A New Carrier for Advanced Cosmeceuticals
Abstract
1. Introduction
2. Cosmetic Products
3. Bionanotechnology and Industrial R&D Innovation
4. Biopolymers from Nature
5. The Cosmetics and Active Ingredients Market
6. Chitin Nanofibrils and Nanolignin to Make Renewable Fabrics
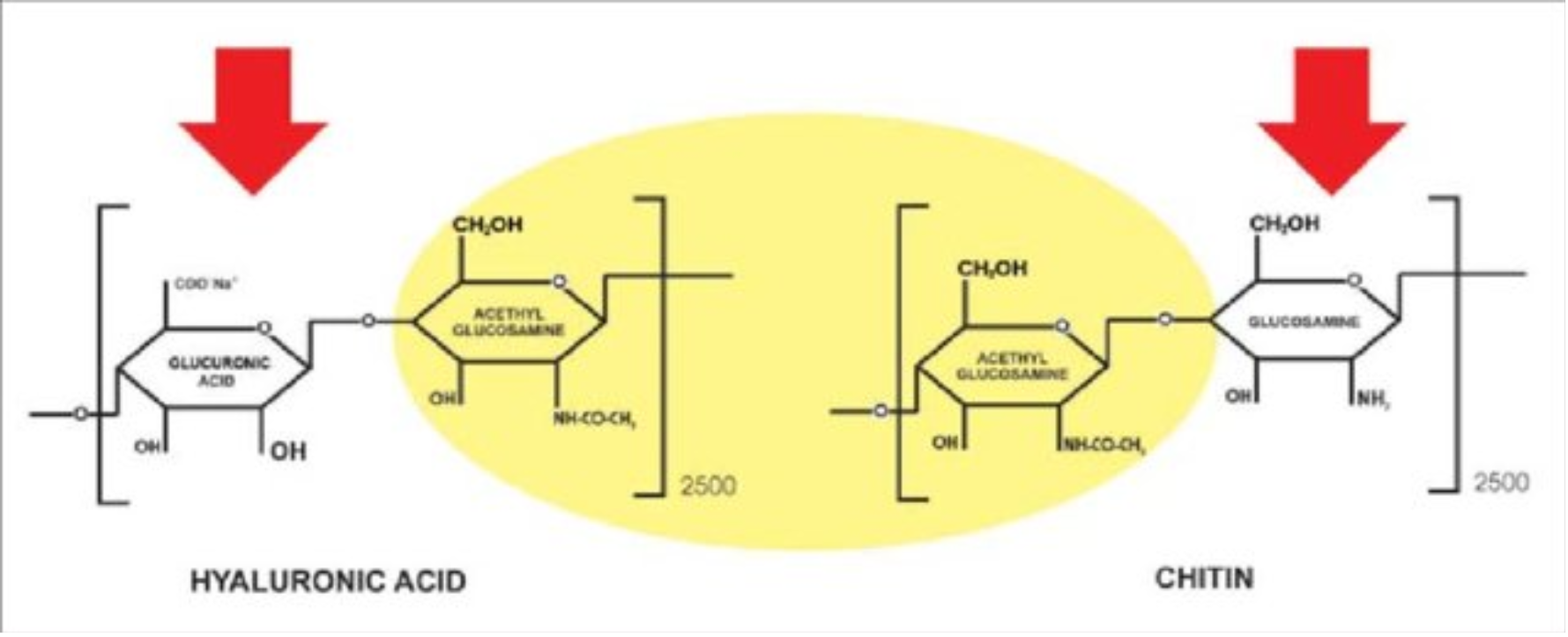
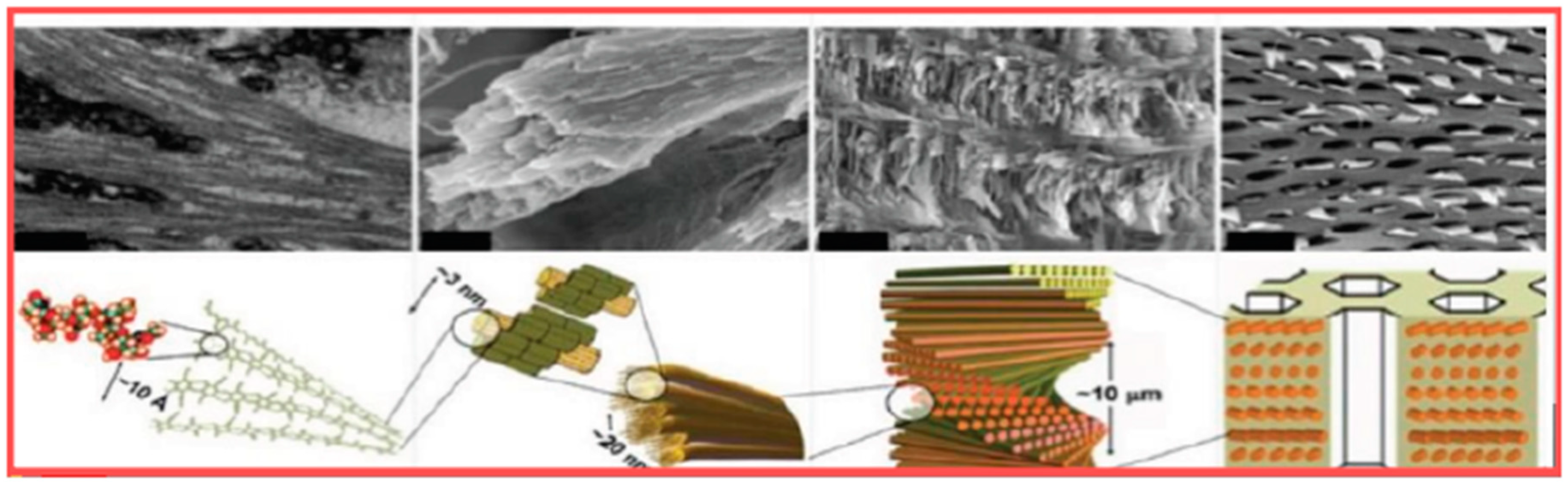
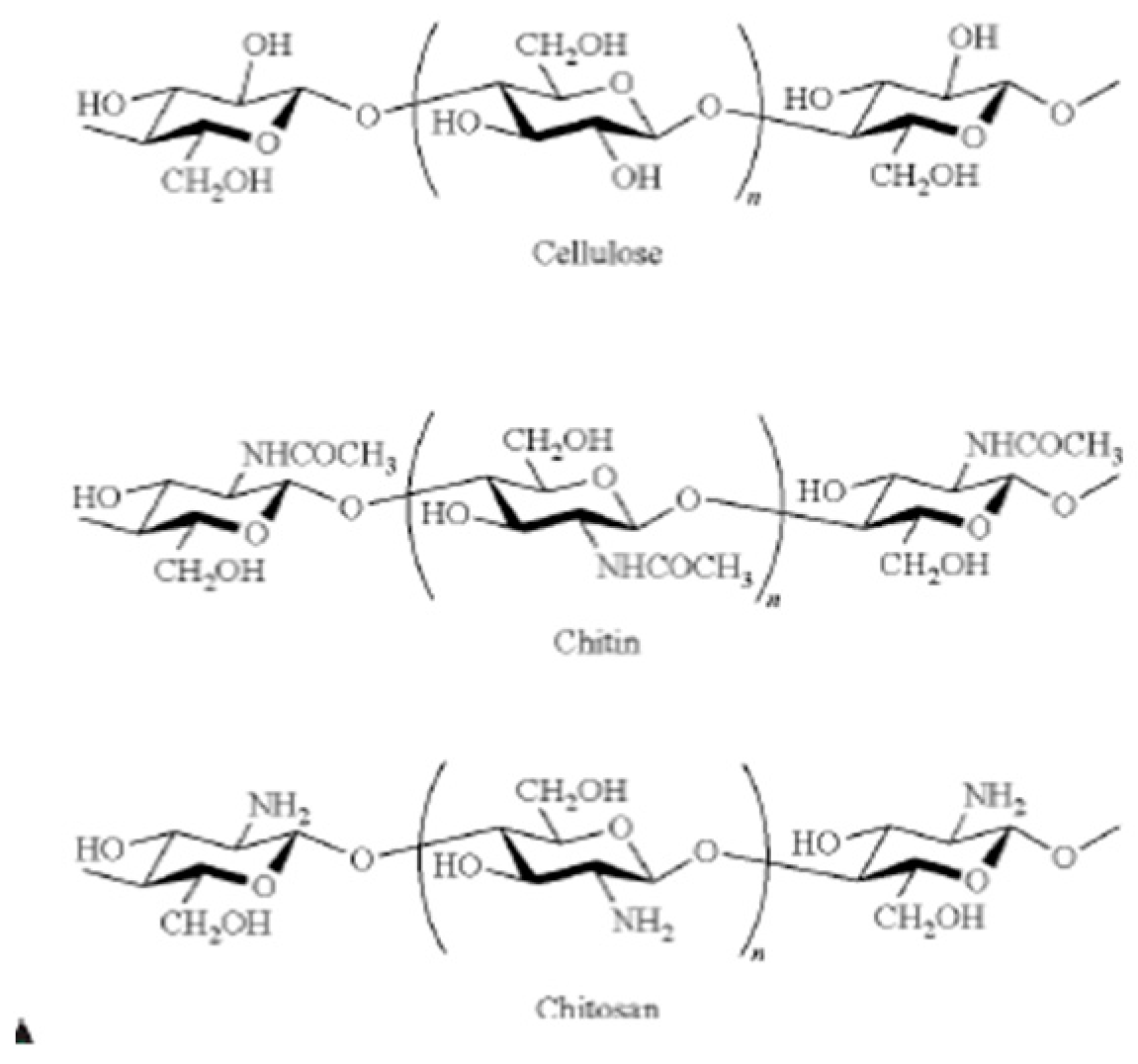
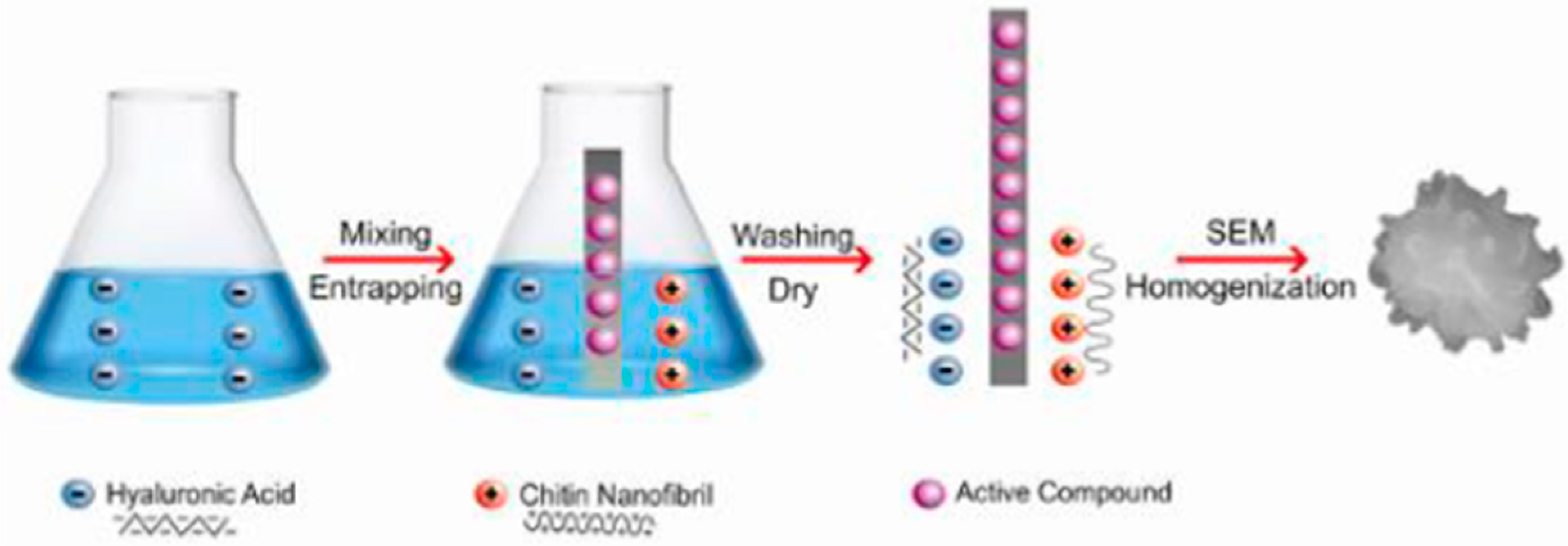
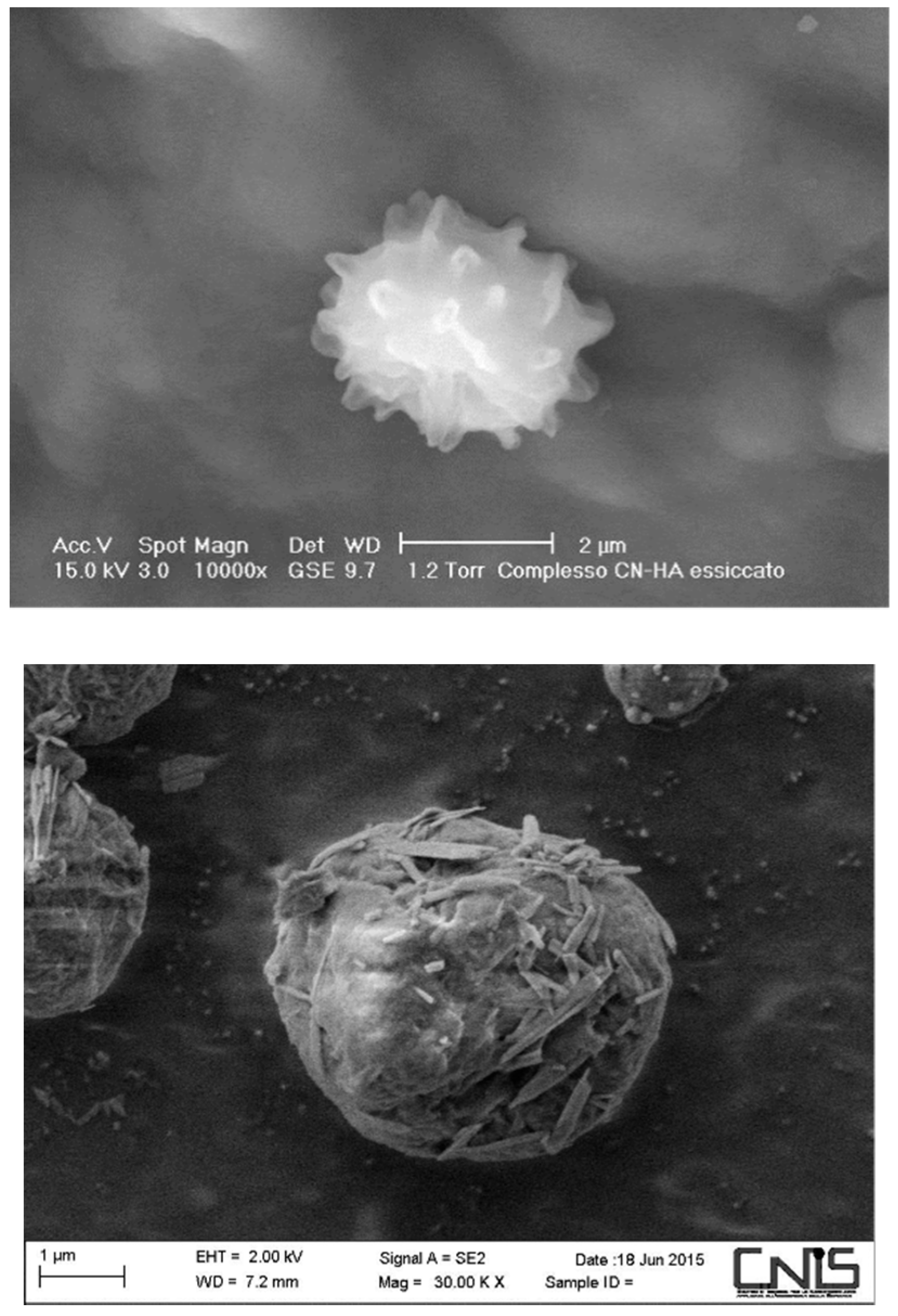
6.1. Chitin
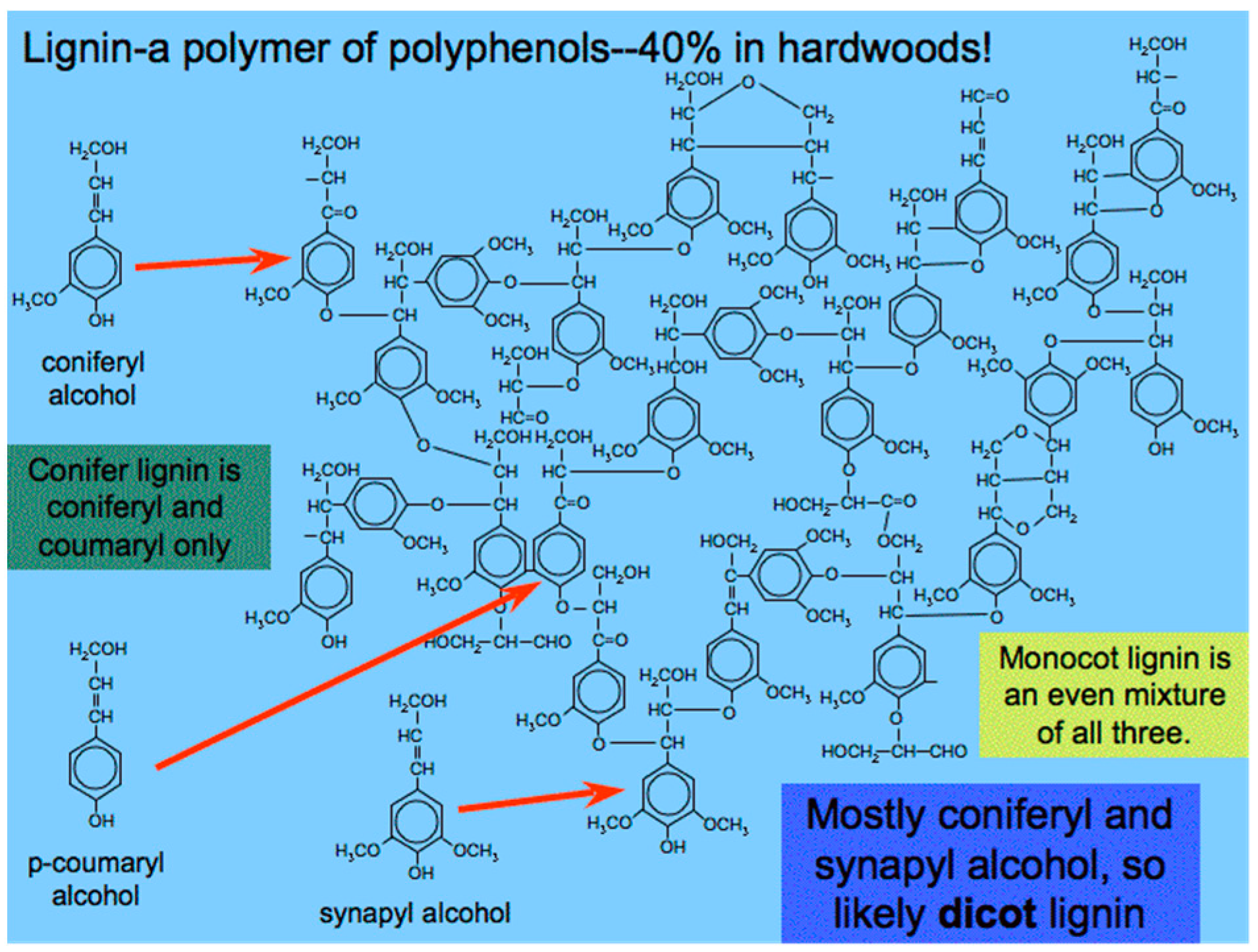
6.2. Lignin
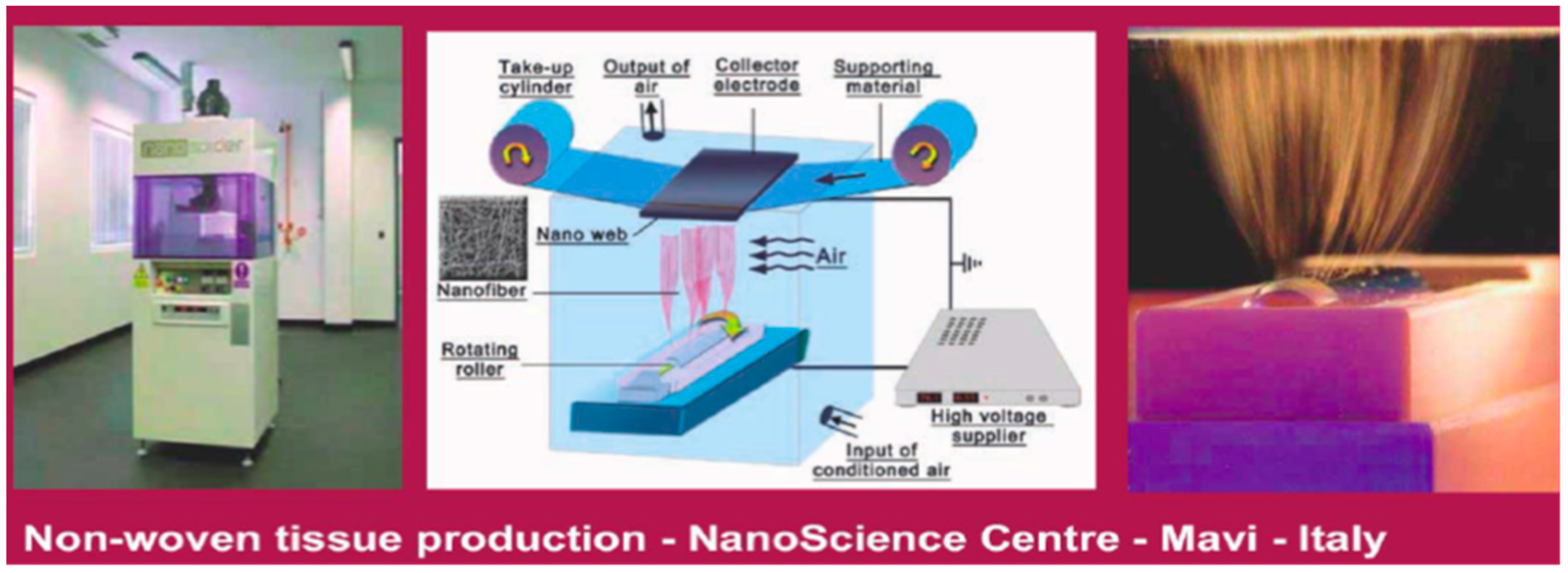
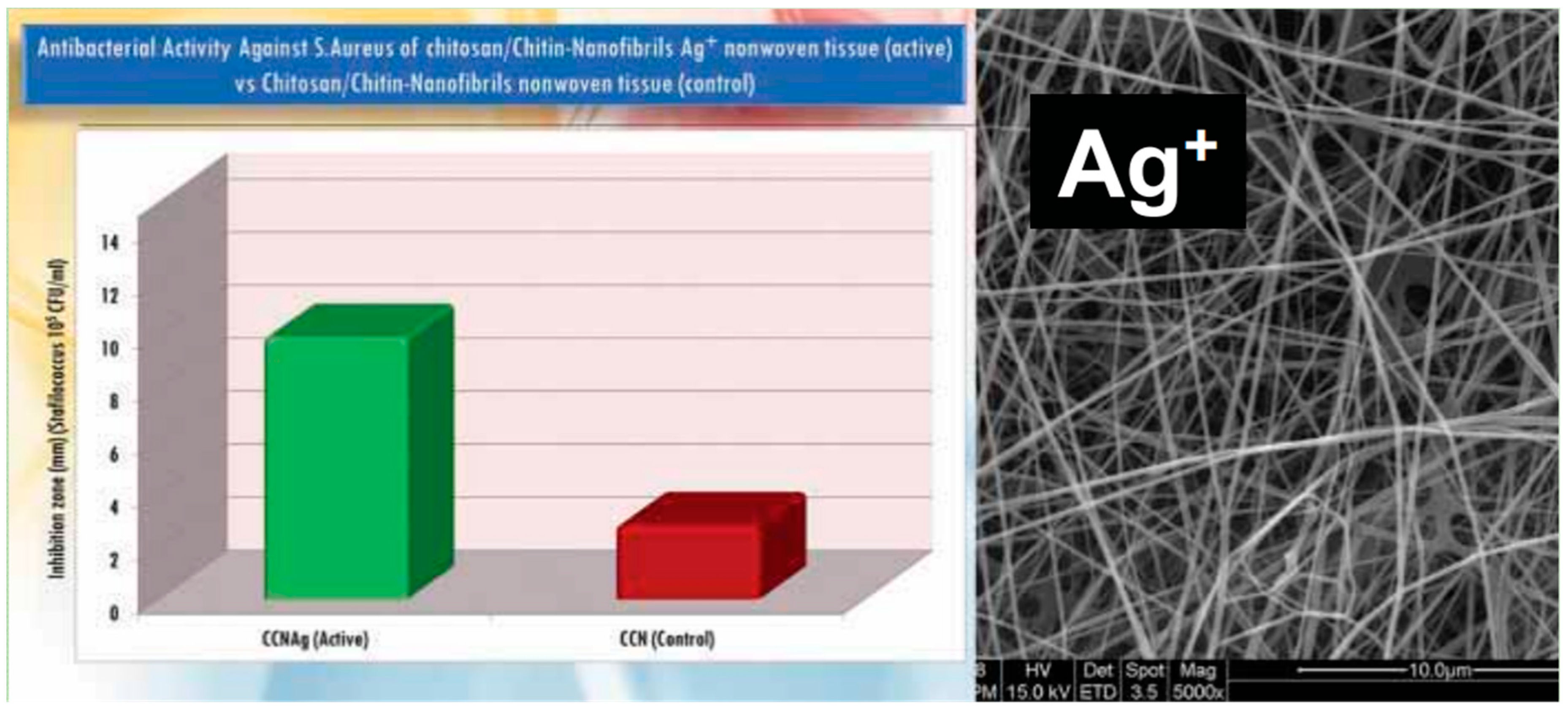
7. Bionanocomposites and Nonwoven Tissues
8. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Schueller, R.; Romanowski, P. Beginning Cosmetic Chemistry; Allured Publishing Co.: Carol Stream, IL, USA, 2003. [Google Scholar]
- Schueller, R.; Romanowski, P. Multifunctional Cosmetics; Marcel Dekker, Inc.: New York, NY, USA, 2003. [Google Scholar]
- Epstein, H. Skin Care Products. In Cosmetic Science and Technology; Barel, A.O., Paye, M., Maibach, H.I., Eds.; Informa Healthcare: New York, NY, USA, 2009; pp. 121–134. [Google Scholar]
- Council Directive 76/768/EEC of 27 July 1976. On the Approximation of the Laws of the Member States Relating to Cosmetic Products. OJEC n. l. 262/169.27.9.1976 (and Following Fmendments), Official Journal of the European Communities. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/PDF/?uri=CELEX:31976L0768&from=HU (accessed on 11 February 2019).
