Pork as a Source of Omega-3 (n-3) Fatty Acids
Abstract
:1. Introduction
2. Pork in Human Diets
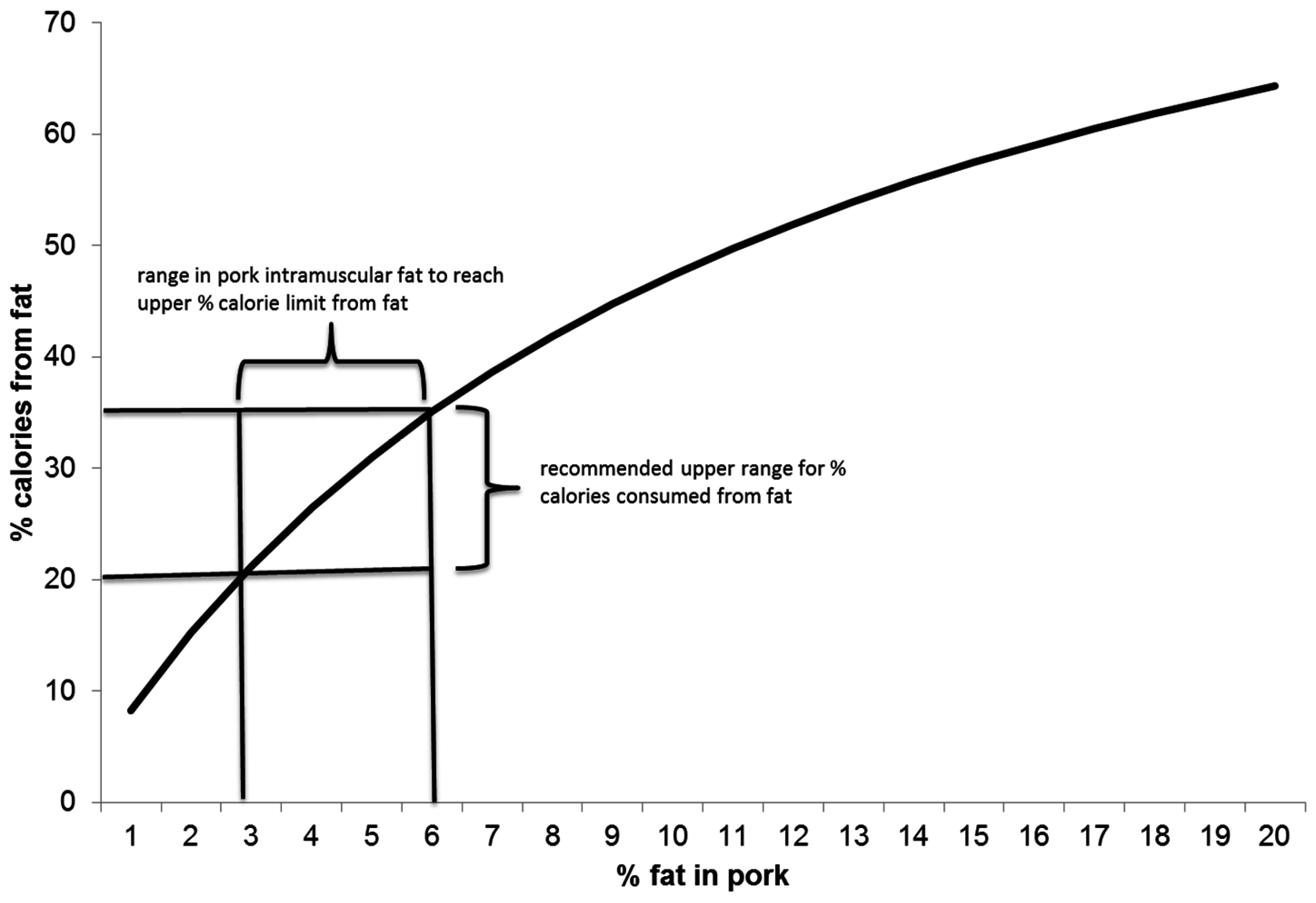
3. Pork Fatty Acid Composition
| Fatty Acid | mg/100 g Tissue | % of Fatty Acids | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| LM | AM + E | AM + E + SF | AM +E + SF + SCF | SEM | LM | AM + E | AM + E + SF | AM + E + SF + SCF | SEM | |
| C16:0 | 718 d | 1523 c | 3033 b | 5782 a | 227 | 24.3 | 24.8 | 25.6 | 25.6 | 0.5 |
| C18:0 | 378 d | 822 c | 1740 b | 3438 a | 128 | 12.8 c | 13.3 bc | 14.6 ab | 15.2 a | 0.5 |
| ΣSFA | 1147 d | 2458 c | 5001 b | 9659 a | 371 | 38.8 b | 39.9 ab | 42.2 a | 42.6 a | 0.9 |
| C16:1-9c | 101 d | 191 c | 313 b | 519 a | 25.8 | 3.39 a | 3.09 a | 2.66 b | 2.29 c | 0.12 |
| C18:1-9c | 1148 d | 2430 c | 4627 b | 9006 a | 357 | 38.6 | 39.4 | 39.2 | 39.9 | 0.8 |
| C18:1-11c | 116 d | 295 c | 420 b | 667 a | 24.0 | 3.96 b | 4.82 a | 3.59 c | 2.97 d | 0.06 |
| ΣMUFA | 1409 d | 3017 c | 5610 b | 10652 a | 415 | 47.4 | 49.0 | 47.5 | 47.2 | 0.9 |
| C18:2n-6 | 189 d | 422 c | 822 b | 1656 a | 51.4 | 7.07 | 7.00 | 7.09 | 7.39 | 0.47 |
| C18:3n-6 | 8.08 c | 13.8 c | 29.8 b | 66.5 a | 2.55 | 0.284 ab | 0.220 c | 0.258 bc | 0.295 a | 0.012 |
| C20:2n-6 | 5.54 d | 15.5 c | 37.2 b | 85.9 a | 2.28 | 0.205 d | 0.262 c | 0.322 b | 0.385 a | 0.019 |
| C20:3n-6 | 6.92 d | 10.1 c | 13.4 b | 21.5 a | 0.761 | 0.263 a | 0.166 b | 0.115 bc | 0.095 c | 0.020 |
| C20:4n-6 | 46.0 d | 59.7 c | 65.8 b | 75.4 a | 2.03 | 1.77 a | 1.00 b | 0.580 c | 0.342 c | 0.120 |
