Nano- and Microplastics in the Cardiovascular System: Current Insights and Biological Implications
Abstract
1. Introduction
2. Micro- and Nanoplastics as Emerging Pollutants: Biological Evidence and Analytical Approaches
2.1. Differences Between Microplastics (>1 µm) and Nanoplastics (<1 µm) in Cellular Uptake and Toxicity
2.2. Clinical Evidence of the Presence of Micro- and Nanoplastics in Cardiac and Vascular Tissue: Clinical Findings
2.3. Detection Techniques for MNPs: Composition, Molecular, Structure, and Function Levels
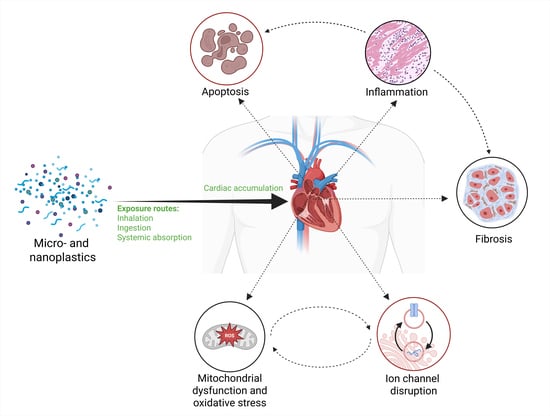
3. Mechanisms of MNPs Translocation, Distribution and Cardiac Accumulations
3.1. Mechanisms of MNPs Translocation and Transport in the Bloodstream
3.2. Mechanisms of MNP Accumulation in the Myocardium
4. Cardiovascular Toxicity Mechanisms
4.1. Integrated Oxidative, Inflammatory, and Cell Death Mechanisms Underlying MNP-Induced Cardiotoxicity
4.2. Oxidative and ER Stress-Mediated Ion Channel Dysregulation by MNPs Causing Cardiac Arrhythmias and Necrosis
4.3. Immune and Inflammatory Mechanisms Triggered by MNPs in the Myocardium
5. Clinical Implications
5.1. Endothelial Damage and Vascular Dysfunction
5.2. Contribution to Atherosclerosis and Arterial Stiffness
5.3. Potential Role of MNPs in Cardiomyopathies
6. Therapeutic Perspectives and Prevention Strategies
7. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ho, K.T.; Bjorkland, R.; Burgess, R.M. Comparing the Definitions of Microplastics Based on Size Range: Scientific and Policy Implications. Mar. Pollut. Bull. 2024, 207, 116907. [Google Scholar] [CrossRef]
- Lett, Z.; Hall, A.; Skidmore, S.; Alves, N.J. Environmental Microplastic and Nanoplastic: Exposure Routes and Effects on Coagulation and the Cardiovascular System. Environ. Pollut. 2021, 291, 118190. [Google Scholar] [CrossRef] [PubMed]
- Irfan, H.; Irfan, H.; Khan, M.A.; Inanc, O.; Hasibuzzaman, M.A. Microplastics and Nanoplastics: Emerging Threats to Cardiovascular Health—A Comprehensive Review. Ann. Med. Surg. 2025, 87, 209–216. [Google Scholar] [CrossRef]
- Wu, Y.; Li, Z.; Shi, L.; Zhu, Y.; Wang, Y.; Yan, N.; Yang, Y.; He, S.; Li, J. Effects of Leachate from Disposable Plastic Takeout Containers on the Cardiovascular System after Thermal Contact. Ecotoxicol. Environ. Saf. 2024, 288, 117383. [Google Scholar] [CrossRef]
- Oraee, S.; Alinejadfard, M.; Mahdavi, S.; Rajai Firouzabadi, S.; Hatami, S.; Mohammadi, I.; Zarinfar, Y.; Khaheshi, I. The Association between Micro- and Nano-Plastics and Cardiovascular Outcomes: A Comprehensive Review. Toxicol. Environ. Health Sci. 2025, 17, 203–218. [Google Scholar] [CrossRef]
- Prattichizzo, F.; Ceriello, A.; Pellegrini, V.; La Grotta, R.; Graciotti, L.; Olivieri, F.; Paolisso, P.; D’Agostino, B.; Iovino, P.; Balestrieri, M.L.; et al. Micro-Nanoplastics and Cardiovascular Diseases: Evidence and Perspectives. Eur. Heart J. 2024, 45, 4099–4110. [Google Scholar] [CrossRef]
- Liang, J.; Ji, F.; Abdullah, A.L.B.; Qin, W.; Zhu, T.; Tay, Y.J.; Li, Y.; Han, M. Micro/Nano-Plastics Impacts in Cardiovascular Systems across Species. Sci. Total Environ. 2024, 942, 173770. [Google Scholar] [CrossRef]
- Symeonides, C.; Aromataris, E.; Mulders, Y.; Dizon, J.; Stern, C.; Barker, T.H.; Whitehorn, A.; Pollock, D.; Marin, T.; Dunlop, S. An Umbrella Review of Meta-Analyses Evaluating Associations between Human Health and Exposure to Major Classes of Plastic-Associated Chemicals. Ann. Glob. Health 2024, 90, 52. [Google Scholar] [CrossRef]
- Hyman, S.; Acevedo, J.; Giannarelli, C.; Trasande, L. Phthalate Exposure from Plastics and Cardiovascular Disease: Global Estimates of Attributable Mortality and Years Life Lost. eBioMedicine 2025, 117, 105730. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.-H.; Zheng, C.-M.; Wang, Y.-J.; Wang, Y.-L.; Chiu, H.-W. Effects of Microplastics and Nanoplastics on the Kidney and Cardiovascular System. Nat. Rev. Nephrol. 2025, 21, 585–596. [Google Scholar] [CrossRef] [PubMed]
