Biobased Anti-Adhesive Marine Coatings from Polyhydroxyalkanoates and Polysaccharides
Abstract
1. Introduction
2. Coatings Based on Poly(hydroxyalkanoates)
2.1. Extraction and Characterization of PHAs
2.2. Advantages and Applications of PHAs
2.3. Anti-Adhesive PHA Coatings
3. Coatings Based on Polysaccharides
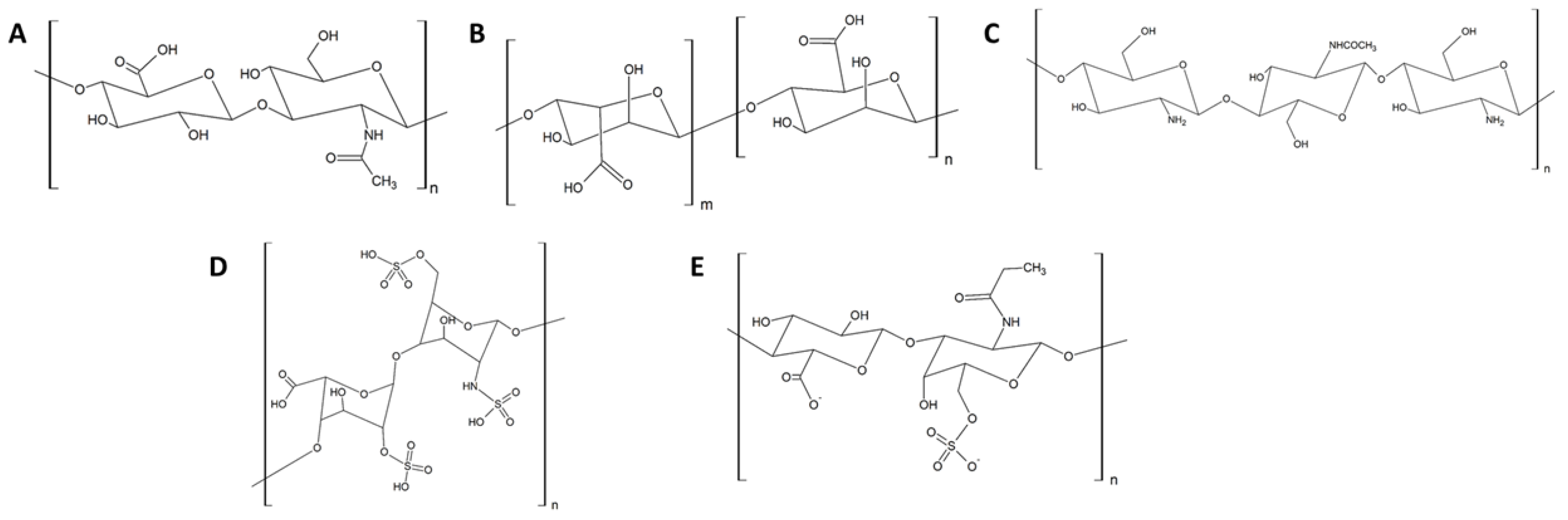
3.1. Anti-Adhesive Polysaccharides
3.1.1. Coordination Bonds
3.1.2. Self-Assembled Monolayers
3.1.3. Covalent Attachment
- Mussel-inspired technology: Synthetic oligomers and polymers inspired by the multifunctional tethering system (byssus) of the common mussel (genus Mytilus) emerged in the 1980s and have since become a very active research domain within the wider bioinspired and biomimetic materials arena [92]. Studies on the remarkable ability of mussels to attach onto virtually any surface underwater has unveiled L-DOPA (3,4-dihydroxy-L-phenylalanine) as the key molecule responsible for this versatile adhesion. Consequently, DOPA or cathecol were conjugated with functional polymers to impart the adhesive properties of mussels to them [69]. An HP-doped coating was developed by combining polysaccharide with mussel-inspired technology [69]. Cathecol-grafted HP was synthetized by an amide bond forming reaction, then the adhesive HP was utilized for surface modification of stainless-steel substrates. A reduction of approximately 75% in the adhesion of A. coffeaformis and N. perminuta was observed and ascribed to the hydration and electrostatic repulsion between the negatively charged HP and the diatom cell walls [92].
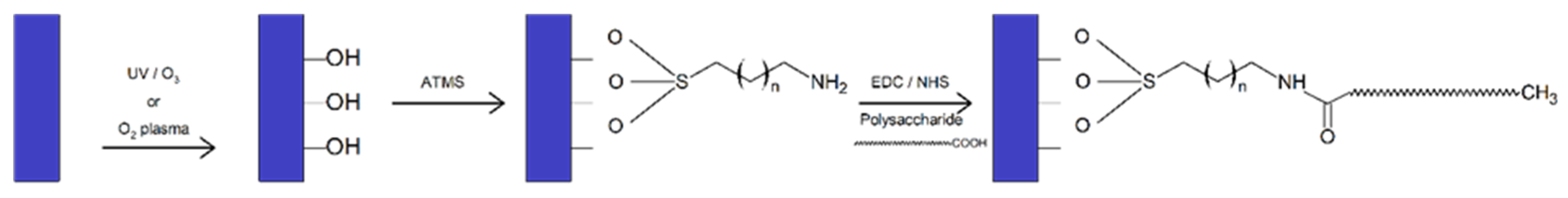
- Silanization: The functionalization of surfaces by a self-assembled aminosilane monolayer to introduce an amino function onto a substrate (titanium plates, silicon wafers, and glass slides) has enabled the covalent grafting of polysaccharides [93]. Covalent binding of polysaccharides was achieved by conversion of carboxylic acids into esters functional groups via reaction with N-hydroxysccinimide (NHS) and carbodiimide (EDC) and formation of amide bonds with amine groups on the surface [93] (Figure 5).