| C22:4n-6 | 1.35 d | 8.66 c | 11.8 b | 17.1 a | 0.59 | 0.048 d | 0.145 a | 0.102 b | 0.076 c | 0.005 |
| Σn-6 | 249 d | 516 c | 950 b | 1856 a | 55.0 | 9.36 | 8.57 | 8.21 | 8.29 | 0.61 |
| C18:3n-3 | 22.7 c | 41.3 c | 89.5 b | 186 a | 8.22 | 0.818 | 0.669 | 0.758 | 0.824 | 0.067 |
| C20:3n-3 | 3.01 c | 5.75 c | 15.6 b | 36.7 a | 1.22 | 0.104 c | 0.092 c | 0.131 b | 0.162 a | 0.008 |
| C20:5n-3 | 6.35 | 5.26 | 5.72 | 7.87 | 0.68 | 0.235 a | 0.0854 b | 0.0484 bc | 0.0349 c | 0.016 |
| C22:3n-3 | 5.40 a | 0.918 c | 1.79 bc | 2.52 b | 0.50 | 0.204 a | 0.0134 b | 0.0141 b | 0.0108 b | 0.011 |
| C22:5n-3 | 11.0 d | 15.4 c | 20.8 b | 30.4 a | 1.1 | 0.422 a | 0.257 b | 0.181 bc | 0.137 c | 0.029 |
| C22:6n-3 | 5.45 c | 6.42 bc | 8.71 b | 12.7 a | 0.88 | 0.209 a | 0.109 b | 0.0773 b | 0.0581 b | 0.024 |
| Σn-3 | 54.0 c | 75.1 c | 142 b | 276 a | 10.8 | 1.99 a | 1.22 b | 1.21 b | 1.22 b | 0.13 |
| ΣPUFA | 306 d | 596 c | 1104 b | 2158 a | 66 | 11.5 | 9.88 | 9.52 | 9.63 | 0.7 |
| TOTAL | 2922 d | 6140 c | 11,795 b | 22,577 a | 821 | 100 | 100 | 100 | 100 | 0 |
| n-6/n-3 | 4.77 b | 7.00 a | 6.81 a | 6.76 a | 0.26 | |||||
| PUFA/SFA | 0.301 | 0.250 | 0.228 | 0.228 | 0.025 | |||||

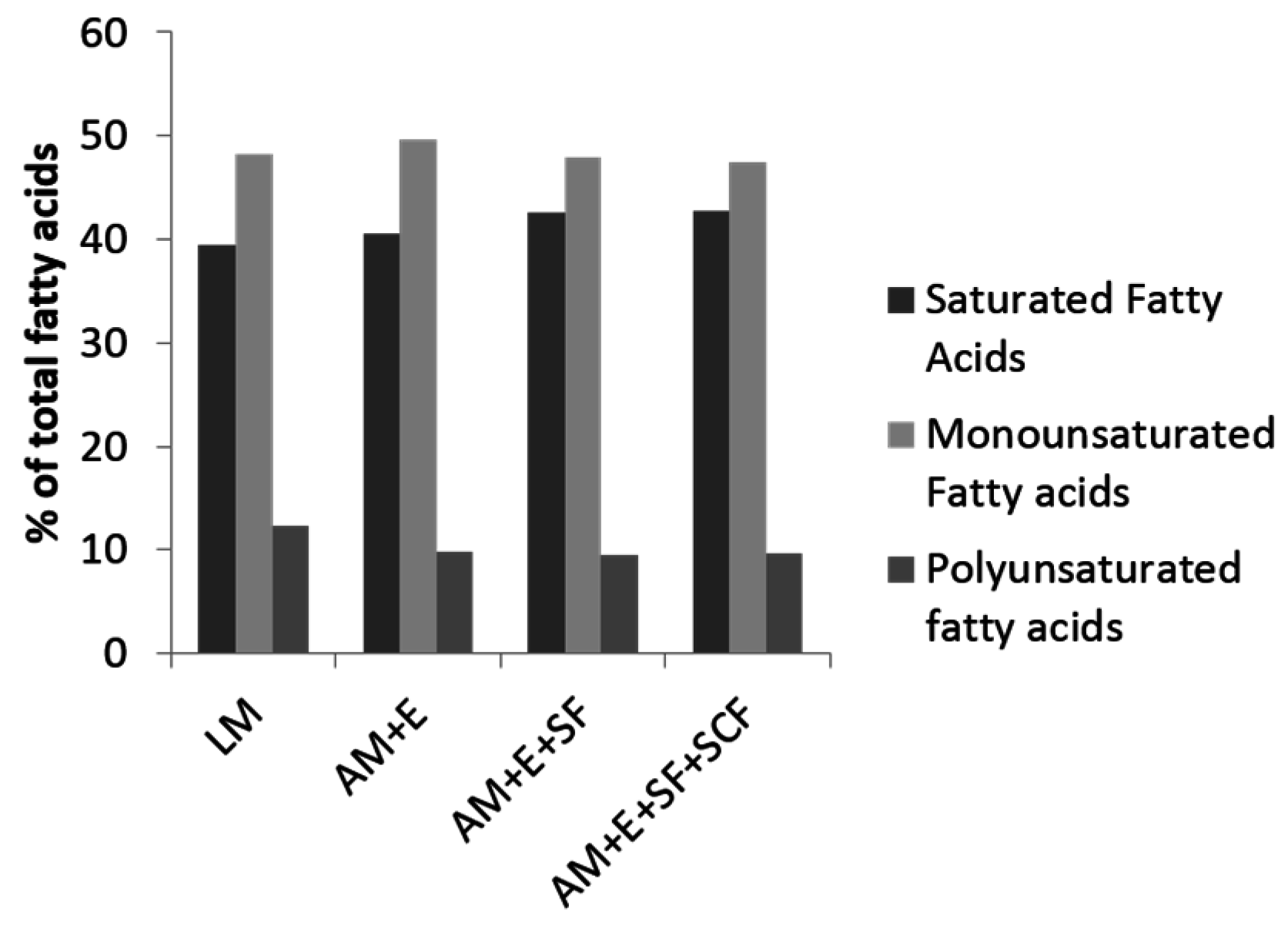
4. Enriching Pork with n-3 Fatty Acids
4.1. Initial Efforts to Improve Pork Fatty Acid Composition
4.2. Efforts to Increase n-3 Fatty Acids in Pork
4.3. An Example of n-3 Enriched Pork and Post-Production Considerations
| Fatty Acid | mg/100 g Tissue | % of Fatty Acids | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| LM | AM + E | AM + E + SF | AM + E + SF + SCF | SEM | LM | AM + E | AM + E + SF | AM + E + SF + SCF | SEM | |
