Microbial Fortification Improved Photosynthetic Efficiency and Secondary Metabolism in Lycopersicon esculentum Plants Under Cd Stress
Abstract
1. Introduction
2. Materials and Methods
2.1. Bacterial Strains
2.2. Plant Growth and Treatments
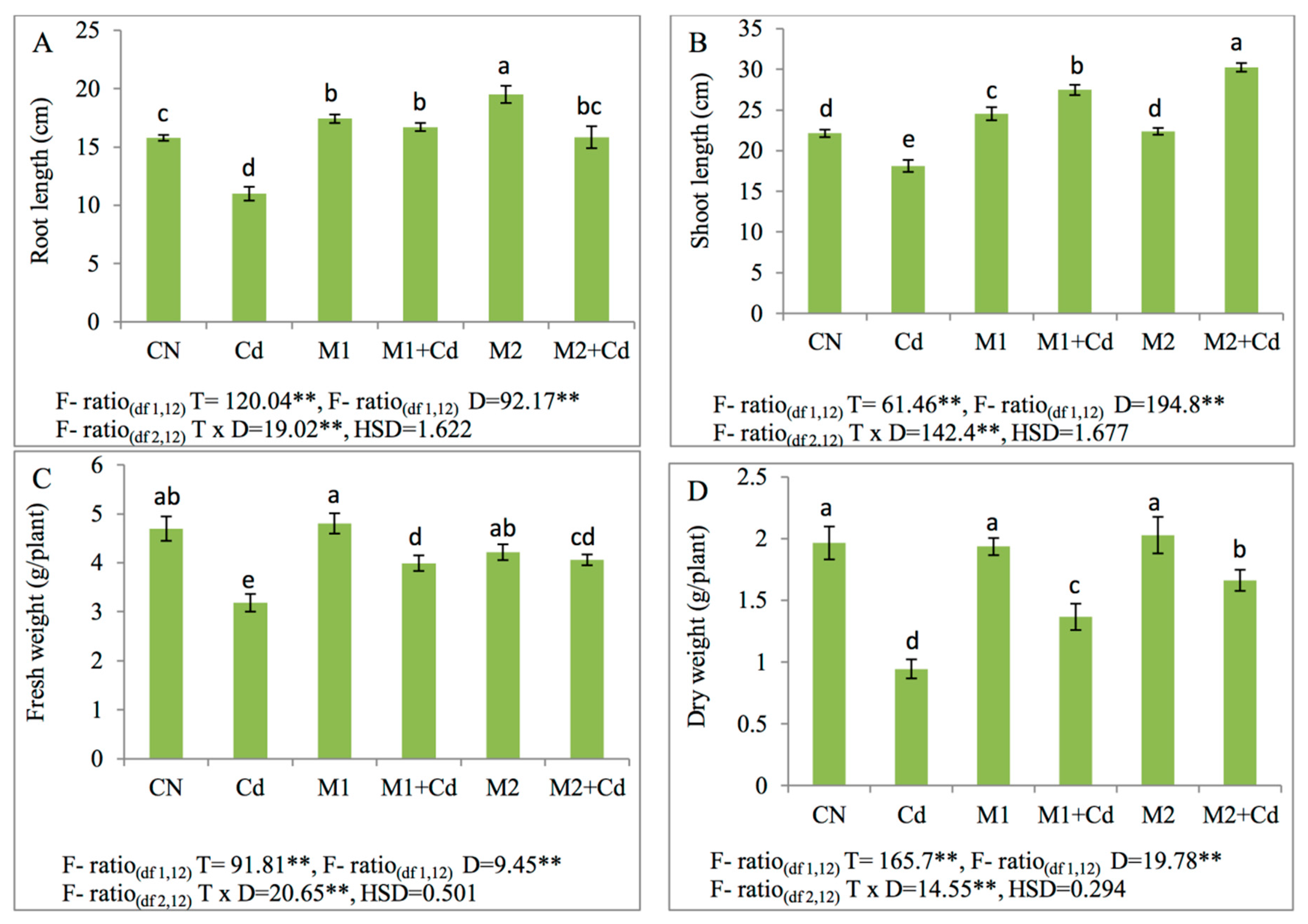
2.3. Growth and Biomass Yield
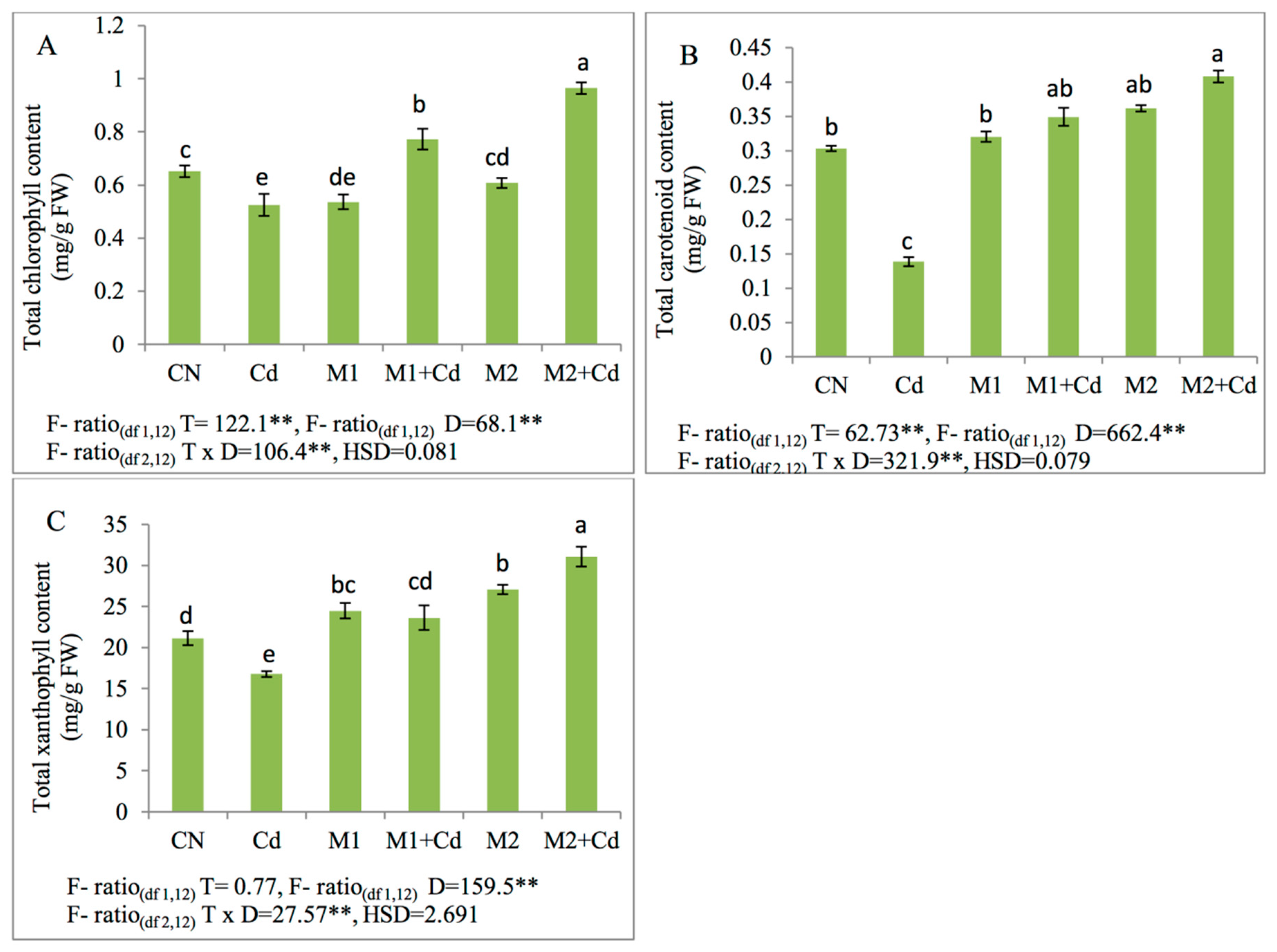
2.4. Total Chlorophyll, Carotenoid Content and Total Xanthophyll Content
2.5. Gas Exchange Parameters
2.6. Phenolic Compounds
2.6.1. Total Phenols
2.6.2. Total Flavonoids
2.6.3. Total Anthocyanins
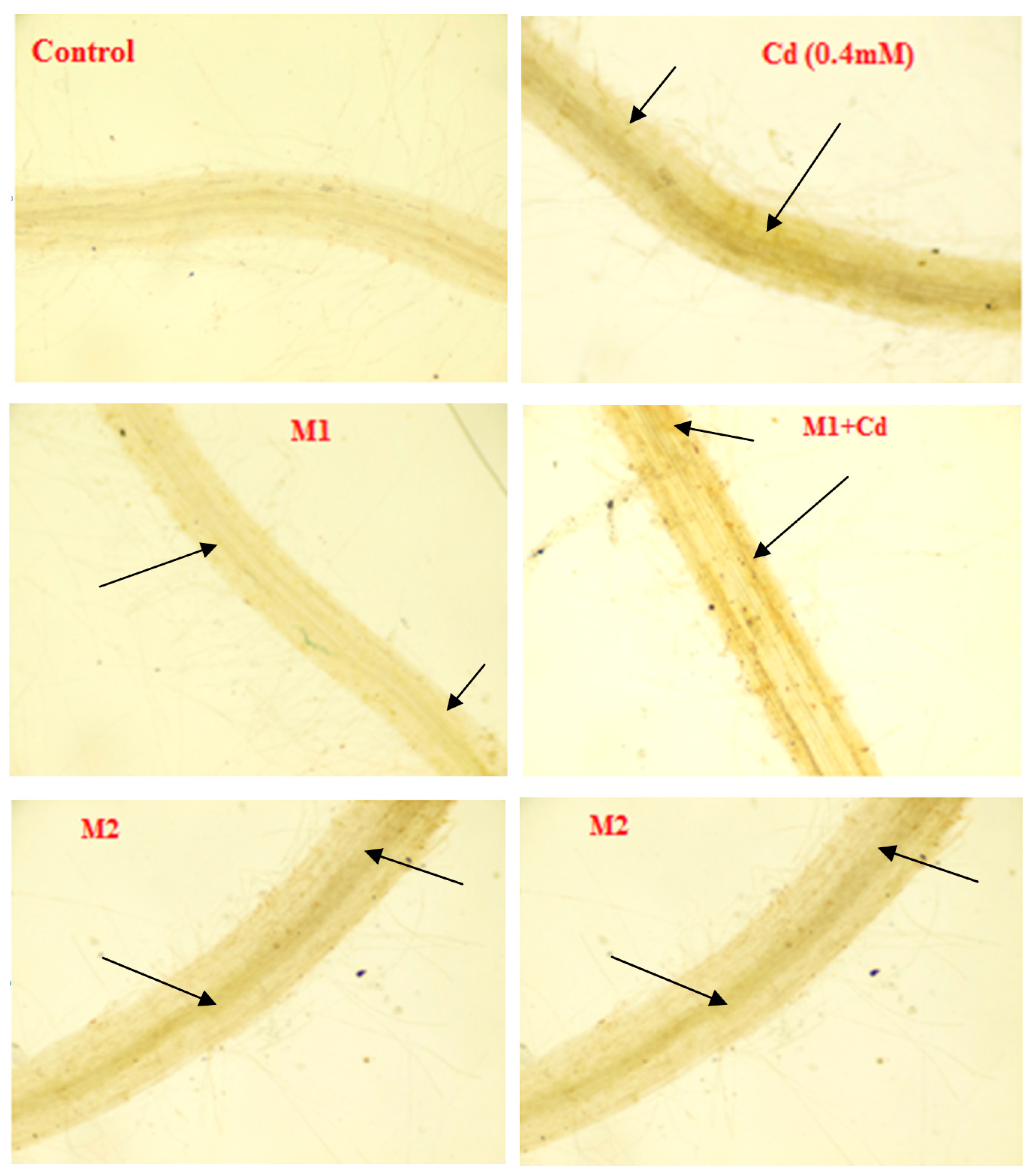
2.6.4. In Situ Localization Studies of Phenols
2.7. Osmoprotectants
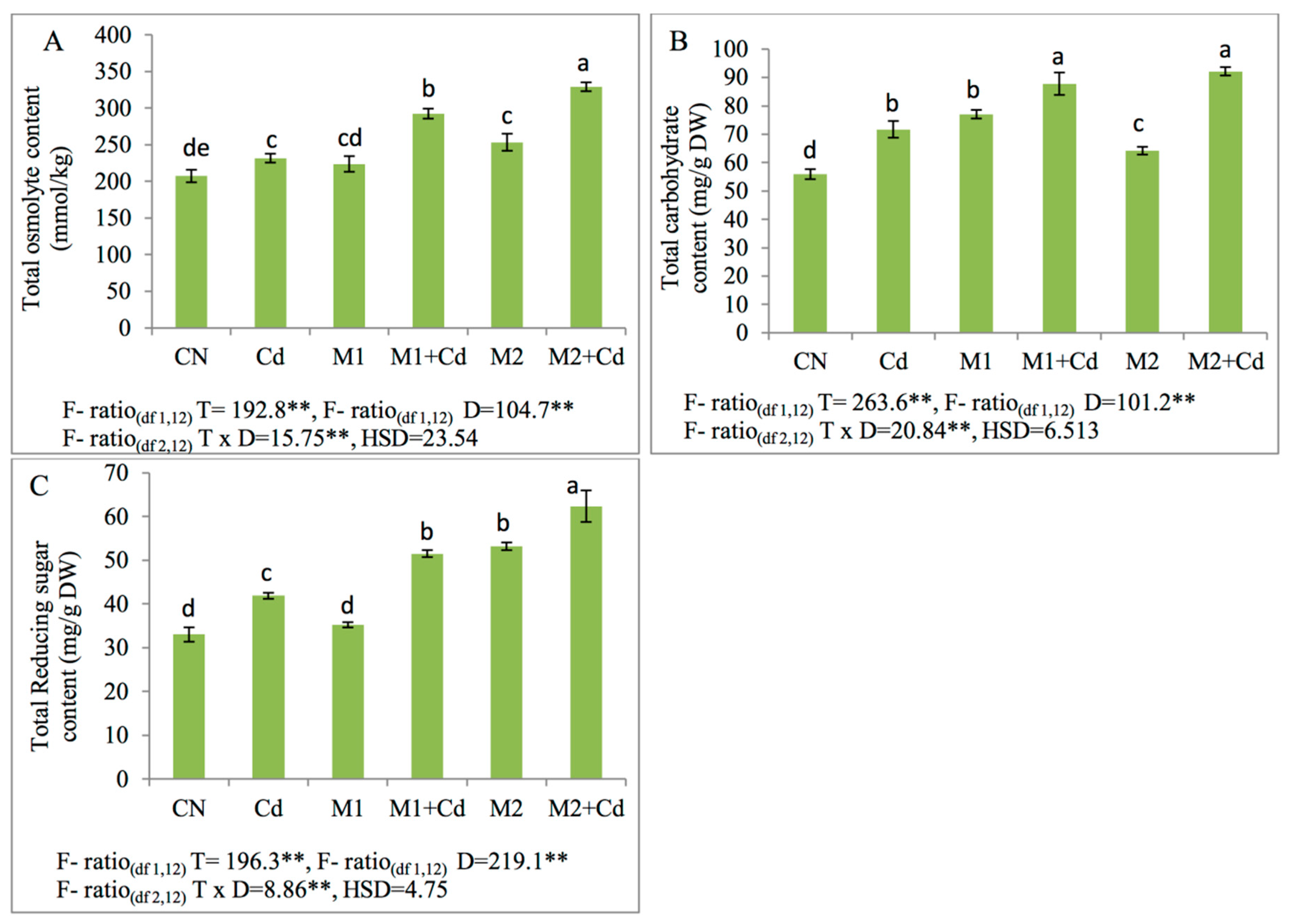
2.7.1. Total Osmolytes
2.7.2. Total Carbohydrates
2.7.3. Total Reducing Sugars
2.7.4. Trehalose Content
2.7.5. Glycine Betaine Content
2.7.6. Proline Content
2.7.7. Free Amino Acid Content
2.8. Statistical Analysis
3. Results
3.1. Growth and Biomass Yield
3.2. Photosynthetic Pigments
3.3. Gas Exchange Attributes
3.4. Phenolic Compounds
3.5. Histochemical Studies of Phenol Tagging
3.6. Osmoprotectants
3.6.1. Effect of Microbial Inoculations Upon Total Osmolytes, Carbohydrates, Reducing Sugars
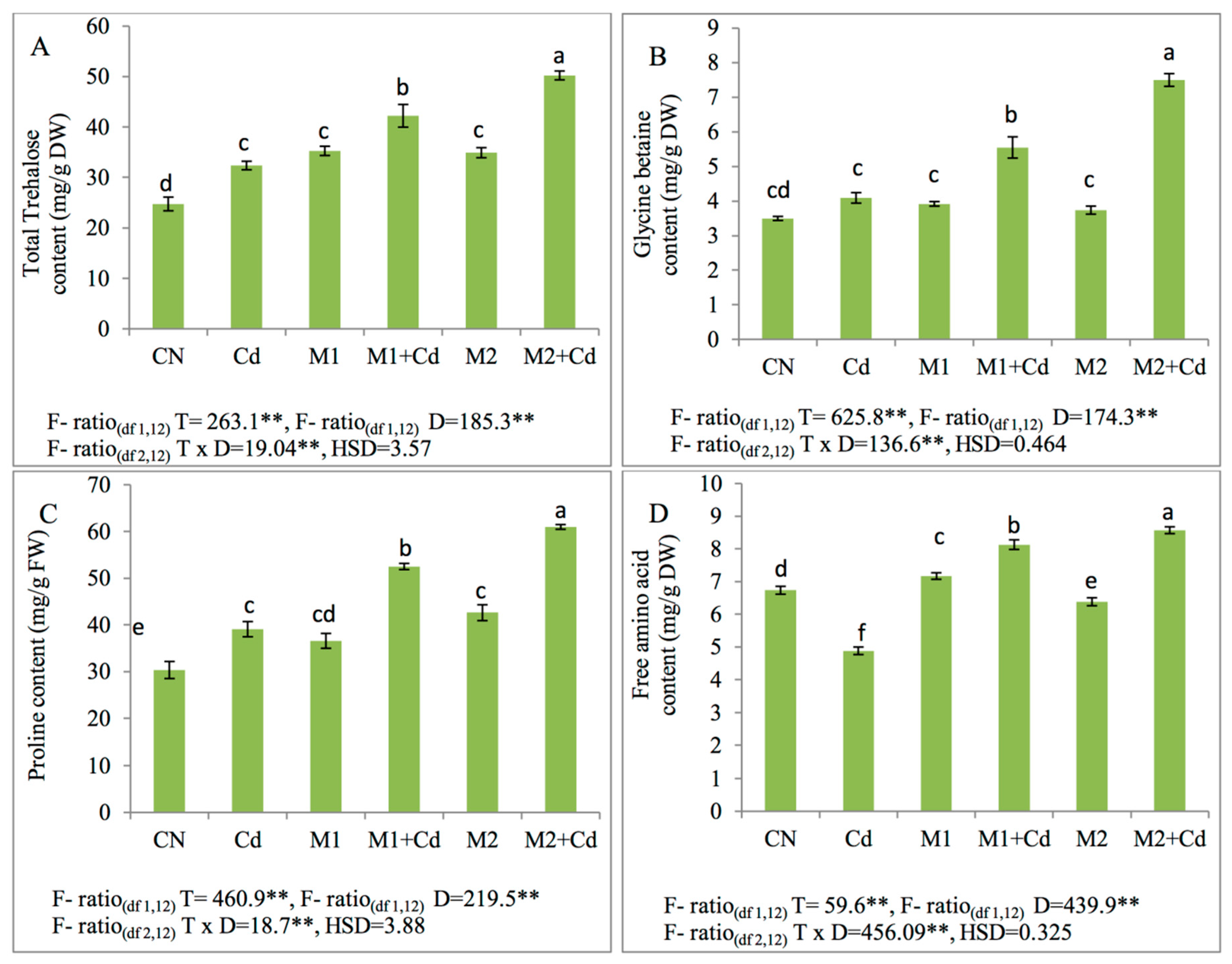
3.6.2. Effect of Micro-Organisms Upon Trehalose, Glycine betaine, Proline, and Free Amino Acid Contents
4. Discussion
5. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Shahid, M.; Javed, M.T.; Mushtaq, A.; Akram, M.S.; Mahmood, F.; Ahmed, T.; Noman, M.; Azeem, M. Microbe-Mediated Mitigation of Cadmium Toxicity in Plants. In Cadmium Toxicity and Tolerance in Plants; Elsevier: Amsterdam, The Netherlands, 2019; pp. 427–449. [Google Scholar] [CrossRef]
- Tran, T.A.; Popova, L.P. Functions and toxicity of cadmium in plants: Recent advances and future prospects. Turk. J. Bot. 2013, 37, 1–13. [Google Scholar]
- Dugar, G.; Herbig, A.; Förstner, K.U.; Heidrich, N.; Reinhardt, R.; Nieselt, K.; Sharma, C.M. High-Resolution Transcriptome Maps Reveal Strain-Specific Regulatory Features of Multiple Campylobacter jejuni Isolates. PLoS Genet. 2013, 9, e1003495. [Google Scholar] [CrossRef]
- Moradi, A.; Abbaspour, K.C.; Afyuni, M. Modelling field-scale cadmium transport below the root zone of a sewage sludge amended soil in an arid region in Central Iran. J. Contam. Hydrol. 2005, 79, 187–206. [Google Scholar] [CrossRef]
- Gupta, A.; Gupta, R.; Singh, R.L. Microbes and environment. In Principles and Applications of Environmental Biotechnology for a Sustainable Future; Singh, R.L., Ed.; Springer: Singapore, 2017; pp. 43–84. [Google Scholar]
- Nymark, M.; Sharma, A.K.; Sparstad, T.; Bones, A.M.; Winge, P. A CRISPR/Cas9 system adapted for gene editing in marine algae. Sci. Rep. 2016, 6. [Google Scholar] [CrossRef]
- Gallego, S.M.; Pena, L.B.; Barcia, R.A.; Azpilicueta, C.E.; Iannone, M.F.; Rosales, E.P.; Zawoznik, M.S.; Groppa, M.D.; Benavides, M.P. Unravelling cadmium toxicity and tolerance in plants: Insight into regulatory mechanisms. Environ. Exp. Bot. 2012, 83, 33–46. [Google Scholar] [CrossRef]
- Du, Y.; Li, H.; Yin, K.; Zhai, H. Cadmium accumulation, subcellular distribution, and chemical forms in Vitis vinifera cv. Chardonnay grapevine. Ying Yong Sheng Tai Xue Bao = J. Appl. Ecol. 2012, 23, 1607–1612. [Google Scholar]
