Cosmetic Packaging to Save the Environment: Future Perspectives
Abstract
1. Introduction
The Bio and Circular Economy Approach in the Current Waste Management
2. Sustainable Cosmetic Market
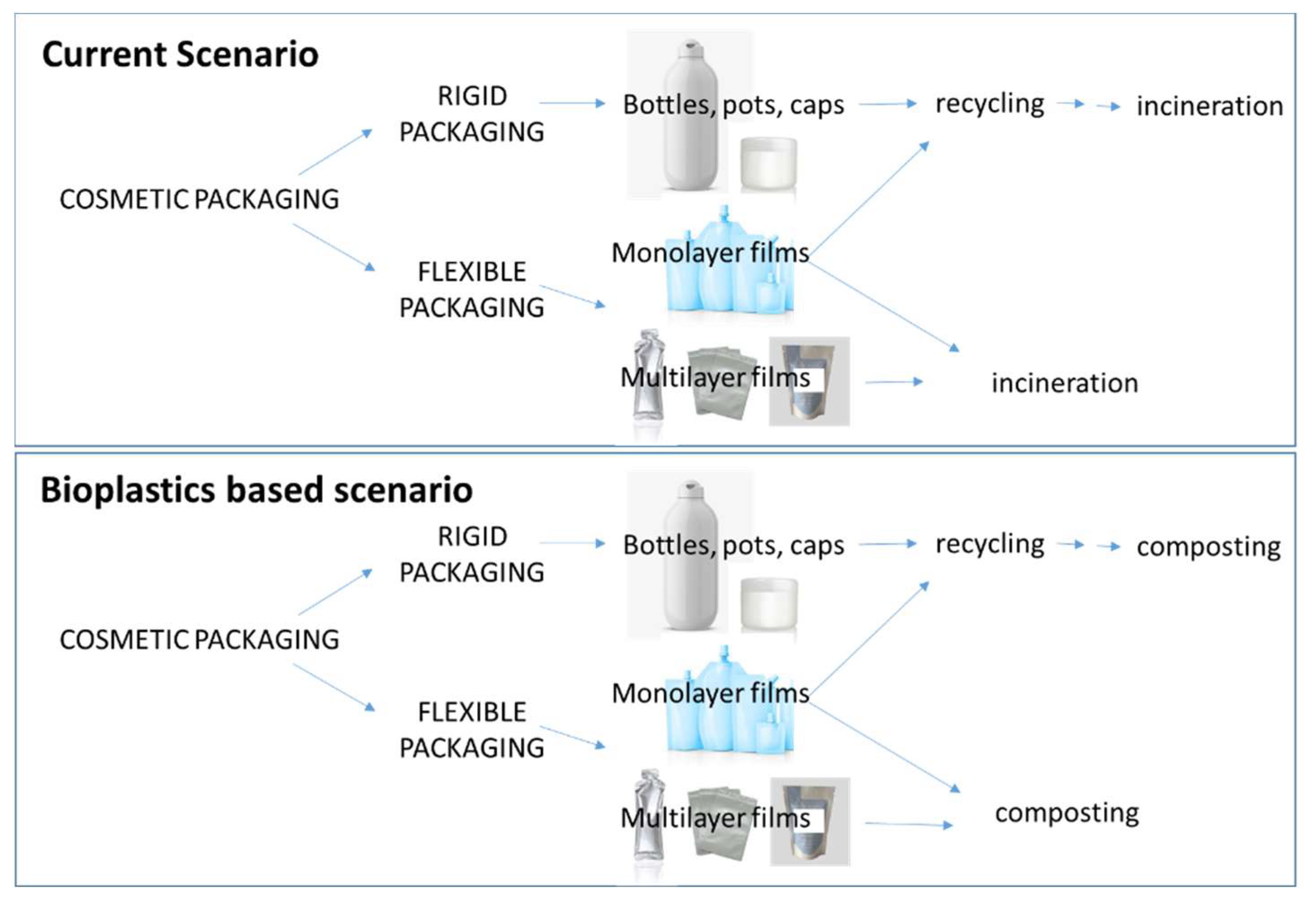
3. Plastic for Packaging of Cosmetics
4. Biodegradable Plastic for Cosmetic Packaging