- Regulation (EC) No 1223/2009 of the European Parliament and of the Council, November 30 2009 on Cosmetic products, Official Journal of the European Union. Available online: https://ec.europa.eu/health/sites/health/files/endocrine_disruptors/docs/cosmetic_1223_2009_regulation_en.pdf (accessed on 11 February 2019).
- Morganti, P.; Paglialunga, S. EU Borderline cosmetic products: Review of current regulatory status. Clin. Dermatol. 2008, 26, 392–397. [Google Scholar] [CrossRef]
- Reed, E. The definition of “cosmeceuticals”. J. Soc. Cosmet. Chem. 1962, 13, 103–116. [Google Scholar]
- Kligman, A.M. Why cosmeceuticals? Cosmet. Toilet. 1995, 108, 37–40. [Google Scholar]
- Skechers, J.W. Cosmetic: Are crossing the final barrier into Dermatology. J. Appl. Cosmetol. 2007, 25, 1–10. [Google Scholar]
- Sivamani, R.V.; Jagdeo, J.; Elsner, P.; Maibach, H.I. Cosmeceuticals and Active Cosmetics: Drugs Versus Cosmetics, 3rd ed.; Tailor& Francis: New York, NY, USA, 2015. [Google Scholar]
- Ames, N.S.; Ranucci, A.; Moriyama, B.; Wallenberg, G.R. Understanding the 16SrRNA Gene in Translational Nursing Science. Nurs. Res. 2017, 6, 184–197. [Google Scholar] [CrossRef]
- Morganti, P.; Del Ciotto, P.; Stoller, M.; Chianese, A. Antibacterial and Anti-inflammatory Green Nanocomposites. Chem. Eng. Trans. 2016, 47, 61–66. [Google Scholar]
