Aggressive Primary Thyroid Mucoepidermoid Carcinoma with Extensive Pulmonary Involvement
Abstract
1. Introduction
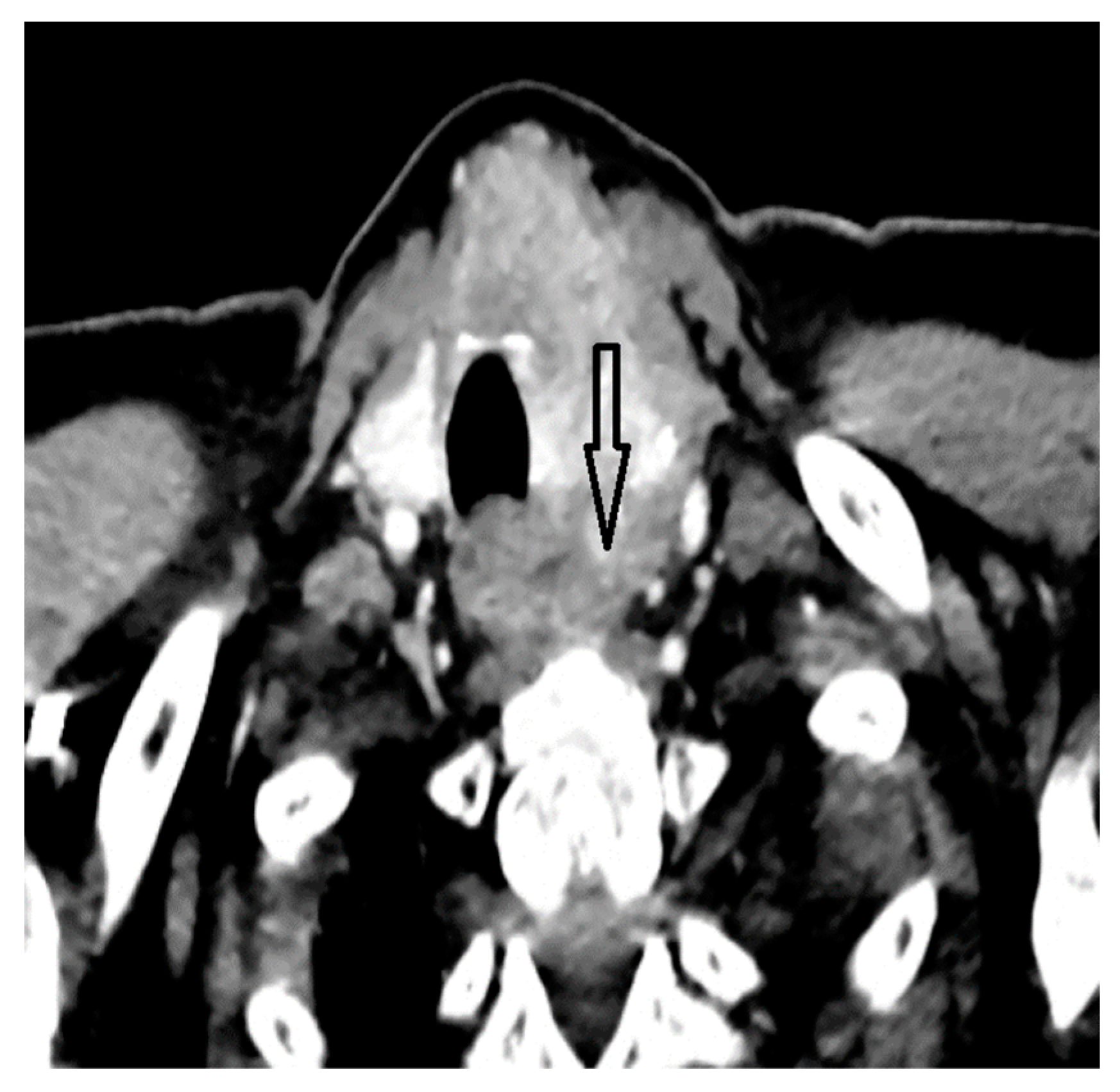
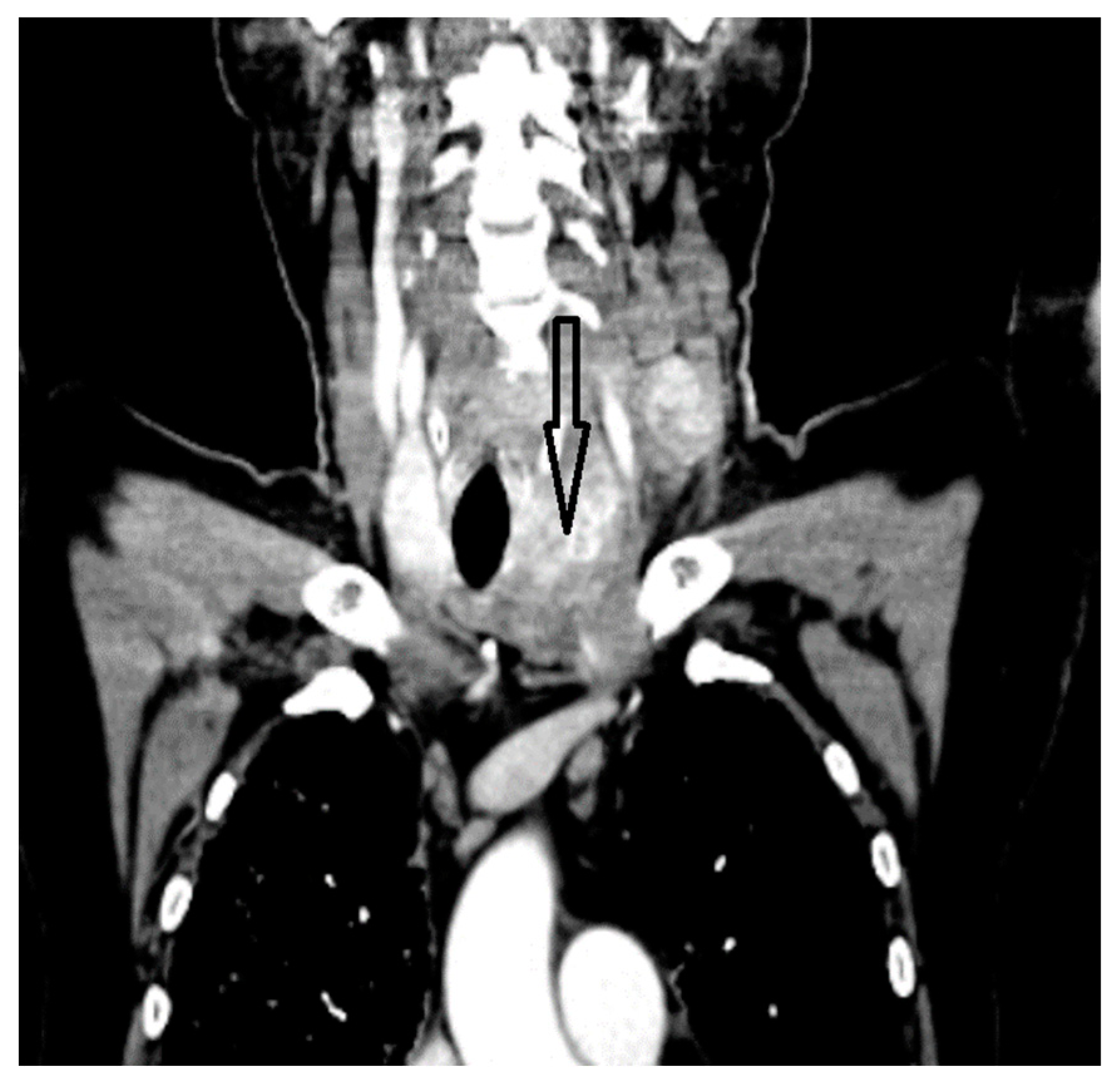
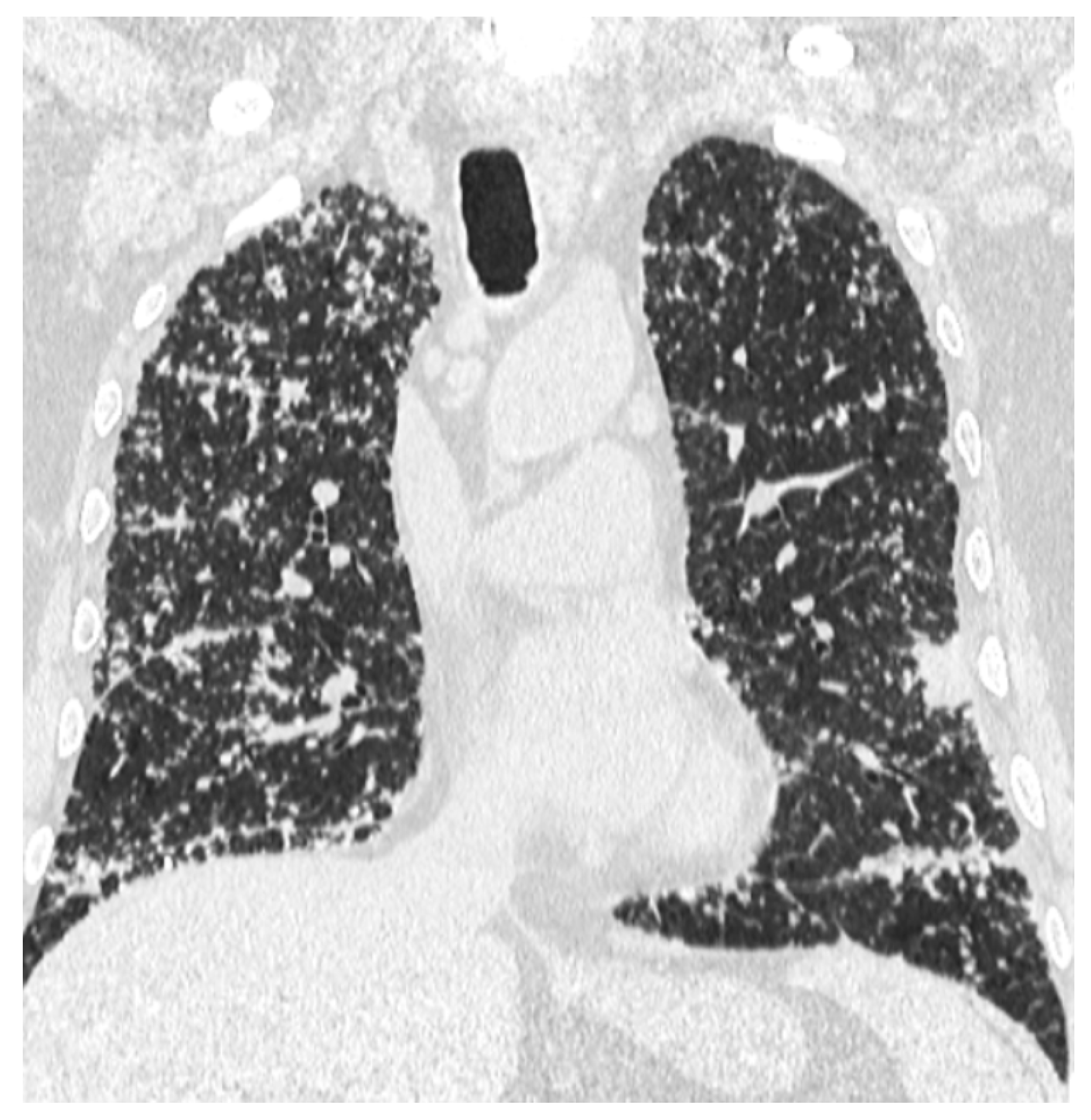
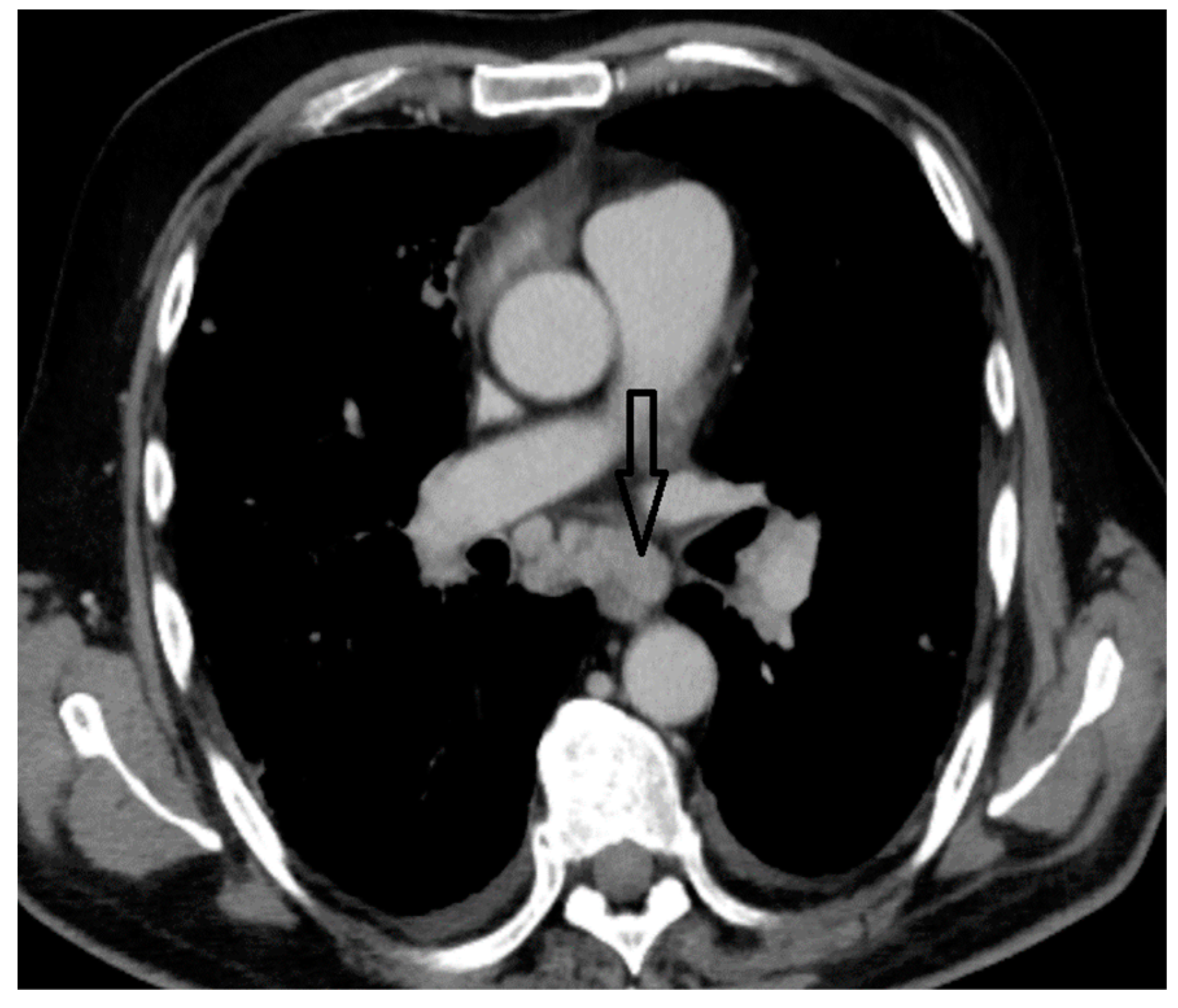
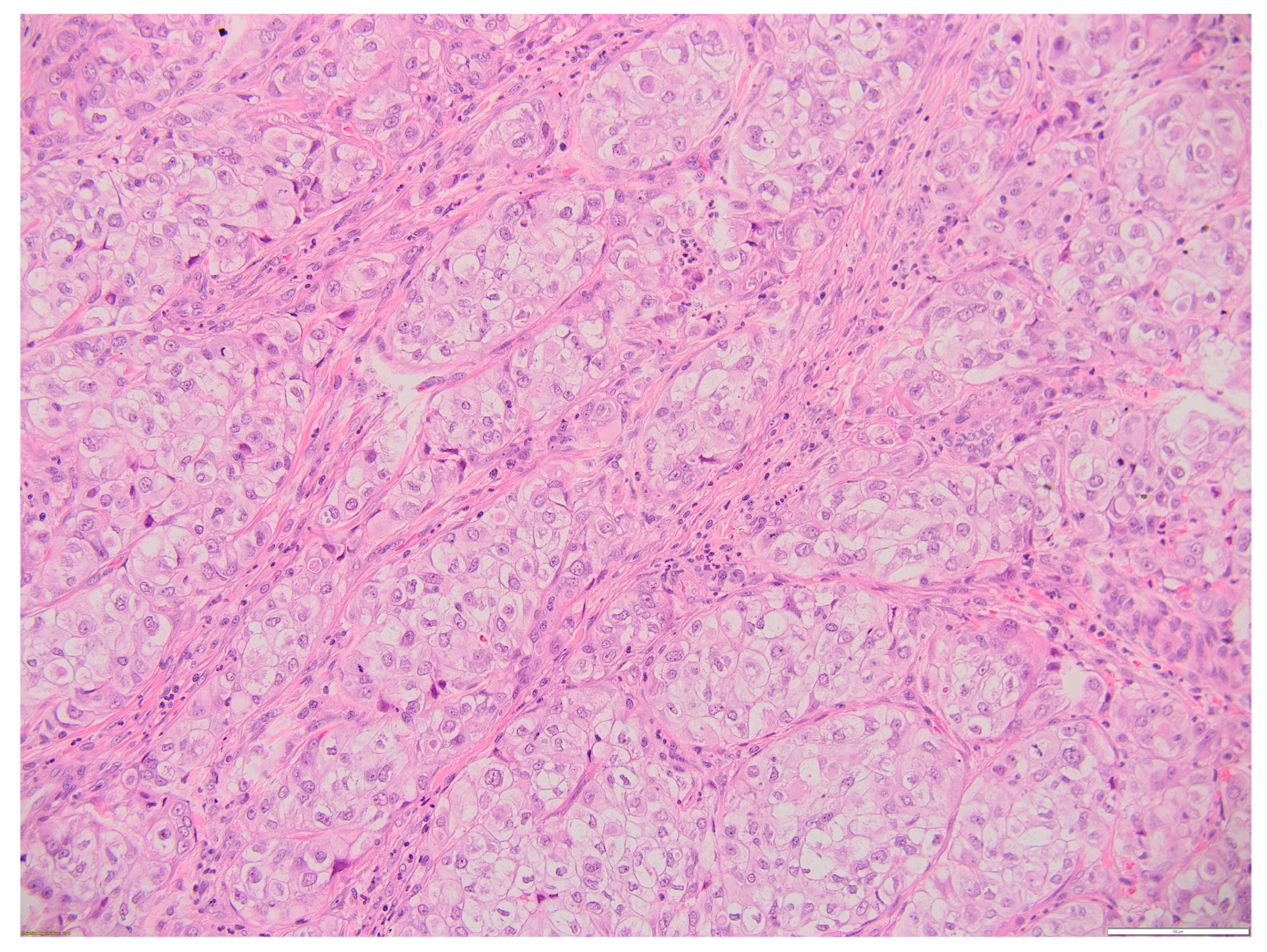
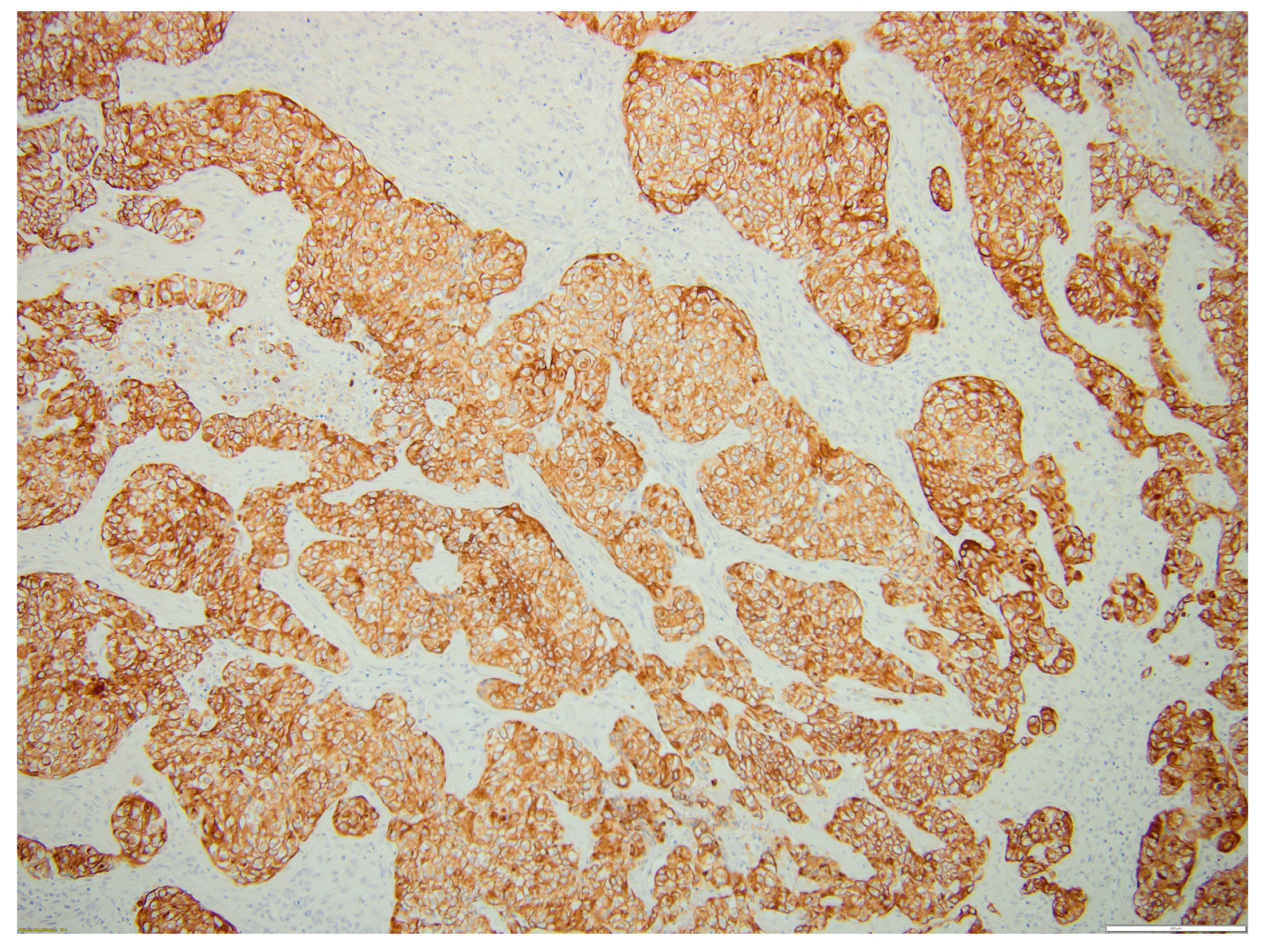
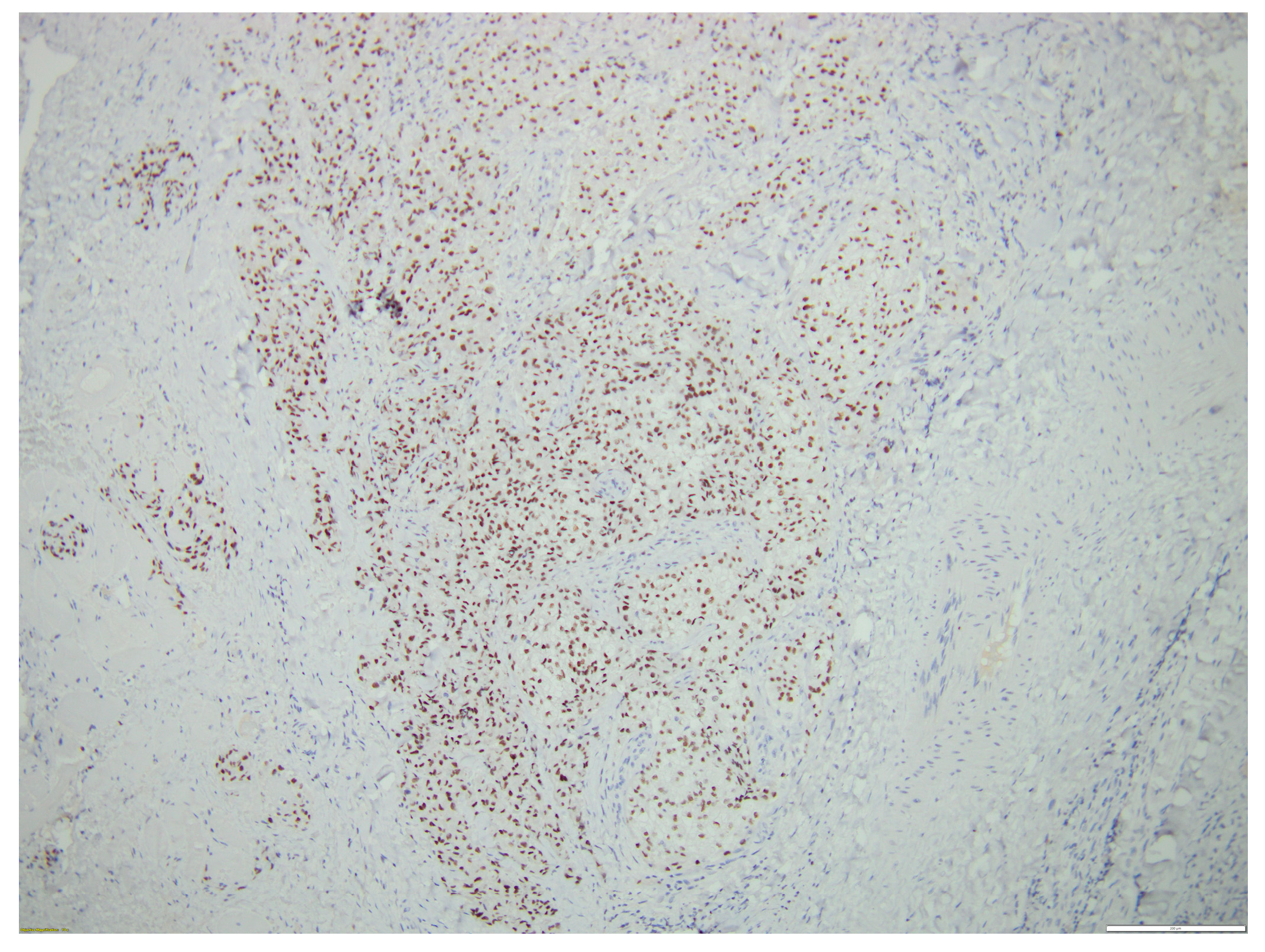
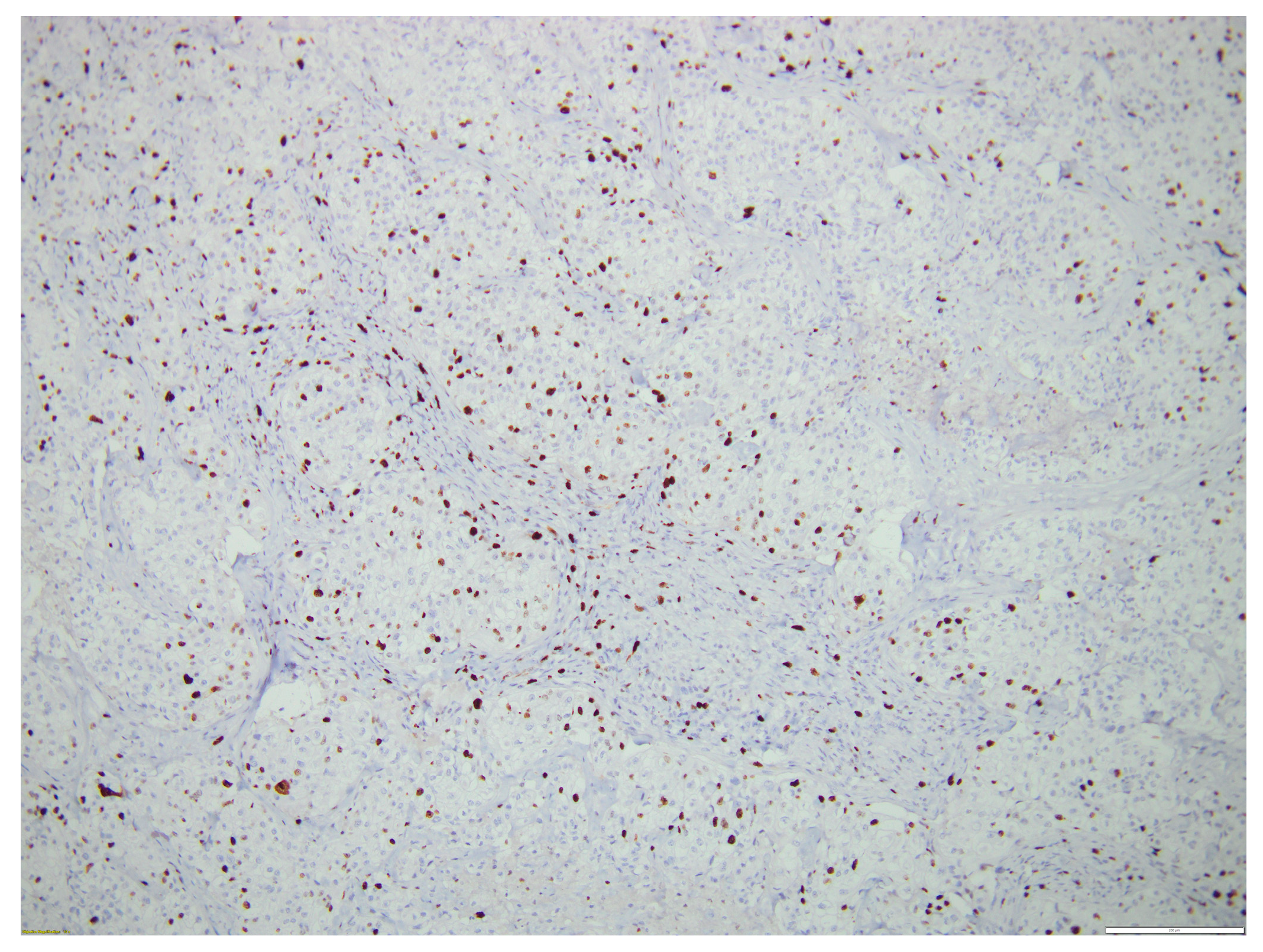
2. Case Report
3. Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics, 2019. CA Cancer J. Clin. 2019, 69, 7–34. [Google Scholar] [CrossRef] [PubMed]
- Cabanillas, M.E.; McFadden, D.G.; Durante, C. Thyroid cancer. Lancet 2016, 388, 2783–2795. [Google Scholar] [CrossRef] [PubMed]
- Speight, P.M.; Barrett, A.W. Salivary gland tumours. Oral. Dis. 2002, 8, 229–240. [Google Scholar] [CrossRef] [PubMed]
- Hastrup, N.; Sehested, M. High-grade mucoepidermoid carcinoma of the breast. Histopathology 1985, 9, 887–892. [Google Scholar] [CrossRef] [PubMed]
- Ohtsuki, Y.; Yoshino, T.; Takahashi, K.; Sonobe, H.; Kohno, K.; Akagi, T. Electron microscopic study of mucoepidermoid carcinoma in the pancreas. Acta Pathol. Jpn. 1987, 37, 1175–1182. [Google Scholar] [CrossRef]
- Kay, S. Mucoepidermoid carcinoma of the esophagus. Report of two cases. Cancer 1968, 22, 1053–1059. [Google Scholar] [CrossRef]
- Green, L.K.; Gallion, T.L.; Gyorkey, F. Peripheral mucoepidermoid tumour of the lung. Thorax 1991, 46, 65–66. [Google Scholar] [CrossRef][Green Version]
- Rhatigan, R.M.; Roque, J.L.; Bucher, R.L. Mucoepidermoid carcinoma of the thyroid gland. Cancer 1977, 39, 210–214. [Google Scholar] [CrossRef] [PubMed]
