Effects of Temperature and Glyphosate on Fatty Acid Composition, Antioxidant Capacity, and Lipid Peroxidation in the Gastropod Lymneae sp.
Abstract
:1. Introduction
2. Materials and Methods
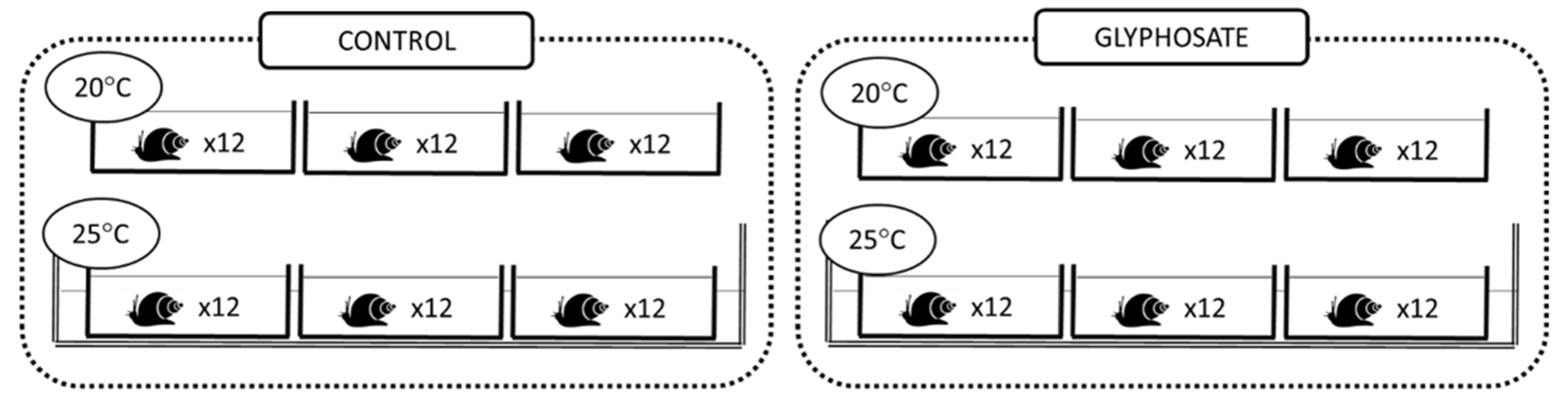
2.1. Experimental Design
2.2. Lipid Extraction and Fatty Acid Analysis
2.3. Enzyme Assays
2.4. Protein Content
2.5. Lipid Peroxidation
2.6. Statistical Analysis
3. Results and Discussion
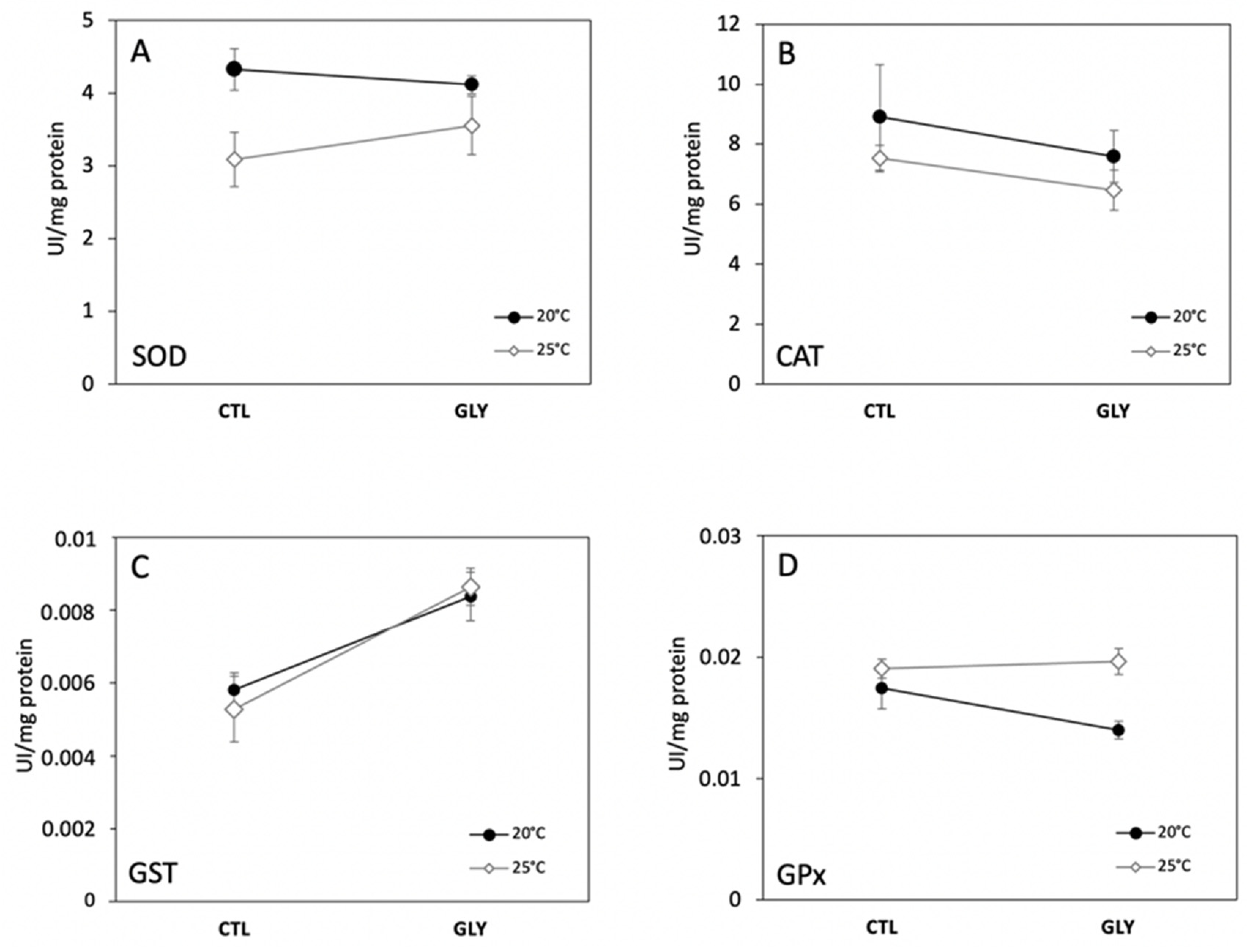
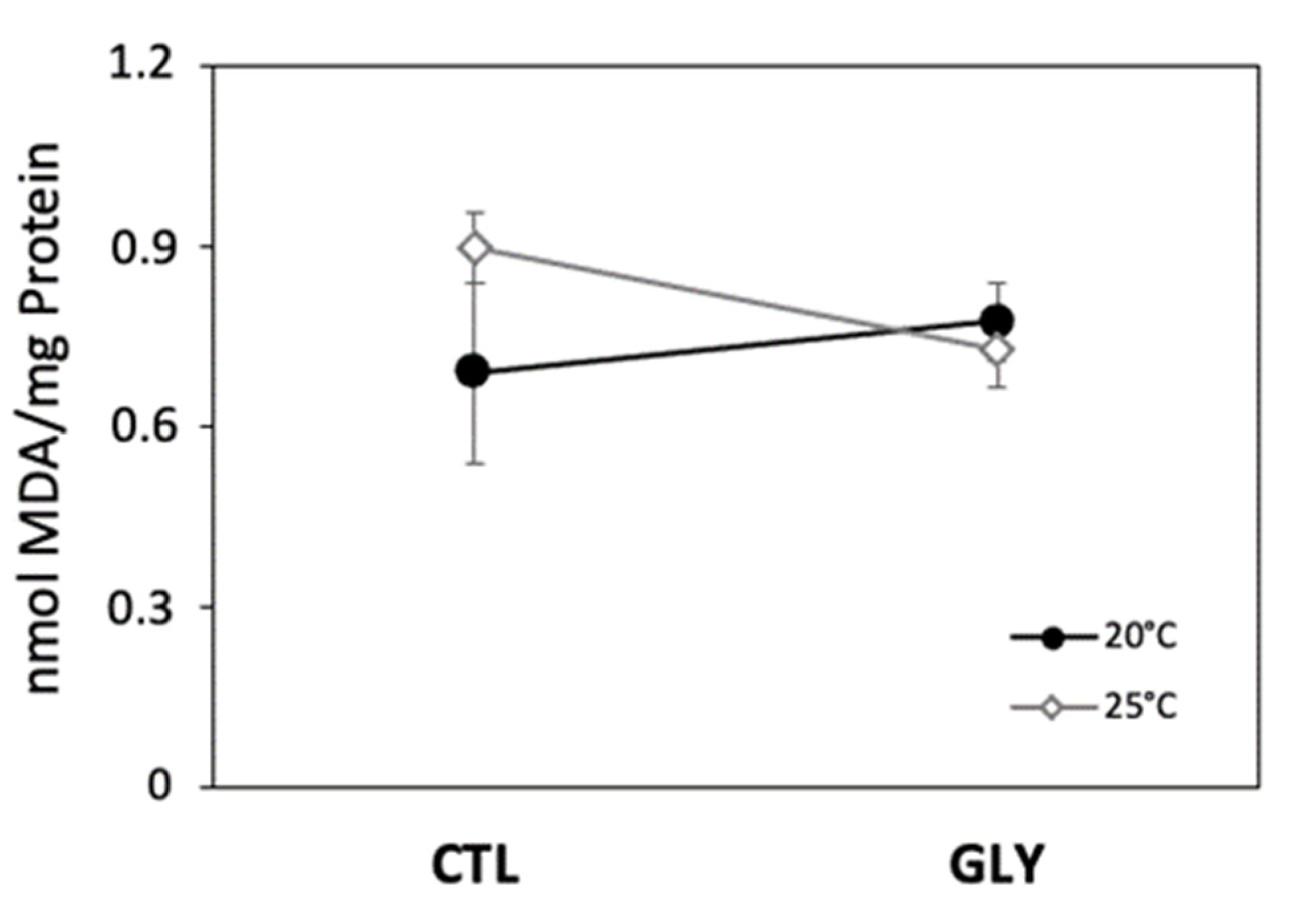
3.1. Effects of Temperature and Glyphosate on Snail Enzyme Activity and Lipid Peroxidation
3.1.1. Temperature Effect on Antioxidant Enzymes and Lipid Peroxidation
3.1.2. Glyphosate Effect on Antioxidant Enzymes and Lipid Peroxidation
3.1.3. Interaction Effects
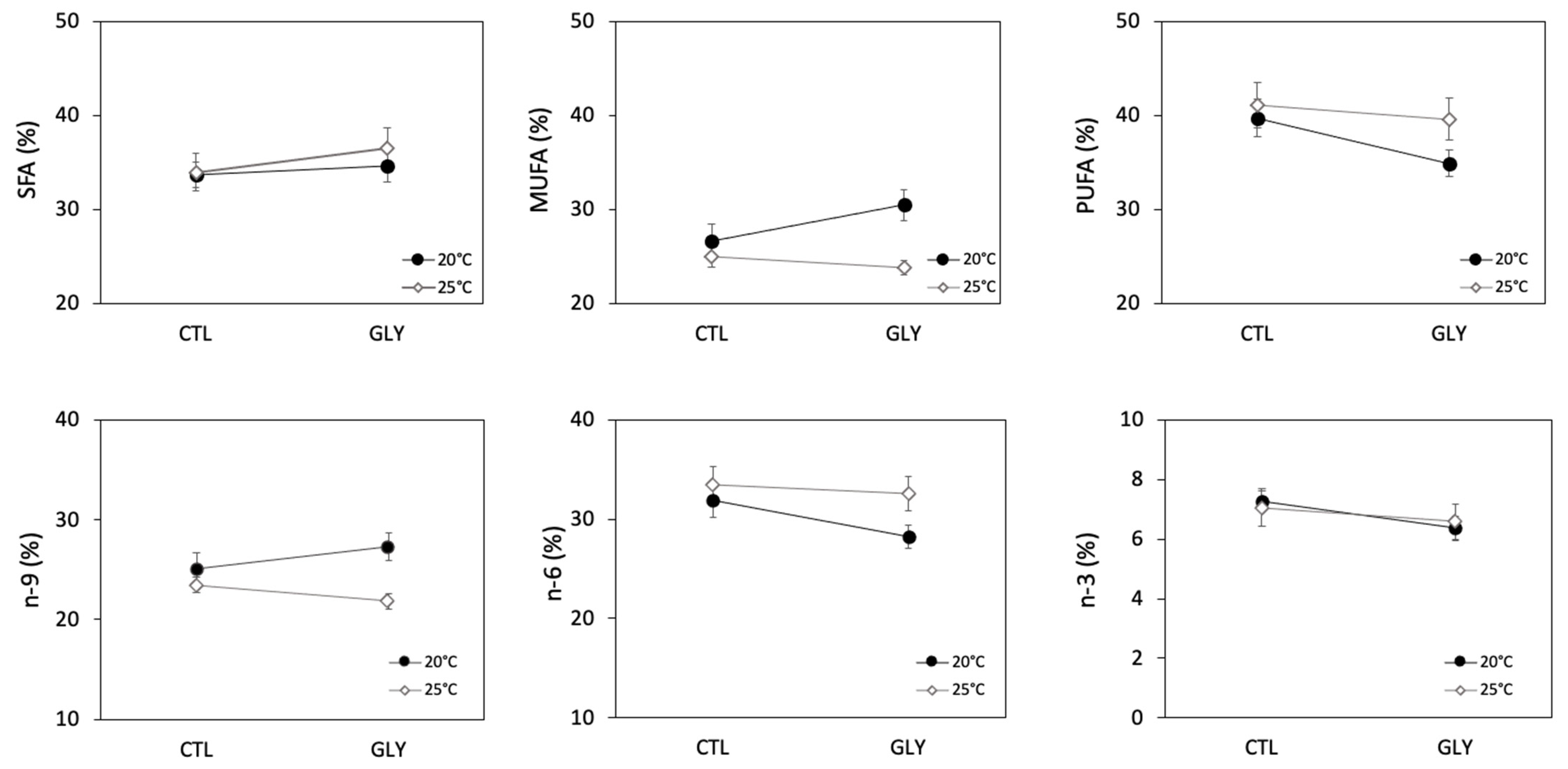
3.2. Snails Fatty Acid Composition as a Response to Glyphosate and Temperature
3.2.1. Temperature Effect on Snails Fatty Acid Composition
3.2.2. Glyphosate Effect on Snails Fatty Acid Composition
3.2.3. Interaction Effects
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
Appendix A
| Fatty Acid | Mean ± SEM |
|---|---|
| 16:0 | 27.88 ± 0.30 |
| 17:0 | 0.60 ± 0.03 |
| 18:0 | 1.52 ± 0.96 |
| 16:1n7 | 0.51 ± 0.23 |
| 18:1n9 | 35.76 ± 1.26 |
| 20:1n9 | 1.63 ± 0.03 |
| 22:1n9 | 1.09 ± 0.05 |
| 18:2n6 | 21.63 ± 0.24 |
| 18:3n3 | 0.96 ± 0.02 |
| 20:5n3 | 3.48 ± 0.15 |
| 22:6n3 | 2.64 ± 0.03 |
| Others | 2.37 ± 0.08 |
References
- Battaglin, W.A.; Kolpin, D.W.; Scribner, E.A.; Kuivila, K.M.; Sandstrom, M.W. Glyphosate, other herbicides, and transformation products in midwestern streams, 20021. JAWRA J. Am. Water Resour. Assoc. 2005, 41, 323–332. [Google Scholar] [CrossRef] [Green Version]
- Blanchoud, H.; Moreau-Guigon, E.; Farrugia, F.; Chevreuil, M.; Mouchel, J. Contribution by urban and agricultural pesticide uses to water contamination at the scale of the Marne watershed. Sci. Total Environ. 2007, 375, 168–179. [Google Scholar] [CrossRef] [PubMed]
- Pline, W.A.; Price, A.J.; Wilcut, J.W.; Edmisten, K.L.; Wells, R. Absorption and translocation of glyphosate in glyphosate-resistant cotton as influenced by application method and growth stage. Weed Sci. 2001, 49, 460–467. [Google Scholar] [CrossRef]
- Struger, J.; Thompson, D.; Staznik, B.; Martin, P.; McDaniel, T.; Marvin, C. Occurrence of Glyphosate in Surface Waters of Southern Ontario. Bull. Environ. Contam. Toxicol. 2008, 80, 378–384. [Google Scholar] [CrossRef]
