Skin Care Formulations and Lipid Carriers as Skin Moisturizing Agents
Abstract
:1. Introduction
2. Skin Care Products—Cosmeceuticals
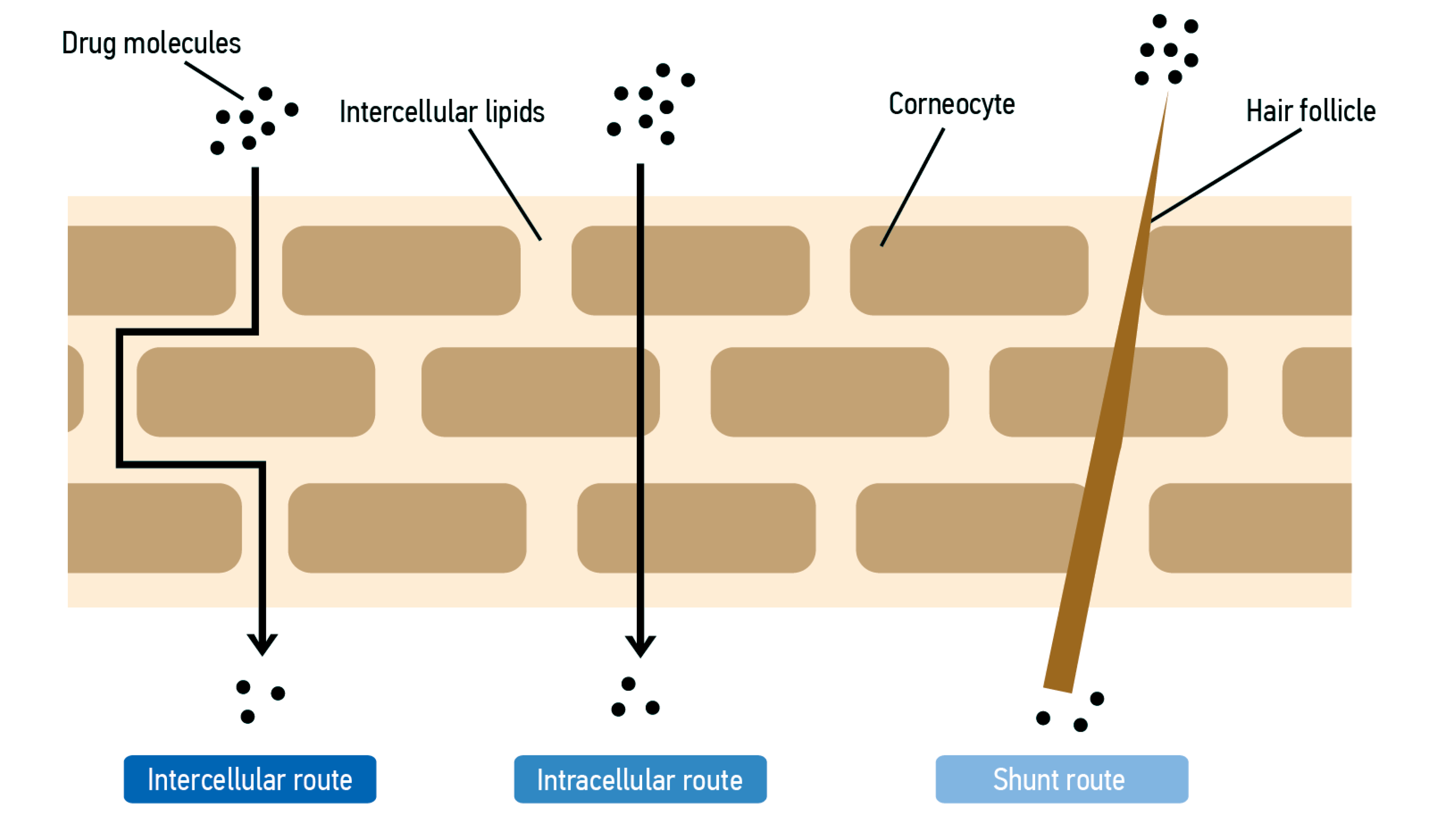
3. Skin Hydration
- by the addition of humectants (glycerol, sorbitol, sodium hyaluronate with low MW, elements of natural hydration factors). These are hygroscopic substances that increase the ability of the SC cells to retain water.