- Morganti, P.; Fusco, A.; Paoletti, I.; Perfetto, B.; Del Ciotto, P.; Palombo, M.; Chianese, A.; Baroni, A.; Donnarumma, G. Antiinflammatory, Immunomodulatory and Tissue Repair Activity on Human Keratinocytes by Green Innovative Nanocomposites. Materials 2017, 10, 843. [Google Scholar] [CrossRef] [PubMed]
- Renner, G.; Aufebert, F.; Burfeindt, J.; Calvet Carhths-Perifan, M. Cosmetic Europe Guidelines on the Management of Undesirable Effects and Reporting of Serious Undesirable Effects from Cosmetics in the European Union. Cosmetics 2017, 4, 1. [Google Scholar] [CrossRef]
- Cornell, E.; Kwa, M.; Pallet, A.S.; Xu, S. Adverse events reported to the Food and Drug Administration from 2004 to 2016 for cosmetics and personal care products marketed to newborns and infants. Pediatr. Dermatol. 2018, 35, 225–229. [Google Scholar] [CrossRef] [PubMed]
- Inetianbor, J.E.; Yakubu, J.M.; Zeonu, S.C. Effects of Food Additives and Preservatives on man-a Review. Asian J. Sci. Technol. 2015, 6, 1118–1135. [Google Scholar]
- Soni, M.G.; Carabin, I.G.; Burdock, G.A. Safety assessment of p-Hydroxy benzoic acid(parabens). Food Chem. Toxicol. 2005, 43, 986–1015. [Google Scholar] [CrossRef]
- Paszkiewicz, W. Parabens: Food Preservatives and Consumer Safety. Med. Weter. 2015, 71, 553–556. [Google Scholar]
- Muse, H.P.; Frost, S.; Ohund, U.; Foscn, K.E. Allergic contact dermatitis from octylthiazolinone. Contact Dermat. 2013, 69, 49–52. [Google Scholar] [CrossRef] [PubMed]
- Isaksson, M.; Hauksson, I.; Hindsen, M.; Ponten, A.; Svedman, C.; Bruze, M. Methylisothiazolinone Contact Allergy is Rising to Alarming Heights also in Southern Sveden. Acta Derm. Venereol. 2015, 96, 32–34. [Google Scholar]