- Peruzzo, P.J.; Porta, A.A.; Ronco, A.E. Levels of glyphosate in surface waters, sediments and soils associated with direct sowing soybean cultivation in north pampasic region of Argentina. Environ. Pollut. 2008, 156, 61–66. [Google Scholar] [CrossRef]
- Coupe, R.H.; Kalkhoff, S.J.; Capel, P.D.; Gregoire, C. Fate and transport of glyphosate and aminomethylphosphonic acid in surface waters of agricultural basins. Pest Manag. Sci. 2011, 68, 16–30. [Google Scholar] [CrossRef]
- Giesy, J.P.; Dobson, S.; Solomon, K.R. Ecotoxicological risk assessment for Roundup (R) Herbicide. Rev. Environ. Contam. Toxicol. 2000, 167, 35–120. [Google Scholar]
- Jiraungkoorskul, W.; Upatham, E.S.; Kruatrachue, M.; Sahaphong, S.; Vichasri-Grams, S.; Pokethitiyook, P. Biochemical and histopathological effects of glyphosate herbicide on Nile tilapia (Oreochromis niloticus). Environ. Toxicol. 2003, 18, 260–267. [Google Scholar] [CrossRef]
- Glusczak, L.; Miron, D.D.S.; Crestani, M.; da Fonseca, M.B.; Pedron, F.D.A.; Duarte, M.F.; Vieira, V.L.P. Effect of glyphosate herbicide on acetylcholinesterase activity and metabolic and hematological parameters in piava (Leporinus obtusidens). Ecotoxicol. Environ. Saf. 2006, 65, 237–241. [Google Scholar] [CrossRef]
- Modesto, K.A.; Martinez, C.B.R. Roundup (R) causes oxidative stress in liver and inhibits acetylcholinesterase in muscle and brain of the fish Prochilodus lineatus. Chemosphere 2010, 78, 294–299. [Google Scholar] [CrossRef]
- Çavaş, T.; Könen, S. Detection of cytogenetic and DNA damage in peripheral erythrocytes of goldfish (Carassius auratus) exposed to a glyphosate formulation using the micronucleus test and the comet assay. Mutagenesis 2007, 22, 263–268. [Google Scholar] [CrossRef] [Green Version]
- Gahl, M.K.; Pauli, B.D.; Houlahan, J.E. Effects of chytrid fungus and a glyphosate-based herbicide on survival and growth of wood frogs (Lithobates sylvaticus). Ecol. Appl. 2011, 21, 2521–2529. [Google Scholar] [CrossRef]
- Howe, C.M.; Berrill, M.; Pauli, B.D.; Helbing, C.C.; Werry, K.; Veldhoen, N. Toxicity of glyphosate-based pesticides to four north american frog species. Environ. Toxicol. Chem. 2004, 23, 1928–1938. [Google Scholar] [CrossRef]
- Jones, D.K.; Hammond, J.I.; Relyea, R.A. Roundup (R) and Amphibians: The Importance of Concentration, Application Time, and Stratification. Environ. Toxicol. Chem. 2010, 29, 2016–2025. [Google Scholar] [CrossRef]