- Goldsworthy, A.; O’Callaghan, L.; Blum, C.; Horobin, J.; Tajouri, L.; Olsen, M.; Bruggen, N.V.D.; McKirdy, S.; Alghafri, R.; Tronstad, O.; et al. Micro-Nanoplastic Induced Cardiovascular Disease and Dysfunction: A Scoping Review. J. Expo. Sci. Environ. Epidemiol. 2025, 35, 746–769. [Google Scholar] [CrossRef]
- Lerchner, T.; Jedlička, J.; Kripnerová, M.; Dejmek, J.; Kuncová, J. Influence of Micro- and Nanoplastics on Mitochondrial Function in the Cardiovascular System: A Review of the Current Literature. Physiol. Res. 2024, 73, S685–S695. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Gao, Q.; Gao, Q.; Xu, M.; Fang, N.; Mu, L.; Han, X.; Yu, H.; Zhang, S.; Li, Y.; et al. Microplastics and Nanoplastics Increase Major Adverse Cardiac Events in Patients with Myocardial Infarction. J. Hazard. Mater. 2025, 489, 137624. [Google Scholar] [CrossRef]
- Rafa, N.; Ahmed, B.; Zohora, F.; Bakya, J.; Ahmed, S.; Ahmed, S.F.; Mofijur, M.; Chowdhury, A.A.; Almomani, F. Microplastics as Carriers of Toxic Pollutants: Source, Transport, and Toxicological Effects. Environ. Pollut. 2024, 343, 123190. [Google Scholar] [CrossRef]
- Lu, Y.; Cao, M.; Tian, M.; Huang, Q. Internalization and Cytotoxicity of Polystyrene Microplastics in Human Umbilical Vein Endothelial Cells. J. Appl. Toxicol. 2023, 43, 262–271. [Google Scholar] [CrossRef] [PubMed]
- Rashid, S.; Majeed, L.R.; Mehta, N.; Radu, T.; Martín-Fabiani, I.; Bhat, M.A. Microplastics in Terrestrial Ecosystems: Sources, Transport, Fate, Mitigation, and Remediation Strategies. Euro-Mediterr. J. Environ. Integr. 2025, 10, 2633–2659. [Google Scholar] [CrossRef]
- Li, Y.; Tao, L.; Wang, Q.; Wang, F.; Li, G.; Song, M. Potential Health Impact of Microplastics: A Review of Environmental Distribution, Human Exposure, and Toxic Effects. Environ. Health 2023, 1, 249–257. [Google Scholar] [CrossRef]
- Ateia, M.; Ersan, G.; Alalm, M.G.; Boffito, D.C.; Karanfil, T. Emerging Investigator Series: Microplastic Sources, Fate, Toxicity, Detection, and Interactions with Micropollutants in Aquatic Ecosystems—A Review of Reviews. Environ. Sci. Process. Impacts 2022, 24, 172–195. [Google Scholar] [CrossRef]
- Karak, P.; Parveen, A.; Modak, A.; Adhikari, A.; Chakrabortty, S. Microplastic Pollution: A Global Environmental Crisis Impacting Marine Life, Human Health, and Potential Innovative Sustainable Solutions. Int. J. Environ. Res. Public. Health 2025, 22, 889. [Google Scholar] [CrossRef]
- McHale, M.E.; Sheehan, K.L. Bioaccumulation, Transfer, and Impacts of Microplastics in Aquatic Food Chains. J. Environ. Expo. Assess. 2024, 3, 15. [Google Scholar] [CrossRef]
- Salehi, M.; Pincus, L.N.; Deng, B.; Peters, C.A. Microplastics: From Intrinsic Properties to Environmental Fate. Environ. Eng. Sci. 2024, 41, 425–435. [Google Scholar] [CrossRef]
- Haque, F.; Fan, C. Fate and Impacts of Microplastics in the Environment: Hydrosphere, Pedosphere, and Atmosphere. Environments 2023, 10, 70. [Google Scholar] [CrossRef]
- Boctor, J.; Hoyle, F.C.; Farag, M.A.; Ebaid, M.; Walsh, T.; Whiteley, A.S.; Murphy, D.V. Microplastics and Nanoplastics: Fate, Transport, and Governance from Agricultural Soil to Food Webs and Humans. Environ. Sci. Eur. 2025, 37, 68. [Google Scholar] [CrossRef]
- Gouin, T.; Ellis-Hutchings, R.; Pemberton, M.; Wilhelmus, B. Addressing the Relevance of Polystyrene Nano- and Microplastic Particles Used to Support Exposure, Toxicity and Risk Assessment: Implications and Recommendations. Part. Fibre Toxicol. 2024, 21, 39. [Google Scholar] [CrossRef]
- Khan, A.; Jia, Z. Recent Insights into Uptake, Toxicity, and Molecular Targets of Microplastics and Nanoplastics Relevant to Human Health Impacts. iScience 2023, 26, 106061. [Google Scholar] [CrossRef]
- Baroni, A.; Moulton, C.; Cristina, M.; Sansone, L.; Belli, M.; Tasciotti, E. Nano- and Microplastics in the Brain: An Emerging Threat to Neural Health. Nanomaterials 2025, 15, 1361. [Google Scholar] [CrossRef]