- Aerts, O.; Goossen, A.; Lambert, J.; Leopoittevin, J.P. Contact Allergy Caused by Isothiazolinone Derivatives: An Overview of non-cosmetic and unusual cosmetic sources. Eur. J. Dermatol. 2017, 27, 115–122. [Google Scholar] [CrossRef] [PubMed]
- Elmonem, M.A.; van de Heuvel, L.P.; Levtchenko, E.N. Immunomodulatory Effects of Chitotriosidase Enzyme. Enzyme Res. 2016. [Google Scholar] [CrossRef] [PubMed]
- Sender, R.; Fuchs, S.; Milo, R. Revised Estimates for the Number of a Human and Bacteria Cells in the Body. PLoS Biol. 2016, 14, e1002533. [Google Scholar] [CrossRef] [PubMed]
- Morganti, P.; Stoller, M. Chitin and Lignin: Natural Ingredients from Waste Materials to Make Innovative and Healthy Products for Humans and Plant. Chem. Eng. Trans. 2017, 60, 319–324. [Google Scholar]
- Vinardell, M.P.; Mityans, M. Lignins and Their Derivatives with Beneficial Effects on Human Health. Int. J. Mol. Sci. 2017, 18, 1219. [Google Scholar] [CrossRef]
- Beisl, S.; Friedl, A.; Miltner, A. Lignin from micro-to Na-nosize: Applications. Int. J. Mol. Sci. 2017, 18, 2367. [Google Scholar] [CrossRef]
- Buscema, M.; Pieri, G. Ricerca Scientifica e Innovazione; Rizzoli: Milano, Italy, 2003. [Google Scholar]
- Braconier, H.; Nicoletti, G.; Westemore, B. Policy Challenges for the Next 50 Years. OECD Economics Department Policy Papers N 9, Paris, France. 2014. Available online: www.oecd.org (accessed on 14 March 2018).