- Hanana, H.; Simon, G.; Kervarec, N.; Mohammadou, B.A.; Cerantola, S. HRMAS NMR as a tool to study metabolic responses in heart clam Ruditapes decussatus exposed to Roundup (R). Talanta 2012, 97, 425–431. [Google Scholar] [CrossRef]
- Iummato, M.M.; Sabatini, S.E.; Cacciatore, L.C.; Cochón, A.C.; Cataldo, D.; de Molina, M.D.; Juárez, Á.B. Biochemical responses of the golden mussel Limnoperna fortunei under dietary glyphosate exposure. Ecotoxicol. Environ. Saf. 2018, 163, 69–75. [Google Scholar] [CrossRef]
- Mottier, A.; Séguin, A.; Devos, A.; Le Pabic, C.; Voiseux, C.; Lebel, J.M.; Serpentini, A.; Fiévet, B.; Costil, K. Effects of subchronic exposure to glyphosate in juvenile oysters (Crassostrea gigas): From molecular to individual levels. Mar. Pollut. Bull. 2015, 95, 665–677. [Google Scholar] [CrossRef]
- Hong, Y.; Yang, X.; Huang, Y.; Yan, G.; Cheng, Y. Assessment of the oxidative and genotoxic effects of the glyphosate-based herbicide roundup on the freshwater shrimp, Macrobrachium nipponensis. Chemosphere 2018, 210, 896–906. [Google Scholar] [CrossRef]
- Hong, Y.; Yang, X.; Yan, G.; Huang, Y.; Zuo, F.; Shen, Y.; Ding, Y.; Cheng, Y. Effects of glyphosate on immune responses and haemocyte DNA damage of Chinese mitten crab, Eriocheir sinensis. Fish Shellfish Immunol. 2017, 71, 19–27. [Google Scholar] [CrossRef]
- Von Fumetti, S.; Blaurock, K. Effects of the herbicide Roundup (R) on the metabolic activity of Gammarus fossarum Koch, 1836 (Crustacea; Amphipoda). Ecotoxicology 2018, 27, 1249–1260. [Google Scholar] [CrossRef]
- Sáenz, M.E.; Di Marzio, W.D.; Alberdi, J.L.; Tortorelli, M.D.C. Effects of technical grade and a commercial formulation of glyphosate on algal population growth. Bull. Environ. Contam. Toxicol. 1997, 59, 638–644. [Google Scholar] [CrossRef]
- Wong, P. Effects of 2,4-D, glyphosate and paraquat on growth, photosynthesis and chlorophyll–a synthesis of Scenedesmus quadricauda Berb 614. Chemosphere 2000, 41, 177–182. [Google Scholar] [CrossRef]
- Slaninova, A.; Smutna, M.; Modra, H.; Svobodova, Z. A review: Oxidative stress in fish induced by pesticides. Neuro Endocrinol. Lett. 2009, 30, 2–12. [Google Scholar]
- Halliwell, B.; Gutteridge, J.M. Free Radicals in Biology and Medicine, 4th ed.; Oxford University Press: Oxford, NY, USA, 2007; p. p. xxxvi. [Google Scholar]