| C16:0 | 571 d | 1180 c | 2227 b | 4169 a | 152 | 22.1 a | 20.1 b | 20.5 b | 19.8 b | 0.3 |
| C18:0 | 318 d | 678 c | 1326 b | 2545 a | 102 | 12.4 | 11.7 | 12.2 | 12.1 | 0.3 |
| ΣSFA | 931 d | 1953 c | 3731 b | 7040 a | 265 | 36.0 a | 33.4 b | 34.3 b | 33.5 b | 0.6 |
| C16:1-9c | 66.6 d | 113 c | 177 b | 295 a | 12.0 | 2.59 a | 1.94 b | 1.68 bc | 1.41 c | 0.10 |
| C18:1-9c | 873 d | 1871 c | 3380 b | 6727 a | 218 | 33.8 a | 32.2 b | 31.7 b | 32.2 b | 0.5 |
| C18:1-11c | 76.7 d | 196 c | 265 b | 419 a | 14.3 | 3.06 b | 3.39 a | 2.53 c | 2.01 d | 0.08 |
| ΣMUFA | 1045 d | 2259 c | 3991 b | 7724 a | 256 | 40.7 a | 38.9 ab | 37.4 bc | 36.9 c | 0.6 |
| C18:2n-6 | 225 d | 652 c | 1207 b | 2470 a | 55.0 | 9.68 b | 11.4 a | 11.5 a | 11.9 a | 0.34 |
| C18:3n-6 | 6.38 d | 12.8 c | 25.1 b | 53.0 a | 1.54 | 0.26 | 0.223 | 0.236 | 0.253 | 0.011 |
| C20:2n-6 | 6.40 d | 24.7 c | 47.1 b | 106 a | 2.75 | 0.27 b | 0.437 a | 0.454 a | 0.509 a | 0.025 |
| C20:3n-6 | 5.86 c | 7.83 bc | 10.0 b | 15.9 a | 0.79 | 0.26 a | 0.141 b | 0.099 c | 0.076 c | 0.012 |
| C20:4n-6 | 28.2 c | 34.3 b | 36.6 b | 42.3 a | 1.9 | 1.35 a | 0.626 b | 0.372 c | 0.205 c | 0.06 |
| C22:4n-6 | 1.23 c | 3.87 b | 5.27 b | 7.95 a | 0.56 | 0.05 ab | 0.067 a | 0.049 ab | 0.037 b | 0.007 |
| Σn-6 | 266 d | 723 c | 1306 b | 2643 a | 59 | 11.6 | 12.7 | 12.5 | 12.7 | 0.4 |
| C18:3n-3 | 145 d | 614 c | 1290 b | 2800 a | 71 | 5.76 d | 10.6 c | 12.1 b | 13.4 a | 0.30 |
| C20:3n-3 | 21.5 d | 82.2 c | 166 b | 381 a | 11.6 | 0.85 c | 1.42 b | 1.55 b | 1.82 a | 0.053 |