4.1. Poly(lactic Acid)
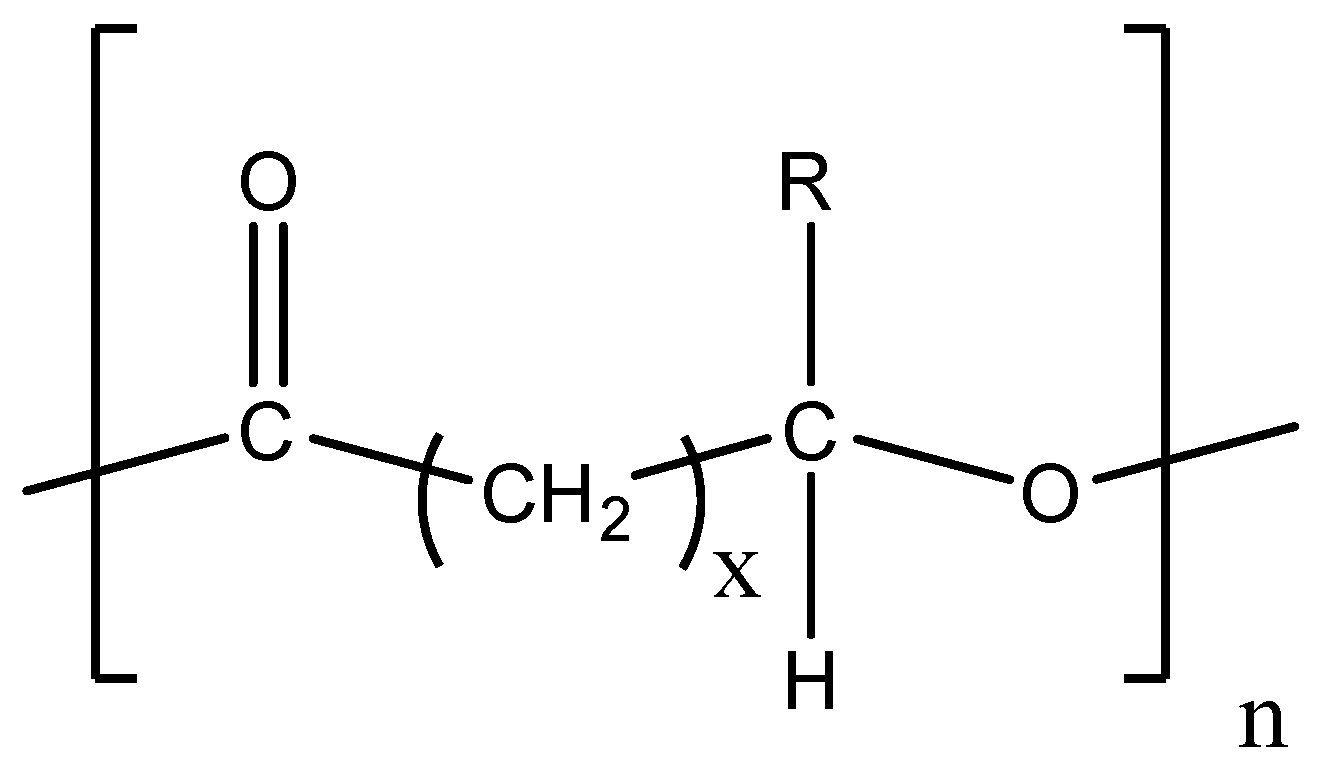
4.2. Polyhydroxyalkanoates
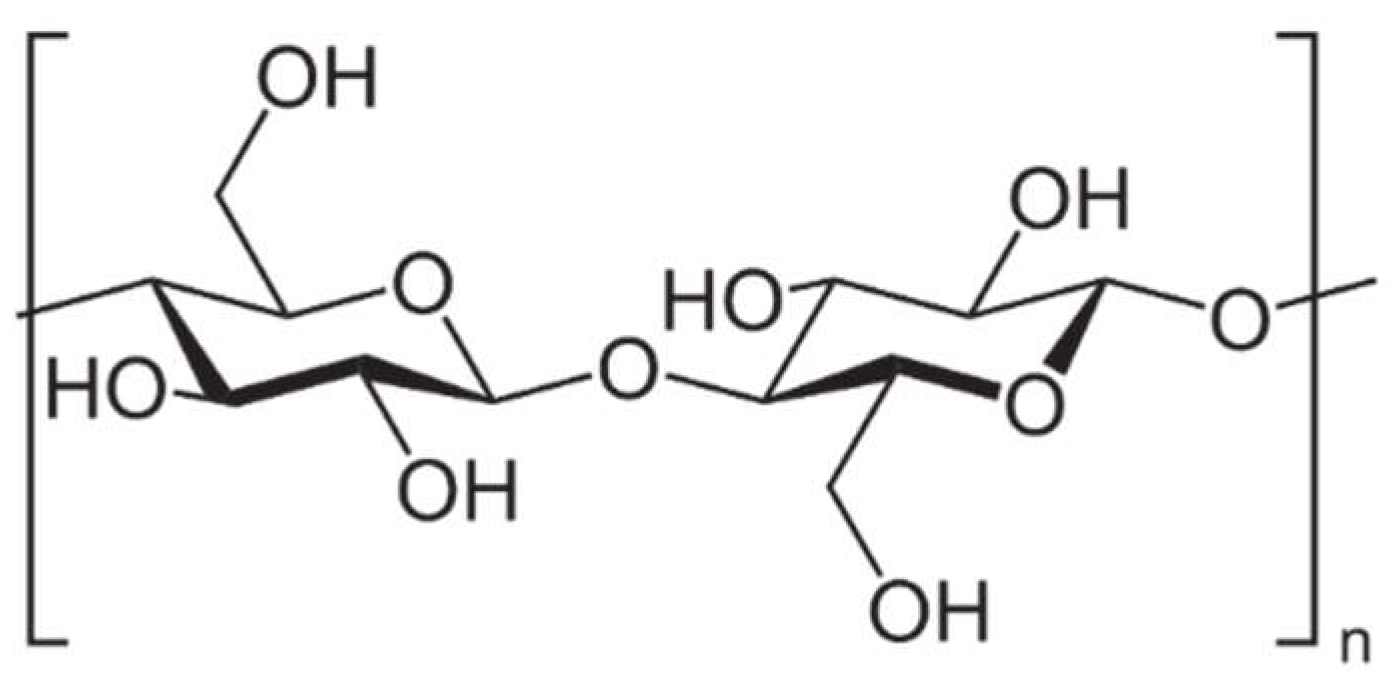
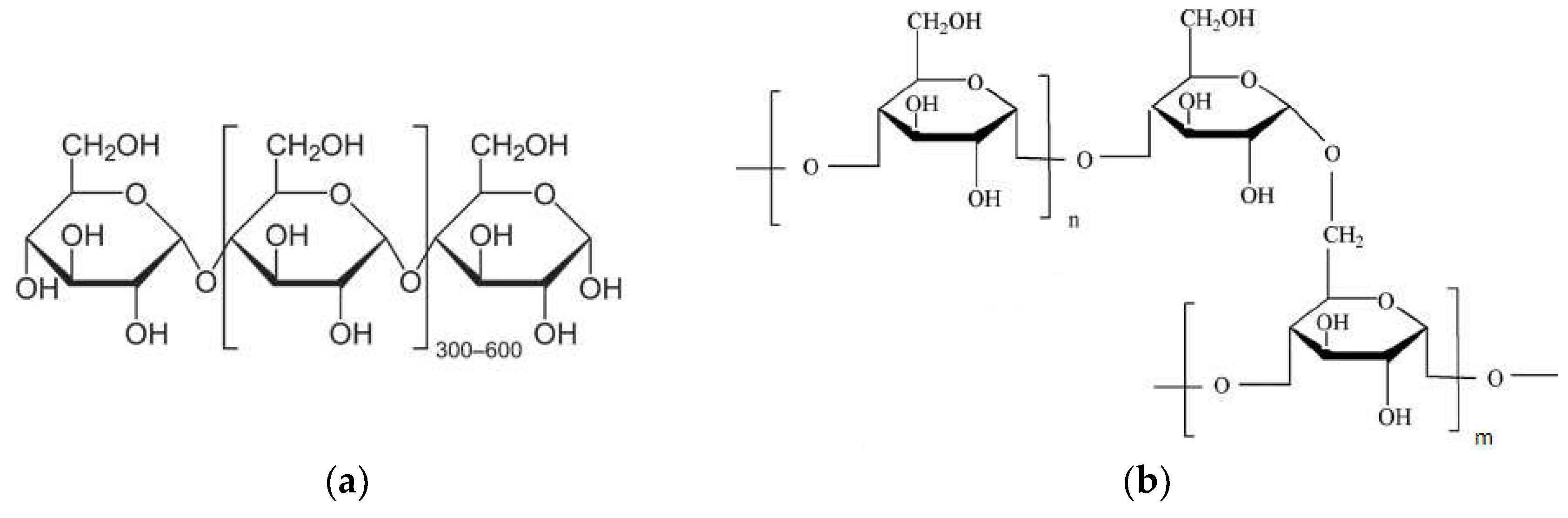
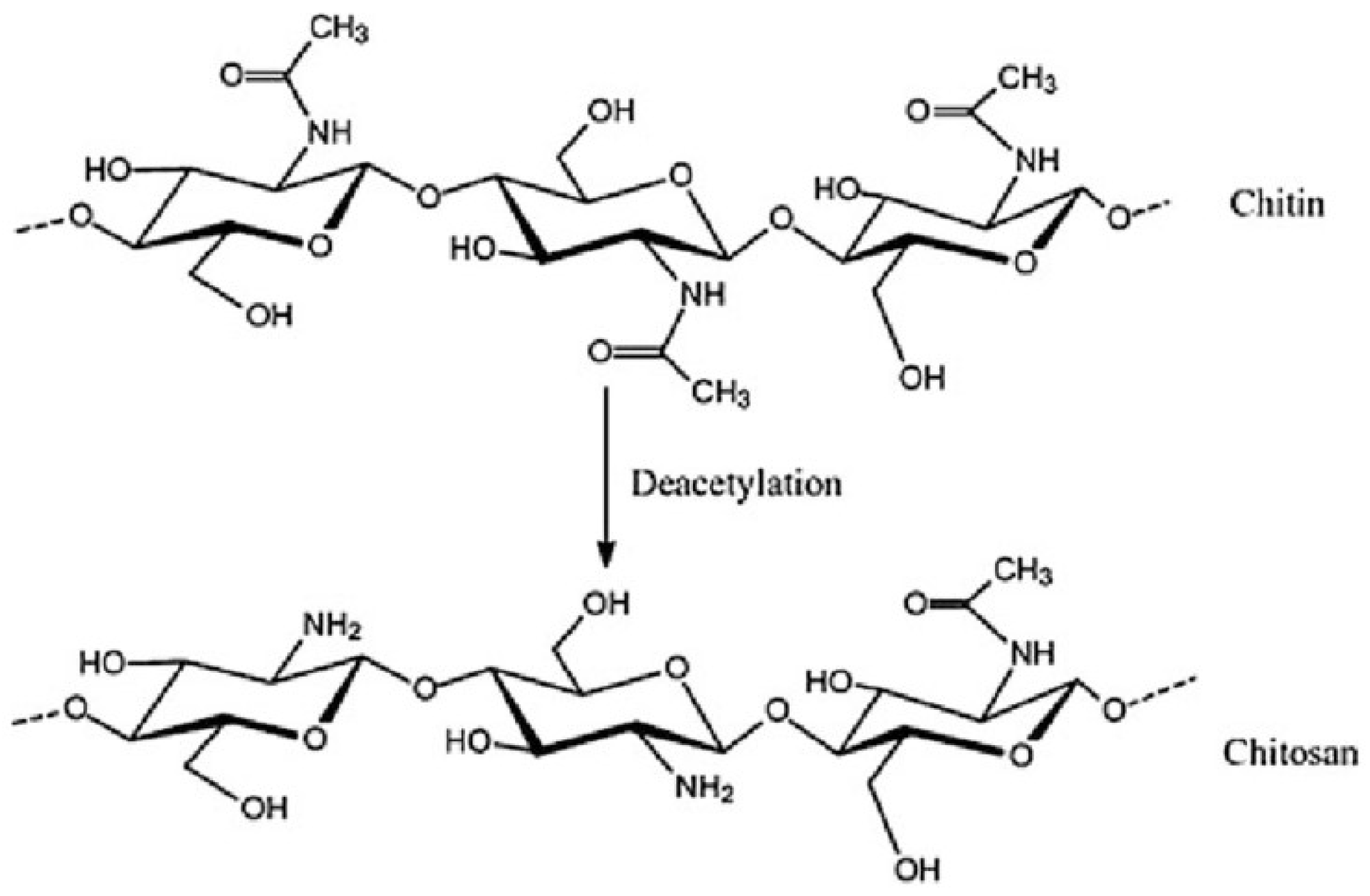
4.3. Polysaccharides
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Communication from the Commission to the European Parliament, the Council, the European Economic and Social Committee and the Committee of the Regions. A European Strategy for Plastics in a Circular Economy. Available online: http://ec.europa.eu/environment/circular-economy/pdf/plastics-strategy.pdf (accessed on 12 April 2019).