- Norris, S.E.; Mitchell, T.W.; Else, P.L.; Hancock, S. Phospholipid Peroxidation: Lack of Effect of Fatty Acid Pairing. Lipids 2012, 47, 451–460. [Google Scholar] [CrossRef]
- Schaur, R.J.; Siems, W.; Bresgen, N.; Eckl, P.M. 4-Hydroxy-nonenal—A Bioactive Lipid Peroxidation Product. Biomolecules 2015, 5, 2247–2337. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Glusczak, L.; Miron, D.D.S.; Moraes, B.S.; Simões, R.R.; Schetinger, M.R.C.; Morsch, V.M.; Loro, V.L. Acute effects of glyphosate herbicide on metabolic and enzymatic parameters of silver catfish (Rhamdia quelen). Comp. Biochem. Physiol. Part C Toxicol. Pharmacol. 2007, 146, 519–524. [Google Scholar] [CrossRef]
- Lushchak, O.V.; Kubrak, O.I.; Storey, J.M.; Storey, K.B.; Lushchak, V.I. Low toxic herbicide Roundup induces mild oxidative stress in goldfish tissues. Chemosphere 2009, 76, 932–937. [Google Scholar] [CrossRef]
- Iummato, M.M.; Di Fiori, E.; Sabatini, S.E.; Cacciatore, L.C.; Cochón, A.C.; de Molina, M.D.; Juárez, Á.B. Evaluation of biochemical markers in the golden mussel Limnoperna fortunei exposed to glyphosate acid in outdoor microcosms. Ecotoxicol. Environ. Saf. 2013, 95, 123–129. [Google Scholar] [CrossRef]
- Matozzo, V.; Munari, M.; Masiero, L.; Finos, L.; Marin, M.G. Ecotoxicological hazard of a mixture of glyphosate and aminomethylphosphonic acid to the mussel Mytilus galloprovincialis (Lamarck 1819). Sci. Rep. 2019, 9, 1–9. [Google Scholar] [CrossRef]
- Guo, F.; Bunn, S.E.; Brett, M.T.; Kainz, M.J. Polyunsaturated fatty acids in stream food webs high dissimilarity among producers and consumers. Freshw. Biol. 2017, 62, 1325–1334. [Google Scholar] [CrossRef] [Green Version]
- Arts, M.T.; Ackman, R.G.; Holub, B.J. “Essential fatty acids” in aquatic ecosystems: A crucial link between diet and human health and evolution. Can. J. Fish Aquat. Sci. 2001, 58, 122–137. [Google Scholar] [CrossRef]
- Tocher, D.R. Metabolism and Functions of Lipids and Fatty Acids in Teleost Fish. Rev. Fish. Sci. 2003, 11, 107–184. [Google Scholar] [CrossRef]
- Brett, M.T.; Muller-Navarra, D.C. The role of highly unsaturated fatty acids in aquatic food web processes. Freshw. Biol. 1997, 38, 483–499. [Google Scholar] [CrossRef]
- Lushchak, V.I. Environmentally induced oxidative stress in aquatic animals. Aquat. Toxicol. 2011, 101, 13–30. [Google Scholar] [CrossRef]
