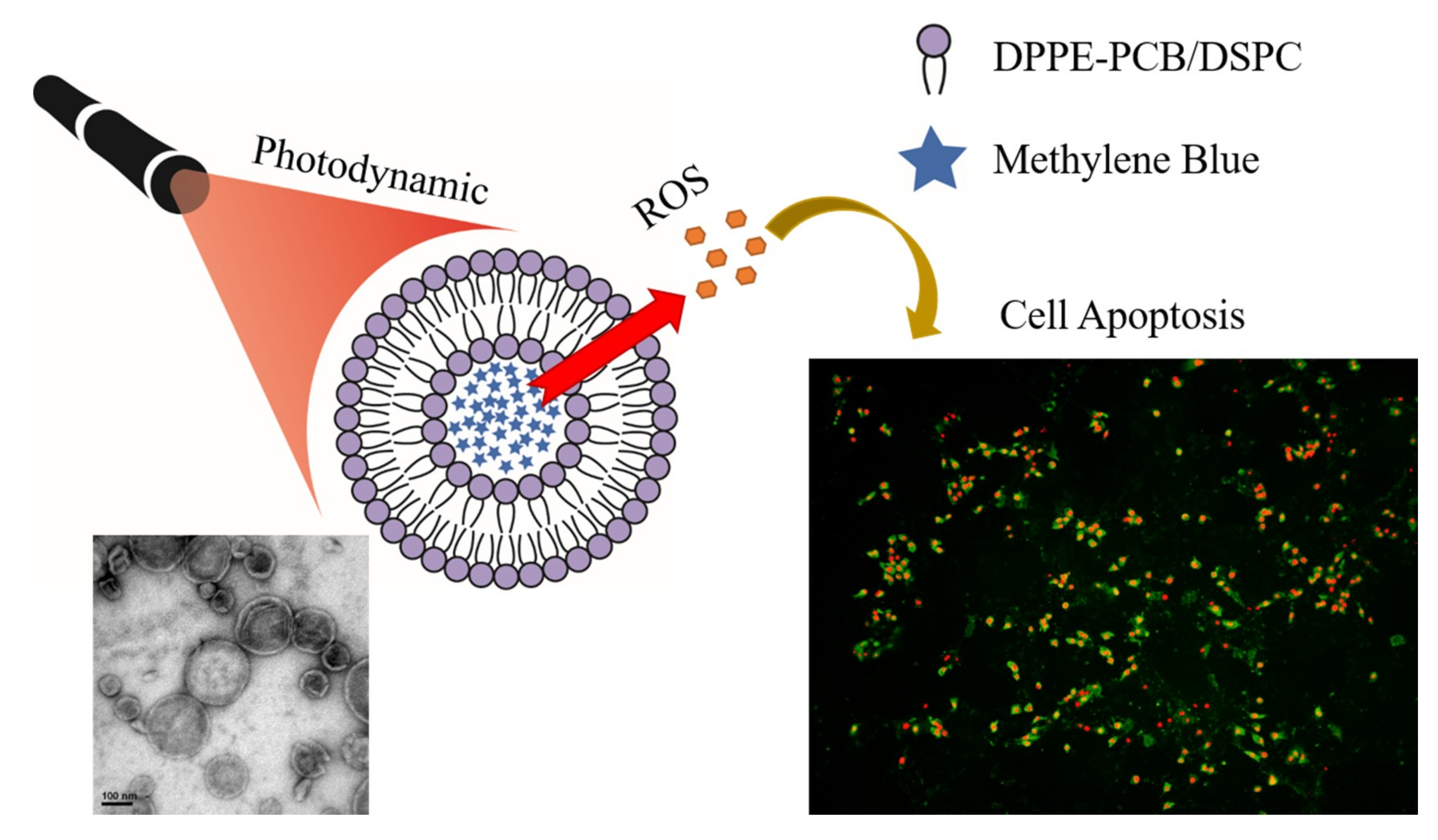
Methylene-Blue-Encapsulated Liposomes as Photodynamic Therapy Nano Agents for Breast Cancer Cells
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. MB-Liposome Synthesis
2.3. MB Release Test
2.4. ROS Generation: RNO Test
2.5. Cell Culture
2.6. Cytotoxicity
2.7. Live and Dead Staining
2.8. Intracellular Uptake
2.9. In Vitro ROS Generation: DCFH-DA Assay
2.10. Identification of Cell Death Pathway
2.11. In Vivo Toxicity Test
2.12. Statistical Analysis
3. Results and Discussion
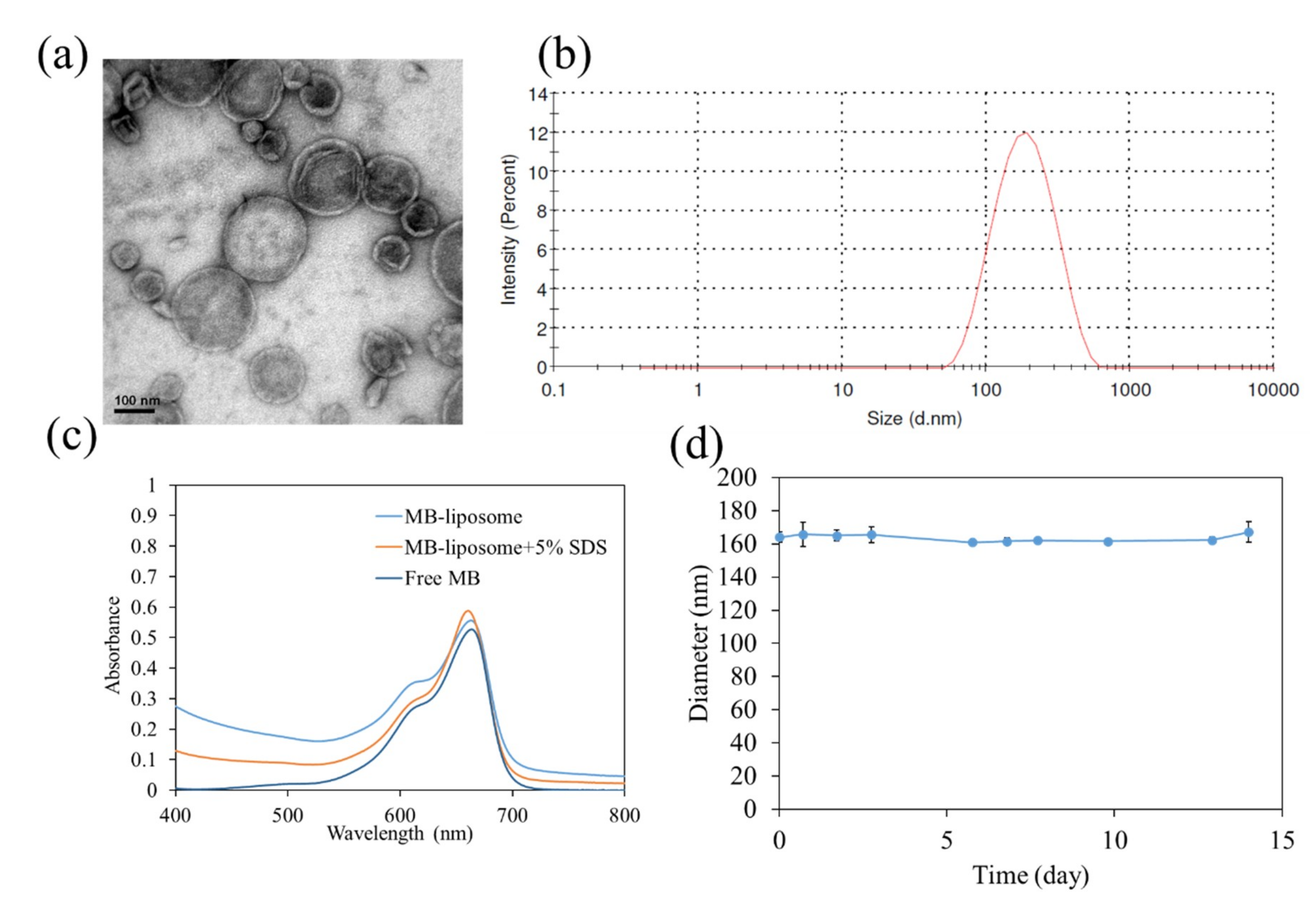
3.1. Characterization of MB-Liposome
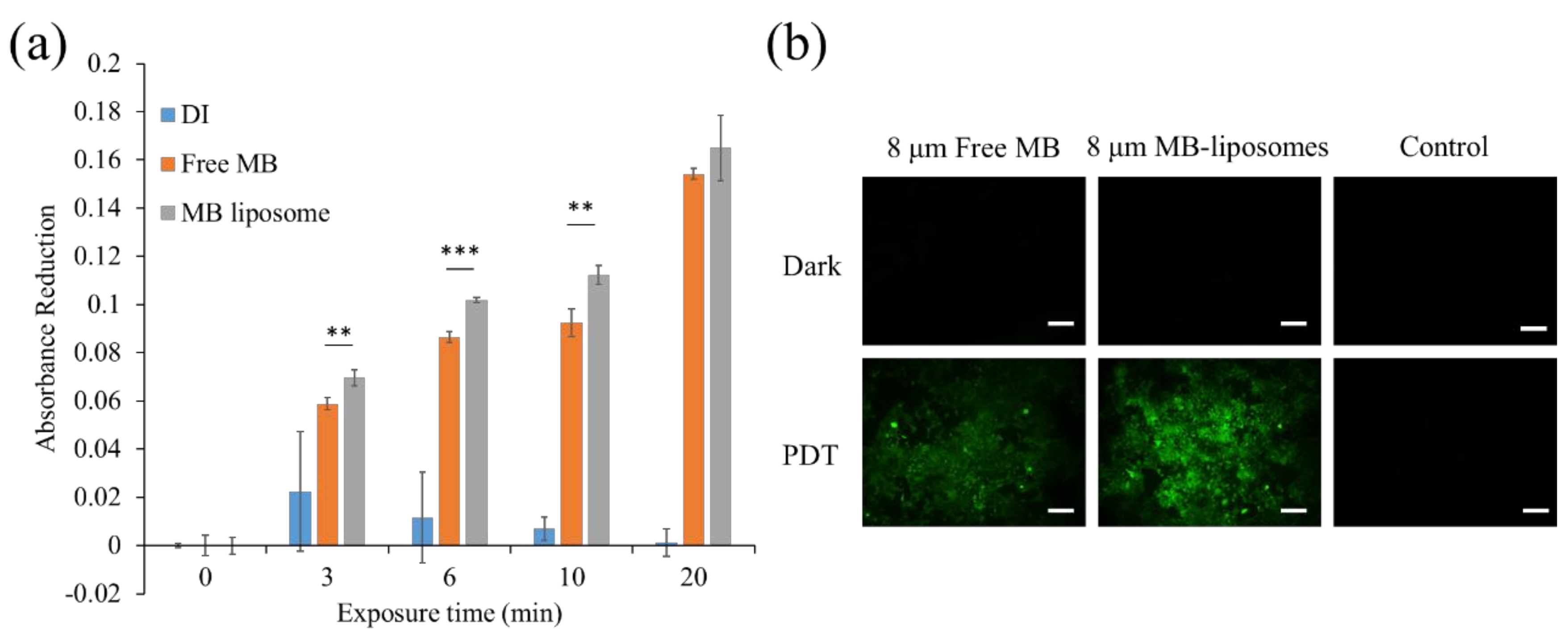
3.2. ROS Generation Ability
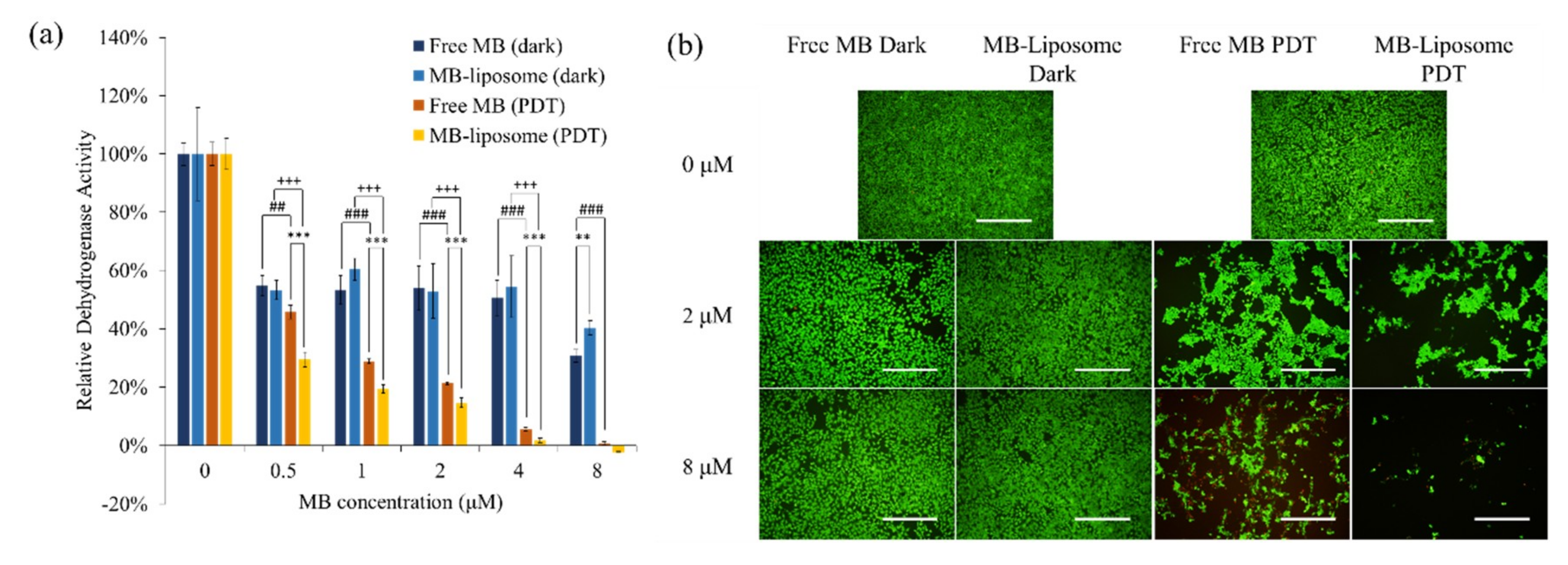
3.3. In Vitro Cytotoxicity Test
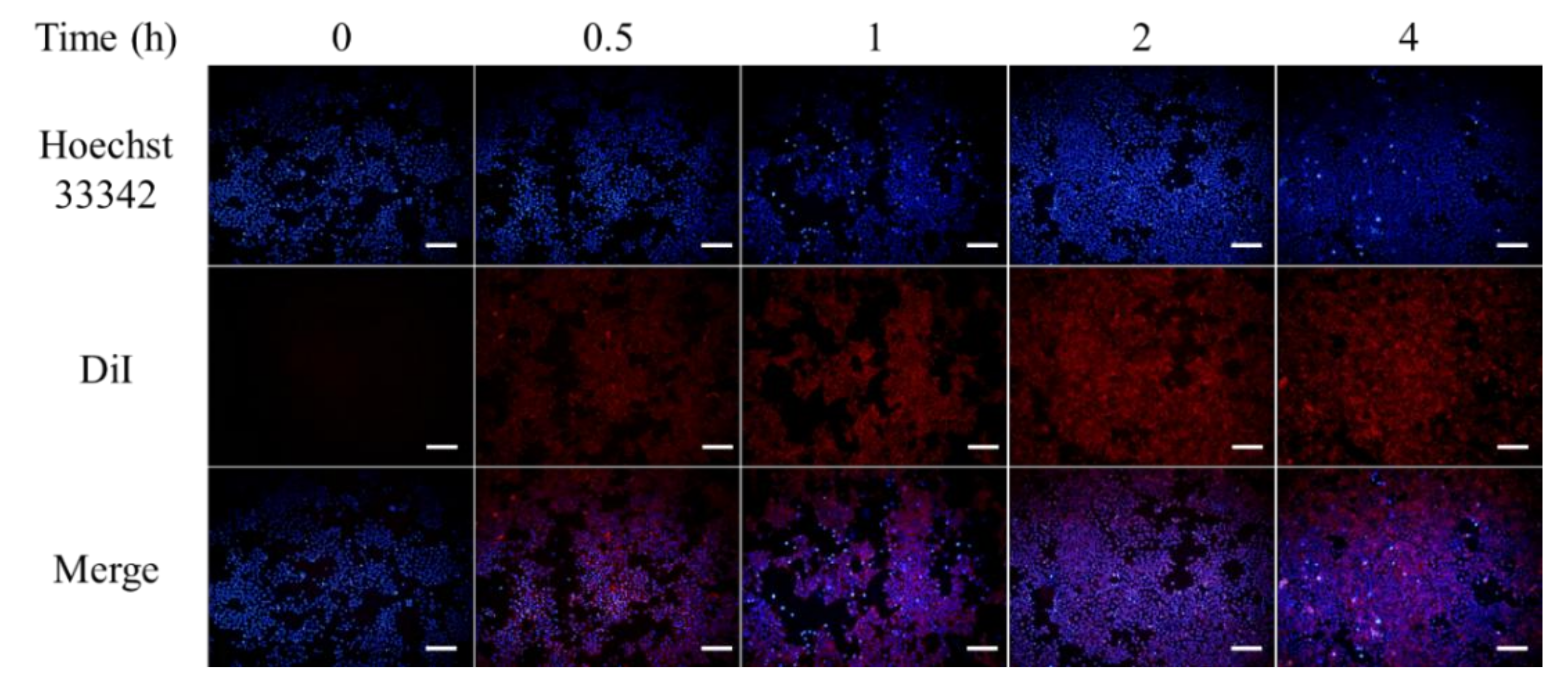
3.4. Intracellular Uptake
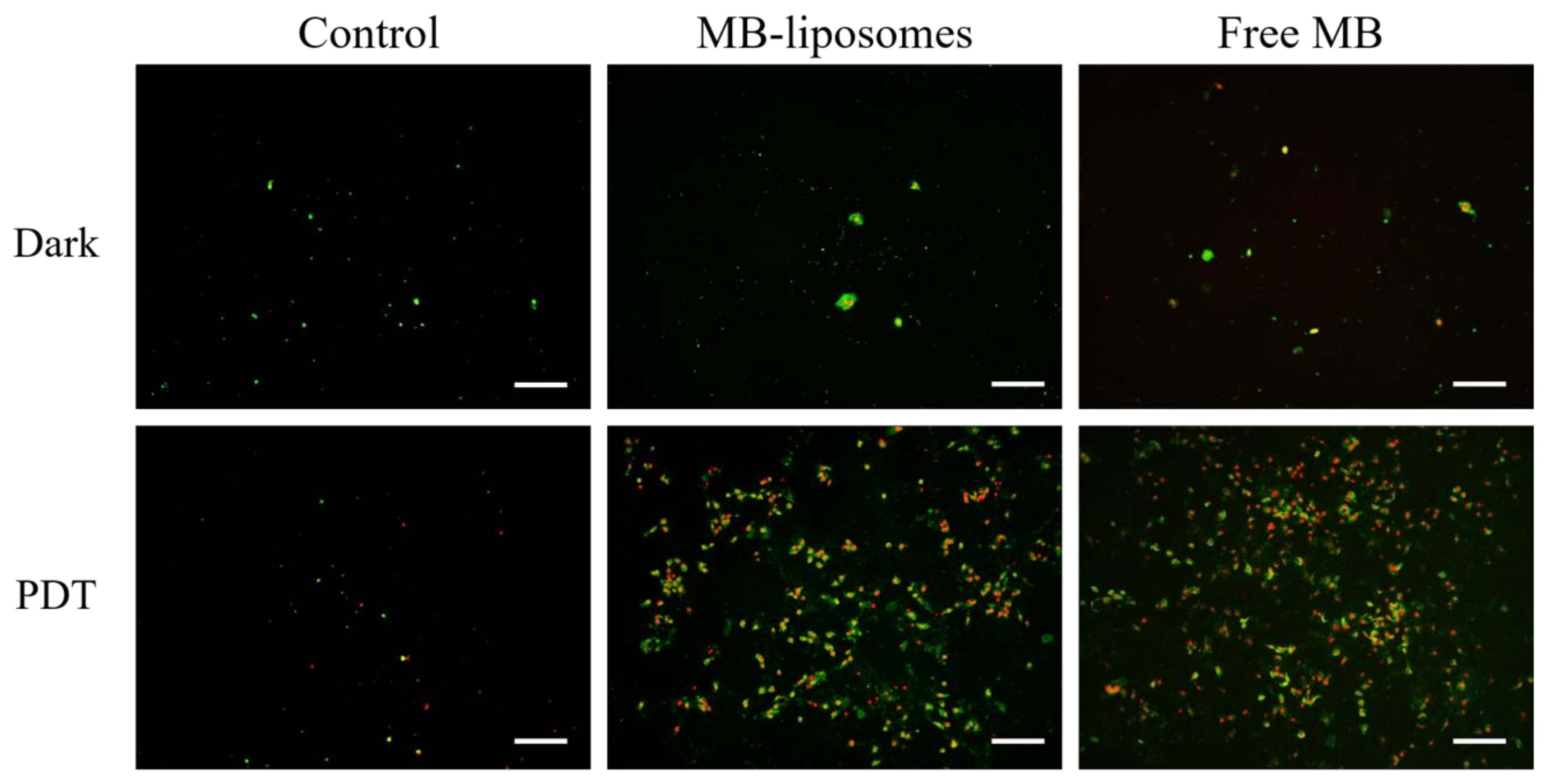
3.5. Identification of Cell Death Pathway
3.6. Survival Rate of Mice Administrated with MB-Liposomes
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Chapman, J.A.; Meng, D.; Shepherd, L.; Parulekar, W.; Ingle, J.N.; Muss, H.B.; Palmer, M.; Yu, C.; Goss, P.E. Competing causes of death from a randomized trial of extended adjuvant endocrine therapy for breast cancer. J. Natl. Cancer Inst. 2008, 100, 252–260. [Google Scholar] [CrossRef] [PubMed]
- Pulaski, B.A.; Ostrand-Rosenberg, S. Mouse 4t1 breast tumor model. Curr. Protoc. Immunol. 2001. [Google Scholar] [CrossRef]
- Liu, L.-H.; Zhang, Y.-H.; Qiu, W.-X.; Zhang, L.; Gao, F.; Li, B.; Xu, L.; Fan, J.-X.; Li, Z.-H.; Zhang, X.-Z. Dual-stage light amplified photodynamic therapy against hypoxic tumor based on an o2 self-sufficient nanoplatform. Small 2017, 13, 1701621. [Google Scholar] [CrossRef] [PubMed]