- OECD, Innovation Strategy 2015. 2015, An Agenda for Policy Action. Meeting of OECD Council at Ministerial Level, Paris, France, 3–4 June. Available online: www.oecd.org (accessed on 11 February 2019).
- Mulhaupt, R. Green Polymers Chemistry and Bio-Based Plastics: Dreams and Reality. Chem. Phys. 2013, 214, 159–174. [Google Scholar] [CrossRef]
- Miscallef, N. Reimaging Growth in the Global Beauty; Euromonitor International: Aberdeen, UK, 2017. [Google Scholar]
- Rai, R.; Roether, J.A.; Knowles, J.C.; Mordan, N.; Roy, I. Highly Elastomeric Poly(3-hydroxy octanoate) Based Natural Polymer Composite for Enhanced Keratinocyte Regeneration. Int. J. Polym. Mater. 2018, in press. [Google Scholar]
- Benkeblla, N. Polysaccharides: Natural Fibers in Food and Nutrition; CRC Press: New York, NY, USA, 2014. [Google Scholar]
- M&M. Active Ingredients Market for Cosmetics by Functionality Magarpatta, Maharashtra, India. 2018. Available online: www.marketsandmarkets.com (accessed on 19 March 2018).
- WHO. Ambient Air Pollution: A Global Assessment of Exposure and Burden of Disease; World Heath Organization: Geneva, Switzerland, 2016. [Google Scholar]
- English, J.S.; Dawe, R.S.; Ferguson, J. Environmental effects and skin disease. Br. Med. Bull. 2003, 68, 129–142. [Google Scholar] [CrossRef] [PubMed]
- Kim, E.K.; Kim, S.; Lee, J.H.; Han, Y.; Kim, Y.M.; Kim, G.B.; Jung, K.; Cheong, H.K.; Ahn, K. Indoor air pollution aggravates symtoms of atopic dermatitis in children. PLoS ONE 2015, 10, e0119501. [Google Scholar]
- Kim, K.E.; Cho, D.; Park, H.J. Air pollution and skin diseases: Adverse effects of airborne particulate matter on various skin diseases. Life Sci. 2016, 152, 126–134. [Google Scholar] [CrossRef] [PubMed]
- US Commercial Service Hong Kong. Cosmetic & Toiletries Market Overviews 2015; Country Commercial Guide and Industrial Report; USA-International Trade Organization: New York, NY, USA, 2016.
- Gerstle, T. Asia Personal Care & Cosmetic Market Guide 2016; Country Commercial Guide and Industrial Report; USA-International Trade Organization: New York, NY, USA, 2016.
- Morganti, P.; Coltelli, M.B.; Danti, S. Biobased tissues for innovative cosmetic products: PolyBioSkin as an EU Research Project. Glob. J. Nanomed. 2018, 3. [Google Scholar] [CrossRef]
- Kwek, S. The Facial Masks Remain a Covered Item among Urban Chinese Consumers; Mintel Global New Products Database: London, UK, 2017. [Google Scholar]
- GIA Inc. Chitin & Chitosan: A Global Strategic Business Report; Global Industry Analists Inc.: New York, NY, USA, 2018. [Google Scholar]
- Stern, R.; Jedrzejas, M. Hyaluronic asset: Their genomical structure and mechanisms of action. Chem Rev. 2006, 106, 818–839. [Google Scholar] [CrossRef] [PubMed]
- Ige, O.O.; Umoru, L.E.; Aribo, S. Natural Products: A Minefield of Biomaterials. ISRN Mater. Sci. 2012. [Google Scholar] [CrossRef]