- Holman, R. Progress in Chemistry of Fats and Other Lipids; Lundberg, W.O., Malkin, T., Eds.; Pergamon Press Ltd.: London, UK, 1957. [Google Scholar]
- Bayir, M.; Sirkeciogl, A.N.; Bayir, A.; Aras, M. Alterations in Fatty Acids of Polar Lipids in Salmo trutta on Long-term Exposure to a Glyphosate-Based Herbicide (Roundup®). Pak. J. Biol. Sci. 2013, 16, 1194–1198. [Google Scholar] [CrossRef] [Green Version]
- De Carvalho, R.C.; Feijão, E.; Matos, A.R.; Cabrita, M.T.; Novais, S.C.; Lemos, M.F.L.; Caçador, I.; Marques, J.C.; Reis-Santos, P.; Fonseca, V.F.; et al. Glyphosate-Based Herbicide Toxicophenomics in Marine Diatoms: Impacts on Primary Production and Physiological Fitness. Appl. Sci. 2020, 10, 7391. [Google Scholar] [CrossRef]
- Sommer, U.; Adrian, R.; Bauer, B.; Winder, M. The response of temperate aquatic ecosystems to global warming: Novel insights from a multidisciplinary project. Mar. Biol. 2012, 159, 2367–2377. [Google Scholar] [CrossRef]
- Hazel, J.R. Thermal Adaptation in Biological-Membranes-Is Homeoviscous Adaptation the Explanation. Annu. Rev. Physiol. 1995, 57, 19–42. [Google Scholar] [CrossRef]
- Bagnyukova, T.V.; Danyliv, S.I.; Zin’Ko, O.S.; Lushchak, V.I. Heat shock induces oxidative stress in rotan Perccottus glenii tissues. J. Therm. Biol. 2007, 32, 255–260. [Google Scholar] [CrossRef]
- Folch, J.; Lees, M.; Stanley, G.S. A simple method for the isolation and purification of total lipides from animal tissues. J. Biol. Chem. 1957, 226, 497–509. [Google Scholar] [CrossRef]
- Fadhlaoui, M.; Laderriere, V.; Lavoie, I.; Fortin, C. Influence of Temperature and Nickel on Algal Biofilm Fatty Acid Composition. Environ. Toxicol. Chem. 2020, 39, 1566–1577. [Google Scholar] [CrossRef]
- Habig, W.H.; Pabst, M.J.; Jakoby, W.B. Glutathione S-transferases. The first enzymatic step in mercapturic acid formation. J. Biol. Chem. 1974, 249, 7130–7139. [Google Scholar] [CrossRef]
- Mubiana, V.K.; Blust, R. Effects of temperature on scope for growth and accumulation of Cd, Co, Cu and Pb by the marine bivalve Mytilus edulis. Mar. Environ. Res. 2007, 63, 219–235. [Google Scholar] [CrossRef] [Green Version]
- Nichols, J.W.; Playle, R.C. Influence of temperature on silver accumulation and depuration in rainbow trout. J. Fish. Biol. 2004, 64, 1638–1654. [Google Scholar] [CrossRef]
