1. Introduction
Citrus limon (L.) Burm. f. is a tree with evergreen leaves and yellow edible fruits from the family Rutaceae. In some languages, C. limon is known as lemon (English), Zitrone (German), le citron (French), limón (Spanish), and níngméng, 檸檬 (Chinese).
The main raw material of
C. limon is the fruit, particularly the essential oil and juice obtained from it. The
C. limon fruit stands out as having well-known nutritional properties, but it is worth remarking that its valuable biological activities are underestimated in modern phytotherapy and cosmetology [
1].
C. limon fruit juice (lemon juice) has traditionally been used as a remedy for scurvy before the discovery of vitamin C [
2]. This common use of
C. limon, known since ancient times, has nowadays been supported by numerous scientific studies. Other uses for lemon juice, known from traditional medicine, include treatment of high blood pressure, the common cold, and irregular menstruation. Moreover, the essential oil of
C. limon is a known remedy for coughs [
3,
4,
5].
In Romanian traditional medicine,
C. limon essential oil was administered on sugar for suppressing coughs [
3]. Aside from being rich in vitamin C, which assists in warding off infections, the juice is traditionally used to treat scurvy, sore throats, fevers, rheumatism, high blood pressure, and chest pain [
6].
In Trinidad, a mixture of lemon juice with alcohol or coconut oil has been used to treat fever, coughs in the common cold, and high blood pressure. Moreover, the juice or grated skin, mixed with molasses, has been used to remove excess water from the body, and the juice mixed with olive oil has been administered for womb infection and kidney stones [
4]. According to Indian traditional medicine,
C. limon juice can induce menstruation; the recommended dose for this is two teaspoons consumed twice a day [
5].
Currently, valuable scientific publications focus on the ever wider pharmacological actions of
C. limon fruit extract, juice and essential oil. They include studies of, for example, antibacterial, antifungal, anti-inflammatory, anticancer, hepatoregenerating and cardioprotective activities [
7,
8,
9,
10,
11].
The pharmacological potential of
C. limon is determined by its rich chemical composition. The most important group of secondary metabolites in the fruit includes flavonoids and also other compounds, such as phenolic acids, coumarins, carboxylic acids, aminoacids and vitamins. The main compounds of essential oil are monoterpenoids, especially D-limonene. These valuable chemical components are the reason for the important position of
C. limon in the food and cosmetics industries [
12,
13,
14].
The aim of this overview is a systematic review of scientific works and in-depth analyses of the latest investigations and promotions related to C. limon as a valuable plant species, important in pharmacy, cosmetology and the food industry. Additionally, relevant biotechnological investigations are presented.
2. The Genus Citrus
The genus
Citrus is one of the most important taxonomic subunits of the family
Rutaceae. Fruits produced by the species belonging to this genus are called ‘citrus’ in colloquial language, or citrus fruits. Citrus fruits are commonly known for their valuable nutritional, pharmaceutical and cosmetic properties. The genus
Citrus includes evergreen plants, shrubs or trees (from 3 to 15 m tall). Their leaves are leathery, ovoid or elliptical in shape. Some of them have spikes. The flowers grow individually in leaf axils. Each flower has five petals, white or reddish. The fruit is a hesperidium berry. The species belonging to the genus
Citrus occurs naturally in areas with a warm and mild climate, mainly in the Mediterranean region. They are usually sensitive to frost [
2].
One of the best known and most used species of the genus
Citrus is the lemon—
Citrus limon (L.) Burm. f. (Latin synonyms:
C. × limonia,
C. limonum). Other important species included in this taxonomic unit are:
Citrus aurantium ssp.
aurantium—bitter orange,
Citrus sinensis—Chinese orange,
Citrus reticulata—mandarin,
Citrus paradise—grapefruit,
Citrus bergamia—bergamot orange,
Citrus medica—citron, and many others. A team of scientists from the University of California (Oakland, California, USA) [
15] analyzed the origin of several species of the genus
Citrus, including
C. limon. They found that
C. limon was a plant that had formed as a result of the combination of two species—
C. aurantium and
C. medica. In the studies of scientists from Southwest University of China (Chongqing, China), the metabolite profiles of
C. limon, C. aurantium and
C. medica were evaluated using gas chromatography–mass spectrometry (GC-MS) and the partial least squares discriminant analysis (PLS-DA) score plot [
16]. They proved that
C. limon has a smaller distance between
C. aurantium and
C. medica in comparison with other
Citrus species. These studies demonstrated that
C. limon was likely a hybrid of
C. medica and
C. aurantium, as previously suspected [
16].
Botanical classification of the species of the genus
Citrus is very difficult due to the frequent formation of hybrids and the introduction of numerous cultivars through cross-pollination. Hybrids are produced to obtain fruit with valuable organoleptic and industrial properties, including seedless fruit, high juiciness, and the required taste. For older varieties, hybrids and cultivars, the latest molecular techniques are often needed to identify them.
