Pharmacist-Led Diagnostics: A New Frontier in Antimicrobial Stewardship
Abstract
1. Introduction
2. Results
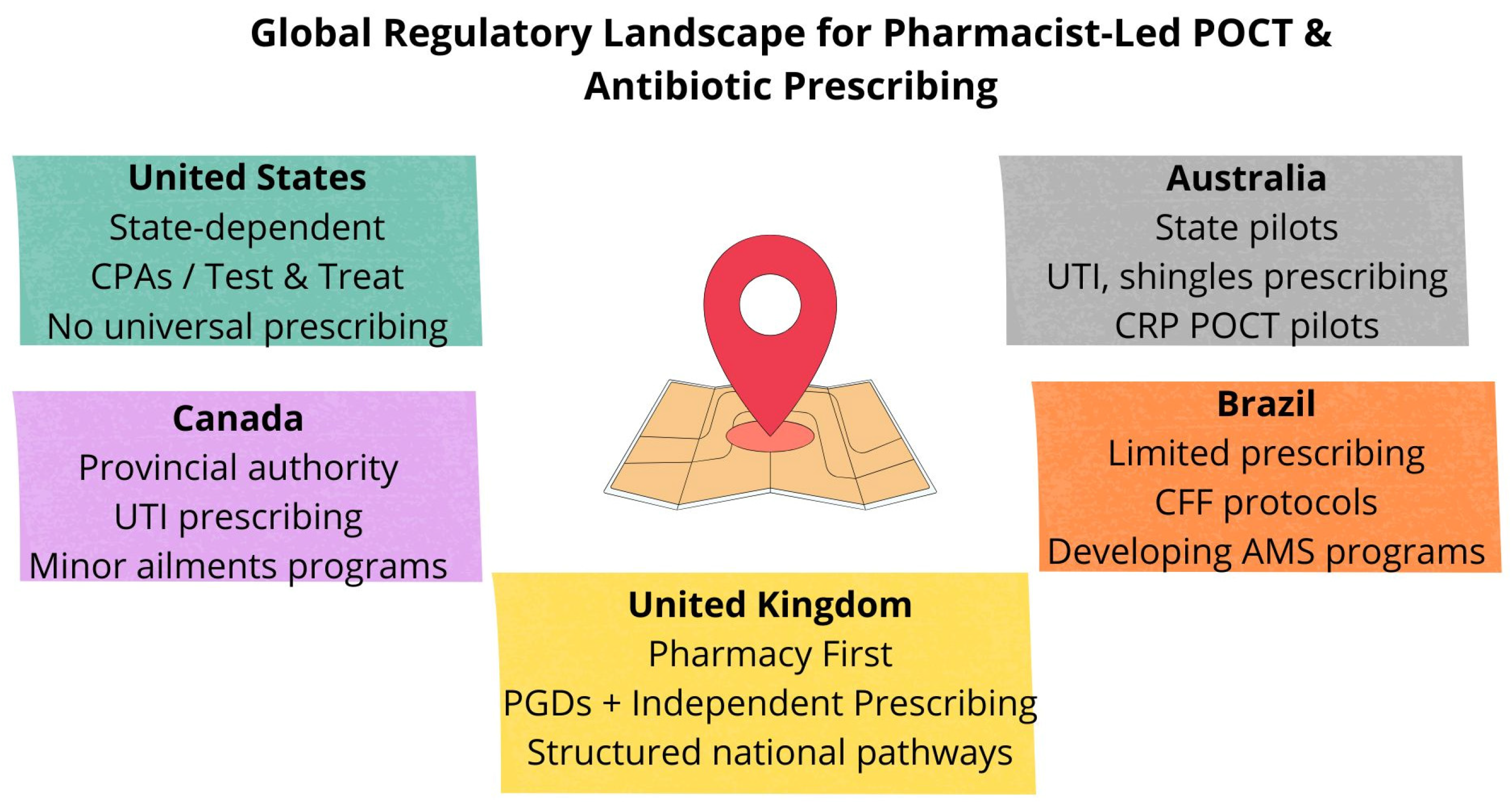
2.1. Regulation by Country
2.1.1. United States
2.1.2. United Kingdom
2.1.3. Australia
2.1.4. Canada
2.1.5. Brazil
2.2. Clinical Data, Monitoring
3. Discussion
4. Methods
4.1. Screening Process
4.2. Data Extraction
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Murray, C.J.L.; Ikuta, K.S.; Sharara, F.; Swetschinski, L.; Aguilar, G.R.; Gray, A.; Han, C.; Bisignano, C.; Rao, P.; Wool, E.; et al. Global burden of bacterial antimicrobial resistance in 2019: A systematic analysis. Lancet 2022, 399, 629–655. [Google Scholar] [CrossRef] [PubMed]
- Kaspute, G.; Zebrauskas, A.; Streckyte, A.; Ivaskiene, T.; Prentice, U. Combining Advanced Therapies with Alternative Treatments: A New Approach to Managing Antimicrobial Resistance? Pharmaceutics 2025, 17, 648. [Google Scholar] [CrossRef] [PubMed]
- Ashiru-Oredope, D.; Doble, A.; Thornley, T.; Saei, A.; Gold, N.; Sallis, A.; McNulty, C.A.M.; Lecky, D.; Umoh, E.; Klinger, C. Improving Management of Respiratory Tract Infections in Community Pharmacies and Promoting Antimicrobial Stewardship: A Cluster Randomised Control Trial with a Self-Report Behavioural Questionnaire and Process Evaluation. Pharmacy 2020, 8, 44. [Google Scholar] [CrossRef] [PubMed]
- Mantzourani, E.; Wasag, D.; Cannings-John, R.; Ahmed, H.; Evans, A. Characteristics of the Sore Throat Test and Treat Service in Community Pharmacies (STREP) in Wales: Cross-Sectional Analysis of 11,304 Consultations Using Anonymized Electronic Pharmacy Records. J. Antimicrob. Chemother. 2023, 78, 84–92. [Google Scholar] [CrossRef]
- Onwunduba, A.; Ekwunife, O.; Onyilogwu, E. Impact of Point-of-Care C-Reactive Protein Testing Intervention on Non-Prescription Dispensing of Antibiotics for Respiratory Tract Infections in Private Community Pharmacies in Nigeria: A Cluster Randomized Controlled Trial. Int. J. Infect. Dis. 2023, 127, 137–143. [Google Scholar] [CrossRef]
- Chan, J.T.N.; Nguyen, V.; Tran, T.N.; Nguyen, N.V.; Do, N.T.T.; van Doorn, H.R.; Lewycka, S. Point-of-Care Testing in Private Pharmacy and Drug Retail Settings: A Narrative Review. BMC Infect. Dis. 2023, 23, 551. [Google Scholar] [CrossRef]
