Application of Wood and Vegetable Waste-Based Biochars in Sustainable Agriculture: Evaluation on Nitrate Leaching, Pesticide Fate, Soil Properties, and Brassica oleracea Growth
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Setup
2.2. Nitrate and Ammonium Quantification in Environmental Matrices
2.3. Pesticides Extraction and Quantification in Environmental Matrices and Cauliflower Curd
2.4. Soil Physicochemical Analyses
2.5. SEM Analyses
2.6. ATR-FTIR Spectral Collection and Data Analysis
2.7. Morphologic Growth Parameters
2.8. Statistical Analysis
3. Results and Discussions
3.1. The Influence of Biochar on Soil’s Physical-Chemical Properties
3.2. Biochar’s Influence on the Protection of Agricultural Pollutants
3.2.1. Effect of Biochar on Nitrogen Compounds Dynamics
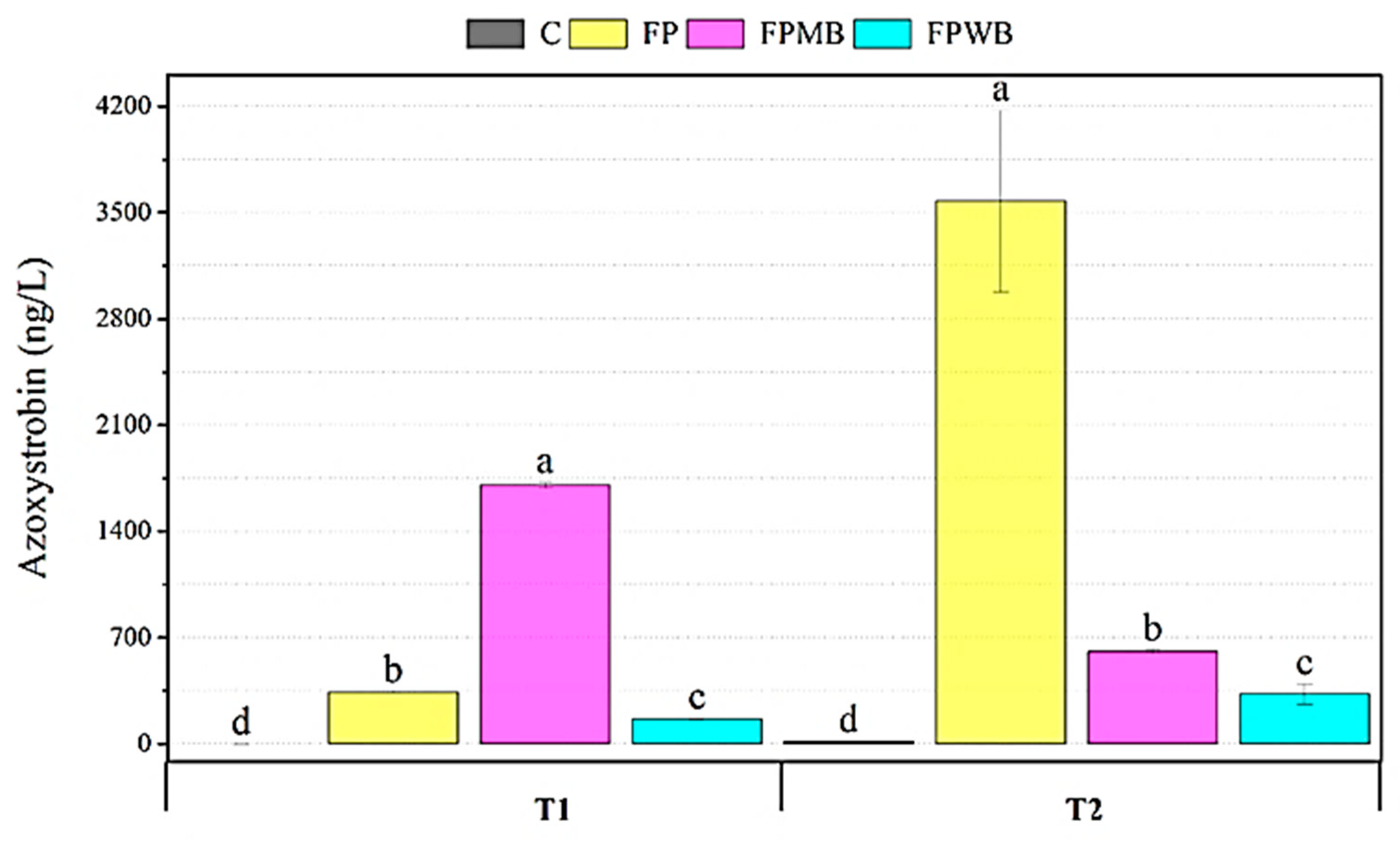
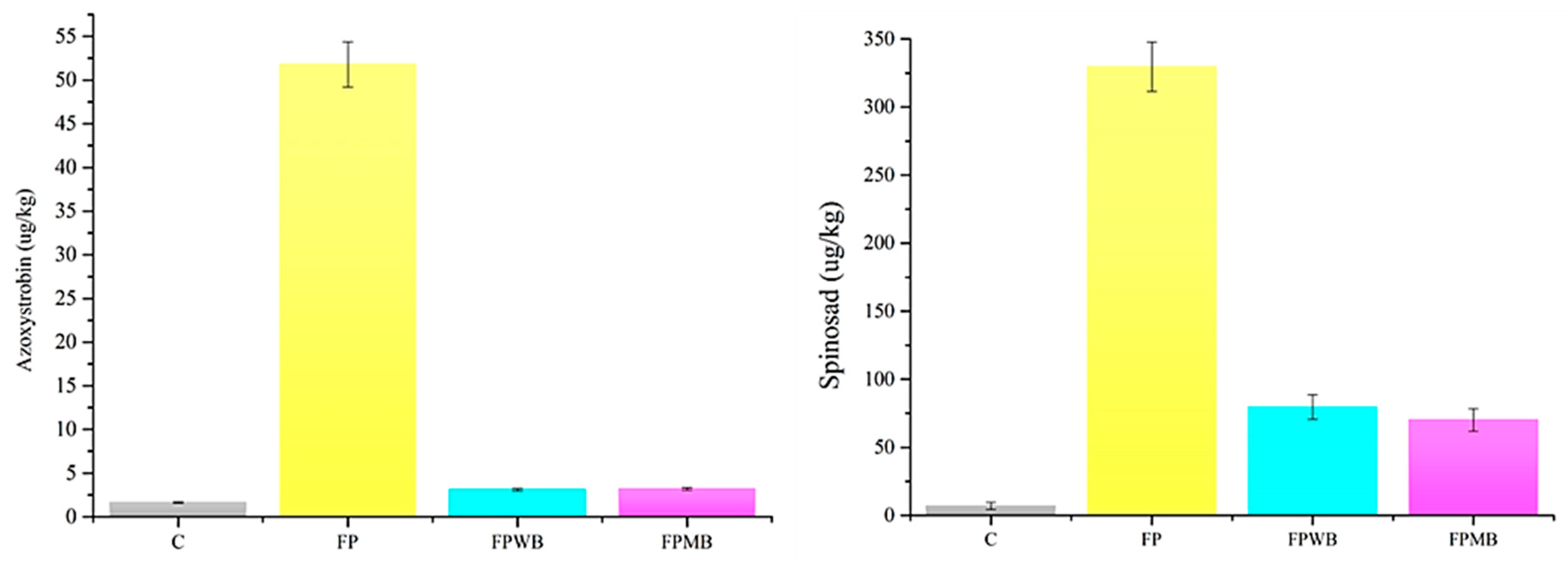
3.2.2. The Impact of Biochar on Pesticide Protection in the Environment and Cauliflower
3.3. Biochar Effect on Brassica oleracea L. var botrytis Growth
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Galloway, J.N.; Townsend, A.R.; Erisman, J.W.; Bekunda, M.; Cai, Z.; Freney, J.R.; Martinelli, L.A.; Seitzinger, S.P.; Sutton, M.A. Transformation of the Nitrogen Cycle: Recent Trends, Questions, and Potential Solutions. Science 2008, 320, 889–892. [Google Scholar] [CrossRef] [PubMed]
