Thoracic UltrasONOgraphy Reporting: The TUONO Consensus
Abstract
1. Introduction
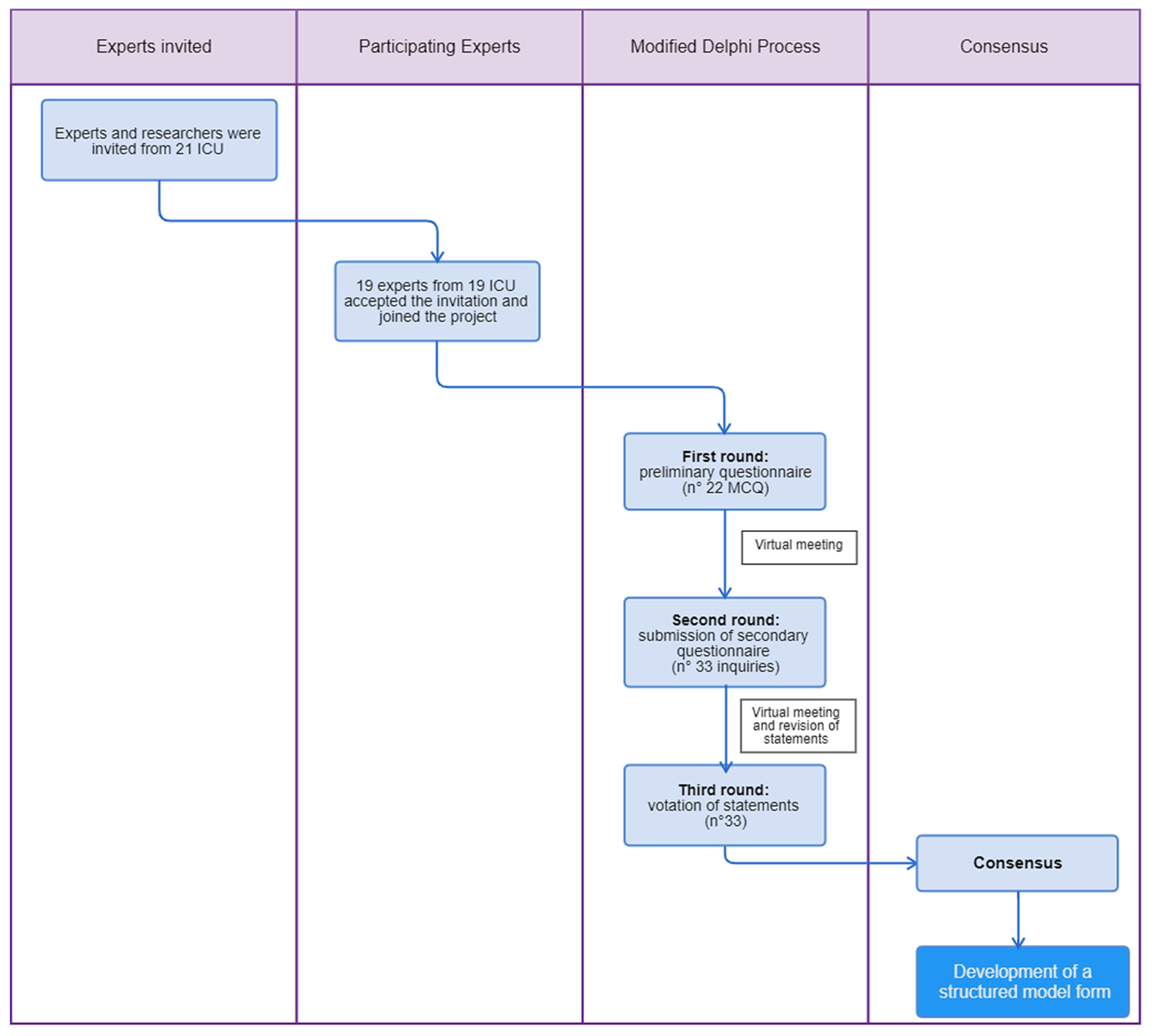
2. Materials and Methods
2.1. 1st Delphi Round
2.2. 1st Virtual Meeting and 2nd Delphi Round
2.3. 2nd Virtual Meeting and 3rd Delphi Round
3. Results
- n° 26: it is useful to report if the examination is performed in the emergency setting.
