Review of Modern Techniques for the Assessment of Skin Hydration
Abstract
1. Introduction
2. Differential Scanning Calorimetry
3. Electrical-Based Methods
4. Transepidermal Water Loss
5. Skin Elasticity
6. Photothermal Radiometry
7. Attenuated Total Reflection Infrared Spectroscopy
8. Nuclear Magnetic Resonance
9. Optical Coherence Tomography
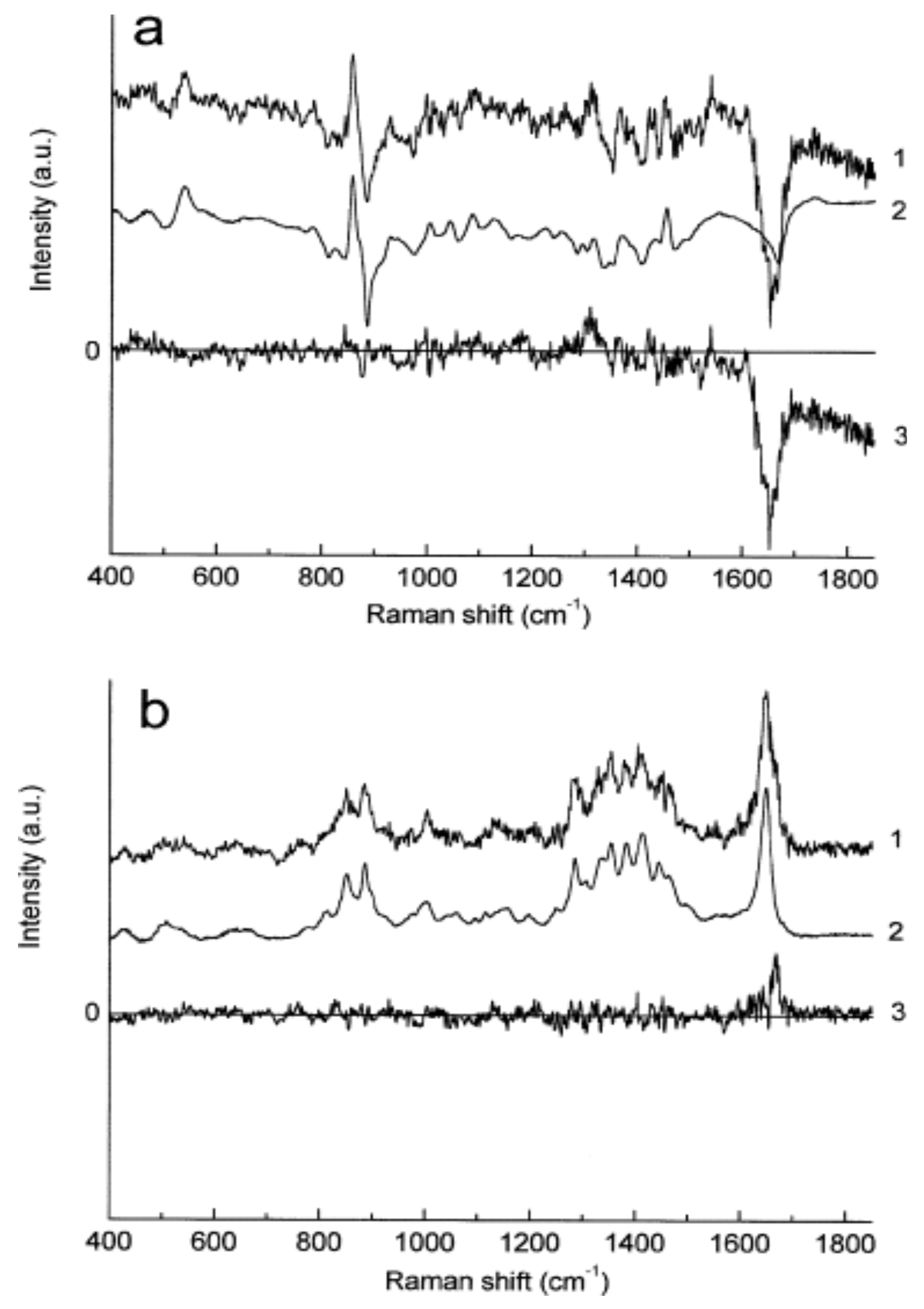
10. Raman Spectroscopy
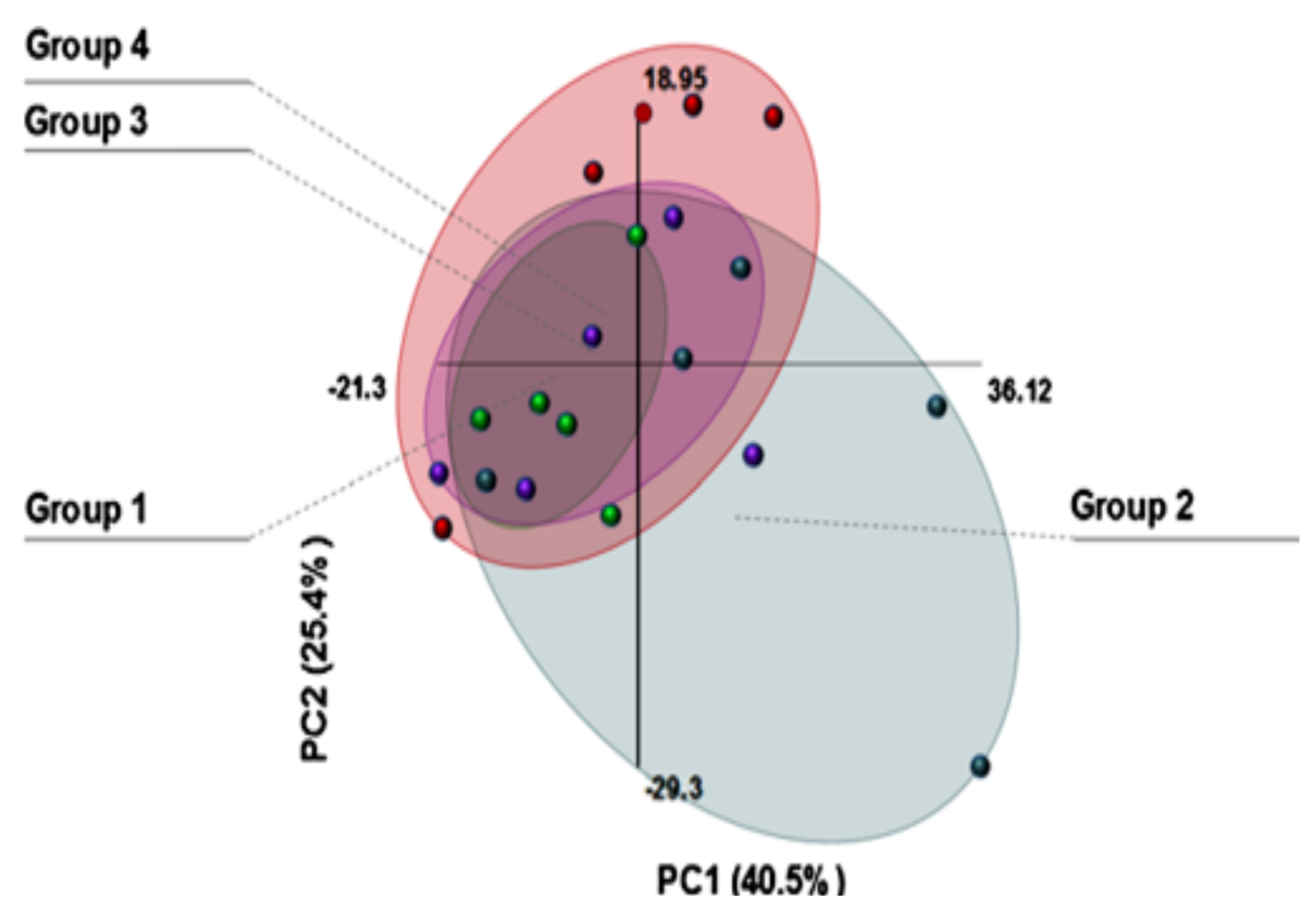
11. Confocal Raman Microscopy
12. Near Infrared Spectroscopy
13. Discussion
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| SC | Stratum Corneum |
| RH | Relative Humidity |
| DSC | Differential Scanning Calorimetry |
| SCIM | Surface-Characterizing Impedance monitor |
| TEWL | Transepidermal Water Loss |
| OTTER | Opto-Thermal Transient Emission Radiometry |
| WVFD | Water vapor flux density |
| ATR-IR | Attenuated Total Reflection Infrared |
| ATR-FTIR | Attenuated Total Reflection Fourier Transform Infrared |
| IRE | Internal Reflectance Element |
| NMR | Nuclear Magnetic Resonance |
| MRI | Magnetic Resonance imaging |
| OCT | Optical Coherence Tomography |
| TDOCT | Time-Domain Optical Coherence Tomography |
| SDOCT | Spectral-Domain Optical Coherence Tomography |
| RS | Raman Spectroscopy |
| CRM | Confocal Raman Microscopy |
| PLS | Partial Least-Squares |
| NIR | Near Infrared |
| PCA | Principal Component Analysis |
| ANN | Artificial Neural Networks |
References
- Marieb, E.N.; Mallatt, J. Human Anatomy, 2nd ed.; Benjamin-Cummings Pub Co.: San Francisco, CA, USA, 1996. [Google Scholar]
- Leyden, J.J.; Rawlings, A.V. Skin Moisturization, 1st ed.; Informa Healthcare: London, UK, 2002. [Google Scholar]
- Chiu, M.H.; Prenner, E.J. Differential scanning calorimetry: An invaluable tool for a detailed thermodynamic characterization of macromolecules and their interactions. J. Pharm. Bioallied Sci. 2011, 3, 39–59. [Google Scholar] [CrossRef] [PubMed]
- Agache, P.G.; Humbert, P.; Maibach, H.I. Measuring the Skin; Springer: Berlin, Germany, 2004. [Google Scholar]
- Golden, G.M.; Guzek, D.B.; Harris, R.R.; McKie, J.E.; Potts, R.O. Lipid thermotropic transitions in human stratum corneum. J. Investig. Dermatol. 1986, 86, 255–259. [Google Scholar] [CrossRef]
- Khan, Z.; Kellaway, I. Differential scanning calorimetry of dimethylsulphoxide-treated human stratum corneum. Int. J. Pharm. 1989, 55, 129–134. [Google Scholar] [CrossRef]
- Golden, G.M.; Guzek, D.B.; Kennedy, A.H.; McKie, J.E.; Potts, R.O. Stratum corneum lipid phase transitions and water barrier properties. Biochemistry 1987, 26, 2382–2388. [Google Scholar] [CrossRef] [PubMed]
- Barry, B. Mode of action of penetration enhancers in human skin. J. Control. Release 1987, 6, 85–97. [Google Scholar] [CrossRef]
- Potts, R.O.; Golden, G.M.; Francoeur, M.L.; Mak, V.H.; Guy, R.H. Mechanism and enhancement of solute transport across the stratum corneum. J. Control. Release 1991, 15, 249–260. [Google Scholar] [CrossRef]
- Al-Saidan, S.; Barry, B.; Williams, A. Differential scanning calorimetry of human and animal stratum corneum membranes. Int. J. Pharm. 1998, 168, 17–22. [Google Scholar] [CrossRef]
- Papir, Y.S.; Hsu, K.H.; Wildnauer, R.H. The mechanical properties of stratum corneum. I. The effect of water and ambient temperature on the tensile properties of newborn rat stratum corneum. Biochim. Biophys. Acta 1975, 399, 170–180. [Google Scholar] [CrossRef]
- Miller, D.L.; Wildnauer, R.H. Thermoanalytical probes for the analysis of physical properties of stratum corneum. J. Investig. Dermatol. 1977, 69, 287–289. [Google Scholar] [CrossRef]
- Inoue, T.; Tsujii, K.; Okamoto, K.; Toda, K. Differential scanning calorimetric studies on the melting behavior of water in stratum corneum. J. Investig. Dermatol. 1986, 86, 689–693. [Google Scholar] [CrossRef]
- Imokawa, G.; Kuno, H.; Kawai, M. Stratum Corneum Lipids Serve as a Bound-Water Modulator. J. Investig. Dermatol. 1991, 96, 845–851. [Google Scholar] [CrossRef] [PubMed]
- Perez, B.; Dahlgaard, S.E.; Bulsara, P.; Rawlings, A.V.; Jensen, M.M.; Dong, M.; Glasius, M.; Clarke, M.J.; Guo, Z. Synthesis and characterization of O-acylated-ω-hydroxy fatty acids as skin-protecting barrier lipids. J. Colloid Interface Sci. 2017, 490, 137–146. [Google Scholar] [CrossRef] [PubMed]
- Perez, B.; Hansen, B.S.; Bulsara, P.A.; Rawlings, A.V.; Clarke, M.J.; Guo, Z. Fractionated aliphatic alcohols as synthetic precursors of ultra long-chain monoacylglycerols for cosmetic applications. Int. J. Cosmet. Sci. 2017, 39, 511–517. [Google Scholar] [CrossRef]
- Libio, I.C.; Demori, R.; Ferrão, M.F.; Lionzo, M.I.Z.; da Silveira, N.P. Films based on neutralized chitosan citrate as innovative composition for cosmetic application. Mater. Sci. Eng. C Mater. Biol. Appl. 2016, 67, 115–124. [Google Scholar] [CrossRef]
- Täuber, A.; Müller-Goymann, C.C. In vitro model of infected stratum corneum for the efficacy evaluation of poloxamer 407-based formulations of ciclopirox olamine against Trichophyton rubrum as well as differential scanning calorimetry and stability studies. Int. J. Pharm. 2015, 494, 304–311. [Google Scholar] [CrossRef]
- Gazga-Urioste, C.; Rivera-Becerril, E.; Pérez-Hernández, G.; Angélica Noguez-Méndez, N.; Faustino-Vega, A.; Tomás Quirino-Barreda, C. Physicochemical characterization and thermal behavior of hexosomes containing ketoconazole as potential topical antifungal delivery system. Drug Dev. Ind. Pharm. 2018, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Rubio, L.; Alonso, C.; Rodríguez, G.; Cócera, M.; Barbosa-Barros, L.; Coderch, L.; de la Maza, A.; Parra, J.L.; López, O. Bicellar systems as vehicle for the treatment of impaired skin. Eur. J. Pharm. Biopharm. 2014, 86, 212–218. [Google Scholar] [CrossRef]
- Montenegro, L.; Castelli, F.; Sarpietro, M.G. Differential Scanning Calorimetry Analyses of Idebenone-Loaded Solid Lipid Nanoparticles Interactions with a Model of Bio-Membrane: A Comparison with In Vitro Skin Permeation Data. Pharmaceuticals 2018, 11, 138. [Google Scholar] [CrossRef]
- Ansari, H.; Singh, P. Formulation and in-vivo Evaluation of Novel Topical Gel of Lopinavir for Targeting HIV. Curr. HIV Res. 2018. [Google Scholar] [CrossRef]
- Pireddu, R.; Sinico, C.; Ennas, G.; Schlich, M.; Valenti, D.; Murgia, S.; Marongiu, F.; Fadda, A.M.; Lai, F. The effect of diethylene glycol monoethyl ether on skin penetration ability of diclofenac acid nanosuspensions. Colloids Surf. B Biointerfaces 2018, 162, 8–15. [Google Scholar] [CrossRef]
- Lauterbach, A.; Mueller-Goymann, C.C. Development, formulation, and characterization of an adapalene-loaded solid lipid microparticle dispersion for follicular penetration. Int. J. Pharm. 2014, 466, 122–132. [Google Scholar] [CrossRef] [PubMed]
- Walters, K.A.; Roberts, M.S. Dermatologic, Cosmeceutic, and Cosmetic Development: Therapeutic and Novel Approaches, 1st ed.; CRC Press: Boca Raton, FL, USA, 2007. [Google Scholar]
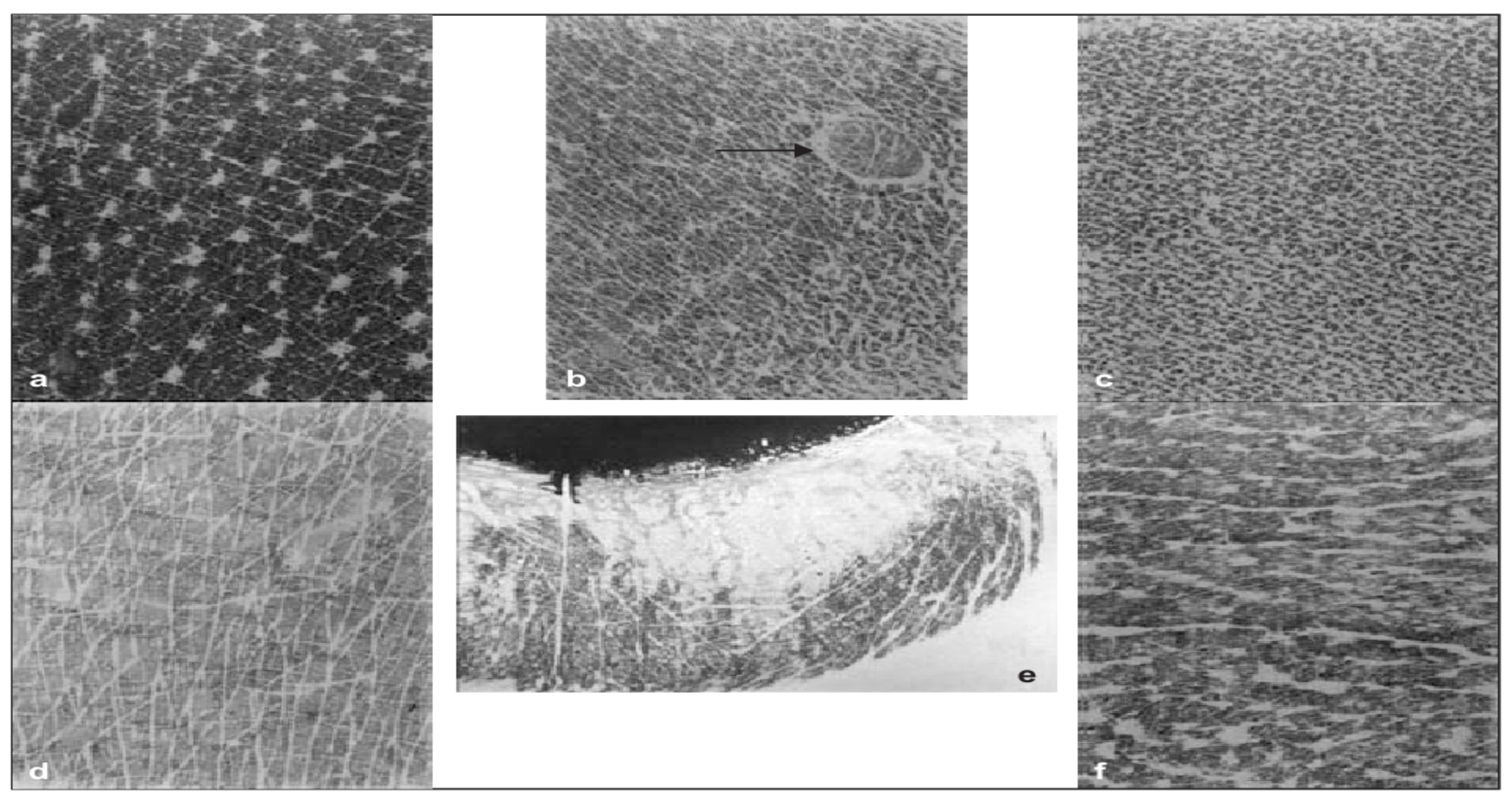
- Pierard, G. Skin capacitance imageing for the dermatologist. Dermatology 2005, 210, 3–7. [Google Scholar]
- Batisse, D.; Giron, F.; Lévêque, J.L. Capacitance imageing of the skin surface. Skin Res. Technol. 2006, 12, 99–104. [Google Scholar] [CrossRef] [PubMed]
- Lévêque, J.L.; Querleux, B. SkinChip, a new tool for investigating the skin surface in vivo. Skin Res. Technol. 2003, 9, 343–347. [Google Scholar] [CrossRef] [PubMed]
- Leveque, J.L.; Xhauflaire-Uhoda, E.; Pierard, G.E. Skin capacitance imageing, a new technique for investigating the skin surface. Eur. J. Dermatol. 2006, 16, 500–506. [Google Scholar] [PubMed]
- Diridollou, S.; de Rigal, J.; Querleux, B.; Leroy, F.; Holloway Barbosa, V. Comparative study of the hydration of the stratum corneum between four ethnic groups: Influence of age. Int. J. Dermatol. 2007, 46 (Suppl. 1), 11–14. [Google Scholar] [CrossRef] [PubMed]