- by adding film-forming/occlusive agents or emollients [hydrocarbons (petrolatum, paraffin oil, perhydrosqualene), silicone oils, animal or vegetable oils, fatty alcohols, especially stearyl and cetyl, waxes (from shea, bees)]. Many of the lipophilic moisturizers act by forming a nearly impermeable layer on the surface of the SC, thereby reducing water evaporation from the SC. Such moisturizers are called occlusive moisturizers and do not normally penetrate the SC. However, some moisturizers appear to increase the water content in the SC without forming an impermeable layer on the surface of the SC. The mode of action of such non-occlusive lipophilic moisturizers is as yet unclear [9].
- by the addition of lipids which mimic the lipids in SC (ceramides).
4. Novel Nanocarriers for Skin Care Products
4.1. Liposomes
4.2. Niosomes
4.3. Nanostructured Lipid Carriers and Solid Lipid Nanoparticles
- mixing SLN/NLC with existing products
- adding viscosity improvers to the aqueous phase
- directly preparing a semi-solid formulation containing only nanoparticles in a one-step process.
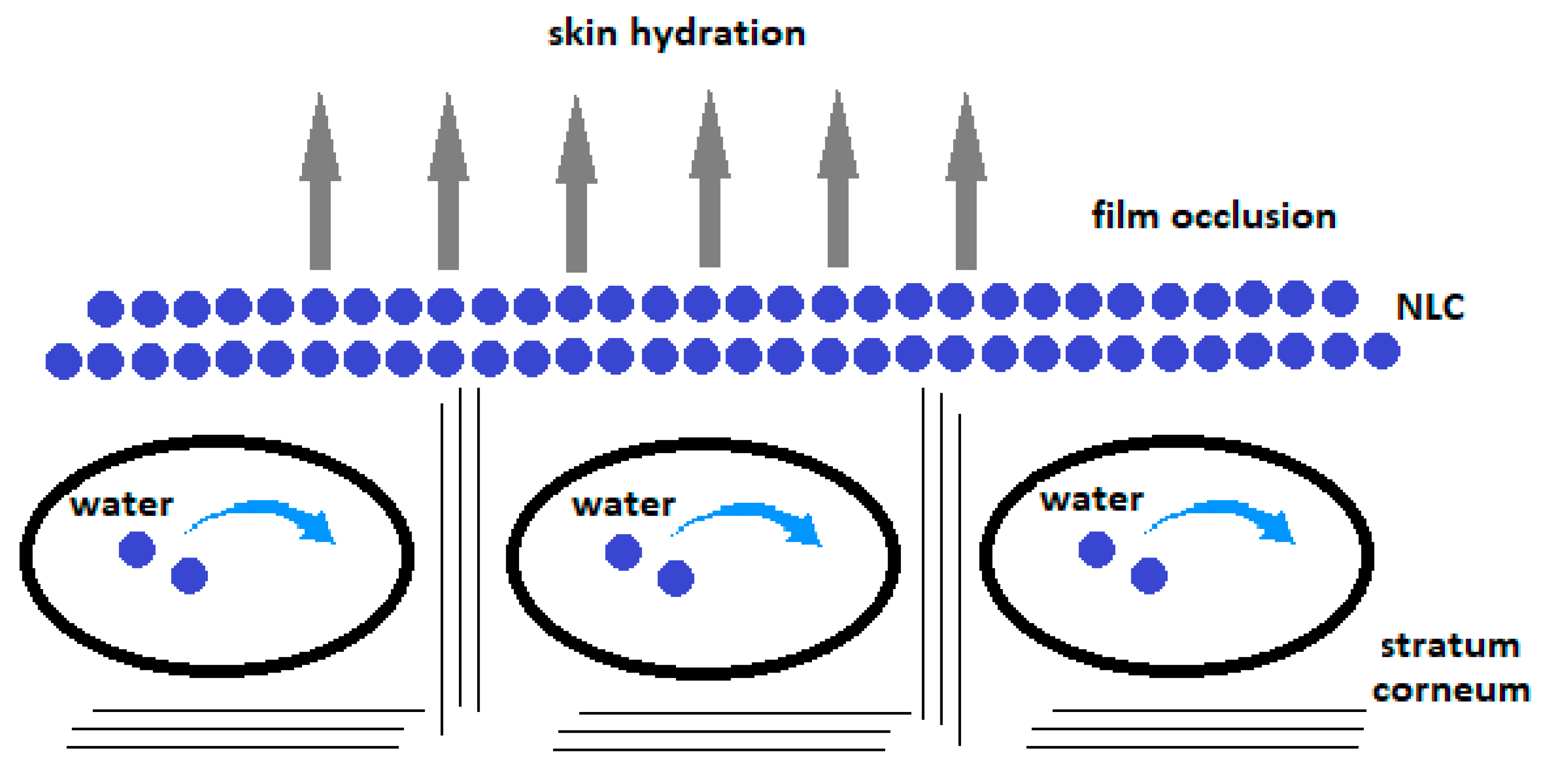
4.3.1. Film Formation and Occlusive Property of NLCs and SLNs
4.3.2. The Effects of Varying Lipid Concentrations, Lipid and Oil Ratio, and the Addition of Propylene Glycol and Lecithin on the Long-Term Physical Stability of NLC, Skin Hydration, and TEWL
4.4. Nanoemulsions
4.5. Gold Nanoparticles
4.6. Nanospheres
5. Recent Studies on Moisturizing Factors Distributed through Lipid Vehicles
5.1. Squalene
5.2. Phyto-Based Nano-Formulations
5.3. Argan/Jojoba Oil Nanostructured Lipid Carriers
5.4. Macro-Sized Particles, Made of a Hydrogel Polymer, Loaded with Olive Oil
5.5. Silver-Nanolipid Complex with Hemp Seed Oil for Application to Atopic Dermatitis Skin
5.6. Exotic Vegetable Oils for Cosmetic o/w Nanoemulsions
5.7. Silicone Vesicles and Matrices
5.8. Hyaluronic Acid
5.9. Ceramides
6. Conclusions/Future Perspectives
Author Contributions
Funding
Conflicts of Interest
References
- Kalia, Y.N.; Guy, R.H. Modeling transdermal drug release. Adv. Drug Deliv. Rev. 2001, 48, 159–172. [Google Scholar] [CrossRef]
- Czajkowska-Kosnik, A.; Szekalska, M.; Winnicka, K. Nanostructured lipid carriers: A potential use for skin drug delivery systems. Pharmacol. Rep. 2019, 71, 156–166. [Google Scholar] [CrossRef]
- Jeon, S.; Yoo, C.Y.; Park, S.N. Improved stability and skin permeability of sodium hyaluronate-chitosan multilayered liposomes by Layer-by-Layer electrostatic deposition for quercetin delivery. Colloids Surf. B Biointerfaces 2015, 129, 7–14. [Google Scholar] [CrossRef] [PubMed]
- Pandey, A.; Jatana, G.K.; Sonthalia, S. Cosmeceuticals. In Stat Pearls; StatPearls Publishing: Treasure Island, FL, USA, 2021. Available online: https://www.ncbi.nlm.nih.gov/books/NBK544223/ (accessed on 1 August 2021).
- Dhapte-Pawar, V.; Kadam, S.; Saptarsi, S.; Kenjale, P.P. Nanocosmeceuticals: Facets and aspects. Future Sci. OA 2020, 6, FSO613. [Google Scholar] [CrossRef] [PubMed]