- Jin, W.J.; Kim, B.; Kim, D.; Park Choo, H.Y.; Kim, H.H.; Ha, H.; Lee, Z.H. Nf-kappab signaling regulates cell-autonomous regulation of cxcl10 in breast cancer 4t1 cells. Exp. Mol. Med. 2017, 49, e295. [Google Scholar] [CrossRef] [PubMed]
- Hsieh, C.C.; Wang, C.H.; Huang, Y.S. Lunasin attenuates obesity-associated metastasis of 4t1 breast cancer cell through anti-inflammatory property. Int. J. Mol. Sci. 2016, 17, 2109. [Google Scholar] [CrossRef] [PubMed]
- Usuda, J.; Tsutsui, H.; Honda, H.; Ichinose, S.; Ishizumi, T.; Hirata, T.; Inoue, T.; Ohtani, K.; Maehara, S.; Imai, K.; et al. Photodynamic therapy for lung cancers based on novel photodynamic diagnosis using talaporfin sodium (npe6) and autofluorescence bronchoscopy. Lung Cancer 2007, 58, 317–323. [Google Scholar] [CrossRef] [PubMed]
- Kiesslich, T.; Gollmer, A.; Maisch, T.; Berneburg, M.; Plaetzer, K. A comprehensive tutorial on in vitro characterization of new photosensitizers for photodynamic antitumor therapy and photodynamic inactivation of microorganisms. BioMed Res. Int. 2013, 2013, 17. [Google Scholar] [CrossRef]
- Debele, T.; Peng, S.; Tsai, H.-C. Drug carrier for photodynamic cancer therapy. Int. J. Mol. Sci. 2015, 16, 22094–22136. [Google Scholar] [CrossRef]
- Moskal, T.L.; Dougherty, T.J.; Urschel, J.D.; Antkowiak, J.G.; Regal, A.-M.; Driscoll, D.L.; Takita, H. Operation and photodynamic therapy for pleural mesothelioma: 6-year follow-up. Ann. Thorac. Surg. 1998, 66, 1128–1133. [Google Scholar] [CrossRef]
- Dougherty, T.J.; Gomer, C.J.; Henderson, B.W.; Jori, G.; Kessel, D.; Korbelik, M.; Moan, J.; Peng, Q. Photodynamic therapy. J. Natl. Cancer Inst. 1998, 90, 889–905. [Google Scholar] [CrossRef]