- Franssila, K.O.; Harach, H.R.; Wasenius, V.M. Mucoepidermoid carcinoma of the thyroid. Histopathology 1984, 8, 847–860. [Google Scholar] [CrossRef] [PubMed]
- Christofer Juhlin, C.; Mete, O.; Baloch, Z.W. The 2022 WHO classification of thyroid tumors: Novel concepts in nomenclature and grading. Endocr Relat Cancer 2023, 30. [Google Scholar] [CrossRef]
- Bhandarkar, N.D.; Chan, J.; Strome, M. A rare case of mucoepidermoid carcinoma of the thyroid. Am. J. Otolaryngol. 2005, 26, 138–141. [Google Scholar] [CrossRef]
- Farhat, N.A.; Faquin, W.C.; Sadow, P.M. Primary mucoepidermoid carcinoma of the thyroid gland: A report of three cases and review of the literature. Endocr. Pathol. 2013, 24, 229–233. [Google Scholar] [CrossRef]
- Iyer, J.; Hariharan, A.; Cao, U.M.N.; Mai, C.T.T.; Wang, A.; Khayambashi, P.; Nguyen, B.H.; Safi, L.; Tran, S.D. An Overview on the Histogenesis and Morphogenesis of Salivary Gland Neoplasms and Evolving Diagnostic Approaches. Cancers 2021, 13, 3910. [Google Scholar] [CrossRef]
- Dardick, I.; Burford-Mason, A.P. Current status of histogenetic and morphogenetic concepts of salivary gland tumorigenesis. Crit. Rev. Oral. Biol. Med. 1993, 4, 639–677. [Google Scholar] [CrossRef]
- Sreeja, C.; Shahela, T.; Aesha, S.; Satish, M.K. Taxonomy of salivary gland neoplasm. J. Clin. Diagn. Res. 2014, 8, 291–293. [Google Scholar] [CrossRef]
- Jung, C.K.; Bychkov, A.; Kakudo, K. Update from the 2022 World Health Organization Classification of Thyroid Tumors: A Standardized Diagnostic Approach. Endocrinol. Metab. 2022, 37, 703–718. [Google Scholar] [CrossRef]