- Saha, S.K.; Promite, S.; Botheras, C.L.; Manias, E.; Mothobi, N.; Robinson, S.; Athan, E. Improving Diagnostic Antimicrobial Stewardship in Respiratory Tract Infections: A Protocol for a Scoping Review Investigating Point-of-Care Testing Programmes in Community Pharmacy. BMJ Open 2023, 13, e068193. [Google Scholar] [CrossRef]
- Cillessen, L.M.; Lyons-Burney, H.; Gubbins, P.O. Chapter 47-Pharmacist Use of Point-of-Care Testing to Improve Access to Care. In Remington, 23rd ed.; Adejare, A., Ed.; Academic Press: Cambridge, MA, USA, 2021; pp. 817–828. ISBN 978-0-12-820007-0. [Google Scholar]
- Blaskovich, M.A.T.; Cooper, M.A. Antibiotics Re-Booted—Time to Kick Back against Drug Resistance. NPJ Antimicrob. Resist. 2025, 3, 47. [Google Scholar] [CrossRef]
- Bishop, C.; Yacoob, Z.; Knobloch, M.J.; Safdar, N. Community Pharmacy Interventions to Improve Antibiotic Stewardship and Implications for Pharmacy Education: A Narrative Overview. Res. Soc. Adm. Pharm. 2019, 15, 627–631. [Google Scholar] [CrossRef]
- Sachdev, G.; Kliethermes, M.A.; Vernon, V.; Leal, S.; Crabtree, G. Current Status of Prescriptive Authority by Pharmacists in the United States. JACCP J. Am. Coll. Clin. Pharm. 2020, 3, 807–817. [Google Scholar] [CrossRef]
- Adams, A.J.; Eid, D.D. Federal Pharmacist Paxlovid Prescribing Authority: A Model Policy or Impediment to Optimal Care? Explor. Res. Clin. Soc. Pharm. 2023, 9, 100244. [Google Scholar] [CrossRef] [PubMed]
- Adams, A.J.; Weaver, K.K. Pharmacists’ Patient Care Process: A State “Scope of Practice” Perspective. Innov. Pharm. 2019, 10. [Google Scholar] [CrossRef] [PubMed]
- Witry, M. The Role of Community Pharmacists in Point-of-Care Testing and Treatment for Influenza and Group A Streptococcus -a Narrative Review Using Ecological Systems Theory. Res. Soc. Adm. Pharm. 2025, 21, 205–214. [Google Scholar] [CrossRef] [PubMed]
- Koski, R.R.; Klepser, N.; Koski, M.; Klepser, M.; Klepser, D. Community Pharmacist-Provided Test and Treat Programs for Acute Infectious Conditions. JACCP J. Am. Coll. Clin. Pharm. 2023, 6, 1030–1040. [Google Scholar] [CrossRef]
- Shrestha, J.; Zahra, F.; Cannady, J. Antimicrobial Stewardship. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2025. [Google Scholar]
