed]
- Laissue, P.; Christin-Maitre, S.; Touraine, P.; Kuttenn, F.; Ritvos, O.; Aittomaki, K.; Bourcigaux, N.; Jacquesson, L.; Bouchard, P.; Frydman, R.; et al. Mutations and sequence variants in GDF9 and BMP15 in patients with premature ovarian failure. Eur. J. Endocrinol. 2006, 154, 739–744. [Google Scholar] [CrossRef] [PubMed]
- Dixit, H.; Rao, L.K.; Padmalatha, V.; Kanakavalli, M.; Deenadayal, M.; Gupta, N.; Chakravarty, B.; Singh, L. Mutational screening of the coding region of growth differentiation factor 9 gene in Indian women with ovarian failure. Menopause 2005, 12, 749–754. [Google Scholar] [CrossRef] [PubMed]
- Zhao, H.; Qin, Y.; Kovanci, E.; Simpson, J.L.; Chen, Z.J.; Rajkovic, A. Analyses of GDF9 mutation in 100 Chinese women with premature ovarian failure. Fertil. Steril. 2007, 88, 1474–1476. [Google Scholar] [CrossRef] [PubMed]
- Lindahl, U.; Kusche-Gullberg, M.; Kjellén, L. Regulated diversity of heparan sulfate. J. Biol. Chem. 1998, 273, 24979–24982. [Google Scholar] [CrossRef]
- Leatherdale, A.; Parker, D.; Tasneem, S.; Wang, Y.; Bihan, D.; Bonna, A.; Hamaia, S.W.; Gross, P.L.; Ni, H.; Doble, B.W.; et al. Multimerin 1 supports platelet function in vivo and binds to specific GPAGPOGPX motifs in fibrillar collagens that enhance platelet adhesion. J. Thromb. Haemost. 2021, 19, 547–561. [Google Scholar] [CrossRef]
- Alhathal, N.; Maddirevula, S.; Coskun, S.; Alali, H.; Assoum, M.; Morris, T.; Deek, H.A.; Hamed, S.A.; Alsuhaibani, S.; Mirdawi, A.; et al. A genomics approach to male infertility. Genet. Med. 2020, 22, 1967–1975. [Google Scholar] [CrossRef]
- Nappi, R.E.; Cucinella, L.; Martini, E.; Rossi, M.; Tiranini, L.; Martella, S.; Bosoni, D.; Cassani, C. Sexuality in premature ovarian insufficiency. Climacteric 2019, 22, 289–295. [Google Scholar] [CrossRef] [PubMed]
| Patient No. | Age (Years) | Family History | Menopause Age (Years) | Married for (Years) | BMI (kg) | Mood Disturbance | Sleep Cycle Changing | Dyspareunia Due to Atrophic Vaginitis | Rule Out PCOS | Other Clinical Symptoms |
|---|---|---|---|---|---|---|---|---|---|---|
| POF-1 | 25 | No | 23 | 7 | 24 | Yes | No | Yes | Yes | Secondary infertility |
| POF-2 | 27 | Yes | 25 | 8 | 31.9 | No | No | No | Yes | Primary infertility |
| POF-3 | 30 | No | 29 | 15 | 27 | Yes | No | No | Yes | Infertility |
| POF-4 | 40 | No | 38 | 2 | 23.4 | No | No | No | Yes | Infertility |
| POF-5 | 35 | Yes | 32 | 12 | 31.56 | No | No | No | Yes | Infertility |
| POF-6 | 27 | No | 25 | 6 | 24.4 | Yes | Yes | Yes | Yes | Infertility and irregular last menstrual period |
| POF-7 | 29 | Yes | 26 | 8 | 24 | Yes | No | Yes | Yes | Infertility |
| POF-8 | 36 | No | 35 | 10 | 31 | Yes | Yes | Yes | Yes | Irregular cycle, infertility, and hypothyroidism |
| POF-9 | 35 | Yes | 33 | 14 | 24 | Yes | Yes | Yes | Yes | Secondary infertility |
| POF-10 | 33 | Yes | 30 | 6 | 23 | Yes | Yes | Yes | Yes | Infertility |
| Patient No. | Estradiol (pg/mL) | PRL (mU/L) | TSH (mlU/L) | FSH (lU/L) | LH (lU/L) | AMHng/mL | Pelvic Ultrasound |
