LipiSensors: Exploiting Lipid Nanoemulsions to Fabricate Ionophore-Based Nanosensors
Abstract
1. Introduction
2. Materials and Methods
3. Results
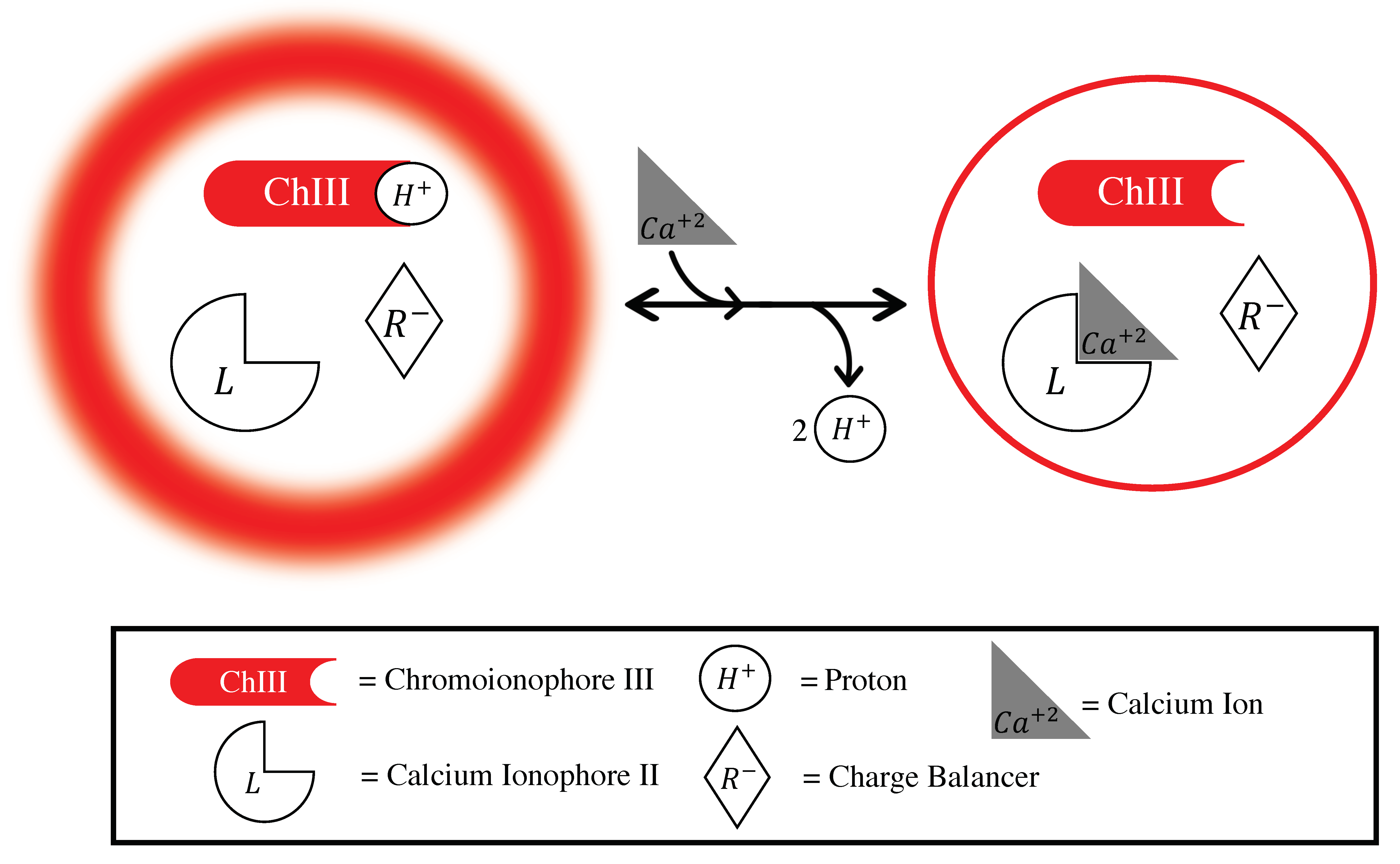
3.1. Mechanism
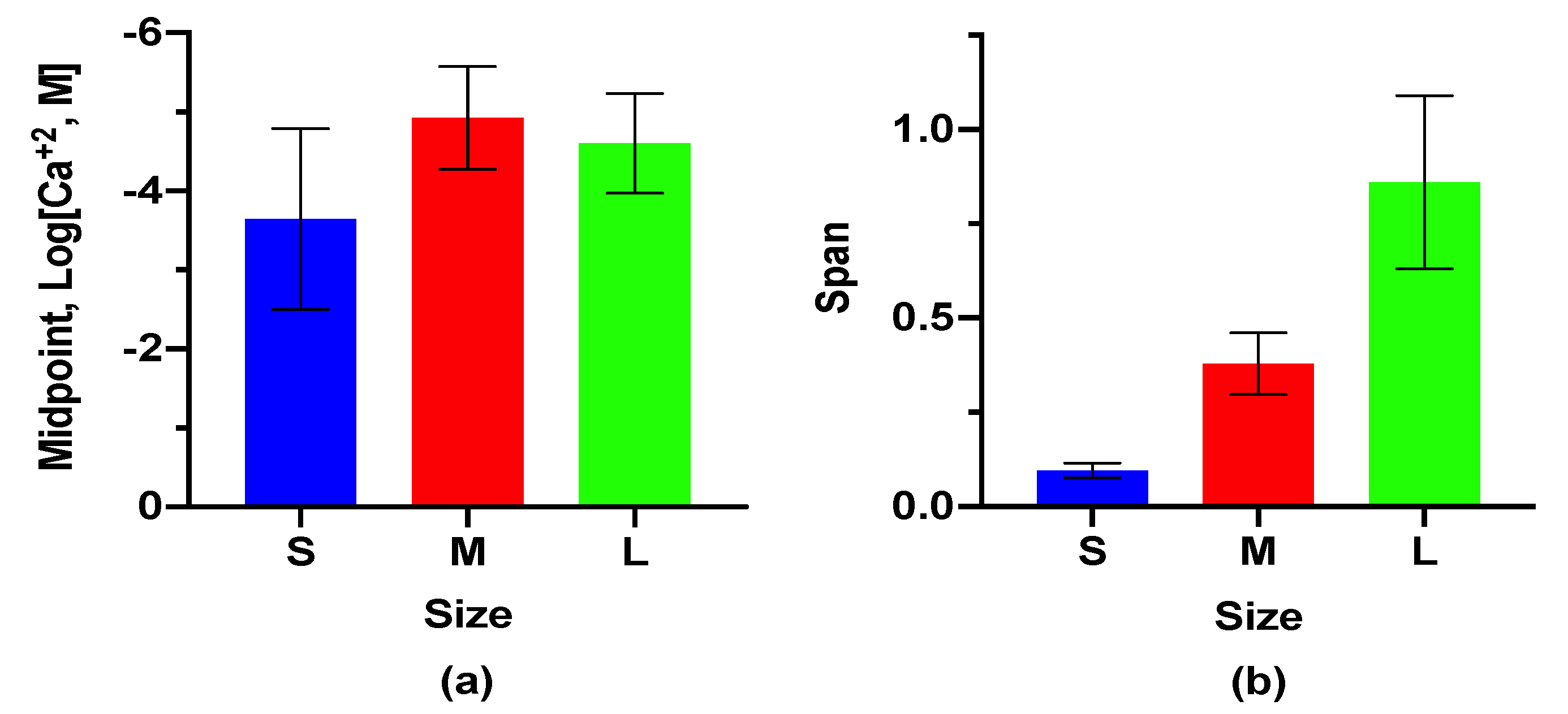
3.2. Ca+2 Response
3.3. Lifetime
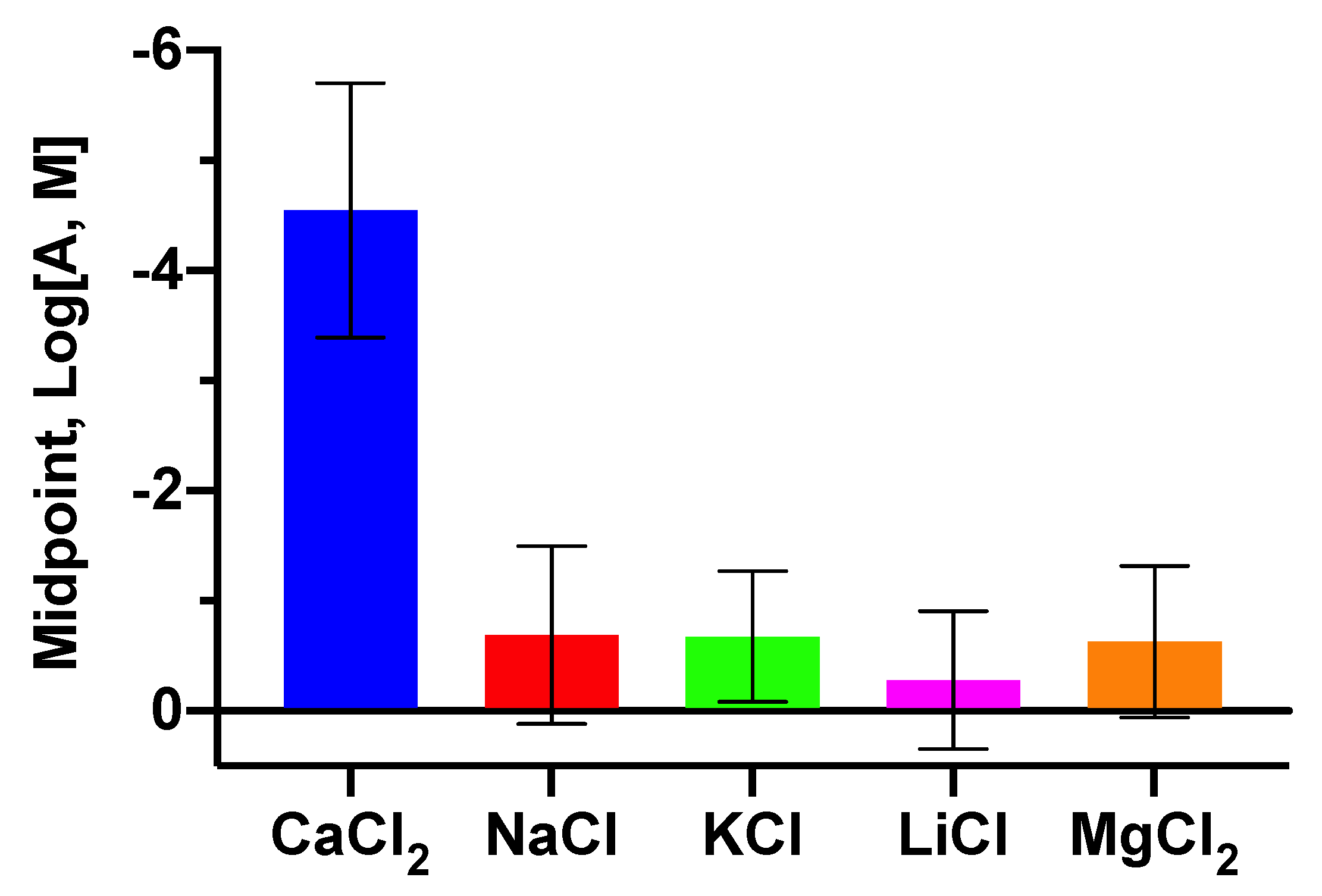
3.4. Selectivity
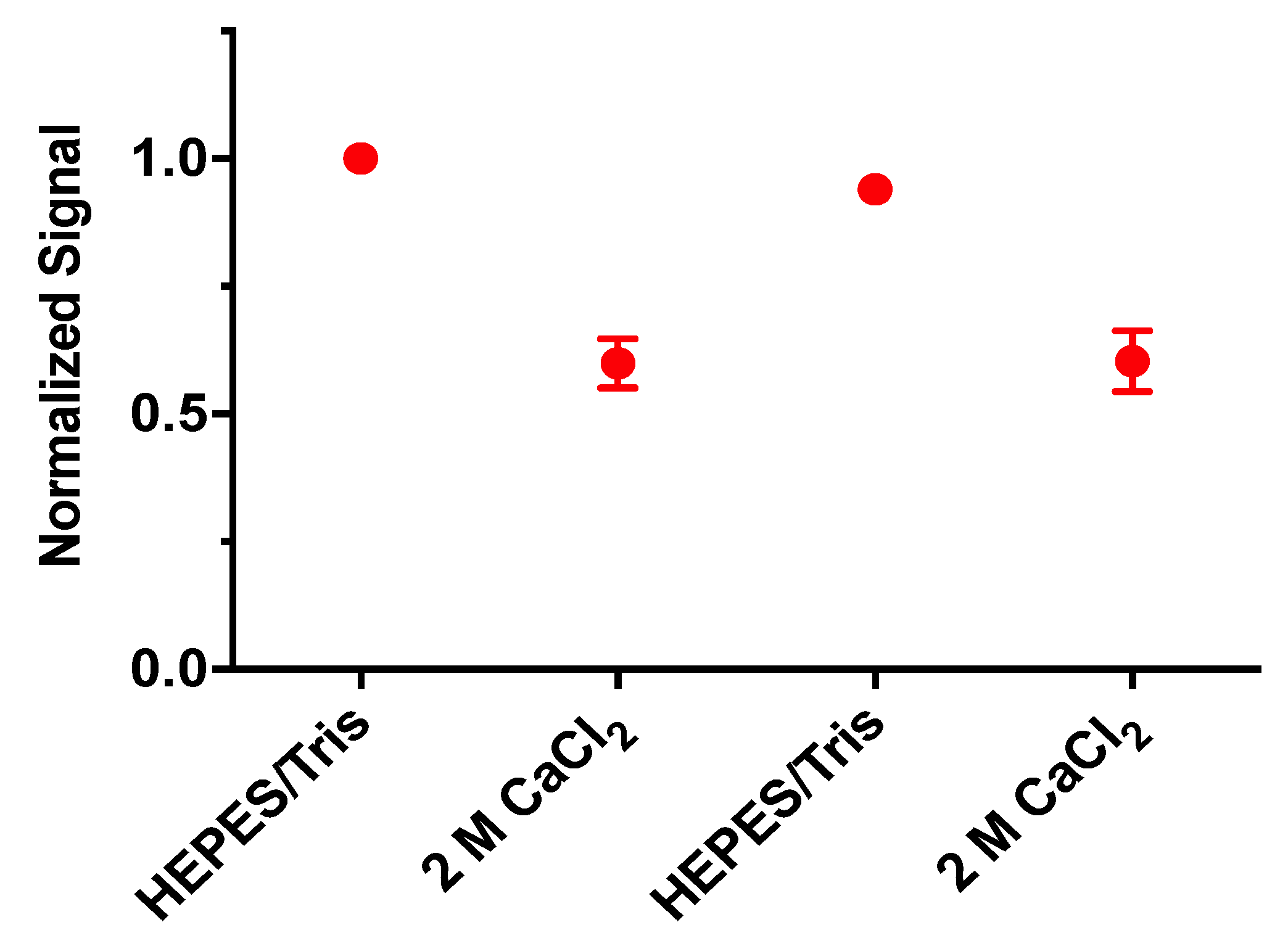
3.5. Reversibility
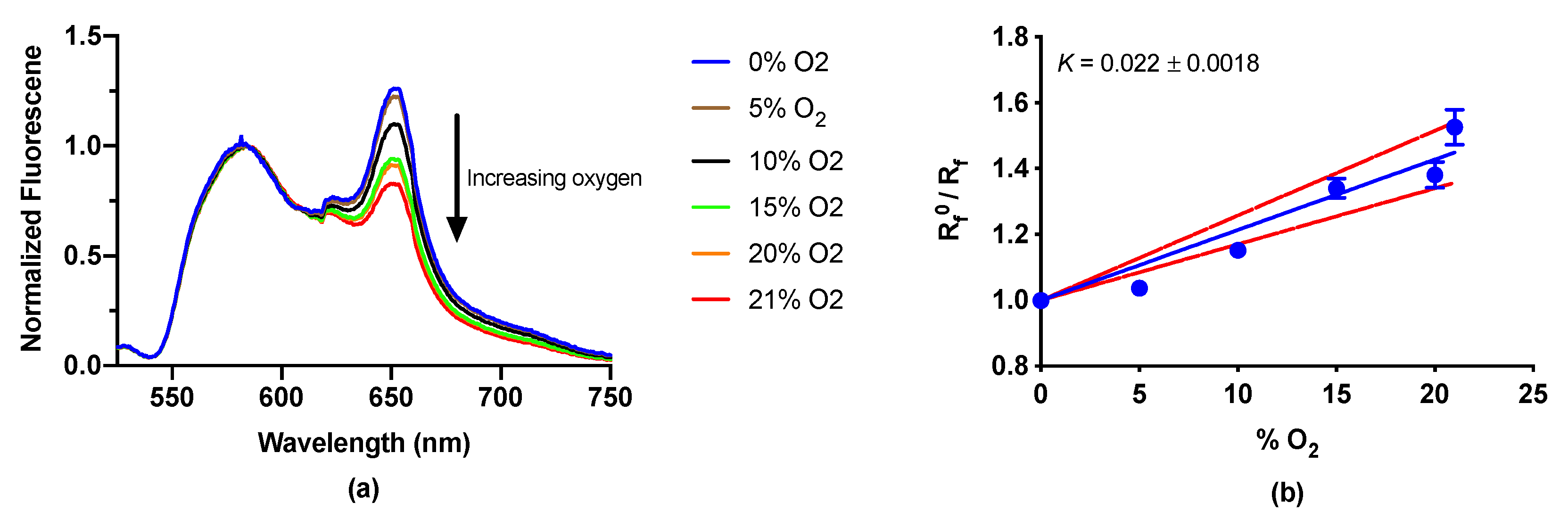
3.6. Oxygen-Sensitive LipiSensor Response and Characterization
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Pendley, B.D.; Lindner, E. Medical Sensors for the Diagnosis and Management of Disease: The Physician Perspective. ACS Sens. 2017, 2, 1549–1552. [Google Scholar] [CrossRef] [PubMed]
- Jett, S.E.; Bonham, A.J. Reusable Electrochemical DNA Biosensor for the Detection of Waterborne Uranium. ChemElectroChem 2017, 4, 843–845. [Google Scholar] [CrossRef]
- Zhang, X.-Y.; Yang, Y.-S.; Wang, W.; Jiao, Q.-C.; Zhu, H.-L. Fluorescent Sensors for the Detection of Hydrazine in Environmental and Biological Systems: Recent Advances and Future Prospects. Coord. Chem. Rev. 2020, 417, 213367. [Google Scholar] [CrossRef]
- Jewell, M.P.; Saccomano, S.C.; David, A.A.; Harris, J.K.; Zemanick, E.T.; Cash, K.J. Nanodiagnostics to Monitor Biofilm Oxygen Metabolism for Antibiotic Susceptibility Testing. Analyst 2020. [Google Scholar] [CrossRef] [PubMed]
- Carter, K.P.; Young, A.M.; Palmer, A.E. Fluorescent Sensors for Measuring Metal Ions in Living Systems. Chem. Rev. 2014, 114, 4564–4601. [Google Scholar] [CrossRef] [PubMed]
- Morales, M.A.; Halpern, J.M. Guide to Selecting a Biorecognition Element for Biosensors. Bioconjug. Chem. 2018, 29, 3231–3239. [Google Scholar] [CrossRef] [PubMed]