- Clemens, S.; Aarts, M.G.M.; Thomine, S.; Verbruggen, N. Plant science: The key to preventing slow cadmium poisoning. Trends Plant Sci. 2013, 18, 92–99. [Google Scholar] [CrossRef]
- Bertin, G.; Averbeck, D. Cadmium: Cellular effects, modifications of biomolecules, modulation of DNA repair and genotoxic consequences (a review). Biochimie 2006, 88, 1549–1559. [Google Scholar] [CrossRef]
- Nawrot, T.; Plusquin, M.; Hogervorst, J.; Roels, H.A.; Celis, H.; Thijs, L.; Vangronsveld, J.; Van Hecke, E.; Staessen, J.A. Environmental exposure to cadmium and risk of cancer: A prospective population-based study. Lancet Oncol. 2006, 7, 119–126. [Google Scholar] [CrossRef]
- DalCorso, G.; Farinati, S.; Maistri, S.; Furini, A. How Plants Cope with Cadmium: Staking All on Metabolism and Gene Expression. J. Integr. Plant Biol. 2008, 50, 1268–1280. [Google Scholar] [CrossRef]
- Sawidis, T. Effect of cadmium on pollen germination and tube growth in Lilium longiflorum and Nicotiana tabacum. Protoplasma 2008, 233, 95–106. [Google Scholar] [CrossRef]
- Sandalio, L.M.; Dalurzo, H.C.; Gómez, M.; Romero-Puertas, M.C.; del Río, L.A. Cadmium-induced changes in the growth and oxidative metabolism of pea plants. J. Exp. Bot. 2001, 52, 2115–2126. [Google Scholar] [CrossRef]
- Zhang, W. Essential and redundant functions of histone acetylation revealed by mutation of target lysines and loss of the Gcn5p acetyltransferase. EMBO J. 1998, 17, 3155–3167. [Google Scholar] [CrossRef]
- Shinozaki, K.; Yamaguchi-Shinozaki, K. Molecular responses to dehydration and low temperature: Differences and cross-talk between two stress signaling pathways. Curr. Opin. Plant Biol. 2000, 3, 217–223. [Google Scholar] [CrossRef]
- Hauser, M.-T. Molecular basis of natural variation and environmental control of trichome patterning. Front. Plant Sci. 2014, 5. [Google Scholar] [CrossRef]
- Dalvi, A.A.; Bhalerao, S.A. Response of plants towards heavy metal toxicity: An overview of avoidance, tolerance and uptake mechanism. Ann. Plant Sci. 2013, 2, 362–368. [Google Scholar]
- Rajkumar, M.; Sandhya, S.; Prasad, M.N.V.; Freitas, H. Perspectives of plant-associated microbes in heavy metal phytoremediation. Biotechnol. Adv. 2012, 30, 1562–1574. [Google Scholar] [CrossRef]
- Sessitsch, A.; Kuffner, M.; Kidd, P.; Vangronsveld, J.; Wenzel, W.W.; Fallmann, K.; Puschenreiter, M. The role of plant-associated bacteria in the mobilization and phytoextraction of trace elements in contaminated soils. Soil Biol. Biochem. 2013, 60, 182–194. [Google Scholar] [CrossRef]
- Merdy, P.; Gharbi, L.T.; Lucas, Y. Pb, Cu and Cr interactions with soil: Sorption experiments and modelling. Colloids Surf. Phys. Eng. Asp. 2009, 347, 192–199. [Google Scholar] [CrossRef]
- Yan, K.; Chen, P.; Shao, H.; Zhang, L.; Xu, G. Effects of Short-Term High Temperature on Photosynthesis and Photosystem II Performance in Sorghum. J. Agron. Crop. Sci. 2011, 197, 400–408. [Google Scholar] [CrossRef]
- Székely, G.; Ábrahám, E.; Cséplő, Á.; Rigó, G.; Zsigmond, L.; Csiszár, J.; Ayaydin, F.; Strizhov, N.; Jásik, J.; Schmelzer, E. Duplicated P5CS genes of Arabidopsis play distinct roles in stress regulation and developmental control of proline biosynthesis. Plant J. 2008, 53, 11–28. [Google Scholar] [CrossRef] [PubMed]