- Wu, J.H.-C.; Khalid, F.; Langford, B.J.; Beahm, N.P.; McIntyre, M.; Schwartz, K.L.; Garber, G.; Leung, V. Community Pharmacist Prescribing of Antimicrobials: A Systematic Review from an Antimicrobial Stewardship Perspective. Can. Pharm. J. 2021, 154, 179–192. [Google Scholar] [CrossRef]
- Maguire, T.; McGuckin, E.; Mawhinney, S.; Kerr, M.; Hamill, M. The Impact of a Community Pharmacy Cough, Cold and Flu Service on Antibiotic Prescribing and Its Acceptability. Pharm. J. 2021. [Google Scholar]
- CDC. Antibiotic Use in the United States. Available online: https://www.cdc.gov/antibiotic-use/hcp/data-research/antibiotic-prescribing.html (accessed on 1 October 2025).
- Outpatient Antibiotic Use: Retail Pharmacy Prescription Data|A.R. & Patient Safety Portal. Available online: https://arpsp.cdc.gov/profile/antibiotic-use/all-classes (accessed on 1 October 2025).
- Nobbs, S. In Practice: Guidance for Pharmacist Prescribers. Available online: https://assets.pharmacyregulation.org/files/2024-01/in-practice-guidance-for-pharmacist-prescribers-february-2020.pdf (accessed on 1 October 2025).
- Patient Group Directions: Who Can Use Them. Available online: https://www.gov.uk/government/publications/patient-group-directions-pgds/patient-group-directions-who-can-use-them (accessed on 1 October 2025).
- England NHS. Pharmacy First. Available online: https://www.england.nhs.uk/primary-care/pharmacy/pharmacy-services/pharmacy-first/ (accessed on 1 October 2025).
- NHS England. Becoming an Independent Prescriber. Available online: https://www.hee.nhs.uk/pharmacy/transforming-pharmacy-education-training/initial-education-training-pharmacists-reform-programme/pharmacy-integration-programme-super/becomingip-2 (accessed on 1 October 2025).
- Janković, S. Pharmacists Provide Antibiotics Only Where Clinically Appropriate, Minor Ailments Scheme Data Show. Pharm. J. 2023. Available online: https://assets.publishing.service.gov.uk/media/6936ac34b612700b2cb73607/ESPAUR-report-2024-to-2025.pdf (accessed on 1 October 2025). [CrossRef]
- UK Health Security Agency (UKHSA). English Surveillance Programme for Antimicrobial Utilisation and Resistance (ESPAUR) Report 2024 to 2025; UKHSA: London, UK, 2025.