3.1.4. Polyelectrolyte Multilayers
3.1.5. Polysaccharide–Silane Hybrid Materials
3.2. The Particular Case of Exopolysaccharides
3.2.1. Antiadhesive Activity of Bacterial Exopolysaccharides
3.2.2. Antiadhesive EPS Coatings
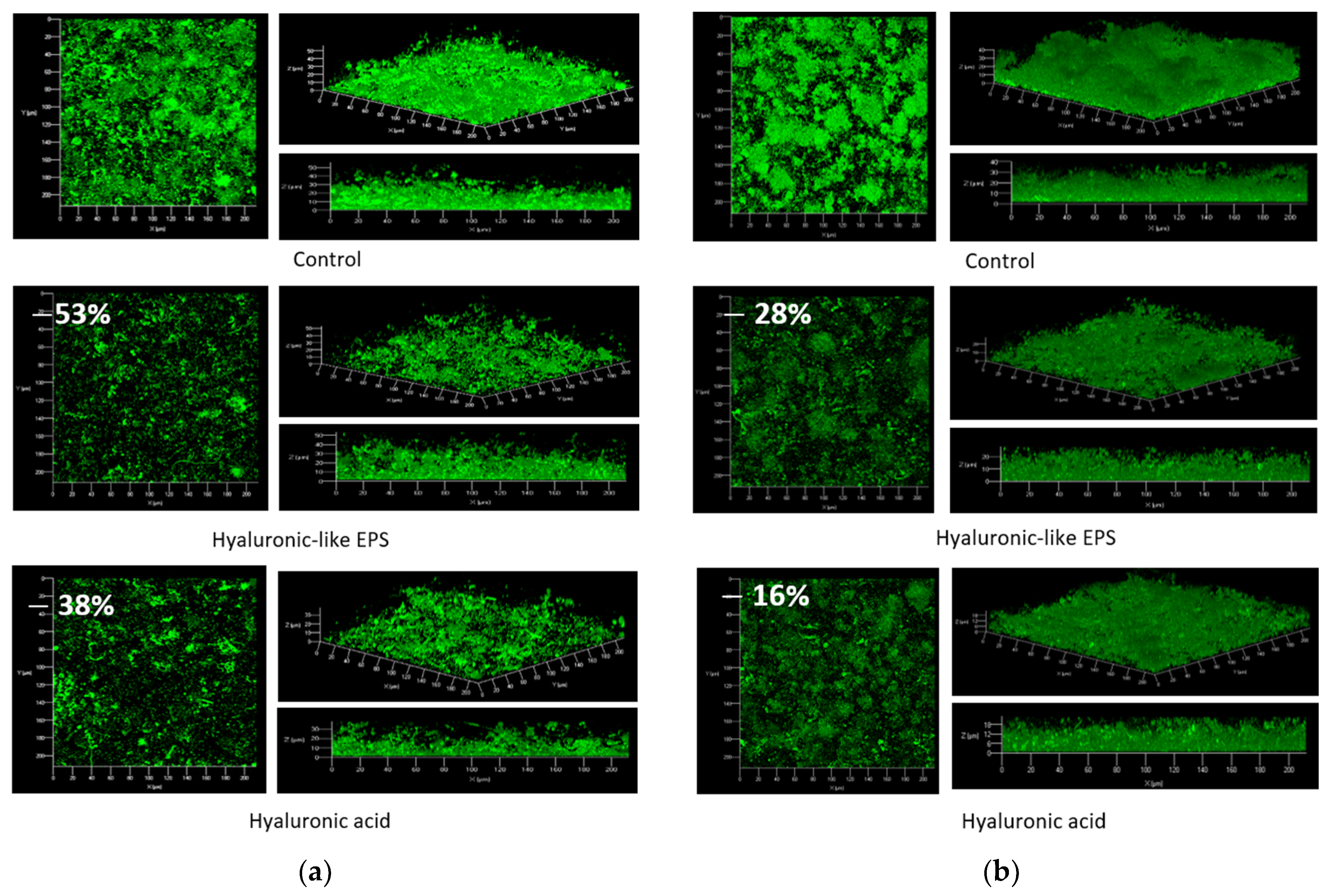
3.2.3. Marine Paints Based on EPSs
4. Conclusions and Future Trends
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Wahl, M. Marine Epibiosis.1. Fouling and antifouling—Some basic aspects. Mar. Ecol. Prog Ser. 1989, 58, 175–189. Available online: https://www.jstor.org/stable/24842178. (accessed on 11 February 2023). [CrossRef]
- Nurioglu, A.G.; Catarina, A.; Esteves, C.; de With, G. Non-toxic, non-biocide-release antifouling coatings based on molecular structure design for marine applications. J. Mater. Chem. B 2015, 3, 6547–6570. [Google Scholar] [CrossRef] [PubMed]
- Farkas, A.; Song, S.; Degiuli, N.; Martić, I.; Demirel, Y.K. Impact of biofilm on the ship propulsion characteristics and the speed reduction. Ocean. Eng. 2020, 1999, 107033. [Google Scholar] [CrossRef]
- Jin, H.; Tian, L.; Bing, W.; Zhao, J.; Ren, L. Bioinspired marine antifouling coatings: Status, prospects, and future. Prog. Mater. Sci. 2022, 124, 100889. [Google Scholar] [CrossRef]
- Donelly, B.; Sammut, K.; Tang, Y. Materials selection for antifouling systems in marine structures. Molecules 2022, 27, 3408. [Google Scholar] [CrossRef]
- Eduok, U.; Faye, O.; Szpunar, J. Recent developments and applications of protective silicone coatings: A review of PDMS functional materials. Prog. Org. Coat. 2017, 111, 124–163. [Google Scholar] [CrossRef]
- Amara, I.; Miled, W.; Ben Slama, R.; Ladhari, N. Antifouling processes and toxicity effects of antifouling paints on marine environment. A review. Env. Toxicol. Pharmacol. 2018, 57, 115–130. [Google Scholar] [CrossRef]
- de Campos, B.G.; Figueiredo, J.; Perina, F.; Abessa, D.M.D.S.; Loureiro, S.; Martins, R. Occurrence, effects and environmental risk of antifouling biocides (EU PT 21); Are marine ecostyems threatened? Crit. Rev. Envir. Sci. Technol. 2022, 52, 3179–3210. [Google Scholar] [CrossRef]
- Murthy, P.S.; Venugopalan, V.; Krishna Mohan, V.P.; Nachariah, Y.V.; Das, A.; Venkatnarayanan, S.; Stahya, S.; Subba Rao, T. Adavancements and modifications to polydimethylsiloxane foul release antifouling coatings. In A Treatise on Corrosiion Sciene, Engineering and Technology; Indian Institute of Metals Series; Springer: Singapore, 2022. [Google Scholar] [CrossRef]
- Holberg, S.; Losada, R.; Blaikie, F.; Hanse, H.H.W.B.; Soreau, S.; Onderwater, R.C.A. Hydrophilic silicone coatings as fouling release: Simple synthesis, comparison to commercial, marine coatings and application on fresh water-cooled heat exchangers. Mater. Today Commun. 2022, 22, 100750. [Google Scholar] [CrossRef]
- Hu, P.; Xie, Q.; Ma, C.; Zhang, G. Silicone-Based Fouling-Release Coatings for Marine Antifouling. Langmuir 2020, 36, 2170–2183. [Google Scholar] [CrossRef]
- Lu, G.; Tian, S.; Li, J.; Xu, X.; Liu, S.; Pu, J. Fabrication of bio-based amphiphilic hydrogel coating with excellent antifouling and mechanical properties. Chem. Eng. J. 2021, 409, 128134. [Google Scholar] [CrossRef]
- Qiu, H.; Feng, K.; Gapeeva, A.; Meurisch, K.; Kaps, S.; Li, X.; Yu, L.; Mishra, Y.K.; Adelung, R.; Baum, M. Functional polymer materials for modern marine biofouling control. Prog. Polym. Sci. 2022, 127, 101516. [Google Scholar] [CrossRef]
- Kuliasha, C.A.; Finlay, J.A.; Franco, S.C.; Clare, A.S.; Stafslien, S.J.; Brennan, A.B. Marine anti-biofouling efficacy of amphiphilic poly(coacrylate) grafted PDMSe: Effect of graft molecular weight. Biofouling 2017, 33, 252–267. [Google Scholar] [CrossRef]
- Sakala, G.P.; Reches, M. Peptide-Based Approaches to Fight Biofouling. Adv. Mater. Interfaces 2018, 5, 1800073. [Google Scholar] [CrossRef]
- Barry, M.E.; Davidson, E.C.; Zhang, C.; Patterson, A.L.; Yu, B.; Leonardi, A.K.; Duzen, N.; Malaviya, K.; Clarke, J.L.; Finlay, J.A.; et al. The Role of Hydrogen Bonding in Peptoid-Based Marine Antifouling Coatings. Macromolecules 2019, 52, 1287–1295. [Google Scholar] [CrossRef]
- Portier, É.; Azemar, F.; Benkhaled, B.T.; Bardeau, J.-F.; Faÿ, F.; Réhel, K.; Lapinte, V.; Linossier, I. Poly(oxazoline) for the design of amphiphilic silicone coatings. Prog. Org. Coat. 2021, 153, 106116. [Google Scholar] [CrossRef]
- Jensen, M.J.; Peel, A.; Horne, R.; Chamberlain, J.; Xu, L.; Hansen, M.R.; Guymon, C.A. Antifouling and Mechanical Properties of Photografted Zwitterionic Hydrogel Thin-Film Coatings Depend on the Cross-Link Density. ACS Biomater. Sci. Eng. 2021, 7, 4494–4502. [Google Scholar] [CrossRef]
- Aktij, S.A.; Taghipour, A.; Rahimpour, A.; Mollahosseini, A.; Tiraferri, A. A critical review on ultrasonic-assisted fouling control and cleaning of fouled membranes. Ultrasonics 2020, 108, 106228. [Google Scholar] [CrossRef]
- Xie, L.; Hong, F.; He, C.; Ma, L.; Liu, J.; Zang, J.; Wu, C. Coatings with a self-generating hydrogel surface for antifouling. Polymer 2011, 52, 3738–3744. [Google Scholar] [CrossRef]