- Rufino, R.D.; Sarubbo, L.A.; Campos-Takaki, G.M. Enhancement of stability of biosurfactant produced by Candida lipolytica using industrial residue as substrate. World J. Microbiol. Biotechnol. 2007, 23, 729–734. [Google Scholar] [CrossRef]
- Moreno, M.M.; Jaime, V.; Sara, G.M.; Carmen, M. Response of healthy local tomato (Solanum lycopersicum L.) populations to grafting in organic farming. Sci. Rep. 2019, 9, 4592. [Google Scholar] [CrossRef] [PubMed]
- Ghaffari, H.; RezaGhassemzadeh, H.; Sadeghi, M.; Alijani, S. Some physical, mechanical and chemical properties of tomato fruit related to mechanical damage and bruising models. Biol. Forum 2015, 7, 712–718. [Google Scholar]
- Abdel-Monaim, M. Induced systemic resistance in tomato plants against Fusarium wilt disease. Int. Res. J. Microbiol. 2012, 3, 14–23. [Google Scholar]
- Arthur, E.; Crews, H.; Morgan, C. Optimizing plant genetic strategies for minimizing environmental contamination in the food chain. Int. J. Phytoremediation 2000, 2, 1–21. [Google Scholar] [CrossRef]
- Khanna, K.; Jamwal, V.L.; Kohli, S.K.; Gandhi, S.G.; Ohri, P.; Bhardwaj, R.; Abd_Allah, E.F.; Hashem, A.; Ahmad, P. Plant growth promoting rhizobacteria induced Cd tolerance in Lycopersicon esculentum through altered antioxidative defense expression. Chemosphere 2019, 217, 463–474. [Google Scholar] [CrossRef]
- Dourado, M.N.; Souza, L.A.; Martins, P.F.; Peters, L.P.; Piotto, F.A.; Azevedo, R.A. Burkholderia sp. SCMS54 Triggers a Global Stress Defense in Tomato Enhancing Cadmium Tolerance. Water Air Soil Pollut. 2014, 225. [Google Scholar] [CrossRef]
- Khanna, K.; Jamwal, V.L.; Sharma, A.; Gandhi, S.G.; Ohri, P.; Bhardwaj, R.; Al-Huqail, A.A.; Siddiqui, M.H.; Ali, H.M.; Ahmad, P. Supplementation with plant growth promoting rhizobacteria (PGPR) alleviates cadmium toxicity in Solanum lycopersicum by modulating the expression of secondary metabolites. Chemosphere 2019, 230, 628–639. [Google Scholar] [CrossRef]
- Arnon, D.I. Copper enzymes in isolated chloroplasts, polyphenoxidase in Beta vulgaris. Plant Physiol. 1949, 24, 1–15. [Google Scholar] [CrossRef]
- Maclachlan, S.; Zalik, S. Plastid structure, chlorophyll concentration and free amino acid composition of a chlorophyll mutant of barley. Can. J. Bot. 1963, 41, 1053–1062. [Google Scholar] [CrossRef]
- Lawrence, J. Determination of total xanthophyll and marigold oleoresin. J. Assoc. Off. Anal. Chem. 1990, 2, 970–975. [Google Scholar]
- Singleton, V.L.; Rossi, J.A. Colorimetry of total phenolics with phosphomolybdic-phosphotungstic acid reagents. Am. J. Enol. Vitic. 1965, 16, 144–158. [Google Scholar]
- Zhishen, J.; Mengcheng, T.; Jianming, W. The determination of flavonoid contents in mulberry and their scavenging effects on superoxide radicals. Food Chem. 1999, 64, 555–559. [Google Scholar] [CrossRef]
- Mancinelli, A.L. Photoregulation of Anthocyanin Synthesis. Plant Physiol. 1984, 75, 447–453. [Google Scholar] [CrossRef]
- Gahan, P.B. Plant Microtechnique; McGraw-Hill Book Company: New York, NY, USA; London, UK, 1984. [Google Scholar]
- Hedge, J.; Hofreiter, B.; Whistler, R. Carbohydrate Chemistry; Academic Press: New York, NY, USA, 1962; p. 17. [Google Scholar]
- Miller, G.L. Use of Dinitrosalicylic Acid Reagent for Determination of Reducing Sugar. Anal. Chem. 1959, 31, 426–428. [Google Scholar] [CrossRef]