- Tsien, R.Y. New Calcium Indicators and Buffers with High Selectivity against Magnesium and Protons: Design, Synthesis, and Properties of Prototype Structures. Biochemistry 1980, 19, 2396–2404. [Google Scholar] [CrossRef]
- Han, J.Y.; Burgess, K. Fluorescent Indicators for Intracellular PH. Chem. Rev. 2010, 110, 2709–2728. [Google Scholar] [CrossRef]
- Chan, J.; Dodani, S.C.; Chang, C.J. Reaction-Based Small-Molecule Fluorescent Probes for Chemoselective Bioimaging. Nat. Chem. 2012, 4, 973–984. [Google Scholar] [CrossRef]
- Qi, J.; Liu, D.Y.; Liu, X.Y.; Guan, S.Q.; Shi, F.L.; Chang, H.X.; He, H.R.; Yang, G.M. Fluorescent PH Sensors for Broad-Range PH Measurement Based on a Single Fluorophore. Anal. Chem. 2015, 87, 5897–5904. [Google Scholar] [CrossRef]
- Deng, F.; Liu, L.; Qiao, Q.; Huang, C.; Miao, L.; Xu, Z. A General Strategy to Develop Cell Membrane Fluorescent Probes with Location- and Target-Specific Fluorogenicities: A Case of a Zn2+ Probe with Cellular Selectivity. Chem. Commun. 2019, 55, 15045–15048. [Google Scholar] [CrossRef] [PubMed]
- Hirata, T.; Terai, T.; Yamamura, H.; Shimonishi, M.; Komatsu, T.; Hanaoka, K.; Ueno, T.; Imaizumi, Y.; Nagano, T.; Urano, Y. Protein-Coupled Fluorescent Probe to Visualize Potassium Ion Transition on Cellular Membranes. Anal. Chem. 2016, 88, 2693–2700. [Google Scholar] [CrossRef]
- Dubach, J.M.; Balaconis, M.K.; Clark, H.A. Fluorescent Nanoparticles for the Measurement of Ion Concentration in Biological Systems. J. Vis. Exp. 2011, 53, 1–5. [Google Scholar] [CrossRef]