- A Sustainable Bioeconomy for Europe: Strengthening the Connection between Economy, Society and the Environment. Available online: https://ec.europa.eu/research/bioeconomy/pdf/ec_bioeconomy_strategy_2018.pdf (accessed on 2 February 2019).
- Coltelli, M.B.; Aglietto, M. Riutilizzo dei Materiali Polimerici; Edizioni Nuova Cultura: Rome, Italy, 2011. [Google Scholar]
- Landfill of Waste. Amendments Adopted by the European Parliament on 14 March 2017 on the Proposal for a Directive of the European Parliament and of the Council Amending Directive 1999/31/EC on the Landfill of Waste. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/PDF/?uri=CELEX:52017AP0071&rid=9 (accessed on 12 April 2019).
- Ehilen, O.; Obadoni, B.; Imade, F.; Eseigbe, D.; Mensah, J.K. The Effect of Detergents on the Germination and Growth of Amaranthus hybridus L. and Solanum lycopersicon L. Niger. Ann. Natural Sci. 2017, 16, 100–108. [Google Scholar]
- Fatoba, P.O.; Olorunmaiye, K.S.; Adepoju, A.O. Effects of soaps and detergents wastes on seed germination, flowering and fruiting of tomato (Lycopersico nesculentum) and Okra (Abelmoschus esculentus) plants. Ecol. Environ. Conserv. 2011, 17, 7–11. [Google Scholar]
- Alvarenga, P.; Palma, P.; Goncalves, A.P.; Fernandes, R.M.; Cunha-Queda, A.C.; Duarte, E.; Vallini, G. Evaluation of chemical and ecotoxicological characteristics of biodegradable organic residues for application to agricultural land. Environ. Int. 2007, 33, 505–513. [Google Scholar] [CrossRef]
- Position of European Bioplastics concerning Bioplastics and the Circular Economy. Available online: https://www.pac.gr/bcm/uploads/bioplastics-and-the-circular-economy.pdf (accessed on 6 January 2019).
- The Global Cosmetic Products Market by Product Type. Available online: https://www.researchandmarkets.com/reports/3329348/global-cosmetics-market-by-product-type (accessed on 8 January 2019).
- Sustainable Cosmetic Summit. Available online: http://www.sustainablecosmeticssummit.com/aboutthesummit.htm (accessed on 6 January 2019).
- Said, P.; Pradhan, R.; Sharma, N.; Naik, B. Protective Coatings for Shelf Life Extension of Fruits and Vegetables. J. Bioresour. Eng. Technol. 2014, 1, 1–6. [Google Scholar]
- Coltelli, M.B.; Coiai, S.; Bronco, S.; Passaglia, E. Nanocomposites Based on Phyllosilicates: From Petrochemicals to Renewable Thermoplastic Matrices. In Advanced Nanomaterials; Geckeler, K.E., Nishide, H., Eds.; WILEY-VCH Verlag GmbH & Co. KGaA: Weinheim, Germany, 2010; pp. 403–458. [Google Scholar]
- Rittigstein, P.; Priestley, R.D.; Broadbelt, L.J.; Torkelson, J.M. Model polymer nanocomposites provide an understanding of confinement effects in real nanocomposites. Nat. Mater. 2007, 6, 257–258. [Google Scholar] [CrossRef] [PubMed]
- Neilson, L.E. Models for the Permeability of Filled Polymer Systems. J. Macromol. Sci. Chem. 1967, 1, 929–942. [Google Scholar]
- Pandora Project, Probing Safety of Nano-Objects by Defining Immune Responses of Environmental Organisms. Available online: www.pandora-h2020.eu (accessed on 6 April 2019).
- EC4SafeNano, European Center for Risk Management and Safe Innovation in Nanomaterials and Nanotechnologies. Available online: http://ec4safenano.eu/ (accessed on 6 April 2019).
- Optinanopro, Processing and Control of Novel Nanomaterials in Packaging, Automotive and Solar Panel Processing Lines. Available online: http://optinanopro.eu/ (accessed on 6 April 2019).
- Requirements for Packaging Recoverable through Composting and Biodegradation—Test Scheme and Evaluation Criteria for the Final Acceptance of Packaging, EN 13432, 2002. Available online: https://www.en-standard.eu/din-en-13432-requirements-for-packaging-recoverable-through-composting-and-biodegradation-test-scheme-and-evaluation-criteria-for-the-final-acceptance-of-packaging-english-version-of-din-en-13432/?gclid=Cj0KCQjw7sDlBRC9ARIsAD-pDFpKF9YMYpOOIiqnfagcQWk8JaDMHbcPENZanf5vWH_0hDPCw7rpwdEaAnIdEALw_wcB (accessed on 12 April 2019).
