Characterization, Stability Assessment, Antioxidant Evaluation and Cell Proliferation Activity of Virgin Coconut Oil-based Nanostructured Lipid Carrier Loaded with Ficus deltoidea Extract
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Methods
2.2.1. Preparation of FD Loaded Nanostructured Lipid Carrier
2.2.2. Characterization FD Loaded Nanostructured Lipid Carrier
Measurement of Particle Size and Polydispersity Index
Encapsulation Efficiency and Drug Loading
Determination of Zeta Potential
Investigation of the Morphological Structure
2.2.3. Stability Study
2.2.4. Antioxidant Activity
2.2.5. Skin Permeation Study
2.2.6. Cell Proliferative Study
3. Results and Discussions
3.1. Physicochemical Properties
3.2. Characterization of Ficus deltoidea Loaded Nanostructured Lipid Carrier
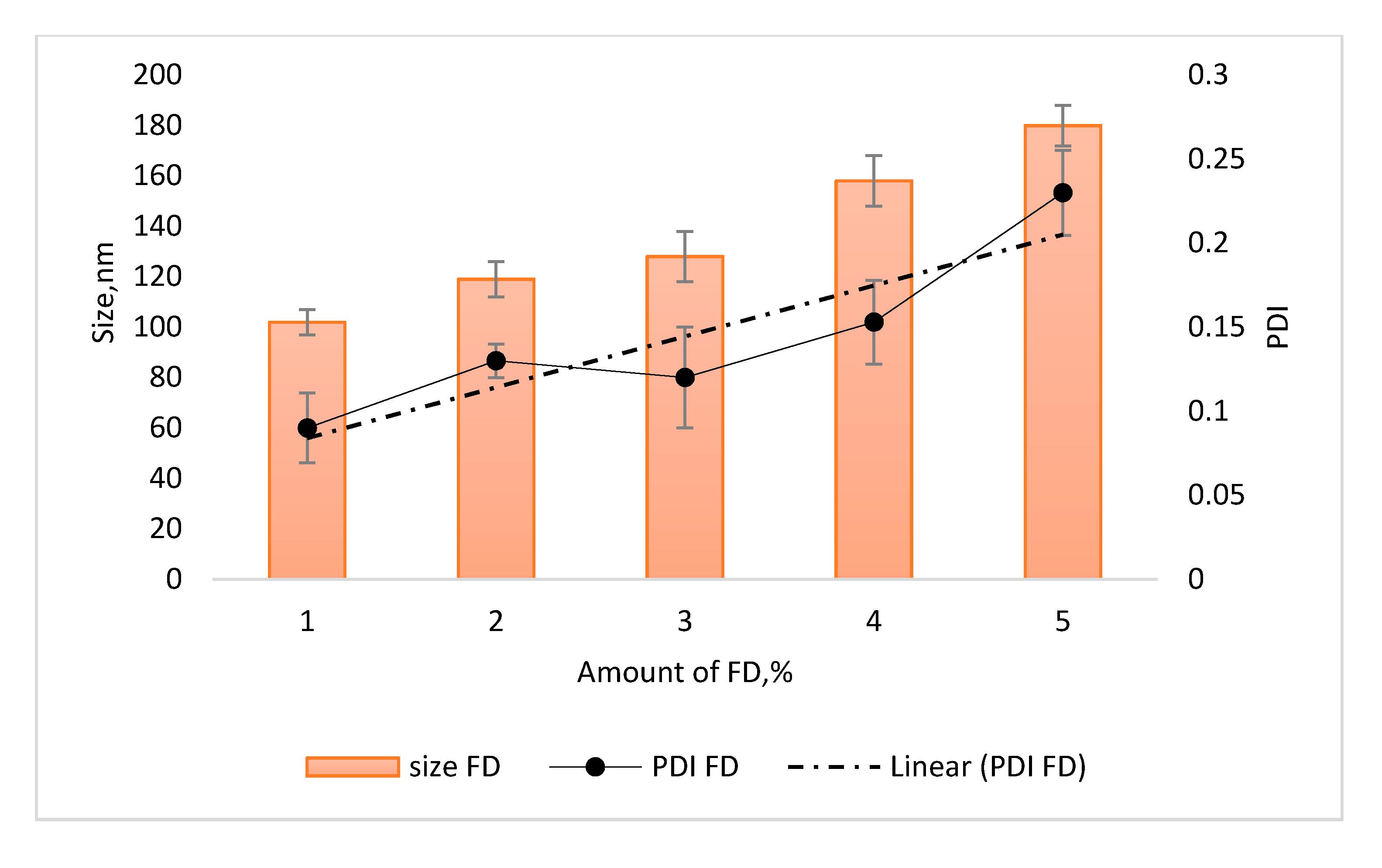
3.2.1. Particle Size and PDI of Ficus deltoidea Loaded Nanostructured Lipid Carrier
3.2.2. Zeta Potential of FD Loaded Nanostructured Lipid Carrier
3.2.3. Active Ingredient Loading and Encapsulation Efficiency of FD Loaded Nanostructured Lipid Carrier
3.2.4. Investigation of Morphological Structure
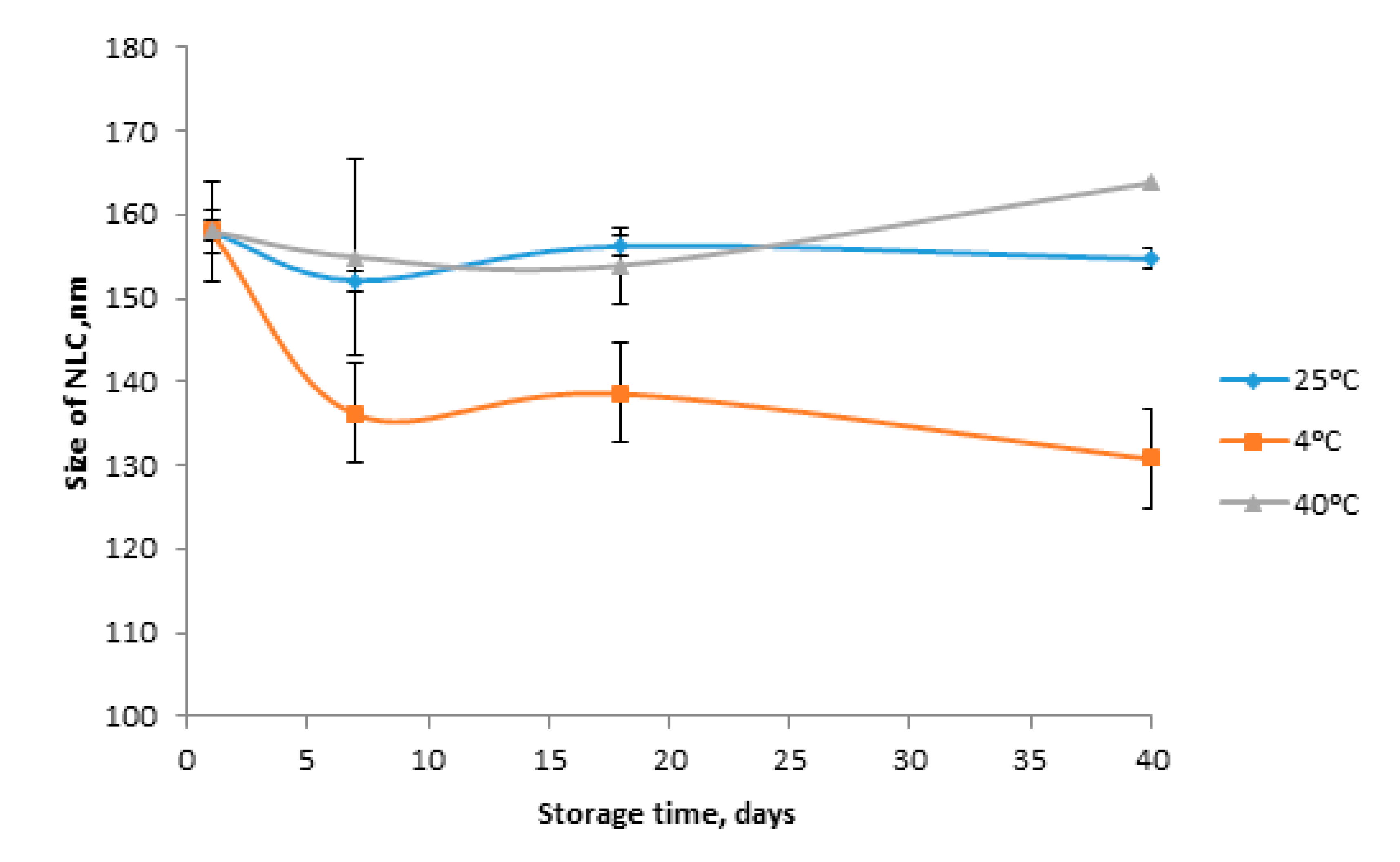
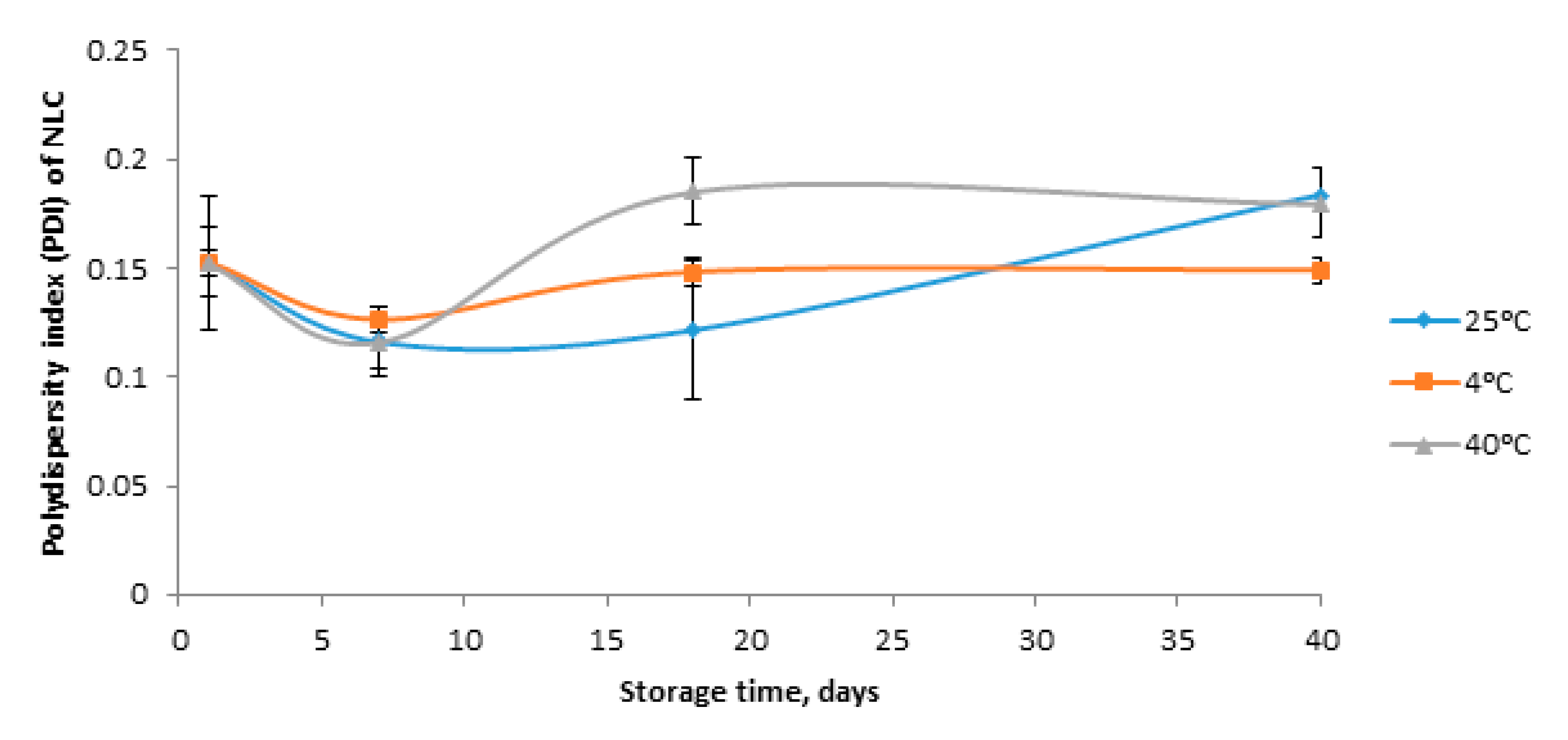
3.3. Stability of FD Loaded Nanostructured Lipid Carrier
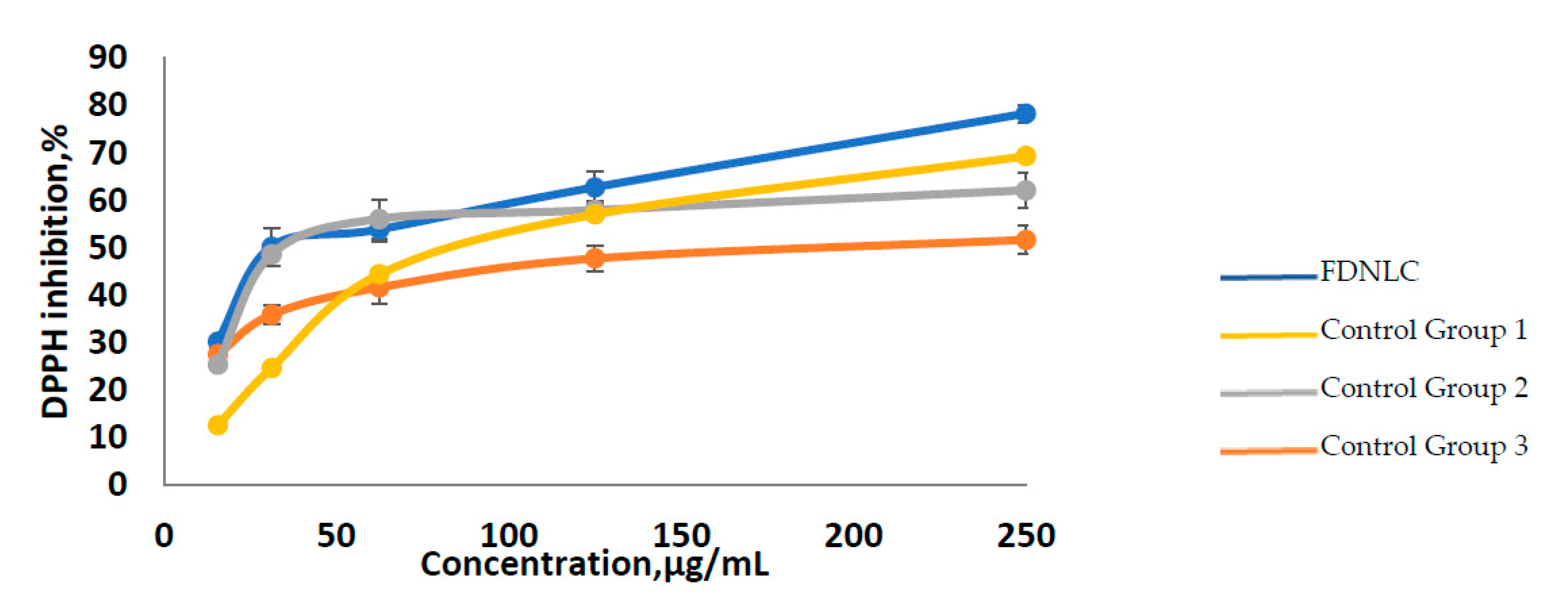
3.4. Antioxidant Activity of FD Loaded Nanostructured Lipid Carrier
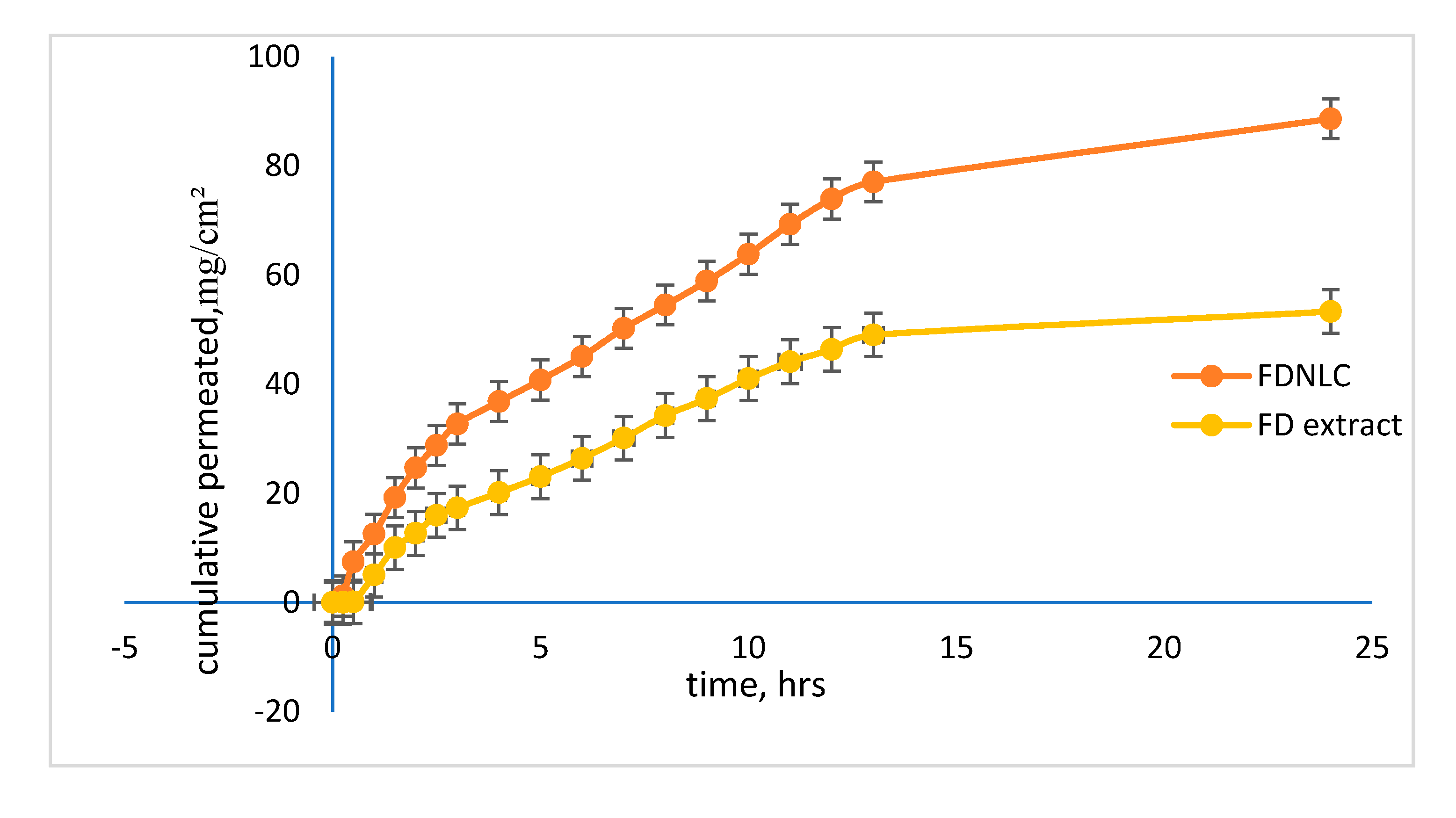
3.5. Skin Permeation
