Characterization of the Barley Net Blotch Pathosystem at the Center of Origin of Host and Pathogen
Abstract
1. Introduction
2. Results
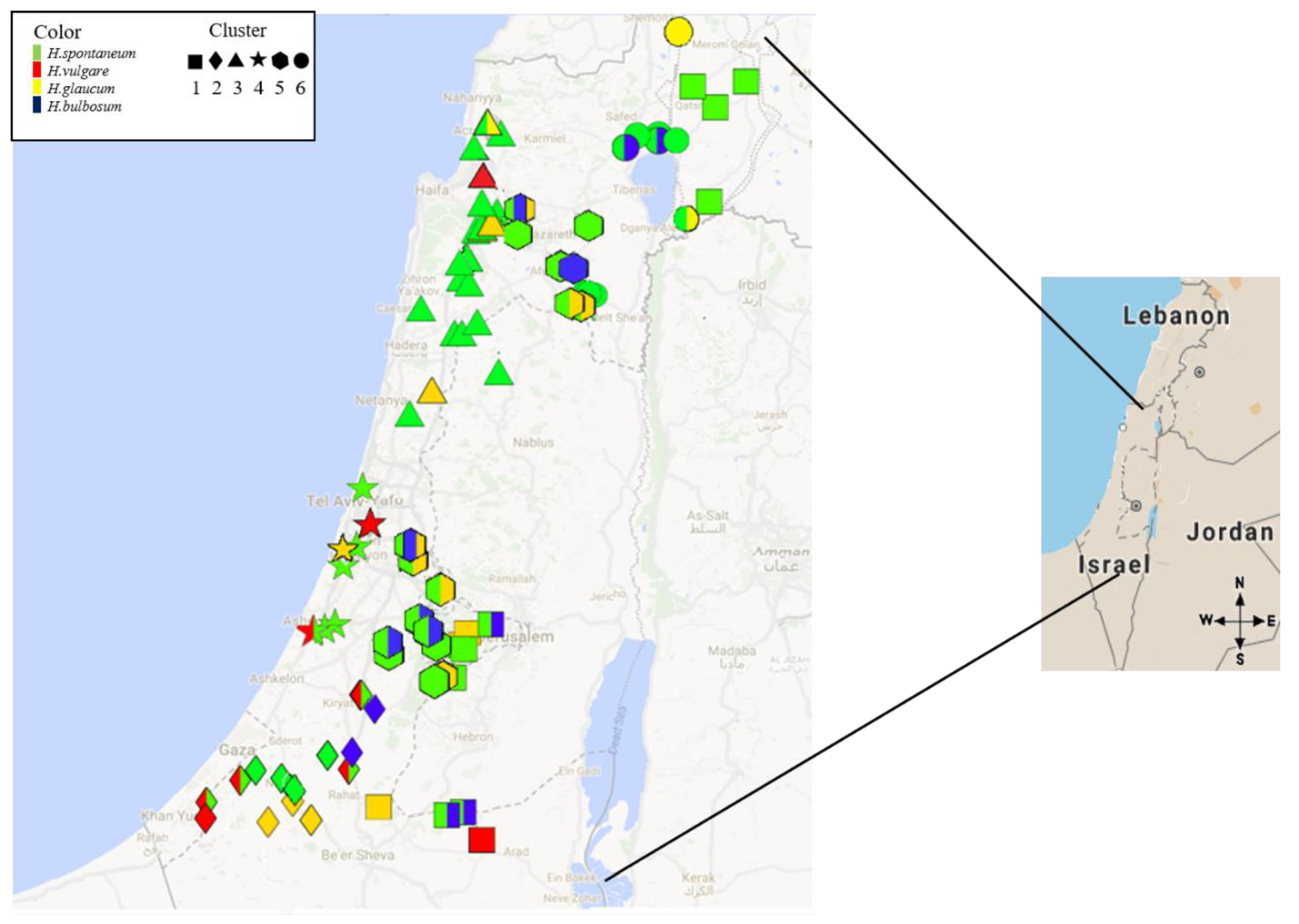
2.1. Spatial Survey Illustrated that Net Blotch is Distributed Across a Range of Hosts and Eco-Geographical Conditions
2.2. High Frequency of Pyrenophora teres f. maculata among P. teres Isolates
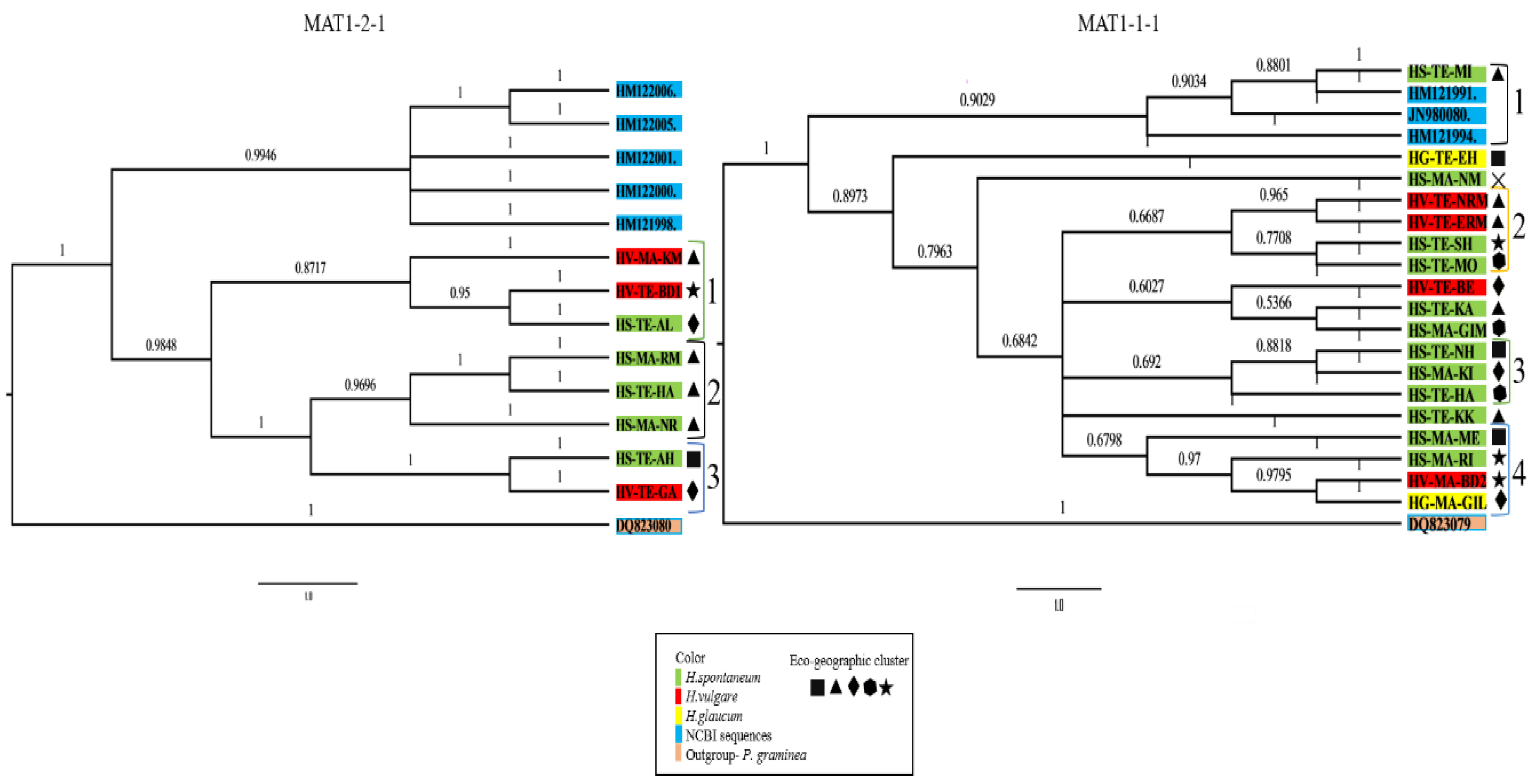
2.3. P. teres Isolates Cluster into Several Phylogenetic Groups
2.4. Substantial Variability in Saprotrophic Growth Rates
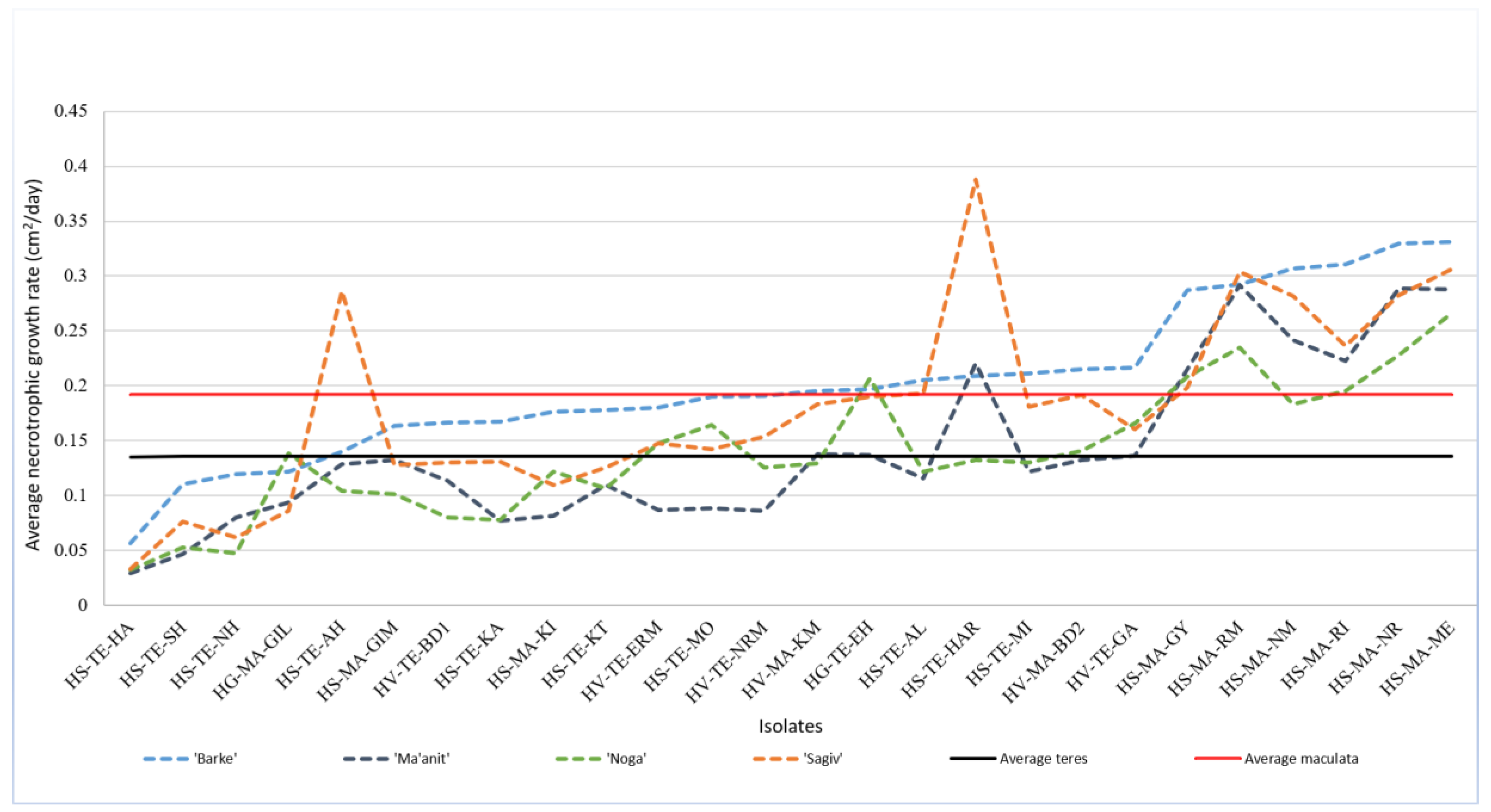
2.5. Necrotrophic Growth Rates Reveal Substantial Variability among Isolates, and Higher Growth Rates of Ptm Isolates
2.5.1. Isolates from Wild Hosts Have More Variable Necrotrophic Growth Rates
2.5.2. Isolate X Host Interactions
2.5.3. Correlation Between Necrotrophic and Saprotrophic Growth Rates among Isolates
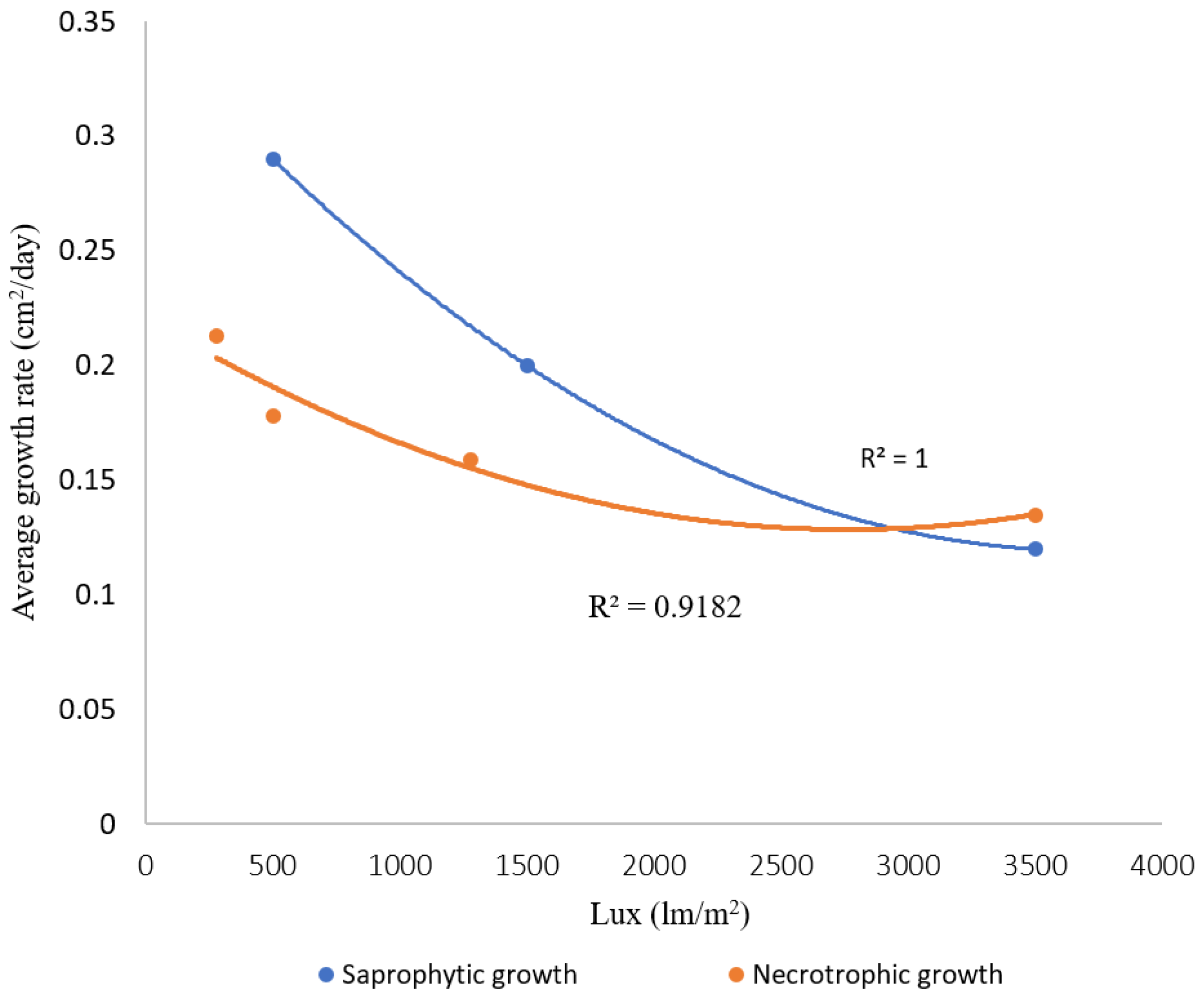
2.6. Light Intensity is Negatively Correlated with Saprotrophic and Necrotrophic Growth Rates
3. Discussion
3.1. Disease Distribution
3.2. Characterization of the NB Isolates Collection
3.2.1. Genetic Characterization
3.2.2. Characterization of Virulence on Wild vs. Domesticated Host
3.2.3. The Prominent Role of P. teres f. maculata
3.2.4. Reduced Light Intensity, a Potential Driver of NB Epidemics
4. Materials and Methods
4.1. Sampling Procedure
4.2. Plant and Pathogen Material
4.3. Genomic DNA Extraction, Primers, PCR, and Sequence Analysis
4.4. Phylogenetic Analysis
4.5. Phenotypic Assay
4.6. Statistical Analyses
5. Conclusions
Supplementary Materials