- Losacco, D.; Ancona, V.; De Paola, D.; Tumolo, M.; Massarelli, C.; Gatto, A.; Uricchio, V.F. Development of Ecological Strategies for the Recovery of the Main Nitrogen Agricultural Pollutants: A Review on Environmental Sustainability in Agroecosystems. Sustainability 2021, 13, 7163. [Google Scholar] [CrossRef]
- Corsini, E.; Sokooti, M.; Galli, C.L.; Moretto, A.; Colosio, C. Pesticide Induced Immunotoxicity in Humans: A Comprehensive Review of the Existing Evidence. Toxicology 2013, 307, 123–135. [Google Scholar] [CrossRef] [PubMed]
- Reeves, W.R.; McGuire, M.K.; Stokes, M.; Vicini, J.L. Assessing the Safety of Pesticides in Food: How Current Regulations Protect Human Health. Adv. Nutr. 2019, 10, 80–88. [Google Scholar] [CrossRef] [PubMed]
- Arora, S.; Arora, S.; Sahni, D.; Sehgal, M.; Srivastava, D.S.; Singh, A. Pesticides Use and Its Effect on Soil Bacteria and Fungal Populations, Microbial Biomass Carbon and Enzymatic Activity. Curr. Sci. 2019, 116, 643–649. [Google Scholar] [CrossRef]
- PAN International List of Highly Hazardous Pesticides (PAN List of HHP)—June 2013. Available online: https://pan-international.org/wp-content/uploads/PAN_HHP_List.pdf?_gl=1*q6qghn*_ga*MTI1MDQ2MDI0MC4xNjcxNjk2MDg1*_ga_PVQKRCXXT2*MTY3MTY5NjA4NS4xLjAuMTY3MTY5NjA4NS4wLjAuMA (accessed on 22 December 2022).