- Bridgeman, L.; Cimbalo, A.; López-Rodríguez, D.; Pamies, D.; Frangiamone, M. Exploring Toxicological Pathways of Microplastics and Nanoplastics: Insights from Animal and Cellular Models. J. Hazard. Mater. 2025, 490, 137795. [Google Scholar] [CrossRef]
- Sun, W.; Jin, C.; Bai, Y.; Ma, R.; Deng, Y.; Gao, Y.; Pan, G.; Yang, Z.; Yan, L. Blood Uptake and Urine Excretion of Nano- and Micro-Plastics after a Single Exposure. Sci. Total Environ. 2022, 848, 157639. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.; Cho, S.; Park, K.; Kim, T.; Kim, J.; Ryu, D.-Y.; Hong, J. Potential Lifetime Effects Caused by Cellular Uptake of Nanoplastics: A Review. Environ. Pollut. 2023, 329, 121668. [Google Scholar] [CrossRef]
- Barreto, A.; Santos, J.; Calisto, V.; Rocha, L.S.; Amorim, M.J.B.; Maria, V.L. Cocktail Effects of Emerging Contaminants on Zebrafish: Nanoplastics and the Pharmaceutical Diphenhydramine. NanoImpact 2023, 30, 100456. [Google Scholar] [CrossRef]
- De Oro-Carretero, P.; Garcia-Ordoñez, M.; Roher, N.; Sanz-Landaluze, J. Complex Combined Effects of Polystyrene Nanoplastics and Phenanthrene in Aquatic Models. J. Hazard. Mater. 2025, 496, 139356. [Google Scholar] [CrossRef]
- Mahmud, F.; Sarker, D.B.; Jocelyn, J.A.; Sang, Q.-X.A. Molecular and Cellular Effects of Microplastics and Nanoplastics: Focus on Inflammation and Senescence. Cells 2024, 13, 1788. [Google Scholar] [CrossRef]
- Marfella, R.; Prattichizzo, F.; Sardu, C.; Fulgenzi, G.; Graciotti, L.; Spadoni, T.; D’Onofrio, N.; Scisciola, L.; La Grotta, R.; Frigé, C.; et al. Microplastics and Nanoplastics in Atheromas and Cardiovascular Events. N. Engl. J. Med. 2024, 390, 900–910. [Google Scholar] [CrossRef] [PubMed]
- Geppner, L.; Hellner, J.; Henjakovic, M. Effects of Micro- and Nanoplastics on Blood Cells in Vitro and Cardiovascular Parameters in Vivo, Considering Their Presence in the Human Bloodstream and Potential Impact on Blood Pressure. Environ. Res. 2025, 273, 121254. [Google Scholar] [CrossRef] [PubMed]
- Nouri, M.; Massahi, T.; Hossini, H. Microplastics in Human Body: A Narrative on Routes of Exposure to Contamination and Potential Health Effects. Environ. Pollut. Bioavailab. 2025, 37, 2538535. [Google Scholar] [CrossRef]
- Santos, F.A.; Andre, R.S.; Alvarenga, A.D.; Alves, A.L.M.M.; Correa, D.S. Micro- and Nanoplastics in the Environment: A Comprehensive Review on Detection Techniques. Environ. Sci. Nano 2025, 12, 3442–3467. [Google Scholar] [CrossRef]
- Nene, A.; Sadeghzade, S.; Viaroli, S.; Yang, W.; Uchenna, U.P.; Kandwal, A.; Liu, X.; Somani, P.; Galluzzi, M. Recent Advances and Future Technologies in Nano-Microplastics Detection. Environ. Sci. Eur. 2025, 37, 7. [Google Scholar] [CrossRef]
- Sansone, L.; De Iure, A.; Cristina, M.; Belli, M.; Vitiello, L.; Marcolongo, F.; Rosellini, A.; Macera, L.; Spezia, P.G.; Tomino, C.; et al. Nicotine in Combination with SARS-CoV-2 Affects Cells Viability, Inflammatory Response and Ultrastructural Integrity. Int. J. Mol. Sci. 2022, 23, 9488. [Google Scholar] [CrossRef]
- Belli, M.; Cristina, M.; Calabrese, V.; Russo, M.; Granato, M.; Russo, M.A.; Sansone, L. Ultrastructural Changes of Neuroendocrine Pheochromocytoma Cell Line PC-12 Exposed In Vitro to Rotenone. Brain Sci. 2024, 14, 476. [Google Scholar] [CrossRef] [PubMed]
- Zhu, H.; Qian, H.; Yang, Z. Healthful Diet and Nutritional Food as a Preventive and Interventional Paradigm in the Face of Microplastic and Nanoplastic Crisis. Food Front. 2025, 6, 2511–2513. [Google Scholar] [CrossRef]
- Pulusu, V.S.; Chilamula, S.; Holkunde, A.; Gunturi, R.; Vidiyala, P.; Anekalla, T.R. Microplastics in the Environment: Sources, Detection Techniques, and Analytical Challenges. Open Access Libr. J. 2025, 12, 1–33. [Google Scholar] [CrossRef]
