Botany, Nutritional Value, Phytochemical Composition and Biological Activities of Quinoa
Abstract
:1. Introduction
Origin and Dissemination
2. Quinoa Botany and Morphological Characteristics
2.1. Leaves
2.2. Root
2.3. Stem
2.4. Inflorescences and Flowers
2.5. Fruit
3. Nutritional Profile
3.1. Protein
3.2. Lipids
3.3. Carbohydrates
3.4. Starch
3.5. Fiber
3.6. Minerals
3.7. Vitamins
4. Secondary Metabolites
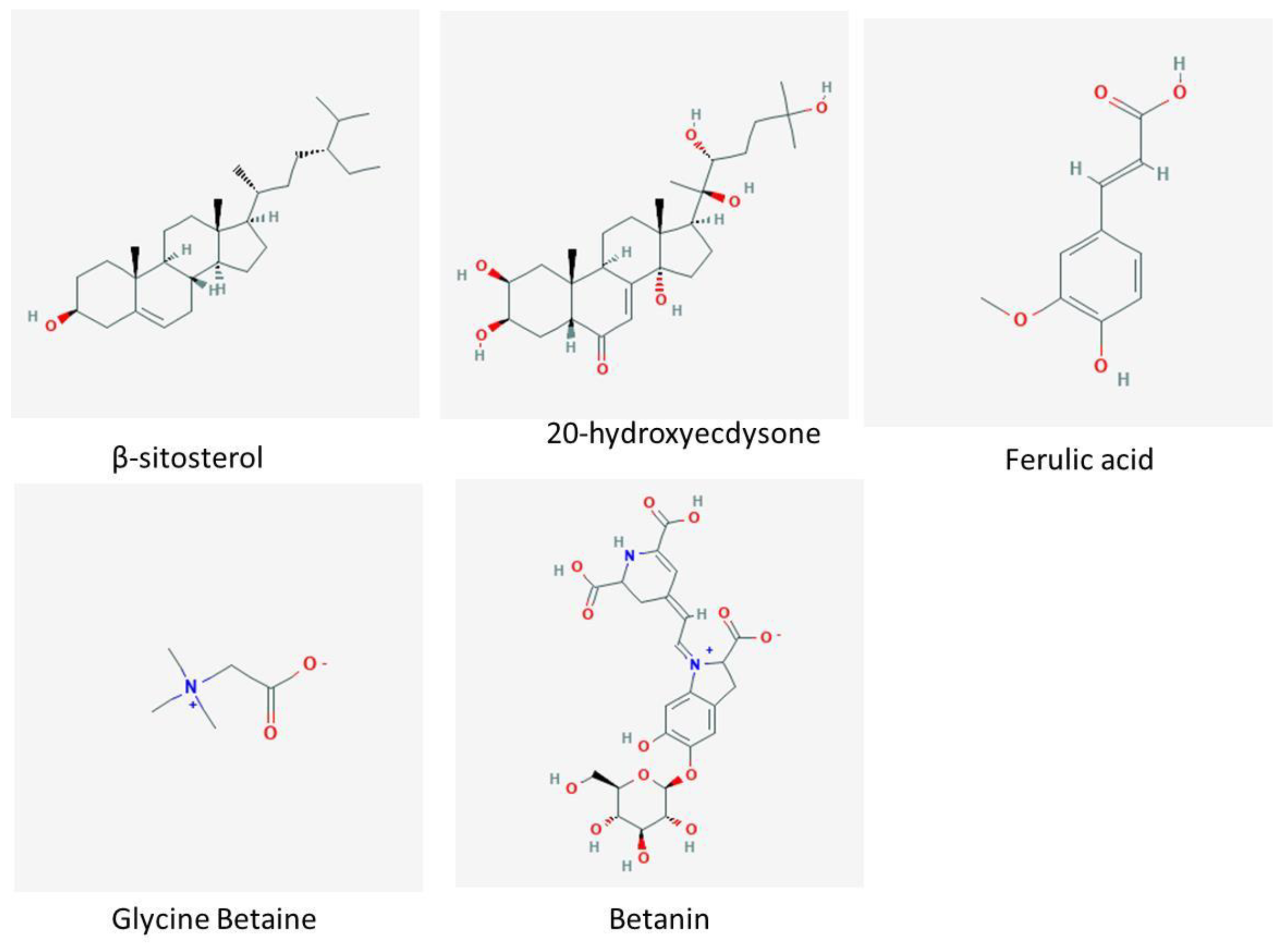
4.1. Phytoecdysteroids
4.2. Saponins
4.3. Phenolic Compounds
4.3.1. Phenolic Acids
4.3.2. Flavonoids
4.3.2.1. Flavonol Glycosides
4.3.2.2. Isoflavones
4.4. Betalains
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Maradini-Filho, A.M. Quinoa: Nutritional aspects. J. Nutrac. Food Sci. 2017, 2, 1–5. [Google Scholar]
- Vega-Galvez, A.; Miranda, M.; Vergara, J.; Uribe, E.; Puente, L.; Martinez, E.A. Nutrition facts and functional potential of quinoa (Chenopodium quinoa Willd.). an ancient Andean grain: A review. J. Sci. Food Agric. 2010, 90, 2541–2547. [Google Scholar] [CrossRef]
- National Research Council. Lost Crops of the Incas: Little-Known Plants of the Andes with Promise for Worldwide Cultivation; The National Academies Press: Washington, DC, USA, 1989. [Google Scholar] [CrossRef]
- Jacobsen, S.-E.; Mujica, A.; Jensen, C.R. The resistance of quinoa (Chenopodium quinoa Willd.) to adverse abiotic factors. Food Rev. Inter. 2003, 19, 99–109. [Google Scholar] [CrossRef]
- Saeidi, S.; Siadat, S.A.; Moshatati, A.; Moradi-Telavat, S.N. Effect of sowing time and nitrogen fertilizer rates on growth, seed yield and nitrogen use efficiency of quinoa (Chenopodium quinoa Willd) in Ahvaz. Iran. J. Crop Sci. 2020, 21, 354–367. [Google Scholar]
- Gamboa, C.; Schuster, M.; Schrevens, E.; Maertens, M. Price volatility and quinoa consumption among smallholder producers in the Andes. Sci. Agropecu. 2020, 11, 113–125. [Google Scholar] [CrossRef]
- Liu, M.; Zhu, K.; Yao, Y.; Chen, Y.; Guo, H.; Ren, G.; Yang, X.; Li, J. Antioxidant, anti-inflammatory, and antitumor activities of phenolic compounds from white, red, and black Chenopodium quinoa seed. Cereal Chem. 2020, 97, 703–713. [Google Scholar] [CrossRef]
- Prego, I.; Maldonado, S.; Otegui, M. Seed structure and localization of reserves in Chenopodium quinoa. Ann. Bot. 1998, 82, 481–488. [Google Scholar] [CrossRef] [Green Version]
- Ballester-Sánchez, J.; Gil, J.V.; Haros, C.M.; Fernández-Espinar, M.T. Effect of incorporating white, red or black quinoa flours on free and bound polyphenol content, antioxidant activity and colour of bread. Plant Foods Human Nutr. 2019, 74, 185–191. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bhargava, A.; Shukla, S.; Ohri, D. Chenopodium quinoa—An Indian perspective. Ind. Crops Prod. 2006, 23, 73–87. [Google Scholar] [CrossRef]
- Katwal, T.B.; Bazile, D. First adaptation of quinoa in the Bhutanese mountain agriculture systems. PLoS ONE 2020, 15, e0219804. [Google Scholar] [CrossRef] [Green Version]
- Sobota, A.; Świeca, M.; Gęsiński, K.; Wirkijowska, A.; Bochnak, J. Yellow-coated quinoa (Chenopodium quinoa Willd)—Physicochemical, nutritional, and antioxidant properties. J. Sci. Food Agric. 2020, 100, 2035–2042. [Google Scholar] [CrossRef]
- Stikić, R.I.; Milinčić, D.D.; Kostić, A.Ž.; Jovanović, Z.B.; Gašić, U.M.; Tešić, Ž.L.; Djordjević, N.Z.; Savić, S.K.; Czekus, B.G.; Pešić, M.B. Polyphenolic profiles, antioxidant, and in vitro anticancer activities of the seeds of Puno and Titicaca quinoa cultivars. Cereal Chem. 2020, 97, 626–633. [Google Scholar] [CrossRef]
- Tang, Y.; Li, X.; Chen, P.X.; Zhang, B.; Hernandez, M.; Zhang, H.; Marcone, M.F.; Liu, R.; Tsao, R. Characterisation of fatty acid, carotenoid, tocopherol/tocotrienol compositions and antioxidant activities in seeds of three Chenopodium quinoa Willd. genotypes. Food Chem. 2015, 174, 502–508. [Google Scholar] [CrossRef]
- Scalbert, A.; Manach, C.; Morand, C.; Remesy, C.; Jimenez, L. Dietary polyphenols and the prevention of diseases. Crit. Rev. Food Sci. Nutr. 2005, 45, 287–306. [Google Scholar] [CrossRef]
