Structural Characterization and Antioxidant Capacity of Quinoa Cultivars Using Techniques of FT-MIR and UHPLC/ESI-Orbitrap MS Spectroscopy
Abstract
:1. Introduction
2. Results
2.1. Quinoa Grain Characteristics
2.2. Infrared Spectroscopy
2.2.1. Secondary Protein Structure by FTIR Spectroscopy
2.2.2. Secondary Starch Structure by FTIR Spectroscopy
2.3. Polyphenols and Antioxidant Capacity by ABTS, DPPH and FRAP
2.4. Phenolic Compounds under UHPLC-ESI-Orbitrap MS
2.5. Phytochemical Multivariate Structure Analysis
3. Discussion
4. Materials and Methods
4.1. Materials
4.2. Proximal Characteristics
4.3. CIEL*a*b* Coordinates Determination
4.4. Mid-Range Infrared Spectroscopy
4.5. Polyphenols
4.6. Antioxidants
4.6.1. FRAP
4.6.2. ABTS
4.6.3. DPPH
4.7. Phenolic Compounds Determination by UHPLC/ESI-Orbitrap MS
4.8. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Bazile, D.; Pulvento, C.; Verniau, A.; Al-Nusairi, M.S.; Ba, D.; Breidy, J.; Hassan, L.; Mohammed, M.I.; Mambetov, O.; Otambekova, M.; et al. Worldwide Evaluations of Quinoa: Preliminary Results from Post International Year of Quinoa FAO Projects in Nine Countries. Front Plant Sci. 2016, 7, 850. [Google Scholar] [CrossRef] [Green Version]
- García-Parra, M.; Zurita-Silva, A.; Stechauner-Rohringer, R.; Roa-Acosta, D.; Jacobsen, S.-E. Quinoa (Chenopodium quinoa Willd.) and its relationship with agroclimatic characteristics: A Colombian perspective. Chil. J. Agric. Res. 2020, 80, 290–302. [Google Scholar] [CrossRef]
- Ruiz, K.B.; Biondi, S.; Oses, R.; Acuña-Rodríguez, I.S.; Antognoni, F.; Martinez-Mosqueira, E.A.; Coulibaly, A.; Canahua-Murillo, A.; Pinto, M.; Zurita-Silva, A.; et al. Quinoa biodiversity and sustainability for food security under climate change. A review. Agron. Sustain. Dev. 2014, 34, 349–359. [Google Scholar] [CrossRef] [Green Version]
- Pinedo-Taco, R.; Gómez-Pando, L.; Julca-Otiniano, A. Sostenibilidad ambiental de la producción de quinua (Chenopodium quinoa Willd.) en los valles interandinos del Perú. Cienc. Tecnol. Agropecu. 2020, 21, 1–17. [Google Scholar]
- Bazile, D.; Martínez, E.A.; Fuentes, F. Diversity of quinoa in a biogeographical Island: A review of constraints and potential from arid to temperate regions of Chile. Not. Bot. Horti Agrobot. 2014, 42, 289–298. [Google Scholar] [CrossRef] [Green Version]