| C20:5n-3 | 23.3 d | 31.9 c | 38.8 b | 55.3 a | 1.7 | 1.08 a | 0.573 b | 0.385 c | 0.268 c | 0.05 |
| C22:3n-3 | 2.48 b | 2.27 b | 3.06 b | 4.75 a | 0.40 | 0.10 a | 0.038 b | 0.029 b | 0.023 b | 0.005 |
| C22:5n-3 | 21.1 d | 39.5 c | 59.7 b | 102 a | 3.0 | 0.95 a | 0.705 b | 0.575 c | 0.491 c | 0.038 |
| C22:6n-3 | 3.85 d | 8.13 c | 10.6 b | 16.2 a | 0.77 | 0.18 a | 0.146 b | 0.105 c | 0.079 c | 0.011 |
| Σn-3 | 217 d | 778 c | 1569 b | 3360 a | 85 | 8.94 d | 13.5 c | 14.7 b | 16.1 a | 0.39 |
| ΣPUFA | 486 d | 1506 c | 2883 b | 6018 a | 143 | 20.6 c | 26.3 b | 27.4 ab | 28.9 a | 0.7 |
| TOTAL | 2519 d | 5788 c | 10,683 b | 20,881 a | 627 | 100 | 100 | 100 | 100 | 0 |
| n-6/n-3 | 1.28 a | 0.935 b | 0.850 c | 0.788 c | ||||||
| PUFA/SFA | 0.581 b | 0.796 a | 0.807 a | 0.869 a | ||||||
5. Practical Barriers Limiting n-3 Pork Development and Entry into the Food Supply
5.1. The Call for n-3 Enriched Meat Unfulfilled to Date
5.2. Why Production of n-3 Enriched Pork Has Not Been Adopted
5.2.1. Visibility
5.2.2. Challenges along the Value Chain
6. Strategies to Encourage Production and Market Availability of n-3 Fatty Acid Enriched Pork
7. Conclusions
Author Contributions
Conflicts of Interest
References
- Simopoulos, A.P. Importance of the omega-6/omega-3 balance in health and disease: Evolutionary aspects of diet. World Rev. Nutr. Diet. 2011, 102, 10–21. [Google Scholar] [PubMed]
- FAO Sources of Meat. Available online: http://www.fao.org/ag/againfo/themes/en/meat/backgr_sources.html (accessed on 2 September 2015).