- Trevelyan, W.E.; Harrison, J.S. Studies on yeast metabolism. 5. The trehalose content of baker’s yeast during anaerobic fermentation. Biochem. J. 1956, 62, 177–183. [Google Scholar] [CrossRef]
- Grieve, C.M.; Grattan, S.R. Rapid assay for determination of water soluble quaternary ammonium compounds. Plant Soil 1983, 70, 303–307. [Google Scholar] [CrossRef]
- Bates, L.S.; Waldren, R.P.; Teare, I.D. Rapid determination of free proline for water-stress studies. Plant Soil 1973, 39, 205–207. [Google Scholar] [CrossRef]
- Lee, Y.P.; Takahashi, T. An improved colorimetric determination of amino acids with the use of ninhydrin. Anal. Biochem. 1966, 14, 71–77. [Google Scholar] [CrossRef]
- Didwania, N.; Jain, S.; Sadana, D. In-vitro Phytotoxic Effects of Cadmium on Morphological Parameters of Allium cepa. Biological 2019, 12, 137. [Google Scholar]
- Slepecka, K.; Kalwa, K.; Wyrostek, J.; Pankiewicz, U. Evaluation of cadmium, lead, zinc and copper levels in selected ecological cereal food products and their non-ecological counterparts. Curr. Issues Pharm. Med Sci. 2017, 30, 147–150. [Google Scholar] [CrossRef]
- Lanier, C.; Bernard, F.; Dumez, S.; Leclercq-Dransart, J.; Lemière, S.; Vandenbulcke, F.; Nesslany, F.; Platel, A.; Devred, I.; Hayet, A.; et al. Combined toxic effects and DNA damage to two plant species exposed to binary metal mixtures (Cd/Pb). Ecotoxicol. Environ. Saf. 2019, 167, 278–287. [Google Scholar] [CrossRef]
- Dresler, S.; Hawrylak-Nowak, B.; Kováčik, J.; Pochwatka, M.; Hanaka, A.; Strzemski, M.; Sowa, I.; Wójciak-Kosior, M. Allantoin attenuates cadmium-induced toxicity in cucumber plants. Ecotoxicol. Environ. Saf. 2019, 170, 120–126. [Google Scholar] [CrossRef]
- Yahaghi, Z.; Shirvani, M.; Nourbakhsh, F.; Pueyo, J.J. Uptake and effects of lead and zinc on alfalfa (Medicago sativa L.) seed germination and seedling growth: Role of plant growth promoting bacteria. S. Afr. J. Bot. 2019, 124, 573–582. [Google Scholar] [CrossRef]
- Vacheron, J.; Desbrosses, G.; Bouffaud, M.-L.; Touraine, B.; Moënne-Loccoz, Y.; Muller, D.; Legendre, L.; Wisniewski-Dyé, F.; Prigent-Combaret, C. Plant growth-promoting rhizobacteria and root system functioning. Front. Plant Sci. 2013, 4. [Google Scholar] [CrossRef]
- Singh, S.; Kumar, V.; Sidhu, G.K.; Datta, S.; Dhanjal, D.S.; Koul, B.; Janeja, H.S.; Singh, J. Plant growth promoting rhizobacteria from heavy metal contaminated soil promote growth attributes of Pisum sativum L. Biocatal. Agric. Biotechnol. 2019, 17, 665–671. [Google Scholar] [CrossRef]
- Houri, T.; Khairallah, Y.; Zahab, A.A.; Osta, B.; Romanos, D.; Haddad, G. Heavy Metals Accumulation Effects on The Photosynthetic Performance of Geophytes in Mediterranean Reserve. J. King Saud Univ. Sci. 2019. [Google Scholar] [CrossRef]
- Conti, M.E.; Cecchetti, G. Biological monitoring: Lichens as bioindicators of air pollution assessment—A review. Environ. Pollut. 2001, 114, 471–492. [Google Scholar] [CrossRef]
- İnanç, A.L. Chlorophyll: Structural Properties, Health Benefits and Its Occurrence in Virgin Olive Oils. Acad. Food J. Akad. Gida 2011, 9, 26–32. [Google Scholar]