- Crowther, J.M. Understanding the effects of topography on skin moisturization measurement via two-dimensional capacitance imageing. Int. J. Cosmet. Sci. 2017, 39, 572–578. [Google Scholar] [CrossRef] [PubMed]
- Xhauflaire-Uhoda, E.; Pierard-Franchimont, C.; Pierard, G.E. Skin capacitance mapping of psoriasis. J. Eur. Acad. Dermatol. Venereol. 2006, 20, 1261–1265. [Google Scholar] [CrossRef]
- Pierard-Franchimont, C.; Pierard, G. Sweat Gland Awakening on Physical Training: A Skin Capacitance Mapping Observation. Clin. Res. Dermatol. 2015, 2, 1–4. [Google Scholar] [CrossRef]
- Klang, V.; Schwarz, J.C.; Haberfeld, S.; Xiao, P.; Wirth, M.; Valenta, C. Skin integrity testing and monitoring of in vitro tape stripping by capacitance-based sensor imageing. Skin Res. Technol. 2013, 19, e259–e272. [Google Scholar] [CrossRef]
- Xhauflaire-Uhoda, E.; Mayeux, G.; Quatresooz, P.; Scheen, A.; Pierard, G.E. Facing up to the imperceptible perspiration. Modulatory influences by diabetic neuropathy, physical exercise and antiperspirant. Skin Res. Technol. 2011, 17, 487–493. [Google Scholar] [CrossRef] [PubMed]
- Xhauflaire-Uhoda, E.; Pierard-Franchimont, C.; Pierard, G.E.; Quatresooz, P. Weathering of the hairless scalp: A study using skin capacitance imageing and ultraviolet light-enhanced visualization. Clin. Exp. Dermatol. 2010, 35, 83–85. [Google Scholar] [CrossRef] [PubMed]
- Leveque, J.L.; Goubanova, E. Influence of age on the lips and perioral skin. Dermatology 2004, 208, 307–313. [Google Scholar] [CrossRef] [PubMed]
- Xhauflaire-Uhoda, E.; Pierard, G.E. Skin capacitance imageing of acne lesions. Skin Res. Technol. 2007, 13, 9–12. [Google Scholar] [CrossRef] [PubMed]
- Xhauflaire-Uhoda, E.; Loussouarn, G.; Haubrechts, C.; Leger, D.S.; Pierard, G.E. Skin capacitance imageing and corneosurfametry. A comparative assessment of the impact of surfactants on stratum corneum. Contact Dermat. 2006, 54, 249–253. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Bontozoglou, C.; Chirikhina, E.; Lane, M.E.; Xiao, P. Capacitive Imageing for Skin Characterizations and Solvent Penetration Measurements. Cosmetics 2018, 5, 52. [Google Scholar] [CrossRef]
- Berardesca, E.; Loden, M.; Serup, J.; Masson, P.; Rodrigues, L.M. The revised EEMCO guidance for the in vivo measurement of water in the skin. Skin Res. Technol. 2018, 24, 351–358. [Google Scholar] [CrossRef]
- Gefen, A. Bioengineering Research of Chronic Wounds: A Multidisciplinary Study Approach; Springer: Berlin, Germany, 2009. [Google Scholar]
- Farahmand, S.; Tien, L.; Hui, X.; Maibach, H.I. Measuring transepidermal water loss: A comparative in vivo study of condenser-chamber, unventilated-chamber and open-chamber systems. Skin Res. Technol. 2009, 15, 392–398. [Google Scholar] [CrossRef]
- Nuutinen, J.; Alanen, E.; Autio, P.; Lahtinen, M.R.; Harvima, I.; Lahtinen, T. A closed unventilated chamber for the measurement of transepidermal water loss. Skin Res. Technol. 2003, 9, 85–89. [Google Scholar] [CrossRef]
- Chilcott, R.; Price, S. Principles and Practice of Skin Toxicology; John Wiley & Sons: Hoboken, NJ, USA, 2008. [Google Scholar]
- De Paepe, K.; Houben, E.; Adam, R.; Wiesemann, F.; Rogiers, V. Validation of the VapoMeter, a closed unventilated chamber system to assess transepidermal water loss vs. the open chamber Tewameter. Skin Res. Technol. 2005, 11, 61–69. [Google Scholar] [CrossRef]
- Zhai, H.; Wilhelm, K.P.; Maibach, H.I. Marzulli and Maibach’s Dermatotoxicology, 7th ed.; CRC Press: Boca Raton, FL, USA, 2007. [Google Scholar]
- Bennett, S.; Jones, C.; Matheson, J.R. Closed chamber and open chamber TEWL measurements: A comparison of Dermalab(R) and Aquaflux AF102 instruments. In Proceedings of the 2005 World Congress on Noninvasive Studies of the Skin, Wilmington, DE, USA, 28 September–1 October 2005; p. 46. [Google Scholar]
- Tian, W. Comparison of three TEWL instruments for in vitro and in vivo measurements. In Proceedings of the 2005 World Congress onNoninvasive Studies of the Skin, Wilmington, DE, USA, 28 September–1 October 2005; p. 75. [Google Scholar]
- Rogiers, V.; EEMCO Group. EEMCO guidance for the assessment of transepidermal water loss in cosmetic sciences. Skin Pharmacol. Appl. Skin Physiol. 2001, 14, 117–128. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Lukic, M.; Savic, S.; Lunter, D.J. Reinforcement of barrier function—Skin repair formulations to deliver physiological lipids into skin. Int. J. Cosmet. Sci. 2018, 40, 494–501. [Google Scholar] [CrossRef] [PubMed]
- De Paepe, K.; Roseeuw, D.; Rogiers, V. Repair of acetone- and sodium lauryl sulphate-damaged human skin barrier function using topically applied emulsions containing barrier lipids. J. Eur. Acad. Dermatol. Venereol. 2002, 16, 587–594. [Google Scholar] [CrossRef] [PubMed]
- Chiba, T.; Nakahara, T.; Kohda, F.; Ichiki, T.; Manabe, M.; Furue, M. Measurement of trihydroxy-linoleic acids in stratum corneum by tape-stripping: Possible biomarker of barrier function in atopic dermatitis. PLoS ONE 2019, 14, e0210013. [Google Scholar] [CrossRef] [PubMed]
- Lunnemann, L.; Ludriksone, L.; Schario, M.; Sawatzky, S.; Stroux, A.; Blume-Peytavi, U.; Garcia Bartels, N. Noninvasive monitoring of plant-based formulations on skin barrier properties in infants with dry skin and risk for atopic dermatitis. Int. J. Women’s Dermatol. 2018, 4, 95–101. [Google Scholar] [CrossRef]
- Yang, J.E.; Ngo, H.T.T.; Hwang, E.; Seo, S.A.; Park, S.W.; Yi, T.H. Dietary enzyme-treated Hibiscus syriacus L. protects skin against chronic UVB-induced photoageing via enhancement of skin hydration and collagen synthesis. Arch. Biochem. Biophys. 2019, 662, 190–200. [Google Scholar] [CrossRef] [PubMed]
- Sundaram, H.; Mackiewicz, N.; Burton, E.; Peno-Mazzarino, L.; Lati, E.; Meunier, S. Pilot Comparative Study of the Topical Action of a Novel, Crosslinked Resilient Hyaluronic Acid on Skin Hydration and Barrier Function in a Dynamic, Three-Dimensional Human Explant Model. J. Drugs Dermatol. 2016, 15, 434–441. [Google Scholar]
- Zheng, Y.; Chen, H.; Lai, W.; Xu, Q.; Liu, C.; Wu, L.; Maibach, H.I. Cathepsin D repairing role in photodamaged skin barrier. Skin Pharmacol. Physiol. 2015, 28, 97–102. [Google Scholar] [CrossRef]
- Calabro, G.; De Vita, V.; Patalano, A.; Mazzella, C.; Lo Conte, V.; Antropoli, C. Confirmed efficacy of topical nifedipine in the treatment of facial wrinkles. J. Dermatol. Treat. 2014, 25, 319–325. [Google Scholar] [CrossRef]
- Campos, P.M.B.G.M.; Gonçalves, G.M.S.; Gaspar, L.R. In vitro antioxidant activity and in vivo efficacy of topical formulations containing vitamin C and its derivatives studied by non-invasive methods. Skin Res. Technol. 2008, 14, 376–380. [Google Scholar] [CrossRef]
- Baran, R. Textbook of Cosmetic Dermatology; CRC Press: Boca Raton, FL, USA, 2005. [Google Scholar]
- Escoffier, C.; de Rigal, J.; Rochefort, A.; Vasselet, R.; Lévêque, J.L.; Agache, P.G. Age-related mechanical properties of human skin: An in vivo study. J. Investig. Dermatol. 1989, 93, 353–357. [Google Scholar] [CrossRef]
- Both, W.; Busch, P. Torsion measurement as a means of assessing skin characteristics. In Proceedings of the Forum Cosmeticum Basel, Basel, Switzerland, 19–20 February 1998; pp. 238–250. [Google Scholar]
- Laden, K.; Morrow, R. Torsional measurements on skin. J. Soc. Cosmet. Chem. 1970, 21, 417. [Google Scholar]
- Leveque, J.L.; De Rigal, J. In vivo measurements of the stratum corneum elasticity. Bioeng. Skin 1985, 1, 13–23. [Google Scholar]
- Wiechers, J.W. A supplier’s contribution to performance testing of personal care ingredients. Sofw. Seifen Ole Fette Wachse 1997, 123, 981–990. [Google Scholar]