- Murosaki, T.; Ahmed, N.; Gong, J.P. Antifouling properties of hydrogels. Sci. Technol. Adv. Mater. 2011, 12, 064706. [Google Scholar] [CrossRef]
- Su, X.; Hao, D.; Xu, X.; Guo, X.; Li, Z.; Jiang, L. Hydrophilic/hydrophobic heterogeneity anti-biofouling hydrogels wirh well-regulated rehydration. ACS Appl. Mater. Interfaces 2020, 12, 25316–25323. [Google Scholar] [CrossRef] [PubMed]
- Chiang, H.Y.; Pan, J.; Ma, C.; Qian, P.Y. Combining a bio-based polymer and a natural antifoulant into an eco-friendly antifouling coating. Biofouling 2020, 36, 200–2009. [Google Scholar] [CrossRef] [PubMed]
- Simon-Colin, C.; Gouin, C.; Lemencko, P.; Schmitt, S.; Senant, A.; Kervarec, N.; Guezennec, J. Biosynthesis and characterization of polyhydroxyalkanoates by Pseudomonas guezennei from alkanoates and glucose. Int. J. Biol. Macromol. 2012, 51, 1063–1069. [Google Scholar] [CrossRef] [PubMed]
- Castro-Sowinski, S.; Burdman, S.; Matan, O.; Okon, Y. Natural Functions of Bacterial Polyhydroxyalkanoates. In Plastics from Bacteria, Chen, G.G.-Q., Eds.; Springer: Berlin/Heidelberg, Germany, 2010; pp. 39–61. [Google Scholar] [CrossRef]
- Kadouri, D.; Jurkevitch, E.; Okon, Y.; Castro-Sowinski, S. Ecological and Agricultural Significance of Bacterial Polyhydroxyalkanoates. Crit. Rev. Microbiol. 2005, 31, 55–67. [Google Scholar] [CrossRef] [PubMed]
- Obruca, S.; Sedlacek, P.; Slaninova, E.; Fritz, I.; Daffert, C.; Meixner, K.; Sedrlova, Z.; Koller, M. Novel unexpected functions of PHA granules. Appl. Microbiol. Biotechnol. 2020, 104, 4795–4810. [Google Scholar] [CrossRef]
- Prieto, M.A.; Bühler, B.; Jung, K.; Witholt, B.; Kessler, B. PhaF, a Polyhydroxyalkanoate-Granule-Associated Protein of Pseudomonas oleovorans GPo1 Involved in the Regulatory Expression System for pha Genes. J. Bacteriol. 1999, 181, 858–868. [Google Scholar] [CrossRef]
- Bachmann, B.M.; Seebach, D. Investigation of the Enzymatic Cleavage of Diastereomeric Oligo(3-hydroxybutanoates) Containing Two to Eight HB Units. A Model for the Stereoselectivity of PHB Depolymerase from Alcaligenes faecalis T1. Macromolecules 1999, 32, 1777–1784. [Google Scholar] [CrossRef]
- Hocking, P.J.; Marchessault, R.H.; Timmins, M.R.; Lenz, R.W.; Fuller, R.C. Enzymatic Degradation of Single Crystals of Bacterial and Synthetic Poly(β-hydroxybutyrate). Macromolecules 1996, 29, 2472–2478. [Google Scholar] [CrossRef]
- Huisman, G.W.; de Leeuw, O.; Eggink, G.; Witholt, B. Synthesis of poly-3-hydroxyalkanoates is a common feature of fluorescent pseudomonads. Appl. Environ. Microbiol 1989, 55, 1949–1954. [Google Scholar] [CrossRef]
- Lageveen, R.G.; Huisman, G.W.; Preusting, H.; Ketelaar, P.; Eggink, G.; Witholt, B. Formation of Polyesters by Pseudomonas oleovorans: Effect of Substrates on Formation and Composition of Poly-( R )-3-Hydroxyalkanoates and Poly-(R)-3-Hydroxyalkenoates. Appl. Environ. Microbiol. 1988, 54, 2924–2932. [Google Scholar] [CrossRef]
- Timm, A.; Steinbüchel, A. Formation of polyesters consisting of medium-chain-length 3-hydroxyalkanoic acids from gluconate by Pseudomonas aeruginosa and other fluorescent pseudomonads. Appl. Environ. Microbiol. 1990, 56, 3360–3367. [Google Scholar] [CrossRef]
- Steinbuchel, A.; Valentin, H.E. Diversity of bacterial polyhydroxyalkanoic acids. FEMS Microbiol. Lett. 1995, 128, 219–228. [Google Scholar] [CrossRef]
- Bear, M.-M.; Leboucher-Durand, M.-A.; Langlois, V.; Lenz, R.W.; Goodwin, S.; Guérin, P. Bacterial poly-3-hydroxyalkenoates with epoxy groups in the side chains. React. Funct. Polym. 1997, 34, 65–77. [Google Scholar] [CrossRef]
- Kurth, N.; Renard, E.; Brachet, F.; Robic, D.; Guerin, P.; Bourbouze, R. Poly(3-hydroxyoctanoate) containing pendant carboxylic groups for the preparation of nanoparticles aimed at drug transport and release. Polymer 2002, 43, 1095–1101. [Google Scholar] [CrossRef]
- Stigers, D.J.; Tew, G.N. Poly(3-hydroxyalkanoate)s Functionalized with Carboxylic Acid Groups in the Side Chain. Biomacromolecules 2003, 4, 193–195. [Google Scholar] [CrossRef]
- Arkin, A.H.; Hazer, B.; Borcakli, M. Chlorination of Poly(3-hydroxy alkanoates) Containing Unsaturated Side Chains. Macromolecules 2000, 33, 3219–3223. [Google Scholar] [CrossRef]
- Babinot, J.; Renard, E.; Langlois, V. Preparation of Clickable Poly(3-hydroxyalkanoate) (PHA): Application to Poly(ethylene glycol) (PEG) Graft Copolymers Synthesis: Preparation of Clickable Poly(3-hydroxyalkanoate). Macromol. Rapid Commun. 2010, 31, 619–624. [Google Scholar] [CrossRef]
- Eroğlu, M.S.; Hazer, B.; Ozturk, T.; Caykara, T. Hydroxylation of pendant vinyl groups of poly(3-hydroxy undec-10-enoate) in high yield. J. Appl. Polym. Sci. 2005, 97, 2132–2139. [Google Scholar] [CrossRef]
- Lee, M.Y.; Park, W.H.; Lenz, R.W. Hydrophilic bacterial polyesters modified with pendant hydroxyl groups. Polymer 2000, 41, 1703–1709. [Google Scholar] [CrossRef]
- Tortajada, M.; Ferreira da Silva, L.; Auxiliadora Prieto, M. Second-generation functionalized mediumchain- length polyhydroxyalkanoates: The gateway to high-value bioplastic applications. Int. Microbiol. 2013, 16, 1–15. [Google Scholar] [CrossRef]