C. limon, like many other prolific citrus species, gives rise to numerous varieties, cultivars and hybrids, which are presented in
Table 1 and
Table 2 acc. to [
17].
One of the oldest preserved botanical sources describing species of the genus
Citrus is the “Monograph on the Oranges of Wên-chou” (in Chinese: 記 嘉 桔 錄, “Citrus records of Ji Jia”) by Han Yanzhi from 1178 [
18,
19]. Other historical works describing the species bearing citrus fruits are “Nürnbergische Hesperides” from 1708 and “Traité du Citrus” from 1811. Historically, one of the best known classifications of citrus species is “Histoire Naturelle des Orangers” from 1818. The American botanist Walter Tennyson Swingle (1871–1952) had a particularly significant impact on the present-day taxonomy of the genus
Citrus. He is the author of as many as 95 botanical names of species of the genus
Citrus. Currently, the systematics of the species of the genus
Citrus are based on studies of molecular markers and other DNA analysis technologies still provide new information [
20].
4. C. limon Pharmacopoeial Monographs and Safety of Use
By cold-pressing the fresh outer parts of the
C. limon pericarp (Lat.
exocarpium), an essential oil is obtained—the lemon oil (lat.
Citrus limon aetheroleum,
Limonis aetheroleum,
Oleum Citri). The oil is colourless or yellow, and has a characteristic, strong lemon scent [
21]. It is considered a pharmacopoeial raw material. Its monographs, entitled ‘
Limonis aetheroleum’, are present in the European Pharmacopoeia 9th [
22], American Pharmacopoeia [
23], and in the Ayurvedic Pharmacopoeia of India [
24].
Another pharmacopoeial raw material obtained from
C. limon is the outer part of the
mesocarp—the
flavedo. A monograph entitled ‘
Citrus limon flavedo’ can be found in older editions of the French Pharmacopoeia, for example, in its 10th edition from 1998 [
25].
The fresh fruit of
C. limon is officially listed for use in phytotherapy and in homeopathy in Germany. According to the German Commission D Monographs for homeopathic medicines,
C. limon fresh fruits can be used for treating gingival bleeding and debilitating diseases [
26].
C. limon also has a positive recommendation in the European Commission’s Cosmetics Ingredients Database (CosIng Database) as a valuable plant for cosmetics’ production [
27].
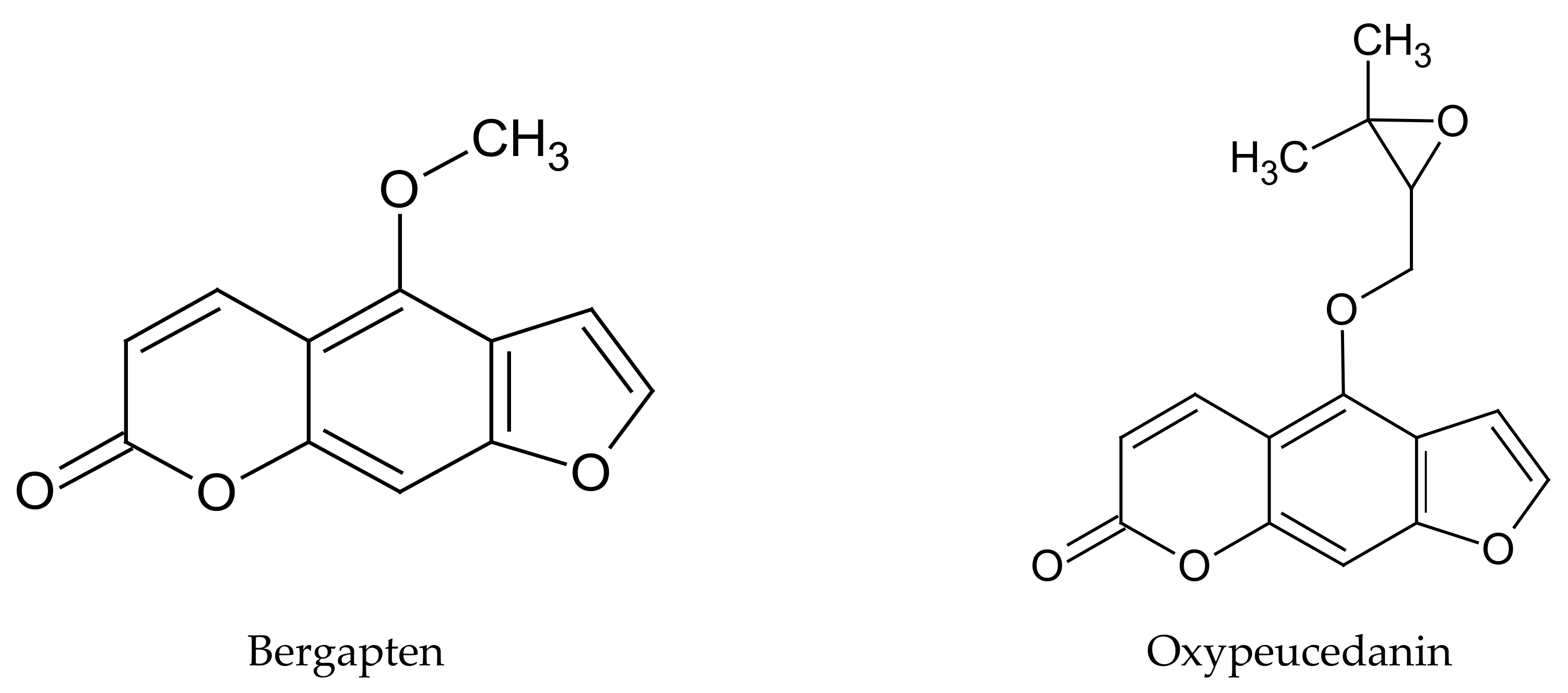
The European Food Safety Authority (EFSA) classified the pericarp, fruit, and leaves of
C. limon as raw materials of plant origin, in which there is presence of naturally occurring ingredients that may pose a threat to human health when used in the production of food and dietary supplements. EFSA has remarked that the toxic substances in these raw materials are photosensitizing compounds belonging to the furanocoumarin group, including bergapten and oxypeucedanin (
Figure 1) [
28].