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Liu, Z.; Ellwood, S.R.; Oliver, R.P.; Friesen, T.L. Pyrenophora teres: Profile of an increasingly damaging barley pathogen. Mol. Plant Pathol. 2011, 12, 1–19. [Google Scholar] [CrossRef]
- McLean, M.S.; Howlett, B.J.; Hollaway, G.J. Erratum to: Epidemiology and control of spot form of net blotch (Pyrenophora teres f. maculata) of barley: A review. Crop Pasture Sci. 2009, 60, 499. [Google Scholar]
- Kenneth, R. Aspects of the Taxonomy, Biology and Epidemiology of Pyrenophora teres Drechsl. (Drechslera teres (SACC.) Shoemaker), the Causal Agent of Net Blotch Disease of Barley. In Doctoral Dissertation; The Hebrew University: Jerusalem, Israel, 1960. [Google Scholar]
- McDonald, B.A.; Linde, C. Pathogen population genetics, evolutionary potential, and durable resistance. Annu. Rev. Phytopathol. 2002, 40, 349–379. [Google Scholar] [CrossRef]
- Ronen, M. The Net Blotch Disease in Barley-Characterization of the Pathogen Pyrenophora teres f. teres and Screening for Resistance in Wild and Domesticated. In MSc thesis; Bar Ilan University: Ramat-Gan, Israel, 2017. [Google Scholar]
- Zaffarano, P.L.; McDonald, B.A.; Zala, M.; Linde, C.C. Global hierarchical gene diversity analysis suggests the fertile crescent is not the center of origin of the barley scald pathogen Rhynchosporium secalis. Phytopathology 2006, 96, 941–950. [Google Scholar] [CrossRef]
- Ben-David, R.; Parks, R.; Dinoor, A.; Kosman, E.; Wicker, T.; Keller, B.; Cowger, C. Differentiation among Blumeria graminis f. sp. tritici isolates originating from wild versus domesticated Triticum Species in Israel. Phytopathology 2016, 106, 861–870. [Google Scholar]