- n° 29: it is useful to report the type of probe.
4. Discussion
4.1. Statements n° 1–5: Systematic Examination of Lung Zones and Localization of Findings
4.2. Statements n° 6–8: Examination of the Pleura
4.3. Statements n° 9–12: LUS Signs
4.4. Statements n° 13–14: Quantification of Findings
4.5. Statements n° 15–18: Pleural Effusion
4.6. Statement n° 19–20: Consolidations and LUS Score
4.7. Statements n° 21–23: Conclusions
4.8. Statements n° 24–29: Context of Performing LUS
4.9. Statements n° 30–33: Ventilatory Setting and Therapeutic Consideration
4.10. Proposal for a Structured Model form for LUS
Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Lichtenstein, D.A. Lung Ultrasound in the Critically Ill. Ann. Intensive Care 2014, 4, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Laursen, C.B.; Sloth, E.; Lassen, A.; Christensen, R.D.; Lambrechtsen, J.; Madsen, P.H.; Henriksen, D.P.; Davidsen, J.R.; Rasmussen, F. Point-of-care ultrasonography in patients admitted with respiratory symptoms: A single-blind, randomised controlled trial. Lancet Respir. Med. 2014, 2, 638–646. [Google Scholar] [CrossRef] [PubMed]
- Davenport, M.S.; Rcc, F.T.S.D.-F.P.O.; Hu, E.M.; Zhang, A.; Shinagare, A.B.; Smith, A.D.; Pedrosa, I.; Kaffenberger, S.D.; Silverman, S.G. Standardized report template for indeterminate renal masses at CT and MRI: A collaborative product of the SAR Disease-Focused Panel on Renal Cell Carcinoma. Abdom. Imaging 2018, 44, 1423–1429. [Google Scholar] [CrossRef] [PubMed]
- Demi, L.; Wolfram, F.; Klersy, C.; De Silvestri, A.; Ferretti, V.V.; Muller, M.; Miller, D.; Feletti, F.; Wełnicki, M.; Buda, N.; et al. New International Guidelines and Consensus on the Use of Lung Ultrasound. J. Ultrasound Med. 2022, 42, 309–344. [Google Scholar] [CrossRef]
- Calamai, I.; Greco, M.; Finazzi, S.; Savi, M.; Vitiello, G.; Garbero, E.; Spina, R.; Montisci, A.; Mongodi, S.; Bertolini, G. Thoracic UltrasONOgraphy Reporting: The TUONO Study. J. Clin. Med. 2022, 11, 7126. [Google Scholar] [CrossRef]
- Boney, O.; Moonesinghe, S.R.; Myles, P.S.; Grocott, M.P.; Bartoszko, J.; Beattie, W.; Bellomo, R.; Buggy, D.; Cabrini, L.; Canet, J.; et al. Core Outcome Measures for Perioperative and Anaesthetic Care (COMPAC): A modified Delphi process to develop a core outcome set for trials in perioperative care and anaesthesia. Br. J. Anaesth. 2021, 128, 174–185. [Google Scholar] [CrossRef]
- Mongodi, S.; Santangelo, E.; De Luca, D.; Rovida, S.; Corradi, F.; Volpicelli, G.; Gargani, L.; Bouhemad, B.; Mojoli, F. Quantitative Lung Ultrasound: Time for a Consensus? Chest 2020, 158, 469–470. [Google Scholar] [CrossRef] [PubMed]
- Calamai, I.; Greco, M.; Bertolini, G.; Spina, R.; Italian Group for the Evaluation of Interventions in Intensive Care Medicine (GiViTI). Current adoption of lung ultrasound in Intensive Care Units: An Italian multi-center survey. Minerva Anestesiol. 2017, 83, 720–727. [Google Scholar] [CrossRef] [PubMed]