In the American Food and Drug Administration (FDA) list,
C. limon essential oil and extracts are classified as safe products [
29].
5. Chemical Composition of C. limon
The chemical composition of C. limon fruit is well known. It has not only been determined for the whole fruit but also separately for the pericarp, juice, pomace, and essential oil. The compositions of the leaves and the fatty oil extracted from C. limon seeds are also known. Due to the large number of C. limon varieties, cultivars and hybrids, various research centres undertake the task of analyzing the chemical composition of the raw materials obtained from them.
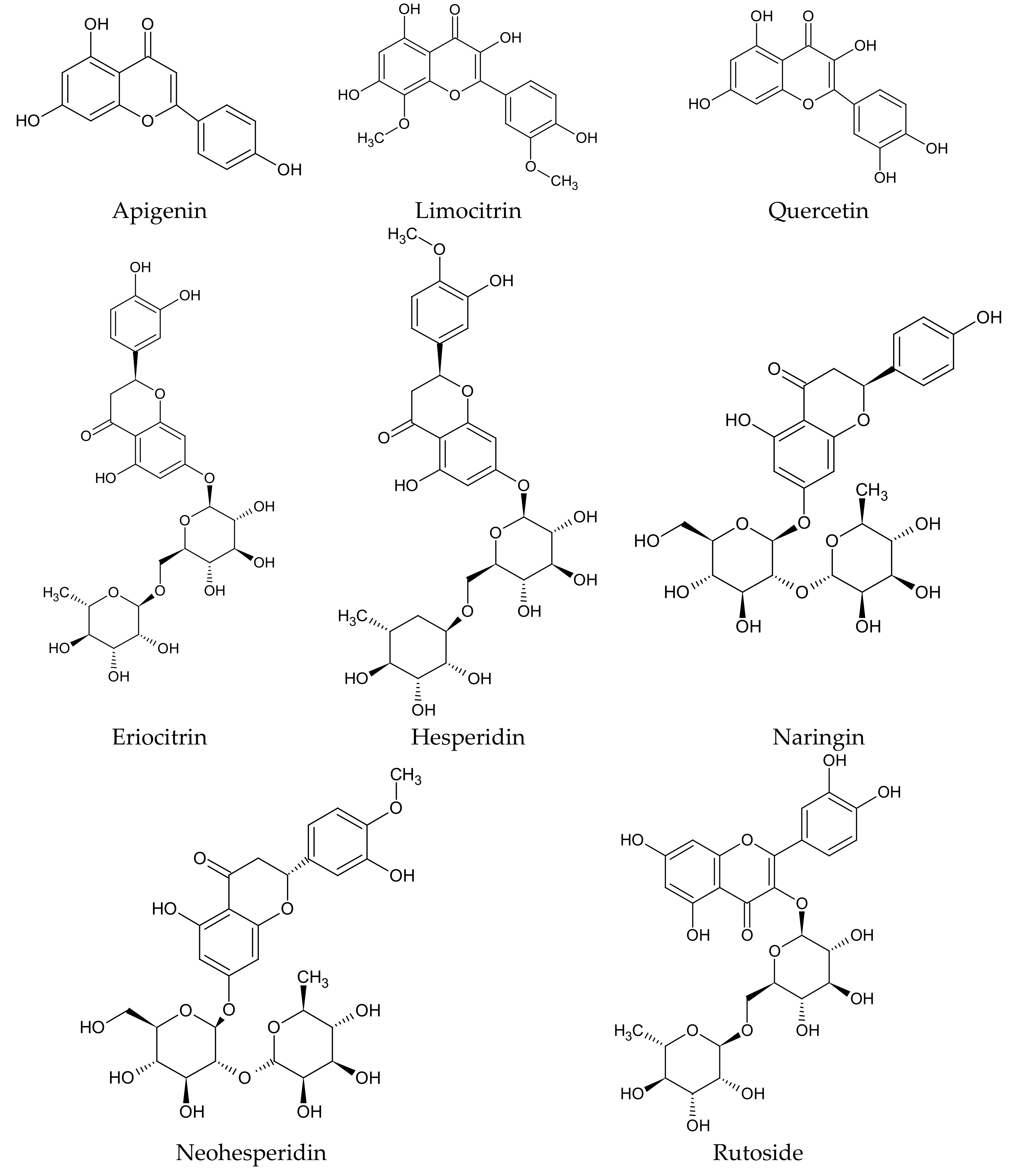
The most important group of bioactive compounds in both
C. limon fruit and its juice, determining their biological activity, are flavonoids such as: flavonones—eriodictyol, hesperidin, hesperetin, naringin; flavones—apigenin, diosmin; flavonols—quercetin; and their derivatives (
Figure 2). In the whole fruit, other flavonoids are additionally detected: flavonols—limocitrin (
Figure 2) and spinacetin, and flavones—orientin and vitexin (
Table 3 and
Table 4). Some flavonoids, such as neohesperidin, naringin and hesperidin (
Figure 2), are characteristic for
C. limon fruit. In comparison to another