- Shah, A.A.; La Fortune, K.; Miller, C.; Mills, S.E.; Baloch, Z.; LiVolsi, V.; Dacic, S.; Mahaffey, A.L.; Nikiforova, M.; Nikiforov, Y.E.; et al. Thyroid sclerosing mucoepidermoid carcinoma with eosinophilia: A clinicopathologic and molecular analysis of a distinct entity. Mod. Pathol. 2017, 30, 329–339. [Google Scholar] [CrossRef]
- Kong, M.; Wang, B.; Wang, X.; Xu, L. A Case Report of Mucoepidermoid Carcinoma With Extensive Keratinization in Parotid: Expanding the Morphologic Spectrum. Int. J. Surg. Pathol. 2022, 30, 422–426. [Google Scholar] [CrossRef] [PubMed]
- Prichard, R.S.; Lee, J.C.; Gill, A.J.; Sywak, M.S.; Fingleton, L.; Robinson, B.G.; Sidhu, S.B.; Delbridge, L.W. Mucoepidermoid carcinoma of the thyroid: A report of three cases and postulated histogenesis. Thyroid 2012, 22, 205–209. [Google Scholar] [CrossRef] [PubMed]
- Shindo, K.; Aishima, S.; Okido, M.; Ohshima, A. A poor prognostic case of mucoepidermoid carcinoma of the thyroid: A case report. Case Rep. Endocrinol. 2012, 2012, 862545. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.; Mirza, O.; Dobbs, S.; Jayaram, S. Poorly differentiated mucoepidermoid carcinoma of the thyroid. BMJ Case Rep. 2020, 13, e236539. [Google Scholar] [CrossRef] [PubMed]
- Le, Q.V.; Ngo, D.Q.; Ngo, Q.X. Primary Mucoepidermoid Carcinoma of the Thyroid: A Report of a Rare Case with Bone Metastasis and Review of the Literature. Case Rep. Oncol. 2019, 12, 248–259. [Google Scholar] [CrossRef]