- European Union EUR-Lex-32009R1107-EN-EUR-Lex. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/?uri=CELEX%3A32009R1107 (accessed on 22 December 2022).
- Van Valkenburg, S. Pan Europe. J. Geog. 1930, 29, 133–140. [Google Scholar] [CrossRef]
- Niazi, N.K.; Bibi, I.; Shahid, M.; Ok, Y.S.; Shaheen, S.M.; Rinklebe, J.; Wang, H.; Murtaza, B.; Islam, E.; Farrakh Nawaz, M.; et al. Arsenic Removal by Japanese Oak Wood Biochar in Aqueous Solutions and Well Water: Investigating Arsenic Fate Using Integrated Spectroscopic and Microscopic Techniques. Sci. Total Environ. 2018, 621, 1642–1651. [Google Scholar] [CrossRef]
- Micoli, L.; Di Rauso Simeone, G.; Turco, M.; Toscano, G.; Rao, M.A. Anaerobic Digestion of Olive Mill Wastewater in the Presence of Biochar. Energies 2023, 16, 3259. [Google Scholar] [CrossRef]
- Khorram, M.S.; Wang, Y.; Jin, X.; Fang, H.; Yu, Y. Reduced Mobility of Fomesafen through Enhanced Adsorption in Biochar-Amended Soil. Environ. Toxicol. Chem. 2015, 34, 1258–1266. [Google Scholar] [CrossRef]
- Chen, Z.; Pei, J.; Wei, Z.; Ruan, X.; Hua, Y.; Xu, W.; Zhang, C.; Liu, T.; Guo, Y. A Novel Maize Biochar-Based Compound Fertilizer for Immobilizing Cadmium and Improving Soil Quality and Maize Growth. Environ. Pollut. 2021, 277, 116455. [Google Scholar] [CrossRef]
- Zhang, L.; He, Y.; Lin, D.; Yao, Y.; Song, N.; Wang, F. Co-Application of Biochar and Nitrogen Fertilizer Promotes Rice Performance, Decreases Cadmium Availability, and Shapes Rhizosphere Bacterial Community in Paddy Soil. Environ. Pollut. 2022, 308, 119624. [Google Scholar] [CrossRef] [PubMed]
- Zhu, T.; Zhang, J.; Cai, Z.; Müller, C. The N Transformation Mechanisms for Rapid Nitrate Accumulation in Soils under Intensive Vegetable Cultivation. J. Soils Sediments 2011, 11, 1178–1189. [Google Scholar] [CrossRef]
- Dai, Y.; Wang, W.; Lu, L.; Yan, L.; Yu, D. Utilization of Biochar for the Removal of Nitrogen and Phosphorus. J. Clean. Prod. 2020, 257, 120573. [Google Scholar] [CrossRef]
- Wang, B.; Lehmann, J.; Hanley, K.; Hestrin, R.; Enders, A. Ammonium Retention by Oxidized Biochars Produced at Different Pyrolysis Temperatures and Residence Times. RSC Adv. 2016, 6, 41907–41913. [Google Scholar] [CrossRef]
- Zhou, X.; Wang, R.; Liu, H.; Wu, S.; Wu, H. Nitrogen Removal Responses to Biochar Addition in Intermittent-Aerated Subsurface Flow Constructed Wetland Microcosms: Enhancing Role and Mechanism. Ecol. Eng. 2019, 128, 57–65. [Google Scholar] [CrossRef]
- Alsewaileh, A.S.; Usman, A.R.; Al-Wabel, M.I. Effects of Pyrolysis Temperature on Nitrate-Nitrogen (NO 3− -N) and Bromate (BrO 3− ) Adsorption onto Date Palm Biochar. J. Environ. Manage. 2019, 237, 289–296. [Google Scholar] [CrossRef] [PubMed]
- Oleszczuk, P.; Rycaj, M.; Lehmann, J.; Cornelissen, G. Influence of Activated Carbon and Biochar on Phytotoxicity of Air-Dried Sewage Sludges to Lepidium Sativum. Ecotoxicol. Environ. Saf. 2012, 80, 321–326. [Google Scholar] [CrossRef]
- Deng, H.; Feng, D.; He, J.X.; Li, F.Z.; Yu, H.M.; Ge, C.J. Influence of Biochar Amendments to Soil on the Mobility of Atrazine Using Sorption-Desorption and Soil Thin-Layer Chromatography. Ecol. Eng. 2017, 99, 381–390. [Google Scholar] [CrossRef]
- Mandal, A.; Singh, N.; Purakayastha, T.J. Characterization of Pesticide Sorption Behaviour of Slow Pyrolysis Biochars as Low Cost Adsorbent for Atrazine and Imidacloprid Removal. Sci. Total Environ. 2017, 577, 376–385. [Google Scholar] [CrossRef]
- Herath, I.; Kumarathilaka, P.; Al-Wabel, M.I.; Abduljabbar, A.; Ahmad, M.; Usman, A.R.A.; Vithanage, M. Mechanistic Modeling of Glyphosate Interaction with Rice Husk Derived Engineered Biochar. Microporous Mesoporous Mater. 2016, 225, 280–288. [Google Scholar] [CrossRef]