- Ma, J.; Zhao, S.; He, K.; Tian, L.; Zhong, G.; Jones, K.C.; Sweetman, A.J.; Li, J.; Zhou, Q.; Chen, D.; et al. Quantification of Micro- and Nano-Plastics in Atmospheric Fine Particles by Pyrolysis-Gas Chromatography-Mass Spectrometry with Chromatographic Peak Reconstruction. J. Hazard. Mater. 2025, 488, 137292. [Google Scholar] [CrossRef]
- Abbas, Q.; Zia-ur-Rehman, M.; Ullah, H.; Mujtaba Munir, M.A.; Ali, M.U.; Ali, A.; Pikon, K.; Yousaf, B. Recent Advances in the Detection and Quantification of Manufactured Nanoparticles (MNPs) in Complex Environmental and Biological Matrices. J. Clean. Prod. 2024, 471, 143454. [Google Scholar] [CrossRef]
- Balaban Hanoglu, S.; Harmanci, D.; Ucar, N.; Evran, S.; Timur, S. Recent Approaches in Magnetic Nanoparticle-Based Biosensors of miRNA Detection. Magnetochemistry 2023, 9, 23. [Google Scholar] [CrossRef]
- Daoutakou, M.; Kintzios, S. Biosensors for Micro- and Nanoplastics Detection: A Review. Chemosensors 2025, 13, 143. [Google Scholar] [CrossRef]
- Rivera-Rivera, D.M.; Quintanilla-Villanueva, G.E.; Luna-Moreno, D.; Sánchez-Álvarez, A.; Rodríguez-Delgado, J.M.; Cedillo-González, E.I.; Kaushik, G.; Villarreal-Chiu, J.F.; Rodríguez-Delgado, M.M. Exploring Innovative Approaches for the Analysis of Micro- and Nanoplastics: Breakthroughs in (Bio)Sensing Techniques. Biosensors 2025, 15, 44. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.; Xie, J.; Gowen, A.; Xu, J.-L. Machine Learning Driven Methodology for Enhanced Nylon Microplastic Detection and Characterization. Sci. Rep. 2024, 14, 3464. [Google Scholar] [CrossRef] [PubMed]
- Beres, N.D.; Burkart, J.; Graf, E.; Zeder, Y.; Dailey, L.A.; Weinzierl, B. Merging Holography, Fluorescence, and Machine Learning for in Situ Continuous Characterization and Classification of Airborne Microplastics. Atmos. Meas. Tech. 2024, 17, 6945–6964. [Google Scholar] [CrossRef]
- Thomas, C.; Spatayeva, T.; Yu, D.; Loh, A.; Yim, U.H.; Yoon, J.-Y. A Comparison of Current Analytical Methods for Detecting Particulate Matter and Micro/Nanoplastics. Appl. Phys. Rev. 2024, 11, 011313. [Google Scholar] [CrossRef]
- Scroccarello, A.; Álvarez-Diduk, R.; Della Pelle, F.; De Carvalho Castro E Silva, C.; Idili, A.; Parolo, C.; Compagnone, D.; Merkoçi, A. One-Step Laser Nanostructuration of Reduced Graphene Oxide Films Embedding Metal Nanoparticles for Sensing Applications. ACS Sens. 2023, 8, 598–609. [Google Scholar] [CrossRef]
- Papamichail, L.; Koch, L.S.; Veerman, D.; Broersen, K.; Van Der Meer, A.D. Organoids-on-a-Chip: Microfluidic Technology Enables Culture of Organoids with Enhanced Tissue Function and Potential for Disease Modeling. Front. Bioeng. Biotechnol. 2025, 13, 1515340. [Google Scholar] [CrossRef] [PubMed]
- Cho, Y.; Kim, H.N. Organoid-Based Platforms for Investigating Microplastic-Induced Human Organ Toxicity. Organoid 2025, 5, e5. [Google Scholar] [CrossRef]
- Crosset-Perrotin, G.; Moraz, A.; Portela, R.; Alcolea-Rodriguez, V.; Burrueco-Subirà, D.; Smith, C.; Bañares, M.A.; Foroutan, H.; Fairbrother, D.H. Production, Labeling, and Applications of Micro- and Nanoplastic Reference and Test Materials. Environ. Sci. Nano 2025, 12, 2911–2964. [Google Scholar] [CrossRef]
- Zhao, B.; Richardson, R.E.; You, F. Advancing Microplastic Analysis in the Era of Artificial Intelligence: From Current Applications to the Promise of Generative AI. Nexus 2024, 1, 100043. [Google Scholar] [CrossRef]
- Khanam, M.M.; Uddin, M.K.; Kazi, J.U. Advances in Machine Learning for the Detection and Characterization of Microplastics in the Environment. Front. Environ. Sci. 2025, 13, 1573579. [Google Scholar] [CrossRef]
- Petersen, E.J.; Barrios, A.C.; Henry, T.B.; Johnson, M.E.; Koelmans, A.A.; Montoro Bustos, A.R.; Matheson, J.; Roesslein, M.; Zhao, J.; Xing, B. Potential Artifacts and Control Experiments in Toxicity Tests of Nanoplastic and Microplastic Particles. Environ. Sci. Technol. 2022, 56, 15192–15206. [Google Scholar] [CrossRef]