- Christensen, S.A.; Pratt, D.B.; Pratt, C.; Nelson, P.T.; Stevens, M.R.; Jellen, E.N.; Coleman, C.E.; Fairbanks, D.J.; Bonifacio, A.; Maughan, P.J. Assessment of genetic diversity in the USDA and CIP-FAO international nursery collections of quinoa (Chenopodium quinoa Willd.) using microsatellite markers. Plant Genetic Resour. 2007, 5, 82–95. [Google Scholar] [CrossRef] [Green Version]
- Mujica, A.; Jacobsen, S.E.; Ezquierdo, J.; Marathee, J.P. Resultados de la Prueba Americana y Europes de la Quinoa; FAO: Rome, Italy, 2001; p. 51. [Google Scholar]
- Mabry, T.J.; Taylor, A.; Turner, B.L. The betacyanins and their distribution. Phytochemistry 1963, 2, 61–64. [Google Scholar] [CrossRef]
- Hunziker, A. Las especies alimenticias de Amaranthus y Chenopodium cultivadas por los indios de América. Rev. Argent Agron. 1943, 10, 297–354. [Google Scholar]
- Risi, J.C.; Galwey, N.W. The pattern of genetic diversity in the Andean grain crop quinoa (Chenopodium quinoa Willd). I. Associations between characteristics. Euphytica 1989, 41, 147–162. [Google Scholar] [CrossRef]
- Ward, S.M.; Johnson, D.L. Cytoplasmic Male Sterile Quinoa. U.S. Patent US5304718A, 19 April 1994. [Google Scholar]
- Gandarillas, H. Botanica. In Quinoa Kaniwa; Tapia, M.E., Ed.; Cultivos Andinos Instituto Interamericano de Ciencias Agricolas: Bogota, Colombia, 1979; pp. 20–44. [Google Scholar]
- Kozioł, M.J. Chemical composition and nutritional evaluation of quinoa (Chenopodium quinoa Willd.). J. Food Compos. Analysis 1992, 5, 35–68. [Google Scholar] [CrossRef]
- Navruz-Varli, S.; Sanlier, N. Nutritional and health benefits of quinoa (Chenopodium quinoa Willd.). J. Cereal Sci. 2016, 69, 371–376. [Google Scholar] [CrossRef]
- Pellegrini, M.; Lucas-Gonzales, R.; Ricci, A.; Fontecha, J.; Fernández-López, J.; Pérez-Álvarez, J.A.; Viuda-Martos, M. Chemical, fatty acid, polyphenolic profile, techno-functional and antioxidant properties of flours obtained from quinoa (Chenopodium quinoa Willd) seeds. Ind. Crops Prod. 2018, 111, 38–46. [Google Scholar] [CrossRef]
- Nowak, V.; Du, J.; Charrondière, U.R. Assessment of the nutritional composition of quinoa (Chenopodium quinoa Willd.). Food Chem. 2016, 193, 47–54. [Google Scholar] [CrossRef]
- Chen, Y.-S.; Aluwi, N.A.; Saunders, S.R.; Ganjyal, G.M.; Medina-Meza, I.G. Metabolic fingerprinting unveils quinoa oil as a source of bioactive phytochemicals. Food Chem. 2019, 286, 592–599. [Google Scholar] [CrossRef] [PubMed]
- Romano, N.; Ureta, M.M.; Guerrero-Sánchez, M.; Gómez-Zavaglia, A. Nutritional and technological properties of a quinoa (Chenopodium quinoa Willd.) spray-dried powdered extract. Food Res. Inter. 2020, 129, 108884. [Google Scholar] [CrossRef] [PubMed]
- Mhada, M.; Metougui, M.L.; El Hazzam, K.; El Kacimi, K.; Yasri, A. Variations of saponins, minerals and total phenolic compounds due to processing and cooking of quinoa (Chenopodium quinoa Willd.) seeds. Foods 2020, 9, 660. [Google Scholar] [CrossRef] [PubMed]
- Lalaleo, L.; Hidalgo, D.; Valle, M.; Calero-Cáceres, W.; Lamuela-Raventós, R.M.; Becerra-Martínez, E. Differentiating, evaluating, and classifying three quinoa ecotypes by washing, cooking and germination treatments, using 1H NMR-based metabolomic approach. Food Chem. 2020, 331, 127351. [Google Scholar] [CrossRef]
- Fotschki, B.; Juśkiewicz, J.; Jurgoński, A.; Amarowicz, R.; Opyd, P.; Bez, J.; Muranyi, I.; Petersen, I.L.; Llopis, M.L. Protein-rich flours from quinoa and buckwheat favourably affect the growth parameters, intestinal microbial activity and plasma lipid profile of rats. Nutrients 2020, 12, 2781. [Google Scholar] [CrossRef]
- Miranda, M.; Vega-Galvez, A.; Martinez, E.; Lopez, J.; Rodriguez, M.J.; Henriquez, K.; Fuentes, F. Genetic diversity and comparison of physiochemical and nutritional characteristics of six quinoa (Chenopodium quinoa Willd.) genotypes cultivated in Chile. Cienc. Technol. Aliment. Campinas 2012, 32, 835–843. [Google Scholar] [CrossRef] [Green Version]