Citrus species,
C. limon has the highest content of eriocitrin (
Figure 2) [
30].
Phenolic acids are another important group of compounds found both in the juice and fruit. There are mainly two such compounds in the juice—ferulic acid and synapic acid, and their derivatives. In contrast, the presence of p-hydroxybenzoic acid has been confirmed in the fruit. In the fruit, there are also coumarin compounds, carboxylic acids, carbohydrates, as well as amino acids, a complex of B vitamins, and, importantly, vitamin C (ascorbic acid) (
Table 3 and
Table 4) [
1,
12,
13,
31,
32,
33,
34,
35,
36].
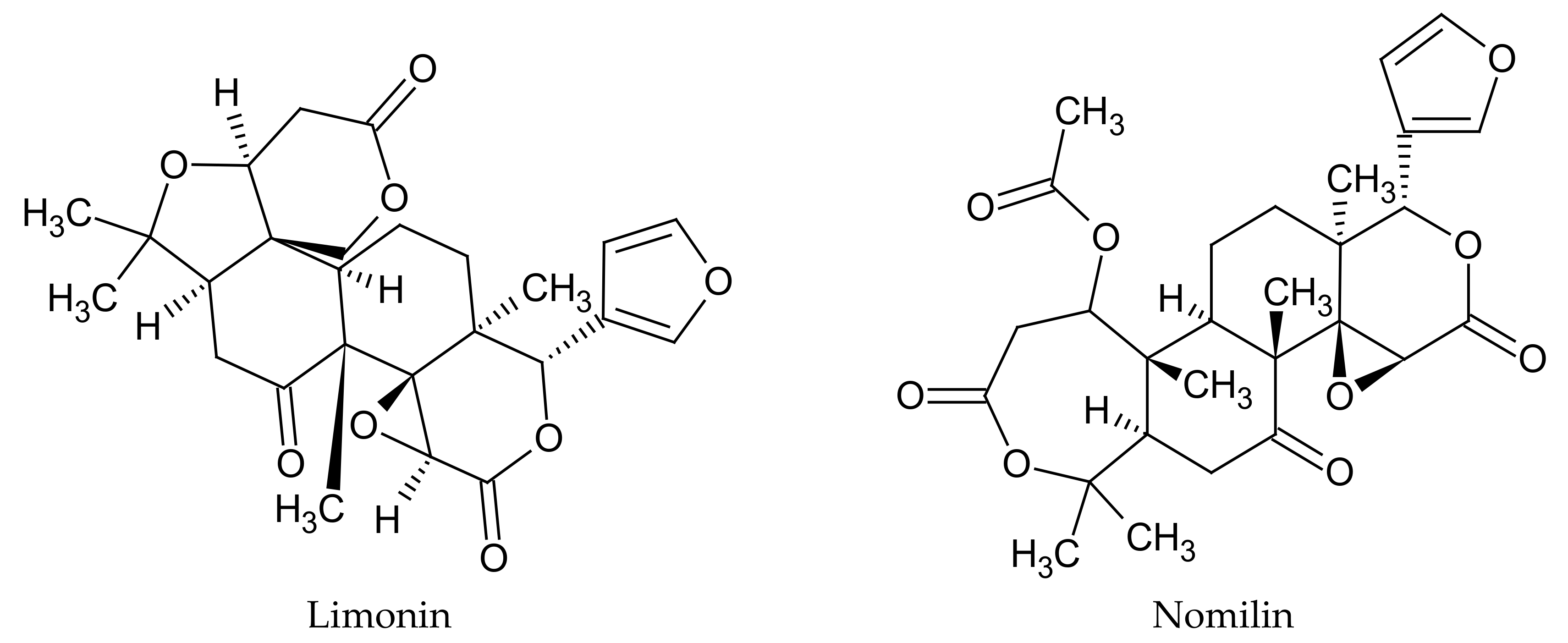
Another interesting group of compounds that are found in
C. limon fruits are limonoids. Limonoids are highly oxidized secondary metabolites with polycyclic triterpenoid backbones. They mainly occur in citrus fruits, including lemons, in which they are found mainly in the seeds, pulp, and peel. There are predominantly two such compounds in
C. limon fruits—limonin and nomilin (
Figure 3) [
37]. Studies have shown that the concentrations of the compounds of this group are dependent on fruit growth and maturation stages. Young citrus fruits contain the highest amounts of these compounds, compared to ripe ones [
38].