- Lamoree, M.H.; Van Boxel, J.; Nardella, F.; Houthuijs, K.J.; Brandsma, S.H.; Béen, F.; Van Duursen, M.B.M. Health Impacts of Microplastic and Nanoplastic Exposure. Nat. Med. 2025, 31, 2873–2887. [Google Scholar] [CrossRef]
- Jahedi, F.; Jaafarzadeh Haghighi Fard, N. Micro- and Nanoplastic Toxicity in Humans: Exposure Pathways, Cellular Effects, and Mitigation Strategies. Toxicol. Rep. 2025, 14, 102043. [Google Scholar] [CrossRef] [PubMed]
- Vojnits, K.; De León, A.; Gibon, J.; Barker, P.; Mahmoudi, M.; Pakpour, S. A Systematic Review of the Potential Neurotoxicity of Micro-and Nanoplastics: The Known and Unknown. Part. Fibre Toxicol. 2025, 22, 29. [Google Scholar] [CrossRef]
- Anik, A.H.; Oishy, M.N.; Murshed, M.F.; Abdullah, R.B.; Khan, M.R. Distribution and Health Impacts of Micro- and Nanoplastics (MNPs) in Human Organs: Accumulation, Translocation, and Toxicity—A Systematic Review of Current Evidence. J. Hazard. Mater. Plast. 2025, 1, 100002. [Google Scholar] [CrossRef]
- Yakovenko, N.; Pérez-Serrano, L.; Segur, T.; Hagelskjaer, O.; Margenat, H.; Le Roux, G.; Sonke, J.E. Human Exposure to PM10 Microplastics in Indoor Air. PLoS ONE 2025, 20, e0328011. [Google Scholar] [CrossRef] [PubMed]
- Eberhard, T.; Casillas, G.; Zarus, G.M.; Barr, D.B. Systematic Review of Microplastics and Nanoplastics in Indoor and Outdoor Air: Identifying a Framework and Data Needs for Quantifying Human Inhalation Exposures. J. Expo. Sci. Environ. Epidemiol. 2024, 34, 185–196. [Google Scholar] [CrossRef] [PubMed]
- Khan, M.S.; Marma, T.U.; Sumi, S.N.; Chisim, A.; Shakib, I.A.; Rana, S. Plastic in Seafood: Are Crustaceans a Gateway to Microplastic Exposure in Humans? Sci. One Health 2025, 4, 100121. [Google Scholar] [CrossRef]
- Al-Mansoori, M.; Harrad, S.; Abdallah, M.A.-E. MPs in Drinking Water and Beverages: Concentrations, Characteristics and Implications for Human Exposure. Emerg. Contam. 2025, 11, 100552. [Google Scholar] [CrossRef]
- Menichetti, A.; Mordini, D.; Montalti, M. Penetration of Microplastics and Nanoparticles Through Skin: Effects of Size, Shape, and Surface Chemistry. J. Xenobiotics 2024, 15, 6. [Google Scholar] [CrossRef]
- Michelini, S.; Mawas, S.; Kurešepi, E.; Barbero, F.; Šimunović, K.; Miremont, D.; Devineau, S.; Schicht, M.; Ganin, V.; Haugen, Ø.P.; et al. Pulmonary Hazards of Nanoplastic Particles: A Study Using Polystyrene in in Vitro Models of the Alveolar and Bronchial Epithelium. J. Nanobiotechnol. 2025, 23, 388. [Google Scholar] [CrossRef]
- Liu, Y.-Y.; Liu, J.; Wu, H.; Zhang, Q.; Tang, X.-R.; Li, D.; Li, C.-S.; Liu, Y.; Cao, A.; Wang, H. Endocytosis, Distribution, and Exocytosis of Polystyrene Nanoparticles in Human Lung Cells. Nanomaterials 2022, 13, 84. [Google Scholar] [CrossRef]
- Wang, Z.; Wang, S.; Liu, S.; Wang, Z.; Li, F.; Bu, Q.; An, X. Polystyrene Microplastics Induce Potential Toxicity through the Gut-Mammary Axis. npj Sci. Food 2025, 9, 139. [Google Scholar] [CrossRef]
- Zhang, L.; Sun, T.; Gong, M.; Zhou, C.; Zhao, Y.; Zhang, W.; Zhang, Z.; Xiao, S.; Yang, X.; Wang, M.; et al. Protein Corona Dynamicity Contributes to Biological Destiny Disparities of Nanoparticles. Mater. Today Bio 2024, 28, 101215. [Google Scholar] [CrossRef]
- Kopatz, V.; Wen, K.; Kovács, T.; Keimowitz, A.S.; Pichler, V.; Widder, J.; Vethaak, A.D.; Hollóczki, O.; Kenner, L. Micro- and Nanoplastics Breach the Blood–Brain Barrier (BBB): Biomolecular Corona’s Role Revealed. Nanomaterials 2023, 13, 1404. [Google Scholar] [CrossRef] [PubMed]
- Chia, S.P.S.; Pang, J.K.S.; Winanto, W.; Soh, B.-S. Nanoplastics Induces Arrhythmia in Human Stem-Cells Derived Cardiomyocytes. Ecotoxicol. Environ. Saf. 2025, 289, 117657. [Google Scholar] [CrossRef]
- Xiong, Z.; Kong, Q.; Hua, J.; Chen, Q.; Wang, D. Cardiotoxicity of Polystyrene Nanoplastics and Associated Mechanism of Myocardial Cell Injury in Mice. Ecotoxicol. Environ. Saf. 2025, 290, 117712. [Google Scholar] [CrossRef]