- Lichtenstein, D.A. BLUE-protocol and FALLS-protocol: Two applications of lung ultrasound in the critically ill. Chest 2015, 147, 1659–1670. [Google Scholar] [CrossRef] [PubMed]
- Gargani, L.; Volpicelli, G. How I do it: Lung ultrasound. Cardiovasc. Ultrasound 2014, 12, 25. [Google Scholar] [CrossRef]
- Mongodi, S.; De Luca, D.; Colombo, A.; Stella, A.; Santangelo, E.; Corradi, F.; Gargani, L.; Rovida, S.; Volpicelli, G.; Bouhemad, B.; et al. Quantitative Lung Ultrasound: Technical Aspects and Clinical Applications. Anesthesiology 2021, 134, 949–965. [Google Scholar] [CrossRef] [PubMed]
- Jambrik, Z.; Monti, S.; Coppola, V.; Agricola, E.; Mottola, G.; Miniati, M.; Picano, E. Usefulness of ultrasound lung comets as a nonradiologic sign of extravascular lung water. Am. J. Cardiol. 2004, 93, 1265–1270. [Google Scholar] [CrossRef] [PubMed]
- Gargani, L. Lung ultrasound: A new tool for the cardiologist. Cardiovasc. Ultrasound 2011, 9, 6. [Google Scholar] [CrossRef] [PubMed]
- Via, G.; Storti, E.; Gulati, G.; Neri, L.; Mojoli, F.; Braschi, A. Lung ultrasound in the ICU: From diagnostic instrument to respiratory monitoring tool. Minerva Anestesiol. 2012, 78, 1282–1296. [Google Scholar] [PubMed]
- Lichtenstein, D.A.; Mezière, G.A. Relevance of lung ultrasound in the diagnosis of acute respiratory failure the BLUE protocol. Chest 2008, 134, 117–125. [Google Scholar] [CrossRef] [PubMed]
- Tung-Chen, Y.; Ossaba-Vélez, S.; Velásquez, K.S.A.; Parra-Gordo, M.L.; Díez-Tascón, A.; Villén-Villegas, T.; Montero-Hernández, E.; Gutiérrez-Villanueva, A.; Trueba-Vicente; Arenas-Berenguer, I.; et al. The Impact of Different Lung Ultrasound Protocols in the Assessment of Lung Lesions in COVID-19 Patients: Is There an Ideal Lung Ultrasound Protocol? Ángela Trueba-Vicente 4 · Isabel Are-nas-Berenguer 5 · Milagros Martí de Gracia 2. J. Ultrasound 2021, 25, 483–491. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Qin, A.; Yang, X.; Zhou, S.; Luo, Y.; Zhu, F.; Hu, B.; Li, J.; Cai, S.; Peng, Z. Findings and Prognostic Value of Lung Ultrasonography in Coronal Virus Disease 2019 (COVID-19) Pneumonia. Shock 2020, 56, 200–205. [Google Scholar] [CrossRef]
- Biswas, A.; Lascano, J.E.; Mehta, H.J.; Faruqi, I. The utility of the “shred sign” in the diagnosis of acute respir-atory distress syndrome resulting from multifocal pneumonia. Am. J. Respir. Crit. Care Med. 2017, 195, e20–e22. [Google Scholar] [CrossRef]
- Dulchavsky, S.A.; Hamilton, D.R.; Diebel, L.N.; Sargsyan, A.E.; Billica, R.D.; Williams, D.R. Thoracic Ultrasound Diagnosis of Pneumothorax. J. Trauma Inj. Infect. Crit. Care 1999, 47, 970–971. [Google Scholar] [CrossRef]
- Lichtenstein, D.A.; Menu, Y. A bedside ultrasound sign ruling out pneumothorax in the critically ill. Lung sliding. Chest 1995, 108, 1345–1348. [Google Scholar] [CrossRef]
- Soummer, A.; Perbet, S.; Brisson, H.; Arbelot, C.; Constantin, J.-M.; Lu, Q.; Rouby, J.-J. Ultrasound assessment of lung aeration loss during a successful weaning trial predicts postextubation distress*. Crit. Care Med. 2012, 40, 2064–2072. [Google Scholar] [CrossRef]