- Rawlings, A.V.; Harding, C.R. Moisturization and skin barrier function. Dermatol. Ther. 2004, 17, 43–48. [Google Scholar] [CrossRef] [PubMed]
- Loden, M. The clinical benefit of moisturizers. J. Eur. Acad. Dermatol. Venereol. 2005, 19, 672–688. [Google Scholar] [CrossRef]
- Duplan, H.; Nocera, T. Skin hydration and hydrating products. Ann. Dermatol. Venereol. 2018, 145, 376–384. [Google Scholar] [CrossRef] [PubMed]
- Caussin, J.; Gooris, G.S.; Bouwstra, J.A. FTIR studies show lipophilic moisturizers to interact with stratum corneum lipids, rendering the more densely packed. Biochim. Biophys. Acta 2008, 1778, 1517–1524. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Touitou, E.; Dayan, N.; Bergelson, L.; Godin, B.; Eliaz, M. Ethosomes-novel vesicular carriers for enhanced delivery: Characterization and skin penetration properties. J. Control. Release 2000, 65, 403–418. [Google Scholar] [CrossRef]
- Gollavilli, H.; Hegde, A.R.; Managuli, R.S.; Bhaskar, K.V.; Dengale, S.J.; Reddy, M.S.; Kalthur, G.; Mutalik, S. Naringin nano-ethosomal novel sunscreen creams: Development and performance evaluation. Colloids Surf. B Biointerfaces 2020, 193, 111122. [Google Scholar] [CrossRef]
- Kazi, K.M.; Mandal, A.S.; Biswas, N.; Guha, A.; Chatterjee, S.; Behera, M.; Kuotsu, K. Niosome: A future of targeted drug delivery systems. J. Adv. Pharm. Technol. Res. 2010, 1, 374–380. [Google Scholar]
- Chena, S.; Hanninga, S.; Falconerb, J.; Lockea, M.; Wena, J. Recent advances in non-ionic surfactant vesicles (niosomes): Fabrication, characterization, pharmaceutical and cosmetic applications. Eur. J. Pharm. Biopharm. 2019, 144, 18–39. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Aziz, Z.A.A.; Mohd-Nasir, H.; Ahmad, A.; Setapar, S.H.M.; Peng, W.L.; Chuo, S.C.; Khatoon, A.; Umar, K.; Yaqoob, A.A.; Nasir, M.; et al. Role of Nanotechnology for Design and Development of Cosmeceutical: Application in Makeup and Skin Care. Front. Chem. 2019, 7, 739. [Google Scholar] [CrossRef] [PubMed]
- Wavikar, P.; Vavia, P. Nanolipidgel for Enhanced Skin Deposition and Improved Antifungal Activity. AAPS PharmSciTech 2013, 14, 222–233. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Müller, R.H.; Petersen, R.D.; Hommoss, A.; Pardeike, J. Nanostructured lipid carriers (NLC) in cosmetic dermal products. Adv. Drug Deliv. Rev. 2007, 59, 522–530. [Google Scholar] [CrossRef]
- Patel, D.; Kumar, V.; Kesharwani, R.; Mazumdar, B. Lipid Nanoparticle a Novel Carrier for Cosmetics and Topical Preparation: A Review. Inventi Rapid Cosmeceuticals 2015, 3, 1–6. [Google Scholar]
- Souto, E.B.; Wissing, S.A.; Barbosa, C.M.; Muller, R.H. Development of a controlled release formulation based on SLN and NLC for topical clotrimazole delivery. Int. J. Pharm. 2004, 278, 71–77. [Google Scholar] [CrossRef]