- Govindaraju, M.; Ganeshkumar, R.S.; Suganthi, P.; Muthukumaran, V.R.; Visvanathan, P. Impact assessment of air pollution stress on plant species through biochemical estimations. Int. J. Environ. Ecol. Eng. 2010, 4, 696–699. [Google Scholar]
- Schelbert, S.; Aubry, S.; Burla, B.; Agne, B.; Kessler, F.; Krupinska, K.; Hörtensteiner, S. Pheophytin Pheophorbide Hydrolase (Pheophytinase) Is Involved in Chlorophyll Breakdown during Leaf Senescence in Arabidopsis. Plant Cell 2009, 21, 767–785. [Google Scholar] [CrossRef] [PubMed]
- Khan, A.H.A.; Nawaz, I.; Yousaf, S.; Cheema, A.S.; Iqbal, M. Soil amendments enhanced the growth of Nicotiana alata L. and Petunia hydrida L. by stabilizing heavy metals from wastewater. J. Environ. Manag. 2019, 242, 46–55. [Google Scholar] [CrossRef]
- You, J.; Chan, Z. ROS Regulation During Abiotic Stress Responses in Crop Plants. Front. Plant Sci. 2015, 6. [Google Scholar] [CrossRef] [PubMed]
- McElroy, J.S.; Kopsell, D.A. Physiological role of carotenoids and other antioxidants in plants and application to turfgrass stress management. N. Z. J. Crop Hortic. Sci. 2009, 37, 327–333. [Google Scholar] [CrossRef]
- Khalid, N.; Noman, A.; Aqeel, M.; Masood, A.; Tufail, A. Phytoremediation potential of Xanthium strumarium for heavy metals contaminated soils at roadsides. Int. J. Environ. Sci. Technol. 2019, 16, 2091–2100. [Google Scholar] [CrossRef]
- El-Meihy, R.M.; Abou-Aly, H.E.; Youssef, A.M.; Tewfike, T.A.; El-Alkshar, E.A. Efficiency of heavy metals-tolerant plant growth promoting bacteria for alleviating heavy metals toxicity on sorghum. Environ. Exp. Bot. 2019, 162, 295–301. [Google Scholar] [CrossRef]
- Guo, D.; Ren, C.; Ali, A.; Du, J.; Zhang, Z.; Li, R.; Zhang, Z. Streptomyces pactum and sulfur mediated the antioxidant enzymes in plant and phytoextraction of potentially toxic elements from a smelter-contaminated soils. Environ. Pollut. 2019, 251, 37–44. [Google Scholar] [CrossRef]
- Pinter, M.I.; Salomon, M.V.; Berli, F.; Gil, R.; Bottini, R.; Piccoli, P. Plant growth promoting rhizobacteria alleviate stress by AsIII in grapevine. Agric. Ecosyst. Environ. 2018, 267, 100–108. [Google Scholar] [CrossRef]
- Kapoor, D.; Kaur, S.; Bhardwaj, R. Physiological and biochemical changes in Brassica juncea plants under Cd-induced stress. BioMed Res. Int. 2014, 2014, 726070. [Google Scholar] [CrossRef]
- Velikova, V.; Tsonev, T.; Loreto, F.; Centritto, M. Changes in photosynthesis, mesophyll conductance to CO2, and isoprenoid emissions in Populus nigra plants exposed to excess nickel. Environ. Pollut. 2011, 159, 1058–1066. [Google Scholar] [CrossRef] [PubMed]
- Salisbury, A.B.; Gallagher, F.J.; Caplan, J.S.; Grabosky, J.C. Maintenance of photosynthesis by Betula populifolia in metal contaminated soils. Sci. Total Environ. 2018, 625, 1615–1627. [Google Scholar] [CrossRef] [PubMed]
- Garcia, J.S.; Dalmolin, Â.C.; Cortez, P.A.; Barbeira, P.S.; Mangabeira, P.A.O.; França, M.G.C. Short-term cadmium exposure induces gas exchanges, morphological and ultrastructural disturbances in mangrove Avicennia schaueriana young plants. Mar. Pollut. Bull. 2017, 131, 122–129. [Google Scholar] [CrossRef] [PubMed]
- Kohli, S.K.; Handa, N.; Sharma, A.; Gautam, V.; Arora, S.; Bhardwaj, R.; Alyemeni, M.N.; Wijaya, L.; Ahmad, P. Combined effect of 24-epibrassinolide and salicylic acid mitigates lead (Pb) toxicity by modulating various metabolites in Brassica juncea L. seedlings. Protoplasma 2017, 255, 11–24. [Google Scholar] [CrossRef] [PubMed]