- Ben-David, R.; Dinoor, A.; Peleg, Z.; Fahima, T. Reciprocal hosts responses to powdery mildew isolates originating from domesticated wheats and their wild progenitor. Front. Plant Sci. 2018, 9, 75. [Google Scholar] [CrossRef] [PubMed]
- Frenkel, O.; Sherman, A.; Abbo, S.; Shtienberg, D. Different ecological affinities and aggressiveness patterns among Didymella rabiei isolates from sympatric domesticated chickpea and wild Cicer judaicum. Phytopathology 2008, 98, 600–608. [Google Scholar] [CrossRef] [PubMed]
- Golani, M.; Abbo, S.; Sherman, A.; Frenkel, O.; Shtienberg, D. The temperature response and aggressiveness of Peyronellaea pinodes Isolates originating from wild and domesticated Pisum sp. in Israel. Phytopathology 2016, 106, 824–832. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Turgeon, B.G. Application of mating type gene technology to problems in fungal biology. Annu. Rev. Phytopathol. 1998, 36, 115–137. [Google Scholar] [CrossRef] [PubMed]
- Rau, D.; Maier, F.J.; Papa, R.; Brown, A.H.D.; Balmas, V.; Saba, E.; Schaefer, W.; Attene, G. Isolation and characterization of the mating-type locus of the barley pathogen Pyrenophora teres and frequencies of mating-type idiomorphs within and among fungal populations collected from barley landraces. Genome 2005, 48, 855–869. [Google Scholar] [CrossRef][Green Version]
- Huelsenbeck, J.P.; Ronquist, F. MRBAYES: Bayesian inference of phylogenetic trees. Bioinformatics 2001, 17, 754–755. [Google Scholar] [CrossRef] [PubMed]