- Ferris, M.S.; Katageri, A.G.; Gohring, G.M.; Cash, K.J. A Dual-Indicator Strategy for Controlling the Response of Ionophore-Based Optical Nanosensors. Sens. Actuators B Chem. 2018, 256, 674–681. [Google Scholar] [CrossRef]
- Mistlberger, G.; Crespo, G.A.; Bakker, E. Ionophore-Based Optical Sensors. Annu. Rev. Anal. Chem. 2014, 7, 483–512. [Google Scholar] [CrossRef]
- Ruckh, T.T.; Clark, H.A. Implantable Nanosensors: Toward Continuous Physiologic Monitoring. Anal. Chem. 2014, 86, 1314–1323. [Google Scholar] [CrossRef]
- Di, W.; Clark, H.A. Optical Nanosensors for: In Vivo Physiological Chloride Detection for Monitoring Cystic Fibrosis Treatment. Anal. Methods 2020, 12, 1441–1448. [Google Scholar] [CrossRef]
- Jewell, M.P.; Galyean, A.A.; Kirk Harris, J.; Zemanick, E.T.; Cash, K.J. Luminescent Nanosensors for Ratiometric Monitoring of Three-Dimensional Oxygen Gradients in Laboratory and Clinical Pseudomonas Aeruginosa Biofilms. Appl. Environ. Microbiol. 2019, 85, e01116-19. [Google Scholar] [CrossRef] [PubMed]
- Xie, X.; Bakker, E. Ion Selective Optodes: From the Bulk to the Nanoscale. Anal. Bioanal. Chem. 2015, 407, 3899–3910. [Google Scholar] [CrossRef]
- Galyean, A.A.; Behr, M.R.; Cash, K.J. Ionophore-Based Optical Nanosensors Incorporating Hydrophobic Carbon Dots and a PH-Sensitive Quencher Dye for Sodium Detection. Analyst 2018, 143, 458–465. [Google Scholar] [CrossRef]
- Xie, X. Renovating the Chromoionophores and Detection Modes in Carrier-Based Ion-Selective Optical Sensors. Anal. Bioanal. Chem. 2016, 408, 2717–2725. [Google Scholar] [CrossRef] [PubMed]
- Balaconis, M.K.; Clark, H.A. Biodegradable Optode-Based Nanosensors for in Vivo Monitoring. Anal. Chem. 2012, 84, 5787–5793. [Google Scholar] [CrossRef] [PubMed]