- Curti, R.; Carmen-Sanahuja, M.; Vidueiros, S.; Curti, C.; Pallaro, A.; Bertero, H. Oil quality in sea level quinoa as determined by cultivar-specific responses to temperature and radiation conditions. J. Sci. Food Agric. 2019, 100, 1358–1361. [Google Scholar] [CrossRef]
- Li, G.; Zhu, F. Quinoa starch: Structure, properties, and applications. Carbohydr. Polym. 2018, 181, 851–861. [Google Scholar] [CrossRef]
- Bazile, D.; Bertero, H.D.; Nieto, C. Estado del Arte de la Quinua en el Mundo 2013; FAO: Santiago, Chile; CIRAD: Montpellier, Francia, 2014; Volume 1, 724p. [Google Scholar]
- Reguera, M.; Conesa, C.M.; Gil-Gómez, A.; Haros, C.M.; Pérez-Casas, M.Á.; Briones-Labarca, V.; Bolaños, L.; Bonilla, I.; Álvarez, R.; Pinto, K.; et al. The impact of different agroecological conditions on the nutritional composition of quinoa seeds. PeerJ 2018, 14, 1–20. [Google Scholar] [CrossRef]
- García-Parra, M.; García-Molano, J.; Deaquiz-Oyola, Y. Physiological performance of quinoa (Chenopodium quinoa Willd.) under agricultural climatic conditions in Boyaca, Colombia. Agron. Colomb. 2019, 37, 160–168. [Google Scholar] [CrossRef]
- Lesjak, J.; Calderini, D.F. Increased Night Temperature Negatively Affects Grain Yield, Biomass and Grain Number in Chilean Quinoa. Front. Plant Sci. 2017, 8, 1–11. [Google Scholar] [CrossRef] [Green Version]
- Miranda, M.; Vega-Gálvez, A.; Martínez, E.; López, J.; Marín, R.; Aranda, M.; Fuentes, F. Influence of contrasting environments on seed composition of two quinoa genotypes: Nutritional and functional properties. Chil. J. Agric. Res. 2013, 73, 108–116. [Google Scholar] [CrossRef]
- García-Salcedo, A.J.; Torres-Vargas, O.L.; Ariza-Calderón, H. Physical-chemical characterization of quinoa (Chenopodium quinoa Willd.), amaranth (Amaranthus caudatus, L.), and chia (Salvia hispanica, L.) flours and seeds. Acta Agron. 2018, 67, 215–222. [Google Scholar]
- Roa-Acosta, D.F.; Bravo-Gómez, J.E.; García-Parra, M.A.; Rodríguez-Herrera, R.; Solanilla-Duque, J.F. Hyper-protein quinoa flour (Chenopodium Quinoa Wild): Monitoring and study of structural and rheological properties. LWT-Food Sci. Technol. 2020, 121, 1–7. [Google Scholar] [CrossRef]
- Kwil, I.; Piwowar-Sulej, K.; Krzywonos, M. Local entrepreneurship in the context of food production: A review. Sustainability 2020, 12, 424. [Google Scholar] [CrossRef] [Green Version]