- Yoshimura, T.; Higashi, S.; Yamada, S.; Noguchi, H.; Nomoto, M.; Suzuki, H.; Ishida, T.; Takayama, H.; Hirano, Y.; Yamashita, M.; et al. PCP4/PEP19 and HER2 Are Novel Prognostic Markers in Mucoepidermoid Carcinoma of the Salivary Gland. Cancers 2021, 14, 54. [Google Scholar] [CrossRef]
- Egebjerg, K.; Harwood, C.D.; Woller, N.C.; Kristensen, C.A.; Mau-Sørensen, M. HER2 Positivity in Histological Subtypes of Salivary Gland Carcinoma: A Systematic Review and Meta-Analysis. Front. Oncol. 2021, 11, 693394. [Google Scholar] [CrossRef]
- Le, H.T.; Nguyen, T.P.X.; Hirokawa, M.; Katoh, R.; Mitsutake, N.; Matsuse, M.; Sako, A.; Kondo, T.; Vasan, N.; Kim, Y.M.; et al. Primary Thyroid Mucoepidermoid Carcinoma (MEC) Is Clinically, Prognostically, and Molecularly Different from Sclerosing MEC with Eosinophilia: A Multicenter and Integrated Study. Endocr. Pathol. 2023, 34, 100–111. [Google Scholar] [CrossRef] [PubMed]
- Butler, R.T.; Spector, M.E.; Thomas, D.; McDaniel, A.S.; McHugh, J.B. An immunohistochemical panel for reliable differentiation of salivary duct carcinoma and mucoepidermoid carcinoma. Head. Neck Pathol. 2014, 8, 133–140. [Google Scholar] [CrossRef] [PubMed]
- Chan, J.K.; Albores-Saavedra, J.; Battifora, H.; Carcangiu, M.L.; Rosai, J. Sclerosing mucoepidermoid thyroid carcinoma with eosinophilia. A distinctive low-grade malignancy arising from the metaplastic follicles of Hashimoto’s thyroiditis. Am. J. Surg. Pathol. 1991, 15, 438–448. [Google Scholar] [CrossRef]
- Ames, E.; Campbell, M.J.; Afify, A.; Krane, J.F.; Huang, E.C. Sclerosing mucoepidermoid carcinoma with eosinophilia: Cytologic characterization of a rare distinct entity in the thyroid. Diagn. Cytopathol. 2018, 46, 632–635. [Google Scholar] [CrossRef]