- Berardesca, E.; de Rigal, J.; Leveque, J.L.; Maibach, H.I. In vivo biophysical characterization of skin physiological differences in races. Dermatologica 1991, 182, 89–93. [Google Scholar] [CrossRef]
- Rawlings, A.V. Ethnic skin types: Are there differences in skin structure and function? Int. J. Cosmet. Sci. 2006, 28, 79–93. [Google Scholar] [CrossRef]
- Tur, E. Physiology of the skin–differences between women and men. Clin. Dermatol. 1997, 15, 5–16. [Google Scholar] [CrossRef]
- Batisse, D.; Bazin, R.; Baldeweck, T.; Querleux, B.; Leveque, J.L. Influence of age on the wrinkling capacities of skin. Skin Res. Technol. 2002, 148–154. [Google Scholar] [CrossRef]
- Salter, D.; McArthur, H.; Crosse, J.; Dickens, A. Skin mechanics measured in vivo using torsion: A new and accurate model more sensitive to age, sex and moisturizing treatment. Int. J. Cosmet. Sci. 1993, 15, 200–218. [Google Scholar] [CrossRef]
- Sanders, R. Torsional elasticity of human skin in vivo. Pflügers Archiv Eur. J. Physiol. 1973, 342, 255–260. [Google Scholar] [CrossRef]
- Leveque, J.L.; Corcuff, P.; de Rigal, J.; Agache, P. In Vivo Studies of the Evolution of Physical Properties of the Human Skin with Age. Int. J. Dermatol. 1984, 23, 322–329. [Google Scholar] [CrossRef] [PubMed]
- Hara, Y.; Masuda, Y.; Hirao, T.; Yoshikawa, N. The relationship between the Young’s modulus of the stratum corneum and age: A pilot study. Skin Res. Technol. 2013, 19, 339–345. [Google Scholar] [CrossRef]
- Baumann, L. Cosmetic Dermatology: Principles and Practice; McGraw-Hill Prof Med/Tech: New York, NY, USA, 2009. [Google Scholar]
- Nedelec, B.; Couture, M.A.; Calva, V.; Poulin, C.; Chouinard, A.; Shashoua, D.; Gauthier, N.; Correa, J.A.; de Oliveira, A.; Mazer, B.; et al. Randomized controlled trial of the immediate and long-term effect of massage on adult postburn scar. Burns J. Int. Soc. Burn Inj. 2019, 45, 128–139. [Google Scholar] [CrossRef] [PubMed]
- Moortgat, P.; Meirte, J.; Maertens, K.; Lafaire, C.; De Cuyper, L.; Anthonissen, M. Can a cohesive silicone bandage outperform an adhesive silicone gel sheet in the treatment of scars? A randomised comparative trial. Plast. Reconstr. Surg. 2018. [Google Scholar] [CrossRef]
- Cortes, H.; Magana, J.J.; Reyes-Hernandez, O.D.; Zacaula-Juarez, N.; Gonzalez-Torres, M.; Diaz-Beltrán, W.; León-Trejo, M.C.; Cariño-Calvo, L.; Leyva-Gómez, G.; González-Del Carmen, M. Non-invasive analysis of skin mechanical properties in patients with lamellar ichthyosis. Skin Res. Technol. 2019. [Google Scholar] [CrossRef] [PubMed]
- Mazzarello, V.; Ferrari, M.; Ena, P. Werner syndrome: Quantitative assessment of skin ageing. Clin. Cosmet. Investig. Dermatol. 2018, 11, 397–402. [Google Scholar] [CrossRef]
- Busche, M.N.; Thraen, A.C.J.; Gohritz, A.; Rennekampff, H.O.; Vogt, P.M. Burn Scar Evaluation Using the Cutometer® MPA 580 in Comparison to “Patient and Observer Scar Assessment Scale” and “Vancouver Scar Scale”. J. Burn Care Res. 2018, 39, 516–526. [Google Scholar] [CrossRef]
- Gardien, K.L.M.; Marck, R.E.; Bloemen, M.C.T.; Waaijman, T.; Gibbs, S.; Ulrich, M.M.W.; Middelkoop, E.; Dutch Outback Study Group. Outcome of Burns Treated With Autologous Cultured Proliferating Epidermal Cells: A Prospective Randomized Multicenter Intrapatient Comparative Trial. Cell Transplant. 2016, 25, 437–448. [Google Scholar] [CrossRef]
- Hansen, B.; Jemec, G.B.E. The mechanical properties of skin in osteogenesis imperfecta. Arch. Dermatol. 2002, 138, 909–911. [Google Scholar] [CrossRef]
- Nedelec, B.; Correa, J.A.; de Oliveira, A.; Lasalle, L.; Perrault, I. Longitudinal burn scar quantification. Burns J. Int. Soc. Burn Inj. 2014. [Google Scholar] [CrossRef]
- Yoon, H.S.; Baik, S.H.; Oh, C.H. Quantitative measurement of desquamation and skin elasticity in diabetic patients. Skin Res. Technol. 2002, 8, 250–254. [Google Scholar] [CrossRef] [PubMed]
- Nam, G.W.; Baek, J.H.; Koh, J.S.; Hwang, J.K. The seasonal variation in skin hydration, sebum, scaliness, brightness and elasticity in Korean females. Skin Res. Technol. 2014. [Google Scholar] [CrossRef]
- Bae, S.H.; Park, J.J.; Song, E.J.; Lee, J.A.; Byun, K.S.; Kim, N.S.; Moon, T.K. The comparison of the melanin content and UV exposure affecting ageing process: Seven countries in Asia. J. Cosmet. Dermatol. 2016, 15, 335–342. [Google Scholar] [CrossRef] [PubMed]
- Coumare, R.; Bouten, L.; Barbier, F. Influence of the menstrual cycle on breast skin elasticity. Comput. Methods Biomech. Biomed. Eng. 2015, 18 (Suppl. 1), 1912–1913. [Google Scholar] [CrossRef] [PubMed]
- Song, E.J.; Lee, J.A.; Park, J.J.; Kim, H.J.; Kim, N.S.; Byun, K.S.; Choi, G.S.; Moon, T.K. A study on seasonal variation of skin parameters in Korean males. Int. J. Cosmet. Sci. 2015, 37, 92–97. [Google Scholar] [CrossRef] [PubMed]
- Jemec, G.B.; Gniadecka, M.; Jemec, B. Measurement of skin mechanics: A study of inter- and intra-individual variation using the Dermaflex A. Skin Res. Technol. 1996, 2, 164–166. [Google Scholar] [CrossRef] [PubMed]
- Jacobs, S.W.; Culbertson, E.J. Effects of Topical Mandelic Acid Treatment on Facial Skin Viscoelasticity. Facial Plast. Surg. 2018, 34, 651–656. [Google Scholar] [CrossRef]
- Lapatina, N.G.; Pavlenko, T. Diluted Calcium Hydroxylapatite for Skin Tightening of the Upper Arms and Abdomen. J. Drugs Dermatol. 2017, 16, 900–906. [Google Scholar]
- Nisbet, S.; Mahalingam, H.; Gfeller, C.F.; Biggs, E.; Lucas, S.; Thompson, M.; Cargill, M.R.; Moore, D.; Bielfeldt, S. Cosmetic benefit of a biomimetic lamellar cream formulation on barrier function or the appearance of fine lines and wrinkles in randomised proof-of-concept clinical studies. Int. J. Cosmet. Sci. 2018. [Google Scholar] [CrossRef]
- Yimam, M.; Lee, Y.C.; Jiao, P.; Hong, M.; Brownell, L.; Jia, Q. A Randomized, Active Comparator-controlled Clinical Trial of a Topical Botanical Cream for Skin Hydration, Elasticity, Firmness, and Cellulite. J. Clin. Aesthet. Dermatol. 2018, 11, 51–57. [Google Scholar]
- Kanlayavattanakul, M.; Lourith, N.; Chaikul, P. Jasmine rice panicle: A safe and efficient natural ingredient for skin ageing treatments. J. Ethnopharmacol. 2016, 193, 607–616. [Google Scholar] [CrossRef]
- Rodrigues, F.; Matias, R.; Ferreira, M.; Amaral, M.H.; Oliveira, M.B.P. In vitro and in vivo comparative study of cosmetic ingredients Coffee silverskin and hyaluronic acid. Exp. Dermatol. 2016, 25, 572–574. [Google Scholar] [CrossRef] [PubMed]
- Bindra, R.M.; Imhof, R.E.; Mochan, A.; Eccleston, G.M. Opto-thermal technique for in-vivo stratum corneum hydration measurement. Le J. Phys. IV 1994, 4, C7-465–C7-468. [Google Scholar] [CrossRef]
- Imhof, R.E.; Birch, D.J.S.; Thornley, F.R.; Gilchrist, J.R.; Strivens, T.A. Optothermal transient emission radiometry. J. Phys. E Sci. Instrum. 1984, 17, 521–525. [Google Scholar] [CrossRef]
- Imhof, R.; Whitters, C.; Birch, D. Opto-thermal in-vivo monitoring of structural breakdown of an emulsion sunscreen on skin. Clin. Mater. 1990, 5, 271–278. [Google Scholar] [CrossRef]
- Xiao, P. Photothermal Radiometry for Skin Research. Cosmetics 2016, 3, 10. [Google Scholar] [CrossRef]
- Guo, X.; Imhof, R.E.; De Rigal, J. Spectroscopic Study of Water-Keratin Interactions in Stratum Corneum. Anal. Sci. 2001, 17, s342–s345. [Google Scholar]
- Xiao, P.; Imhof, R.E. Optothermal skin-water concentration gradient measurement. Proc. SPIE 1996, 2681, 31–41. [Google Scholar] [CrossRef]
- Xiao, P.; Packham, H.; Zheng, X.; Singh, H.; Elliott, C.; Berg, E.P.; Imhof, R.E. Opto-thermal radiometry and condenser-chamber method for stratum corneum water concentration measurements. Appl. Phys. B 2007, 86, 715–719. [Google Scholar] [CrossRef]