- Xie, X.; Mistlberger, G.; Bakker, E. Ultrasmall Fluorescent Ion-Exchanging Nanospheres Containing Selective Ionophores. Anal. Chem. 2013, 85, 9932–9938. [Google Scholar] [CrossRef] [PubMed]
- Ruckh, T.T.; Mehta, A.A.; Dubach, J.M.; Clark, H.A. Polymer-Free Optode Nanosensors for Dynamic, Reversible and Ratiometric Sodium Imaging in the Physiological Range. Sci. Rep. 2013, 3, 3366. [Google Scholar] [CrossRef]
- Du, X.F.; Yang, L.Y.; Hu, W.C.; Wang, R.J.; Zhai, J.Y.; Xie, X.J. A Plasticizer-Free Miniaturized Optical Ion Sensing Platform with Ionophores and Silicon-Based Particles. Anal. Chem. 2018, 90, 5818–5824. [Google Scholar] [CrossRef]
- Rao, J.P.; Geckeler, K.E. Polymer Nanoparticles: Preparation Techniques and Size-Control Parameters. Prog. Polym. Sci. 2011, 36, 887–913. [Google Scholar] [CrossRef]
- Du, X.; Wang, R.; Zhai, J.; Li, X.; Xie, X. Ionophore-Based Ion-Selective Nanosensors from Brush Block Copolymer Nanodots. ACS Appl. Nano Mater. 2020, 3, 782–788. [Google Scholar] [CrossRef]
- Gravier, J.J.; Garcia, F.P.N.Y.; Delmas, T.; Mittler, F.; Couffin, A.-C.; Vinet, F.; Texier-Nogues, I. Lipidots: Competitive Organic Alternative to Quantum Dots for in Vivo Fluorescence Imaging. J. Biomed. Opt. 2011, 16, 1–11. [Google Scholar] [CrossRef]
- Fortin, P.-Y.; Genevois, C.; Koenig, A.; Heinrich, E.; Texier-Nogues, I.; Couillaud, F. Detection of Brain Tumors Using Fluorescence Diffuse Optical Tomography and Nanoparticles as Contrast Agents. J. Biomed. Opt. 2012, 17, 1–6. [Google Scholar] [CrossRef]
- Hinger, D.; Navarro, F.; Käch, A.; Thomann, J.-S.; Mittler, F.; Couffin, A.-C.; Maake, C. Photoinduced Effects of M-Tetrahydroxyphenylchlorin Loaded Lipid Nanoemulsions on Multicellular Tumor Spheroids. J. Nanobiotechnol. 2016, 14, 68. [Google Scholar] [CrossRef]