- Schoch, C.L.; Seifert, K.A.; Huhndorf, S.; Robert, V.; Spouge, J.L.; Levesque, C.A.; Chen, W.; Bolchacova, E.; Voigt, K.; Crous, P.W.; et al. Nuclear ribosomal internal transcribed spacer (ITS) region as a universal DNA barcode marker for Fungi. Proc. Natl. Acad. Sci. USA. 2012, 109, 6241–6246. [Google Scholar] [CrossRef] [PubMed]
- Srivastava, D.A.; Yakubov, M.; Feldbaum, R.; Tish, N.; Shoyhet, H.; Manasherova, E.; Pandaranayaka, E.P.J.; Rav-David, D.; Elad, Y.; Harel, A. Multiparametric analysis of diversity in Botrytis cinerea isolates from Israel. Phytoparasitica 2018, 46, 569–581. [Google Scholar] [CrossRef]
- Lev-Yadun, S.; Gopher, A.; Abbo, S. The cradle of agriculture. Science 2000, 288, 1602–1603. [Google Scholar] [CrossRef] [PubMed]
- Hübner, S.; HÖffken, M.; Oren, E.; Haseneyer, G.; Stein, N.; Graner, A.; Schmid, K.; Fridman, E. Strong correlation of wild barley (Hordeum spontaneum) population structure with temperature and precipitation variation. Mol. Ecol. 2009, 18, 1523–1536. [Google Scholar] [CrossRef]
- Salamini, F.; Özkan, H.; Brandolini, A.; Schäfer-Pregl, R.; Martin, W. Genetics and geography of wild cereal domestication in the near east. Nat. Rev. Genet. 2002, 3, 429–441. [Google Scholar] [CrossRef]
- Dinoor, A. Role of wild and cultivated plants in the epidemiology of plant diseases in Israel. Annu. Rev. Phytopathol. 1974, 12, 413–436. [Google Scholar] [CrossRef]
- Walther, U.; Rapke, H.; Proeseler, G.; Szigat, G. Hordeum bulbosum—A new source of disease resistance—Transfer of resistance to leaf rust and mosaic viruses from H. bulbosum into winter barley. Plant Breed. 2000, 119, 215–218. [Google Scholar] [CrossRef]