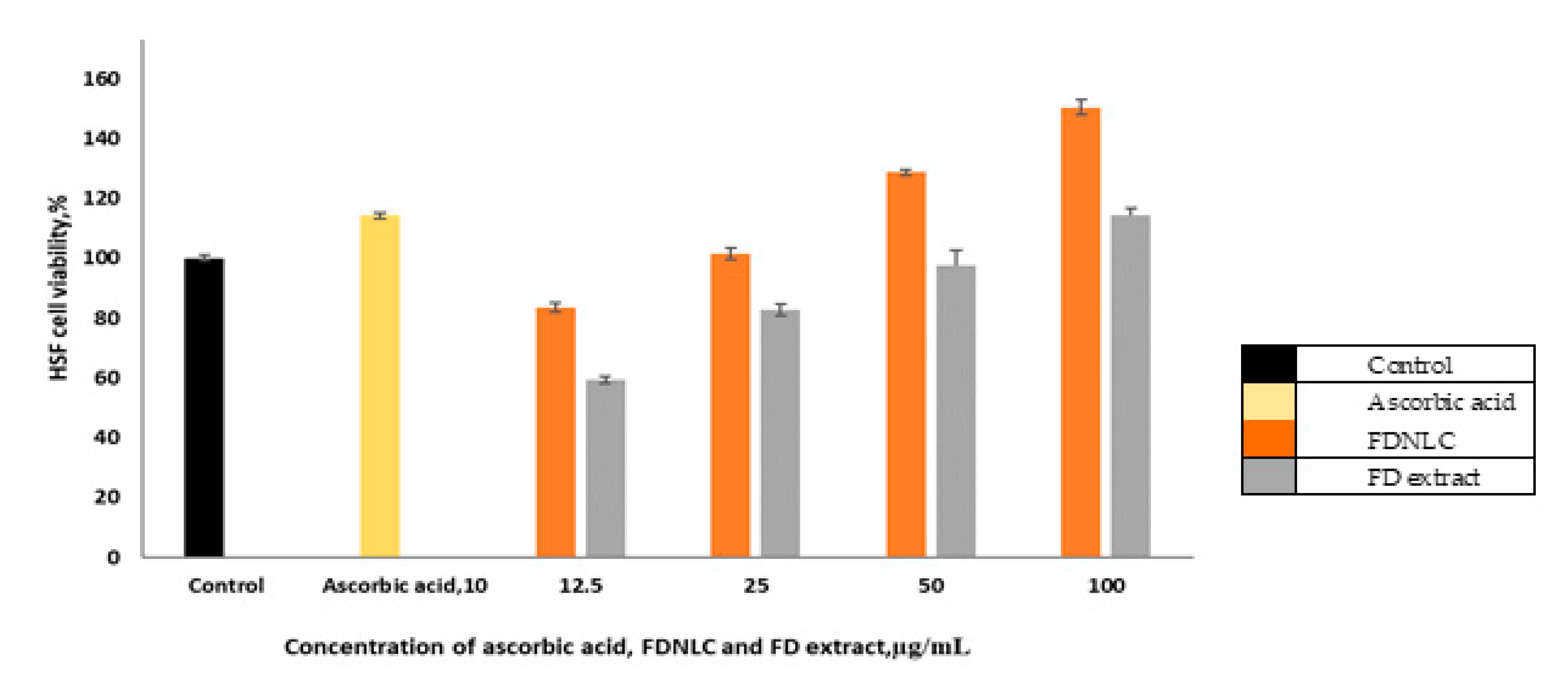
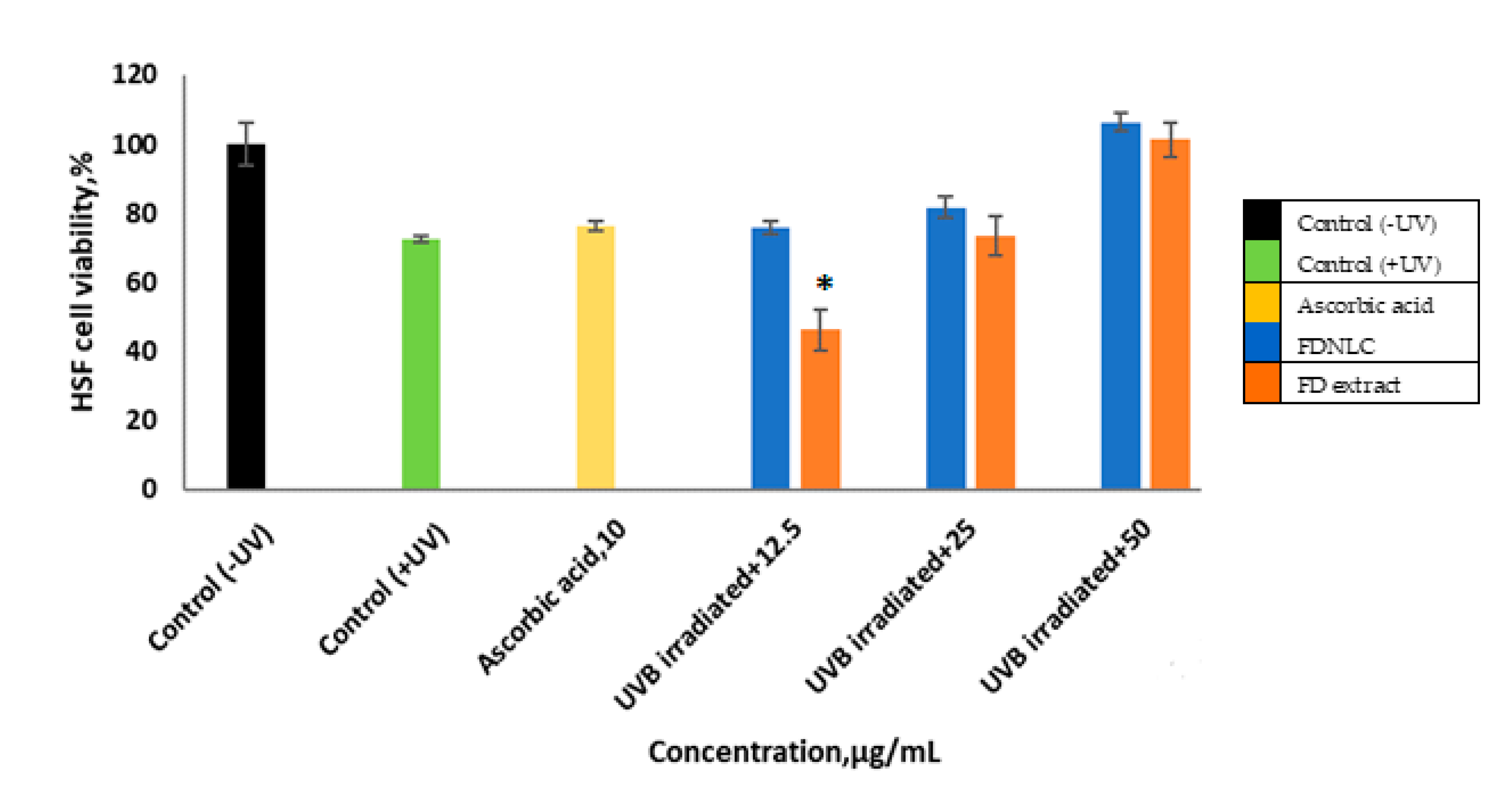
3.6. Cell Proliferative Assay
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Bunawan, H.; Amin, N.M.; Bunawan, S.N.; Baharum, S.N.; Mohd Noor, N. Ficus deltoidea jack: A review on its phytochemical and pharmacological importance. Evid. Based Complement. Altern. Med. 2014, 2014, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Haron, S.H. Enzymatic Extraction of Ficus deltoidea for Better Enhancement of Extracted Yield. Bachelor’s Thesis, University Putra Malaysia, Seri Kembangan, Malaysia, 2014. [Google Scholar]
- Hasham, R.; Choi, H.K.; Sarmidi, M.R.; Park, C.S. Protective effects of a Ficus deltoidea (Mas cotek) extract against UVB-induced photoageing in skin cells. Biotechnol. Bioprocess Eng. 2013, 18, 185–193. [Google Scholar] [CrossRef]
- Abdulla, M.A.; Ahmed, K.A.-A.; Abu-Luhoom, F.M.; Muhanid, M. Role of Ficus deltoidea extract in the enhancement of wound healing in experimental rats. Biomed. Res. 2010, 21, 241–245. [Google Scholar]
- Vinardell, M.P.; Mitjans, M. Nanocarriers for Delivery of Antioxidants on the Skin. Cosmetics 2015, 2, 342–354. [Google Scholar] [CrossRef]
- Desai, P.; Patlolla, R.R.; Singh, M. Interaction of nanoparticles and cell-penetrating peptides with skin for transdermal drug delivery. Mol. Membr. Biol. 2010, 27, 247–259. [Google Scholar] [CrossRef] [PubMed]