- Reddy, V.U.N.; Ramanaiah, S.V.; Reddy, M.V.; Chang, Y.C. Review of the Developments of Bacterial Medium-Chain-Length Polyhydroxyalkanoates (mcl-PHAs). Bioengineering 2022, 9, 225. [Google Scholar] [CrossRef] [PubMed]
- Gregory, D.A.; Taylor, C.S.; Fricker, A.T.; Asare, E.; Tetali, S.S.; Haycock, J.W.; Roy, I. Polyhydroxyalkanoates and their advances for biomedical applications. Trends Mol. Med. 2022, 28, 331–342. [Google Scholar] [CrossRef] [PubMed]
- Vicente, D.; Proença, D.N.; Morais, P.V. The Role of Bacterial Polyhydroalkanoate (PHA) in a Sustainable Future: A Review on the Biological Diversity. Int. J. Environ. Res. Public Health 2023, 20, 2959. [Google Scholar] [CrossRef] [PubMed]
- Ashby, R.D.; Solaiman, D.K.Y.; Foglia, T.A. Synthesis of Short-/Medium-Chain-Length Poly(hydroxyalkanoate) Blends by Mixed Culture Fermentation of Glycerol. Biomacromolecules 2005, 6, 2106–2112. [Google Scholar] [CrossRef] [PubMed]
- Koller, M.; Bona, R.; Braunegg, G.; Hermann, C.; Horvat, P.; Kroutil, M.; Martinz, J.; Neto, J.; Pereira, L.; Varila, P. Production of Polyhydroxyalkanoates from Agricultural Waste and Surplus Materials. Biomacromolecules 2005, 6, 561–565. [Google Scholar] [CrossRef]
- Alves, A.A.; Siqueira, E.C.; Barros, M.P.S.; Silva, P.E.C.; Houllou, L.M. Polyhydroxyalkanoates: A review of microbial production and technology application. Int. J. Environ. Sci. Technol. 2023, 20, 3409–3420. [Google Scholar] [CrossRef]
- Tsuge, T. Metabolic improvements and use of inexpensive carbon sources in microbial production of polyhydroxyalkanoates. J. Biosci. Bioeng. 2002, 94, 579–584. [Google Scholar] [CrossRef]
- Chathalingath, N.; Kingsly, J.S.; Gunasekar, A. Biosynthesis and biodegradation of poly(3-hydroxybutyrate) from Priestiaflexa; A promising mangrove halophyte towards the development of sustainable eco-friendly bioplastics. Microbiol. Res. 2023, 267, 127270. [Google Scholar] [CrossRef]
- Ohura, T.; Aoyagi, Y.; Takagi, K.; Yoshida, Y.; Kasuya, K.; Doi, Y. Biodegradation of poly(3-hydroxyalkanoic acids) fibers and isolation of poly(3-hydroxybutyric acid)-degrading microorganisms under aquatic environments. Polym. Degrad. Stab. 1999, 63, 23–29. [Google Scholar] [CrossRef]
- Chou, H.; Chen, C.; Huang, C.; Wang, H.; Hsiung, Y.-C.; Liang, C.-H.; Ou, C.-M.; Guo, G. Screening potential polyhydroxyalkanoate-producing bacteria from wastewater sludge. Arch. Microbiol. 2023, 205, 120. [Google Scholar] [CrossRef]
- Sudesh, K.; Abe, H.; Doi, Y. Synthesis, structure and properties of polyhydroxyalkanoates: Biological polyesters. Prog. Polym. Sci. 2000, 25, 1503–1555. [Google Scholar] [CrossRef]
- Che, L.; Jin, W.; Zhou, X.; Han, W.; Chen, Y.; Chen, C.; Jiang, G. Current Status and future perspectives on the biological production of polyhydroxyalkanoates. Asia-Pac. J. Chem. Eng. 2023, e2899. [Google Scholar] [CrossRef]
- Rathbone, S.; Furrer, P.; Lübben, J.; Zinn, M.; Cartmell, S. Biocompatibility of polyhydroxyalkanoate as a potential material for ligament and tendon scaffold material: Biocompatibility of Polyhydroxyalkanoate. J. Biomed. Mater. Res. 2010, 93A, 1391–1403. [Google Scholar] [CrossRef]
- Srivastava, S.K.; Tripathi, A.D. Effect of saturated and unsaturated fatty acid supplementation on bio-plastic production under submerged fermentation. Biotech 2013, 3, 389–397. [Google Scholar] [CrossRef]
- Ladhari, S.; Vu, N.N.; Boisvertn, C.; Saidi, A.; Nguyen-Tri, P. Recent Development of Polyhydroxyalkanoates (PHA)-based Materials for Antibacterial applications: A review. ACS Appl. Bio. Mater. 2023. [Google Scholar] [CrossRef]
- Prakash, P.; Lee, W.-H.; Loo, C.-Y.; Wong, H.S.J.; Parumasivam, T. Advances in Polyhydroxyalkanoate Nanocarriers for Effective Drug Delivery: An Overview and Challenges. Nanomaterials 2022, 12, 175. [Google Scholar] [CrossRef]
- Chen, G.-Q.; Wu, Q. The application of polyhydroxyalkanoates as tissue engineering materials. Biomaterials 2005, 26, 6565–6578. [Google Scholar] [CrossRef]
- Phuegyod, S.; Pramual, S.; Wattanavichean, N.; Assawajaruwan, S.; Amornsakchai, T.; Sukho, P.; Svasti, J.; Surarit, R.; Niamsiri, N. Microbial Poly(hydroxybutyrate-cohydroxyvalerate) Scaffold for Periodontal Tissue Engineering. Polymers 2023, 15, 855. [Google Scholar] [CrossRef]
- Zhang, J.; Shishatskaya, E.I.; Volova, T.G.; da Silva, L.F.; Chen, G.-Q. Polyhydroxyalkanoates (PHA) for therapeutic applications. Mater. Sci. Eng. C 2018, 86, 144–150. [Google Scholar] [CrossRef]
- Guennec, A.; Brelle, L.; Balnois, E.; Linossier, I.; Renard, E.; Langlois, V.; Faÿ, F.; Chen, G.Q.; Simon-Colin, C.; Vallée-Réhel, K. Antifouling properties of amphiphilic poly(3-hydroxyalkanoate): An environmentally-friendly coating. Biofouling 2021, 37, 894–910. [Google Scholar] [CrossRef]
- Mauclaire, L.; Brombacher, E.; Bünger, J.D.; Zinn, M. Factors controlling bacterial attachment and biofilm formation on medium-chain-length polyhydroxyalkanoates (mcl-PHAs). Colloids Surf. B Biointerfaces 2010, 76, 104–111. [Google Scholar] [CrossRef] [PubMed]