- Saneesh Babu, P.S.; Manu, P.M.; Dhanya, T.J.; Tapas, P.; Meera, R.N.; Surendran, A.; Aneesh, K.A.; Vadakkancheril, S.J.; Ramaiah, D.; Nair, S.A.; et al. Bis(3,5-diiodo-2,4,6-trihydroxyphenyl)squaraine photodynamic therapy disrupts redox homeostasis and induce mitochondria-mediated apoptosis in human breast cancer cells. Sci. Rep. 2017, 7, 42126. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, A.; Ohtani, N.; Yamakoshi, K.; Iida, S.-i.; Tahara, H.; Nakayama, K.; Nakayama, K.I.; Ide, T.; Saya, H.; Hara, E. Mitogenic signalling and the p16ink4a–rb pathway cooperate to enforce irreversible cellular senescence. Nat. Cell Biol. 2006, 8, 1291. [Google Scholar] [CrossRef] [PubMed]
- Ramsey, M.R.; Sharpless, N.E. Ros as a tumour suppressor? Nat. Cell Biol. 2006, 8, 1213. [Google Scholar] [CrossRef] [PubMed]
- May, J.M.; Qu, Z.-C.; Cobb, C.E. Reduction and uptake of methylene blue by human erythrocytes. Am. J. Physiol. Cell Physiol. 2004, 286, C1390–C1398. [Google Scholar] [CrossRef] [PubMed]
- Konan, Y.N.; Gurny, R.; Allémann, E. State of the art in the delivery of photosensitizers for photodynamic therapy. J. Photochem. Photobiol. B Biol. 2002, 66, 89–106. [Google Scholar] [CrossRef]
- Yan, F.; Zhang, Y.; Kim, K.S.; Yuan, H.-K.; Vo-Dinh, T. Cellular uptake and photodynamic activity of protein nanocages containing methylene blue photosensitizing drug. Photochem. Photobiol. 2010, 86, 662–666. [Google Scholar] [CrossRef] [PubMed]
- Weissleder, R. A clearer vision for in vivo imaging. Nat. Biotechnol. 2001, 19, 316. [Google Scholar] [CrossRef] [PubMed]
- Boccalini, G.; Conti, L.; Montis, C.; Bani, D.; Bencini, A.; Berti, D.; Giorgi, C.; Mengoni, A.; Valtancoli, B. Methylene blue-containing liposomes as new photodynamic anti-bacterial agents. J. Mater. Chem. B 2017, 5, 2788–2797. [Google Scholar] [CrossRef]