- Müller, R.H.; Radtke, M.; Wissing, S.A. Solid lipid nanoparticles (SLN) and nanostructured lipid carriers (NLC) in cosmetic and dermatological preparations. Adv. Drug Deliv. Rev. 2002, 54 (Suppl. S1), S131–S155. [Google Scholar]
- Pardeshi, C.; Rajput, P.; Belgamwar, V.; Tekade, A.; Patil, G.; Chaudhary, K.; Sonje, A. Solid lipid based nanocarriers: An overview. Acta Pharm. 2012, 62, 433–472. [Google Scholar] [CrossRef]
- Lacatusu, I.; Niculae, G.; Badea, N.; Stan, R.; Popa, O.; Oprea, O.; Meghea, A. Design of soft lipid nanocarriers based on bioactive vegetable oils with multiple health benefits. Chem. Eng. J. 2014, 246, 311–321. [Google Scholar] [CrossRef]
- Tichota, D.M.; Silva, A.C.; Sousa Lobo, J.M.; Amaral, M.H. Design, characterization, and clinical evaluation of argan oil nanostructured lipid carriers to improve skin hydration. Int. J. Nanomed. 2014, 9, 3855–3864. [Google Scholar]
- Li, B.; Ge, Z.Q. Nanostructured lipid carriers improve skin permeation and chemical stability of idebenone. AAPS PharmSciTech 2012, 13, 276–283. [Google Scholar] [CrossRef]
- Müller, R.H.; Petersen, R.D.; Hommoss, A.; Pardeike, J. Nanostructured lipid carriers (NLC) in cosmetic dermal products. Adv. Drug Deliv. Rev. 2007, 59, 522–530. [Google Scholar] [CrossRef] [PubMed]
- Mansor, T.S.T.; Che Man, Y.B.; Shuhaimi, M.; Abdul Afiq, M.J.; Ku Nurul, F.K.M. Physicochemical properties of virgin coconut oil extracted from different processing methods. Int. Food Res. J. 2012, 19, 837–845. [Google Scholar]