- Hinger, D.; Gräfe, S.; Navarro, F.; Spingler, B.; Pandiarajan, D.; Walt, H.; Couffin, A.-C.; Maake, C. Lipid Nanoemulsions and Liposomes Improve Photodynamic Treatment Efficacy and Tolerance in CAL-33 Tumor Bearing Nude Mice. J. Nanobiotechnol. 2016, 14, 71. [Google Scholar] [CrossRef] [PubMed]
- Delmas, T.; Piraux, H.; Couffin, A.-C.; Texier, I.; Vinet, F.; Poulin, P.; Cates, M.E.; Bibette, J. How to Prepare and Stabilize Very Small Nanoemulsions. Langmuir 2011, 27, 1683–1692. [Google Scholar] [CrossRef] [PubMed]
- Delmas, T.; Couffin, A.-C.; Bayle, P.A.; de Crécy, F.; Neumann, E.; Vinet, F.; Bardet, M.; Bibette, J.; Texier, I. Preparation and Characterization of Highly Stable Lipid Nanoparticles with Amorphous Core of Tuneable Viscosity. J. Colloid Interface Sci. 2011, 360, 471–481. [Google Scholar] [CrossRef]
- Jacquart, A.; Keramidas, M.; Vollaire, J.; Boisgard, R.; Pottier, G.; Rustique, E.; Mittler, F.; Navarro, F.; Boutet, J.; Coll, J.-L.; et al. LipImageTM 815: Novel Dye-Loaded Lipid Nanoparticles for Long-Term and Sensitive in Vivo near-Infrared Fluorescence Imaging. J. Biomed. Opt. 2013, 18, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Gravier, J.; Sancey, L.; Coll, J.L.; Hirsjärvi, S.; Benoît, J.P.; Vinet, F.; Texier, I. FRET as a Tool for the Investigation of the Fate of Lipidots Contrast Agents in Vivo. In Proceedings of the SPIE, San Francisco, CA, USA, 22–27 January 2011; Volume 7910. [Google Scholar] [CrossRef]