- Ollis, T.E.; Meyer, B.J.; Howe, P.R. Australian food sources and intakes of omega–6 and omega–3 polyunsaturated fatty acids. Ann. Nutr. Metab. 1999, 43, 346–355. [Google Scholar] [CrossRef] [PubMed]
- Teicholz, N. The Big Fat Surprise: Why Butter, Meat and Cheese belong in a Healthy Diet; Simon and Schuster: New York, NY, USA, 2014. [Google Scholar]
- Fortin, A.; Robertson, W.; Tong, A. The eating quality of canadian pork and its relationship with intramuscular fat. Meat Sci. 2005, 69, 297–305. [Google Scholar] [CrossRef] [PubMed]
- U.S. Department of Health and Human Services; U.S. Department of Agriculture. Dietary Guidelines for Americans; US Government Printing Office: Washington, DC, USA, 2010.
- Siri-Tarino, P.W.; Sun, Q.; Hu, F.B.; Krauss, R.M. Meta-analysis of prospective cohort studies evaluating the association of saturated fat with cardiovascular disease. Am. J. Clin. Nutr. 2010, 98, 535–546. [Google Scholar] [CrossRef] [PubMed]
- Chowdhury, R.; Warnakula, S.; Kunutsor, S.; Crowe, F.; Ward, H.A.; Johnson, L.; Franco, O.H.; Butterworth, A.S.; Forouhi, N.G.; Thompson, S.G.; et al. Association of dietary, circulating, and supplement fatty acids with coronary risk: A systematic review and meta-analysis. Ann. Intern. Med. 2014, 160, 398–406. [Google Scholar] [CrossRef] [PubMed]
- Barendse, W. Should animal fats be back on the table? A critical review of the human health effects of animal fat. Anim. Prod. Sci. 2014, 54, 831–855. [Google Scholar]
- Binnie, M.A.; Barlow, K.; Johnson, V.; Harrison, C. Red meats: Time for a paradigm shift in dietary advice. Meat Sci. 2014, 98, 445–451. [Google Scholar] [CrossRef] [PubMed]
- Heinz, G.; Hautzinger, P. Meat Processing Technology for Small to Medium Scale Producers. Available online: http://www.fao.org/docrep/010/ai407e/ai407e00.htm (accessed on 2 September 2015).
- Enser, M.; Hallett, K.; Hewitt, B.; Fursey, G.A.J.; Wood, J.D. Fatty acid content and composition of english beef, lamb and pork at retail. Meat Sci. 1996, 42, 443–456. [Google Scholar] [CrossRef]
- Cardiovascular Review Group, Great Britain—Department of Health. Nutritional Aspects of Cardiovascular Disease; HMSO: Richmond, UK, 1994.
- Juárez, M.; Dugan, M.E.R.; Aldai, N.; Aalhus, J.L.; Patience, J.F.; Zijlstra, R.T.; Beaulieu, A.D. Increasing omega-3 levels through dietary co-extruded flaxseed supplementation negatively affects pork palatability. Food Chem. 2011, 126, 1716–1723. [Google Scholar] [CrossRef] [PubMed]
- Romans, J.R.; Johnson, R.C.; Wulf, D.M.; Libal, G.W.; Costello, W.J. Effects of ground flaxseed in swine diets on pig performance and on physical and sensory characteristics and omega-3 fatty acid content of pork: I. Dietary level of flaxseed. J. Anim. Sci. 1995, 73, 1982–1986. [Google Scholar]
- Wood, J.D.; Enser, M.; Fisher, A.V.; Nute, G.R.; Sheard, P.R.; Richardson, R.I.; Hughes, S.I.; Whittington, F.M. Fat deposition, fatty acid composition and meat quality: A review. Meat Sci. 2008, 78, 343–358. [Google Scholar] [CrossRef] [PubMed]
- Savell, J.; Cross, H. The role of fat in the palatability of beef, pork, and lamb. In Designing Foods: Animal Product Options in the Marketplace; National Academy Press: Washington, DC, USA, 1988; pp. 345–355. [Google Scholar]
- Raes, K.; de Smet, S.; Demeyer, D. Effect of dietary fatty acids on incorporation of long chain polyunsaturated fatty acids and conjugated linoleic acid in lamb, beef and pork meat: A review. Anim. Feed Sci. Technol. 2004, 113, 199–221. [Google Scholar] [CrossRef]
- Woods, V.B.; Fearon, A.M. Dietary sources of unsaturated fatty acids for animals and their transfer into meat, milk and eggs: A review. Livest. Sci. 2009, 126. [Google Scholar] [CrossRef]