- Pantani, R.; Sorrentino, A. Influence of crystallinity on the biodegradation rate of injection-moulded poly(lactic acid) samples in controlled composting conditions. Polym. Degrad. Stab. 2013, 98, 1089–1096. [Google Scholar] [CrossRef]
- Fortunati, E.; Armentano, I.; Iannoni, A.; Barbale, M.; Zaccheo, S.; Scavone, M.; Visai, L.; Kenny, J.M. New multifunctional poly(lactide acid) composites: Mechanical, antibacterial, and degradation properties. J. Appl. Polym. Sci. 2012, 124, 87–98. [Google Scholar] [CrossRef]
- Castro-Aguirre, E.; Iniguez-Franco, F.; Samsudin, H.; Fang, X.; Auras, R. Poly(lactic acid)-Mass production, processing, industrial applications, and end of life. Adv. Drug Deliv. Rev. 2016, 107, 333–366. [Google Scholar] [CrossRef]
- Signori, F.; Coltelli, M.B.; Bronco, S. Thermal degradation of poly(lactic acid) (PLA) and poly(butylene adipate-co-terephthalate) (PBAT) and their blends upon melt processing. Polym. Degrad. Stab. 2009, 94, 74–82. [Google Scholar] [CrossRef]
- Aliotta, L.; Cinelli, P.; Coltelli, M.B.; Lazzeri, A. Rigid filler toughening in PLA-Calcium Carbonate composites: Effect of particle surface treatment and matrix plasticization. Eur. Polym. J. 2019, 113, 78–88. [Google Scholar] [CrossRef]
- Fehri, M.K.; Mugoni, C.; Cinelli, P.; Anguillesi, I.; Coltelli, M.B.; Fiori, S.; Montorsi, M.; Lazzeri, A. Composition dependence of the synergistic effect of nucleating agent and plasticizer in poly(lactic acid): A Mixture Design study. Express Polym. Lett. 2016, 10, 274–288. [Google Scholar] [CrossRef]
- Aliotta, L.; Cinelli, P.; Coltelli, M.B.; Righetti, M.C.; Gazzano, M.; Lazzeri, A. Effect of nucleating agents on crystallinity and properties of poly (lactic acid) (PLA). Eur. Polym. J. 2017, 93, 822–832. [Google Scholar] [CrossRef]
- Mallegni, N.; Phuong, V.T.; Coltelli, M.-B.; Cinelli, P.; Lazzeri, A. Poly(lactic acid) (PLA) Based Tear Resistant and Biodegradable Flexible Films by Blown Film Extrusion. Materials 2018, 11. [Google Scholar] [CrossRef]
- Coltelli, M.-B.; Bronco, S.; Chinea, C. The effect of free radical reactions on structure and properties of poly(lactic acid) (PLA) based blends. Polym. Degrad. Stab. 2010, 95, 332–341. [Google Scholar] [CrossRef]
- Farsetti, S.; Cioni, B.; Lazzeri, A. Physico-Mechanical Properties of Biodegradable Rubber Toughened Polymers. Macromol. Symp. 2011, 301, 82–89. [Google Scholar] [CrossRef]
- Coltelli, M.-B.; Toncelli, C.; Ciardelli, F.; Bronco, S. Compatible blends of biorelated polyesters through catalytic transesterification in the melt. Polym. Degrad. Stab. 2011, 96, 982–990. [Google Scholar] [CrossRef]
- Phuong, V.T.; Coltelli, M.-B.; Cinelli, P.; Cifelli, M.; Verstichel, S.; Lazzeri, A. Compatibilization and property enhancement of poly(lactic acid)/polycarbonate blends through triacetin-mediated interchange reactions in the melt. Polymer 2014, 55, 4498–4513. [Google Scholar] [CrossRef]
- Phuong, V.T.; Gigante, V.; Aliotta, L.; Coltelli, M.-B.; Cinelli, P.; Lazzeri, A. Reactively extruded ecocomposites based on poly(lactic acid)/bisphenol A polycarbonate blends reinforced with regenerated cellulose microfibers. Compos. Sci. Technol. 2017, 139, 127–137. [Google Scholar] [CrossRef]
- Gigante, V.; Aliotta, L.; Phuong, V.T.; Coltelli, M.B.; Cinelli, P.; Lazzeri, A. Effects of waviness on fiber-length distribution and interfacial shear strength of natural fibers reinforced composites. Compos. Sci. Technol. 2017, 152, 129–138. [Google Scholar] [CrossRef]
- Phuong, V.; Verstichel, S.; Cinelli, P.; Anguillesi, I.; Coltelli, M.; Lazzeri, A. Cellulose Acetate Blends—Effect of Plasticizers on Properties and Biodegradability; Ingenta: London, UK, 2014; Volume 2. [Google Scholar]
- Coltelli, M.-B.; Mallegni, N.; Rizzo, S.; Cinelli, P.; Lazzeri, A. Improved Impact Properties in Poly(lactic acid) (PLA) Blends Containing Cellulose Acetate (CA) Prepared by Reactive Extrusion. Materials 2019, 12. [Google Scholar] [CrossRef] [PubMed]
- Coltelli, M.-B.; Maggiore, I.D.; Bertoldo, M.; Signori, F.; Bronco, S.; Ciardelli, F. Poly(lactic acid) properties as a consequence of poly(butylene adipate-co-terephthalate) blending and acetyl tributyl citrate plasticization. J. Appl. Polym. Sci. 2008, 110, 1250–1262. [Google Scholar] [CrossRef]
- Fehri, S.; Cinelli, P.; Coltelli, M.B.; Anguillesi, I.; Lazzeri, A. Thermal Properties of Plasticized Poly (Lactic Acid) (PLA) Containing Nucleating Agent. Int. J. Chem. Eng. Appl. 2016, 7, 85–88. [Google Scholar] [CrossRef]
- Sinha Ray, S.; Bousmina, M. Biodegradable polymers and their layered silicate nanocomposites: In greening the 21st century materials world. Prog. Mater. Sci. 2005, 50, 962–1079. [Google Scholar] [CrossRef]
- Castiello, S.; Coltelli, M.-B.; Conzatti, L.; Bronco, S. Comparative study about preparation of poly(lactide)/Organophilic montmorillonites nanocomposites through melt blending or ring opening polymerization methods. J. Appl. Polym. Sci. 2012, 125, E413–E428. [Google Scholar] [CrossRef]
- Scatto, M.; Salmini, E.; Castiello, S.; Coltelli, M.-B.; Conzatti, L.; Stagnaro, P.; Andreotti, L.; Bronco, S. Plasticized and nanofilled poly(lactic acid)-based cast films: Effect of plasticizer and organoclay on processability and final properties. J. Appl. Polym. Sci. 2013, 127, 4947–4956. [Google Scholar] [CrossRef]
- Jorda-Beneyto, M.; Ortuño, N.; Devis, A.; Aucejo, S.; Puerto, M.; Gutiérrez-Praena, D.; Houtman, J.; Pichardo, S.; Maisanaba, S.; Jos, A. Use of nanoclay platelets in food packaging materials: Technical and cytotoxicity approach. Food Addit. Contam. Part A 2014, 31, 354–363. [Google Scholar] [CrossRef] [PubMed]
- Maisanaba, S.; Prieto, A.I.; Pichardo, S.; Jordá-Beneyto, M.; Aucejo, S.; Jos, Á. Cytotoxicity and mutagenicity assessment of organomodified clays potentially used in food packaging. Toxicol. In Vitro 2015, 29, 1222–1230. [Google Scholar] [CrossRef]
- Development of a Bioplastic Package for Organic Cosmetic Creams-BIOBEAUTY-. Available online: https://cordis.europa.eu/project/rcn/188537/reporting/en (accessed on 2 February 2019).