- Ruales, J.; de Grijalva, Y.; Lopez-Jaramillo, P.; Nair, B.M. The nutritional quality of an infant food from quinoa and its effect on the plasma level of insulin-like growth factor-1 (IGF-1) in undernourished children. Int. J. Food Sci. Nutr. 2002, 53, 143–154. [Google Scholar] [CrossRef]
- Farinazzi-Machado, F.M.V.; Barbalho, S.M.; Oshiiwa, M.; Goulart, R.; Pessan Junior, O. Use of cereal bars with quinoa (Chenopodium quinoa W.) to reduce risk factors related to cardiovascular diseases. Cienc. Technol. Aliment. Campinas 2012, 32, 239–244. [Google Scholar] [CrossRef] [Green Version]
- De Carvalho, F.G.; Ovidio, P.P.; Padovan, G.J.; Jordao Junior, A.A.; Marchini, J.S.; Navarro, A.M. Metabolic parameters of postmenopausal women after quinoa or corn flakes intake-a prospective and double-blind study. Int. J. Food Sci. Nutr. 2014, 65, 380–385. [Google Scholar] [CrossRef]
- Zevallos, V.F.; Herencia, I.L.; Chang, F.; Donnelly, S.; Ellis, J.H.; Ciclitira, P.J. Gastrointestinal effects of eating quinoa (Chenopodium quinoa Willd.) in celiac patients. Am. J. Gastroenterol. 2014, 109, 270–278. [Google Scholar] [CrossRef] [PubMed]
- Navarro-Perez, D.; Radcliffe, J.; Tierney, A.; Jois, M. Quinoa seed lowers serum triglycerides in overweight and obese subjects: A dose-response randomized controlled clinical trial. Curr. Dev. Nutr. 2017, 1, e001321. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, L.; Lietz, G.; Bal, W.; Watson, A.; Morfey, B.; Seal, C. Effects of quinoa (Chenopodium quinoa Willd.) consumption on markers of CVD risk. Nutrients 2018, 10, 777. [Google Scholar] [CrossRef] [Green Version]
- Brinegar, C.; Sine, B.; Nwokocha, L. High-cysteine 2S seed storage proteins from quinoa (Chenopodium quinoa). J. Agric. Food Chem. 1996, 44, 1621–1623. [Google Scholar] [CrossRef]
- Brinegar, C.; Goundan, S. Isolation and characterization of chenopodin, the 11S seed storage protein from quinoa (Chenopodium quinoa Willd). J. Agric. Food Chem. 1993, 41, 182–185. [Google Scholar] [CrossRef]
- Wang, J.; de Wit, M.; Schutyser, M.A.I.; Boom, R.M. Analysis of electrostatic powder charging for fractionation of foods. Innov. Food Sci. Emerg. Technol. 2014, 26, 360–365. [Google Scholar] [CrossRef]
- Pelgrom, P.J.M.; Vissers, A.M.; Boom, R.M.; Schutyser, M.A.I. Dry fractionation for production of functional pea protein concentrates. Food Res. Int. 2013, 3, 232–239. [Google Scholar] [CrossRef]
- Motta, C.; Castanheira, I.; Gonzales, G.B.; Delgado, I.; Torres, D.; Santos, M.; Matos, A.S. Impact of cooking methods and malting on amino acids content in amaranth, buckwheat and quinoa. J. Food Comp. Anal. 2019, 76, 58–65. [Google Scholar] [CrossRef]
- Benito-Román, O.; Rodríguez-Perrino, M.; Sanz, M.T.; Melgosa, R.; Beltrán, S. Supercritical carbon dioxide extraction of quinoa oil: Study of the influence of process parameters on the extraction yield and oil quality. J. Supercritical Fluids 2018, 139, 62–71. [Google Scholar] [CrossRef] [Green Version]
- Graf, B.L.; Rojo, L.E.; Delatorre-Herrera, J.; Poulev, A.; Calfio, C.; Raskin, I. Innovations in health value and functional food development of quinoa (Chenopodium quinoa Willd.). Compr. Rev. Food Sci Safety. 2015, 14, 431–445. [Google Scholar] [CrossRef] [Green Version]
- Yao, Y.; Yang, X.; Shi, Z.; Ren, G. Anti-inflammatory activity of saponins from quinoa (Chenopodium quinoa Willd.) seeds in lipopolysaccharide-stimulated RAW 264.7 macrophages cells. J. Food Sci. 2014, 79, H1018–H1023. [Google Scholar] [CrossRef]
- Jancurová, M.; Minarovičová, L.; Dandár, A. Quinoa—A review. Czech J. Food Sci. 2009, 27, 71–79. [Google Scholar] [CrossRef] [Green Version]
- Valencia-Chamorro, S.A. Quinoa. In Encyclopedia of Food Science and Nutrition; Caballero, B., Ed.; Academic Press: Amsterdam, The Netherlands, 2003; pp. 4895–4902. [Google Scholar]
- Jan, K.N.; Panesar, P.S.; Singh, S. Effect of moisture content on the physical and mechanical properties of quinoa seeds. Int. Agrophys. 2019, 33, 41–48. [Google Scholar] [CrossRef]
