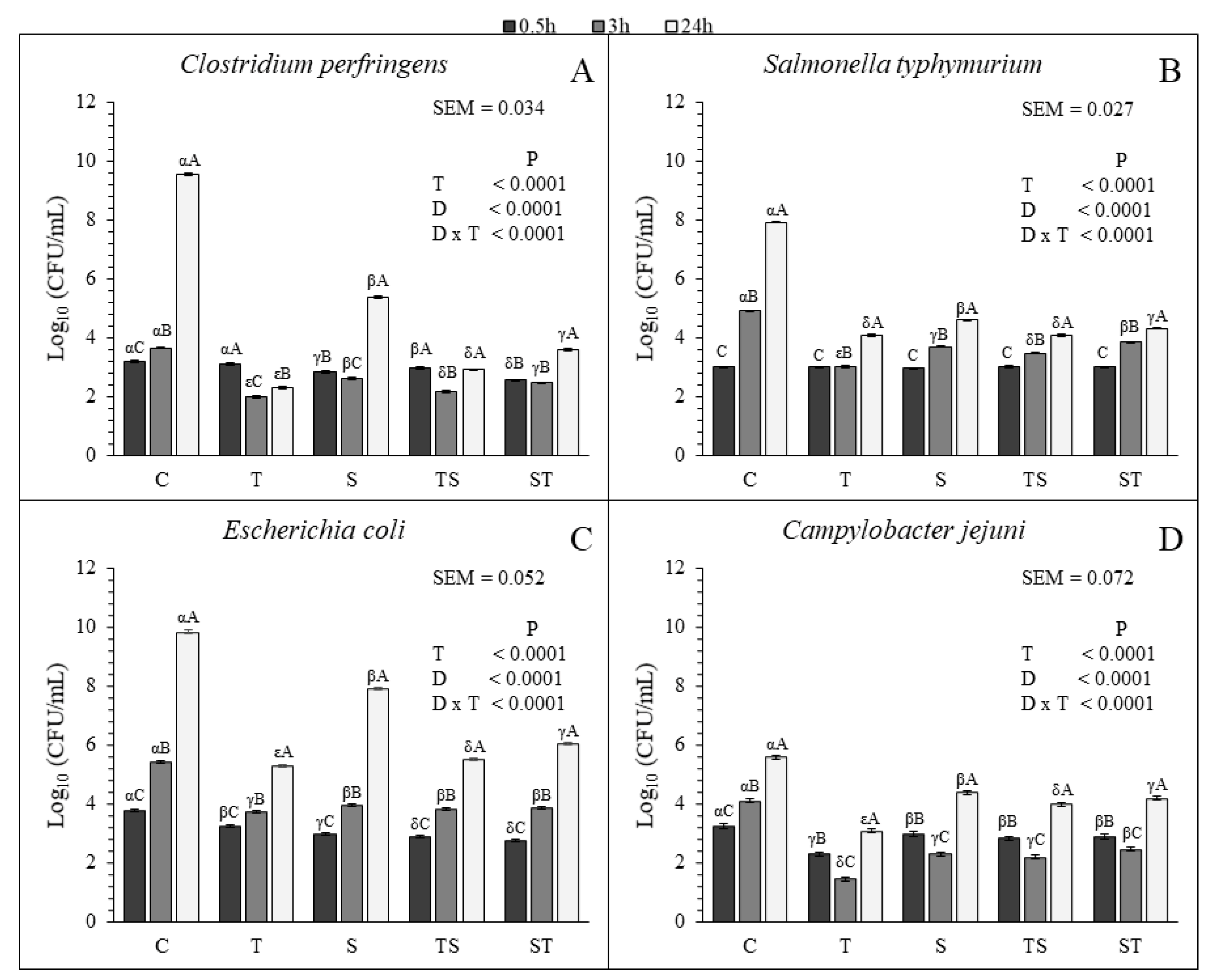
3.1. Microbiological Assay
All the treatments were efficient in decreasing the bacterial growth of each species compared to the control (
Figure 1). The T resulted the most effective treatment in controlling the growth of each bacterial species at 3 h and 24 h. For the other treatments, the behavior of the tested bacteria was different.
C. perfringens and
S. typhymurium resulted more sensitive to the TS than to the ST and S at 3 h and 24 h. At 3 h no significant differences were found for the S, TS and ST for
E. coli but at 24 h the TS was more efficient in limiting the growth compared to the other two treatments (
Figure 1C). No significant different growth was observed for
C. jejuni with S and TS at 3 h. However, at the same sampling time, the growth with ST was higher than the growth with S and TS. At 24 h the growth of
C. jejuni was lower with the TS than with the S and ST (
Figure 1D).