|---|---|---|---|---|---|---|---|
| POF-1 | 40 | 106 | 4.6 | 77 | 15.38 | Not performed | Normal uterus with poor ovarian reserves |
| POF-2 | 5 | 208.6 | 1.23 | 39.52 | 16.45 | Not performed | Normal |
| POF-3 | 136.2 | 217.24 | 0.991 | 49.64 | 38.46 | Not performed | Poor ovarian reserves |
| POF-4 | 16.51 | 173.8 | 1.79 | 54.51 | 21.3 | 0.010 | Poor ovaries reserves |
| POF-5 | 15.25 | 694.77 | 7.68 | 44.78 | 25 | Not performed | Normal ovaries |
| POF-6 | 18 | 227 | 4.5 | 60 | 13.18 | 0.05 | Normal ovaries |
| POF-7 | 16.31 | 753.42 | 3.566 | 20.49 | 70.15 | Not performed | Poor ovarian reserves |
| POF-8 | 47.13 | 248.4 | 5.19 | 12.94 | 3.43 | Not performed | Poor ovarian reserves |
| POF-9 | 13 | 53 | 2.07 | 16.1 | 2.11 | Not performed | Poor ovarian reserves |
| POF-10 | 19 | 1091 | 4.8 | 98 | 80 | Not performed | Poor ovarian reserves |
| Patient No. | POF01 | POF01 | POF03/04 | POF05 | POF06 | POF07 |
|---|---|---|---|---|---|---|
| Gene | HS6ST1 | HS6ST1 | BNC1 | MEIOB | GDF9 | MMRN1 |
| Chromosome | 2 | 2 | 15 | 16 | 5 | 4 |
| Nucleotide variant | c.261C>A | c.341T>G | c.2319C>A | c.520G>A | c.1121C>T | c.3578T>C |
| SNP | rs200979099 | rs199993343 | rs1453233120 | rs1025418947 | rs373477788 | rs201798101 |
| Protein variant | p.Asp87Glu | p.Val114Gly | p.Asn773Lys | p.Val174Met | p.Pro374Leu | p.Leu1193Ser |
| Zygosity | Homo | Homo | Hetero | Homo | Homo | Homo |
| Exon | 1 | 1 | 5 | 6 | 2 | 8 |
| gnomAD Freq | 0.00941 | 0.0018 | 0.00000657 | 0.0000197 | 0.0000591 | 0.0000723 |
| ACMG classification | VUS | VUS | VUS | VUS | VUS | VUS |
| Prediction bioinformatic tools | ||||||
| SIFT | P | BM | P | U | P | U |
| Revel | P | U | B | U | P | P |
| PROVEAN | U | BM | U | U | PM | B |
| Mutation Taster | U | U | B | U | VUS | BM |
| CADD | 25 | 21 | 2.584 | 26.3 | 27.4 | 27 |
| Mutation assessor | P | BM | U | U | P | P |
| FATHMM | U | BM | B | BM | VUS | U |
| DANN | U | U | U | U | U | U |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Almatrafi, A.M.; Hibshi, A.M.; Basit, S. Exome Sequencing to Identify Novel Variants Associated with Secondary Amenorrhea and Premature Ovarian Insufficiency (POI) in Saudi Women. Biomedicines 2024, 12, 785. https://doi.org/10.3390/biomedicines12040785
Almatrafi AM, Hibshi AM, Basit S. Exome Sequencing to Identify Novel Variants Associated with Secondary Amenorrhea and Premature Ovarian Insufficiency (POI) in Saudi Women. Biomedicines. 2024; 12(4):785. https://doi.org/10.3390/biomedicines12040785
Chicago/Turabian StyleAlmatrafi, Ahmed M., Ali M. Hibshi, and Sulman Basit. 2024. "Exome Sequencing to Identify Novel Variants Associated with Secondary Amenorrhea and Premature Ovarian Insufficiency (POI) in Saudi Women" Biomedicines 12, no. 4: 785. https://doi.org/10.3390/biomedicines12040785
APA StyleAlmatrafi, A. M., Hibshi, A. M., & Basit, S. (2024). Exome Sequencing to Identify Novel Variants Associated with Secondary Amenorrhea and Premature Ovarian Insufficiency (POI) in Saudi Women. Biomedicines, 12(4), 785. https://doi.org/10.3390/biomedicines12040785