- Marina, A.M.; Che Man, Y.B.; Nazimah, S.A.H.; Amin, I. Chemical Properties of Virgin Coconut Oil. J. Am. Oil Chem. Soc. 2009, 86, 301–307. [Google Scholar] [CrossRef]
- Rohman, A.; Man, Y.C. Sismindari Quantitative analysis of virgin coconut oil in cream cosmetics preparations using fourier transform infrared (FTIR) spectroscopy. Pak. J. Pharm. Sci. 2009, 22, 415–420. [Google Scholar] [PubMed]
- Kim, S.; Jang, J.E.; Kim, J.; Lee, Y.I.; Lee, D.W.; Song, S.Y.; Lee, J.H. Enhanced barrier functions and anti-inflammatory effect of cultured coconut extract on human skin. Food Chem. Toxicol. 2017, 106, 367–375. [Google Scholar] [CrossRef]
- Rosli, N.A.; Hasham, R.; Aziz, A. Design and physicochemical evaluation of lipid encapsulated Zingiber Zerumbut Oil by D-optimal mixture design. J. Teknol. 2018, 3, 105–113. [Google Scholar]
- Uprit, S.; Kumar Sahu, R.; Roy, A.; Pare, A. Preparation and characterization of minoxidil loaded nanostructured lipid carrier gel for effective treatment of alopecia. Saudi Pharm. J. 2013, 21, 379–385. [Google Scholar] [CrossRef]
- Dangi, R.S.; Shakya, S. Preparation, optimization and characterization of PLGA nanoparticle. Int. J. Pharm. Life Sci. 2013, 4, 2810–2818. [Google Scholar]
- Noor, N.M.; Khan, A.A.; Hasham, R.; Talib, A.; Sarmidi, M.R.; Aziz, R.; Abd, A. Empty nano and micro-structured lipid carriers of virgin coconut oil for skin moisturisation. IET Nanobiotechnol. 2016, 10, 195–199. [Google Scholar] [CrossRef]