- Klymchenko, A.S.; Roger, E.; Anton, N.; Anton, H.; Shulov, I.; Vermot, J.; Mely, Y.; Vandamme, T.F. Highly Lipophilic Fluorescent Dyes in Nano-Emulsions: Towards Bright Non-Leaking Nano-Droplets. RSC Adv. 2012, 2, 11876–11886. [Google Scholar] [CrossRef]
- Mérian, J.; Boisgard, R.; Bayle, P.-A.; Bardet, M.; Tavitian, B.; Texier, I. Comparative Biodistribution in Mice of Cyanine Dyes Loaded in Lipid Nanoparticles. Eur. J. Pharm. Biopharm. 2015, 93, 1–10. [Google Scholar] [CrossRef]
- Cash, K.J.; Clark, H.A. Phosphorescent Nanosensors for in Vivo Tracking of Histamine Levels. Anal. Chem. 2013, 85, 6312–6318. [Google Scholar] [CrossRef]
- Schindelin, J.; Arganda-Carreras, I.; Frise, E.; Kaynig, V.; Longair, M.; Pietzsch, T.; Preibisch, S.; Rueden, C.; Saalfeld, S.; Schmid, B.; et al. Fiji: An Open-Source Platform for Biological-Image Analysis. Nat. Methods 2012, 9, 676–682. [Google Scholar] [CrossRef]
- Lee, S.-K.; Okura, I. Photostable Optical Oxygen Sensing Material: Platinum Tetrakis (Pentafluorophenyl) Porphyrin Immobilized in Polystyrene. Anal. Commun. 1997, 34, 185–188. [Google Scholar] [CrossRef]
- Papkovsky, D.B.; Ponomarev, G.V.; Trettnak, W.; Oleary, P. Phosphorescent Complexes of Porphyrin Ketones: Optical Properties and Application to Oxygen Sensing. Anal. Chem. 1995, 67, 4112–4117. [Google Scholar] [CrossRef]
- Quaranta, M.; Borisov, S.M.; Klimant, I. Indicators for Optical Oxygen Sensors. Bioanal. Rev. 2012, 4, 115–157. [Google Scholar] [CrossRef] [PubMed]
- Clapham, D.E. Calcium Signaling. Cell 2007, 131, 1047–1058. [Google Scholar] [CrossRef] [PubMed]
- Ozaydin-Ince, G.; Dubach, J.M.; Gleason, K.K.; Clark, H.A. Microworm Optode Sensors Limit Particle Diffusion to Enable in Vivo Measurements. Proc. Natl. Acad. Sci. USA 2011, 108, 2656–2661. [Google Scholar] [CrossRef] [PubMed]