- Department of Health; Victoria, A. Community Pharmacist Program. Available online: https://www.health.vic.gov.au/primary-care/community-pharmacist-program (accessed on 1 October 2025).
- Stehlik, P.; Moles, R.; Jones, M.; Murray, A.; El-Den, S.; Morgan, M.; Mar, C.D. Community Pharmacists’ Referrals to General Practice with Suspected Need of Antibiotics: A National Prospective Pilot. medRxiv 2024. medRxiv:2024.06.19.24309200. [Google Scholar] [CrossRef]
- Rizvi, T.; Zaidi, S.T.R.; Williams, M.; Thompson, A.; Peterson, G.M. Factors Influencing Community Pharmacists’ Participation in Antimicrobial Stewardship: A Qualitative Inquiry. Pharmacy 2025, 13, 56. [Google Scholar] [CrossRef]
- Llor, C.; Benkő, R.; Bjerrum, L. Global Restriction of the Over-the-Counter Sale of Antimicrobials: Does It Make Sense? Front. Public Health 2024, 12, 1412644. [Google Scholar] [CrossRef]
- Ministry of Health. Pharmacy Services in B.C. Available online: https://www2.gov.bc.ca/gov/content/health/accessing-health-care/pharmacy-services (accessed on 1 October 2025).
- Saskatchewan College of Pharmacy Professionals. Available online: https://saskpharm.ca/ (accessed on 1 October 2025).
- Patel, T.S.; Sati, H.; Lessa, F.C.; Patel, P.K.; Srinivasan, A.; Hicks, L.A.; Neuhauser, M.M.; Tong, D.; van der Heijden, M.; Alves, S.C.; et al. Defining Access without Excess: Expanding Appropriate Use of Antibiotics Targeting Multidrug-Resistant Organisms. Lancet Microbe 2024, 5, e93–e98. [Google Scholar] [CrossRef] [PubMed]
- Conselho Federal de Farmacia. Pharmaceutical Prescription and the Clinical Attributions of the Pharmacists. Available online: https://www.cff.org.br/userfiles/file/prescri%C3%A7%C3%A3o%20farmaceutica_AZUL%20(2).pdf (accessed on 1 October 2025).
- Menezes, R.M.; Gonçalves, M.R.S.; Costa, M.M.d.M.; Krumennauer, E.C.; Carneiro, G.M.; Reuter, C.P.; Renner, J.D.P.; Carneiro, M. Antimicrobial Stewardship Programmes in Brazil: Introductory Analysis. Res. Soc. Dev. 2022, 11, e51011729444. [Google Scholar] [CrossRef]
- Cunha, J.P.A.; Rodrigues, J.P.V.; Rocha, K.S.S.; Gonçalves, A.M.R.F.; Varallo, F.R.; Pereira, L.R.L. National Assessment of Brazilian Community Pharmacists’ Knowledge in Medication Dispensing: A Cross-Sectional Survey. medRxiv 2025. medRxiv:2025.06.07.25329194. [Google Scholar] [CrossRef]
- Hohmeier, K.C.; McKeirnan, K.; Akers, J.; Klepser, M.; Klepser, S.A.; Chen, C.; Klepser, D.G. Implementing Community Pharmacy-Based Influenza Point-of-Care Test-and-Treat under Collaborative Practice Agreement. Implement Sci. Commun. 2022, 3, 77. [Google Scholar] [CrossRef]
- Edokpayi, K.; Aluko, P.; Cheng, F.K.K.; Attar-Zadeh, D.; Dawson, A. Streptococcus A Rapid Diagnostic Testing in England Community Pharmacies: Clinical and Economic Impact of Empowering Pharmacists in Management of Sore Throat. J. Prim. Care Community Health 2025, 16, 21501319251340836. [Google Scholar] [CrossRef]
- Thursky, K.A.; Hardefeldt, L.Y.; Rajkhowa, A.; Ierano, C.; Bishop, J.; Hawes, L.; Biezen, R.; Saha, S.K.; Dowson, L.; Bailey, K.E.; et al. Antimicrobial Stewardship in Australia: The Role of Qualitative Research in Programme Development. JAC Antimicrob. Resist. 2021, 3, dlab166. [Google Scholar] [CrossRef]