- Pickering, R.; Ruge-Wehling, B.; Johnston, P.A.; Schweizer, G.; Ackermann, P.; Wehling, P. The transfer of a gene conferring resistance to scald (Rhynchosporium secalis) from Hordeum bulbosum into H. vulgare chromosome 4HS. Plant Breed. 2006, 125, 576–579. [Google Scholar] [CrossRef]
- Scholz, M.; Ruge-Wehling, B.; Habekuss, A.; Schrader, O.; Pendinen, G.; Fischer, K.; Wehling, P. Ryd4 (Hb): A novel resistance gene introgressed from Hordeum bulbosum into barley and conferring complete and dominant resistance to the barley yellow dwarf virus. Theor. Appl. Genet. 2009, 119, 837–849. [Google Scholar] [CrossRef]
- Stevens, R.B. Cultural practices in disease controltle. In Plant Pathology; Academic Press: Cambridge, MA, USA, 1960; pp. 357–429. [Google Scholar]
- Rau, D.; Rodriguez, M.; Leonarda Murgia, M.; Balmas, V.; Bitocchi, E.; Bellucci, E.; Nanni, L.; Attene, G.; Papa, R. Co-evolution in a landrace meta-population: Two closely related pathogens interacting with the same host can lead to different adaptive outcomes. Sci. Rep. 2015, 5, 12834. [Google Scholar] [CrossRef] [PubMed]
- Rau, D.; Attene, G.; Brown, A.H.D.; Nanni, L.; Maier, F.J.; Balmas, V.; Saba, E.; Schäfer, W.; Papa, R. Phylogeny and evolution of mating-type genes from Pyrenophora teres, the causal agent of barley “net blotch” disease. Curr. Genet. 2007, 51, 377–392. [Google Scholar] [CrossRef] [PubMed]
- Lu, S.; Edwards, M.C.; Friesen, T.L. Genetic variation of single nucleotide polymorphisms identified at the mating type locus correlates with form-specific disease phenotype in the barley net blotch fungus Pyrenophora teres. Eur. J. Plant Pathol. 2013, 135, 49–65. [Google Scholar] [CrossRef]