- Chauhan, I.; Yasir, M.; Verma, M.; Singh, A.P. Nanostructured Lipid Carriers: A Groundbreaking Approach for Transdermal Drug Delivery. Adv. Pharm. Bull. 2020, 10, 150–165. [Google Scholar] [CrossRef]
- Pardeike, J.; Schwabe, K.; Müller, R.H. Influence of nanostructured lipid carriers (NLC) on the physical properties of the Cutanova Nanorepair Q10 cream and the in vivo skin hydration effect. Adv. Drug Deliv. Rev. 2010, 396, 166–173. [Google Scholar] [CrossRef]
- Khater, D.; Nsairat, H.; Odeh, F.; Saleh, M.; Jaber, A.; Alshaer, W.; Al Bawab, A.; Mubarak, M.S. Design, Preparation, and Characterization of Effective Dermal and Transdermal Lipid Nanoparticles: A Review. Cosmetics 2021, 8, 39. [Google Scholar] [CrossRef]
- Loo, C.H.; Basri, M.; Ismail, R.; Lau, H.L.N.; Tejo, B.A.; Kanthimathi, M.S.; Hassan, H.A.; Choo, Y.M. Effect of compositions in nanostructured lipid carriers (NLC) on skin hydration and occlusion. Int. J. Nanomed. 2013, 8, 13–22. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kaul, S.; Gulati, N.; Verma, D.; Mukherjee, S.; Nagaich, U. Role of Nanotechnology in Cosmeceuticals: A Review of Recent Advances. Hindawi J. Pharm. 2018, 2018, 3420204. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ashaolu, T.J. Environ Nanoemulsions for health, food, and cosmetics: A review. Chem. Lett. 2021, 15, 1–15. [Google Scholar]
- Zhou, H.; Luo, D.; Chen, D.; Tan, X.; Bai, X.; Liu, Z.; Yang, X.; Liu, W. Current Advances of Nanocarrier Technology-Based Active Cosmetic Ingredients for Beauty Applications. Clin. Cosmet. Investig. Dermatol. 2021, 14, 867–887. [Google Scholar] [CrossRef]
- Chung, H.; Kim, T.W.; Kwon, M.; Kwon, I.C.; Jeong, S.Y. Oil components modulate physical characteristics and function of the natural oil emulsions as drug or gene delivery system. J. Control. Release 2001, 71, 339–350. [Google Scholar] [CrossRef]
- Fang, J.Y.; Fang, C.L.; Liu, C.H.; Su, Y.H. Lipid nanoparticles as vehicles for topical psoralen delivery: Solid lipid nanoparticles (SLN) versus nanostructured lipid carriers (NLC). Eur. J. Pharm. Biopharm. 2008, 70, 633–640. [Google Scholar] [CrossRef] [PubMed]
- Huang, Z.R.; Lin, Y.K.; Fang, J.Y. Biological and pharmacological activities of squalene and related compounds: Potential uses in cosmetic dermatology. Molecules 2009, 23, 540–554. [Google Scholar] [CrossRef]
- Ganesan, P.; Choi, D.K. Current application of phytocompound-based nanocosmeceuticals for beauty and skin therapy. Int. J. Nanomed. 2016, 11, 1987–2007. [Google Scholar] [CrossRef] [Green Version]
- Khameneh, B.; Halimi, V.; Jaafari, M.R.; Golmohammadzadeh, S. Safranal-loaded solid lipid nanoparticles: Evaluation of sunscreen and moisturizing potential for topical applications. Iran. J. Basic Med. Sci. 2015, 18, 58–63. [Google Scholar]
- Bernardi, D.S.; Pereira, T.A.; Maciel, N.R.; Bortoloto, J.; Viera, G.S.; Oliveira, G.C.; Rocha-Filho, P.A. Formation and stability of oil-in-water nanoemulsions containing rice bran oil: In vitro and in vivo assessments. J. Nanobiotechnol. 2011, 9, 44. [Google Scholar] [CrossRef]