- Lai, C.Y.; Chao, T.C.; Lin, J.D.; Hsueh, C. Sclerosing mucoepidermoid carcinoma with eosinophilia of thyroid gland in a male patient: A case report and literature review. Int. J. Clin. Exp. Pathol. 2015, 8, 5947–5951. [Google Scholar]
- Hirokawa, M.; Takada, N.; Abe, H.; Suzuki, A.; Higuchi, M.; Miya, A.; Hayashi, T.; Fukushima, M.; Kawahara, A.; Miyauchi, A. Thyroid sclerosing mucoepidermoid carcinoma with eosinophilia distinct from the salivary type. Endocr. J. 2018, 65, 427–436. [Google Scholar] [CrossRef]
- Wiles, A.B.; Kraft, A.O.; Mueller, S.M.; Powers, C.N. Sclerosing mucoepidermoid carcinoma with eosinophilia of the thyroid: Case report of a rare lesion with novel genetic mutation. Diagn. Cytopathol. 2019, 47, 589–593. [Google Scholar] [CrossRef] [PubMed]
- Quiroga-Garza, G.; Lee, J.H.; El-Naggar, A.; Black, J.O.; Amrikachi, M.; Zhai, Q.J.; Ayala, A.G.; Ro, J.Y. Sclerosing mucoepidermoid carcinoma with eosinophilia of the thyroid: More aggressive than previously reported. Hum. Pathol. 2015, 46, 725–731. [Google Scholar] [CrossRef] [PubMed]
- Shehadeh, N.J.; Vernick, J.; Lonardo, F.; Madan, S.K.; Jacobs, J.R.; Yoo, G.H.; Kim, H.E.; Ensley, J.F. Sclerosing mucoepidermoid carcinoma with eosinophilia of the thyroid: A case report and review of the literature. Am. J. Otolaryngol. 2004, 25, 48–53. [Google Scholar] [CrossRef] [PubMed]
- Raveendran Nair, A.K.A.; George, N.A.; Kumar, R.; Sreekumar, A.; Jayasree, K. Sclerosing mucoepidermoid carcinoma with eosinophilia of thyroid gland: Not so indolent a neoplasm? Indian. J. Pathol. Microbiol. 2018, 61, 242–244. [Google Scholar] [CrossRef]