- Zhang, H.; Cao, H.; Luo, Y.; Cao, Y.; Meng, X.; Wang, X.; Du, R.; Yan, X.; Gao, Y.; Fang, Z.; et al. Polystyrene Nanoplastics Trigger Mitochondrial and Metabolic Reprogramming in Cardiomyocytes: Evidence from Integrated Transcriptomic and Metabolomic Analysis. Ecotoxicol. Environ. Saf. 2026, 312, 119921. [Google Scholar] [CrossRef]
- Zhang, T.; Liao, Y.; Ling, J.; Zhang, J.; Zhang, D.; Yin, X.; Yu, P.; Liu, X. Tiny Trouble: Microplastics, Nanoplastics, and Their Heartfelt Impact on Cardiovascular Health. Cardiovasc. Res. 2025, 121, 992–1010. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Xie, E.; Du, Z.; Peng, Z.; Han, Z.; Li, L.; Zhao, R.; Qin, Y.; Xue, M.; Li, F.; et al. Detection of Various Microplastics in Patients Undergoing Cardiac Surgery. Environ. Sci. Technol. 2023, 57, 10911–10918. [Google Scholar] [CrossRef] [PubMed]
- Chang, J.; Zhu, Y.; Yang, Z.; Wang, Z.; Wang, M.; Chen, L. Airborne Polystyrene Nanoplastics Exposure Leads to Heart Failure via ECM-Receptor Interaction and PI3K/AKT/BCL-2 Pathways. Sci. Total Environ. 2024, 954, 176469. [Google Scholar] [CrossRef]
- Forest, V.; Pourchez, J. Can the Impact of Micro- and Nanoplastics on Human Health Really Be Assessed Using in Vitro Models? A Review of Methodological Issues. Environ. Int. 2023, 178, 108115. [Google Scholar] [CrossRef]
- Li, J.; Weng, H.; Liu, S.; Li, F.; Xu, K.; Wen, S.; Chen, X.; Li, C.; Nie, Y.; Liao, B.; et al. Embryonic Exposure of Polystyrene Nanoplastics Affects Cardiac Development. Sci. Total Environ. 2024, 906, 167406. [Google Scholar] [CrossRef]
- Roshanzadeh, A.; Oyunbaatar, N.-E.; Ganjbakhsh, S.E.; Park, S.; Kim, D.-S.; Kanade, P.P.; Lee, S.; Lee, D.-W.; Kim, E.-S. Exposure to Nanoplastics Impairs Collective Contractility of Neonatal Cardiomyocytes under Electrical Synchronization. Biomaterials 2021, 278, 121175. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Zhu, S.; Liu, Q.; Wei, J.; Jin, Y.; Wang, X.; Zhang, L. Polystyrene Microplastics Cause Cardiac Fibrosis by Activating Wnt/β-Catenin Signaling Pathway and Promoting Cardiomyocyte Apoptosis in Rats. Environ. Pollut. 2020, 265, 115025. [Google Scholar] [CrossRef]
- Wang, K.; Du, Y.; Li, P.; Guan, C.; Zhou, M.; Wu, L.; Liu, Z.; Huang, Z. Nanoplastics Causes Heart Aging/Myocardial Cell Senescence through the Ca2+/mtDNA/cGAS-STING Signaling Cascade. J. Nanobiotechnol. 2024, 22, 96. [Google Scholar] [CrossRef]
- Wei, J.; Wang, X.; Liu, Q.; Zhou, N.; Zhu, S.; Li, Z.; Li, X.; Yao, J.; Zhang, L. The Impact of Polystyrene Microplastics on Cardiomyocytes Pyroptosis through NLRP3 /Caspase-1 Signaling Pathway and Oxidative Stress in Wistar Rats. Environ. Toxicol. 2021, 36, 935–944. [Google Scholar] [CrossRef]
- Liu, G.; Bao, Q.; Zhang, C.; Zhong, Y.; Deng, M.; Huang, Y.; Ye, Z.; Jing, J. PVC Nanoplastics Impair Cardiac Function via Lysosomal and Mitochondrial Dysfunction. Biochem. Biophys. Res. Commun. 2025, 762, 151736. [Google Scholar] [CrossRef] [PubMed]
- Ali, N.; Katsouli, J.; Marczylo, E.L.; Gant, T.W.; Wright, S.; Bernardino De La Serna, J. The Potential Impacts of Micro-and-Nano Plastics on Various Organ Systems in Humans. eBioMedicine 2024, 99, 104901. [Google Scholar] [CrossRef]
- Zhu, X.; Wang, C.; Duan, X.; Liang, B.; Genbo Xu, E.; Huang, Z. Micro- and Nanoplastics: A New Cardiovascular Risk Factor? Environ. Int. 2023, 171, 107662. [Google Scholar] [CrossRef]
- Wang, T.; Yi, Z.; Liu, X.; Cai, Y.; Huang, X.; Fang, J.; Shen, R.; Lu, W.; Xiao, Y.; Zhuang, W.; et al. Multimodal Detection and Analysis of Microplastics in Human Thrombi from Multiple Anatomically Distinct Sites. eBioMedicine 2024, 103, 105118. [Google Scholar] [CrossRef] [PubMed]
- Smyth, E.; Solomon, A.; Vydyanath, A.; Luther, P.K.; Pitchford, S.; Tetley, T.D.; Emerson, M. Induction and Enhancement of Platelet Aggregation in Vitro and in Vivo by Model Polystyrene Nanoparticles. Nanotoxicology 2015, 9, 356–364. [Google Scholar] [CrossRef]