- Rodríguez, S.D.; López-Fernández, M.P.; Maldonado, S.; Buera, M.P. Evidence on the discrimination of quinoa grains with a combination of FT-MIR and FT-NIR spectroscopy. J. Food Sci. Technol. 2019, 56, 4457–4464. [Google Scholar] [CrossRef]
- Abdelaleem, M.A.; Elbassiony, K.R.A. Evaluation of phytochemicals and antioxidant activity of gamma irradiated quinoa (Chenopodium quinoa). Braz. J. Biol. 2021, 81, 806–813. [Google Scholar] [CrossRef]
- Carrasco-Sandoval, J.; Rebolledo, P.; Peterssen-Fonseca, D.; Fischer, S.; Wilckens, R.; Aranda, M.; Henríquez-Aedo, K. A fast and selective method to determine phenolic compounds in quinoa (Chenopodium quinoa Will) seeds applying ultrasound-assisted extraction and high-performance liquid chromatography. Chem. Pap. 2020, 75, 431–438. [Google Scholar] [CrossRef]
- Bazile, D.; Jacobsen, S.-E.; Verniau, A. The Global Expansion of Quinoa: Trends and Limits. Front. Plant Sci. 2016, 7, 1–6. [Google Scholar] [CrossRef] [Green Version]
- Barth, A. Infrared spectroscopy of proteins. Biochim. Biophys. Acta Bioenerg. 2007, 1767, 1073–1101. [Google Scholar] [CrossRef] [Green Version]
- Sadat, A.; Joye, I.J. Peak fitting applied to fourier transform infrared and raman spectroscopic analysis of proteins. Appl. Sci. 2020, 10, 5918. [Google Scholar] [CrossRef]
- Escribano, J.; Cabanes, J.; Jiménez-Atiénzar, M.; Ibañez-Tremolada, M.; Gómez-Pando, L.R.; García-Carmona, F.; Gandía-Herrero, F. Characterization of betalains, saponins and antioxidant power in differently colored quinoa (Chenopodium quinoa) varieties. Food Chem. 2017, 234, 285–294. [Google Scholar] [CrossRef]
- Medina, W.; Skurtys, O.; Aguilera, J.M. Study on image analysis application for identification Quinoa seeds (Chenopodium quinoa Willd) geographical provenance. LWT-Food Sci. Technol. 2010, 43, 238–246. [Google Scholar] [CrossRef]
- Abderrahim, F.; Huanatico, E.; Segura, R.; Arribas, S.; Gonzalez, M.C.; Condezo-Hoyos, L. Physical features, phenolic compounds, betalains and total antioxidant capacity of coloured quinoa seeds (Chenopodium quinoa Willd.) from Peruvian Altiplano. Food Chem. 2015, 183, 83–90. [Google Scholar] [CrossRef]
- Li, G.; Zhu, F. Rheological properties in relation to molecular structure of quinoa starch. Int. J. Biol. Macromol. 2018, 114, 767–775. [Google Scholar] [CrossRef]