- Andrzejewska, A.; Mazurkiewicz, A.; Ligaj, B. Investigation of mechanical properties the polylactide in function its degradation rate. IOP Conf. Ser. Mater. Sci. Eng. 2018, 393, 012033. [Google Scholar] [CrossRef]
- Iñiguez-Franco, F.; Auras, R.; Rubino, M.; Dolan, K.; Soto-Valdez, H.; Selke, S. Effect of nanoparticles on the hydrolytic degradation of PLA-nanocomposites by water-ethanol solutions. Polym. Degrad. Stab. 2017, 146, 287–297. [Google Scholar] [CrossRef]
- Zhang, P.; He, Y.-Y.; Gao, D.; Cai, Y.; Liu, B. Hydrolytic and thermal degradation of polyethylene glycol compatibilized poly(lactic acid)-nanocrystalline cellulose bionanocomposites. J. Appl. Polym. Sci. 2019, 136, 46933. [Google Scholar] [CrossRef]
- Bugnicourt, E.; Cinelli, P.; Lazzeri, A.; Alvarez, V. Polyhydroxyalkanoate (PHA): Review of synthesis, characteristics, processing and potential applications in packaging. Express Polym. Lett. 2014, 8, 791–808. [Google Scholar] [CrossRef]
- Sudesh, K.; Abe, H.; Doi, Y. Synthesis, structure and properties of polyhydroxyalkanoates: Biological polyesters. Prog. Polym. Sci. 2000, 25, 1503–1555. [Google Scholar] [CrossRef]
- Yoon, J.-S.; Lee, W.-S.; Jin, H.-J.; Chin, I.-J.; Kim, M.-N.; Go, J.-H. Toughening of poly(3-hydroxybutyrate) with poly(cis-1,4-isoprene). Eur. Polym. J. 1999, 35, 781–788. [Google Scholar] [CrossRef]
- Jabeen, N.; Majid, I.; Nayik, G.A. Bioplastics and food packaging: A review. Cogent Food Agric. 2015, 1, 1117749. [Google Scholar] [CrossRef]
- Haugaard, V.K.; Danielsen, B.; Bertelsen, G. Impact of polylactate and poly(hydroxybutyrate) on food quality. Eur. Food Res. Technol. 2003, 216, 233–240. [Google Scholar] [CrossRef]
- Koller, I.; Owen, A.J. Starch-Filled PHB and PHB/HV copolymer. Polym. Int. 1996, 39, 175–181. [Google Scholar] [CrossRef]
- Singh, R. Facts, Growth, and Opportunities in Industrial Biotechnology. Org. Process Res. Dev. 2011, 15, 175–179. [Google Scholar] [CrossRef]
- Yu, J.; Chen, L.X.L. The Greenhouse Gas Emissions and Fossil Energy Requirement of Bioplastics from Cradle to Gate of a Biomass Refinery. Environ. Sci. Technol. 2008, 42, 6961–6966. [Google Scholar] [CrossRef]
- Chen, Y.; Tsai, Y.-H.; Chou, I.N.; Tseng, S.-H.; Wu, H.-S. Application of Biodegradable Polyhydroxyalkanoates as Surgical Films for Ventral Hernia Repair in Mice. Int. J. Polym. Sci. 2014, 2014. [Google Scholar] [CrossRef]
- Deroiné, M.; Le Duigou, A.; Corre, Y.-M.; Le Gac, P.-Y.; Davies, P.; César, G.; Bruzaud, S. Seawater accelerated ageing of poly(3-hydroxybutyrate-co-3-hydroxyvalerate). Polym. Degrad. Stab. 2014, 105, 237–247. [Google Scholar] [CrossRef]
- Volova, T.G.; Boyandin, A.N.; Vasiliev, A.D.; Karpov, V.A.; Prudnikova, S.V.; Mishukova, O.V.; Boyarskikh, U.A.; Filipenko, M.L.; Rudnev, V.P.; Bá Xuân, B.; et al. Biodegradation of polyhydroxyalkanoates (PHAs) in tropical coastal waters and identification of PHA-degrading bacteria. Polym. Degrad. Stab. 2010, 95, 2350–2359. [Google Scholar] [CrossRef]
- Seggiani, M.; Cinelli, P.; Balestri, E.; Mallegni, N.; Stefanelli, E.; Rossi, A.; Lardicci, C.; Lazzeri, A. Novel Sustainable Composites Based on Poly(hydroxybutyrate-co-hydroxyvalerate) and Seagrass Beach-CAST Fibers: Performance and Degradability in Marine Environments. Materials 2018, 11. [Google Scholar] [CrossRef]