- Piarali, S.; Marlinghaus, L.; Viebahn, R.; Lewis, H.; Ryadnov, M.G.; Groll, J.; Salber, J.; Roy, I. Activated Polyhydroxyalkanoate Meshes Prevent Bacterial Adhesion and Biofilm Development in Regenerative Medicine Applications. Front. Bioeng. Biotechnol. 2020, 8, 442. [Google Scholar] [CrossRef] [PubMed]
- Che, X.M.; Wei, D.X.; Chen, G.Q. Superhydrophobic Polyhydroxyalkanoates: Preparation and applications. Biomacromolecules 2019, 20, 618–624. [Google Scholar] [CrossRef] [PubMed]
- Brelle, L.; Faÿ, F.; Ozturk, T.; Didier, N.; Renard, E.; Langlois, V. Hydrogel based on Polyhydroxyalkanoate Sulfonate: Control of the Swelling Rate by the Ionic Group content. Biomacromolecules 2023, in press. [Google Scholar] [CrossRef]
- Arias, S.L.; Devorkin, J.; Civantos, A.; Allain, J.P. Escherichia coli Adhesion and Biofilm Formation on Polydimethylsiloxane are Independent of Substrate Stiffness. Langmuir 2021, 37, 16–25. [Google Scholar] [CrossRef]
- Kolewe, K.W.; Peyton, S.R.; Schiffman, J.D. Fewer Bacteria Adhere to Softer Hydrogels. ACS Appl. Mater. Interfaces 2015, 7, 19562–19569. [Google Scholar] [CrossRef]
- Kim, S.; Ko, S.; Kang, S.M. Adhesive heparin coating for marine antifouling applications. Macromol. Res. 2016, 24, 645–649. [Google Scholar] [CrossRef]
- Junter, G.-A.; Thébault, P.; Lebrun, L. Polysaccharide-based antibiofilm surfaces. Acta Biomater. 2016, 30, 13–25. [Google Scholar] [CrossRef]
- Wen, C.; Guo, H.; Yang, J.; Li, Q.; Zhang, X.; Sui, X.; Cao, M.; Zhang, L. Zwitteronic hydrogel coated superhydrophilic hierarchical antifouling floater enables unimpeded interfacial steam generation and multi-contamination resistance in complex conditions. Chem. Eng. J. 2021, 421, 130344. [Google Scholar] [CrossRef]
- Yu, W.; Wang, Y.; Gnutt, P.; Wanka, R.; Krause, L.M.K.; Finlay, J.A.; Clare, S.A.; Rosenhaln, A. Layer-by-layer deposited hybrid polymer Coatings based on polysaccharides and zwitteronic silanes with marine antifouling properties. ACS Appl. Bio. Mater. 2021, 4, 2385–2397. [Google Scholar] [CrossRef]
- Aghajani, M.; Esmaeili, F. Anti-biofouling assembly strategies for protein & cell repellent surfaces: A mini-review. J. Biomater. Sci. Polym. Ed. 2021, 32, 1770–1789. [Google Scholar] [CrossRef]
- Cao, X.; Pettit, M.E.; Conlan, S.L.; Wagner, W.; Ho, A.D.; Clare, A.S.; Callow, J.A.; Callow, M.E.; Grunze, M.; Rosenhaln, A. Resistance of polysachharide coatings to proteins, hematopoietic cells, and marine organisms. ACS Biomacromol. 2009, 10, 907–915. [Google Scholar] [CrossRef]
- Bauer, S.; Arpa-Sancet, M.P.; Finlay, J.A.; Callow, M.E.; Callow, J.A.; Rosenhahn, A. Adhesion of Marine Fouling Organisms on Hydrophilic and Amphiphilic Polysaccharides. Langmuir 2013, 29, 4039–4047. [Google Scholar] [CrossRef]
- Shutava, T.G.; Livanovich, K.S.; Sharamet, A.A. Layer-by-layer films of polysaccharides modified with polyethylene glycol and dextran. Colloids Surf. B Biointerfaces 2019, 173, 412–420. [Google Scholar] [CrossRef]
- Mohan, T.; Cas, A.; Bracic, M.; Plohl, O.; Vesel, A.; Rupnik, M.; Zemljic, L.F.; Rebol, J. Highly protein reppellent and antiadhesive polysaccharide biomaterial coating for urinary catheter applications. ACS Biomater. Sci. Eng. 2019, 5, 5825–5832. [Google Scholar] [CrossRef]
- Yazdi, M.K.; Sajadi, S.M.; Seidi, F.; Rabiee, N.; Fatahi, Y.; Rabiee, M.; Midhun Dominic, C.D.; Zarrintaj, P.; Formela, K.; Reza Saeb, M.; et al. Clockable polysaccharides for biomedical applications: A comprehensive review. Prog. Polym. Sci. 2022, 133, 101590. [Google Scholar] [CrossRef]
- Lee, Y.E.; Kim, H.; Seo, C.; Park, T.; Lee, K.B.; Yoo, S.Y.; Hong Tae Kim, J.; Lee, J. Marine polysaccharides: Therapeutic efficacy and biomedical applications. Arch. Pharm. Res. 2017, 40, 1006–1020. [Google Scholar] [CrossRef]
- Ruocco, N.; Costantini, S.; Guareniello, S.; Costantini, M. Polysaccharides from the marine environment with pharmacological, cosmeceutical and nutraceutical potential. Molecules 2016, 21, 551. [Google Scholar] [CrossRef]
- Murugappan, V.; Muthadhi, A. Studies on the influence of alginate as a natural polymer in mechanical and long-lasting properties of concrete-review. Mater. Proc. 2022, 65, 839–845. [Google Scholar] [CrossRef]
- Jiménez-Gomez, C.; Cecilia, J.A. Chitosan: A natural Biopolymer with a wide and varied range of applications. Molecules 2020, 25, 3981. [Google Scholar] [CrossRef]
- Crini, G. Historical review on chitin and chitosan biopolymers. Env. Chem Lett 2019, 17, 1623–1643. [Google Scholar] [CrossRef]
- Pokhrel, S.; Yadav, P.N. Functinalization of chitosan polymer and their application. J. Macromol. Sci. A 2019, 56, 450–475. [Google Scholar] [CrossRef]
- Jin, Y.; Zhu, Z.; Liang, L.; Zheng, O.; Wang, Y.; Guo, Y.; Zhu, K.; Mehmood, R.; Wang, B. A facile heparin/carboxymethyl chitosan coating mediated by polydopamine on implants for hemocompatibility and antibacterial properties. Appl. Surf. Sci. 2020, 528, 146539. [Google Scholar] [CrossRef]