- Doreau, M.; Chilliard, Y. Digestion and metabolism of dietary fat in farm animals. Br. J. Nutr. 1997, 78, S15–S35. [Google Scholar] [CrossRef] [PubMed]
- Shorland, F.B. Effect of the dietary fat on the composition of depot fats of animals. Nature 1950, 165. [Google Scholar] [CrossRef]
- Turner, T.D.; Mapiye, C.; Aalhus, J.L.; Beaulieu, A.D.; Patience, J.F.; Zijlstra, R.T.; Dugan, M.E.R. Flaxseed fed pork: N-3 fatty acid enrichment and contribution to dietary recommendations. Meat Sci. 2014, 96, 541–547. [Google Scholar] [CrossRef] [PubMed]
- Narayan, B.; Miyashita, K.; Hosakawa, M. Physiological effects of eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA)—A review. Food Rev. Int. 2006, 22, 291–307. [Google Scholar] [CrossRef]
- Swanson, D.; Block, R.; Mousa, S.A. Omega-3 fatty acids EPA and DHA: Health benefits throughout life. Adv. Nutr. 2012, 3. [Google Scholar] [CrossRef] [PubMed]
- Howe, P.; Meyer, B.; Record, S.; Baghurst, K. Dietary intake of long-chain omega-3 polyunsaturated fatty acids: Contribution of meat sources. Nutrition 2006, 22, 47–53. [Google Scholar] [CrossRef] [PubMed]
- Miller, E.; Kaur, G.; Larsen, A.; Loh, S.P.; Linderborg, K.; Weisinger, H.S.; Turchini, G.M.; Cameron-Smith, D.; Sinclair, A.J. A short-term n-3 DPA supplementation study in humans. Eur. J. Nutr. 2013, 52, 895–904. [Google Scholar] [CrossRef] [PubMed]
- Vahmani, P.; Mapiye, C.; Prieto, N.; Rolland, D.C.; McAllister, T.A.; Aalhus, J.L.; Dugan, M.E. The scope for manipulating the polyunsaturated fatty acid content of beef: A review. J. Anim. Sci. Biotechnol. 2015, 6. [Google Scholar] [CrossRef] [PubMed]
- Canadian Food Inspection Agency (CFIA): Omega-3 and Omega-6 Polyunsaturated Fatty Acid Claims. Available online: http://www.inspection.gc.ca/food/labelling/food-labelling-for-industry/nutrient-content/specific-claim-requirements/eng/1389907770176/1389907817577?chap=7 (accessed on 9 September 2015).
- Kris-Etherton, P.M. Monounsaturated fatty acids and risk of cardiovascular disease. Circulation 1999, 100, 1253–1258. [Google Scholar] [CrossRef] [PubMed]
- Yu, S.; Derr, J.; Etherton, T.D.; Kris-Etherton, P. Plasma cholesterol-predictive equations demonstrate that stearic acid is neutral and monounsaturated fatty acids are hypocholesterolemic. Am. J. Clin. Nutr. 1995, 61, 1129–1139. [Google Scholar] [PubMed]
- Koch, D.E.; Pearson, A.M.; Magee, W.T.; Hoefer, J.A.; Schweigert, B.S. Effect of diet on the fatty acid composition of pork fat. J. Anim. Sci. 1968, 27, 360–365. [Google Scholar]
- Stewart, J.W.; Kaplan, M.L.; Beitz, D.C. Pork with a high content of polyunsaturated fatty acids lowers LDL cholesterol in women. Am. J. Clin. Nutr. 2001, 74, 179–187. [Google Scholar] [PubMed]
- Anderson, D.B.; Kauffman, R.G.; Benevenga, N.J. Estimate of fatty acid turnover in porcine adipose tissue. Lipids 1972, 7, 488–489. [Google Scholar] [CrossRef] [PubMed]
- Cunnane, S.C.; Stitt, P.A.; Sujata, G.; Armstrong, J.K. Raised omega-3 fatty acid levels in pigs fed flax. Can. J. Anim. Sci. 1990, 70, 251–254. [Google Scholar] [CrossRef]
- Kouba, M.; Enser, M.; Whittington, F.M.; Nute, G.R.; Wood, J.D. Effect of a high-linolenic acid diet on lipogenic enzyme activities, fatty acid composition, and meat quality in the growing pig. J. Anim. Sci. 2003, 81, 1967–1979. [Google Scholar] [PubMed]
- Corino, C.; Rossi, R.; Cannata, S.; Ratti, S. Effect of dietary linseed on the nutritional value and quality of pork and pork products: Systematic review and meta-analysis. Meat Sci. 2014, 98, 679–688. [Google Scholar] [CrossRef] [PubMed]
- Cherian, G.; Sim, J.S. Dietary alpha-linolenic acid alters the fatty acid composition of lipid classes in swine tissues. J. Agric. Food Chem. 1995, 43, 2911–2916. [Google Scholar] [CrossRef]