- González, J.A.; Roldán, A.; Gallardo, M.; Escudero, T.; Prado, F.E. Quantitative determinations of chemical compounds with nutritional value from Inca crops: Chenopodium quinoa ("quinoa"). Plant Foods Hum. Nutr. 1989, 39, 331–337. [Google Scholar] [CrossRef] [PubMed]
- Gross, R.; Koch, F.; Malaga, I.; De Miranda, A.F.; Schoeneberger, H.; Trugo, L.C. Chemical composition and protein quality of some local Andean food sources. Food Chem. 1989, 34, 25–34. [Google Scholar] [CrossRef]
- Contreras-Jiménez, B.; Torres-Vargas, O.L.; Rodríguez-García, M.E. Physicochemical characterization of quinoa (Chenopodium quinoa) flour and isolated starch. Food Chem. 2019, 298, 124982. [Google Scholar] [CrossRef]
- Kierulf, A.; Whaley, J.; Liu, W.; Enayati, M.; Tan, C.; Perez-Herrera, M.; You, Z.; Abbaspourrad, A. Protein content of amaranth and quinoa starch plays a key role in their ability as pickering emulsifiers. Food Chem. 2020, 315, 126246. [Google Scholar] [CrossRef]
- Lorenz, K. Quinoa (Chenopodium quinoa Willd) starch: Physico-chemical properties and functional characteristics. Starch/Staerke 1990, 42, 81–86. [Google Scholar] [CrossRef]
- Ruales, J.; Nair, B. Properties of starch and dietary fibre in quinoa (Chenopodium quinoa Willd) seeds. Plant Foods Hum. Nutr. 1994, 45, 223–246. [Google Scholar] [CrossRef]
- Goyat, J.; Rudra, S.G.; Suri, S.; Passi, S.J.; Dutta, H. Nutritional, functional and sensory properties of ready-to-eat chia and quinoa mix enriched low amylose rice based porridge mixes. Curr. Res. Nutr. Food Sci. J. 2019, 7, 399–414. [Google Scholar] [CrossRef]
- Ranhotra, G.; Gelroth, J.; Glaser, B.; Lorenz, K.; Johnson, D. Composition and protein nutritional quality of quinoa. Cereal Chem. 1993, 3, 303–305. [Google Scholar]
- Alvarez-Jubete, L.; Arendt, E.K.; Gallagher, E. Nutritive value and chemical composition of pseudocereals as gluten-free ingredients. Inter. J. Food Sci. Nutr. 2009, 60, 240–257. [Google Scholar] [CrossRef] [PubMed]
- Lamothe, L.M.; Srichuwong, S.; Reuhs, B.L.; Hamaker, B.R. Quinoa (Chenopodium quinoa W.) and amaranth (Amaranthus caudatus L.) provide dietary fibres high in pectic substances and xyloglucans. Food Chem. 2015, 15, 490–496. [Google Scholar] [CrossRef]
- Ruales, J.; Nair, B. Content of fat, vitamins and minerals in quinoa (Chenopodium quinoa, Willd) seeds. Food Chem. 1993, 48, 131–136. [Google Scholar] [CrossRef]
- Basantes-Morales, E.R.; Alconada, M.M.; Pantoja, J.L. Quinoa (Chenopodium quinoa Willd) Production in the Andean Region: Challenges and potentials. J. Exper. Agric. Int. 2019, 36, 1–18. [Google Scholar] [CrossRef]
- Abugoch, L.E. Quinoa (Chenopodium quinoa Willd.): Composition, chemistry, nutritional and functional properties. Adv. Food Nutr. Res. 2009, 58, 1–31. [Google Scholar]
- Mohyuddin, S.G.; Riaz, A.; Qamar, A.; Ali, S.H.; Hu, C.; Wu, L.; Yu, T.; Ju, X.H. Quinoa is beneficial to the comprehensive nutritional value of potential health. Pakistan J. Sci. 2019, 70, 69–74. [Google Scholar]
- Lorenz, K.; Nyanzi, F. Enzyme activities in quinoa (Chenopodium quinoa Willd). Int. J. Food Sci. Technol. 1989, 24, 543–551. [Google Scholar] [CrossRef]
- Dinan, L.; Lafont, R. Innovative and future applications for ecdysteroids. In Ecdysone: Structures and Functions; Smagghe, G., Ed.; Springer: Dordrecht, The Netherlands, 2009; pp. 551–578. [Google Scholar]
- Bajguz, A.; Bąkała, I.; Talarek, M. Ecdysteroids in plants and their pharmacological effects in vertebrates and humans. In Studies in Natural Products Chemistry; Rahman, A., Ed.; Elsevier: New York, NY, USA, 2015; Volume 45, pp. 121–145. [Google Scholar]
- Kumpun, S.; Maria, A.; Crouzet, S.; Evrard-Todeschi, N.; Girault, J.P.; Lafont, R. Ecdysteroids from Chenopodium quinoa Willd., an ancient Andean crop of high nutritional value. Food Chem. 2011, 125, 1226–1234. [Google Scholar] [CrossRef]