- Xiao, P.; Wong, W.; Cottenden, A.M.; Imhof, R.E. In vivo stratum corneum over-hydration and water diffusion coefficient measurements using opto-thermal radiometry and TEWL Instruments. Int. J. Cosmet. Sci. 2012, 34, 328–331. [Google Scholar] [CrossRef]
- Milner, T.E.; Smithies, D.J.; Goodman, D.M.; Lau, A.; Nelson, J.S. Depth determination of chromophores in human skin by pulsed photothermal radiometry. Appl. Opt. 1996, 35, 3379–3385. [Google Scholar] [CrossRef]
- Choi, B.; Majaron, B.; Nelson, J.S. Computational model to evaluate port wine stain depth profiling using pulsed photothermal radiometry. J. Biomed. Opt. 2004, 9, 299–307. [Google Scholar] [CrossRef]
- Jacques, S.L.; Nelson, J.S.; Wright, W.H.; Milner, T.E. Pulsed photothermal radiometry of port-wine-stain lesions. Appl. Opt. 1993, 32, 2439–2446. [Google Scholar] [CrossRef]
- Xiao, P.; Ciortea, L.I.; Singh, H.; Berg, E.P.; Imhof, R.E. Opto-thermal radiometry for in-vivo nail measurements. J. Phys. Conf. Ser. 2010, 214, 012008. [Google Scholar] [CrossRef]
- Xiao, P.; Ou, X.; Ciortea, L.I.; Berg, E.P.; Imhof, R.E. In Vivo Skin Solvent Penetration Measurements Using Opto-thermal Radiometry and Fingerprint Sensor. Int. J. Thermophys. 2012, 33, 1787–1794. [Google Scholar] [CrossRef]
- Xiao, P.; Zheng, X.; Imhof, R.E.; Hirata, K.; McAuley, W.J.; Mateus, R.; Hadgraft, J.; Lane, M.E. Opto-Thermal Transient Emission Radiometry (OTTER) to image diffusion in nails in vivo. Int. J. Pharm. 2011, 406, 111–113. [Google Scholar] [CrossRef] [PubMed]
- Vidovic, L.; Milanic, M.; Majaron, B. Objective characterization of bruise evolution using photothermal depth profiling and Monte Carlo modeling. J. Biomed. Opt. 2015, 20, 017001. [Google Scholar] [CrossRef] [PubMed]
- Milanic, M.; Majaron, B. Spectral filtering in pulsed photothermal temperature profiling of collagen tissue phantoms. J. Biomed. Opt. 2009, 14, 064024. [Google Scholar] [CrossRef]
- Majaron, B.; Milanic, M. Effective infrared absorption coefficient for photothermal radiometric measurements in biological tissues. Phys. Med. Biol. 2008, 53, 255–268. [Google Scholar] [CrossRef] [PubMed]
- Leveque, J.L.; Escoubez, M.; Rasseneur, L. Water-keratin interaction in human stratum corneum. Bioeng. Skin 1887, 227–242. [Google Scholar]
- Bindra, R.M.; Imhof, R.E.; Eccleston, G.M. In-vivo opto-thermal measurement of epidermal thickness. Le J. Phys. IV 1994, 04, C7-445–C7-448. [Google Scholar] [CrossRef]
- Cowen, J.A.; Imhof, R.E.; Xiao, P. Opto-thermal Measurement of Stratum Corneum Renewal Time. Anal. Sci. 2001, 17, s353–s356. [Google Scholar]
- Notingher, I.; Imhof, R.E.; Xiao, P.; Pascut, F.C. Near-surface depth-resolved midinfrared emission spectroscopy. Rev. Sci. Instrum. 2003, 74, 346–348. [Google Scholar] [CrossRef]
- Notingher, I.; Imhof, R.E.; Xiao, P.; Pascut, F.C. Spectral depth profiling of arbitrary surfaces by thermal emission decay-Fourier transform infrared spectroscopy. Appl. Spectrosc. 2003, 57, 1494–1501. [Google Scholar] [CrossRef]
- Potts, R.O.; Guzek, D.B.; Harris, R.R.; McKie, J.E. A noninvasive, in vivo technique to quantitatively measure water concentration of the stratum corneum using attenuated total-reflectance infrared spectroscopy. Arch. Dermatol. Res. 1985, 277, 489–495. [Google Scholar] [CrossRef] [PubMed]
- Bommannan, D.; Potts, R.; Guy, R. Examination of stratum corneum barrier function in vivo by infrared spectroscopy. J. Investig. Dermatol. 1990, 95, 403–408. [Google Scholar] [CrossRef]
- Boncheva, M.; Damien, F.; Normand, V. Molecular organization of the lipid matrix in intact Stratum corneum using ATR-FTIR spectroscopy. Biochim. Biophys. Acta 2008, 1778, 1344–1355. [Google Scholar] [CrossRef]
- Berthaud, F.; Boncheva, M. Correlation between the properties of the lipid matrix and the degrees of integrity and cohesion in healthy human Stratum corneum. Exp. Dermatol. 2011, 20, 255–262. [Google Scholar] [CrossRef]
- Watkinson, A.; Lee, R.S.; Moore, A.E.; Pudney, P.D.A.; Paterson, S.E.; Rawlings, A.V. Reduced barrier efficiency in axillary stratum corneum. Int. J. Cosmet. Sci. 2002, 24, 151–161. [Google Scholar] [CrossRef]
- Wang, C.; Zhu, J.; Zhang, D.; Yang, Y.; Zheng, L.; Qu, Y.; Yang, X.; Cui, X. Ionic liquid—microemulsions assisting in the transdermal delivery of Dencichine: Preparation, in-vitro and in-vivo evaluations, and investigation of the permeation mechanism. Int. J. Pharm. 2018, 535, 120–131. [Google Scholar] [CrossRef]
- Binder, L.; Kulovits, E.M.; Petz, R.; Ruthofer, J.; Baurecht, D.; Klang, V.; Valenta, C. Penetration monitoring of drugs and additives by ATR-FTIR spectroscopy/tape stripping and confocal Raman spectroscopy—A comparative study. Eur. J. Pharm. Biopharm. 2018, 130, 214–223. [Google Scholar] [CrossRef] [PubMed]
- Cilurzo, F.; Vistoli, G.; Selmin, F.; Gennari, C.G.M.; Musazzi, U.M.; Franzé, S.; Lo Monte, M.; Minghetti, P. An insight into the skin penetration enhancement mechanism of N-methylpyrrolidone. Mol. Pharm. 2014, 11, 1014–1021. [Google Scholar] [CrossRef] [PubMed]
- Csizmazia, E.; Eros, G.; Berkesi, O.; Berko, S.; Szabo-Revesz, P.; Csanyi, E. Ibuprofen penetration enhance by sucrose ester examined by ATR-FTIR in vivo. Pharm. Dev. Technol. 2012, 17, 125–128. [Google Scholar] [CrossRef]
- Obata, Y.; Utsumi, S.; Watanabe, H.; Suda, M.; Tokudome, Y.; Otsuka, M.; Takayama, K. Infrared spectroscopic study of lipid interaction in stratum corneum treated with transdermal absorption enhancers. Int. J. Pharm. 2010, 389, 18–23. [Google Scholar] [CrossRef] [PubMed]
- Goh, C.F.; Craig, D.Q.M.; Hadgraft, J.; Lane, M.E. The application of ATR-FTIR spectroscopy and multivariate data analysis to study drug crystallisation in the stratum corneum. Eur. J. Pharm. Biopharm. 2017, 111, 16–25. [Google Scholar] [CrossRef]
- Wolf, M.; Halper, M.; Pribyl, R.; Baurecht, D.; Valenta, C. Distribution of phospholipid based formulations in the skin investigated by combined ATR-FTIR and tape stripping experiments. Int. J. Pharm. 2017, 519, 198–205. [Google Scholar] [CrossRef]
- Vyumvuhore, R.; Tfayli, A.; Manfait, M.; Baillet-Guffroy, A. Vibrational spectroscopy coupled to classical least square analysis, a new approach for determination of skin moisturizing agents’ mechanisms. Skin Res. Technol. 2014, 20, 282–292. [Google Scholar] [CrossRef]
- Caussin, J.; Rozema, E.; Gooris, G.S.; Wiechers, J.W.; Pavel, S.; Bouwstra, J.A. Hydrophilic and lipophilic moisturisers have similar penetration profiles but different effects on SC water distribution in vivo. Exp. Dermatol. 2009, 18, 954–961. [Google Scholar] [CrossRef] [PubMed]
- Kilpatrick-Liverman, L.; Kazmi, P.; Wolff, E.; Polefka, T. The use of near-infrared spectroscopy in skin care applications. Skin Res. Technol. 2006, 12, 162–169. [Google Scholar] [CrossRef] [PubMed]
- Hansen, J.R.; Yellin, W. NMR and Intrared Spectroscopic Studies of Stratum Corneum Hydration. In Water Structure at the Water-Polymer Interface; Jellinek, H.H.G., Ed.; Springer: Boston, MA, USA, 1972; pp. 19–28. [Google Scholar]
- Gilard, V.; Martino, R.; Malet-Martino, M.; Riviere, M.; Gournay, A.; Navarro, R. Measurement of total water and bound water contents in human stratum corneum by in vitro proton nuclear magnetic resonance spectroscopy. Int. J. Cosmet. Sci. 1998, 20, 117–125. [Google Scholar] [CrossRef]
- Yamamura, T.; Tezuka, T. The Water-Holding Capacity of the Stratum Corneum Measured by 1 H-NMR. J. Investig. Dermatol. 1989, 93, 160–164. [Google Scholar] [CrossRef] [PubMed]
- Jokura, Y.; Ishikawa, S.; Tokuda, H.; Imokawa, G. Molecular Analysis of Elastic Properties of the Stratum Corneum by Solid-State 13C-Nuclear Magnetic Resonance Spectroscopy. J. Investig. Dermatol. 1995, 104, 806–812. [Google Scholar] [CrossRef]