Analysis of macroelements in
C. limon fruit showed the presence in pulp and peel of: calcium (Ca), magnesium (Mg), phosphorus (P), potassium (K) and sodium (Na) [
36].
In
C. limon seed oil, the main ingredients are fatty acids, such as arachidonic acid, behenic acid and linoleic acid, and also tocopherols and carotenoids (
Table 5) [
33,
35]. The latest studies showed that
C. limon fruit pulp oil contains more fatty acids compared to other
Citrus species, such as
C. aurantium,
C. reticulata and
C. sinensis. The following fatty acids have been identified in
C. limon pulp oil: behenic acid, erucic acid, gondoic acid, lauric acid, linoleic acid, α-linolenic acid, margaric acid, palmitic acid, palmitoleic acid, pentadecanoic acid, and stearic acid [
39].
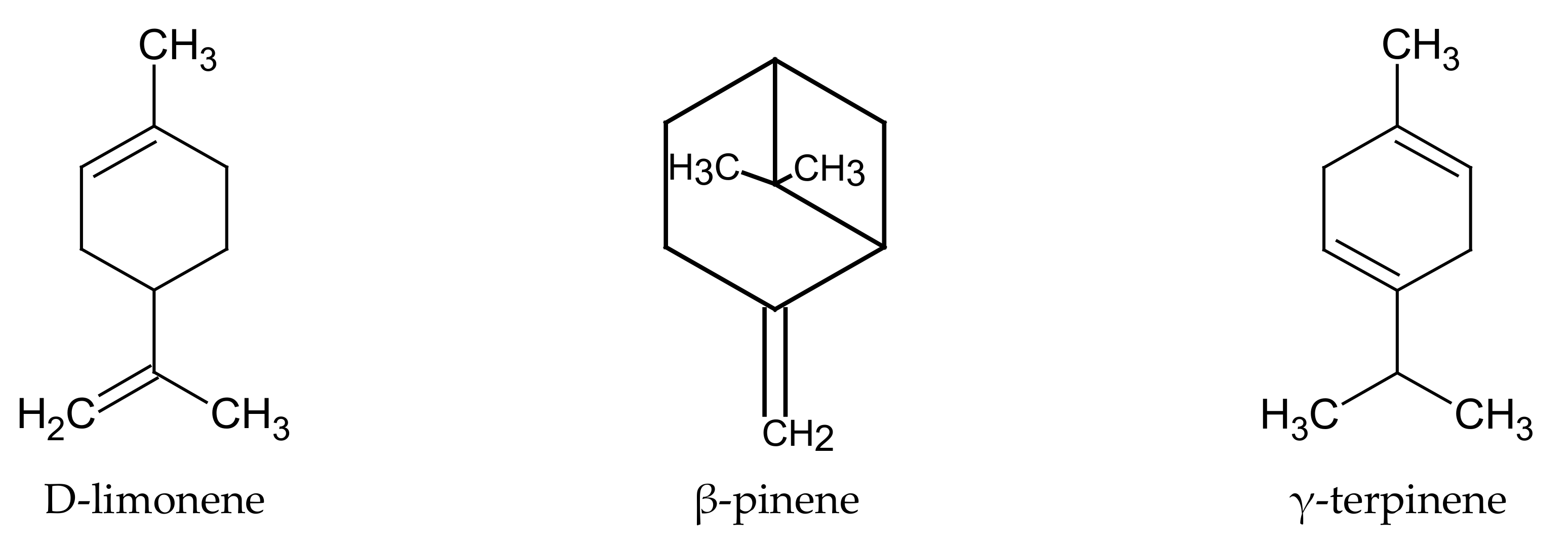
The main components of the
C. limon essential oil are monoterpenoids. Among them, quantitatively dominant in the essential oil obtained from pericarp are: limonene (69.9%), β-pinene (11.2%), γ-terpinene (8.21%), (
Figure 4), sabinene (3.9%), myrcene (3.1%), geranial (E-citral, 2.9%), neral (Z-citral, 1.5%), linalool (1.41%). In addition to terpenoids, the essential oil also contains linear furanocoumarins (psoralens) and polymethoxylated flavones (
Table 6) [
14,
40,
41].