- Yang, H.; Yang, S.; Kong, J.; Dong, A.; Yu, S. Obtaining information about protein secondary structures in aqueous solution using Fourier transform IR spectroscopy. Nat. Protoc. 2015, 10, 382–396. [Google Scholar] [CrossRef]
- Roa-Acosta, D.F.; Solanilla-Duque, J.F.; Agudelo-Laverde, L.; Villada-Castillo, H.S.; Tolaba, M.P. Structural and thermal properties of the amaranth starch granule obtained by high-impact wet milling. Int. J. Food Eng. 2020, 16, 1–12. [Google Scholar] [CrossRef]
- Wang, X.; Zhao, R.; Yuan, W. Composition and secondary structure of proteins isolated from six different quinoa varieties from China. J. Cereal Sci. 2020, 95, 103036. [Google Scholar] [CrossRef]
- Wolkers, W.F.; Bochicchio, A.; Selvaggi, G.; Hoekstra, F.A. Fourier Transform Infrared Microspectroscopy Detects Changes in Protein Secondary Structure Associated with Desiccation Tolerance in Developing Maize Embryos. Plant Physiol. 1998, 116, 1169–1177. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zeng, H.Y.; Cai, L.H.; Cai, X.L.; Wang, Y.J.; Li, Y.Q. Structure characterization of protein fractions from lotus (Nelumbo nucifera) seed. J. Mol. Struct. 2011, 1001, 139–144. [Google Scholar] [CrossRef]
- Sánchez-Mendoza, N.A.; Ruiz-Ruiz, J.C.; Dávila, G.; Jiménez-Martínez, C. Propiedades tecnofuncionales y biológicas de harina, aislado y fracciones proteicas mayoritarias de semillas de Inga paterno. CyTAJ Food 2017, 15, 400–408. [Google Scholar] [CrossRef] [Green Version]
- Taiz, L.; Zeiger, E. Fisiología Vegetal; Universitat Jaume: Castellón de la Plana, Spain, 2006; Volume 1, 782p. [Google Scholar]
- Smith, M.R.; Rao, I.M.; Merchant, A. Source-Sink Relationships in Crop Plants and Their Influence on Yield Development and Nutritional Quality. Front. Plant Sci. 2018, 9, 1–10. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hussain, M.I.; Al-Dakheel, A.J.; Reigosa, M.J. Genotypic differences in agro-physiological, biochemical and isotopic responses to salinity stress in quinoa (Chenopodium quinoa Willd.) plants: Prospects for salinity tolerance and yield stability. Plant Physiol. Biochem. 2018, 129, 411–420. [Google Scholar] [CrossRef] [PubMed]
- Cai, Z.-Q.; Gao, Q. Comparative physiological and biochemical mechanisms of salt tolerance in five contrasting highland quinoa cultivars. BMC Plant Biol. 2020, 20, 70. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, L.; Lietz, G.; Seal, C.J. Phenolic, apparent antioxidant and nutritional composition of quinoa (Chenopodium quinoa Willd.) seeds. Int. J. Food Sci. Technol. 2021, 56, 3245–3254. [Google Scholar] [CrossRef]