- Laule, C.; Tahir, S.; Chia, C.L.L.; Vavasour, I.M.; Kitson, N.; MacKay, A.L. A proton NMR study on the hydration of normal versus psoriatic stratum corneum: Linking distinguishable reservoirs to anatomical structures. NMR Biomed. 2010, 23, 1181–1190. [Google Scholar] [CrossRef] [PubMed]
- Silva, C.L.; Topgaard, D.; Kocherbitov, V.; Sousa, J.J.S.; Pais, A.A.C.C.; Sparr, E. Stratum corneum hydration: Phase transformations and mobility in stratum corneum, extracted lipids and isolated corneocytes. Biochim. Biophys. Acta 2007, 1768, 2647–2659. [Google Scholar] [CrossRef] [PubMed]
- Bjorklund, S.; Nowacka, A.; Bouwstra, J.A.; Sparr, E.; Topgaard, D. Characterization of Stratum Corneum Molecular Dynamics by Natural-Abundance 13C Solid-State NMR. PLoS ONE 2013, 8, e61889. [Google Scholar] [CrossRef] [PubMed]
- Muta, K.; Inomata, S.; Fukuhara, T.; Nomura, J.; Nishiyama, T.; Tagawa, Y.I.; Amano, S. Inhibitory effect of the extract of rhizome of Curcuma longa L in gelatinase activity and its effect on human skin. J. Biosci. Bioeng. 2018, 125, 353–358. [Google Scholar] [CrossRef]
- Tessema, E.N.; Gebre-Mariam, T.; Lange, S.; Dobner, B.; Neubert, R.H.H. Potential application of oat-derived ceramides in improving skin barrier function: Part 1. Isolation and structural characterization. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2017, 1065–1066, 87–95. [Google Scholar] [CrossRef]
- Hoppel, M.; Reznicek, G.; Kahlig, H.; Kotisch, H.; Resch, G.P.; Valenta, C. Topical delivery of acetyl hexapeptide-8 from different emulsions: Influence of emulsion composition and internal structure. Eur. J. Pharm. Sci. 2015, 68, 27–35. [Google Scholar] [CrossRef]
- Pham, Q.D.; Topgaard, D.; Sparr, E. Tracking solvents in the skin through atomically resolved measurements of molecular mobility in intact stratum corneum. Proc. Natl. Acad. Sci. USA 2017, 114, E112–E121. [Google Scholar] [CrossRef]
- Voegeli, D. The effect of washing and drying practices on skin barrier function. J. Wound Ostomy Cont. Nurs. 2008, 35, 84–90. [Google Scholar] [CrossRef]
- Bittoun, J.; Saint-Jalmes, H.; Querleux, B.; Darrasse, L.; Jolivet, O.; Idy-Peretti, I.; Wartski, M.; Richard, S.; Leveque, J. In vivo high-resolution MR imageing of the skin in a whole-body system at 1.5 T. Radiology 1990, 176, 457–460. [Google Scholar] [CrossRef]
- Richard, S.; Querleux, B.; Bittoun, J.; Idy-Peretti, I.; Jolivet, O.; Cermakova, E.; Lévêque, J. In vivo proton relaxation times analysis of the skin layers by magnetic resonance imageing. J. Investig. Dermatol. 1991, 97, 120–125. [Google Scholar] [CrossRef] [PubMed]
- Franconi, F.; Akoka, S.; Guesnet, J.; Baret, J.; Dersigny, D.; Breda, B.; Muller, C.; Beau, P. Measurement of epidermal moisture content by magnetic resonance imageing: Assessment of a hydration cream. Br. J. Dermatol. 1995, 132, 913–917. [Google Scholar] [CrossRef] [PubMed]
- Szayna, M.; Kuhn, W. In vivo and in vitro investigations of hydration effects of beauty care products by high-field MRI and NMR microscopy. J. Eur. Acad. Dermatol. Venereol. 1998, 11, 122–128. [Google Scholar] [CrossRef] [PubMed]
- Mesrar, J.; Ognard, J.; Garetier, M.; Chechin, D.; Misery, L.; Ben Salem, D. In vivo skin moisturizing measurement by high-resolution 3 Tesla magnetic resonance imageing. Skin Res. Technol. 2017, 23, 289–294. [Google Scholar] [CrossRef] [PubMed]
- Ablett, S.; Burdett, N.G.; Carpenter, T.A.; Hall, L.D.; Salter, D.C. Short echo time MRI enables visualisation of the natural state of human stratum corneum water in vivo. Magn. Reson. Imageing 1996, 14, 357–360. [Google Scholar] [CrossRef]
- Mirrashed, F.; Sharp, J.C. In vivo quantitative analysis of the effect of hydration (immersion and Vaseline treatment) in skin layers using high-resolution MRI and magnetisation transfer contrast. Skin Res. Technol. 2004, 10, 14–22. [Google Scholar] [CrossRef]
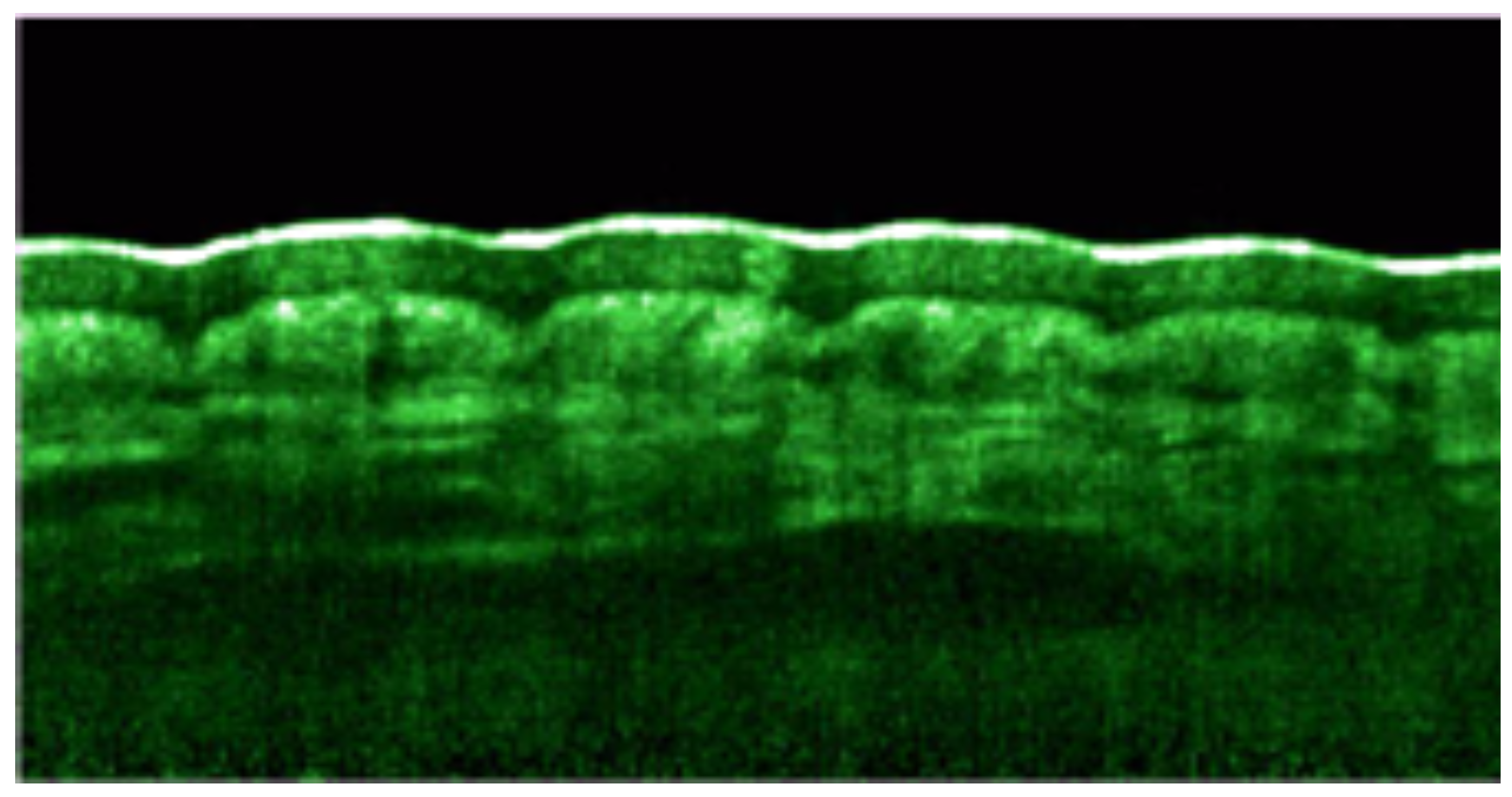
- Welzel, J.; Bruhns, M.; Wolff, H. Optical coherence tomography in contact dermatitis and psoriasis. Arch. Dermatol. Res. 2003, 295, 50–55. [Google Scholar] [CrossRef]
- Mogensen, M.; Morsy, H.; Thrane, L.; Jemec, G. Morphology and epidermal thickness of normal skin imaged by optical coherence tomography. Dermatology 2008, 217, 14–20. [Google Scholar] [CrossRef]
- Welzel, J.; Lankenau, E.; Birngruber, R.; Engelhardt, R. Optical coherence tomography of the human skin. J. Am. Acad. Dermatol. 1997, 37, 958–963. [Google Scholar] [CrossRef]
- Mogensen, M.; Thrane, L.; Joergensen, T.; Andersen, P.; Jemec, G. Optical Coherence Tomography for Imageing of Skin and Skin Diseases. Semin. Cutan. Med. Surg. 2009, 28, 196–202. [Google Scholar] [CrossRef] [PubMed]
- Tearney, G.; Brezinski, M.; Southern, J.; Bouma, B.; Hee, M.; Fujimoto, J. Determination of the refractive index of highly scattering human tissue by optical coherence tomography. Opt. Lett. 1995, 20, 2258–2260. [Google Scholar] [CrossRef]
- Wilhelm, K.P.; Elsner, P.; Berardesca, E. Bioengineering of the Skin: Skin Imageing and Analysis; CRC Press: Boca Raton, FL, USA, 2006. [Google Scholar]
- Knüttel, A.; Boehlau-Godau, M. Spatially confined and temporally resolved refractive index and scattering evaluation in human skin performed with optical coherence tomography. J. Biomed. Opt. 2000, 5, 83–93. [Google Scholar] [CrossRef]
- Abuzahra, F.; Baron, J. Optical coherence tomography of the skin: A diagnostic light look. Der Hautarzt; Zeitschrift für Dermatologie, Venerologie, und verwandte Gebiete 2006, 57, 646. [Google Scholar] [CrossRef] [PubMed]
- Xu, W.; Ranger-Moore, J.; Saboda, K.; Salasche, S.; Warneke, J.; Alberts, D. Investigating sun-damaged skin and actinic keratosis with optical coherence tomography: A pilot study. Technol. Cancer Res. Treat. 2003, 2, 525–535. [Google Scholar]
- Gladkova, N.; Petrova, G.; Nikulin, N.; Radenska-Lopovok, S.; Snopova, L.; Chumakov, Y.; Nasonova, V.; Gelikonov, V.; Gelikonov, G.; Kuranov, R.; et al. In vivo optical coherence tomography imageing of human skin: Norm and pathology. Skin Res. Technol. 2000, 6, 6–16. [Google Scholar] [CrossRef] [PubMed]
- Jensen, L.; Thrane, L.; Andersen, P.; Tycho, A.; Pedersen, F.; Andersson-Engels, S.; BendsU00D8e, N.; Svanberg, S.; Svanberg, K. Optical coherence tomography in clinical examination of non-pigmented skin malignancies. In Proceedings of the European Conference on Biomedical Optics, Munich, Germany, 22 June 2003. [Google Scholar]
- Olmedo, J.; Warschaw, K.; Schmitt, J.; Swanson, D. Optical coherence tomography for the characterization of basal cell carcinoma in vivo: A pilot study. J. Am. Acad. Dermatol. 2006, 55, 408–412. [Google Scholar] [CrossRef] [PubMed]
- Olmedo, J.; Warschaw, K.; Schmitt, J.; Swanson, D. Correlation of thickness of basal cell carcinoma by optical coherence tomography in vivo and routine histologic findings: A pilot study. Dermatol. Surg. 2007, 33, 421–426. [Google Scholar]
- Strasswimmer, J.; Pierce, M.; Park, B. Characterization of basal cell carcinoma by multifunctional optical coherence tomography. J. Investig. Dermatol. 2003, 121, 0156. [Google Scholar]
- Gambichler, T.; Orlikov, A.; Vasa, R.; Moussa, G.; Hoffmann, K.; Stucker, M.; Altmeyer, P.; Bechara, F. In vivo optical coherence tomography of basal cell carcinoma. J. Dermatol. Sci. 2007, 45, 167–173. [Google Scholar] [CrossRef]
- Ulrich, M.; Stockfleth, E.; Roewert-Huber, J.; Astner, S. Noninvasive diagnostic tools for nonmelanoma skin cancer. Br. J. Dermatol. 2007, 157, 56–58. [Google Scholar] [CrossRef]
- Sattler, E.; Kastle, R.; Welzel, J. Optical coherence tomography in dermatology. J. Biomed. Opt. 2013, 18, 061224. [Google Scholar] [CrossRef] [PubMed]
- Schuh, S.; Kaestle, R.; Sattler, E.; Welzel, J. Comparison of different optical coherence tomography devices for diagnosis of non-melanoma skin cancer. Skin Res. Technol. 2016, 22, 395–405. [Google Scholar] [CrossRef]