- Skálová, A.; Vanecek, T.; Sima, R.; Laco, J.; Weinreb, I.; Perez-Ordonez, B.; Starek, I.; Geierova, M.; Simpson, R.H.; Passador-Santos, F.; et al. Mammary analogue secretory carcinoma of salivary glands, containing the ETV6-NTRK3 fusion gene: A hitherto undescribed salivary gland tumor entity. Am. J. Surg. Pathol. 2010, 34, 599–608. [Google Scholar] [CrossRef]
- Pittala, K.; Hall, S.; Huff, M.L.; Sheikh, H.; Wallace, S.J. Primary Cutaneous Secretory Carcinoma: A Case Report and Literature Review. Cureus 2023, 15, e34203. [Google Scholar] [CrossRef]
- Wang, J.; Xie, L.; Ma, L.; Miao, Y.; Guo, J.; Xu, E.; Yang, X.; Su, W.; Xi, Y. Two cases report of secretory carcinoma of the salivary gland in the lung: One primary and one metastatic after many years. Gland. Surg. 2022, 11, 2013–2020. [Google Scholar] [CrossRef]
- Desai, M.A.; Mehrad, M.; Ely, K.A.; Bishop, J.A.; Netterville, J.; Aulino, J.M.; Lewis, J.S. Secretory Carcinoma of the Thyroid Gland: Report of a Highly Aggressive Case Clinically Mimicking Undifferentiated Carcinoma and Review of the Literature. Head. Neck Pathol. 2019, 13, 562–572. [Google Scholar] [CrossRef]
- Wu, E.Y.; Lebastchi, J.; Marqusee, E.; Lorch, J.H.; Krane, J.F.; Barletta, J.A. A case of primary secretory carcinoma of the thyroid with high-grade features. Histopathology 2017, 71, 665–669. [Google Scholar] [CrossRef]
- Rodríguez-Urrego, P.A.; Dogan, S.; Lin, O. Cytologic findings of mammary analogue secretory carcinoma arising in the thyroid. Diagn. Cytopathol. 2017, 45, 552–556. [Google Scholar] [CrossRef]