The essential oil of the
C. limon leaf differs in composition from oil obtained from pericarp. Its main compounds include: limonene (31.5%), sabinene (15.9%), citronellal (11.6%), linalool (4.6%), neral (4.5%), geranial (4.5%), (E)-β-ocimene (3.9%), myrcene (2.9%), citronellol (2.3%), β-caryophyllene (1.7%), terpne-4-ol (1.4%), geraniol (1.3%) and α-pinene (1.2%) (
Table 6) [
14,
16,
40,
41,
42,
43].
6. Metabolomic Profile Studies
The team of Mucci et al. [
35] investigated the metabolic profile of different parts of
C. limon fruit. Flavedo, albedo, pulp, oil glands, and the seeds of lemon fruit and citron were studied through high resolution magic angle spinning nuclear magnetic resonance (HR-MAS NMR) spectroscopy. The analyses were made directly on intact tissues without any physicochemical manipulation. In
C. limon flavedo were detected: terpenoids (limonene, β-pinene and γ-terpinene), aminoacids (asparagine, arginine, glutamine, proline), organic acids (malic acid and quinic acid), osmolites (stachydrine), and fatty acid chains and sugars (glucose, fructose, β-fructofuranose, myoinositol, scylloinositol and sucrose) (
Table 3). The albedo of
C. limon fruit showed the presence of low signals from: aminoacids (alanine, threonine, valine, glutamine), sugars (glucose, sucrose, β-fructofuranose, myoinosytol, scylloinositol and β-fructopyranose), and osmolites (stachydrine, β-hydroxybutyrate, ethanol) (
Table 3). In albedo, clear signals from flavonoids were detected, such as hesperidin and rutoside, that have been identified also by high performance liquid chromatography (HPLC) analyses. Oil glands’ HR-MAS NMR composition analysis showed the presence of terpenoids (limonene, γ-terpinene, β-pinene, α-pinene, geranial, neral, citronellal, myrcene, sabinene, α-thujene, nerol and geraniol esters) and sugars (glucose, sucrose, β-fructofuranose and β-fructopyranose). The analysis of
C. limon pulp showed the presence of aminoacids (asparagine, proline, alanine, γ-aminobutyric acid (GABA), glutamine, threonine and valine), organic acids (citric acid and malic acid), sugars (glucose, sucrose, β-fructofuranose, β-fructopyranose, myoinosytol and scylloinosytol) and osmolites (stachydrine, ethanol and methanol) (
Table 3). HR-MAS NMR seeds analysis indicated that their composition is dominated by triglyceride signals (linoleic acid, linolenic acid and their derivatives), sugars (glucose and sucrose), osmolites (stachydrine) and trigonelline [
35].
In another metabolomic study, the peel extracts of ripened
C. limon fruit was characterized as containing nonfluorescent chlorophyll catabolites (NCCs) and dioxobilane-type nonfluorescent chlorophyll catabolite (DNCC) [
44]. In the peels of
C. limon fruit, four chlorophyll catabolites were detected: Cl-NCC1, Cl-NCC2, Cl-NCC3 and Cl-NCC4 [
44].
The metabolomic profile of
C. limon leaf was investigated by Asai et al. [
45]. The studies showed that
C. limon leaves contain 26 different organic acids and their derivatives (aconitic acid, 2-aminobutyric acid, 4-aminobutyric acid, ascorbic acid, benzoic acid, citramalic acid, citric acid, p-coumaric acid, ferulic acid, fumaric acid, glucaric acid, glycolic acid, 3-hydroxybutyric acid, 2-isopropylmalic acid, malic acid, malonic acid, 3-methylglutaric acid, oxamic acid, D-3-phenyllacetic acid, pipecolic acid, pyruvic acid, quinic acid, shikimic acid, succinic acid, threonic acid, urocanic acid), 21 aminoacids (alanine, γ-aminobutyric acid, anthranilic acid, asparagine, aspartic acid, glutamic acid, glutamine, glycine, histidine, isoleucine, leucine, lysine, methionine, phenylalanine, proline, pyroglutamic acid, serine, threonine, tryptophan, tyrosine, valine), and 13 sugars and sugar alcohols (arabinose, fructose, galactose, glucose, glycerol, inositol, lyxose, maltose, rhamnose, ribose, sorbose, sucrose, xylitol). Additionally, studied leaves have been exposed to stress conditions (leaves were placed in such a way that the edge of the petiole was in contact with the bottom of a glass bottle, soaked with 0.2 mM jasmonic acid and salicylic acid aqueous solutions, and incubated at 25 °C for 24 h). The content of aminoacids, such as, tyrosine, tryptophan, phenylalanine, valine, leucine, isoleucine, lysine, methionine, threonine, histidine, and γ-aminobutyric acid, was increased after this stress treatment [
45].