- Lee, S.E.; Yi, Y.; Moon, S.; Yoon, H.; Park, Y.S. Impact of Micro- and Nanoplastics on Mitochondria. Metabolites 2022, 12, 897. [Google Scholar] [CrossRef]
- Gu, X.; Zhang, Z.; Zhao, L.; Lu, L.; Lu, X.; Li, Y.; Gu, T.; Huang, X.; Huang, G.; Liang, Y.; et al. Exposure to Polyethylene Terephthalate Micro(Nano)Plastics Exacerbates Inflammation and Fibrosis after Myocardial Infarction by Reprogramming the Gut and Lung Microbiota and Metabolome. J. Hazard. Mater. 2025, 488, 137410. [Google Scholar] [CrossRef]
- Persiani, E.; Cecchettini, A.; Amato, S.; Ceccherini, E.; Gisone, I.; Sgalippa, A.; Ippolito, C.; Castelvetro, V.; Lomonaco, T.; Vozzi, F. Virgin and Photo-Degraded Microplastics Induce the Activation of Human Vascular Smooth Muscle Cells. Sci. Rep. 2025, 15, 4263. [Google Scholar] [CrossRef] [PubMed]
- Sivakumar, R.; Senghor Kadalangudi Aravaanan, A.; Vellore Mohanakrishnan, V.; Kumar, J. From Environment to Endothelium: The Role of Microplastics in Vascular Aging. Microplastics 2025, 4, 52. [Google Scholar] [CrossRef]
- Li, R.; Wang, X.; Zhang, J.; Hu, Y.; Yang, Y.; Zhang, Y.; Li, B.; Shen, M.; Wang, C.; Yang, Y.; et al. HIF-1α/HO-1-Mediated Ferroptosis Participates in Polystyrene Nanoplastics-Induced Intergenerational Cardiotoxicity. Nano Lett. 2025, 25, 2226–2235. [Google Scholar] [CrossRef]
- Niu, H.; Li, W. Exposure of Polystyrene Nanoplastics Led to Ferroptosis on Cardiomyocytes. Environ. Pollut. 2025, 382, 126685. [Google Scholar] [CrossRef]
- Zhang, T.; Yang, S.; Ge, Y.; Yin, L.; Pu, Y.; Gu, Z.; Chen, Z.; Liang, G. Unveiling the Heart’s Hidden Enemy: Dynamic Insights into Polystyrene Nanoplastic-Induced Cardiotoxicity Based on Cardiac Organoid-on-a-Chip. ACS Nano 2024, 18, 31569–31585. [Google Scholar] [CrossRef]
- Cheng, W.; Chen, H.; Zhou, Y.; You, Y.; Feng, Y.; Wang, Y. Low Dose of Micro-/Nano-Plastics Mixture Induced Cardiac Hypertrophy and Reductive Stress: The Liver-Heart Crosstalk and Hepatic-Cardiac Organoids-on-a-Chip. J. Hazard. Mater. 2025, 490, 137686. [Google Scholar] [CrossRef]
- Weber, A.; Schwiebs, A.; Solhaug, H.; Stenvik, J.; Nilsen, A.M.; Wagner, M.; Relja, B.; Radeke, H.H. Nanoplastics Affect the Inflammatory Cytokine Release by Primary Human Monocytes and Dendritic Cells. Environ. Int. 2022, 163, 107173. [Google Scholar] [CrossRef] [PubMed]
- Mierzejewski, K.; Kurzyńska, A.; Golubska, M.; Całka, J.; Gałęcka, I.; Szabelski, M.; Paukszto, Ł.; Andronowska, A.; Bogacka, I. New Insights into the Potential Effects of PET Microplastics on Organisms via Extracellular Vesicle-Mediated Communication. Sci. Total Environ. 2023, 904, 166967. [Google Scholar] [CrossRef]
- Sulistomo, H.W.; Aditya, M.R.; Muhammad, A.R.; Rahma, O.A. Impacts of Micro- and Nanoplastic Exposure on the Cardiovascular System: A Systematic Review Focused on in Vivo Studies. Life 2024, 17, 2409835. [Google Scholar] [CrossRef]
- Tan, S.; Liang, S.; Zhao, Y.; Pan, X.; Wang, W.; Liao, W.; Yang, X.; Wu, W.; He, Q. The Evolving Interface of Aged Microplastics and Heavy Metals: Implications for Environmental Fate and Toxicity. Environ. Geochem. Health 2026, 48, 98. [Google Scholar] [CrossRef] [PubMed]
- Kovacs, K.; Bodis, J.; Vass, R.A. Microplastics, Endocrine Disruptors, and Oxidative Stress: Mechanisms and Health Implications. Int. J. Mol. Sci. 2025, 27, 399. [Google Scholar] [CrossRef] [PubMed]
- Vlacil, A.-K.; Bänfer, S.; Jacob, R.; Trippel, N.; Kuzu, I.; Schieffer, B.; Grote, K. Polystyrene Microplastic Particles Induce Endothelial Activation. PLoS ONE 2021, 16, e0260181. [Google Scholar] [CrossRef]
- Zhou, Y.; Wu, Q.; Li, Y.; Feng, Y.; Wang, Y.; Cheng, W. Low-Dose of Polystyrene Microplastics Induce Cardiotoxicity in Mice and Human-Originated Cardiac Organoids. Environ. Int. 2023, 179, 108171. [Google Scholar] [CrossRef] [PubMed]