- Andrie, R.M.; Schoch, C.L.; Hedges, R.; Spatafora, J.W.; Ciuffetti, L.M. Homologs of ToxB, a host-selective toxin gene from Pyrenophora tritici-repentis, are present in the genome of sister-species Pyrenophora bromi and other members of the Ascomycota. Fungal Genet. Biol. 2008, 45, 363–377. [Google Scholar] [CrossRef] [PubMed]
- Hübner, S.; Günther, T.; Flavell, A.; Fridman, E.; Graner, A.; Korol, A.; Schmid, K.J. Islands and streams: Clusters and gene flow in wild barley populations from the Levant. Mol. Ecol. 2012, 21, 1115–1129. [Google Scholar] [CrossRef] [PubMed]
- Vanderplank, J. Disease Resistance in Plants, 2nd ed.; Academic Press: Orlando, FL, USA, 1984. [Google Scholar]
- Afanasenko, O.S.; Jalli, M.; Pinnschmidt, H.O.; Filatova, O.; Platz, G.J. Development of an international standard set of barley differential genotypes for Pyrenophora teres f. teres. Plant Pathol. 2009, 58, 665–676. [Google Scholar] [CrossRef]
- McLean, M.S.; Howlett, B.J.; Hollaway, G.J. Spot form of net blotch, caused by Pyrenophora teres f. maculata, is the most prevalent foliar disease of barley in Victoria, Australia. Australas. Plant Pathol. 2010, 39, 46–49. [Google Scholar]
- Marshall, J.M.; Kinzer, K.; Brueggeman, R.S. First report of Pyrenophora teres f. maculata the cause of spot form net blotch of barley in Idaho. Plant Dis. 2015, 99, 1860. [Google Scholar]
- NOAA (National Oceanic and Atmospheric Administration). Available online: https://www.wrh.noaa.gov/boi/climo/average%20annual%20temperatures%20at%20boise,%20airport.txt (accessed on 29 November 2019).