- Sharma, O.P.; Bhat, T.K. DPPH antioxidant assay revisited. Food Chem. 2009, 113, 1202–1205. [Google Scholar] [CrossRef]
- Azlim Almey, A.J.K.S.Z.S. Total phenolic content and primary antioxidant activity of methanolic and ethanolic extracts of aromatic plants’ leafs. Int. Food Res. J. 2010, 17, 1077–1084. [Google Scholar]
- Da Costa, S.; Basri, M.; Shamsudin, N.; Basri, H. Stability of Positively Charged Nanoemulsion Formulation Containing Steroidal Drug for Effective Transdermal Application. J. Chem. 2014, 2014, 1–8. [Google Scholar] [CrossRef]
- Zirak, M.B.; Pezeshki, A. Effect of Surfactant Concentration on the Particle Size, Stability and Potential Zeta of Beta carotene Nano Lipid Carrier. Int. J. Curr. Microbiol. Appl. Sci. 2015, 4, 924–932. [Google Scholar]
- Das, S.; Ng, W.K.; Kanaujia, P.; Kim, S.; Tan, R.B.H. Formulation design, preparation and physicochemical characterizations of solid lipid nanoparticles containing a hydrophobic drug: Effects of process variables. Colloids Surfaces B Biointerfaces 2011, 88, 483–489. [Google Scholar] [CrossRef]
- Mitri, K.; Shegokar, R.; Gohla, S.; Anselmi, C.; Müller, R.H. Lipid nanocarriers for dermal delivery of lutein: Preparation, characterization, stability and performance. Int. J. Pharm. 2011, 414, 267–275. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Mira, E.; Egea, M.A.; Garcia, M.L.; Souto, E.B. Design and ocular tolerance of flurbiprofen loaded ultrasound-engineered NLC. Colloids Surfaces B Biointerfaces 2010, 81, 412–421. [Google Scholar] [CrossRef]