- Zhang, P.; Sun, H.; Min, L.; Ren, C. Biochars Change the Sorption and Degradation of Thiacloprid in Soil: Insights into Chemical and Biological Mechanisms. Environ. Pollut. 2018, 236, 158–167. [Google Scholar] [CrossRef] [PubMed]
- Mukherjee, S.; Weihermüller, L.; Tappe, W.; Hofmann, D.; Köppchen, S.; Laabs, V.; Vereecken, H.; Burauel, P. Sorption-Desorption Behaviour of Bentazone, Boscalid and Pyrimethanil in Biochar and Digestate Based Soil Mixtures for Biopurification Systems. Sci. Total Environ. 2016, 559, 63–73. [Google Scholar] [CrossRef]
- Yavari, S.; Malakahmad, A.; Sapari, N.B.; Yavari, S. Synthesis Optimization of Oil Palm Empty Fruit Bunch and Rice Husk Biochars for Removal of Imazapic and Imazapyr Herbicides. J. Environ. Manage. 2017, 193, 201–210. [Google Scholar] [CrossRef] [PubMed]
- Manna, S.; Singh, N.; Purakayastha, T.J.; Berns, A.E. Effect of Deashing on Physico-Chemical Properties of Wheat and Rice Straw Biochars and Potential Sorption of Pyrazosulfuron-Ethyl. Arab. J. Chem. 2020, 13, 1247–1258. [Google Scholar] [CrossRef]
- Alahabadi, A.; Moussavi, G. Preparation, Characterization and Atrazine Adsorption Potential of Mesoporous Carbonate-Induced Activated Biochar (CAB) from Calligonum Comosum Biomass: Parametric Experiments and Kinetics, Equilibrium and Thermodynamic Modeling. J. Mol. Liq. 2017, 242, 40–52. [Google Scholar] [CrossRef]
- Petter, F.A.; Ferreira, T.S.; Sinhorin, A.P.; de Lima, L.B.; de Morais, L.A.; Pacheco, L.P. Sorption and Desorption of Diuron in Oxisol under Biochar Application. Bragantia 2016, 75, 487–496. [Google Scholar] [CrossRef]
- Mayakaduwa, S.S.; Vithanage, M.; Karunarathna, A.; Mohan, D.; Ok, Y.S. Interface Interactions between Insecticide Carbofuran and Tea Waste Biochars Produced at Different Pyrolysis Temperatures. Chem. Speciat. Bioavailab. 2016, 28, 110–118. [Google Scholar] [CrossRef]
- Wu, C.; Liu, X.; Wu, X.; Dong, F.; Xu, J.; Zheng, Y. Sorption, Degradation and Bioavailability of Oxyfluorfen in Biochar-Amended Soils. Sci. Total Environ. 2019, 658, 87–94. [Google Scholar] [CrossRef]
- Kookana, R.S.; Sarmah, A.K.; Van Zwieten, L.; Krull, E.; Singh, B. Biochar Application to Soil. Agronomic and Environmental Benefits and Unintended Consequences. Adv. Agron. 2011, 112, 103–143. [Google Scholar] [CrossRef]
- García-Jaramillo, M.; Cox, L.; Cornejo, J.; Hermosín, M.C. Effect of Soil Organic Amendments on the Behavior of Bentazone and Tricyclazole. Sci. Total Environ. 2014, 466–467, 906–913. [Google Scholar] [CrossRef]
- Khalid, S.; Shahid, M.; Murtaza, B.; Bibi, I.; Natasha; Asif Naeem, M.; Niazi, N.K. A Critical Review of Different Factors Governing the Fate of Pesticides in Soil under Biochar Application. Sci. Total Environ. 2020, 711, 134645. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.B.; Ying, G.G.; Peng, P.A.; Wang, L.; Zhao, J.L.; Zhang, L.J.; Yuan, P.; He, H.P. Influence of Biochars on Plant Uptake and Dissipation of Two Pesticides in an Agricultural Soil. J. Agric. Food Chem. 2010, 58, 7915–7921. [Google Scholar] [CrossRef] [PubMed]
- Sun, K.; Gao, B.; Zhang, Z.; Zhang, G.; Zhao, Y.; Xing, B. Sorption of Atrazine and Phenanthrene by Organic Matter Fractions in Soil and Sediment. Environ. Pollut. 2010, 158, 3520–3526. [Google Scholar] [CrossRef] [PubMed]
- Losacco, D.; Tumolo, M.; Cotugno, P.; Leone, N.; Massarelli, C.; Convertini, S.; Tursi, A.; Uricchio, V.F.; Ancona, V. Use of Biochar to Improve the Sustainable Crop Production of Cauliflower (Brassica Oleracea L.). Plants 2022, 11, 1182. [Google Scholar] [CrossRef] [PubMed]
- Bigham: Method of Soil Analysis. Part 3. Chemical Methods. Available online: https://scholar.google.com/scholar_lookup?title=Methods+of+Soil+Analysis:+Part+3+Chemical+Methods&author=Bigham,+J.M.&publication_year=1996 (accessed on 3 January 2023).