- Svetoslav, N.; Petrov, M.; Lymperkis, L.; Friak, M.; Sachs, C.; Fabritius, H.O.; Raabe, D.; Negeubaues, J. Revealing the Deign Principles of High-Performance Biological Composites using Ab initio and Multiscale Simulations: The example of Loabster cuticle. Adv. Mater. 2010, 22, 519–626. [Google Scholar]
- Morganti, P. Biomimetic Materials Mimicking Nature at the Base of EU Projects. J. Sci. Res. Rep. 2014, 3, 532–544. [Google Scholar] [CrossRef]
- Qu, T.; Verma, D.; Alucozai, M.; Toman, R. Influence of Interactions on Deformation Mechanism and Interface Viscosity in a Chitin-Calcite Interfaces. Acta Biomater. 2015, 25. [Google Scholar] [CrossRef] [PubMed]
- Bueter, C.L.; Specht, C.A.; Levitz, S.M. Innate Sensing of Chitin and Chitosan. PLoS Pathog. 2013, 9, e1003080. [Google Scholar] [CrossRef]
- Muzzarelli, R.A.A.; El Mehtedi, M.; Mattioli/Belmonte, M. Emerging Biomedical Applications of Nano/Chitins and Nano-Chitosans Obtained via Advanced Eco-Friendly Technologies from Marine Resources. Mar. Drugs 2014, 12, 5468–5502. [Google Scholar] [CrossRef] [PubMed]
- Zhao, D.; Yu, S.; Sun, B.; Gao, S.; Guo, S.; Zhao, K. Biomedical Applications of Chitosan and Its Derivative Nanoparticles. Polymers 2018, 10, 462. [Google Scholar] [CrossRef]
- Crisponi, G.; Nurchi, V.M.; Lachowicz, J.I.; Peana, M.; Medici, S.; Zoruddu, M.A. Toxicity of Nanoparticles: Etiology and Mechanisms. In Antimicrobial Architectonics from Synthesis to Applications; Grumezescu, A., Ed.; Elsevier: New York, NY, USA, 2017; pp. 512–537. [Google Scholar]
- Morganti, P.; Palombo, M.; Carezzi, F.; Nunziata, M.L.; Morganti, G.; Cardillo, M.; Chianese, A. Green Nanotechnology Bioeconomy Serving: Natural Beauty Masks to Save the Environment. Cosmetics 2016, 3, 41. [Google Scholar] [CrossRef]
- Donnarumma, G.; Fusco, A.; Morganti, P.; Palombo, M.; Anniboletti, T.; Del Ciotto, P.; Baroni, A.; Chianese, A. Advanced Medications made by Green Nanocomposites. Int. J. Res. Nano Sci. 2018, 5, 261–270. [Google Scholar]
- Eide, K.B.; Norberg, A.L.; Heggset, E.B.; Lindbom, A.R.; Varum, K.M.; Eijsink, V.G.H.; Sorlie, M. Human Chitotriosidase-Catalyzed Hydrolosis of Chitosan. Biochemistry 2012, 52, 487–495. [Google Scholar] [CrossRef] [PubMed]
- Morganti, P.; Carezzi, F.; Del Ciotto, P.; Morganti, G.; Nunziata, M.L.; Gao, X.H.; Chen, H.D.; Tischenko, G.; Yudin, V.E. Chitin Nanofibrils: A Natural Multifunctional Polymer. Physicochemical characteristics, effectiveness and safeness. In Nanobiotechnology; Phoenix, D.A., Waqar, A., Eds.; One Central Press: Manchester, UK, 2014; pp. 1–37. [Google Scholar]
- Martinez, V.; Mitjans, M.; Binardell, M.P. Pharmacological Applications of lignins and lignins related compounds: An overview. Curr. Organ. Chem. 2012, 16, 1863–1870. [Google Scholar] [CrossRef]