- Riley, P.A.; Enser, M.; Nute, G.R.; Wood, J.D. Effects of dietary linseed on nutritional value and other quality aspects of pig muscle and adipose tissue. Anim. Sci. 2000, 71, 483–500. [Google Scholar]
- Ahn, D.U.; Lutz, S.; Sim, J.S. Effects of dietary alpha-linolenic acid on the fatty acid composition, storage stability and sensory characteristics of pork loin. Meat Sci. 1996, 43, 291–299. [Google Scholar] [CrossRef]
- Romans, J.R.; Wulf, D.M.; Johnson, R.C.; Libal, G.W.; Costello, W.J. Effects of ground flaxseed in swine diets on pig performance and on physical and sensory characteristics and omega-3 fatty acid content of pork: II. Duration of 15% dietary flaxseed. J. Anim. Sci. 1995, 73, 1987–1999. [Google Scholar] [PubMed]
- Juárez, M.; Dugan, M.; Aldai, N.; Aalhus, J.; Patience, J.; Zijlstra, R.; Beaulieu, A. Feeding co-extruded flaxseed to pigs: Effects of duration and feeding level on growth performance and backfat fatty acid composition of grower–finisher pigs. Meat Sci. 2010, 84, 578–584. [Google Scholar] [CrossRef] [PubMed]
- Fontanillas, R.; Barroeta, A.; Baucells, M.D.; Guardiola, F. Backfat fatty acid evolution in swine fed diets high in either cis-monounsaturated, trans, or (n-3) fats. J. Anim. Sci. 1998, 76, 1045–1055. [Google Scholar] [PubMed]
- Huang, F.R.; Zhan, Z.P.; Luo, J.; Liu, Z.X.; Peng, J. Duration of dietary linseed feeding affects the intramuscular fat, muscle mass and fatty acid composition in pig muscle. Livest. Sci. 2008, 118, 132–139. [Google Scholar] [CrossRef]
- Martínez-Ramírez, H.R.; Kramer, J.K.G.; de Lange, C.F.M. Retention of n-3 polyunsaturated fatty acids in trimmed loin and belly is independent of timing of feeding ground flaxseed to growing-finishing female pigs. J. Anim. Sci. 2014, 92, 238–249. [Google Scholar] [CrossRef] [PubMed]
- Enser, M.; Richardson, R.I.; Wood, J.D.; Gill, B.P.; Sheard, P.R. Feeding linseed to increase the n-3 pufa of pork: Fatty acid composition of muscle, adipose tissue, liver and sausages. Meat Sci. 2000, 55, 201–212. [Google Scholar] [CrossRef]
- Nguyen, L.Q.; Nuijens, M.C.G.A.; Everts, H.; Salden, N.; Beynen, A.C. Mathematical relationships between the intake of n-6 and n-3 polyunsaturated fatty acids and their contents in adipose tissue of growing pigs. Meat Sci. 2003, 65, 1399–1406. [Google Scholar] [CrossRef]
- Htoo, J.K.; Meng, X.; Patience, J.F.; Dugan, M.E.R.; Zijlstra, R.T. Effects of coextrusion of flaxseed and field pea on the digestibility of energy, ether extract, fatty acids, protein, and amino acids in grower-finisher pigs. J. Anim. Sci. 2008, 86, 2942–2951. [Google Scholar] [CrossRef] [PubMed]
- U.S. Department of Health and Human Services (HHS). Food labeling: Nutrient content claims; alpha-linolenic acid, eicosapentaenoic acid, and docosahexaenoic acid omega-3 fatty acids. Fed. Regist. 2014, 79, 23262–23273. [Google Scholar]
- European Food Safety Authority (EFSA). Scientific opinion: Labelling reference intake values for n-3 and n-6 polyunsaturated fatty acids. EPSA J. 2009, 1176, 1–11. [Google Scholar]
- Kim, E.J.; Kim, M.-K.; Jin, X.-J.; Oh, J.-H.; Kim, J.E.; Chung, J.H. Skin aging and photoaging alter fatty acids composition, including 11,14,17-eicosatrienoic acid, in the epidermis of human skin. J. Korean Med. Sci. 2010, 25, 980–983. [Google Scholar] [CrossRef] [PubMed]
- Schenck, P.A.; Rakoff, H.; Emken, E.A. δ8 desaturation in vivo of deuterated eicosatrienoic acid by mouse liver. Lipids 1996, 31, 593–600. [Google Scholar] [CrossRef] [PubMed]
- Coates, A.M.; Sioutis, S.; Buckley, J.D.; Howe, P.R. Regular consumption of n-3 fatty acid-enriched pork modifies cardiovascular risk factors. Br. J. Nutr. 2009, 101, 592–597. [Google Scholar] [CrossRef] [PubMed]