- Dogan, S.; Wang, L.; Ptashkin, R.N.; Dawson, R.R.; Shah, J.P.; Sherman, E.J.; Michael Tuttle, R.; Fagin, J.A.; Klimstra, D.S.; Katabi, N.; et al. Mammary analog secretory carcinoma of the thyroid gland: A primary thyroid adenocarcinoma harboring ETV6-NTRK3 fusion. Mod. Pathol. 2016, 29, 985–995. [Google Scholar] [CrossRef] [PubMed]
- Reynolds, S.; Shaheen, M.; Olson, G.; Barry, M.; Wu, J.; Bocklage, T. A Case of Primary Mammary Analog Secretory Carcinoma (MASC) of the Thyroid Masquerading as Papillary Thyroid Carcinoma: Potentially More than a One Off. Head. Neck Pathol. 2016, 10, 405–413. [Google Scholar] [CrossRef]
- Liao, H.; Khan, A.; Miron, P.M.; Cornejo, K.M. Mammary Analogue Secretory Carcinoma of the Thyroid Mimicking Locally Advanced Papillary Thyroid Carcinoma: A Rare Case Report. Int. J. Surg. Pathol. 2018, 26, 459–463. [Google Scholar] [CrossRef]
- Wakely, P.E. Mucoepidermoid carcinoma: Analysis of 76 cytologic cases and correlation with histologic grade. Cancer Cytopathol. 2022, 130, 783–799. [Google Scholar] [CrossRef] [PubMed]
- Cameselle-Teijeiro, J.; Febles-Pérez, C.; Sobrinho-Simões, M. Cytologic features of fine needle aspirates of papillary and mucoepidermoid carcinoma of the thyroid with anaplastic transformation. A case report. Acta Cytol. 1997, 41, 1356–1360. [Google Scholar] [CrossRef]
- Larson, R.S.; Wick, M.R. Primary mucoepidermoid carcinoma of the thyroid: Diagnosis by fine-needle aspiration biopsy. Diagn. Cytopathol. 1993, 9, 438–443. [Google Scholar] [CrossRef]