- Drozd, N.; Lunkov, A.P.; Shagdarova, B.T.; Zhuikova, Y.V.; Il’ina, A.V.; Varlamov, V.P. Chitosan/heparin layer-by-layer coatings for improving thromboresistance of polyurethane. Surf. Interfaces 2022, 28, 101674. [Google Scholar] [CrossRef]
- Bauer, S.; Alles, M.; Arpa-Sancet, M.P.; Ralston, E.; Swain, G.W.; Aldred, N.; Clare, A.S.; Finlay, J.A.; Callow, M.E.; Callow, J.A.; et al. Resistance of Amphiphilic Polysaccharides against Marine Fouling Organisms. Biomacromolecules 2016, 17, 897–904. [Google Scholar] [CrossRef]
- Jakobi, V.; Schwarze, J.; Finlay, J.A.; Nolte, K.A.; Spöllmann, S.; Becker, H.-W.; Clare, A.S.; Rosenhahn, A. Amphiphilic Alginates for Marine Antifouling Applications. Biomacromolecules 2018, 19, 402–408. [Google Scholar] [CrossRef]
- Kim, D.; Kang, S.M. Red Algae-Derived Carrageenan Coatings for Marine Antifouling Applications. Biomacromolecules 2020, 21, 5086–5092. [Google Scholar] [CrossRef]
- Fyrner, T.; Lee, H.-H.; Mangone, A.; Ekblad, T.; Pettitt, M.E.; Callow, M.E.; Callow, J.A.; Conlan, S.L.; Mutton, R.; Clare, A.S.; et al. Saccharide-Functionalized Alkanethiols for Fouling-Resistant Self-Assembled Monolayers: Synthesis, Monolayer Properties, and Antifouling Behavior. Langmuir 2011, 27, 15034–15047. [Google Scholar] [CrossRef]
- Nugraha, R.; Finlay, J.A.; Hill, S.; Fyrner, T.; Yandi, W.; Callow, M.E.; Callow, J.A.; Ederth, T. Antifouling properties of oligo(lactose)-based self-assembled monolayers. Biofouling 2015, 31, 123–134. [Google Scholar] [CrossRef]
- Ederth, T.; Ekblad, T.; Pettitt, M.E.; Conlan, S.L.; Du, C.-X.; Callow, M.E.; Callow, J.A.; Mutton, R.; Clare, A.S.; D’Souza, F.; et al. Resistance of Galactoside-Terminated Alkanethiol Self-Assembled Monolayers to Marine Fouling Organisms. ACS Appl. Mater. Interfaces 2011, 3, 3890–3901. [Google Scholar] [CrossRef]
- Gadenne, V.; Lebrun, L.; Jouenne, T.; Thebault, P. Antiadhesive activity of ulvan polysaccharides covalently immobilized onto titanium surface. Colloids Surf. B Biointerfaces 2013, 112, 229–236. [Google Scholar] [CrossRef]
- Jeong, Y.; Yoo, J.S.; Kang, S.M. Marine Fouling Resistance of Ulvan-grafted Solid Surface. Bull. Korean Chem. Soc. 2018, 39, 1459–1462. [Google Scholar] [CrossRef]
- Gnanasampanthan, T.; Beyer, C.D.; Yu, W.; Karthäuser, J.F.; Wanka, R.; Spöllmann, S.; Becker, H.-W.; Aldred, N.; Clare, A.S.; Rosenhahn, A. Effect of Multilayer Termination on Nonspecific Protein Adsorption and Antifouling Activity of Alginate-Based Layer-by-Layer Coatings. Langmuir 2021, 37, 5950–5963. [Google Scholar] [CrossRef]
- Yu, W.; Koc, J.; Finlay, J.A.; Clarke, J.L.; Clare, A.S.; Rosenhahn, A. Layer-by-layer constructed hyaluronic acid/chitosan multilayers as antifouling and fouling-release coatings. Biointerphases 2019, 14, 051002. [Google Scholar] [CrossRef]
- Yu, W.; Wanka, R.; Finlay, J.A.; Clarke, J.L.; Clare, A.S.; Rosenhahn, A. Degradable hyaluronic acid/chitosan polyelectrolyte multilayers with marine fouling-release properties. Biofouling 2020, 36, 1049–1064. [Google Scholar] [CrossRef]
- Xu, G.; Liu, P.; Pranantyo, D.; Neoh, K.-G.; Kang, E.-T. Dextran- and Chitosan-Based Antifouling, Antimicrobial Adhesion, and Self-Polishing Multilayer Coatings from pH-Responsive Linkages-Enabled Layer-by-Layer Assembly. ACS Sustain. Chem. Eng. 2018, 6, 3916–3926. [Google Scholar] [CrossRef]
- Gnanasampanthan, T.; Karthäuser, J.F.; Spöllmann, S.; Wanka, R.; Becker, H.-W.; Rosenhahn, A. Amphiphilic Alginate-based layer-by-layer coatings exhibiting resistance against nonspecific protein adsorption and marine biofouling. ACS Appl. Mater. Interfaces 2022, 14, 16062–16073. [Google Scholar] [CrossRef]
- Lakhan, M.N.; Chen, R.; Liu, F.; Shar, A.H.; Soomro, I.A.; Chand, K.; Ahmed, M.; Hanan, A.; Khan, A.; Maitlo, A.A.; et al. Construction of antifouling marine coatings via layer-by-layer assembly of chitosan and acid siloxane resin. J. Polym. Res. 2023, 30, 136. [Google Scholar] [CrossRef]
- Wanka, R.; Koc, J.; Clarke, J.; Hunsucker, K.Z.; Swain, G.W.; Aldred, N.; Finlay, J.A.; Clare, A.S.; Rosenhahn, A. Sol–Gel-Based Hybrid Materials as Antifouling and Fouling-Release Coatings for Marine Applications. ACS Appl. Mater. Interfaces 2020, 12, 53286–53296. [Google Scholar] [CrossRef]
- Guezennec, J. Deep-sea hydrothermal vents: A new source of innovative bacterial exopolysaccharides of biotechnological interest? J. Ind. Microbiol. Biotechnol. 2002, 29, 204–208. [Google Scholar] [CrossRef]
- Freitas, F.; Torres, C.; Reis, M. Engineering aspects of microbial exopolysaccharide production. Bioresour. Technol. 2017, 245 Pt B, 1674–1683. [Google Scholar] [CrossRef]
- Delattre, C.; Pierre, G.; Laroche, C.; Michaud, P. Production, extraction and characterization of microalgal and cyanobacterial exopolysaccharides. Biotechnol. Adv. 2016, 34, 1159–1179. [Google Scholar] [CrossRef] [PubMed]
- Donot, F.; Fontana, A.; Baccou, J.C.; Schorr-Galindo, S. Microbial exopolysaccharides: Main examples of synthesis, excretion, genetics and extraction. Carbohydr. Polym. 2012, 87, 951–962. [Google Scholar] [CrossRef]