- Stachniuk, A.; Szmagara, A.; Czeczko, R.; Fornal, E. LC-MS/MS Determination of Pesticide Residues in Fruits and Vegetables. J. Environ. Sci. Heal. Part B Pestic. Food Contam. Agric. Wastes 2017, 52, 446–457. [Google Scholar] [CrossRef]
- Olsen, S.R.; Cole, C.V.; Watanabe, S.; Dean, L.A. Estimation of Available Phosphorus in Soils by Extraction with Sodium bicarbonate. Sterling Robertson Olsen - Google Libros. Available online: https://books.google.it/books?hl=it&lr=&id=d-oaM88x5agC&oi=fnd&pg=PA3&ots=zZ0g-CkVXD&sig=qS9FwZLDvZrY9vshp3H6FTjt-Is&redir_esc=y#v=onepage&q&f=false (accessed on 3 January 2023).
- Losacco, D.; Campanale, C.; Tumolo, M.; Ancona, V.; Massarelli, C.; Uricchio, V.F. Evaluating the Influence of Nitrogen Fertilizers and Biochar on Brassica Oleracea L. Var. Botrytis by the Use of Fourier Transform Infrared (FTIR) Spectroscopy. Sustain. 2022, 14, 11985. [Google Scholar] [CrossRef]
- ImageJ Public Domain License. Available online: https://imagej.nih.gov/ij/docs/intro.html (accessed on 22 December 2022).
- Hossain, M.Z.; Bahar, M.M.; Sarkar, B.; Donne, S.W.; Wade, P.; Bolan, N. Assessment of the Fertilizer Potential of Biochars Produced from Slow Pyrolysis of Biosolid and Animal Manures. J. Anal. Appl. Pyrolysis 2021, 155, 105043. [Google Scholar] [CrossRef]
- Pukalchik, M.; Mercl, F.; Terekhova, V.; Tlustoš, P. Biochar, Wood Ash and Humic Substances Mitigating Trace Elements Stress in Contaminated Sandy Loam Soil: Evidence from an Integrative Approach. Chemosphere 2018, 203, 228–238. [Google Scholar] [CrossRef]
- Bandara, T.; Xu, J.; Potter, I.D.; Franks, A.; Chathurika, J.B.A.J.; Tang, C. Mechanisms for the Removal of Cd(II) and Cu(II) from Aqueous Solution and Mine Water by Biochars Derived from Agricultural Wastes. Chemosphere 2020, 254, 126745. [Google Scholar] [CrossRef]
- Chintala, R.; Mollinedo, J.; Schumacher, T.E.; Malo, D.D.; Julson, J.L. Effect of Biochar on Chemical Properties of Acidic Soil. Arch. Agron. Soil Sci. 2013, 60, 393–404. [Google Scholar] [CrossRef]
- Li, X.; Song, B.; Yin, D.; Lal, M.K.; Riaz, M.; Song, X.; Huang, W. Influence of Biochar on Soil Properties and Morphophysiology of Sugar Beet Under Fomesafen Residues. J. Soil Sci. Plant Nutr. 2023, 23, 1619–1632. [Google Scholar] [CrossRef]
- Buss, W.; Shepherd, J.G.; Heal, K.V.; Mašek, O. Spatial and Temporal Microscale PH Change at the Soil-Biochar Interface. Geoderma 2018, 331, 50–52. [Google Scholar] [CrossRef]
- Lentz, R.D.; Ippolito, J.A. Biochar and Manure Affect Calcareous Soil and Corn Silage Nutrient Concentrations and Uptake. J. Environ. Qual. 2012, 41, 1033–1043. [Google Scholar] [CrossRef]
- Chan, K.Y.; Van Zwieten, L.; Meszaros, I.; Downie, A.; Joseph, S.; Chan, K.Y.; Van Zwieten, L.; Meszaros, I.; Downie, A.; Joseph, S. Agronomic Values of Greenwaste Biochar as a Soil Amendment. Soil Res. 2007, 45, 629–634. [Google Scholar] [CrossRef]
- Lehmann, J.; Rillig, M.C.; Thies, J.; Masiello, C.A.; Hockaday, W.C.; Crowley, D. Biochar Effects on Soil Biota - A Review. Soil Biol. Biochem. 2011, 43, 1812–1836. [Google Scholar] [CrossRef]
- Phares, C.A.; Amoakwah, E.; Danquah, A.; Akaba, S.; Frimpong, K.A.; Mensah, T.A. Improved Soil Physicochemical, Biological Properties and Net Income Following the Application of Inorganic NPK Fertilizer and Biochar for Maize Production. Acta Ecol. Sin. 2022, 42, 289–295. [Google Scholar] [CrossRef]
- Wu, J.; Jin, L.; Wang, N.; Wei, D.; Pang, M.; Li, D.; Wang, J.; Li, Y.; Sun, X.; Wang, W.; et al. Effects of Combined Application of Chemical Fertilizer and Biochar on Soil Physio-Biochemical Properties and Maize Yield. Agriculture 2023, 13, 1200. [Google Scholar] [CrossRef]