- Yu, J.; Hsu, C.-H.; Huang, C.-C.; Chang, P.-Y. Development of therapeutic au–methylene blue nanoparticles for targeted photodynamic therapy of cervical cancer cells. ACS Appl. Mater. Interfaces 2015, 7, 432–441. [Google Scholar] [CrossRef] [PubMed]
- Aoyama, T.; Fujikawa, H.; Cho, H.; Ogata, T.; Shirai, J.; Hayashi, T.; Rino, Y.; Masuda, M.; Oba, M.S.; Morita, S.; et al. A methylene blue-assisted technique for harvesting lymph nodes after radical surgery for gastric cancer: A prospective, randomized, controlled study. Am. J. Surg. Pathol. 2015, 39, 266–273. [Google Scholar] [CrossRef]
- Lyon, J.P.; Rezende, R.R.; Rabelo, M.P.; de Lima, C.J.; Moreira, L.M. Synergic effect of photodynamic therapy with methylene blue and surfactants in the inhibition of candida albicans. Mycopathologia 2013, 175, 159–164. [Google Scholar] [CrossRef] [PubMed]
- Konig, K. Multiphoton microscopy in life sciences. J. Microsc. 2000, 200, 83–104. [Google Scholar] [CrossRef] [PubMed]
- Tian, G.; Gu, Z.; Zhou, L.; Yin, W.; Liu, X.; Yan, L.; Jin, S.; Ren, W.; Xing, G.; Li, S.; et al. Mn2+ dopant-controlled synthesis of nayf4:Yb/er upconversion nanoparticles for in vivo imaging and drug delivery. Adv. Mater. 2012, 24, 1226–1231. [Google Scholar] [CrossRef] [PubMed]
- Guan, J.; Lai, X.; Wang, X.; Leung, A.W.; Zhang, H.; Xu, C. Photodynamic action of methylene blue in osteosarcoma cells in vitro. Photodiagnosis Photodyn. Ther. 2014, 11, 13–19. [Google Scholar] [CrossRef] [PubMed]
- Liu, T.-M.; Yu, J.; Chang, C.A.; Chiou, A.; Chiang, H.K.; Chuang, Y.-C.; Wu, C.-H.; Hsu, C.-H.; Chen, P.-A.; Huang, C.-C. One-step shell polymerization of inorganic nanoparticles and their applications in sers/nonlinear optical imaging, drug delivery, and catalysis. Sci. Rep. 2014, 4, 5593. [Google Scholar] [CrossRef] [PubMed]