- Hijmans, R.J.; Cameron, S.E.; Parra, J.L.; Jones, P.G.; Jarvis, A. Very high resolution interpolated climate surfaces for global land areas. Int. J. Climatol. 2005, 25, 1965–1978. [Google Scholar] [CrossRef]
- Canessa, P.; Schumacher, J.; Hevia, M.A.; Tudzynski, P.; Larrondo, L.F. Assessing the effects of light on differentiation and virulence of the plant pathogen botrytis cinerea: Characterization of the white collar complex. PLoS ONE 2013, 8, e84223. [Google Scholar] [CrossRef]
- Cox, P.M.; Betts, R.A.; Jones, C.D.; Spall, S.A.; Totterdell, I.J. Acceleration of global warming due to carbon-cycle feedbacks in a coupled climate model. Nature 2000, 408, 184–187. [Google Scholar] [CrossRef] [PubMed]
- Hevia, M.A.; Canessa, P.; Müller-Esparza, H.; Larrondo, L.F. A circadian oscillator in the fungus Botrytis cinerea regulates virulence when infecting Arabidopsis thaliana. Proc. Natl. Acad. Sci. USA 2015, 112, 8744–8749. [Google Scholar] [CrossRef] [PubMed]
- König, J.; Perovic, D.; Kopahnke, D.; Ordon, F. Mapping seedling resistance to net form of net blotch (Pyrenophora teres f. teres) in barley using detached leaf assay. Plant Breed. 2014, 133, 356–365. [Google Scholar]
- Williams, K.J.; Smyl, C.; Lichon, A.; Wong, K.Y.; Wallwork, H. Development and use of an assay based on the polymerase chain reaction that differentiates the pathogens causing spot form and net form of net blotch of barley. Australas. Plant Pathol. 2001, 30, 37–44. [Google Scholar] [CrossRef]
- Möller, E.M.; Bahnweg, G.; Sandermann, H.; Geiger, H.H. A simple and efficient protocol for isolation of high molecular weight DNA from filamentous fungi, fruit bodies, and infected plant tissues. Nucleic Acids Res. 1992, 20, 6115–6116. [Google Scholar] [CrossRef] [PubMed]
- Katoh, K.; Misawa, K.; Kuma, K.; Miyata, T. MAFFT: A novel method for rapid multiple sequence alignment based on fast Fourier transform. Nucleic Acids Res. 2002, 30, 3059–3066. [Google Scholar] [CrossRef] [PubMed]
- Rambaut, A. FigTree, A Graphical Viewer of Phylogenetic Trees. (Ver 1.4.3). 2009. Available online: http://tree.bio.ed.ac.uk/software/figtree/ (accessed on 4 October 2016).
- Baldwin, B.G.; Sanderson, M.J.; Porter, J.M.; Wojciechowski, M.F.; Campbell, C.S.; Donoghue, M.J. Erratum: The ITS region of nuclear ribosomal DNAZ: A valuable source of evidence on angiosperm phylogeny. Ann. Mo. Bot. Gard. 1996, 83, 151. [Google Scholar] [CrossRef]
- Schneider, C.A.; Rasband, W.S.; Eliceiri, K.W. NIH Image to ImageJ: 25 years of image analysis. Nat. Methods 2012, 9, 671–675. [Google Scholar] [CrossRef]
| Isolate Name 1 | Collection Site | Host Specie | MAT Idiomorph | Forma | Phylogenetic Group |
|---|---|---|---|---|---|
| H.S-MA-RI | Rishon | H. spontaneum | 1-1-1 | Ptm | 4 |
| H.V-TE-NRM | Ramot Menashe | H. vulgare | 1-1-1 | Ptt | 2 |
| H.V-TE-ERM | Ramot Menashe | H. vulgare | 1-1-1 | Ptt | 2 |
| H.V-TE-BE 2 | Beeri | H. vulgare | 1-1-1 | Ptt | - |
| H.V-MA-BD2 | Beit Dagan | H. vulgare | 1-1-1 | Ptm | 4 |
| H.S-TE-SH | Shtulim | H. spontaneum | 1-1-1 | Ptt | 2 |
| H.S-MA-NM | Neve Michae | H. spontaneum | 1-1-1 | Ptm | - |
| H.S-TE-NH | Netiv Halamed He | H. spontaneum | 1-1-1 | Ptt | 3 |
| H.S-TE-MO | Modi'in | H. spontaneum | 1-1-1 | Ptt | 2 |
| H.S-TE-MI | Mitzpe Ilan | H. spontaneum | 1-1-1 | Ptt | 1 |
| H.S-MA-ME | Meytzar | H. spontaneum | 1-1-1 | Ptm | 4 |