- Graf, B.L.; Rojo, L.E.; Delatorre-Herrera, J.; Poulev, A.; Calfio, C.; Raskin, I. Phytoecdysteroids and flavonoid glycosides among Chilean and commercial sources of Chenopodium quinoa: Variation and correlation to physico-chemical characteristics. J. Sci. Food Agric. 2016, 96, 633–643. [Google Scholar] [CrossRef] [Green Version]
- Lin, M.; Han, P.; Li, Y.; Wang, W.; Lai, D.; Zhou, L. Quinoa secondary metabolites and their biological activities or functions. Molecules 2019, 24, 2512. [Google Scholar] [CrossRef] [Green Version]
- Nsimba, Y.R.; Kikuzaki, H.; Konishi, Y. Ecdysteroids act as inhibitors of calf skin collagenase and oxidative stress. J. Biochem. Mol. Toxicol. 2008, 22, 240–250. [Google Scholar] [CrossRef]
- Bathori, M.; Tóth, N.; Hunyadi, A.; Márki, A.; Zádor, E. Phtoecdysteroids and anabolic-androgenic steroids—Structure and effects in humans. Curr. Med. Chem. 2008, 15, 75–91. [Google Scholar] [CrossRef] [Green Version]
- Gorelick-Feldman, M.D.; Maclean, D.; Ilic, N.; Poulev, A.; Lila, M.A.; Cheng, D.; Raskin, I. Phytoecdysteroids increase protein synthesis in skeletal muscle cells. J. Agric. Food Chem. 2008, 56, 3532–3537. [Google Scholar] [CrossRef]
- Foucault, A.S.; Even, P.; Lafont, R.; Dioh, W.; Veillet, S.; Tomé, D.; Huneau, J.F.; Hermier, D.; Quignard-Boulangé, A. Quinoa extract enriched in 20-hydroxyecdysone affects energy homeostasis and intestinal fat absorption in mice fed a high-fat diet. Physiol. Behav. J. 2014, 128, 226–231. [Google Scholar] [CrossRef] [PubMed]
- Xia, X.; Zhang, Q.; Liu, R.; Wang, Z.; Tang, N.; Liu, F.; Huang, G.; Jiang, X.; Gui, G.; Wang, L.; et al. Effects of 20-hydroxyecdysone on improving memory deficits in streptozotocin-induced type 1 diabetes mellitus in rat. Eur. J. Pharmacol. 2014, 740, 45–52. [Google Scholar] [CrossRef] [PubMed]
- Yendo, A.C.A.; De Costa, F.; Gosmann, G.; Fett-Neto, A.G. Production of plant bioactive triterpenoid saponins: Elicitation strategies and target genes to improve yields. Mol. Biotechnol. 2010, 46, 94–104. [Google Scholar] [CrossRef]
- Dini, I.; Tenore, G.C.; Dini, A. Antioxidant compound contents and antioxidant activity before and after cooking in sweet and bitter Chenopodium quinoa seeds. LWT-Food Sci. Tech. 2010, 43, 447–451. [Google Scholar] [CrossRef]
- Gomez-Caravaca, A.M.; Iafelice, G.; Lavini, A.; Pulvent, C.; Caboni, M.F.; Marconi, E. Phenolic compounds and saponins in quinoa samples (Chenopodium quinoa Willd.) grown under different saline and nonsaline irrigation regimens. J. Agric. Food Chem. 2012, 60, 4620–4627. [Google Scholar] [CrossRef] [PubMed]
- Macarena, S.; Ricardo, S.N. Antifungal properties of quinoa (Chenopodium quinoa Willd) alkali treated saponins against Botrytis cinerea. Ind. Crops Prod. 2008, 27, 296–302. [Google Scholar] [CrossRef]
- Vilche, C.; Gely, M.; Santalla, E. Physical properties of quinoa seeds. Biosyst. Engin. 2003, 86, 59–65. [Google Scholar] [CrossRef]
- Woldemichael, G.M.; Wink, M. Identification and biological activities of triterpenoid saponins from Chenopodium quinoa. J. Agric. Food Chem. 2001, 49, 2327–2332. [Google Scholar] [CrossRef]
- Harborne, J.B.; Williams, C.A. Advances in flavonoid research since 1992. Phytochemistry 2000, 55, 481–504. [Google Scholar] [CrossRef]
- Han, Y.; Chi, J.; Zhang, M.; Zhang, R.; Fan, S.; Dong, L.; Huang, F.; Liu, L. Changes in saponins, phenolics and antioxidant activity of quinoa (Chenopodium quinoa willd) during milling process. LWT Food Sci. Technol. 2019, 114, 108381. [Google Scholar] [CrossRef]
- Okarter, N.; Liu, R.H. Health benefits of whole grain phytochemicals. Crit. Rev. Food Sci. Nutr. 2010, 50, 193–208. [Google Scholar] [CrossRef]
- Da-Silva, W.S.; Harney, J.W.; Kim, B.W.; Li, J.; Bianco, S.D.; Crescenzi, A.; Christoffolete, M.A.; Huang, S.A.; Bianco, A.C. The small polyphenolic molecule kaempferol increases cellular energy expenditure and thyroid hormone activation. Diabetes 2007, 56, 767–776. [Google Scholar] [CrossRef] [Green Version]
- Renard, C.M.; Wende, G.; Booth, E.J. Cell wall phenolics and polysaccharides in different tissues of quinoa (Chenopodium quinoa Willd). J. Sci. Food Agric. 1999, 79, 2029–2034. [Google Scholar] [CrossRef]