- Jiang, W.; Kim, B.Y.S.; Rutka, J.T.; Chan, W.C.W. Nanoparticle-mediated cellular response is size-dependent. Nat. Nanotechnol. 2008, 3, 145. [Google Scholar] [CrossRef]
- Asati, A.; Santra, S.; Kaittanis, C.; Perez, J.M. Surface-charge-dependent cell localization and cytotoxicity of cerium oxide nanoparticles. ACS Nano 2010, 4, 5321–5331. [Google Scholar] [CrossRef]
- El-Sayed, I.H.; Huang, X.; El-Sayed, M.A. Surface plasmon resonance scattering and absorption of anti-EGFR antibody conjugated gold nanoparticles in cancer diagnostics: Applications in oral cancer. Nano Lett. 2005, 5, 829–834. [Google Scholar] [CrossRef]
- Zhang, S.; Li, J.; Lykotrafitis, G.; Bao, G.; Suresh, S. Size-dependent endocytosis of nanoparticles. Adv. Mater. 2009, 21, 419–424. [Google Scholar] [CrossRef]
- Liu, X.; Chen, Y.; Li, H.; Huang, N.; Jin, Q.; Ren, K.; Ji, J. Enhanced retention and cellular uptake of nanoparticles in tumors by controlling their aggregation behavior. ACS Nano 2013, 7, 6244–6257. [Google Scholar] [CrossRef]
- Tai, Y.-W.; Chiu, Y.-C.; Wu, P.-T.; Yu, J.; Chin, Y.-C.; Wu, S.-P.; Chuang, Y.-C.; Hsieh, H.-C.; Lai, P.-S.; Yu, H.-P.; et al. Degradable nir-ptt nanoagents with a potential cu@cu2o@polymer structure. ACS Appl. Mater. Interfaces 2018, 10, 5161–5174. [Google Scholar] [CrossRef] [PubMed]
- Pisanic Ii, T.R.; Zhang, Y.; Wang, T.H. Quantum dots in diagnostics and detection: Principles and paradigms. Analyst 2014, 139, 2968–2981. [Google Scholar] [CrossRef] [PubMed]
- Zhao, C.; Bai, Z.; Liu, X.; Zhang, Y.; Zou, B.; Zhong, H. Small gsh-capped cuins2 quantum dots: Mpa-assisted aqueous phase transfer and bioimaging applications. ACS Appl. Mater. Interfaces 2015, 7, 17623–17629. [Google Scholar] [CrossRef] [PubMed]