- Wu, K.; Li, J.; Luo, J.; Liu, Y.; Song, Y.; Liu, N.; Rafiq, M.T.; Li, T. Effects of elevated CO2 and endophytic bacterium on photosynthetic characteristics and cadmium accumulation in Sedum alfredii. Sci. Total Environ. 2018, 643, 357–366. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, I.; Akhtar, M.J.; Mehmood, S.; Akhter, K.; Tahir, M.; Saeed, M.F.; Hussain, M.B.; Hussain, S. Combined application of compost and Bacillus sp. CIK-512 ameliorated the lead toxicity in radish by regulating the homeostasis of antioxidants and lead. Ecotoxicol. Environ. Saf. 2018, 148, 805–812. [Google Scholar] [CrossRef]
- Wei, T.; Lv, X.; Jia, H.; Hua, L.; Xu, H.; Zhou, R.; Zhao, J.; Ren, X.; Guo, J. Effects of salicylic acid, Fe(II) and plant growth-promoting bacteria on Cd accumulation and toxicity alleviation of Cd tolerant and sensitive tomato genotypes. J. Environ. Manag. 2018, 214, 164–171. [Google Scholar] [CrossRef]
- Santos-Sánchez, N.F.; Salas-Coronado, R.; Hernández-Carlos, B.; Villanueva-Cañongo, C. Shikimic Acid Pathway in Biosynthesis of Phenolic Compounds. In Plant Physiological Aspects of Phenolic Compounds; IntechOpen: London, UK, 2019. [Google Scholar]
- Williams, R.J.; Spencer, J.P.E.; Rice-Evans, C. Flavonoids: Antioxidants or signalling molecules? Free Radic. Biol. Med. 2004, 36, 838–849. [Google Scholar] [CrossRef]
- Michalak, A. Phenolic compounds and their antioxidant activity in plants growing under heavy metal stress. Pol. J. Environ. Stud. 2006, 15, 523–530. [Google Scholar]
- Posmyk, M.M.; Bałabusta, M.; Wieczorek, M.; Sliwinska, E.; Janas, K.M. Melatonin applied to cucumber (Cucumis sativus L.) seeds improves germination during chilling stress. J. Pineal Res. 2009, 46, 214–223. [Google Scholar] [CrossRef]
- Chen, S.; Wang, Q.; Lu, H.; Li, J.; Yang, D.; Liu, J.; Yan, C. Phenolic metabolism and related heavy metal tolerance mechanism in Kandelia Obovata under Cd and Zn stress. Ecotoxicol. Environ. Saf. 2019, 169, 134–143. [Google Scholar] [CrossRef] [PubMed]
- Manquián-Cerda, K.; Escudey, M.; Zúñiga, G.; Arancibia-Miranda, N.; Molina, M.; Cruces, E. Effect of cadmium on phenolic compounds, antioxidant enzyme activity and oxidative stress in blueberry (Vaccinium corymbosum L.) plantlets grown in vitro. Ecotoxicol. Environ. Saf. 2016, 133, 316–326. [Google Scholar] [CrossRef]
- Bota, C.; Deliu, C. The effect of copper sulphate on the production of flavonoids in Digitalis lanata cell cultures. Farmacia 2011, 59, 113–118. [Google Scholar]
- Hawrylak, B.; Matraszek, R.; Szymańska, M. Response of Lettuce (Lactuca Sativa L.) to Selenium in Nutrient Solution Contaminated with Nickel. Veg. Crop. Res. Bull. 2007, 67, 63–70. [Google Scholar] [CrossRef]
- Islam, F.; Yasmeen, T.; Arif, M.S.; Riaz, M.; Shahzad, S.M.; Imran, Q.; Ali, I. Combined ability of chromium (Cr) tolerant plant growth promoting bacteria (PGPB) and salicylic acid (SA) in attenuation of chromium stress in maize plants. Plant Physiol. Biochem. 2016, 108, 456–467. [Google Scholar] [CrossRef] [PubMed]
- Zaets, I.; Kramarev, S.; Kozyrovska, N. Inoculation with a bacterial consortium alleviates the effect of cadmium overdose in soybean plants. Open Life Sci. 2010, 5. [Google Scholar] [CrossRef]