- Choque-Quispe, D.; Ligarda-Samanez, C.A.; Ramos-Pacheco, B.S.; Leguía-Damiano, S.; Calla-Florez, M.; Zamalloa-Puma, L.M.; Colque-Condeña, L. Phenolic compounds, antioxidant capacity, and protein content of three varieties of germinated quinoa (Chenopodium quinoa willd). Ing. Investig. 2021, 41, 1–7. [Google Scholar]
- Paśko, P.; Bartoń, H.; Zagrodzki, P.; Gorinstein, S.; Fołta, M.; Zachwieja, Z. Anthocyanins, total polyphenols and antioxidant activity in amaranth and quinoa seeds and sprouts during their growth. Food Chem. 2009, 115, 994–998. [Google Scholar] [CrossRef]
- Ballester-Sánchez, J.; Gil, J.V.; Haros, C.M.; Fernández-Espinar, M.T. Effect of Incorporating White, Red or Black Quinoa Flours on Free and Bound Polyphenol Content, Antioxidant Activity and Colour of Bread. Plant Foods Hum. Nutr. 2019, 74, 185–191. [Google Scholar] [CrossRef] [Green Version]
- Tang, Y.; Tsao, R. Phytochemicals in quinoa and amaranth grains and their antioxidant, anti-inflammatory, and potential health beneficial effects: A review. Mol. Nutr. Food Res. 2017, 61, 1–16. [Google Scholar] [CrossRef]
- Hinojosa, L.; González, J.; Barrios-Masias, F.; Fuentes, F.; Murphy, K. Quinoa Abiotic Stress Responses: A Review. Plants 2018, 7, 106. [Google Scholar] [CrossRef] [Green Version]
- Šamec, D.; Karalija, E.; Šola, I.; Vujcic, V.; Salopek-Sondi, B. The Role of Polyphenols in Abiotic Stress Response: The Influence of Molecular Structure. Plants 2021, 10, 118. [Google Scholar] [CrossRef]
- Rasul, A.; Millimouno, F.M.; Eltayb, W.A.; Ali, M.; Li, J.; Li, X. Pinocembrin: A Novel Natural Compound with Versatile Pharmacological and Biological Activities. BioMed Res. Int. 2013, 2013, 379850. [Google Scholar] [CrossRef]
- Sarker, U.; Oba, S. Salinity stress enhances color parameters, bioactive leaf pigments, vitamins, polyphenols, flavonoids and antioxidant activity in selected Amaranthus leafy vegetables. J. Sci. Food Agric. 2018, 99, 2275–2284. [Google Scholar] [CrossRef]
- Antognoni, F.; Potente, G.; Biondi, S.; Mandrioli, R.; Marincich, L.; Ruiz, K.B. Free and conjugated phenolic profiles and antioxidant activity in quinoa seeds and their relationship with genotype and environment. Plants 2021, 10, 1046. [Google Scholar] [CrossRef] [PubMed]