- Jia, L.J.; Zhang, D.R.; Li, Z.Y.; Feng, F.F.; Wang, Y.C.; Dai, W.T.; Duan, C.X.; Zhang, Q. Preparation and characterization of silybin-loaded nanostructured lipid carriers. Drug Deliv. 2010, 17, 11–18. [Google Scholar] [CrossRef] [PubMed]
- Chu, B.; Zhang, L.; Qu, Y.; Chen, X.; Peng, J.; Huang, Y.; Qian, Z. Synthesis, characterization and drug loading property of monomethoxy-poly(ethylene glycol)-poly(ϵ-caprolactone)-poly(D,L-lactide) (MPEG-PCLA) copolymers. Sci. Rep. 2016, 6, 1–15. [Google Scholar] [CrossRef]
- Jenning, V.; Thünemann, A.F.; Gohla, S.H. Characterisation of a novel solid lipid nanoparticle carrier system based on binary mixtures of liquid and solid lipids. Int. J. Pharm. 2000, 199, 167–177. [Google Scholar] [CrossRef]
- Venkateswarlu, V.; Manjunath, K. Preparation, characterization and in vitro release kinetics of clozapine solid lipid nanoparticles. J. Control. Release 2004, 95, 627–638. [Google Scholar] [CrossRef]
- Huang, J.; Wang, Q.; Li, T.; Xia, N.; Xia, Q. Nanostructured lipid carrier (NLC) as a strategy for encapsulation of quercetin and linseed oil: Preparation and in vitro characterization studies. J. Food Eng. 2017, 215, 1–12. [Google Scholar] [CrossRef]
- Antolovich, M.; Prenzler, P.D.; Patsalides, E.; McDonald, S.; Robards, K. Methods for testing antioxidant activity. Analyst 2002, 127, 183–198. [Google Scholar] [CrossRef] [PubMed]