- Kim, S.; Fernandes, M.M.; Matama, T.; Loureiro, A.; Gomes, A.C.; Cavaco-Paulo, A. Chitosan-lignosulfonates sono-chemically prepared Nanoparticles: Characterization and potential applications. Colloids Surf. B Biointerfaces 2013, 103, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Kumar Gupta, A.; Mohanty, S.; Nayak, S.K. Synthesis, Characterization and Application of Lignin Nanoparticles (LNPs). Mater. Focus 2014, 3, 444–454. [Google Scholar] [CrossRef]
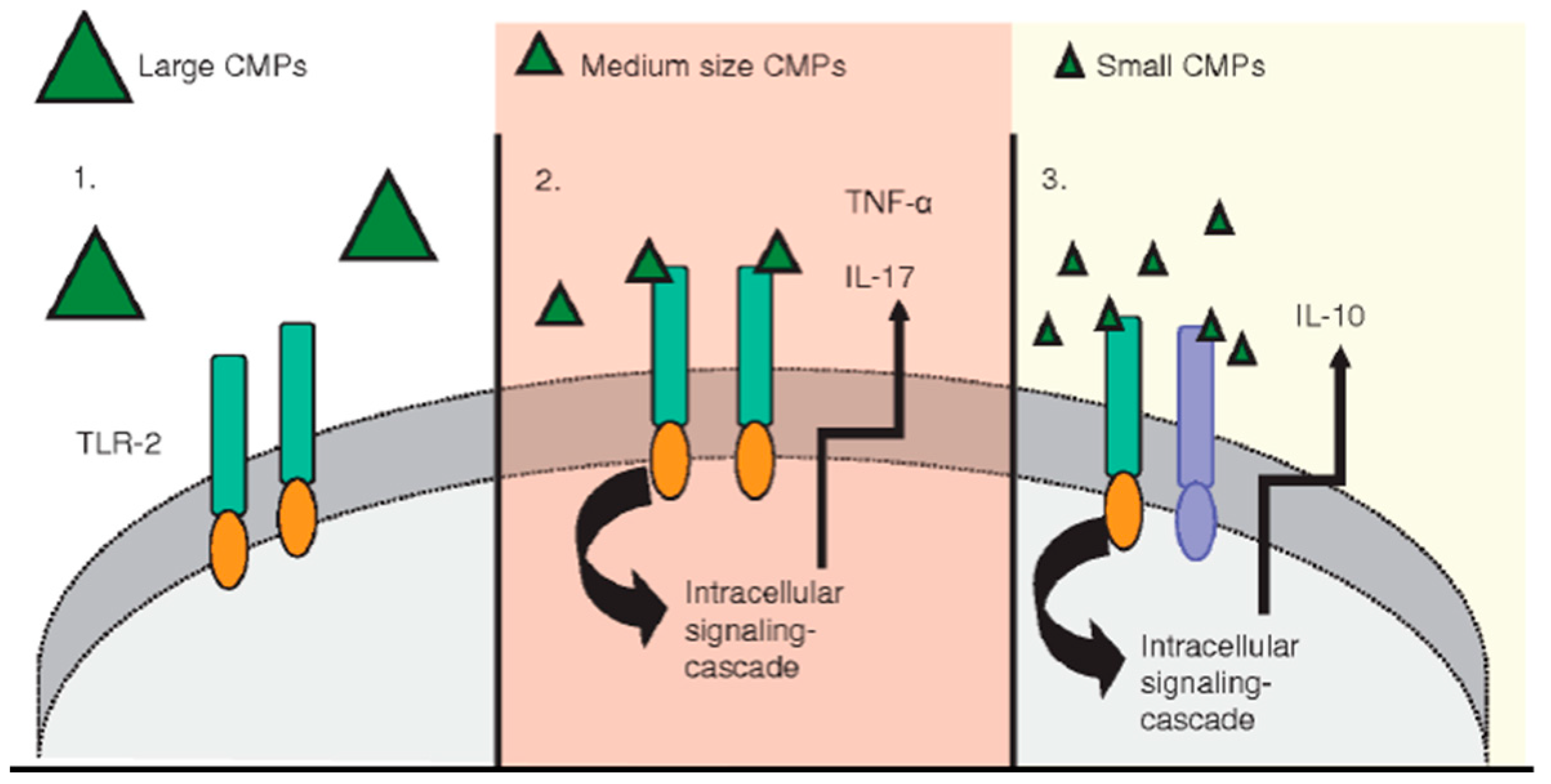
- Da Silva, C.A.; Chalouni, C.; Williams, A.; Hartl, D.; Lee, C.G.; Elias, J.A. Chitin is a size-dependent regulator of machrophage TNF and IL-10 production. J. Immunol. 2009, 182, 3573–3582. [Google Scholar] [CrossRef] [PubMed]
- Brinchmann, B.C.; Bayat, M.; Brogger, T.; Muttuvelu, D.V.; Tjonneland, A.; Sigsgaard, T. A possible role of chitin in the pathogenesis of asthma and allergy. Ann. Agric. Environ. Med. 2011, 18, 7–12. [Google Scholar] [PubMed]
- Lee, C.G.; Da Silva, C.A.; Dela Cruz, C.S.; Ahangari, F.; Ma, B.; Kang, M.J.; He, C.H.; Takyar, S.; Elias, J.A. Role of Chitin and Chitinase/Chitinase-Like Proteins in Inflammation, Tissue Remodeling, and Injury. Annu. Rev. Physiol. 2011, 73. [Google Scholar] [CrossRef] [PubMed]
- Mushi, N.E.; Utsen, S.; Berglund, L.A. Nanostructured biocomposite film of high toughness based on native Chitin nanofibrils and chitosan. Front. Chem. 2014. [Google Scholar] [CrossRef] [PubMed]
- Hamed, I.; Ozogul, F.; Regenstein, L.M. Industrial Applications of Crustacean by-products (Chitin, Chitosan and Chitooligosaccgarides): A review. Trends Food Sci. Technol. 2016, 48, 40–50. [Google Scholar] [CrossRef]
- Qian, Y.; Deng, Y.; Qiu, X.; Li, H.; Yang, D. Formation of uniform colloidal spheres from lignin, a renewable resource recovered from pulping spent liquor. Green. Chem. 2014, 16, 2156–2163. [Google Scholar] [CrossRef]
- Calvin-Flores, F.G.; Dobado, J.A. Lignin as renewable raw material. ChemSusChem 2010, 3, 1227–1235. [Google Scholar] [CrossRef] [PubMed]
- Henriksson, G. What are the biological functions of lignin and its complex action with carbohydrates? Nord. Pulp Pap. Res. J. 2017, 32, 527–541. [Google Scholar] [CrossRef]
- Tian, D.; Hu, J.; Bao, J.; Chandra, R.P.; Saddler, J.N.; Lu, C. Lignin valorization: Lignin nanoparticles as high-value bio-additive for multifunctional nanocomposites. Biotechnol. Biofuels 2017, 10, 192. [Google Scholar] [CrossRef] [PubMed]
- Penna, M.J.; Mijajlovjc, M.; Biggs, M.J. Molecular level understanding of protein adsorption of the interface between water and a strongly interacting uncharged solid surface. J. Am. Chem. Sci. 2013, 135, 5323–5331. [Google Scholar] [CrossRef]
- Beisl, S.; Miltner, A.; Friedl, A. Lignin from Micro- to Nano size: Production Methods. Mol. Sci. 2017, 18, 1244. [Google Scholar] [CrossRef] [PubMed]
- Coltelli, M.B.; Cinelli, P.; Gigante, V.; Aliotta, L.; Morganti, P.; Panariello, L.; Lazzeri, A. Chitin Nanofibrils in Poly(Lactic acid)(PLA) Nanocomposites: Dispersion and Thetmo-Mechanical Properties. Int. J. Mol. Sci. 2019, 20, 504. [Google Scholar] [CrossRef] [PubMed]