- Kao, C.-W.; Wu, P.-T.; Liao, M.-Y.; Chung, I.-J.; Yang, K.-C.; Tseng, W.-Y.; Yu, J. Magnetic nanoparticles conjugated with peptides derived from monocyte chemoattractant protein-1 as a tool for targeting atherosclerosis. Pharmaceutics 2018, 10, 62. [Google Scholar] [CrossRef] [PubMed]
- Jayaram, D.T.; Ramos-Romero, S.; Shankar, B.H.; Garrido, C.; Rubio, N.; Sanchez-Cid, L.; Gómez, S.B.; Blanco, J.; Ramaiah, D. In vitro and in vivo demonstration of photodynamic activity and cytoplasm imaging through tpe nanoparticles. ACS Chem. Biol. 2016, 11, 104–112. [Google Scholar] [CrossRef] [PubMed]
- Un, K.; Sakai-Kato, K.; Oshima, Y.; Kawanishi, T.; Okuda, H. Intracellular trafficking mechanism, from intracellular uptake to extracellular efflux, for phospholipid/cholesterol liposomes. Biomaterials 2012, 33, 8131–8141. [Google Scholar] [CrossRef] [PubMed]
- Sessa, G.; Weissmann, G. Phospholipid spherules (liposomes) as a model for biological membranes. J. Lipid Res. 1968, 9, 310–318. [Google Scholar]
- Sercombe, L.; Veerati, T.; Moheimani, F.; Wu, S.Y.; Sood, A.K.; Hua, S. Advances and challenges of liposome assisted drug delivery. Front. Pharmacol. 2015, 6. [Google Scholar] [CrossRef]
- Rajendrakumar, S.; Chang, N.-C.; Mohapatra, A.; Uthaman, S.; Lee, B.-I.; Tsai, W.-b.; Park, I.-K. A lipophilic ir-780 dye-encapsulated zwitterionic polymer-lipid micellar nanoparticle for enhanced photothermal therapy and nir-based fluorescence imaging in a cervical tumor mouse model. Int. J. Mol. Sci. 2018, 19, 1189. [Google Scholar] [CrossRef]