According to Mehl et al. [
46], the identification of volatile and non-volatile metabolites in
C. limon essential oil is dependent on geographic origin and the analytical methods used. To evaluate the potential of volatile and non-volatile fractions for classification purposes, volatile compounds of cold-pressed lemon oils were analyzed, using modern methods like gas chromatography-flame ionization detector-mass spectrometer (GC-FID/MS) and fourier transform mid-infrared spectroscopy (FT-MIR), while the non-volatile residues were studied using FT-MIR with proton nuclear magnetic resonance (
1H-NMR) and ultra-high performance liquid chromatography-quadrupole time-of-flight mass spectrometry (UHPLC-TOF-MS). The studies lead to very good differentiation and classification of samples regarding their geographic origin and extraction process modalities. The essential oil from the Italian-originated
C. limon fruit was enriched in α-thujene, α-pinene, α-terpinene, sesquiterpenoids (i.e., β-caryophyllene) and furocoumarins (i.e., bergamottin). The essential oil from Spanish and Argentinian
C. limon fruit was characterized by significant terpene contents, such as limonene, but differed in imperatorin, and byakangelicol contents. The studies showed that essential oil from Spanish
C. limon fruit contained more camphor and 4-terpineol, while Argentinian
C. limon fruit contained more sabinene and cis-sabinene hydrate [
46].
The studies performed by Jing et al. [
16] focused on the identification of components in the essential oil of different
Citrus species, including
C. limon. In general, most of the studied essential oil components were identified as monoterpenoids. The major monoterpenes in
C. limon essential oil were: limonene (70.37%), p-mentha-3,8-diene (18.00%), myrcene (4.40%), α-pinene (3.24%), α-thujene (1.05%) and terpinolene (0.90%) (
Table 6). Other monoterpenoids, which were identified as characteristic of
C. limon, were: sabinene (0.28%), α-terpinene (0.22%), trans-muurola-4(14), 5-diene (0.18%), eucalyptol (0.12%), octanol acetate (0.03%), β-curcumene (0.03%), zonarene (0.03%), 7-epi-sesquithujene (0.02%), citronellyl acetate (0.02%), α-farnesene (0.01%) (
Table 6). The shown metabolite-based profiling model can be used to clearly discriminate the basic
Citrus species. Limonene, α-pinene, sabinene and α-terpinene were the major characteristic components of the analyzed metabolomes of
Citrus genotypes that contributed to their taxonomy [
16].
Studies performed by Masson et al. [
43] deal with furanocoumarin’s and coumarin’s metabolomic profile in essential oil from
C. limon fruit peel.
C. limon essential oil contained large amounts of both furanocoumarins and coumarins compared to another tested
Citrus essential oils. In
C. limon essential oil, 13 furanocoumarins were detected (bergamottin, bergapten, byakangelicol, byakangelicin, epoxybergamottin, 8-geranyloxypsoralen, heraclenin, imperatorin, isoimperatorin, isopimpinellin, oxypeucedanin, oxypeucedanin hydrate, phellopterin) and two coumarins (citropten and herniarin) (
Table 6) [
43].
8. C. limon in the Food Industry
Due to the rich chemical composition of
C. limon fruit and other lemon-derived raw materials, they have applications in the food industry and in food processing. The lemon fruit is used mainly as a fresh fruit, but it is also processed to make juices, jams, jellies, molasses, etc. [
41]. Fresh lemon fruit can be kept for several months, maintaining their levels of juice, vitamins, minerals, fibre, and carbohydrates. The vitamin C (ascorbic acid) content in lemon fruits and juices decreases during storage and industrial processing. The factors lowering this content are: oxygen, heat, light, time, storage temperature and storage duration. To prevent the reduction in the ascorbic acid levels and antioxidant capacity of both the lemon fruit and lemon juice, they should be kept at 0–5 °C and protected from water loss by proper packaging, with high relative humidity during distribution. Under such conditions, lemon products show a good retention of vitamin C and antioxidant capacity [
41,
74].
C. limon peel is rich in pectin, which is used in a wide range of food industrial processes as a gelling agent, including the production of jams and jellies, and as thickener, texturizer, emulsifier and stabilizer in dairy products. Due to its jellifying properties, the pectin is also used in pharmaceutical, dental and cosmetic formulations [
75].
Lemon juice is used as an ingredient in beverages, particularly lemonade and soft drinks, and in other foods, such as salad dressings, sauces, and baked products. Lemon juice is a natural flavouring and preservative, and it is also used to add an acidic, or sour, taste to foods and soft drinks [
41,
76].
C. limon is the most suitable, being free from pesticide residues, raw material for enhancing the flavour of liqueurs, e.g., “limoncello”, the traditional liqueur of Sicily. It is made by the maceration of lemon peel in ethanol, water and sugar [
41,
76].