- Monari, M.; Matozzo, V.; Foschi, J.; Cattani, O.; Serrazanetti, G.P.; Marin, M.G. Effects of high temperatures on functional responses of haemocytes in the clam Chamelea gallina. Fish Shellfish Immunol. 2007, 22, 98–114. [Google Scholar] [CrossRef]
- Verlecar, X.; Jena, K.; Chainy, G. Biochemical markers of oxidative stress in Perna viridis exposed to mercury and temperature. Chem. Interact. 2007, 167, 219–226. [Google Scholar] [CrossRef]
- Wang, J.; Ren, R.-M.; Yao, C.-L. Oxidative stress responses of Mytilus galloprovincialis to acute cold and heat during air exposure. J. Molluscan Stud. 2018, 84, 285–292. [Google Scholar] [CrossRef]
- Heise, K.; Puntarulo, S.; Portner, H.O.; Abele, D. Production of reactive oxygen species by isolated mitochondria of the Antarctic bivalve Laternula elliptica (King and Broderip) under heat stress. Comp. Biochem. Physiol. C-Toxicol. Pharmacol. 2003, 134, 79–90. [Google Scholar] [CrossRef]
- Taylor, A.M.; Maher, W.A.; Ubrihien, R.P. Mortality, condition index and cellular responses of Anadara trapezia to combined salinity and temperature stress. J. Exp. Mar. Biol. Ecol. 2017, 497, 172–179. [Google Scholar] [CrossRef]
- Madeira, D.; Narciso, L.; Cabral, H.N.; Diniz, M.S.; Vinagre, C. Role of thermal niche in the cellular response to thermal stress: Lipid peroxidation and HSP70 expression in coastal crabs. Ecol. Indic. 2014, 36, 601–606. [Google Scholar] [CrossRef]
- Farombi, E.O.; Adelowo, O.A.; Ajimoko, Y.R. Biomarkers of Oxidative Stress and Heavy Metal Levels as Indicators of Environmental Pollution in African Cat Fish (Clarias gariepinus) from Nigeria Ogun River. Int. J. Environ. Res. Public Health. 2007, 4, 158–165. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cavalli, V.L.D.L.O.; Cattani, D.; Rieg, C.E.H.; Pierozan, P.; Zanatta, L.; Parisotto, E.B.; Filho, D.W.; Silva, F.R.M.B.; Pessoa-Pureur, R.; Zamoner, A. Roundup disrupts male reproductive functions by triggering calcium-mediated cell death in rat testis and Sertoli cells. Free Radic. Biol. Med. 2013, 65, 335–346. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Olorunsogo, O.O. Modification of the transport of protons and Ca2+ ions across mitochondrial coupling membrane by N-(phosphonomethyl)glycine. Toxicology 1990, 61, 205–209. [Google Scholar] [CrossRef]
- Jokanović, M. Biotransformation of organophosphorus compounds. Toxicology 2001, 166, 139–160. [Google Scholar] [CrossRef]