- Gómez-Caravaca, A.M.; Segura-Carretero, A.; Fernández-Gutiérrez, A.; Caboni, M.F. Simultaneous determination of phenolic compounds and saponins in quinoa (Chenopodium quinoa Willd) by a liquid chromatography–diode array detection–electrospray ionization–time-of-flight mass spectrometry methodology. J. Agric. Food Chem. 2011, 59, 10815–10825. [Google Scholar] [CrossRef]
- Lutz, M.; Martínez, A.; Martínez, E.A. Daidzein and genistein contents in seeds of quinoa (Chenopodium quinoa Willd.) from local ecotypes grown in arid Chile. Ind. Crops Prod. 2013, 49, 117–121. [Google Scholar] [CrossRef]
- Samtiya, M.; Aluko, R.E.; Dhewa, T.; Moreno-Rojas, J.M. Potential health benefits of plant food-derived bioactive components: An overview. Foods 2021, 10, 839. [Google Scholar] [CrossRef] [PubMed]
- Escribano, J.; Cabanes, J.; Jiménez-Atiénzar, M.; Ibañez-Tremolada, M.; Gómez-Pando, L.R.; García-Carmona, F.; Gandía-Herrero, F. Characterization of betalains, saponins and antioxidant power in differently colored quinoa (Chenopodium quinoa) varieties. Food Chem. 2017, 234, 285–294. [Google Scholar] [CrossRef] [PubMed]
- Abderrahim, F.; Huanatico, E.; Segura, R.; Arribas, S.; Gonzalez, M.C.; Condezo-Hoyos, L. Physical features, phenolic compounds, betalains and total antioxidant capacity of coloured quinoa seeds (Chenopodium quinoa Willd.) from Peruvian Altiplano. Food Chem. 2015, 183, 83–90. [Google Scholar] [CrossRef] [PubMed]
- Ann-Mari Repo-Carrasco-Valencia, R.; Lesli Astuhuaman, S. Quinoa (Chenopodium quinoa, Willd.) as a source of dietary fiber and other functional components. Food Sci. Technol. 2011, 31, 225–230. [Google Scholar] [CrossRef] [Green Version]
- Neagu, C.; Barbu, V. Principal component analysis of the factors involved in the extraction of beetroot betalains. J. Agroaliment. Proc. Technol. 2014, 20, 311–318. [Google Scholar]
- Gengatharan, A.; Dykes, G.A.; Choo, W.S. Betalains: Natural plant pigments with potential application in functional foods. LWT Food Sci. Technol. 2015, 64, 645–649. [Google Scholar] [CrossRef]
- Aguilar-Tuesta, S.; Mamani-Navarro, W.; Espinoza-Silva, C.; Basilio-Atencio, J.; Condezo-Hoyos, L. Microencapsulated betacyanin from colored organic quinoa (Chenopodium quinoa Willd.): Optimization, physicochemical characterization and accelerated storage stability. J. Sci. Food Agric. 2018, 98, 5873–5883. [Google Scholar] [CrossRef] [PubMed]
- Esatbeyoglu, T.; Wagner, A.E.; Schiniatbeyo, V.B.; Rimbach, G. Betanin—A food colorant with biological activity. Mol. Nutr. Food Res. 2015, 59, 36–47. [Google Scholar] [CrossRef] [PubMed]
- Laqui-Vilca, C.; Aguilar-Tuesta, S.; Mamani-Navarro, W.; Montaño-Bustamante, J.; Condezo-Hoyos, L. Ultrasound-assisted optimal extraction and thermal stability of betalains from colored quinoa (Chenopodium quinoa Willd) hulls. Ind. Crops Prod. 2018, 111, 606–614. [Google Scholar] [CrossRef]
- Tang, Y.; Zhang, B.; Li, X.; Chen, P.X.; Zhang, H.; Liu, R.; Tsao, R. Bound phenolics of quinoa seeds released by acid, alkaline, and enzymatic treatments and their antioxidant and α-glucosidase and pancreatic lipase inhibitory effects. J. Agric. Food Chem. 2016, 64, 1712–1719. [Google Scholar] [CrossRef]
- Chen, P.X.; Tang, Y.; Marcone, M.F.; Pauls, P.K.; Zhang, B.; Liu, R.; Tsao, R. Characterization of free, conjugated and bound phenolics and lipophilic antioxidants in regular-and non-darkening cranberry beans (Phaseolus vulgaris L.). Food Chem. 2015, 185, 298–308. [Google Scholar] [CrossRef]
- Acosta-Estrada, B.A.; Gutiérrez-Uribe, J.A.; Serna-Saldívar, S.O. Bound phenolics in foods, a review. Food Chem. 2014, 152, 46–55. [Google Scholar] [CrossRef] [PubMed]
- Multari, S.; Marsol-Vall, A.; Keskitalo, M.; Yang, B.; Suomela, J.P. Effects of different drying temperatures on the content of phenolic compounds and carotenoids in quinoa seeds (Chenopodium quinoa) from Finland. J. Food Compos. Anal. 2018, 72, 75–82. [Google Scholar] [CrossRef]