- Leonard, S.V.L.; Liddle, C.R.; Atherall, C.A.; Chapman, E.; Watkins, M.; Calaminus, S.D.J.; Rotchell, J.M. Microplastics in Human Blood: Polymer Types, Concentrations and Characterisation Using μFTIR. Environ. Int. 2024, 188, 108751. [Google Scholar] [CrossRef] [PubMed]
- Mashayekhi-Sardoo, H.; Sepahi, S.; Ghorani-Azam, A.; Askarpour, H.; Johnston, T.P.; Sahebkar, A. Protective Effect of Curcumin against Microplastic and Nanoplastics Toxicity. Int. J. Environ. Health Res. 2025, 35, 1314–1353. [Google Scholar] [CrossRef]
- Żwierełło, W.; Maruszewska, A.; Skórka-Majewicz, M.; Goschorska, M.; Baranowska-Bosiacka, I.; Dec, K.; Styburski, D.; Nowakowska, A.; Gutowska, I. The Influence of Polyphenols on Metabolic Disorders Caused by Compounds Released from Plastics—Review. Chemosphere 2020, 240, 124901. [Google Scholar] [CrossRef] [PubMed]
- Busch, M.; Bredeck, G.; Waag, F.; Rahimi, K.; Ramachandran, H.; Bessel, T.; Barcikowski, S.; Herrmann, A.; Rossi, A.; Schins, R.P.F. Assessing the NLRP3 Inflammasome Activating Potential of a Large Panel of Micro- and Nanoplastics in THP-1 Cells. Biomolecules 2022, 12, 1095. [Google Scholar] [CrossRef]



| Model/Study Type | MNPs Type and Exposure | Possible Cardiac Effects | Mechanisms Involved | Reference |
|---|---|---|---|---|
| In vitro (neonatal ventricular myocytes) | Polystyrene nanoparticles (1–100 nm) | Decreased intracellular Ca2+, mitochondrial membrane potential, and contractile force | Mitochondrial dysfunction and metabolic impairment | [79] |
| In vivo (rat model) | Polystyrene microplastics (oral exposure) | Cardiac fibrosis, apoptosis, and electrical abnormalities | Activation of Wnt/β-catenin and NLRP3/caspase-1 pathways | [80] |
| In vivo (rat, human-equivalent dose) | Mixed MNPs (polystyrene, polyethylene) | Elevated troponin I and CK-MB; increased septal thickness | Mitochondrial DNA damage and activation of the cGAS–STING pathway | [81,82] |
| In vitro (HL-1 cardiomyocytes) | PVC nanoplastics (1–100 nm; 1–5 µg/mL) | Decreased ATP levels and mitochondrial potential; vacuolization | Lysosomal and mitochondrial dysfunction; impaired autophagic flux | [83] |
| In vivo (mouse, 4-month exposure) | PVC nanoplastics (oral gavage) | Bradycardia, sarcomeric disarray, interstitial fibrosis | Mitochondrial cristae loss and oxidative stress | [83,84] |
| In vitro (endothelial cells) | Polystyrene nanoplastics | Increased ROS generation, apoptosis, and endothelial dysfunction | NADPH oxidase activation and p53 upregulation | [85] |
| Human studies | Polyethylene and PVC identified in carotid plaques, myocardium, and thrombi | Increased risk of myocardial infarction, stroke, and all-cause mortality | Chronic inflammation, oxidative stress, and immune activation | [86,87] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Cristina, M.; Belli, M.; Baroni, A.; Moulton, C.; Carinci, E.; Gatti, M.; Tasciotti, E.; Russo, M.A.; Russo, P.; Sansone, L. Nano- and Microplastics in the Cardiovascular System: Current Insights and Biological Implications. Nanomaterials 2026, 16, 589. https://doi.org/10.3390/nano16100589
Cristina M, Belli M, Baroni A, Moulton C, Carinci E, Gatti M, Tasciotti E, Russo MA, Russo P, Sansone L. Nano- and Microplastics in the Cardiovascular System: Current Insights and Biological Implications. Nanomaterials. 2026; 16(10):589. https://doi.org/10.3390/nano16100589
Chicago/Turabian StyleCristina, Mario, Manuel Belli, Anna Baroni, Chantalle Moulton, Emily Carinci, Marta Gatti, Ennio Tasciotti, Matteo Antonio Russo, Patrizia Russo, and Luigi Sansone. 2026. "Nano- and Microplastics in the Cardiovascular System: Current Insights and Biological Implications" Nanomaterials 16, no. 10: 589. https://doi.org/10.3390/nano16100589
APA StyleCristina, M., Belli, M., Baroni, A., Moulton, C., Carinci, E., Gatti, M., Tasciotti, E., Russo, M. A., Russo, P., & Sansone, L. (2026). Nano- and Microplastics in the Cardiovascular System: Current Insights and Biological Implications. Nanomaterials, 16(10), 589. https://doi.org/10.3390/nano16100589