- Hirsjärvi, S.; Dufort, S.; Gravier, J.; Texier, I.; Yan, Q.; Bibette, J.; Sancey, L.; Josserand, V.; Passirani, C.; Benoit, J.-P.; et al. Influence of Size, Surface Coating and Fine Chemical Composition on the in Vitro Reactivity and in Vivo Biodistribution of Lipid Nanocapsules versus Lipid Nanoemulsions in Cancer Models. Nanomed. Nanotechnol. Biol. Med. 2013, 9, 375–387. [Google Scholar] [CrossRef] [PubMed]
- Rong, G.; Kim, E.H.; Poskanzer, K.E.; Clark, H.A. A Method for Estimating Intracellular Ion Concentration Using Optical Nanosensors and Ratiometric Imaging. Sci. Rep. 2017, 7, 10819. [Google Scholar] [CrossRef]
- Dubach, J.M.; Das, S.; Rosenzweig, A.; Clark, H.A. Visualizing Sodium Dynamics in Isolated Cardiomyocytes Using Fluorescent Nanosensors. Proc. Natl. Acad. Sci. USA 2009, 106, 16145–16150. [Google Scholar] [CrossRef]
- Ferris, M.S.; Elms, M.K.; Cash, K.J. Enzyme-Conjugated Nanosensors with Tunable Detection Limits for Small Biomolecule Determination. AIChE J. 2019, 65, e16698. [Google Scholar] [CrossRef]
- Cash, K.J.; Li, C.; Xia, J.; Wang, L.V.; Clark, H.A. Optical Drug Monitoring: Photoacoustic Imaging of Nanosensors to Monitor Therapeutic Lithium in Vivo. ACS Nano 2015, 9, 1692–1698. [Google Scholar] [CrossRef]
- Suzuki, K.; Hirayama, E.; Sugiyama, T.; Yasuda, K.; Okabe, H.; Citterio, D. Ionophore-Based Lithium Ion Film Optode Realizing Multiple Color Variations Utilizing Digital Color Analysis. Anal. Chem. 2002, 74, 5766–5773. [Google Scholar] [CrossRef]
- Erenas, M.M.; Cantrell, K.; Ballesta-Claver, J.; de Orbe-Payá, I.; Capitán-Vallvey, L.F. Use of Digital Reflection Devices for Measurement Using Hue-Based Optical Sensors. Sens. Actuators B Chem. 2012, 174, 10–17. [Google Scholar] [CrossRef]