- Carciochi, R.A.; Manrique, G.D.; Dimitrov, K. Changes in phenolic composition and antioxidant activity during germination of quinoa seeds (Chenopodium quinoa Willd.). Int. Food Res. J. 2014, 21, 767–773. [Google Scholar]
- Manjarres-Hernández, E.; Morillo-Coronado, A.; Ojeda-perez, Z.; Cárdenas-Chaparro, A.; Arias-Moreno, D. Characterizacion of the yield components and selection of materials for breeding programs of quinoa (Chenopodium quinoa Willd.). Euphytica 2021, 217, 101. [Google Scholar] [CrossRef]
- Landinez-Torres, A.; Panelli, S.; Picco, A.M.; Comandatore, F.; Tosi, S.; Capelli, E. A meta-barcoding analysis of soil mycobiota of the upper Andean Colombian agro-environment. Sci. Rep. 2019, 9, 10085. [Google Scholar] [CrossRef] [PubMed] [Green Version]

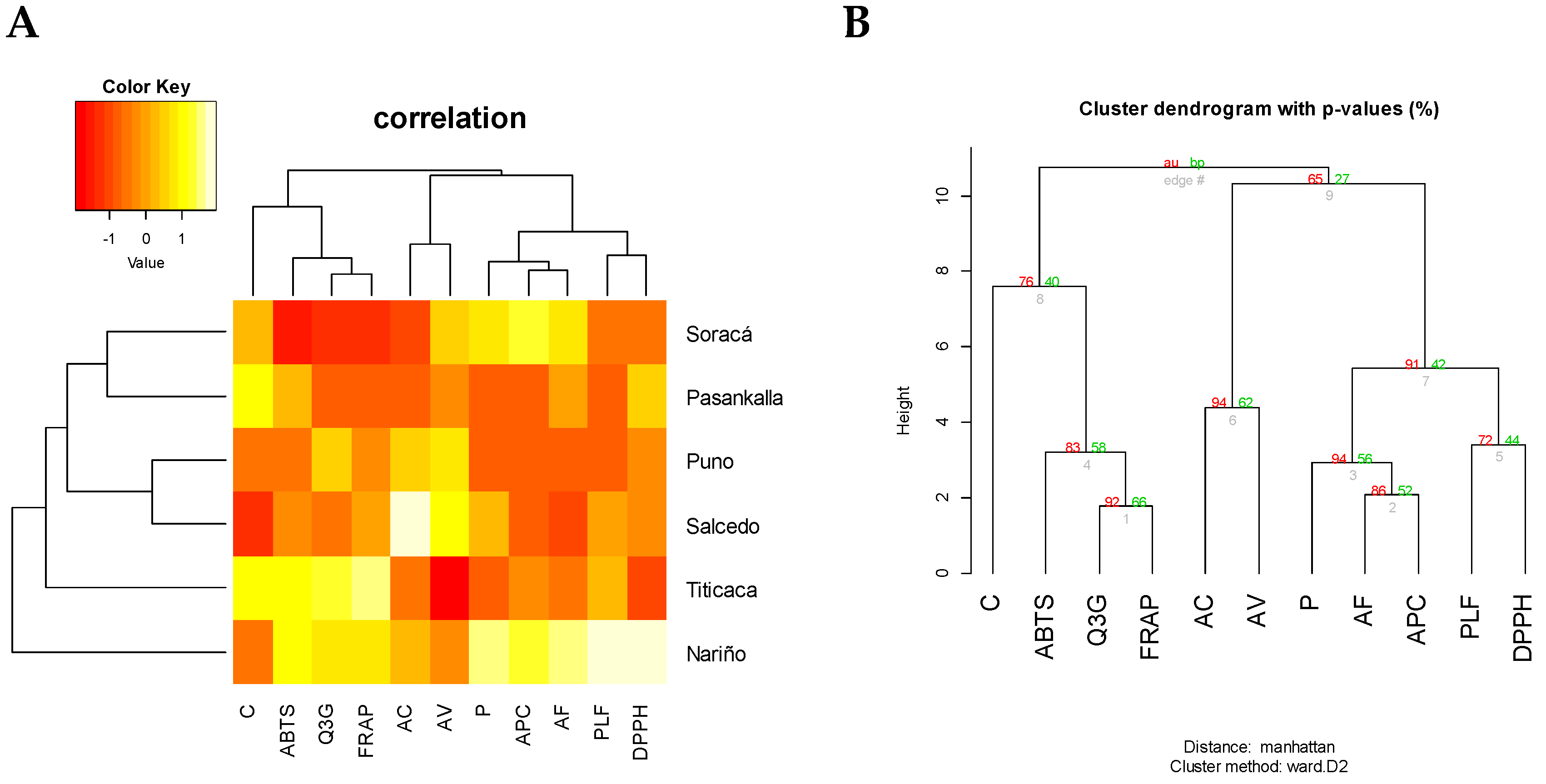
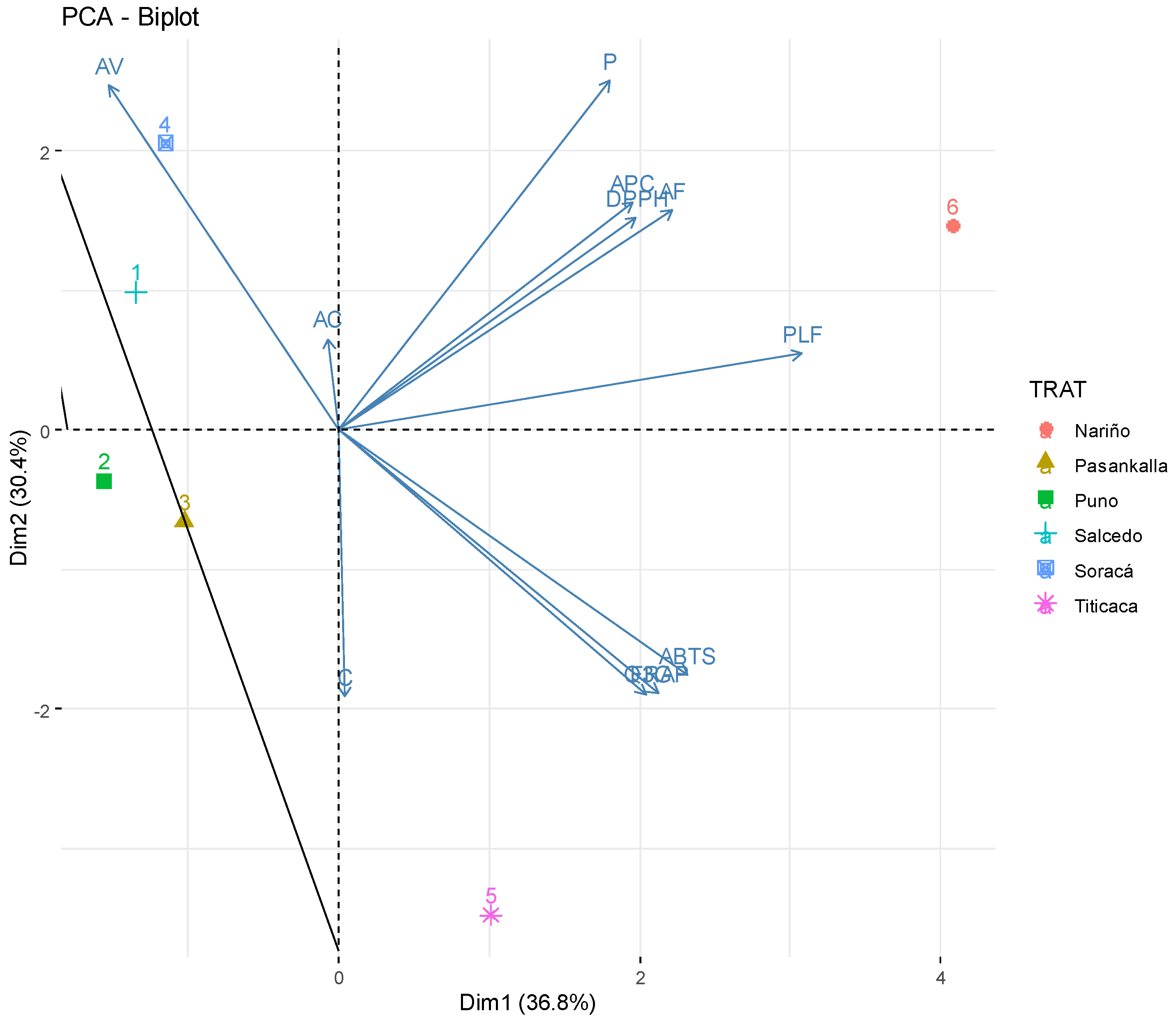


| Cultivars | L* | a* | b* | Protein | Carbohydrates | Fat |
|---|---|---|---|---|---|---|
| Titicaca | 53.50 ± 0.58 d | 4.05 ± 0.08 b | 23.84 ± 0.04 b | 14.63 ± 0.30 a | 49.36 ± 0.70 c | 5.7 ± 0.30 c |
| Salcedo | 73.59 ± 0.52 a | 1.51 ± 0.03 e | 18.14 ± 0.05 e | 13.36 ± 0.47 b | 59.63 ± 0.47 ab | 6.76 ± 0.25 ab |
| Soracá | 69.40 ± 0.90 b | 1.93 ± 0.16 d | 21.12 ± 0.21 c | 14.10 ± 0.26 ab | 57.30 ± 0.70 b | 6.1 ± 0.1 c |
| Pasankalla | 30.78 ± 0.46 e | 10.4 ± 0.24 a | 11.92 ± 0.26 f | 14. 46 ± 0.14 a | 57.30 ± 1.05 b | 6.76 ± 0.25 ab |
| Puno | 54.03 ± 0.22 d | 1.32 ± 0.19 e | 18.99 ± 0.16 d | 11.73 ± 0.30 c | 61.06 ± 0.51 a | 7 ± 0.1 a |
| Nariño | 62.29 ± 0.34 c | 3.56 ± 0.04 c | 25.59 ± 0.04 a | 11.36 ± 0.30 c | 62.53 ± 2.05 a | 6.26 ± 0.15 bc |
| Cultivars | C-H Stretching (2922 cm−1) | C-H Stretching; C-O-C; C-O Bending (1016 cm−1) | C=O Stretching (1633 cm−1) |
|---|---|---|---|
| Titicaca | 0.65 ± 0.02 b | 0.0011 ± 0.00006 c | 0.47 ± 0.04 b |
| Salcedo | 0.68 ± 0.01 ab | 0.0021 ± 0.0001 b | 0.34 ± 0.06 c |
| Soracá | 0.69 ± 0.008 ab | 0.003 ± 0.0004 a | 0.55 ± 0.04 ab |
| Puno | 0.68 ± 0.04 ab | 0.0009 ± 0.0004 c | 0.30 ± 0.02 c |
| Pasankalla | 0.76 ± 0.04 a | 0.0031 ± 0.0001 a | 0.61 ± 0.03 a |
| Nariño | 0.68 ± 0.01 ab | 0.0021 ± 0.0001 b | 0.32 ± 0.005 c |
| Cultivars | β-Sheet-1 (1624 cm−1) | β-Sheet-2 (1627 cm−1) | β-Sheet-3 (1635 cm−1) | Random Coil (1648 cm−1) | α Elice (1656 cm−1) | β-Turns-1 (1667 cm−1) | β-Turns-2 (1675 cm−1) | β-Turns-3 (1680 cm−1) |