- Vossen, E.; Raes, K.; Maertens, L.; Vandenberge, V.; Haak, L.; Chiers, K.; Ducatelle, R.; de Smet, S. Diets containing n-3 fatty acids-enriched pork: Effect on blood lipids, oxidative status and atherosclerosis in rabbits. J. Food Biochem. 2012, 36, 359–368. [Google Scholar] [CrossRef]
- Simopoulos, A.P. New products from the agri-food industry: The return of n-3 fatty acids into the food supply. Lipids 1999, 34, S297–S301. [Google Scholar] [CrossRef] [PubMed]
- Lewis, N.M.; Seburg, S.; Flanagan, N.L. Enriched eggs as a source of n-3 polyunsaturated fatty acids for humans. Poult. Sci. 2000, 79, 971–974. [Google Scholar] [CrossRef] [PubMed]
- Givens, D.; Gibbs, R. Very long chain n-3 polyunsaturated fatty acids in the food chain in the uk and the potential of animal-derived foods to increase intake. Nutr. Bull. 2006, 31, 104–110. [Google Scholar] [CrossRef]
- Bryhni, E.A.; Kjos, N.P.; Ofstad, R.; Hunt, M. Polyunsaturated fat and fish oil in diets for growing-finishing pigs: Effects on fatty acid composition and meat, fat, and sausage quality. Meat Sci. 2002, 62. [Google Scholar] [CrossRef]
- Damez, J.-L.; Clerjon, S. Quantifying and predicting meat and meat products quality attributes using electromagnetic waves: An overview. Meat Sci. 2013, 95, 879–896. [Google Scholar] [CrossRef] [PubMed]
- Prieto, N.; Dugan, M.E.R.; López-Campos, O.; McAllister, T.A.; Aalhus, J.L.; Uttaro, B. Near infrared reflectance spectroscopy predicts the content of polyunsaturated fatty acids and biohydrogenation products in the subcutaneous fat of beef cows fed flaxseed. Meat Sci. 2012, 90, 43–51. [Google Scholar] [CrossRef] [PubMed]
- Prieto, N.; Uttaro, B.; Mapiye, C.; Turner, T.; Dugan, M.; Zamora, V.; Young, M.; Beltranena, E. Predicting fat quality from pigs fed reduced-oil corn dried distillers grains with solubles by near infrared reflectance spectroscopy: Fatty acid composition and iodine value. Meat Sci. 2014, 98, 585–590. [Google Scholar] [CrossRef] [PubMed]
- Berhe, D.T.; Eskildsen, C.E.; Lametsch, R.; Hviid, M.S.; van den Berg, F.; Engelsen, S.B. Prediction of total fatty acid parameters and individual fatty acids in pork backfat using raman spectroscopy and chemometrics: Understanding the cage of covariance between highly correlated fat parameters. Meat Sci. 2016, 111, 18–26. [Google Scholar] [CrossRef] [PubMed]
- Cecchini, M.; Sassi, F.; Lauer, J.A.; Lee, Y.Y.; Guajardo-Barron, V.; Chisholm, D. Tackling of unhealthy diets, physical inactivity, and obesity: Health effects and cost-effectiveness. Lancet 2010, 376, 1775–1784. [Google Scholar] [CrossRef]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dugan, M.E.R.; Vahmani, P.; Turner, T.D.; Mapiye, C.; Juárez, M.; Prieto, N.; Beaulieu, A.D.; Zijlstra, R.T.; Patience, J.F.; Aalhus, J.L. Pork as a Source of Omega-3 (n-3) Fatty Acids. J. Clin. Med. 2015, 4, 1999-2011. https://doi.org/10.3390/jcm4121956
Dugan MER, Vahmani P, Turner TD, Mapiye C, Juárez M, Prieto N, Beaulieu AD, Zijlstra RT, Patience JF, Aalhus JL. Pork as a Source of Omega-3 (n-3) Fatty Acids. Journal of Clinical Medicine. 2015; 4(12):1999-2011. https://doi.org/10.3390/jcm4121956
Chicago/Turabian StyleDugan, Michael E.R., Payam Vahmani, Tyler D. Turner, Cletos Mapiye, Manuel Juárez, Nuria Prieto, Angela D. Beaulieu, Ruurd T. Zijlstra, John F. Patience, and Jennifer L. Aalhus. 2015. "Pork as a Source of Omega-3 (n-3) Fatty Acids" Journal of Clinical Medicine 4, no. 12: 1999-2011. https://doi.org/10.3390/jcm4121956
APA StyleDugan, M. E. R., Vahmani, P., Turner, T. D., Mapiye, C., Juárez, M., Prieto, N., Beaulieu, A. D., Zijlstra, R. T., Patience, J. F., & Aalhus, J. L. (2015). Pork as a Source of Omega-3 (n-3) Fatty Acids. Journal of Clinical Medicine, 4(12), 1999-2011. https://doi.org/10.3390/jcm4121956