- Okonogi, S.; Duangrat, C.; Anuchpreeda, S.; Tachakittirungrod, S.; Chowwanapoonpohn, S. Comparison of antioxidant capacities and cytotoxicities of certain fruit peels. Food Chem. 2007, 103, 839–846. [Google Scholar] [CrossRef]
- Mishra, B.; Patel, B.B.; Tiwari, S. Colloidal nanocarriers: A review on formulation technology, types and applications toward targeted drug delivery. Nanomed. Nanotechnol. Biol. Med. 2010, 6, 9–24. [Google Scholar] [CrossRef] [PubMed]
- Dzolin, S.; Sharipah Ruzaina Syed Aris; Ahmad, R.; Zain, M.M. Radical scavenging and neurotoxicity of four varieties of Ficus deltoidea. In Proceedings of the CSSR 2010—2010 International Conference on Science and Social Research, Kuala Lumpur, Malaysia, 5–7 December 2010; pp. 11–15. [Google Scholar]


| Blank | FDNLC | |
|---|---|---|
| Size (nm) | 153.1 ± 2.5 | 158.0 ± 1.3 |
| PDI | 0.19 ± 0.01 * | 0.15 ± 0.02 |
| Sample/Standard | IC50 Value (µg/mL) | Equation | R2 Value |
|---|---|---|---|
| FDNLC | 45.41 | y = 15.694ln(x) − 9.8861 | R2 = 0.9516 |
| FD extract | 92.20 | y = 21.075ln(x) − 45.628 | R2 = 0.9924 |
| Trolox | 62.90 | y = 11.967ln(x) + 0.4327 | R2 = 0.8024 |
| Ascorbic acid | 180.00 | y = 8.6905ln(x) + 4.8673 | R2 = 0.9845 |
| Samples | J (mg/cm/h) | Kp (cm/h) | Tl (hrs) |
|---|---|---|---|
| FDNLC | 12.727 | 9.8 × 10−2 | 0.023 |