| H.S-MA-KI3 | Kisufim | H. spontaneum | 1-1-1 | Ptm | 3 |
| H.S-TE-KA | Katzir | H. spontaneum | 1-1-1 | Ptt | - |
| H.S-TE-KT | Kiryat Tivon | H. spontaneum | 1-1-1 | Ptt | - |
| H.S-TE-HAR3 | Haruvit | H. spontaneum | 1-1-1 | Ptt | 3 |
| H.S-MA-GIM | Gimzue | H. spontaneum | 1-1-1 | Ptm | - |
| H.G-MA-GIL | Gilat | H. glaucum | 1-1-1 | Ptm | 4 |
| H.G-TE-EH | El Hahar | H. glaucum | 1-1-1 | Ptt | - |
| H.S-TE-AH | Aloney habashan | H. spontaneum | 1-2-1 | Ptt | 3 |
| H.V-MA-KM 3 | Kefar Masarik | H. vulgare | 1-2-1 | Ptm | 1 |
| H.V-TE-GA | Gat | H. vulgare | 1-2-1 | Ptt | 3 |
| H.V-TE-BD1 | Beit Dagan | H. vulgare | 1-2-1 | Ptt | 1 |
| H.S-MA-RM | Ramot Menashe | H. spontaneum | 1-2-1 | Ptm | 2 |
| H.S-MA-NR | Nahal Raz | H. spontaneum | 1-2-1 | Ptm | 2 |
| H.S-TE-HA | Harish | H. spontaneum | 1-2-1 | Ptt | 2 |
| H.S-TE-AL | Alumim | H. spontaneum | 1-2-1 | Ptt | 1 |
| H.S-MA-GY | Gan Yavne | H. spontaneum | n/a | Ptm | n/a |
| H.S-MA-TI 3 | Timrat | H. spontaneum | n/a | Ptm | n/a |
| No. of Isolates | CV | Mean4 | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Nec. Growth Rate 1 | Sap. Growth Rate 2 | Nec. / Sap. 3 | Nec. Growth Rate | Sap. Growth Rate | Nec. / Sap. | ||||
| Category | forma | Ptm | 12 | 15.09 | 34.76 | 26. 53 | 0.22 A,* | 0.55 A,* | 0.39 A,* |
| Ptt | 15 | 34.13 | 34.48 | 42.41 | 0.13 B | 0.45 B | 0.33 B | ||
| Host | Domesticated | 6 | 12.92 A,** | 26.93 | 37.77 | 0.15 | 0.48 | 0.36 | |
| Wild | 21 | 45.95 B | 28.28 | 42.09 | 0.17 | 0.50 | 0.35 | ||
| Variety, medium | |||||
|---|---|---|---|---|---|
| Variety, medium1 | Noga'' | 'Ma'anit' | 'Sagiv' | 'Barke' | |
| ‘Noga’ | |||||
| 'Ma'anit' | 0.682* | ||||
| 'Sagiv' | 0.684* | 0.761* | |||
| 'Barke' | 0.760* | 0.763* | 0.740* | ||
| 'Czapek' | 0.424* | 0.448* | 0.405* | 0.507* | |
| Variety | CV | Average 1 | N |
|---|---|---|---|
| 'Barke' | 47.989 | 0.211A | 105 |
| 'Sagiv' | 55.479 | 0.18AB | 105 |
| 'Ma'anit' | 61.267 | 0.15AB | 105 |
| ‘Noga’ | 55.146 | 0.145B | 108 |
| Primer Name | Sequence (5′→3′) | Amplicon Size (In Base Pairs) |
|---|---|---|
| ptt-MAT1F1 | TGGAAGGATCGCAGACTGGAA | 1933-2161 |
| ptt-MAT1R1 | TTCGTCGCGGAGGAGGCTTGT | |
| ptt-MAT1-1F4952 | GGCAACAAGAGGTGAAGGTG | 1273 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ronen, M.; Sela, H.; Fridman, E.; Perl-Treves, R.; Kopahnke, D.; Moreau, A.; Ben-David, R.; Harel, A. Characterization of the Barley Net Blotch Pathosystem at the Center of Origin of Host and Pathogen. Pathogens 2019, 8, 275. https://doi.org/10.3390/pathogens8040275
Ronen M, Sela H, Fridman E, Perl-Treves R, Kopahnke D, Moreau A, Ben-David R, Harel A. Characterization of the Barley Net Blotch Pathosystem at the Center of Origin of Host and Pathogen. Pathogens. 2019; 8(4):275. https://doi.org/10.3390/pathogens8040275
Chicago/Turabian StyleRonen, Moshe, Hanan Sela, Eyal Fridman, Rafael Perl-Treves, Doris Kopahnke, Alexandre Moreau, Roi Ben-David, and Arye Harel. 2019. "Characterization of the Barley Net Blotch Pathosystem at the Center of Origin of Host and Pathogen" Pathogens 8, no. 4: 275. https://doi.org/10.3390/pathogens8040275
APA StyleRonen, M., Sela, H., Fridman, E., Perl-Treves, R., Kopahnke, D., Moreau, A., Ben-David, R., & Harel, A. (2019). Characterization of the Barley Net Blotch Pathosystem at the Center of Origin of Host and Pathogen. Pathogens, 8(4), 275. https://doi.org/10.3390/pathogens8040275