|---|---|---|---|---|---|---|---|---|
| Titicaca | 2.99 ± 0.13 a | 2.87 ± 0.07 a | 1.09 ± 0.08 c | 3.08 ± 0.07 b | 1.33 ± 0.07 e | 3.53 ± 0.06 a | 1.36 ± 0.05 b | 0.26 ± 0.05 e |
| Salcedo | 2.14 ± 0.05 b | 1.89 ± 0.06 c | 3.08 ± 0.07 b | 3.35 ± 0.14 ab | 3.07 ± 0.07 b | 2.14 ± 0.14 b | 0.92 ± 0.08 d | 0.77 ± 0.05 c |
| Soracá | 0.37 ± 0.04 e | 1.49 ± 0.04 d | 3.44 ± 0.12 a | 3.59 ± 0.14 a | 4.97 ± 0.09 a | 1.46 ± 0.07 c | 0.59 ± 0.01 e | 0.33 ± 0.01 de |
| Puno | 0.75 ± 0.04 d | 0.17 ± 0.02 e | 1.11 ± 0.19 c | 1.24 ± 0.1 d | 2.56 ± 0.14 c | 1.66 ± 0.09 c | 1.07 ± 0.04 c | 1.31 ± 0.01 b |
| Pasankalla | 0.58 ± 0.02 de | 0.08 ± 0.009 e | 0.95 ± 0.04 c | 2.53 ± 0.1 c | 0.14 ± 0.009 f | 1.47 ± 0.05 c | 0.51 ± 0.03 e | 0.5 ± 0.08 d |
| Nariño | 1.1 ± 0.12 c | 2.36 ± 0.1 b | 3.38 ± 0.13 ab | 0.72 ± 0.09 e | 1.73 ± 0.07 e | 0.57 ± 0.1 d | 1.9 ± 0.01 a | 3.37 ± 0.09 a |
| Cultivars | 996 cm−1 | 1014 cm−1 | 1041 cm−1 | 1076 cm−1 | 1099 cm−1 | 1145 cm−1 | 996/1014 | 1041/1014 |
|---|---|---|---|---|---|---|---|---|
| Titicaca | 0.9 ± 0.01 b | 0.11 ± 0.01 c | 0.14 ± 0.004 c | 0.99 ± 0.007 a | 0.39 ± 0.01 ab | 0.53 ± 0.01 a | 8.23 ± 0.79 b | 1.3 ± 0.08 d |
| Salcedo | 0.98 ± 0.01 a | 0.06 ± 0.004 d | 0.13 ± 0.006 c | 0.96 ± 0.02 b | 0.38 ± 0.01 ab | 0.46 ± 0.01 b | 15.28 ± 1.1 a | 2.04 ± 0.12 b |
| Soracá | 0.82 ± 0.01 c | 0.06 ± 0.01 d | 0.26 ± 0.01 b | 0.98 ± 0.005 a | 0.33 ± 0.03 c | 0.53 ± 0.02 a | 14 ± 2.1 a | 4.02 ± 0.27 a |
| Puno | 0.97 ± 0.01 a | 0.16 ± 0.01 b | 0.23 ± 0.01 b | 0.86 ± 0.005 c | 0.34 ± 0.01 bc | 0.4 ± 0.01 c | 6.11 ± 0.32 bc | 1.48 ± 0.16 cd |
| Pasankalla | 0.81 ± 0.01 c | 0.17 ± 0.01 b | 0.32 ± 0.004 a | 0.99 ± 0.002 a | 0.31 ± 0.05 c | 0.53 ± 0.01 a | 4.81 ± 0.34 c | 1.91 ± 0.1 bc |
| Nariño | 0.98 ± 0.01 a | 0.22 ± 0.02 a | 0.34 ± 0.01 a | 0.97 ± 0.005 ab | 0.41 ± 0.01 a | 0.52 ± 0.01 a | 4.51 ± 0.53 c | 1.58 ± 0.16 bcd |
| Cultivars | Polyphenols (mg AG/g) | ABTS (µmol de T/g) | DPPH (µmol de T /g) | FRAP (µmol de AA/g) |
|---|---|---|---|---|
| Titicaca | 1.0409 ± 0.0202 b | 10.8536 ± 0.1317 a | 2.3488 ± 0.0177 d | 3.2626 ± 0.0585 a |
| Salcedo | 0.9303 ± 0.0103 c | 8.2387 ± 0.2718 c | 2.7893 ± 0.0627 c | 1.9886 ± 0.0643 c |
| Soracá | 0.735 ± 0.0212 d | 5.6546 ± 0.1075 d | 2.6638 ± 0.0648 c | 0.8063 ± 0.0215 f |
| Puno | 0.6782 ± 0.0198 e | 7.9468 ± 0.1222 c | 2.8127 ± 0.0830 c | 1.6469 ± 0.0159 d |