- Mlalila, N.; Hilonga, A.; Swai, H.; Devlieghere, F.; Ragaert, P. Antimicrobial packaging based on starch, poly(3-hydroxybutyrate) and poly(lactic-co-glycolide) materials and application challenges. Trends Food Sci. Technol. 2018, 74, 1–11. [Google Scholar] [CrossRef]
- Avérous, L.; Fringant, C.; Moro, L. Plasticized starch–cellulose interactions in polysaccharide composites. Polymer 2001, 42, 6565–6572. [Google Scholar] [CrossRef]
- Gennadios, A.; Hanna, M.A.; Kurth, L.B. Application of Edible Coatings on Meats, Poultry and Seafoods: A Review. LWT—Food Sci. Technol. 1997, 30, 337–350. [Google Scholar] [CrossRef]
- Cyras, V.P.; Commisso, M.S.; Mauri, A.N.; Vázquez, A. Biodegradable double-layer films based on biological resources: Polyhydroxybutyrate and cellulose. J. Appl. Polym. Sci. 2007, 106, 749–756. [Google Scholar] [CrossRef]
- Demirgöz, D.; Elvira, C.; Mano, J.F.; Cunha, A.M.; Piskin, E.; Reis, R.L. Chemical modification of starch based biodegradable polymeric blends: Effects on water uptake, degradation behaviour and mechanical properties. Polym. Degrad. Stab. 2000, 70, 161–170. [Google Scholar] [CrossRef]
- Mensitieri, G.; Di Maio, E.; Buonocore, G.G.; Nedi, I.; Oliviero, M.; Sansone, L.; Iannace, S. Processing and shelf life issues of selected food packaging materials and structures from renewable resources. Trends Food Sci. Technol. 2011, 22, 72–80. [Google Scholar] [CrossRef]
- Chiellini, E.; Cinelli, P.; Ilieva, V.I.; Imam, S.H.; Lawton, J.W. Environmentally Compatible Foamed Articles Based on Potato Starch, Corn Fiber, and Poly(Vinyl Alcohol). J. Cell. Plast. 2009, 45, 17–32. [Google Scholar] [CrossRef]
- Coltelli, M.B.; Danti, S.; Trombi, L.; Morganti, P.; Donnarumma, G.; Baroni, A.; Fusco, A.; Lazzeri, A. Preparation of Innovative Skin Compatible Films to Release Polysaccharides for Biobased Beauty Masks. Cosmetics 2018, 5, 70. [Google Scholar] [CrossRef]
- Verlee, A.; Mincke, S.; Stevens, C.V. Recent developments in antibacterial and antifungal chitosan and its derivatives. Carbohydr. Polym. 2017, 164, 268–283. [Google Scholar] [CrossRef]
- Devlieghere, F.; Vermeulen, A.; Debevere, J. Chitosan: Antimicrobial activity, interactions with food components and applicability as a coating on fruit and vegetables. Food Microbiol. 2004, 21, 703–714. [Google Scholar] [CrossRef]
- Higueras Contreras, L.; López-Carballo, G.; Hernández-Muñoz, P.; Catala, R.; Gavara, R. Antimicrobial packaging of chicken fillets based on the release of carvacrol from chitosan/cyclodextrin films. Int. J. Food Microbiol. 2014, 188C, 53–59. [Google Scholar] [CrossRef]
- Zhonga, Y.; Cavenderb, G.; Zhao, Y. Investigation of different coating application methods on the performance of edible coatings on Mozzarella cheese. LWT—Food Sci. Tecnnol. 2014, 56, 1–8. [Google Scholar] [CrossRef]
- Arancibia, M.Y.; Lòpez-Caballero, M.E.; Gòmez-Guillen, M.C.; Montero, P. Chitosan coatings enriched with active shrimp waste for shrimp preservation. Food Control 2015, 54, 259–266. [Google Scholar] [CrossRef]
- Valenzuela, C.; Tapia, C.; López, L.; Bunger, A.; Escalona, V.; Abugoch, L. Effect of edible quinoa protein-chitosan based films on refrigerated strawberry (Fragaria × ananassa) quality. Electron. J. Biotechnol. 2015, 18, 406–411. [Google Scholar] [CrossRef]
- Cinelli, P.; Coltelli, M.; Mallegni, N.; Morganti, P.; Lazzeri, A. Degradability and Sustainability of Nanocomposites Based on Polylactic Acid and Chitin Nano Fibrils. Chem. Eng. 2017, 60. [Google Scholar]