- Mongodi, S.; Bouhemad, B.; Orlando, A.; Stella, A.; Tavazzi, G.; Via, G.; Iotti, G.A.; Braschi, A.; Mojoli, F. Modified Lung Ultrasound Score for Assessing and Monitoring Pulmonary Aeration. Ultraschall Med.-Eur. J. Ultrasound 2017, 38, 530–537. [Google Scholar] [CrossRef] [PubMed]
- Robba, C.; Wong, A.; Poole, D.; Al Tayar, A.; Arntfield, R.T.; Chew, M.S.; Corradi, F.; Douflé, G.; Goffi, A.; Lamperti, M.; et al. Basic ultrasound head-to-toe skills for intensivists in the general and neuro intensive care unit population: Consensus and expert recommendations of the European Society of Intensive Care Medicine. Intensive Care Med. 2021, 47, 1347–1367. [Google Scholar] [CrossRef] [PubMed]
- Chiumello, D.; Mongodi, S.; Algieri, I.; Vergani, G.L.; Orlando, A.; Via, G.; Crimella, F.; Cressoni, M.; Mojoli, F. Assessment of Lung Aeration and Recruitment by CT Scan and Ultrasound in Acute Respiratory Distress Syndrome Patients*. Crit. Care Med. 2018, 46, 1761–1768. [Google Scholar] [CrossRef] [PubMed]
- Mongodi, S.; De Vita, N.; Salve, G.; Bonaiti, S.; Daverio, F.; Cavagnino, M.; Siano, G.; Amatu, A.; Maggio, G.; Musella, V.; et al. The Role of Lung Ultrasound Monitoring in Early Detection of Ventilator-Associated Pneumonia in COVID-19 Patients: A Retrospective Observational Study. J. Clin. Med. 2022, 11, 3001. [Google Scholar] [CrossRef] [PubMed]
- Rouby, J.-J.; Arbelot, C.; Gao, Y.; Zhang, M.; Lv, J.; Wang, C.; Chunyao, W.; Bin, D.; Barbas, C.S.V.; Neto, F.L.D.; et al. Training for Lung Ultrasound Score Measurement in Critically Ill Patients. Am. J. Respir. Crit. Care Med. 2018, 198, 398–401. [Google Scholar] [CrossRef]
- Smit, M.R.; de Vos, J.; Pisani, L.; Hagens, L.A.; Almondo, C.; Heijnen, N.F.; Schnabel, R.M.; van der Horst, I.C.; Bergmans, D.C.; Schultz, M.J.; et al. Comparison of Linear and Sector Array Probe for Handheld Lung Ultrasound in Invasively Ventilated ICU Patients. Ultrasound Med. Biol. 2020, 46, 3249–3256. [Google Scholar] [CrossRef]
- Volpicelli, G.; Elbarbary, M.; Blaivas, M.; Lichtenstein, D.A.; Mathis, G.; Kirkpatrick, A.W.; Melniker, L.; Gargani, L.; Noble, V.E.; Via, G.; et al. International evidence-based recommendations for point-of-care lung ultrasound conference reports and expert panel. Intensive Care Med. 2012, 38, 577–591. [Google Scholar] [CrossRef]
- Bouhemad, B.; Brisson, H.; Le-Guen, M.; Arbelot, C.; Lu, Q.; Rouby, J.-J. Bedside Ultrasound Assessment of Positive End-Expiratory Pressure–induced Lung Recruitment. Am. J. Respir. Crit. Care Med. 2011, 183, 341–347. [Google Scholar] [CrossRef]
- Xirouchaki, N.; Kondili, E.; Prinianakis, G.; Malliotakis, P.; Georgopoulos, D. Impact of lung ultrasound on clinical decision making in critically ill patients. Intensive Care Med. 2013, 40, 57–65. [Google Scholar] [CrossRef] [PubMed]
- Mongodi, S.; Via, G.; Girard, M.; Rouquette, I.; Misset, B.; Braschi, A.; Mojoli, F.; Bouhemad, B. Lung Ultrasound for Early Diagnosis of Ventilator-Associated Pneumonia. Chest 2016, 149, 969–980. [Google Scholar] [CrossRef] [PubMed]