- Farcy, E.; Gagne, F.; Martel, L.; Fortier, M.; Trépanier, S.; Brousseau, P.; Fournier, M. Short-term physiological effects of a xenobiotic mixture on the freshwater mussel Elliptio complanata exposed to municipal effluents. Environ. Res. 2011, 111, 1096–1106. [Google Scholar] [CrossRef]
- Damiens, G.; His, E.; Gnassia-Barelli, M.; Quiniou, F.; Roméo, M. Evaluation of biomarkers in oyster larvae in natural and polluted conditions. Comp. Biochem. Physiol. Part C Toxicol. Pharmacol. 2004, 138, 121–128. [Google Scholar] [CrossRef] [Green Version]
- Dos Santos, K.C.; Martinez, C.B. Genotoxic and biochemical effects of atrazine and Roundup((R)), alone and in combination, on the Asian clam Corbicula fluminea. Ecotoxicol. Environ. Saf. 2014, 100, 7–14. [Google Scholar] [CrossRef]
- Barky, F.A.; Abdelsalam, H.A.; Mahmoud, M.B.; Hamdi, S.A. Influence of Atrazine and Roundup pesticides on biochemical and molecular aspects of Biomphalaria alexandrina snails. Pestic. Biochem. Physiol. 2012, 104, 9–18. [Google Scholar] [CrossRef]
- Séguin, A.; Mottier, A.; Perron, C.; Lebel, J.M.; Serpentini, A.; Costil, K. Sub-lethal effects of a glyphosate-based commercial formulation and adjuvants on juvenile oysters (Crassostrea gigas) exposed for 35 days. Mar. Pollut. Bull. 2017, 117, 348–358. [Google Scholar] [CrossRef]
- Kraffe, E.; Soudant, P.; Marty, Y. Fatty acids of serine, ethanolamine, and choline plasmalogens in some marine bivalves. Lipids 2004, 39, 59–66. [Google Scholar] [CrossRef]
- Pazos, A.J.; Sánchez, J.L.; Román, G.; Pérez-Parallé, M.L.; Abad, M. Seasonal changes in lipid classes and fatty acid composition in the digestive gland of Pecten maximus. Comp. Biochem. Physiol. Part B Biochem. Mol. Biol. 2003, 134, 367–380. [Google Scholar] [CrossRef]
- Saito, H. Lipid and FA composition of the pearl oyster Pinctada fucata martensii: Influence of season and maturation. Lipids 2004, 39, 997–1005. [Google Scholar] [CrossRef]
- Fried, B.; Rao, K.; Sherma, J.; Huffman, J.E. Fatty acid composition of Goniobasis virginica, Physa sp. and Viviparus malleatus (Mollusca: Gastropoda) from Lake Musconetcong, New Jersey. Biochem. Syst. Ecol. 1993, 21, 809–812. [Google Scholar] [CrossRef]
- Kelly, J.R.; Scheibling, R.E. Fatty acids as dietary tracers in benthic food webs. Mar. Ecol. Prog. Ser. 2012, 446, 1–22. [Google Scholar] [CrossRef] [Green Version]
- Hochachka, P.; Somero, G. Biochemical Adaptation: Mechanism and Process in Physiological Evolution; Oxford University Press: New York, NY, USA, 2002. [Google Scholar]