- Minagawa, A.; Iitaka, M.; Suzuki, M.; Yasuda, S.; Kameyama, K.; Shimada, S.; Kitahama, S.; Wada, S.; Katayama, S. A case of primary mucoepidermoid carcinoma of the thyroid: Molecular evidence of its origin. Clin. Endocrinol. 2002, 57, 551–556. [Google Scholar] [CrossRef]
- Katoh, R.; Sugai, T.; Ono, S.; Takayama, K.; Tomichi, N.; Kurihara, H.; Takamatsu, M. Mucoepidermoid carcinoma of the thyroid gland. Cancer 1990, 65, 2020–2027. [Google Scholar] [CrossRef]
- Baloch, Z.W.; Solomon, A.C.; LiVolsi, V.A. Primary mucoepidermoid carcinoma and sclerosing mucoepidermoid carcinoma with eosinophilia of the thyroid gland: A report of nine cases. Mod. Pathol. 2000, 13, 802–807. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mitrache, M.; Terzea, D.; Sirbu, A.; Fica, S. Aggressive Primary Thyroid Mucoepidermoid Carcinoma with Extensive Pulmonary Involvement. Biomedicines 2024, 12, 285. https://doi.org/10.3390/biomedicines12020285
Mitrache M, Terzea D, Sirbu A, Fica S. Aggressive Primary Thyroid Mucoepidermoid Carcinoma with Extensive Pulmonary Involvement. Biomedicines. 2024; 12(2):285. https://doi.org/10.3390/biomedicines12020285
Chicago/Turabian StyleMitrache, Marius, Dana Terzea, Anca Sirbu, and Simona Fica. 2024. "Aggressive Primary Thyroid Mucoepidermoid Carcinoma with Extensive Pulmonary Involvement" Biomedicines 12, no. 2: 285. https://doi.org/10.3390/biomedicines12020285
APA StyleMitrache, M., Terzea, D., Sirbu, A., & Fica, S. (2024). Aggressive Primary Thyroid Mucoepidermoid Carcinoma with Extensive Pulmonary Involvement. Biomedicines, 12(2), 285. https://doi.org/10.3390/biomedicines12020285