- Dong, X.; Singh, B.P.; Li, G.; Lin, Q.; Zhao, X. Biochar Application Constrained Native Soil Organic Carbon Accumulation from Wheat Residue Inputs in a Long-Term Wheat-Maize Cropping System. Agric. Ecosyst. Environ. 2018, 252, 200–207. [Google Scholar] [CrossRef]
- Gross, A.; Bromm, T.; Glaser, B. Soil Organic Carbon Sequestration after Biochar Application: A Global Meta-Analysis. Agronomy 2021, 11, 2474. [Google Scholar] [CrossRef]
- Hui, D. Effects of Biochar Application on Soil Properties, Plant Biomass Production, and Soil Greenhouse Gas Emissions: A Mini-Review. Agric. Sci. 2021, 12, 213–236. [Google Scholar] [CrossRef]
- Amin, F.R.; Huang, Y.; He, Y.; Zhang, R.; Liu, G.; Chen, C. Biochar Applications and Modern Techniques for Characterization. Clean Technol. Environ. Policy 2016, 18, 1457–1473. [Google Scholar] [CrossRef]
- Mohan, D.; Singh, P.; Sarswat, A.; Steele, P.H.; Pittman, C.U. Lead Sorptive Removal Using Magnetic and Nonmagnetic Fast Pyrolysis Energy Cane Biochars. J. Colloid Interface Sci. 2015, 448, 238–250. [Google Scholar] [CrossRef]
- Chia, C.H.; Gong, B.; Joseph, S.D.; Marjo, C.E.; Munroe, P.; Rich, A.M. Imaging of Mineral-Enriched Biochar by FTIR, Raman and SEM-EDX. Vib. Spectrosc. 2012, 62, 248–257. [Google Scholar] [CrossRef]
- Gao, X.; Yang, J.; Liu, W.; Li, X.; Zhang, W.; Wang, A. Effects of Alkaline Biochar on Nitrogen Transformation with Fertilizer in Agricultural Soil. Environ. Res. 2023, 233, 116084. [Google Scholar] [CrossRef]
- Wang, X.; Zhao, Y.; Wang, H.; Zhao, X.; Cui, H.; Wei, Z. Reducing Nitrogen Loss and Phytotoxicity during Beer Vinasse Composting with Biochar Addition. Waste Manag. 2017, 61, 150–156. [Google Scholar] [CrossRef]
- Heaney, N.; Ukpong, E.; Lin, C. Low-Molecular-Weight Organic Acids Enable Biochar to Immobilize Nitrate. Chemosphere 2020, 240, 124872. [Google Scholar] [CrossRef]
- Cederlund, H.; Börjesson, E.; Stenström, J. Effects of a Wood-Based Biochar on the Leaching of Pesticides Chlorpyrifos, Diuron, Glyphosate and MCPA. J. Environ. Manage. 2017, 191, 28–34. [Google Scholar] [CrossRef]
- Palangi, S.; Bahmani, O.; Atlassi-pak, V. Effects of Wheat Straw Biochar Amendments to Soil on the Fate of Deltamethrin and Soil Properties. Environ. Technol. Innov. 2021, 23, 101681. [Google Scholar] [CrossRef]
- Cara, I.G.; Țopa, D.; Puiu, I.; Jităreanu, G. Biochar a Promising Strategy for Pesticide-Contaminated Soils. Agriculture 2022, 12, 1579. [Google Scholar] [CrossRef]
- Dai, Y.; Zhang, N.; Xing, C.; Cui, Q.; Sun, Q. The Adsorption, Regeneration and Engineering Applications of Biochar for Removal Organic Pollutants: A Review. Chemosphere 2019, 223, 12–27. [Google Scholar] [CrossRef]
- Singh, N.; Singh, S.B.; Mukerjee, I.; Gupta, S.; Gajbhiye, V.T.; Sharma, P.K.; Goel, M.; Dureja, P. Metabolism of 14C-Azoxystrobin in Water at Different PH. J. Environ. Sci. Health-Part B Pestic. Food Contam. Agric. Wastes 2010, 45, 123–127. [Google Scholar] [CrossRef] [PubMed]
- Bending, G.D.; Lincoln, S.D.; Edmondson, R.N. Spatial Variation in the Degradation Rate of the Pesticides Isoproturon, Azoxystrobin and Diflufenican in Soil and Its Relationship with Chemical and Microbial Properties. Environ. Pollut. 2006, 139, 279–287. [Google Scholar] [CrossRef]
- Yang, Y.; Chun, Y.; Shang, G.; Huang, M. PH-Dependence of Pesticide Adsorption by Wheat-Residue-Derived Black Carbon. Langmuir 2004, 20, 6736–6741. [Google Scholar] [CrossRef] [PubMed]
- Rasool, S.; Rasool, T.; Gani, K.M. A Review of Interactions of Pesticides within Various Interfaces of Intrinsic and Organic Residue Amended Soil Environment. Chem. Eng. J. Adv. 2022, 11, 100301. [Google Scholar] [CrossRef]