All the treatments lowered the growth rate of each microbial species, except for
C. jejuni that did not show significant decreasing with the tested compounds between 3 h and 24 h, compared to the control. Furthermore, both
C. perfringens and
C. jejuni decreased in all the treatments between 0.5 h and 3 h, and increased between 3 h and 24 h (
Table 5 and
Figure 1). This observation suggested that the bactericidal effect of CT and SN1, supplied alone or in combination, is stronger at the beginning of the treatment. For
E. coli and
S. typhymurium only a bacteriostatic effect was observed in the treated cultures (
Table 5 and
Figure 1).
Our results are in accordance with Tosi et al. [
2] and Redondo et al. [
3] who demonstrated that the CT can inhibit the growth of
C. perfringens. The antimicrobial activity of tannins seems to be due to their ability to bind microbial enzymes and proteins, in ion deprivation and in inhibiting the topoisomerase, fundamental for the DNA replication [
36,
37,
38]. Moreover, Ramìrez et al. [
6] and Timbermont et al. [
10] showed that the SN1 of SMCFA were efficient in controlling the growth of
S. typhimurium and
C. perfringens, respectively, consistent with our study. Their antimicrobial effect was explained with their ability to penetrate through the bacterial wall, because of their affinity with lipoteichoic acid, present in microbial membrane. Their ability to destroy the inner the membrane is probably due to their compatibility with hydrophilic and hydrophobic moieties [
6,
39,
40,
41]. These results suggest that the CT and SN1 alone or in combination could be useful to control the proliferation of pathogenic bacteria tested in this trial. Hence, these molecules could represent a valid alternative to antibiotics both used alone or in mixture.
3.2. In Vivo Trial
No differences among the groups were found for feed intake (
Table 6), both in each single growth period and in the whole period of bird life, showing that the supplementation with CT and SN1 blends did not affect the palatability of the diets. No differences were found for weight gain and feed efficiency among groups, suggesting that the blends of CT and SN1, at the inclusion level adopted in this study, did not interfere with nutrient absorption, with respect to the single supplementation or to the control diet (
Table 6). Hence, no synergic effect was found when the CT and SN1 mixtures were included in the diets. The results showed a higher feed/gain ratio during the first two weeks, compared to the growing and finisher periods. Usually, young chicks have higher feed efficiency than old birds. This trend could be due to an adaptation period of birds to the rearing condition because also the control group, fed with only the basal diet, did not show significant differences with the other feeding groups. The literature reports information on the effect of CT and SN1 when they are included alone in the diets, but few data are available on the effect of blends composed by a mixture of tannin extracts and monoglycerides. The results of this trial are in accordance with Jamroz et al. [
42] and Antongiovanni et al. [
13,
14,
43] who studied respectively the effect of CT (inclusion level of 0, 250, 500 and 1000 mg/kg on DM) and of several monoglycerides (inclusion level of 200, 350, 500 mg/kg of DM) separately, as dietary supplementation on the performance and histological characteristics of the intestine wall in chickens. No impairment of the growth performance emerged, despite a slight modification on the small intestine wall, due to the introduction in the diet of chestnut tannins and monobutyrin (CT degrades enterocytes while monobutyrin modifies positively villi, microvilli and crypts), was observed. Moreover, previous results, reported by Schiavone et al. [
17], showed that the inclusion of a natural extract of chestnut wood did not affect the apparent digestibility of CP and that this supplement had a positive effect on average daily gain and feed intake in the first two weeks of addition. For monoglycerides, especially with butyric acid, the literature confirms that they can ameliorate growth performances and health in broilers [
12,
13,
14]. In contrast, for the CT, several studies reported that polyphenols reduce protein digestibility in monogastrics, decreasing the productive performances in accordance with a lower availability of this nutrient for the animal nutritional requirements [
5,
44,
45,
46]. In particular, tannins stimulate hypersecretion of endogenous enzymes leading to losses of sulphur aminoacids in poultry species [
47,
48,
49]. The inconsistence of the results reported in many papers, including those shown here, is probably due to the kind of tannin used as dietary supplement, the animal species and the dietary dose formulation. In this study, by an accurate observation, an astringent effect of tannin has been noted in T, TS and ST groups, whose litters resulted drier than the litters of S group (at 35th day: C = 1; T = 0; TS = 0; ST = 0 and S = 1).