- Gambichler, T.; Moussa, G.; Sand, M.; Sand, D.; Orlikov, A.; Altmeyer, P.; Hoffmann, K. Correlation between clinical scoring of allergic patch test reactions and optical coherence tomography. J. Biomed. Opt. 2005, 10, 064030. [Google Scholar] [CrossRef] [PubMed]
- Ropke, M.A.; Alonso, C.; Jung, S.; Norsgaard, H.; Richter, C.; Darvin, M.E.; Litman, T.; Vogt, A.; Lademann, J.; Blume-Peytavi, U.; et al. Effects of glucocorticoids on stratum corneum lipids and function in human skin-A detailed lipidomic analysis. J. Dermatol. Sci. 2017, 88, 330–338. [Google Scholar] [CrossRef] [PubMed]
- Jung, S.; Lademann, J.; Darvin, M.E.; Richter, C.; Pedersen, C.B.; Richter, H.; Schanzer, S.; Kottner, J.; Blume-Peytavi, U.; Ropke, M.A. In vivo characterization of structural changes after topical application of glucocorticoids in healthy human skin. J. Biomed. Opt. 2017, 22, 76018. [Google Scholar] [CrossRef] [PubMed]
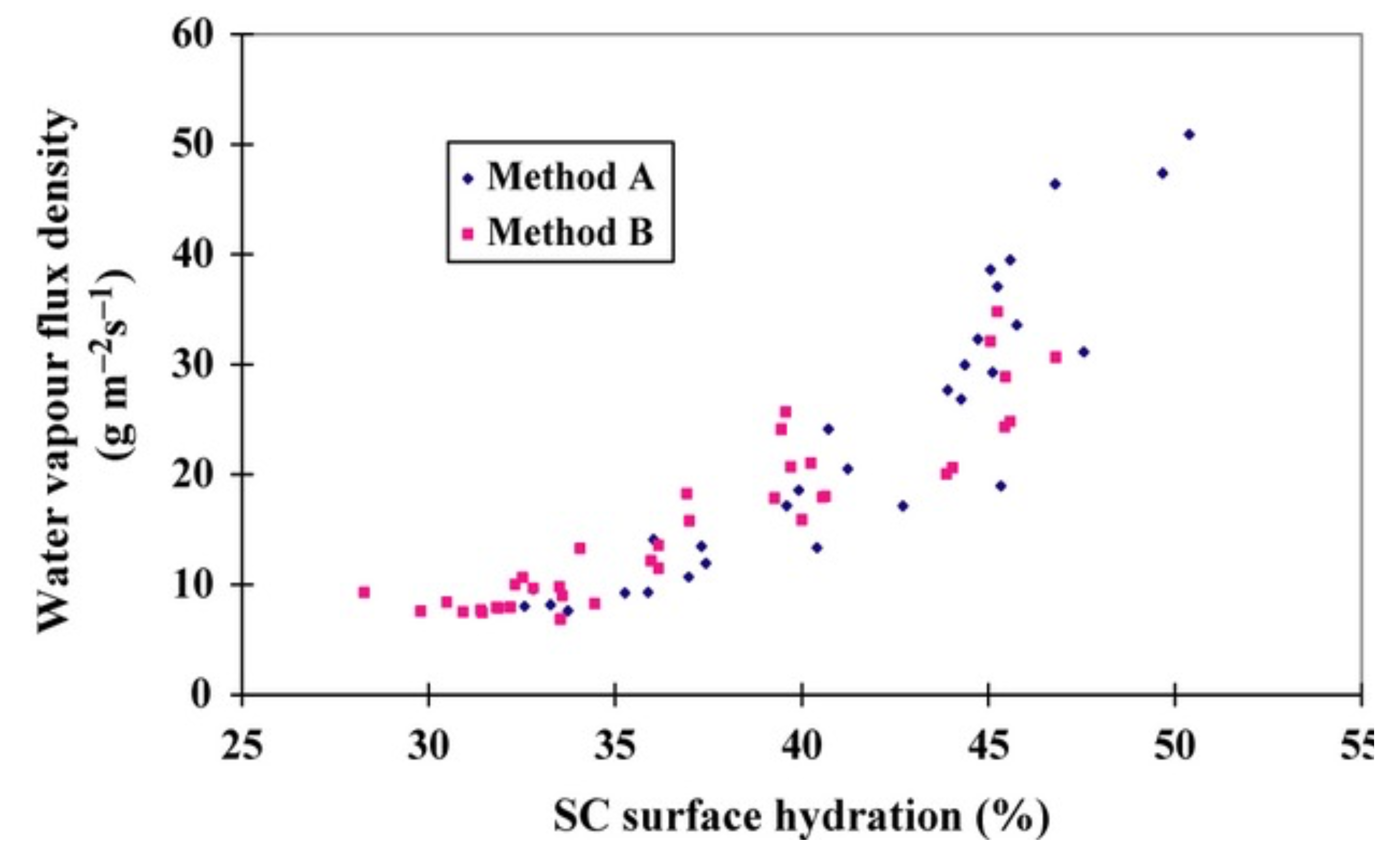
- Crowther, J.; Sieg, A.; Blenkiron, P.; Marcott, C.; Matts, P.; Kaczvinsky, J.; Rawlings, A. Measuring the effects of topical moisturisers on changes in stratum corneum thickness, water gradients and hydration in vivo. Br. J. Dermatol. 2008, 159, 567–577. [Google Scholar] [CrossRef]
- Trojahn, C.; Dobos, G.; Richter, C.; Blume-Peytavi, U.; Kottner, J. Measuring skin ageing using optical coherence tomography in vivo: A validation study. J. Biomed. Opt. 2015, 20, 045003. [Google Scholar] [CrossRef]
- Florence, P.; Cornillon, C.; Darrasse, M.F.; Flament, F.; Panhard, S.; Diridollou, S.; Loussouarn, G. Functional and structural age-related changes in the scalp skin of Caucasian women. Skin Res. Technol. 2013, 19, 384–393. [Google Scholar] [CrossRef]
- Mizuno, A.; Kitajima, H.; Kawauchi, K.; Muraishi, S.; Ozaki, Y. Near-infrared Fourier transform Raman spectroscopic study of human brain tissues and tumours. J. Raman Spectrosc. 1994, 25, 25–29. [Google Scholar] [CrossRef]
- Frank, C.; McCreery, R.; Redd, D. Raman spectroscopy of normal and diseased human breast tissues. Anal. Chem. 1995, 67, 777–783. [Google Scholar] [CrossRef]
- Alfano, R.; Liu, C.; Sha, W.; Zhu, H.; Akins, D.; Cleary, J.; Prudente, R.; Cellmer, E. Human breast tissues studied by IR Fourier transform Raman spectroscopy. Lasers Life Sci. 1991, 4, 23–28. [Google Scholar]
- Feld, M.; Manoharan, R.; Salenius, J.; Orenstein-Carndona, J.; Roemer, T.; Brennan, J., III; Dasari, R.; Wang, Y. Detection and characterization of human tissue lesions with near-infrared Raman spectroscopy. Proc. SPIE 1995, 2388, 99. [Google Scholar]
- Liu, C.; Das, B.; Glassman, W.; Tang, G.; Yoo, K.; Zhu, H.; Akins, D.; Lubicz, S.; Cleary, J.; Prudente, R.; et al. Raman, fluorescence, and time-resolved light scattering as optical diagnostic techniques to separate diseased and normal biomedical media. J. Photochem. Photobiol. B Biol. 1992, 16, 187–209. [Google Scholar] [CrossRef]
- Mahadevan-Jansen, A.; Mitchell, M.; Ramanujamf, N.; Malpica, A.; Thomsen, S.; Utzinger, U.; Richards-Kortumt, R. Near-Infrared Raman Spectroscopy for In Vitro Detection of Cervical Precancers. Photochem. Photobiol. 1998, 68, 123–132. [Google Scholar] [CrossRef] [PubMed]
- Gniadecka, M.; Wulf, H.; Nielsen, O.; Christensen, D.; Hercogova, J. Distinctive molecular abnormalities in benign and malignant skin lesions: Studies by Raman spectroscopy. Photochem. Photobiol. 1997, 66, 418–423. [Google Scholar] [CrossRef]
- Stone, N.; Stavroulaki, P.; Kendall, C.; Birchall, M.; Barr, H. Raman spectroscopy for early detection of laryngeal malignancy: Preliminary results. Laryngoscope 2000, 110, 1756–1763. [Google Scholar] [CrossRef]
- Duindam, H.; Vrensen, G.; Otto, C.; Puppels, G.; Greve, J. New approach to assess the cholesterol distribution in the eye lens: Confocal Raman microspectroscopy and filipin cytochemistry. J. Lipid Res. 1995, 36, 1139–1146. [Google Scholar]
- Siebinga, I.; Vrensen, G.; De Mul, F.; Greve, J. Age-related changes in local water and protein content of human eye lenses measured by Raman microspectroscopy. Exp. Eye Res. 1991, 53, 233–239. [Google Scholar] [CrossRef]
- Duindam, J.; Vrensen, G.; Otto, C.; Greve, J. Cholesterol, phospholipid, and protein changes in focal opacities in the human eye lens. Investig. Ophthalmol. Vis. Sci. 1998, 39, 94. [Google Scholar]
- Romer, T.; Brennan, J.; Fitzmaurice, M.; Feldstein, M.; Deinum, G.; Myles, J.; Kramer, J.; Lees, R.; Feld, M. Histopathology of human coronary atherosclerosis by quantifying its chemical composition with Raman spectroscopy. Circulation 1998, 97, 878–885. [Google Scholar] [CrossRef]
- Salenius, J.; Brennan, J.; Miller, A.; Wang, Y.; Aretz, T.; Sacks, B.; Dasari, R.; Feld, M. Biochemical composition of human peripheral arteries examined with nearinfrared Raman spectroscopy. J. Vasc. Surg. 1998, 27, 710–719. [Google Scholar] [CrossRef]
- Barry, B.; Edwards, H.; Williams, A. Fourier transform Raman and infrared vibrational study of human skin: Assignment of spectral bands. J. Raman Spectrosc. 1992, 23, 641–645. [Google Scholar] [CrossRef]
- Williams, A.; Edwards, H.; Barry, B. Raman spectra of human keratotic biopolymers: Skin, callus, hair and nail. J. Raman Spectrosc. 1994, 25, 95–98. [Google Scholar] [CrossRef]
- Anigbogu, A.; Williams, A.; Barry, B.; Edwards, H. Fourier transform Raman spectroscopy of interactions between the penetration enhancer dimethyl sulfoxide and human stratum corneum. Int. J. Pharm. 1995, 125, 265–282. [Google Scholar] [CrossRef]
- Lawson, E.; Anigbogu, A.; Williams, A.; Barry, B.; Edwards, H. Thermally induced molecular disorder in human stratum corneum lipids compared with a model phospholipid system; FT-Raman spectroscopy. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 1998, 54, 543–558. [Google Scholar] [CrossRef]
- Caspers, P.; Williams, A.; Carter, E.; Edwards, H.; Barry, B.; Bruining, H.; Puppels, G. Monitoring the penetration enhancer dimethyl sulfoxide in human stratum corneum in vivo by confocal Raman spectroscopy. Pharm. Res. 2002, 19, 1577–1580. [Google Scholar] [CrossRef] [PubMed]
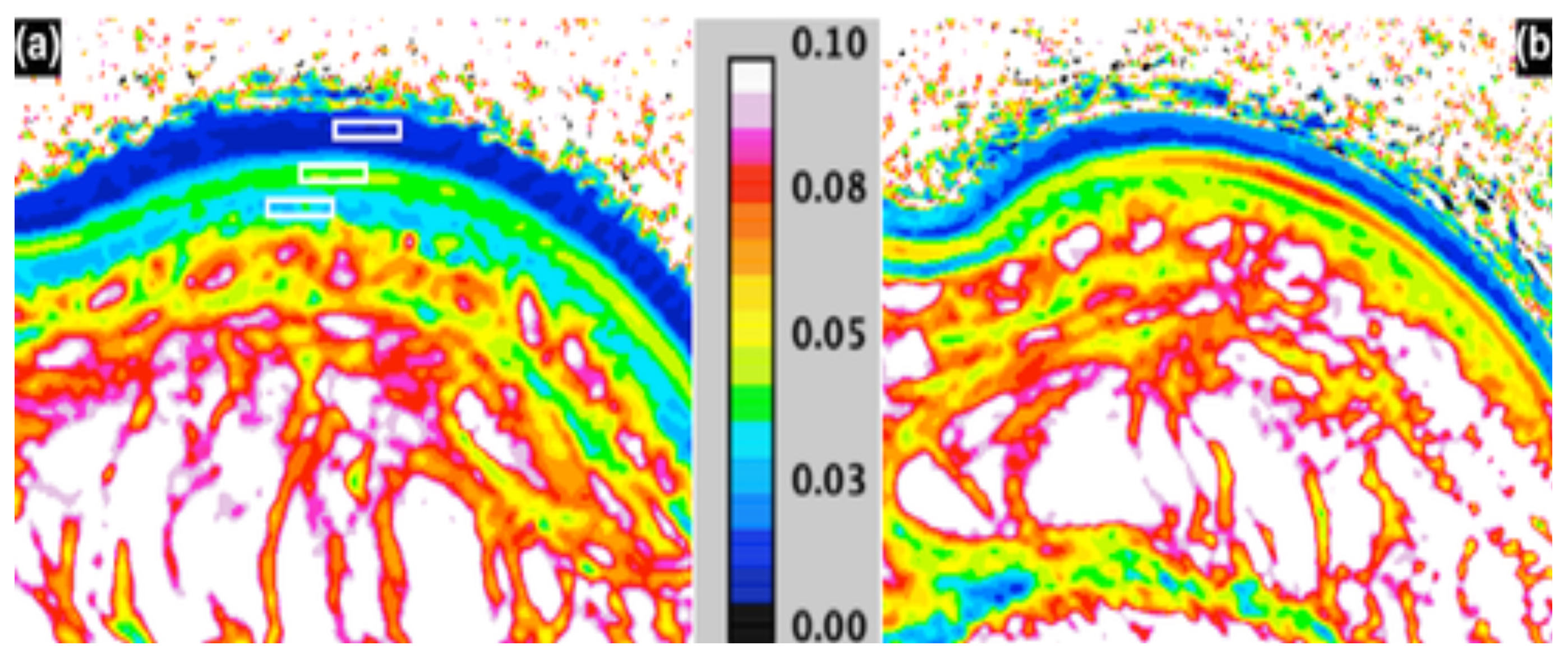
- Caspers, P.; Lucassen, G.; Carter, E.; Bruining, H.; Puppels, G. In vivo confocal Raman microspectroscopy of the skin: Noninvasive determination of molecular concentration profiles. J. Investig. Dermatol. 2001, 116, 434–442. [Google Scholar] [CrossRef] [PubMed]
- Caspers, P.; Lucassen, G.; Wolthuis, R.; Bruining, H.; Puppels, G. In vitro and in vivo Raman spectroscopy of human skin. Biospectroscopy 1998, 4, S31–S39. [Google Scholar] [CrossRef]
- Caspers, P.; Lucassen, G.; Puppels, G. Combined in vivo confocal Raman spectroscopy and confocal microscopy of human skin. Biophys. J. 2003, 85, 572–580. [Google Scholar] [CrossRef]
- Schallreuter, K.; Wood, J.; Farwell, D.; Moore, J.; Edwards, H. Oxybenzone oxidation following solar irradiation of skin: Photoprotection versus antioxidant inactivation. J. Investig. Dermatol. 1996, 106, 583–586. [Google Scholar] [CrossRef]
- Gniadecka, M.; Nielsen, O.; Wessel, S.; Heidenheim, M.; Christensen, D.; Wulf, H. Water and protein structure in photoaged and chronically aged skin. J. Investig. Dermatol. 1998, 111, 1129–1132. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.; Won, K.; Kim, E.J.; Hwang, J.S.; Lee, H.K. Comparison of stratum corneum thickness between two proposed methods of calculation using Raman spectroscopic depth profiling of skin water content. Skin Res. Technol. 2018, 24, 504–508. [Google Scholar] [CrossRef]
- Janssens, M.; van Smeden, J.; Puppels, G.J.; Lavrijsen, A.P.M.; Caspers, P.J.; Bouwstra, J.A. Lipid to protein ratio plays an important role in the skin barrier function in patients with atopic eczema. Br. J. Dermatol. 2014, 170, 1248–1255. [Google Scholar] [CrossRef] [PubMed]