- Kawanishi, M.; Hashimoto, Y.; Shimizu, T.; Sagawa, I.; Ishida, T.; Kiwada, H. Comprehensive analysis of pegylated liposome-associated proteins relating to the accelerated blood clearance phenomenon by combination with shotgun analysis and conventional methods. Biotechnol. Appl. Biochem. 2015, 62, 547–555. [Google Scholar] [CrossRef]
- Akbarzadeh, A.; Rezaei-Sadabady, R.; Davaran, S.; Joo, S.W.; Zarghami, N.; Hanifehpour, Y.; Samiei, M.; Kouhi, M.; Nejati-Koshki, K. Liposome: Classification, preparation, and applications. Nanoscale Res. Lett. 2013, 8, 102. [Google Scholar] [CrossRef] [PubMed]
- Allen, T.M.; Cullis, P.R. Liposomal drug delivery systems: From concept to clinical applications. Adv. Drug Del. Rev. 2013, 65, 36–48. [Google Scholar] [CrossRef] [PubMed]
- Qiao, C.; Liu, J.; Yang, J.; Li, Y.; Weng, J.; Shao, Y.; Zhang, X. Enhanced non-inflammasome mediated immune responses by mannosylated zwitterionic-based cationic liposomes for hiv DNA vaccines. Biomaterials 2016, 85, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.-R.; Bang, D.; Ahn, D.-R. Nano-formulation of a photosensitizer using a DNA tetrahedron and its potential for in vivo photodynamic therapy. Biomater. Sci. 2016, 4, 605–609. [Google Scholar] [CrossRef] [PubMed]
- Dos Santos, M.S.C.; Gouvêa, A.L.; de Moura, L.D.; Paterno, L.G.; de Souza, P.E.N.; Bastos, A.P.; Damasceno, E.A.M.; Veiga-Souza, F.H.; de Azevedo, R.B.; Báo, S.N. Nanographene oxide-methylene blue as phototherapies platform for breast tumor ablation and metastasis prevention in a syngeneic orthotopic murine model. J. Nanobiotechnol. 2018, 16, 9. [Google Scholar] [CrossRef] [PubMed]
- Baran, T.M.; Giesselman, B.R.; Hu, R.; Biel, M.A.; Foster, T.H. Factors influencing tumor response to photodynamic therapy sensitized by intratumor administration of methylene blue. Lasers Surg. Med. 2010, 42, 728–735. [Google Scholar] [CrossRef] [PubMed]