- Ribeiro, R.C.; Barreto, S.M.; Ostrosky, E.A.; da Rocha-Filho, P.A.; Veríssimo, L.M.; Ferrari, M. Production and characterization of cosmetic nanoemulsions containing Opuntia ficus-indica (L.) mill extract as moisturizing agent. Molecules 2015, 20, 2492–2509. [Google Scholar] [CrossRef] [Green Version]
- Tichota, D.M.; Silva, A.C.; Lobo, J.M.S.; Amara, M.H. Design, characterization, and clinical evaluation of argan oil nanostructured lipid carriers to improve skin hydration. Int. J. Nanomed. 2014, 9, 3855–3864. [Google Scholar]
- Estanqueiro, M.; Conceição, J.; Amaral, M.H.; Sousa Lobo, J.M. Characterization, sensorial evaluation and moisturizing efficacy of nanolipidgel formulations. Int. J. Cosmet Sci. 2014, 36, 159–166. [Google Scholar] [CrossRef]
- Korać, R.R.; Khambholja, K.M. Potential of herbs in skin protection from ultraviolet radiation. Pharmacogn. Rev. 2011, 5, 164–173. [Google Scholar] [CrossRef] [Green Version]
- Lee, K.Y.; Mooney, D.J. Alginate: Properties and biomedical applications. Prog. Polym. Sci. 2012, 37, 106–126. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schmitt, A.; Rödel, P.; Anamur, C.; Seeliger, C.; Imhoff, A.B.; Herbst, E.; Vogt, S.; van Griensven, M.; Winter, G.; Engert, J. Calcium Alginate Gels as Stem Cell Matrix—Making Paracrine Stem Cell Activity Available for Enhanced Healing after Surgery. PLoS ONE 2015, 10, e0118937. [Google Scholar] [CrossRef]
- Mota, A.H.; Silva, C.O.; Nicolai, M.; Baby, A.; Palma, L.; Rijo, P.; Ascensão, L.; Reis, C.P. Design and evaluation of novel topical formulation with olive oil as natural functional active. Pharm. Dev. Technol. 2017, 23, 794–805. [Google Scholar] [CrossRef] [PubMed]
- Keck, M.; Schwabe, K. Silver-Nanolipid Complex for Application to Atopic Dermatitis Skin: Rheological Characterization, In Vivo Efficiency and Theory of Action Cornelia. J. Biomed. Nanotechnol. 2009, 5, 428–436. [Google Scholar] [CrossRef]
- Schlossman, M.L.; McCarthy, J.P. Lanolin and its derivatives. J. Amer. Oil Chem. Soc. 1978, 55, 447–450. [Google Scholar] [CrossRef]
- Pereira, T.A.; Guerreiro, C.M.; Maruno, M.; Ferrari, M.; Rocha-Filho, P.A. Exotic Vegetable Oils for Cosmetic O/W Nanoemulsions: In Vivo Evaluation. Molecules 2016, 21, 248. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dini, I.; Laneri, S. The New Challenge of Green Cosmetics: Natural Food Ingredients for Cosmetic Formulations. Molecules 2021, 26, 3921. [Google Scholar] [CrossRef] [PubMed]
- Patravale, V.B.; Mandawgade, S.D. Novel cosmetic delivery systems: An application update. Int. J. Cosmet Sci. 2008, 30, 19–33. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.; Gupta, V.; Anselmo, A.C.; Muraski, J.A.; Mitragotri, S. Topical delivery of hyaluronic acid into skin using SPACE-peptide carriers. J. Control. Release 2014, 173, 67–74. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ming, K.; Chen, X.G.; Kweon, D.K.; Park, H.J. Investigations on skin permeation of hyaluronic acid based nanoemulsion as transdermal carrier. Carbohydr. Polym. 2011, 86, 837–843. [Google Scholar]