- Ayton, D.; Watson, E.; Betts, J.M.; Doyle, J.; Teh, B.; Valoppi, G.; Cotta, M.; Robertson, M.; Peel, T. Implementation of an Antimicrobial Stewardship Program in the Australian Private Hospital System: Qualitative Study of Attitudes to Antimicrobial Resistance and Antimicrobial Stewardship. BMC Health Serv. Res. 2022, 22, 1554. [Google Scholar] [CrossRef]
- Sim, T.F.; Chalmers, L.; Czarniak, P.; Hughes, J.; Iacob, R.; Lee, Y.P.; Parsons, K.; Parsons, R.; Sunderland, B. Point-of-Care C-Reactive Protein Testing to Support the Management of Respiratory Tract Infections in Community Pharmacy: A Feasibility Study. Res. Soc. Adm. Pharm. 2021, 17, 1719–1726. [Google Scholar] [CrossRef]
- Czarniak, P.; Chalmers, L.; Hughes, J.; Iacob, R.; Lee, Y.P.; Parsons, K.; Parsons, R.; Sunderland, B.; Sim, T.F. Point-of-Care C-Reactive Protein Testing Service for Respiratory Tract Infections in Community Pharmacy: A Qualitative Study of Service Uptake and Experience of Pharmacists. Int. J. Clin. Pharm. 2022, 44, 466–479. [Google Scholar] [CrossRef]
- Wakeman, M.; Cork, T.; Wakeman, D.W. Point-of-Care C-Reactive Protein Testing in Community Pharmacy to Deliver Appropriate Interventions in Respiratory Tract Infections. Clin. Pharm. 2018, 10, 149–153. [Google Scholar]

| Ref. | Type of Study | Country | Intervention | Result | Opportunities | Limitations |
|---|---|---|---|---|---|---|
| [5] | Parallel cluster RCT | Nigeria |
| Reduced non-prescription antibiotic dispensing for RTIs. |
|
|
| [3] | Cluster RCT with self-report behavioural questionnaire and process evaluation | UK | AMS behaviour change intervention:
|
|
|
|
| [41] | Feasibility study/pilot | Australia |
|
|
|
|
| [42] | Service pilot, qualitative evaluation | Australia | Implemented CRP service | Reduced perceived need for antibiotics. |
|
|
| [43] | Pilot feasibility evaluation | UK |
| Feasible; suggested potential to reduce unnecessary doctor visits and to guide appropriate interventions. | Small-scale evidence that POC CRP in pharmacy can inform antibiotic decisions locally. |
|
| [4] | Service evaluation/program evaluation (real-world roll-out) | Wales |
|
|
|
|
| Inclusion | Exclusion |
|---|---|
|
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kaspute, G.; Ivaskiene, T. Pharmacist-Led Diagnostics: A New Frontier in Antimicrobial Stewardship. Antibiotics 2025, 14, 1286. https://doi.org/10.3390/antibiotics14121286
Kaspute G, Ivaskiene T. Pharmacist-Led Diagnostics: A New Frontier in Antimicrobial Stewardship. Antibiotics. 2025; 14(12):1286. https://doi.org/10.3390/antibiotics14121286
Chicago/Turabian StyleKaspute, Greta, and Tatjana Ivaskiene. 2025. "Pharmacist-Led Diagnostics: A New Frontier in Antimicrobial Stewardship" Antibiotics 14, no. 12: 1286. https://doi.org/10.3390/antibiotics14121286
APA StyleKaspute, G., & Ivaskiene, T. (2025). Pharmacist-Led Diagnostics: A New Frontier in Antimicrobial Stewardship. Antibiotics, 14(12), 1286. https://doi.org/10.3390/antibiotics14121286