- Zhang, Q.; Xing, B.; Sun, M.; Zhou, B.; Ren, G.; Qin, P. Changes in bio-accessibility, polyphenol profile and antioxidants of quinoa and djulis sprouts during in vitro simulated gastrointestinal digestion. Food Sci. Nutr. 2020, 8, 4232–4241. [Google Scholar] [CrossRef] [PubMed]
- De Simone, F.; Dini, A.; Pizza, C.; Saturnino, P.; Schettino, O. Two flavonol glycosides from Chenopodium quinoa. Phytochemistry 1990, 29, 3690–3692. [Google Scholar] [CrossRef]
- Sampaio, S.L.; Fernandes, A.; Pereira, C.; Calhelha, R.C.; Sokovic, M.; Santos-Buelga, C.; Ferreira, I.C.F.R. Nutritional value, physicochemical characterization and bioactive properties of Brazilian quinoa BRS Piabiru. Food Funct. 2020, 11, 2969–2977. [Google Scholar] [CrossRef] [PubMed]
- Gorinstein, S.; Lojek, A.; Číž, M.; Pawelzik, E.; Delgado-Licon, E.; Medina, O.J.; Moreno, M.; Salas, I.A.; Goshev, I. Comparison of composition and antioxidant capacity of some cereals and pseudocereals. Inter. J. Food Sci. Technol. 2008, 43, 629–637. [Google Scholar] [CrossRef]
- Tsao, R. Chemistry and biochemistry of dietary polyphenols. Nutrients 2010, 2, 1231–1246. [Google Scholar] [CrossRef]

| Study Participants | Treatment | Duration | Outcomes | Conclusions | References |
|---|---|---|---|---|---|
| 50–65-month-old boys | 100 g quinoa-added to baby food/day | 15 days | Increase in plasma level of insulin-like growth factor-1 | Potential role in reducing childhood malnutrition | [33] |
| 18–45-year-old students | Quinoa cereal bars | 30 days | Decrease in total cholesterol, triglycerides and low-density lipoprotein levels | Potential role in preventing cardiovascular disease | [34] |
| Post-menopausal, overweight women | 25 g quinoa flakes and cornflakes/day | 4 weeks | Reduction of total cholesterol, triglycerides and low-density lipoprotein levels and thiobarbituric acid reactive substances Increase of urinary secretion of enterolignans | Beneficial effect on metabolic parameter modulation | [35] |
| Celiac patients | 50 g quinoa/day | 6 weeks | Improved histological and serological parameters Mild hypocholesterolemic effect | Quinoa consumption is safe for celiac individuals | [36] |
| Overweight and obese participants | 50 g quinoa/day | 12 weeks | Reduction of serum triglyceride level Reduction of metabolic syndrome | Potential role in preventing obesity | [37] |
| 35–70-year-old healthy, overweight males | Quinoa-enriched bread (with 20 g quinoa flour)/day | 4 weeks | Reduction in blood glucose and low-density lipoprotein levels | Potential role in preventing cardiovascular disease | [38] |
| Quinoa Genotypes | Moisture | Proteins | Fat | Crude Fiber | Ash | Carbohydrates |
|---|---|---|---|---|---|---|
| Ccoito | 8.47 ± 0.08 | 14.72 ± 0.11 | 5.33 ± 0.06 | 1.81 ± 0.02 | 2.83 ± 0.00 | 68.1 |
| INIA-415 Pasankalla | 9.76 ± 0.07 | 12.69 ± 0.06 | 6.85 ± 0.10 | 2.2 ± 0.02 | 2.49 ± 0.03 | 67 |
| Roja de Copotaque | 8.3 ± 0.07 | 11.51 ± 0.10 | 5.22 ± 0.08 | 2.26 ± 0.02 | 2.93 ± 0.05 | 70.8 |
| Witulla | 8.81 ± 0.08 | 12.28 ± 0.00 | 5.32 ± 0.01 | 2.62 ± 0.02 | 2.57 ± 0.04 | 69.5 |
| 03-21-0093 | 8.47 ± 0.07 | 11.79 ± 0.11 | nd | Nd | 2.76 ± 0.02 | nd |
| Salcedo INIA | 8.26 ± 0.05 | 13.23 ± 0.01 | 5.3 ± 0.09 | 5.3 ± 0.2 | 2.37 ± 0.05 | 70 |
| Commercial 1 | 10.13 ± 0.05 | 13.18 ± 0.01 | 6.51 ± 0.04 | 6.51 ± 0.03 | 2.34 ± 0.10 | 63.6 |
| Commercial 2 | 11.51 ± 0.04 | 13.48 ± 0.06 | 6.34 ± 0.07 | 6.34 ± 0.03 | 2.27 ± 0.10 | 59.4 |
| Huaripongo | 10.34 ± 0.02 | 11.32 ± 0.01 | 6.14 ± 0.01 | 6.14 ± 0.01 | 2.92 ± 0.04 | 67.8 |
| 03-21-1181 | 9.37 ± 0.06 | 11.89 ± 0.02 | 3.95 ± 0.03 | 3.95 ± 0.01 | 3.12 ± 0.02 | 69.8 |
| Mean ± SD | 9.34 ± 1.1 a | 12.61 ± 1.1 a | 5.66 ± 0.09 a | 5.66 ± 1.7 a | 2.66 ± 0.3 a | 67.3 ± 3.7 a |
| Variety | Observed Compounds/Reports | References |
|---|---|---|
| Minttumatilda | Vanillic acid, gallic acid, p-benzoic acid, syringaldehyde, ferulic acid | [99] |