- Ponce, S.C.; Prado, C.; Pagano, E.; Prado, F.E.; Rosa, M. Effect of pH on Cr(III) accumulation, biomass production, and phenolic profile in 2 Salvinia species. Environ. Toxicol. Chem. 2018, 38, 167–176. [Google Scholar] [CrossRef]
- Ahmad, P.; Ahanger, M.A.; Alyemeni, M.N.; Wijaya, L.; Alam, P. Exogenous application of nitric oxide modulates osmolyte metabolism, antioxidants, enzymes of ascorbate-glutathione cycle and promotes growth under cadmium stress in tomato. Protoplasma 2018, 255, 79–93. [Google Scholar] [CrossRef]
- Jan, S.; Alyemeni, M.N.; Wijaya, L.; Alam, P.; Siddique, K.H.; Ahmad, P. Interactive effect of 24-epibrassinolide and silicon alleviates cadmium stress via the modulation of antioxidant defense and glyoxalase systems and macronutrient content in Pisum sativum L. seedlings. BMC Plant Biol. 2018, 18. [Google Scholar] [CrossRef]
- Hasanuzzaman, M.; Alam, M.M.; Nahar, K.; Mohsin, S.M.; Bhuyan, M.B.; Parvin, K.; Hawrylak-Nowak, B.; Fujita, M. Silicon-induced antioxidant defense and methylglyoxal detoxification works coordinately in alleviating nickel toxicity in Oryza sativa L. Ecotoxicology 2019, 28, 261–276. [Google Scholar] [CrossRef]
- Nahar, K.; Hasanuzzaman, M.; Alam, M.M.; Rahman, A.; Suzuki, T.; Fujita, M. Polyamine and nitric oxide crosstalk: Antagonistic effects on cadmium toxicity in mung bean plants through upregulating the metal detoxification, antioxidant defense and methylglyoxal detoxification systems. Ecotoxicol. Environ. Saf. 2016, 126, 245–255. [Google Scholar] [CrossRef] [PubMed]
- Shen, X.; Li, R.; Chai, M.; Cheng, S.; Niu, Z.; Qiu, G.Y. Interactive effects of single, binary and trinary trace metals (lead, zinc and copper) on the physiological responses of Kandelia obovata seedlings. Environ. Geochem. Health 2019, 41, 135–148. [Google Scholar] [CrossRef] [PubMed]
- Desoky, E.-S.M.; Elrys, A.S.; Rady, M.M. Integrative moringa and licorice extracts application improves Capsicum annuum fruit yield and declines its contaminant contents on a heavy metals-contaminated saline soil. Ecotoxicol. Environ. Saf. 2019, 169, 50–60. [Google Scholar] [CrossRef] [PubMed]
- Howladar, S.M. A novel Moringa oleifera leaf extract can mitigate the stress effects of salinity and cadmium in bean (Phaseolus vulgaris L.) plants. Ecotoxicol. Environ. Saf. 2014, 100, 69–75. [Google Scholar] [CrossRef]
- Herbers, K.; Sonnewald, U. Altered gene expression brought about by inter- and intracellularly formed hexoses and its possible implications for plant-pathogen interactions. J. Plant Res. 1998, 111, 323–328. [Google Scholar] [CrossRef]
- Babaeian, M.; Tavassoli, A.; Ghanbari, A.; Esmaeilian, Y.; Fahimifard, M. Effects of foliar micronutrient application on osmotic adjustments, grain yield and yield components in sunflower (Alstar cultivar) under water stress at three stages. Afr. J. Agric. Res. 2011, 6, 1204–1208. [Google Scholar]
- Garg, N.; Singh, S. Mycorrhizal inoculations and silicon fortifications improve rhizobial symbiosis, antioxidant defense, trehalose turnover in pigeon pea genotypes under cadmium and zinc stress. Plant Growth Regul. 2018, 86, 105–119. [Google Scholar] [CrossRef]
- Verma, S.; Dubey, R. Effect of cadmium on soluble sugars and enzymes of their metabolism in rice. Biol. Plant. 2001, 44, 117–123. [Google Scholar] [CrossRef]