Currently, the essential oil from lemon, i.e., pure isolated linalol and citral, are used mainly as a flavouring and natural preservative due to their functional properties (antimicrobial, antifungal, etc.) [
52,
53]. In particular, they are often used to extend the short shelf-life of seafood products and in the production of some types of cheese because they significantly reduces populations of microorganisms, especially those from the family
Enterobacteriaceae [
41,
76].
9. Cosmetological Applications
C. limon fruit extracts and essential oil, as well as the active compounds isolated from these raw materials, have become the object of numerous scientific studies aimed at proving the possibility of their use in cosmetology. Lemon-derived products have long been credited with having a positive effect on acne-prone skin that is easily affected by sunburn or mycosis. In this regard, traditional uses of this raw materials are known in various parts of the world. In Tanzania, the fruit juice of
C. limon is mixed with egg albumin, honey and cucumber, and applied to the skin every day at night to smooth the facial skin and treat acne [
77]. Juice from freshly squeezed fruit of
C. limon mixed with olive oil is used as a natural remedy for the treatment of hair and scalp disorders in the West Bank in Palestine [
78]. Currently, knowledge of the cosmetic activity of
C. limon is constantly expanding.
C. limon essential oil shows antibiotic and flavouring properties, and for this reason it is used in formulations of shampoos, toothpaste, disinfectants, topical ointments and other cosmetics [
41].
Scientific studies have shown a significant antioxidant effect of
C. limon fruit extracts, which is the reason they are recommended for use in anti-ageing cosmetics [
8,
48]. The use of different carriers for
C. limon extracts (e.g., hyalurosomes, glycerosomes) in cosmetics production technology contributes to an even greater inhibition of oxidative stress in skin-building structures, including keratinocytes and fibroblasts (
Table 9) [
79]. In addition, vitamin C from
C. limon is used as an ingredient in specialized dermocosmetics. Its external use increases collagen production, which makes the skin smoother and more tense. It is used in anti-aging products, to reduce shallow wrinkles, and as a synergistic antioxidant in combination with vitamin E [
48].
The ingredients of
C. limon essential oil (including citral, β-pinene, D-limonene), due to the inhibiting activity of tyrosinase and the inhibition of L-dihydroxyphenylalanine (L-DOPA) oxidation, have a depigmenting effect [
80]. In addition, the essential oil has been proven to support the penetration of lipids as well as water-soluble vitamins. It can be used as a promoter of penetration of active substances through the skin [
81]. Moreover, besides the direct effect on the skin, the essential oil can also be used as a natural preservative and as a corrigent in cosmetic products. Studies have confirmed its antibacterial and fungistatic effects (
Table 9) [
7,
52,
53].
Furthermore,
C. limon pericarp extracts, too, exhibit scientifically proven activity that helps to accelerate the regeneration of diabetic wounds. In addition, the essential oil derived from
C. limon pericarp has shown anti-inflammatory, anti-allergic and slimming properties [
49,
59,
60,
62].
According to the CosIng Database (Cosmetic Ingredient Database),
C. limon can be used in twenty-three forms. It can be used in cosmetics in the form of oils obtained from various organs, in the form of extracts, hydrolates, powdered parts of the plant, wax and juice [
27]. The most common activity defined by CosIng for the raw material of this species is to keep the skin in good condition, to improve the odour of cosmetic products, and to mask the smell of other ingredients of cosmetic preparations [
27]. The approved forms of raw materials and their potential effects, as well as their use as corrigents, presented in the CosIng Database, are summarized in
Table 10 [
27].
C. limon essential oil has been used since the 18th century in the production of the famous ‘Eau de Cologne’. In aromatherapy, it is used to treat numerous diseases and lifestyle-related ailments: hypertension, neurosis, anxiety, varicose veins, arthritis, rheumatism and mental heaviness. It also alleviates symptoms characteristic of menopause.
C. limon essential oil is also used in aromatherapy massages to relax muscles, and for calming down and deep relaxation [
21].
C. limon fruit extracts and essential oil should not be used in high concentrations in baths or directly on the skin. Recent studies have shown that D-limonene contained in the oil has an allergenic and irritating effect on the skin. It may cause cross-allergy with balsam of Peru. After applying cosmetics containing
C. limon oil, it is forbidden to expose the skin to sunlight
. C. limon essential oil contains photosensitizing compounds belonging to the linear furanocoumarin group. The lemon pericarp contains: bergapten, phellopterin, 5- and 8-geranoxypsoralen, and the essential oil contains: bergapten, imperatorin, isopimpinellin, xanthotoxin, oxypeucedanin and psoralen [
21,
82].
The International Fragrance Association (IFRA) has issued restrictions on the use of
C. limon essential oil. In preparations remaining on the skin, the concentration of that oil should not exceed 2%. In addition,
C. limon essential oil should not be used in preparations remaining on skin exposed to UV rays. They should not contain more than 15 ppm of bergapten [
83].