- Buragohain, M.K. Composite Structures; CRC Press: New York, NY, USA, 2017. [Google Scholar]
- Kang, J.H.; Castano, O.; Kim, H.W. Electrospun materials as potential platforms for bone tissue engineering. Adv. Drug Deliv. Rev. 2009, 62, 1065–1083. [Google Scholar]
- Keeney, M.; Han, L.H.; Onyah, S.; Yang, F. Tissue Engineering: Focus on the Musculoskeletal System. In Biomaterials Science; Rosen, Y., Elman, N., Eds.; CRC Press: New York, NY, USA, 2012; pp. 191–221. [Google Scholar]
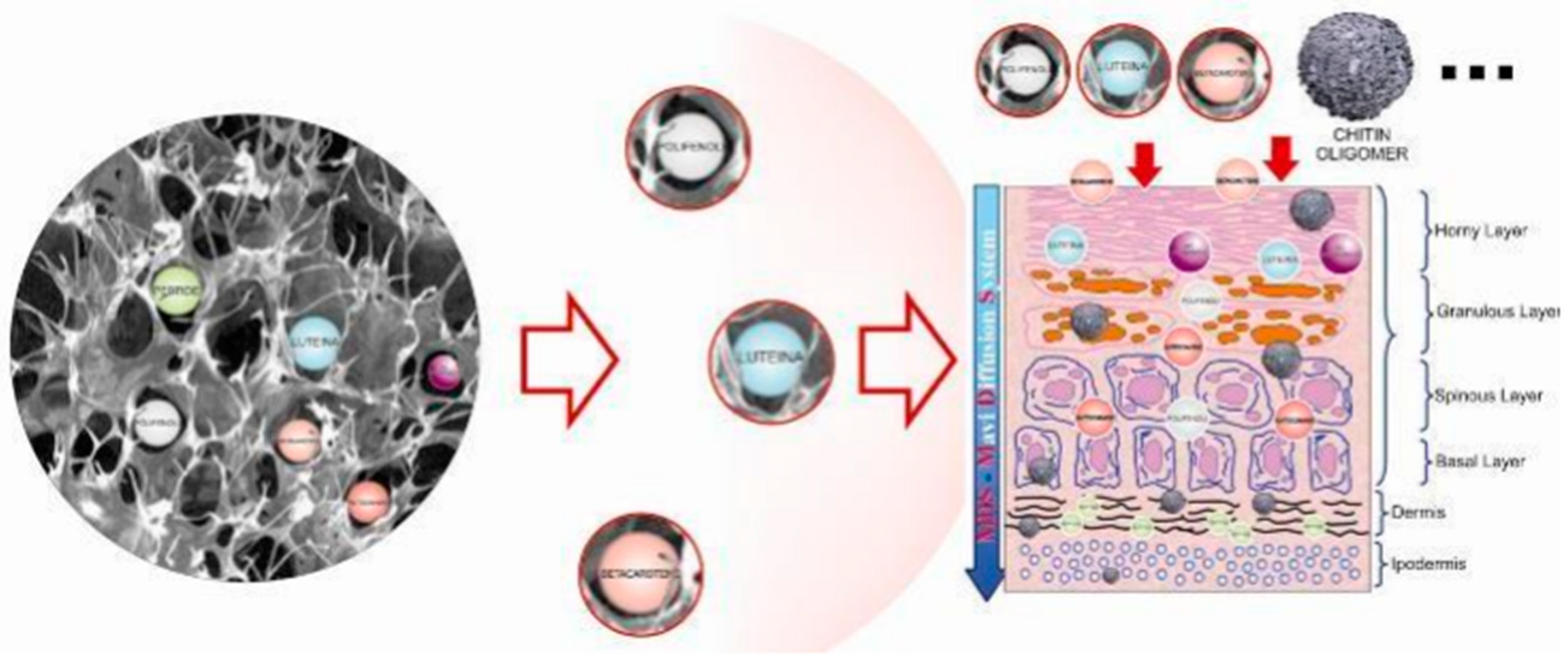
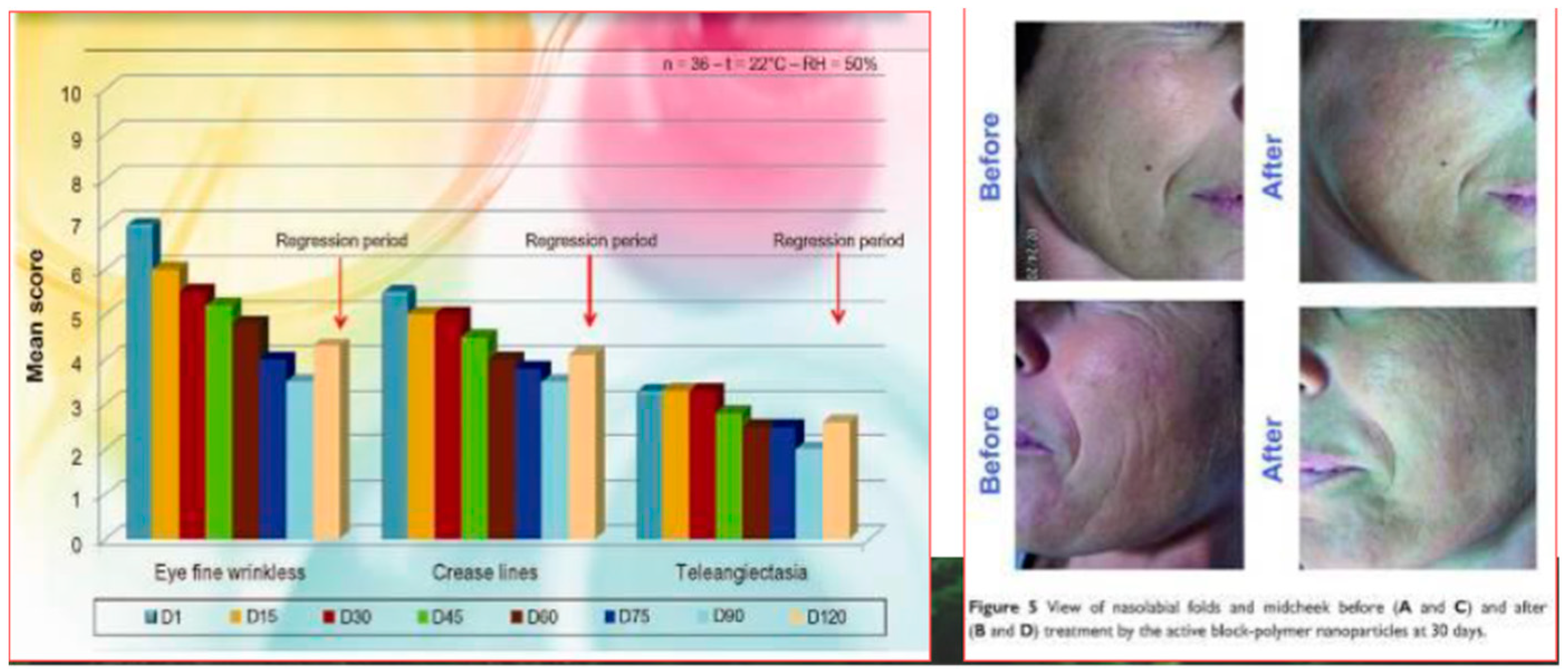
- Morganti, P.; Palombo, M.; Tishchenko, G.; Yudin, V.E.; Guarneri, F.; Cardillo, M.; Del Ciotto, L. Chitin-hyaluronan Nanoparticles to deliver Anti-aging Active Ingredients Through the Skin. Cosmetics 2014, 1, 140–158. [Google Scholar] [CrossRef]
- Morganti, P.; Del Ciotto, P.; Carezzi, F.; Guarneri, F.; Yeo, Y.J. Skin Lightening Efficacy of New Formulations Enhanced by Chitin Nanoparticles Delivery System. Note I. J. Appl. Cosmetol. 2014, 32, 57–71. [Google Scholar]
- Morganti, P.; Fabrizi, G.; Palombo, M.; Cardillo, M.; Cardillo, A.; Del Ciotto, P.; Carezzi, F.; Morganti, G. Activity of Chitin Nanofibrils Block-Copolymers Entrapping Zn/Al/SA/Allantoin on Seborrheic Dermatitis. A randomized double-blind placebo controlled study. J. Appl. Cosmetol. 2014, 32, 3–19. [Google Scholar]
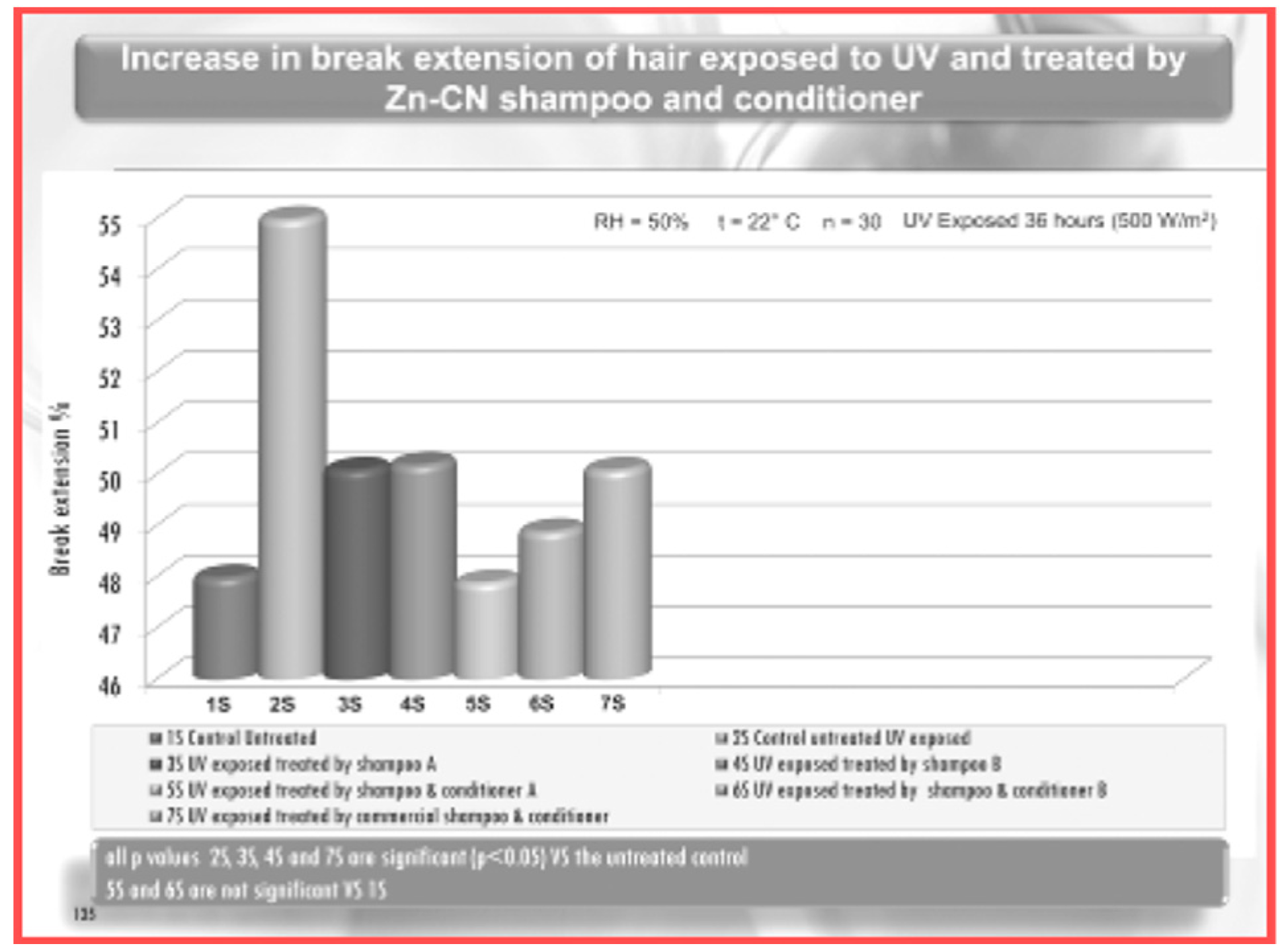
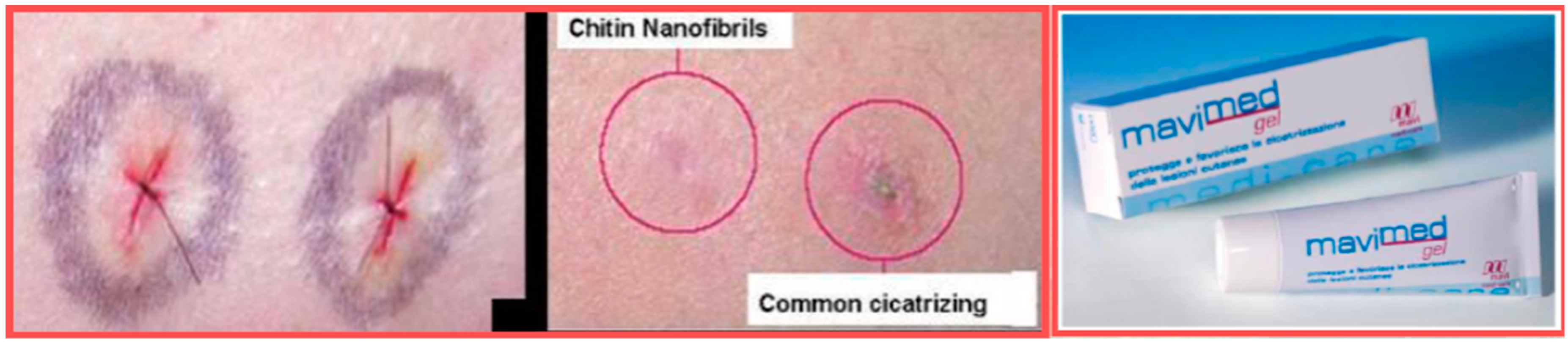
- Morganti, P.; Fabrizi, G.; Guarneri, F.; Palombo, M.; Palombo, P.; Cardillo, M.; Ruocco, E.; Del Ciotto, P.; Morganti, G. Repair Activity of Skin Barrier by Chitin-Nanofibrils Complexes. Sofw. J. 2011, 137, 10–26. [Google Scholar]
- Morganti, P.; Morganti, G.; Morganti, A. Transforming Nanostructured Chitin from Crustacean Waste into Benefical Health Products: A Must of our Society. Nanotechnol. Sci. Appl. 2011, 4, 123–129. [Google Scholar] [CrossRef]
- Jim, J. The Facial Mask Remains a Covered Beauty Item among Urban Chinese Consumers; Mintel Global New Product Database: London, UK, 2017. [Google Scholar]





© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Morganti, P.; Coltelli, M.-B. A New Carrier for Advanced Cosmeceuticals. Cosmetics 2019, 6, 10. https://doi.org/10.3390/cosmetics6010010
Morganti P, Coltelli M-B. A New Carrier for Advanced Cosmeceuticals. Cosmetics. 2019; 6(1):10. https://doi.org/10.3390/cosmetics6010010
Chicago/Turabian StyleMorganti, Pierfrancesco, and Maria-Beatrice Coltelli. 2019. "A New Carrier for Advanced Cosmeceuticals" Cosmetics 6, no. 1: 10. https://doi.org/10.3390/cosmetics6010010
APA StyleMorganti, P., & Coltelli, M.-B. (2019). A New Carrier for Advanced Cosmeceuticals. Cosmetics, 6(1), 10. https://doi.org/10.3390/cosmetics6010010