- Zheng, L.-Y.; Zhu, J.-F. Study on antimicrobial activity of chitosan with different molecular weights. Carbohydr. Polym. 2003, 54, 527–530. [Google Scholar] [CrossRef]
- Morganti, P.; Fusco, A.; Paoletti, I.; Perfetto, B.; Del Ciotto, P.; Palombo, M.; Chianese, A.; Baroni, A.; Donnarumma, G. Anti-Inflammatory, Immunomodulatory, and Tissue Repair Activity on Human Keratinocytes by Green Innovative Nanocomposites. Materials 2017, 10, 843. [Google Scholar] [CrossRef]
- Coltelli, M.-B.; Gigante, V.; Panariello, L.; Aliotta, L.; Morganti, P.; Danti, S.; Cinelli, P.; Lazzeri, A. Chitin nanofibrils in renewable materials for packaging and personal care applications. Adv. Mater. Lett. 2019. [Google Scholar] [CrossRef]
- Coltelli, M.-B.; Cinelli, P.; Gigante, V.; Aliotta, L.; Morganti, P.; Panariello, L.; Lazzeri, A. Chitin Nanofibrils in Poly(Lactic Acid) (PLA) Nanocomposites: Dispersion and Thermo-Mechanical Properties. Int. J. Mol. Sci. 2019, 20, 504. [Google Scholar] [CrossRef]
- Panariello, L.; Coltelli, M.-B.; Buchignani, M.; Lazzeri, A. Chitosan and nano-structured chitin for biobased anti-microbial treatments onto cellulose based materials. Eur. Polym. J. 2019, 113, 328–339. [Google Scholar] [CrossRef]
- Morganti, P.; Danti, S.; Coltelli, M.B. Chitin and lignin to produce biocompatible tissues. Res. Clin. Dermatol. 2018, 1, 5–11. [Google Scholar]
- Morganti, P.; Coltelli, M.B. A New Carrier for Advanced Cosmeceuticals. Cosmetics 2019, 6, 10. [Google Scholar] [CrossRef]
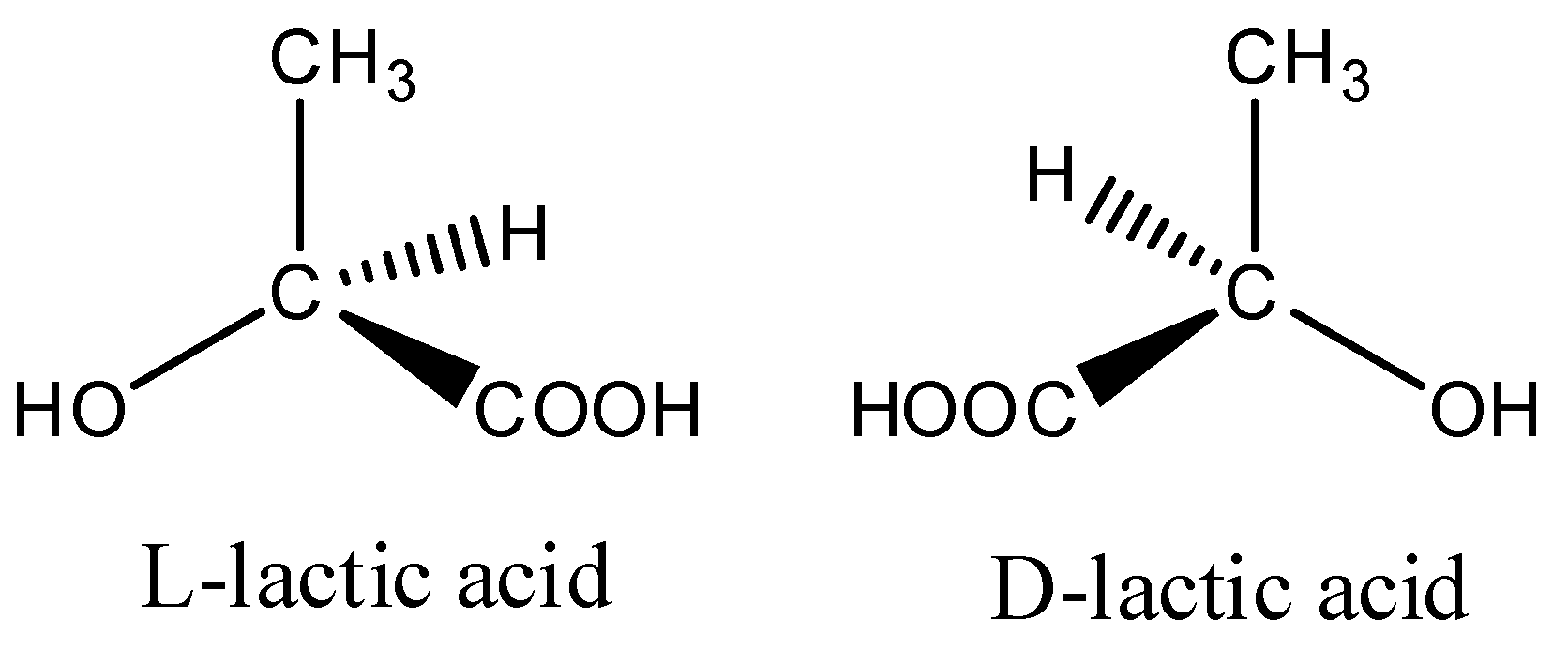
| Material | Share % |
|---|---|
| Packaging | 59 |
| Others | 14 |
| Electrical and electronics equipment | 8 |
| Agriculture | 5 |
| Automotive | 5 |
| Construction | 5 |
| Household (non packaging) | 4 |
| Additives | Properties | Applications | Processing Technologies | References |
|---|---|---|---|---|
| Plasticizers | Decreased Modulus and increased strain at break | Flexible packaging | Flat die extrusion, blow extrusion | [25,27,36,37] |
| Nucleant agents | Increased crystallinity and elastic Modulus | Rigid packaging | Injection molding | [25,26] |
| Polymers | Increased impact properties, increased strain at break | Rigid packaging | Flat die extrusion (followed by thermoforming), injection molding | [28,29,30,31,35] |
| Fillers | Increased Elastic Modulus and tensile strength | Rigid packaging | Flat die extrusion (followed by thermoforming), injection molding | [32,33,38,39,40] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cinelli, P.; Coltelli, M.B.; Signori, F.; Morganti, P.; Lazzeri, A. Cosmetic Packaging to Save the Environment: Future Perspectives. Cosmetics 2019, 6, 26. https://doi.org/10.3390/cosmetics6020026
Cinelli P, Coltelli MB, Signori F, Morganti P, Lazzeri A. Cosmetic Packaging to Save the Environment: Future Perspectives. Cosmetics. 2019; 6(2):26. https://doi.org/10.3390/cosmetics6020026
Chicago/Turabian StyleCinelli, Patrizia, Maria Beatrice Coltelli, Francesca Signori, Pierfrancesco Morganti, and Andrea Lazzeri. 2019. "Cosmetic Packaging to Save the Environment: Future Perspectives" Cosmetics 6, no. 2: 26. https://doi.org/10.3390/cosmetics6020026
APA StyleCinelli, P., Coltelli, M. B., Signori, F., Morganti, P., & Lazzeri, A. (2019). Cosmetic Packaging to Save the Environment: Future Perspectives. Cosmetics, 6(2), 26. https://doi.org/10.3390/cosmetics6020026