- Tfayli, A.; Jamal, D.; Vyumvuhore, R.; Manfait, M.; Baillet-Guffroy, A. Hydration effects on the barrier function of stratum corneum lipids: Raman analysis of ceramides 2, III and 5. Analyst 2013, 138, 6582–6588. [Google Scholar] [CrossRef] [PubMed]
- Ogawa-Fuse, C.; Morisaki, N.; Shima, K.; Hotta, M.; Sugata, K.; Ichihashi, T.; Oguri, M.; Yoshida, O.; Fujimura, T. Impact of water exposure on skin barrier permeability and ultrastructure. Contact Dermat. 2018. [Google Scholar] [CrossRef] [PubMed]
- Biniek, K.; Tfayli, A.; Vyumvuhore, R.; Quatela, A.; Galliano, M.F.; Delalleau, A.; Baillet-Guffroy, A.; Dauskardt, R.H.; Duplan, H. Measurement of the biomechanical function and structure of ex vivo drying skin using raman spectral analysis and its modulation with emollient mixtures. Exp. Dermatol. 2018, 27, 901–908. [Google Scholar] [CrossRef]
- Essendoubi, M.; Gobinet, C.; Reynaud, R.; Angiboust, J.F.; Manfait, M.; Piot, O. Human skin penetration of hyaluronic acid of different molecular weights as probed by Raman spectroscopy. Skin Res. Technol. 2016, 22, 55–62. [Google Scholar] [CrossRef]
- Stettler, H.; Kurka, P.; Wagner, C.; Sznurkowska, K.; Czernicka, O.; Böhling, A.; Bielfeldt, S.; Wilhelm, K.P.; Lenz, H. A new topical panthenol-containing emollient: Skin-moisturizing effect following single and prolonged usage in healthy adults, and tolerability in healthy infants. J. Dermatol. Treat. 2017, 28, 251–257. [Google Scholar] [CrossRef]
- Zeranska, J.; Pasikowska, M.; Szczepanik, B.; Mlosek, K.; Malinowska, S.; Debowska, R.M.; Eris, I. A study of the activity and effectiveness of recombinant fibroblast growth factor (Q40P/S47I/H93G rFGF-1) in anti-ageing treatment. Postepy Dermatol. I Alergol. 2016, 33, 28–36. [Google Scholar] [CrossRef]
- Chrit, L.; Bastien, P.; Sockalingum, G.; Batisse, D.; Leroy, F.; Manfait, M.; Hadjur, C. An in vivo randomized study of human skin moisturization by a new confocal Raman fiber-optic microprobe: Assessment of a glycerol-based hydration cream. Skin Pharmacol. Physiol. 2006, 19, 207–215. [Google Scholar] [CrossRef] [PubMed]
- Egawa, M.; Hirao, T.; Takahashi, M. In vivo estimation of stratum corneum thickness from water concentration profiles obtained with Raman spectroscopy. Acta Dermato-Venereol. 2007, 87, 4–8. [Google Scholar] [CrossRef]
- Sieg, A.; Crowther, J.; Blenkiron, P.; Marcott, C.; Matts, P. Confocal Raman microspectroscopy: Measuring the effects of topical moisturisers on stratum corneum water gradient in vivo. Proc. SPIE 2006, 6093, 60930N. [Google Scholar]
- Choe, C.; Schleusener, J.; Lademann, J.; Darvin, M.E. In vivo confocal Raman microscopic determination of depth profiles of the stratum corneum lipid organization influenced by application of various oils. J. Dermatol. Sci. 2017, 87, 183–191. [Google Scholar] [CrossRef] [PubMed]
- Choe, C.; Lademann, J.; Darvin, M.E. Depth profiles of hydrogen bound water molecule types and their relation to lipid and protein interaction in the human stratum corneum in vivo. Analyst 2016, 141, 6329–6337. [Google Scholar] [CrossRef] [PubMed]
- Eklouh-Molinier, C.; Gaydou, V.; Froigneux, E.; Barlier, P.; Couturaud, V.; Manfait, M.; Piot, O. In vivo confocal Raman microspectroscopy of the human skin: Highlighting of spectral markers associated to ageing via a research of correlation between Raman and biometric mechanical measurements. Anal. Bioanal. Chem. 2015, 407, 8363–8372. [Google Scholar] [CrossRef]
- Boireau-Adamezyk, E.; Baillet-Guffroy, A.; Stamatas, G.N. Age-dependent changes in stratum corneum barrier function. Skin Res. Technol. 2014, 20, 409–415. [Google Scholar] [CrossRef]
- Kikuchi, S.; Aosaki, T.; Bito, K.; Naito, S.; Katayama, Y. In vivo evaluation of lateral lipid chain packing in human stratum corneum. Skin Res. Technol. 2015, 21, 76–83. [Google Scholar] [CrossRef]
- Quatela, A.; Miloudi, L.; Tfayli, A.; Baillet-Guffroy, A. In vivo Raman Microspectroscopy: Intra- and Intersubject Variability of Stratum Corneum Spectral Markers. Skin Pharmacol. Physiol. 2016, 29, 102–109. [Google Scholar] [CrossRef]
- Richters, R.J.H.; Falcone, D.; Uzunbajakava, N.E.; Varghese, B.; Caspers, P.J.; Puppels, G.J.; van Erp, P.E.J.; van de Kerkhof, P.C.M. Sensitive Skin: Assessment of the Skin Barrier Using Confocal Raman Microspectroscopy. Skin Pharmacol. Physiol. 2017, 30, 1–12. [Google Scholar] [CrossRef]
- Darlenski, R.; Fluhr, J.W. In vivo Raman Confocal Spectroscopy in the Investigation of the Skin Barrier. Curr. Probl. Dermatol. 2016, 49, 71–79. [Google Scholar] [CrossRef]
- Tippavajhala, V.K.; Magrini, T.D.; Matsuo, D.C.; Silva, M.G.P.; Favero, P.P.; De Paula, L.R.; Martin, A.A. In Vivo Determination of moisturisers Efficacy on Human Skin Hydration by Confocal Raman Spectroscopy. AAPS PharmSciTech 2018, 19, 3177–3186. [Google Scholar] [CrossRef] [PubMed]
- Souza, C.; Maia Campos, P.; Schanzer, S.; Albrecht, S.; Lohan, S.B.; Lademann, J.; Darvin, M.E.; Meinke, M.C. Radical-Scavenging Activity of a Sunscreen Enriched by Antioxidants Providing Protection in the Whole Solar Spectral Range. Skin Pharmacol. Physiol. 2017, 30, 81–89. [Google Scholar] [CrossRef] [PubMed]
- Pudney, P.; Melot, M.; Caspers, P.; Van Der Pol, A.; Puppels, G. An In Vivo Confocal Raman Study of the Delivery of Trans Retinol to the Skin. Appl. Spectrosc. 2007, 61, 804–811. [Google Scholar] [CrossRef] [PubMed]
- Egawa, M.; Sato, Y. In vivo evaluation of two forms of urea in the skin by Raman spectroscopy after application of urea-containing cream. Skin Res. Technol. 2015, 21, 259–264. [Google Scholar] [CrossRef]
- Mélot, M.; Pudney, P.; Williamson, A.; Caspers, P.; Van Der Pol, A.; Puppels, G. Studying the effectiveness of penetration enhancers to deliver retinol through the stratum cornum by in vivo confocal Raman spectroscopy. J. Control. Release 2009, 138, 32–39. [Google Scholar] [CrossRef] [PubMed]
- Weigmanna, J.; Barthelmesa, C.; Schaeferc, H.; Sterrya, G. Penetration of titanium dioxide microparticles in a sunscreen formulation into the horny layer and the follicular orifice. Skin Pharmacol. Appl. Skin Physiol. 1999, 12, 247–256. [Google Scholar]
- Tfayli, A.; Piot, O.; Pitre, F.; Manfait, M. Follow-up of drug permeation through excised human skin with confocal Raman microspectroscopy. Eur. Biophys. J. 2007, 36, 104–1058. [Google Scholar] [CrossRef]
- Song, Y.; Xiao, C.; Mendelsohn, R.; Zheng, T.; Strekowski, L.; Michniak, B. Investigation of iminosulfuranes as novel transdermal penetration enhancers: Enhancement activity and cytotoxicity. Pharm. Res. 2005, 22, 1918–1925. [Google Scholar] [CrossRef]
- Ashtikar, M.; Langelüddecke, L.; Fahr, A.; Deckert, V. Tip-enhanced Raman scattering for tracking of invasomes in the stratum corneum. Biochim. Biophys. Acta Gen. Subj. 2017, 1861, 2630–2639. [Google Scholar] [CrossRef]
- Lohan, S.B.; Saeidpour, S.; Solik, A.; Schanzer, S.; Richter, H.; Dong, P.; Darvin, M.E.; Bodmeier, R.; Patzelt, A.; Zoubari, G.; et al. Investigation of the cutaneous penetration behavior of dexamethasone loaded to nano-sized lipid particles by EPR spectroscopy, and confocal Raman and laser scanning microscopy. Eur. J. Pharm. Biopharm. 2017, 116, 102–110. [Google Scholar] [CrossRef] [PubMed]
- Dos Santos, L.; Téllez S, C.A.; Sousa, M.P.J.; Azoia, N.G.; Cavaco-Paulo, A.M.; Martin, A.A.; Favero, P.P. In vivo confocal Raman spectroscopy and molecular dynamics analysis of penetration of retinyl acetate into stratum corneum. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2017, 174, 279–285. [Google Scholar] [CrossRef] [PubMed]