No significant differences in carcass quality were found among groups (
Table 7), except for the liver that was smaller in the animals fed the T and S diets than the other feeding groups. This result is consistent with the findings reported by Jamroz et al. [
42] and Antongiovanni et al. [
13] who noted that the supplementation with polyphenols or the monoglycerides of butyric acid did not affect carcass quality, even though monoglycerides represented an energy source for animal growth and tannins are considered to be antinutritive. Unfortunately, in literature, no information is available on blends of CT and SN1, which were not able to affect carcass traits at the tested levels in our study.
Changes in the a* and b* values are related to changes in meat color because these parameters are markers of browning [
50]. Where the SN1 was present in the diet alone an increase of the L* and b* values occurred (
Table 8). Specifically, the S group showed the highest L* and b* values and it significantly differed compared to the other groups. Despite the statistical differences, ΔE calculation for the samples of chicken breast can be useful to understand how the values of color can be perceived by human eyes (
Table 9). As suggested by Mokrzycki and Tatol [
26], a standard observer is able to see the difference in color as follows: 0 < ∆E < 1 observer does not notice the difference, 1 < ∆E < 2 only experienced observers can notice the difference, 2 < ∆E < 3.5 unexperienced observer also notices the difference, 3.5 < ∆E < 5 clear difference in color is noticed, ∆E > 5 observer notices two different colors. Regarding the ∆E, from
Table 9 it emerged that the T and TS were the most similar groups (1 < ∆E < 2), while the ST assumed a 2 < ∆E < 3.5 when compared with C, T, and TS. Interestingly, the S group was found to be the only one with a ∆E > 5 when compared to the other experimental groups, thus underlining that Sn1 monoglycerides strongly impacted meat coloration. Whether this modification could be accepted or not by consumers should be further investigated.
Data reported in this study showed that both the tested blends of CT and SN1 could be utilized as dietary supplements without impairing animal welfare, growing performances and meat quality, thus representing valid alternatives to antibiotics in poultry rearing. Contrariwise, the S diet deeply modified the color of breast meat which could result in a modification of consumers’ acceptance.
In this trial, data related to the antioxidant status of the breast meat did not show significant differences among the groups (
Table 10). Several studies demonstrated the antioxidant power of tannins and of polyunsaturated fatty acids but not for SMCFA, because of the lack of double bounds on carbon chain [
51,
52,
53]. Indeed, several authors reported that ellagic tannins in humans and rats are gradually metabolized by the intestinal microbiota to produce different metabolites with antioxidant effects [
54,
55,
56,
57]. Luciano et al. [
53] found that the inclusion at 8.96% (DM basis) of quebracho tannins in lamb diet produced an improvement in the antioxidant status of
Longissimus dorsi muscle, measured as both its ferric reducing ability and its radical scavenging ability. Other authors reported similar results in beef meat [
30,
58,
59]. In contrast, Gladine et al. [
60] found no effect of polyphenols in rat muscle for the radical scavenging activity. The inability of many polyphenols to be metabolized by the gastrointestinal tract of animals is strongly linked to their molecular structure and solubility, the dose of inclusion and the animal species.
Nowadays, in conventional and intensive poultry production, antibiotics are used to control diseases and to prevent the mortality of birds, responsible for a huge economical loss. This approach conflicts with the sustainability of animal productions because several issues about the development of bacterial resistance, dejection disposal, food safety and human health occur. Therefore, the poultry industry needs alternatives able to replace antibiotics with natural or synthetic compounds able to simulate natural molecules. Chestnut tannins are a by-product of wood industry because they are obtained by distillation of wood used in the building industry. SN1 is obtained by recycling glycerol derived from biodiesel production. Hence, CT and SN1 are part of the concept of bio-economy. Moreover, FAO reported that livestock support the livelihoods and food supply of almost 1.3 billion people, being one of the fastest growing areas of the agricultural economy in the world. In developing countries, poultry production plays an important role in food, and pathogen proliferation represents an important public health problem that cannot be underestimated. At the same time, environmental sustainability must be ensured [
61]. Data reported in this study showed that CT, SN1 or their blends could represent a valid alternative to antibiotics in poultry rearing. Although literature shows several studies in which tannins exert antinutritional effects in monogastric, in the present in vivo trial, no detrimental effects were observed on animal welfare, performance or meat quality. However, it is well known that the kind of polyphenols and the dietary inclusion level are fundamental to explain the biological and nutritional effects of dietary tannins. Besides, the literature shows unequivocal positive effects of SN1 monoglycerides in protecting gut from pathogens, by providing energy to enterocytes and by favoring the development of gut villi [
12,
13,
16]. Hence, the blends of CT and SN1 could represent a good compromise among antimicrobial activities, animal gut protection, meat quality and production sustainability.