- Vetrugno, L.; Mojoli, F.; Boero, E.; Berchialla, P.; Bignami, E.G.; Orso, D.; Cortegiani, A.; Forfori, F.; Corradi, F.; Cammarota, G.; et al. Level of Diffusion and Training of Lung Ultrasound during the COVID-19 Pandemic—A National Online Italian Survey (ITALUS) from the Lung Ultrasound Working Group of the Italian Society of Anesthesia, Analgesia, Resuscitation, and Intensive Care (SIAARTI). Ultraschall Med.-Eur. J. Ultrasound 2021, 43, 464–472. [Google Scholar] [CrossRef] [PubMed]
- Lopes, A.J.; Mafort, T.T.; da Costa, C.H.; Rufino, R.; Firmida, M.C.; Kirk, K.M.; Cobo, C.G.; Costa, H.D.S.B.D.; da Cruz, C.M.B.Q.; Mogami, R. Comparison Between Lung Ultrasound and Computed Tomographic Findings in Patients with COVID-19 Pneumonia. J. Ultrasound Med. 2021, 40, 1391–1399. [Google Scholar] [CrossRef] [PubMed]
| Experts’ Consensus Statements | Agree (%) | Disagree (%) | Neutral (%) |
|---|---|---|---|
| Systematic examination of lung zones | |||
| 100 | 0 | 0 |
| 86.4 | 13.6 | 0 |
| 92.9 | 7.1 | 0 |
| 81.0 | 19 | 0 |
| 92.9 | 7.1 | 0 |
| Examination of the pleura | |||
| 81.0 | 19 | 0 |
| 100 | 0 | 0 |
| 100 | 0 | 0 |
| LUS signs | |||
| 100 | 0 | 0 |
| 100 | 0 | 0 |
| 85.7 | 7.1 | 7.1 |
| 92.9 | 7.1 | 0 |
| 86.4 | 13.6 | 0 |
| 92.9 | 7.1 | 0 |
| Pleural effusion | |||
| 100 | 0 | 0 |
| 100 | 0 | 0 |
| 100 | 0 | 0 |
| 100 | 0 | 0 |
| Consolidations | |||
| 100 | 0 | 0 |
| LUS score | |||
| 92.9 | 7.1 | 0 |
| Conclusions | |||
| 81.8 | 18.2 | 0 |
| 100 | 0 | 0 |
| 100 | 0 | 0 |
| Context of performing a LUS | |||
| 92.9 | 7.1 | 0 |
| 92.9 | 7.1 | 0 |
| 63.2 | 15.8 | 21.1 |
| 95.5 | 4.5 | 0 |
| 90.9 | 8.1 | 0 |
| 70 | 30 | 0 |
| Ventilatory settings | |||
| 91.0 | 9 | 0 |
| 95.5 | 4.5 | 0 |
| Therapeutic considerations | |||
| 100 | 0 | 0 |
| 100 | 0 | 0 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Calamai, I.; Greco, M.; Savi, M.; Vitiello, G.; Garbero, E.; Spina, R.; Pisani, L.; Mongodi, S.; Finazzi, S.; on behalf of the TUONO Study Investigators. Thoracic UltrasONOgraphy Reporting: The TUONO Consensus. Diagnostics 2023, 13, 1535. https://doi.org/10.3390/diagnostics13091535
Calamai I, Greco M, Savi M, Vitiello G, Garbero E, Spina R, Pisani L, Mongodi S, Finazzi S, on behalf of the TUONO Study Investigators. Thoracic UltrasONOgraphy Reporting: The TUONO Consensus. Diagnostics. 2023; 13(9):1535. https://doi.org/10.3390/diagnostics13091535
Chicago/Turabian StyleCalamai, Italo, Massimiliano Greco, Marzia Savi, Gaia Vitiello, Elena Garbero, Rosario Spina, Luigi Pisani, Silvia Mongodi, Stefano Finazzi, and on behalf of the TUONO Study Investigators. 2023. "Thoracic UltrasONOgraphy Reporting: The TUONO Consensus" Diagnostics 13, no. 9: 1535. https://doi.org/10.3390/diagnostics13091535
APA StyleCalamai, I., Greco, M., Savi, M., Vitiello, G., Garbero, E., Spina, R., Pisani, L., Mongodi, S., Finazzi, S., & on behalf of the TUONO Study Investigators. (2023). Thoracic UltrasONOgraphy Reporting: The TUONO Consensus. Diagnostics, 13(9), 1535. https://doi.org/10.3390/diagnostics13091535