- Pot, L.M.; Coenraads, P.J.; Blomeke, B.; Puppels, G.J.; Caspers, P.J. Real-time detection of p-phenylenediamine penetration into human skin by in vivo Raman spectroscopy. Contact Dermat. 2016, 74, 152–158. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Cambron, T.; Niu, Y.; Xu, Z.; Su, N.; Zheng, H.; Wei, K.; Ray, P. A MCR approach revealing protein, water and lipid depth profile in atopic dermatitis patients’ stratum corneum via in vivo confocal Raman spectroscopy. Anal. Chem. 2019. [Google Scholar] [CrossRef] [PubMed]
- Perticaroli, S.; Yeomans, D.J.; Wireko, F.C.; Webber, J.T.; Werchowski, K.M.; Cambron, R.T.; Ray, P.J. Translating chemometric analysis into physiological insights from in vivo confocal Raman spectroscopy of the human stratum corneum. Biochim. Biophys. Acta Biomembr. 2019, 1861, 403–409. [Google Scholar] [CrossRef] [PubMed]
- Martin, K. In vivo measurements of water in skin by near-infrared reflectance. Appl. Spectrosc. 1998, 52, 1001–1007. [Google Scholar] [CrossRef]
- Martin, K. Direct measurement of moisture in skin by NIR spectroscopy. J. Soc. Cosmet. Chem. 1993, 44, 249–261. [Google Scholar]
- Walling, P.; Dabney, J. Moisture in skin by near-infrared reflectance spectroscopy. J. Soc. Cosmet. Chem. 1989, 40, 151–171. [Google Scholar]
- Arimoto, H.; Egawa, M. Non-contact skin moisture measurement based on near-infrared spectroscopy. Appl. Spectrosc. 2004, 58, 1439–1446. [Google Scholar] [CrossRef]
- Arimoto, H.; Egawa, M. Skin moisture measurement based on near-infrared spectroscopy and regression analysis. In Proceedings of the 25th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (IEEE Cat. No.03CH37439), Cancun, Mexico, 17–21 September 2003; Volume 4, pp. 3438–3441. [Google Scholar] [CrossRef]
- Egawa, M.; Fukuhara, T.; Takahashi, M.; Ozaki, Y. Determining water content in human nails with a portable near-infrared spectrometer. Appl. Spectrosc. 2003, 57, 473–478. [Google Scholar] [CrossRef]
- Egawa, M.; Arimoto, H.; Hirao, T.; Takahashi, M.; Ozaki, Y. Regional difference of water content in human skin studied by diffuse-reflectance near-infrared spectroscopy: Consideration of measurement depth. Appl. Spectrosc. 2006, 60, 24–28. [Google Scholar] [CrossRef]
- McIntosh, L.M.; Jackson, M.; Mantsch, H.H.; Mansfield, J.R.; Crowson, A.N.; Toole, J.W.P. Near-infrared spectroscopy for dermatological applications. Vib. Spectrosc. 2002, 28, 53–58. [Google Scholar] [CrossRef]
- Takiwaki, H.; Miyaoka, Y.; Arase, S. Analysis of the absorbance spectra of skin lesions as a helpful tool for detection of major pathophysiological changes. Skin Res. Technol. 2004, 10, 130–135. [Google Scholar] [CrossRef] [PubMed]
- Atencio, J.A.D.; Rodriguez, M.C.; Montiel, S.V.y.; Gutierrez, J.L.; Martinez, F.; Gutierrez, B.; Orozco, E.; Castro, J.; Rodriguez, A.C. Diffuse Reflectance Spectroscopy of Human Skin Using a Commercial Fiber Optic Spectrometer. AIP Conf. Proc. 2008, 1032, 105–107. [Google Scholar] [CrossRef]
- Dreassi, E.; Ceramelli, G.; Fabbri, L.; Vocioni, F.; Bartalini, P.; Corti, P. Application of Near-infrared Reflectance Spectrometry in the Studyof AtopyPart 1. Investigation of Skin Spectra. Analyst 1997, 122, 767–770. [Google Scholar] [CrossRef] [PubMed]
- Dreassi, E.; Ceramelli, G.; Mura, P.; Perruccio, P.L.; Vocioni, F.; Bartalini, P.; Corti, P. Near-infrared Reflectance Spectrometry in the Studyof AtopyPart2. Interactions Between the Skin and Polyethylene Glycol400, Isopropyl Myristate and Hydrogel. Analyst 1997, 122, 771–776. [Google Scholar] [CrossRef]
- Corti, P.; Ceramelli, G.; Dreassi, E.; Njine, M. Near infrared reflectance spectroscopy in the study of atopy Part 3.† Interactions between the skin and fomblins. Analyst 1998, 123, 2313–2317. [Google Scholar] [CrossRef]
- Boden, I.; Nilsson, D.; Naredi, P.; Lindholm-Sethson, B. Characterization of healthy skin using near infrared spectroscopy and skin impedance. Med. Biol. Eng. Comput. 2008, 46, 985–995. [Google Scholar] [CrossRef]
- Greve, T.M.; Kamp, S.; Jemec, G.B.E. Disease quantification in dermatology: In vivo near-infrared spectroscopy measures correlate strongly with the clinical assessment of psoriasis severity. J. Biomed. Opt. 2013, 18, 037006. [Google Scholar] [CrossRef]
- De Rigal, J.; Losch, M.; Bazin, R.; Camus, C.; Sturelle, C.; Descamps, V.; Leveque, J. Near infrared spectroscopy: A new approach to the characterization of dry skin. J. Soc. Cosmet. Chem. 1993, 44, 197–209. [Google Scholar]
- Egawa, M. In vivo simultaneous measurement of urea and water in the human stratum corneum by diffuse-reflectance near-infrared spectroscopy. Skin Res. Technol. 2009, 15, 195–199. [Google Scholar] [CrossRef] [PubMed]
- Mohamad, M.; Msabbri, A.R.; MatJafri, M.Z. Conceptual design of near infrared spectroscopy instrumentation for skin moisture measurement. In Proceedings of the 2011 IEEE Colloquium on Humanities Science and Engineering, Penang, Malaysia, 5–6 December 2011; pp. 801–804. [Google Scholar] [CrossRef]
- Wichrowski, K.; Sore, G.; Khaïat, A. Use of infrared spectroscopy for in vivo measurement of the stratum corneum moisturization after application of costmetic preparations. Int. J. Cosmet. Sci. 1995, 17, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Qassem, M.; Kyriacou, P. Use of reflectance near-infrared spectroscopy to investigate the effects of daily moisturiser application on skin optical response and barrier function. J. Biomed. Opt. 2014, 19. [Google Scholar] [CrossRef] [PubMed]
- Qassem, M.; Kyriacou, P. In vivo investigation of short term skin water contact and moisturiser application using NIR Spectroscopy. In Proceedings of the 2013 35th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC), Osaka, Japan, 3–7 July 2013. [Google Scholar]
- Qassem, M.; Kyriacou, P.A. Reflectance near-infrared measurements for determining changes in skin barrier function and scattering in relation to moisturiser application. J. Biomed. Opt. 2015, 20, 095008. [Google Scholar] [CrossRef] [PubMed]
- Qassem, M.; Kyriacou, P. Investigating skin barrier function utilizing reflectance NIR Spectroscopy. In Proceedings of the 2014 36th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC), Chicago, IL, USA, 26–30 August 2014; pp. 3735–3738. [Google Scholar] [CrossRef]
- Huang, X.; Yeo, W.H.; Liu, Y.; Rogers, J.A. Epidermal differential impedance sensor for conformal skin hydration monitoring. Biointerphases 2012, 7, 52. [Google Scholar] [CrossRef] [PubMed]
- Webb, R.C.; Bonifas, A.P.; Behnaz, A.; Zhang, Y.; Yu, K.J.; Cheng, H.; Shi, M.; Bian, Z.; Liu, Z.; Kim, Y.S.; et al. Ultrathin conformal devices for precise and continuous thermal characterization of human skin. Nat. Mater. 2013, 12, 938–944. [Google Scholar] [CrossRef]
- Krishnan, S.; Shi, Y.; Webb, R.C.; Ma, Y.; Bastien, P.; Crawford, K.E.; Wang, A.; Feng, X.; Manco, M.; Kurniawan, J.; et al. Multimodal epidermal devices for hydration monitoring. Microsyst. Nanoeng. 2017, 3, 17014. [Google Scholar] [CrossRef]
- Woo, Y.; Ahn, J.; Chun, I.; Kim, H. Development of a method for the determination of human skin moisture using a portable near-infrared system. Anal. Chem. 2001, 73, 4964–4971. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Qassem, M.; Kyriacou, P. Review of Modern Techniques for the Assessment of Skin Hydration. Cosmetics 2019, 6, 19. https://doi.org/10.3390/cosmetics6010019
Qassem M, Kyriacou P. Review of Modern Techniques for the Assessment of Skin Hydration. Cosmetics. 2019; 6(1):19. https://doi.org/10.3390/cosmetics6010019
Chicago/Turabian StyleQassem, Meha, and Panayiotis Kyriacou. 2019. "Review of Modern Techniques for the Assessment of Skin Hydration" Cosmetics 6, no. 1: 19. https://doi.org/10.3390/cosmetics6010019
APA StyleQassem, M., & Kyriacou, P. (2019). Review of Modern Techniques for the Assessment of Skin Hydration. Cosmetics, 6(1), 19. https://doi.org/10.3390/cosmetics6010019