- You, X.; Zheng, H.; Ge, J.; Fang, S.; Suo, F.; Kong, Q.; Zhao, P.; Zhang, G.; Zhang, C.; Li, Y. Effect of Biochar on the Enantioselective Soil Dissipation and Lettuce Uptake and Translocation of the Chiral Pesticide Metalaxyl in Contaminated Soil. J. Agric. Food Chem. 2019, 67, 13550–13557. [Google Scholar] [CrossRef] [PubMed]
- Egamberdieva, D.; Jabbarov, Z.; Arora, N.K.; Wirth, S.; Bellingrath-Kimura, S.D. Biochar Mitigates Effects of Pesticides on Soil Biological Activities. Environ. Sustain. 2021, 4, 335–342. [Google Scholar] [CrossRef]
- Oleszczuk, P.; Jośko, I.; Futa, B.; Pasieczna-Patkowska, S.; Pałys, E.; Kraska, P. Effect of Pesticides on Microorganisms, Enzymatic Activity and Plant in Biochar-Amended Soil. Geoderma 2014, 214–215, 10–18. [Google Scholar] [CrossRef]
- Rafael, R.B.A.; Fernández-Marcos, M.L.; Cocco, S.; Ruello, M.L.; Fornasier, F.; Corti, G. Benefits of Biochars and NPK Fertilizers for Soil Quality and Growth of Cowpea (Vigna Unguiculata L. Walp.) in an Acid Arenosol. Pedosphere 2019, 29, 311–333. [Google Scholar] [CrossRef]
- Hussain, F.; Hussain, I.; Khan, A.H.A.; Muhammad, Y.S.; Iqbal, M.; Soja, G.; Reichenauer, T.G.; Zeshan; Yousaf, S. Combined Application of Biochar, Compost, and Bacterial Consortia with Italian Ryegrass Enhanced Phytoremediation of Petroleum Hydrocarbon Contaminated Soil. Environ. Exp. Bot. 2018, 153, 80–88. [Google Scholar] [CrossRef]
- You, X.; Yin, S.; Suo, F.; Xu, Z.; Chu, D.; Kong, Q.; Zhang, C.; Li, Y.; Liu, L. Biochar and Fertilizer Improved the Growth and Quality of the Ice Plant (Mesembryanthemum Crystallinum L.) Shoots in a Coastal Soil of Yellow River Delta, China. Sci. Total Environ. 2021, 775, 144893. [Google Scholar] [CrossRef]
- Kammann, C.I.; Schmidt, H.P.; Messerschmidt, N.; Linsel, S.; Steffens, D.; Müller, C.; Koyro, H.W.; Conte, P.; Stephen, J. Plant Growth Improvement Mediated by Nitrate Capture in Co-Composted Biochar. Sci. Rep. 2015, 5, 1–13. [Google Scholar] [CrossRef]
- Tarar, O.F.; Asghar, A.; Qayyum, S.A.; Kanwal, H.; Lateef, A.; Nazir, R.; Imam Abidi, S.H.; Naeem, M.K.; Shahid, B. Synthesis and Surface Morphology of Banana Biochar-Based Nano-Fertilizer and Its Effect on First Stages of Growth Parameters of Cucumber, Broccoli, and Red Okra. J. Saudi Soc. Agric. Sci. 2023, 22, 535–545. [Google Scholar] [CrossRef]
- Yan, B.; Zhang, Y.; Wang, Y.; Rong, X.; Peng, J.; Fei, J.; Luo, G. Biochar Amendments Combined with Organic Fertilizer Improve Maize Productivity and Mitigate Nutrient Loss by Regulating the C–N–P Stoichiometry of Soil, Microbiome, and Enzymes. Chemosphere 2023, 324, 138293. [Google Scholar] [CrossRef] [PubMed]
- Doan, T.T.; Henry-Des-Tureaux, T.; Rumpel, C.; Janeau, J.L.; Jouquet, P. Impact of Compost, Vermicompost and Biochar on Soil Fertility, Maize Yield and Soil Erosion in Northern Vietnam: A Three Year Mesocosm Experiment. Sci. Total Environ. 2015, 514, 147–154. [Google Scholar] [CrossRef]
- Pan, X.; Zhang, Y.; Wang, X.; Liu, G. Effect of Adding Biochar with Wood Vinegar on the Growth of Cucumber. IOP Conf. Ser. Earth Environ. Sci. 2017, 61, 012149. [Google Scholar] [CrossRef]



| Biochar Type | Pesticide | Pesticide Type | % Decrease | Reference |
|---|---|---|---|---|
| Cassava wastes | Atrazine | H | 96% | [20] |
| Biochar mix | Atrazine | H | 90% | [21] |
| Rice husk | Glyphosate | H | 82% | [22] |
| Maize straw and pig manure | Thiacloprid | I | 81% | [23] |
| Pine woodchips | Pyrimethanil | F | 72% | [24] |
| Oil palm and rice husk | Imazapyr | H | 70% | [25] |