- Malook, I.; Rehman, S.U.; Khan, M.D.; El-Hendawy, S.E.; Al-Suhaibani, N.A.; Aslam, M.M.; Jamil, M. Heavy metals induced lipid peroxidation in spinach mediated with microbes. Pak. J. Bot. 2017, 49, 2301–2308. [Google Scholar]
| Treatments | Net Photosynthetic Rate (µmol m2 s−1) (Mean ± SD) | Stomatal Conductance (mmol CO2 m2 s−1) (Mean ± SD) | Intracellular CO2 (µmol mol1) (Mean ± SD) | Transpiration Rate (mmol m2 s−1) (Mean ± SD) |
|---|---|---|---|---|
| CN | 25.54 ± 0.952 c | 0.474 ± 0.02 d | 436 ± 8.42 ab | 2.453 ± 0.11 b |
| Cd | 17.36 ± 0.67 e | 0.324 ± 0.007 e | 392.8 ± 4.72 d | 1.563 ± 0.10 e |
| M1 | 28.33 ± 0.83 b | 0.524 ± 0.012 c | 455.6 ± 5.51 a | 2.75 ± 0.12 a |
| M1+Cd | 22.44 ± 0.55 d | 0.573 ± 0.008 b | 422.9 ± 5.74 c | 1.843 ± 0.057 c |
| M2 | 27.73 ± 0.53 b | 0.496 ± 0.009 cd | 442.3 ± 3.14 ab | 2.97 ± 0.09 a |
| M2+Cd | 30.44 ± 0.90 a | 0.604 ± 0.007 a | 462.6 ± 7.10 a | 1.763 ± 0.04 d |
| F-ratio(df 1,12) T | 112.3 ** | 0.238 | 42.6 ** | 523.06 ** |
| F-ratio(df 2,12) D | 152.3 ** | 399.6 ** | 61.94 ** | 25.10 ** |
| F-ratio(df 2,12) T x D | 86.09 * | 242.5 ** | 47.94 ** | 5.525 * |
| HSD | 2.078 | 0.0287 | 16.49 | 0.254 |
| Treatments | Total Phenols (Mean ± SD) | Total Flavonoids (Mean ± SD) | Total Anthocyanins (Mean ± SD) |
|---|---|---|---|
| CN | 15.25 ± 0.78 e | 0.772 ± 0.041 e | 2.453 ± 0.35 d |
| Cd | 19.66 ± 0.70 d | 1.009 ± 0.017 c | 4.372 ± 0.27 b |
| M1 | 25.61 ± 0.65 c | 0.861 ± 0.012 d | 4.050 ± 0.20 bc |
| M1+Cd | 30.64 ± 0.61 b | 1.343 ± 0.019 b | 6.542 ± 0.31 a |
| M2 | 21.69 ± 0.73 d | 0.946 ± 0.044 c | 5.084 ± 0.15 b |
| M2+Cd | 36.02 ± 0.92 a | 1.586 ± 0.025 a | 7.154 ± 0.33 a |
| F-ratio(df 1,12) T | 515.8 ** | 1005.7 ** | 279.6 ** |
| F-ratio(df 2,12) D | 445.9 ** | 230.9 ** | 153.7 ** |
| F-ratio(df 2,12) T x D | 84.66 * | 67.47 ** | 1.762 |
| HSD | 2.029 | 0.0830 | 0.751 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Khanna, K.; Kohli, S.K.; Ohri, P.; Bhardwaj, R.; Al-Huqail, A.A.; Siddiqui, M.H.; Alosaimi, G.S.; Ahmad, P. Microbial Fortification Improved Photosynthetic Efficiency and Secondary Metabolism in Lycopersicon esculentum Plants Under Cd Stress. Biomolecules 2019, 9, 581. https://doi.org/10.3390/biom9100581
Khanna K, Kohli SK, Ohri P, Bhardwaj R, Al-Huqail AA, Siddiqui MH, Alosaimi GS, Ahmad P. Microbial Fortification Improved Photosynthetic Efficiency and Secondary Metabolism in Lycopersicon esculentum Plants Under Cd Stress. Biomolecules. 2019; 9(10):581. https://doi.org/10.3390/biom9100581
Chicago/Turabian StyleKhanna, Kanika, Sukhmeen Kaur Kohli, Puja Ohri, Renu Bhardwaj, Asma A. Al-Huqail, Manzer H. Siddiqui, Ghada Saleh Alosaimi, and Parvaiz Ahmad. 2019. "Microbial Fortification Improved Photosynthetic Efficiency and Secondary Metabolism in Lycopersicon esculentum Plants Under Cd Stress" Biomolecules 9, no. 10: 581. https://doi.org/10.3390/biom9100581
APA StyleKhanna, K., Kohli, S. K., Ohri, P., Bhardwaj, R., Al-Huqail, A. A., Siddiqui, M. H., Alosaimi, G. S., & Ahmad, P. (2019). Microbial Fortification Improved Photosynthetic Efficiency and Secondary Metabolism in Lycopersicon esculentum Plants Under Cd Stress. Biomolecules, 9(10), 581. https://doi.org/10.3390/biom9100581