- Uche, L.E.; Gooris, G.S.; Beddoes, C.M.; Bouwstra, J.A. New insight into phase behavior and permeability of skin lipid models based on sphingosine and phytosphingosine ceramides. Biochim. Biophys. Acta Biomembr. 2019, 1861, 1317–1328. [Google Scholar] [CrossRef]
- Yilmaz, E.; Borchert, H.H. Design of a phytosphingosine-containing, positively-charged nanoemulsion as a colloidal carrier system for dermal application of ceramides. Eur. J. Pharm. Biopharm. 2005, 60, 91–98. [Google Scholar] [CrossRef]
| Product | Company | Use |
|---|---|---|
| Liposomes | ||
| Rehydrating Liposome day Creme | Kerstin Florian | Rehydrate the skin and acts as moisturizer |
| Dermosome | Microfluidics | Retaining and prevent loss of moisture from the skin |
| Moisture Liposome Eye Cream | Decorte | Whitening the black skin around the eyes and acts as moisturizer |
| Niosomes | ||
| MayuNiosome Base Cream | Laon Cosmetics | Moisturizing and whitening actions |
| Solid lipid nanoparticles | ||
| Allure Body Cream | Chanel | Moisturizing the skin and body |
| Nanoemulsions | ||
| Precision-Solution Destressante Solution Nano Emulsion Peaux Sensitivity | Chanel | Moisturizing the skin |
| Hyaluronic Acid and Nanoemulsion Intensive Hydration Toner | Coni Beauty | Hydrating skin and moisture retention |
| Phyto-Endorphin Hand Cream | Rhonda Allison | Sooth and moisturize the skin |
| Vita-Herb Nona-Vital Skin Toner | Vitacos Cosmetics | Moisturizing the skin |
| Gold nanoparticles | ||
| Nano Gold Foil Liquid | Ameizii | Repairing damaged skin and acts as moisturizer |
| 24K Nano Ultra Silk Serum | Orogold | Prevent moisture loss and keep a healthy skin |
| Nanospheres | ||
| Fresh As A Daisy Body Lotion | Kara Vita | Moisturizing the skin and also prevent water loss |
| Ultra Moisturizing Day Cream | Hydralane Paris | Moisturizing agent and in retaining moisture inside skin |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pavlou, P.; Siamidi, A.; Varvaresou, A.; Vlachou, M. Skin Care Formulations and Lipid Carriers as Skin Moisturizing Agents. Cosmetics 2021, 8, 89. https://doi.org/10.3390/cosmetics8030089
Pavlou P, Siamidi A, Varvaresou A, Vlachou M. Skin Care Formulations and Lipid Carriers as Skin Moisturizing Agents. Cosmetics. 2021; 8(3):89. https://doi.org/10.3390/cosmetics8030089
Chicago/Turabian StylePavlou, Panagoula, Angeliki Siamidi, Athanasia Varvaresou, and Marilena Vlachou. 2021. "Skin Care Formulations and Lipid Carriers as Skin Moisturizing Agents" Cosmetics 8, no. 3: 89. https://doi.org/10.3390/cosmetics8030089
APA StylePavlou, P., Siamidi, A., Varvaresou, A., & Vlachou, M. (2021). Skin Care Formulations and Lipid Carriers as Skin Moisturizing Agents. Cosmetics, 8(3), 89. https://doi.org/10.3390/cosmetics8030089