- Linetsky, M.; Ortwerth, B.J. Quantitation of the reactive oxygen species generated by the uva irradiation of ascorbic acid-glycated lens proteins. Photochem. Photobiol. 1996, 63, 649–655. [Google Scholar] [CrossRef] [PubMed]
- Curtin, J.F.; Donovan, M.; Cotter, T.G. Regulation and measurement of oxidative stress in apoptosis. J. Immunol. Methods 2002, 265, 49–72. [Google Scholar] [CrossRef]
- Yoo, D.; Jeong, H.; Preihs, C.; Choi, J.-s.; Shin, T.-H.; Sessler, J.L.; Cheon, J. Double-effector nanoparticles: A synergistic approach to apoptotic hyperthermia. Angew. Chem. Int. Ed. 2012, 51, 12482–12485. [Google Scholar] [CrossRef]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wu, P.-T.; Lin, C.-L.; Lin, C.-W.; Chang, N.-C.; Tsai, W.-B.; Yu, J. Methylene-Blue-Encapsulated Liposomes as Photodynamic Therapy Nano Agents for Breast Cancer Cells. Nanomaterials 2019, 9, 14. https://doi.org/10.3390/nano9010014
Wu P-T, Lin C-L, Lin C-W, Chang N-C, Tsai W-B, Yu J. Methylene-Blue-Encapsulated Liposomes as Photodynamic Therapy Nano Agents for Breast Cancer Cells. Nanomaterials. 2019; 9(1):14. https://doi.org/10.3390/nano9010014
Chicago/Turabian StyleWu, Po-Ting, Chih-Ling Lin, Che-Wei Lin, Ning-Chu Chang, Wei-Bor Tsai, and Jiashing Yu. 2019. "Methylene-Blue-Encapsulated Liposomes as Photodynamic Therapy Nano Agents for Breast Cancer Cells" Nanomaterials 9, no. 1: 14. https://doi.org/10.3390/nano9010014
APA StyleWu, P.-T., Lin, C.-L., Lin, C.-W., Chang, N.-C., Tsai, W.-B., & Yu, J. (2019). Methylene-Blue-Encapsulated Liposomes as Photodynamic Therapy Nano Agents for Breast Cancer Cells. Nanomaterials, 9(1), 14. https://doi.org/10.3390/nano9010014