| Rice straw | Pyrazosulfuron-ethyl | H | 66% | [26] |
| Oil palm and rice husk | Imazapic | H | 60% | [25] |
| Pine chip | Atrazine | H | 52% | [20] |
| Pine woodchips | Boscalid | F | 49% | [24] |
| Cotton straw | Fipronil | I | 48% | [20] |
| Wheat straw | Pyrazosulfuron-ethyl | H | 47% | [26] |
| Calligonum comosum | Atrazine | H | 45% | [27] |
| Vegetable waste | Diuron | H | 45% | [28] |
| Rice husk | Methamidophos, phorate, isocarbophos, terbufos, malathion, parathion | I | 32% | [26] |
| Cotton straw | Chlorpyrifos | I | 19% | [26] |
| Red gum, woodchips | Pyrimethanil | F | 14% | [26] |
| Cotton straw | Carbofuran, chlorpyrifos | I | / | [22] |
| Tea waste | Carbofuran | I | / | [29] |
| Parameters | C | FP | FPMB | FPWB | p |
|---|---|---|---|---|---|
| pH (H2O) | 6.9 b ± 0.03 | 6.4 c ± 0.03 | 7.2 a ± 0.01 | 5.7 d ± 0.01 | <0.001 |
| Electrical Conductivity (µS) | 1001.8 c ± 1.03 | 1492.5 a ± 0.87 | 1029.3 b ± 0.47 | 1027.8 b ±3.09 | 0.0 |
| Water Content (g kg−1) | 151.1 b ± 0.59 | 142.2 c ± 2.3 | 112.8 d± 0.12 | 191.8 a ± 0.35 | <0.001 |
| Available P (mg kg−1) | 46.0 b ± 0.27 | 13.0 d ± 0.18 | 64.8 a ± 0.81 | 25.7 c ± 0.2 | 0.0 |
| Organic C (g kg−1) | 225 b ± 0.4 | 213.7 c ± 0.37 | 178.3 d ±0.25 | 272.8 a ± 0.25 | 0.0 |
| Total C (%) | 34.8 b ± 0.96 | 27.5 d ± 0.67 | 42.1 a ± 0.48 | 28.6 c ± 1.81 | <0.001 |
| Total N (%) | 0.77 b ± 0.04 | 0.7 b ± 0.03 | 0.93 a ± 0.01 | 0.64 c ± 0.00 | <0.001 |
| Treatments | -N Concentrations (mg/L) (One-Way ANOVA) |
-N Concentrations (mg/L) (One-Way ANOVA) | ||
|---|---|---|---|---|
| 10 Days Post-I° N Fertilization | 10 Days Post-II° N Fertilization | 10 Days Post-I° N Fertilization | 10 Days Post-II° N Fertilization | |
| Soil without treatment (C) | 0.54 d ± 0.03 | 1.71 b ± 0.04 | 1.83 d ± 0.10 | 5.88 b ± 0.16 |
| Fertilized soil and treated with Pesticides (FP) | 31.09 a ± 0.74 | 37.90 a ± 1.20 | 106.76 a ± 2.53 | 130.14 a ± 4.12 |
| Soil FP with the application of Wood Biochar (FPWB) | 22.37 b ± 0.69 | 38.99 a ± 1.01 | 76.82 b ± 2.38 | 133.88 a ± 3.50 |
| Soil FP with the application of Mixed Biochar (FPMB) | 12.75 c ± 0.21 | 37.08 a ± 0.61 | 43.78 c ± 0.72 | 127 a ± 2.09 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Losacco, D.; Campanale, C.; Triozzi, M.; Massarelli, C.; Uricchio, V.F. Application of Wood and Vegetable Waste-Based Biochars in Sustainable Agriculture: Evaluation on Nitrate Leaching, Pesticide Fate, Soil Properties, and Brassica oleracea Growth. Environments 2024, 11, 13. https://doi.org/10.3390/environments11010013
Losacco D, Campanale C, Triozzi M, Massarelli C, Uricchio VF. Application of Wood and Vegetable Waste-Based Biochars in Sustainable Agriculture: Evaluation on Nitrate Leaching, Pesticide Fate, Soil Properties, and Brassica oleracea Growth. Environments. 2024; 11(1):13. https://doi.org/10.3390/environments11010013
Chicago/Turabian StyleLosacco, Daniela, Claudia Campanale, Mariangela Triozzi, Carmine Massarelli, and Vito Felice Uricchio. 2024. "Application of Wood and Vegetable Waste-Based Biochars in Sustainable Agriculture: Evaluation on Nitrate Leaching, Pesticide Fate, Soil Properties, and Brassica oleracea Growth" Environments 11, no. 1: 13. https://doi.org/10.3390/environments11010013
APA StyleLosacco, D., Campanale, C., Triozzi, M., Massarelli, C., & Uricchio, V. F. (2024). Application of Wood and Vegetable Waste-Based Biochars in Sustainable Agriculture: Evaluation on Nitrate Leaching, Pesticide Fate, Soil Properties, and Brassica oleracea Growth. Environments, 11(1), 13. https://doi.org/10.3390/environments11010013