| Unencapsulated FD extract | 7.1004 | 4 × 10−2 | 0.223 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Azmi, N.A.N.; Hasham, R.; Ariffin, F.D.; Elgharbawy, A.A.M.; Salleh, H.M. Characterization, Stability Assessment, Antioxidant Evaluation and Cell Proliferation Activity of Virgin Coconut Oil-based Nanostructured Lipid Carrier Loaded with Ficus deltoidea Extract. Cosmetics 2020, 7, 83. https://doi.org/10.3390/cosmetics7040083
Azmi NAN, Hasham R, Ariffin FD, Elgharbawy AAM, Salleh HM. Characterization, Stability Assessment, Antioxidant Evaluation and Cell Proliferation Activity of Virgin Coconut Oil-based Nanostructured Lipid Carrier Loaded with Ficus deltoidea Extract. Cosmetics. 2020; 7(4):83. https://doi.org/10.3390/cosmetics7040083
Chicago/Turabian StyleAzmi, Nor Azrini Nadiha, Rosnani Hasham, Farah Diana Ariffin, Amal A. M. Elgharbawy, and Hamzah Mohd Salleh. 2020. "Characterization, Stability Assessment, Antioxidant Evaluation and Cell Proliferation Activity of Virgin Coconut Oil-based Nanostructured Lipid Carrier Loaded with Ficus deltoidea Extract" Cosmetics 7, no. 4: 83. https://doi.org/10.3390/cosmetics7040083
APA StyleAzmi, N. A. N., Hasham, R., Ariffin, F. D., Elgharbawy, A. A. M., & Salleh, H. M. (2020). Characterization, Stability Assessment, Antioxidant Evaluation and Cell Proliferation Activity of Virgin Coconut Oil-based Nanostructured Lipid Carrier Loaded with Ficus deltoidea Extract. Cosmetics, 7(4), 83. https://doi.org/10.3390/cosmetics7040083