- Chan, K.W.Y.; Liu, G.; Song, X.; Kim, H.; Yu, T.; Arifin, D.R.; Gilad, A.A.; Hanes, J.; Walczak, P.; van Zijl, P.C.M.; et al. MRI-Detectable pH Nanosensors Incorporated into Hydrogels for In Vivo Sensing of Transplanted-Cell Viability. Nat. Mater. 2013, 12, 268–275. [Google Scholar] [CrossRef] [PubMed]
- Rong, G.; Tuttle, E.E.; Neal Reilly, A.; Clark, H.A. Recent Developments in Nanosensors for Imaging Applications in Biological Systems. Annu. Rev. Anal. Chem. 2019, 12, 109–128. [Google Scholar] [CrossRef] [PubMed]

© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dailey, A.L.; Greer, M.D.; Sodia, T.Z.; Jewell, M.P.; Kalin, T.A.; Cash, K.J. LipiSensors: Exploiting Lipid Nanoemulsions to Fabricate Ionophore-Based Nanosensors. Biosensors 2020, 10, 120. https://doi.org/10.3390/bios10090120
Dailey AL, Greer MD, Sodia TZ, Jewell MP, Kalin TA, Cash KJ. LipiSensors: Exploiting Lipid Nanoemulsions to Fabricate Ionophore-Based Nanosensors. Biosensors. 2020; 10(9):120. https://doi.org/10.3390/bios10090120
Chicago/Turabian StyleDailey, Alexandra L., Meredith D. Greer, Tyler Z. Sodia, Megan P. Jewell, Tabitha A. Kalin, and Kevin J. Cash. 2020. "LipiSensors: Exploiting Lipid Nanoemulsions to Fabricate Ionophore-Based Nanosensors" Biosensors 10, no. 9: 120. https://doi.org/10.3390/bios10090120
APA StyleDailey, A. L., Greer, M. D., Sodia, T. Z., Jewell, M. P., Kalin, T. A., & Cash, K. J. (2020). LipiSensors: Exploiting Lipid Nanoemulsions to Fabricate Ionophore-Based Nanosensors. Biosensors, 10(9), 120. https://doi.org/10.3390/bios10090120