| Four varieties of white (Q-w1), red (Q-r1 and Q-r2) and black (Q-b1) and three varieties of white (Q-w2), red (Q-r3) and black (Q-b2) | Gallic acid, caffeic acid, ferulic acid, p-coumaric acid, p-OH benzoic acid, vanillic acid, protocatechuic acid | [99] |
| Q25, Q50, Q100, Q25S, Q50S, Q100S | 1-O-galloyl-β-d-glucose, acacetin/questin/apigenin-7-methyl ether, protocatechuic acid 4-O-glucoside, vanillic glucoside, penstebiosided, canthoside A/2-hydroxybenzoate 2-O-β-d-apiofuranosyl-(1→6)-O-β-Dglucopyranoside, ferulic acid 4-O-glucoside, ethyl-m-digallate, (epi) gallocatechin, quercetin 3-O-(2,6-di-α-l-rhamnopyranosyl)-β-d-galactopyranoside, kaempferol 3-O-β-d-apiofuranosyl(1‴→2″-O-[α-l-rhamnopyranosyl(1‴→6″]-β-d-galactopyranoside, kaempferol 3-O-β-d-apiofuranosyl(1‴→2″-O-[α-l-rhamnopyranosyl(1‴→ 6″]-β-d-galactopyranoside isomer, kaempferol 3-O-(2,6-di-α-l-rhamnopyranosyl)-β-d-galactopyranoside (mauritianin), quercetin 3-O-[ β-d-apiofuranosyl(1‴→2″)]-β-d-galactopyranoside, rutin, quercetin glucuronide, quercetin 3-O-glucoside | [85] |
| Red quinoa (RQ), white quinoa (WQ), Mengli 1 (gray quinoa, GQ), Altiplano, djulis cultivar | Protocatechuic acid, p-hydroxybenzoic acid, vanillic acid, caffeic acid, syringic acid, p-coumaric acid, ferulic acid, isoferulic acid | [100] |
| White and red quinoa | Gentisic acid, L-alpha-hydroxy isovaleric acid, 3-hydroxybenzoic acid, p-coumaric acid, methyl-b-d-galactopyranoside, 3-(3,4-dihydroxyphenyl)propionic acid hesperidin | [100] |
| Puno, Titicaca | Ferulic acid, 5-O-caffeoylquinic acid, gentisic acid, p-coumaric acid, ellagic acid, pterostilbene, coniferyl aldehyde | [13] |
| White quinoa, red quinoa, black quinoa | Protocatechuic acid, p-hydroxybenzoic acid, vanillic acid, syringic acid, p-coumaric acid, ferulic acid, sinapic acid, isoferulic acid | [6] |
| References | Flavonoids | |
|---|---|---|
| [101] | Quercetin 3-O-(2,6-di-α-Lrhamnopyranosyl)-β-Dgalactopyranoside | |
| [85] | Quercetin 3-O-(2,6-di-α-Lrhamnopyranosyl)-β-Dgalactopyranoside | |
| [85] | Quercetin-3-O-(2″-apiosyl)-rutinoside | |
| [85] | Quercetin glucuronide | |
| [101,102] | Kaempferol 3-O-(2,6-di-α-Lrhamnopyranosyl)-β-Dgalactopyranoside | |
| [101,102] | Kaempferol 3-O-(β-d-apiofuranosyl (1″→2″)-α-l-rhamnopyranosyl) β- Dgalactopyranoside | |
| [101] | Kaempferol 3-O-[β-d-apiofuranosyl (1″→2″)] β-d-galactopyranoside | |
| [101] | Kaempferol-3-O-(α-Lrhamnopyranosyl) -β- Dgalactopyranoside, kaempferol glucuronide | |
| [14] | Kaempferol 3-O-glucoside, kaempferol pentosyl rhamnoside, kaempferol | |
| [13] | Puno, Titicaca | Quercetin, isorhamnetin, quercetin−3-Ogalactoside, isorhamnetin−3-Orutinoside, rutin, naringin, aesculin, phlorizin, eriodictyol |
| [103] | BRS Piabiru | Quercetin, kaempferol 3-O-rutinoside, quercetin 3-O-(2″,6″-di-O-α-l-rhamnoside)-β-d-galactoside, quercetin 3-O-(2″-O-β-apioside-6″-O-α-rhamnoside)-β-galactoside, kaempferol 3-O-(2″,6″-di-O-α-rhamnoside)-β-galactoside, kaempferol 3-O-(2″,6″-di-O-α-rhamnoside)-β-glucoside |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hussain, M.I.; Farooq, M.; Syed, Q.A.; Ishaq, A.; Al-Ghamdi, A.A.; Hatamleh, A.A. Botany, Nutritional Value, Phytochemical Composition and Biological Activities of Quinoa. Plants 2021, 10, 2258. https://doi.org/10.3390/plants10112258
Hussain MI, Farooq M, Syed QA, Ishaq A, Al-Ghamdi AA, Hatamleh AA. Botany, Nutritional Value, Phytochemical Composition and Biological Activities of Quinoa. Plants. 2021; 10(11):2258. https://doi.org/10.3390/plants10112258
Chicago/Turabian StyleHussain, M. Iftikhar, Muhammad Farooq, Qamar Abbas Syed, Anum Ishaq, Abdullah Ahmed Al-Ghamdi, and Ashraf A. Hatamleh. 2021. "Botany, Nutritional Value, Phytochemical Composition and Biological Activities of Quinoa" Plants 10, no. 11: 2258. https://doi.org/10.3390/plants10112258
APA StyleHussain, M. I., Farooq, M., Syed, Q. A., Ishaq, A., Al-Ghamdi, A. A., & Hatamleh, A. A. (2021). Botany, Nutritional Value, Phytochemical Composition and Biological Activities of Quinoa. Plants, 10(11), 2258. https://doi.org/10.3390/plants10112258