| Pasankalla | 0.6681 ± 0.001 e | 9.4534 ± 0.3435 b | 3.2587 ± 0.0697 b | 1.2685 ± 0.0373 e |
| Nariño | 1.7737 ± 0.009 a | 11.0023 ± 0.0857 a | 3.8935 ± 0.0454 a | 2.6356 ± 0.0212 b |
| Cultivars | Caffeine | Caffeic Acid | Vanillic Acid | p-Coumaric Acid | Ferulic Acid | Pinocembrin | Quercetin-3-Glucoside |
|---|---|---|---|---|---|---|---|
| Salcedo | 0.12 ± 0.002 d | 2.41 ± 0.02 a | 4.02 ± 0.01 a | 1.92 ± 0.02 d | 5.58 ± 0.1 f | 0.04 ± 0.01 bc | 0.53 ± 0.09 c |
| Puno | 0.13 ± 0.003 c | 1.88 ± 0.01 b | 3.66 ± 0.01 b | 2.04 ± 0.02 d | 6.71 ± 0.09 e | 0.01 ± 0.01 c | 1.23 ± 0.11 b |
| Pasankalla | 0.15 ± 0.003 a | 1.43 ± 0.02 d | 3.02 ± 0.01 e | 2.6 ± 0.02 c | 9.26 ± 0.1 c | 0.01 ± 0.01 c | 0.37 ± 0.14 cd |
| Soracá | 0.14 ± 0.003 b | 1.26 ± 0.01 e | 3.49 ± 0.01 c | 14 ± 0.2 a | 11.83 ± 0.09 b | 0.05 ± 0.02 ab | 0.1 ± 0.1 d |
| Titicaca | 0.15 ± 0.003 a | 1.49 ± 0.01 c | 1.94 ± 0.02 f | 5.53 ± 0.2 b | 7.42 ± 0.1 d | 0.01 ± 0.009 c | 1.79 ± 0.12 a |
| Nariño | 0.13 ± 0.002 c | 1.84 ± 0.01 b | 3.07 ± 0.01 d | 14.41 ± 0.02 a | 14.67 ± 0.1 a | 0.07 ± 0.01 a | 1.49 ± 0.11 b |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
García-Parra, M.; Roa-Acosta, D.; García-Londoño, V.; Moreno-Medina, B.; Bravo-Gomez, J. Structural Characterization and Antioxidant Capacity of Quinoa Cultivars Using Techniques of FT-MIR and UHPLC/ESI-Orbitrap MS Spectroscopy. Plants 2021, 10, 2159. https://doi.org/10.3390/plants10102159
García-Parra M, Roa-Acosta D, García-Londoño V, Moreno-Medina B, Bravo-Gomez J. Structural Characterization and Antioxidant Capacity of Quinoa Cultivars Using Techniques of FT-MIR and UHPLC/ESI-Orbitrap MS Spectroscopy. Plants. 2021; 10(10):2159. https://doi.org/10.3390/plants10102159
Chicago/Turabian StyleGarcía-Parra, Miguel, Diego Roa-Acosta, Víctor García-Londoño, Brigitte Moreno-Medina, and Jesús Bravo-Gomez. 2021. "Structural Characterization and Antioxidant Capacity of Quinoa Cultivars Using Techniques of FT-MIR and UHPLC/ESI-Orbitrap MS Spectroscopy" Plants 10, no. 10: 2159. https://doi.org/10.3390/plants10102159
APA StyleGarcía-Parra, M., Roa-Acosta, D., García-Londoño, V., Moreno-Medina, B., & Bravo-Gomez, J. (2021). Structural Characterization and Antioxidant Capacity of Quinoa Cultivars Using Techniques of FT-MIR and UHPLC/ESI-Orbitrap MS Spectroscopy. Plants, 10(10), 2159. https://doi.org/10.3390/plants10102159