- Moscovici, M. Present and future medical applications of microbial exopolysaccharides. Front. Microbiol. 2015, 6, 1012. [Google Scholar] [CrossRef] [PubMed]
- Osińska-Jaroszuk, M.; Jarosz-Wilkołazka, A.; Jaroszuk-Ściseł, J.; Szałapata, K.; Nowak, A.; Jaszek, M.; Ozimek, E.; Majewska, M. Extracellular polysaccharides from Ascomycota and Basidiomycota: Production conditions, biochemical characteristics, and biological properties. World J. Microbiol. Biotechnol. 2015, 31, 1823–1844. [Google Scholar] [CrossRef]
- Rehm, B.H. Bacterial polymers: Biosynthesis, modifications and applications. Nature reviews. Microbiology 2010, 8, 578–592. [Google Scholar] [CrossRef]
- Rehm, B.H.; Moradali, M.F. (Eds.) Biopolymers for Biomedical and Biotechnological Applications; John Wiley & Sons: Weinheim, Germany, 2021. [Google Scholar]
- Freitas, F.; Alves, V.D.; Reis, M.A. Advances in bacterial exopolysaccharides: From production to biotechnological applications. Trends Biotechnol. 2011, 29, 388–398. [Google Scholar] [CrossRef]
- Papinutti, L. Effects of nutrients, pH and water potential on exopolysaccharides production by a fungal strain belonging to Ganoderma lucidum complex. Bioresour. Technol. 2010, 101, 1941–1946. [Google Scholar] [CrossRef]
- Barcelos, M.; Vespermann, K.; Pelissari, F.M.; Molina, G. Current status of biotechnological production and applications of microbial exopolysaccharides. Crit. Rev. Food Sci. Nutr. 2020, 60, 1475–1495. [Google Scholar] [CrossRef]
- Martin-Pastor, M.; Ferreira, A.S.; Moppert, X.; Nunes, C.; Coimbra, M.A.; Reis, R.L.; Guezennec, J.; Novoa-Carballal, R. Structure, rheology, and copper-complexation of a hyaluronan-like exopolysaccharide from Vibrio. Carbohydr. Polym. 2019, 222, 114999. [Google Scholar] [CrossRef]
- Raguenes, G.; Christen, R.; Guezennec, J.; Pignet, P.; Barbier, G. Vibrio diabolicus sp. nov., a new polysaccharide-secreting organism isolated from a deep-sea hydrothermal vent polychaete annelid, Alvinella pompejana. Int. J. Syst. Bacteriol. 1997, 47, 989–995. [Google Scholar] [CrossRef]
- Cambon-Bonavita, M.A.; Raguénès, G.; Jean, J.; Vincent, P.; Guezennec, J. A novel polymer produced by a bacterium isolated from a deep-sea hydrothermal vent polychaete annelid. J. Appl. Microbiol. 2002, 93, 310–315. [Google Scholar] [CrossRef]
- Concórdio-Reis, P.; Alves, V.D.; Moppert, X.; Guézennec, J.; Freitas, F.; Reis, M. Characterization and Biotechnological Potential of Extracellular Polysaccharides Synthesized by Alteromonas Strains Isolated from French Polynesia Marine Environments. Mar. Drugs 2021, 19, 522. [Google Scholar] [CrossRef]
- Sayem, S.A.; Manzo, E.; Ciavatta, L.; Tramice, A.; Cordone, A.; Zanfardino, A.; De Felice, M.; Varcamonti, M. Anti-biofilm activity of an exopolysaccharide from a sponge-associated strain of Bacillus licheniformis. Microb. Cell Factories 2011, 10, 74. [Google Scholar] [CrossRef]
- Spanò, A.; Laganà, P.; Visalli, G.; Maugeri, T.L.; Gugliandolo, C. In Vitro Antibiofilm Activity of an Exopolysaccharide from the Marine Thermophilic Bacillus licheniformis T14. Curr. Microbiol. 2016, 72, 518–528. [Google Scholar] [CrossRef]
- Muras, A.; Romero, M.; Mayer, C.; Otero, A. Biotechnological applications of Bacillus licheniformis. Crit. Rev. Biotechnol. 2021, 41, 609–627. [Google Scholar] [CrossRef]
- Jurášková, D.; Ribeiro, S.C.; Silva, C.C.G. Exopolysaccharides Produced by Lactic Acid Bacteria: From Biosynthesis to Health-Promoting Properties. Foods 2022, 11, 156. [Google Scholar] [CrossRef]
- Le Costaouëc, T.; Cérantola, S.; Ropartz, D.; Ratiskol, J.; Sinquin, C.; Colliec-Jouault, S.; Boisset, C. Structural data on a bacterial exopolysaccharide produced by a deep-sea Alteromonas macluodii strain. Carbohydr. Polym. 2012, 90, 49–59. [Google Scholar] [CrossRef]
- Rendueles, O.; Kaplan, J.B.; Ghigo, J.-M. Antibiofilm polysaccharides: Bacterial antibiofilm polysaccharides. Environ. Microbiol 2013, 15, 334–346. [Google Scholar] [CrossRef]
- Jha, N.; Madasamy, S.; Prasad, P.; Lakra, A.K.; Tilwani, Y.M.; Arul, V. Physico-chemical and functional characterization of polysaccharide purified from mangrove Rhizophora mucronata leaves having potent biological activity. S. Afr. J. Bot. 2022, 147, 659–669. [Google Scholar] [CrossRef]
- Malakar, C.; Deka, S.; Kalita, M.C. Role of Biosurfactants in Biofilm Prevention and Disruption. In Advancements in Biosurfactants Research; Aslam, R., Mobin, M., Aslam, J., Zehra, S., Eds.; Springer: Cham, Switzerland, 2023. [Google Scholar] [CrossRef]
- Guezennec, J.; Herry, J.M.; Kouzayha, A.; Bachere, E.; Mittelman, M.W.; Bellon Fontaine, M.N. Exopolysaccharides from unusual marine environments inhibit early stages of biofouling. Int. Biodeterior. Biodegrad. 2012, 66, 1–7. [Google Scholar] [CrossRef]
- Jiang, P.; Li, J.; Han, F.; Duan, G.; Lu, X.; Gu, Y.; Yu, W. Antibiofilm Activity of an Exopolysaccharide from Marine Bacterium Vibrio sp. QY101. PLoS ONE 2011, 6, e18514. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Li, Q.; Hao, D.; Jiang, D.; Luo, Y.; Liu, Y.; Zhao, Z. Production, Purification, and Antibiofilm Activity of a Novel Exopolysaccharide from Arthrobacter sp. B4. Prep. Biochem. Biotechnol. 2015, 45, 192–204. [Google Scholar] [CrossRef] [PubMed]
- Pradeepa Shetty, A.D.; Matthews, K.; Hegde, A.R.; Akshatha, B.; Mathias, A.B.; Mutalik, S.; Vidya, S.M. Multidrug resistant pathogenic bacterial biofilm inhibition by Lactobacillus plantarum exopolysaccharide. Bioact. Carbohydr. Diet. Fibre 2016, 8, 7–14. [Google Scholar] [CrossRef]
- Valle, J.; Re, S.D.; Henry, N.; Fontaine, T.; Balestrino, D.; Latour-Lambert, P.; Ghigo, J.-M. Broad-spectrum biofilm inhibition by a secreted bacterial polysaccharide. PNAS 2006, 103, 12558–12563. [Google Scholar] [CrossRef]
- Vishwakarma, J.; Sirisha, V.L. Unraveling the anti-biofilm potential of green algal sulfated polysaccharides against Salmonella enterica and Vibrio harveyi. Appl. Microbiol. Biotechnol. 2020, 104, 6299–6314. [Google Scholar] [CrossRef]
- Wu, S.; Liu, G.; Jin, W.; Xiu, P.; Sun, C. Antibiofilm and Anti-Infection of a Marine Bacterial Exopolysaccharide Against Pseudomonas aeruginosa. Front. Microbiol. 2016, 7, 102. [Google Scholar] [CrossRef]
- Champion, M.; Portier, E.; Vallée-Réhel, K.; Linossier, I.; Balnois, E.; Vignaud, G.; Moppert, X.; Hellio, C.; Faÿ, F. Anti-Biofilm Activity of a Hyaluronan-like Exopolysaccharide from the Marine Vibrio MO245 against Pathogenic Bacteria. Mar. Drugs 2022, 20, 728. [Google Scholar] [CrossRef]
- Su, D.; Bai, X.; He, X. Research progress on hydrogel materials and their antifouling properties. Eur. Polym. J. 2022, 181, 111665. [Google Scholar] [CrossRef]
- Liu, J.; Qu, S.; Suo, Z.; Yang, W. Functional hydrogel coatings. Natl. Sci. Rev. 2015, 8, nwaa254. [Google Scholar] [CrossRef]
- Gokaltun, A.; Kang, Y.B.; Yarmush, M.L.; Usta, O.B.; Asatekin, A. Simple surface modification of poly(dimethylsiloxane) via surface segregating smart polymers for biomicrofluidics. Sci. Rep. 2019, 9, 7377. [Google Scholar] [CrossRef]
- Guégan, C.; Garderes, J.; Le Pennec, G.; Gaillard, F.; Faÿ, F.; Linossier, I.; Herry, J.-M.; Fontaine, M.-N.B.; Réhel, K.V. Alteration of bacterial adhesion induced by the substrate stiffness. Colloids Surf. B Biointerfaces 2014, 114, 193–200. [Google Scholar] [CrossRef]
- Kolewe, K.W.; Zhu, J.; Mako, N.R.; Nonnenman, S.S.; Shiffman, J.D. Bacterial adhesion is affected by the thickness and stiffness of poly(ethylene glycol) hydrogels, ACS Appl. Mater. Interfaces 2018, 10, 2275–2281. [Google Scholar] [CrossRef]
- Finlay, J.A.; Callow, M.E.; Ista, L.K.; Lopez, G.P.; Callow, J.A. The Influence of Surface Wettability on the Adhesion Strength of Settled Spores of the Green Alga Enteromorpha and the Diatom Amphora. Integr. Comp. Biol. 2002, 42, 1116–1122. [Google Scholar] [CrossRef]
- Magin, C.M.; Finlay, J.A.; Clay, G.; Callow, M.E.; Callow, J.A.; Brennan, A.B. Antifouling Performance of cross-linked hydrogel: Refinement of an attachment model. Biomacromolecules 2011, 12, 915–922. [Google Scholar] [CrossRef]
- Rajasree, V.; Satheesh, S.; Vincent, S.P. Antifouling activity of a marine epibiotic bacterium from the seaweed sargassum wightii. Thalassas 2012, 28, 37–43. [Google Scholar]
- Siqueira Melo, R.; Brasil, S.; Carvalho, L.; Limaverde Filho, A.; Pereira, C. Assessment of the Antifouling Effect of Exopolysaccharides Incorporated into Copper Oxide-Based Organic Paint. Int. J. Electrochem. Sci. 2016, 11, 7750. [Google Scholar] [CrossRef]
- Koc, J.; Simovich, T.; Schönemann, E.; Chilkoti, A.; Gardner, H.; Swain, G.W.; Hunsucker, K.; Laschewsky, A.; Rosenhahn, A. Sediment challenge to promising ultra-low fouling hydrophilic surfaces in the marine environment. Biofouling 2019, 35, 454–462. [Google Scholar] [CrossRef]
- Zhao, C.; Zhou, L.; Chiao, M.; Yang, W. Antibacterial hydrogel coating: Strategies in surface chemistry. Adv. Colloid Interface Sci. 2020, 285, 102280. [Google Scholar] [CrossRef]




Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Faÿ, F.; Champion, M.; Guennec, A.; Moppert, X.; Simon-Colin, C.; Elie, M. Biobased Anti-Adhesive Marine Coatings from Polyhydroxyalkanoates and Polysaccharides. Coatings 2023, 13, 766. https://doi.org/10.3390/coatings13040766
Faÿ F, Champion M, Guennec A, Moppert X, Simon-Colin C, Elie M. Biobased Anti-Adhesive Marine Coatings from Polyhydroxyalkanoates and Polysaccharides. Coatings. 2023; 13(4):766. https://doi.org/10.3390/coatings13040766
Chicago/Turabian StyleFaÿ, Fabienne, Marie Champion, Alexandra Guennec, Xavier Moppert, Christelle Simon-Colin, and Mathieu Elie. 2023. "Biobased Anti-Adhesive Marine Coatings from Polyhydroxyalkanoates and Polysaccharides" Coatings 13, no. 4: 766. https://doi.org/10.3390/coatings13040766
APA StyleFaÿ, F., Champion, M., Guennec, A., Moppert, X., Simon-Colin, C., & Elie, M. (2023). Biobased Anti-Adhesive Marine Coatings from Polyhydroxyalkanoates and Polysaccharides. Coatings, 13(4), 766. https://doi.org/10.3390/coatings13040766