- Pernet, F.; Tremblay, R.; Comeau, L.; Guderley, H. Temperature adaptation in two bivalve species from different thermal habitats: Energetics and remodelling of membrane lipids. J. Exp. Biol. 2007, 210, 2999–3014. [Google Scholar] [CrossRef] [Green Version]
- Anacleto, P.; Maulvault, A.L.; Bandarra, N.M.; Repolho, T.; Nunes, M.L.; Rosa, R.; Marques, A. Effect of warming on protein, glycogen and fatty acid content of native and invasive clams. Food Res. Int. 2014, 64, 439–445. [Google Scholar] [CrossRef]
- Cohen, Z.; Norman, H.A.; Heimer, Y.M. Potential use of substituted pyridazinones for selecting polyunsaturated fatty acid overproducing cell lines of algae. Phytochemistry 1993, 32, 259–264. [Google Scholar] [CrossRef]
- Robert, S.; Mansour, M.P.; Blackburn, S.I. Metolachlor-Mediated Selection of a Microalgal Strain Producing Novel Polyunsaturated Fatty Acids. Mar. Biotechnol. 2006, 9, 146–153. [Google Scholar] [CrossRef]
- Barnathan, G. Non-methylene-interrupted fatty acids from marine invertebrates: Occurrence, characterization and biological properties. Biochimie 2009, 91, 671–678. [Google Scholar] [CrossRef]
- Gonçalves, A.; Mesquita, A.; Verdelhos, T.; Coutinho, J.; Marques, J.; Gonçalves, F. Fatty acids’ profiles as indicators of stress induced by of a common herbicide on two marine bivalves species: Cerastoderma edule (Linnaeus, 1758) and Scrobicularia plana (da Costa, 1778). Ecol. Indic. 2016, 63, 209–218. [Google Scholar] [CrossRef]
- Signa, G.; Di Leonardo, R.; Vaccaro, A.; Tramati, C.D.; Mazzola, A.; Vizzini, S. Lipid and fatty acid biomarkers as proxies for environmental contamination in caged mussels Mytilus galloprovincialis. Ecol. Indic. 2015, 57, 384–394. [Google Scholar] [CrossRef]
| GLY | TEMP | GLY*TEMP | |
|---|---|---|---|
| GPx | *** | * | |
| SOD | ** | ||
| GST | *** | ||
| CAT | |||
| MDA |
| 20 °C | 25 °C | Two-Way ANOVA | |||||
|---|---|---|---|---|---|---|---|
| CTL | GLY | CTL | GLY | GLY | TEMP | GLY * TEMP | |
| 14:0 | 0.56 ± 0.05 | 0.50 ± 0.10 | 0.57 ± 0.04 | 0.43 ± 0.05 | |||
| 15:0 | 0.32 ± 0.03 | 0.26 ± 0.01 | 0.35 ± 0.03 | 0.34 ± 0.02 | ** | ||
| 16:0 | 17.13 ± 0.83 | 18.01 ± 0.61 | 15.60 ± 1.11 | 16.71 ± 0.81 | * | ||
| 17:0 | 0.73 ± 0.07 | 0.66 ± 0.08 | 0.80 ± 0.04 | 0.90 ± 0.04 | ** | ||
| 18:0 | 11.27 ± 0.94 | 10.56 ± 1.05 | 13.37 ± 1.05 | 17.26 ± 0.92 | *** | ||
| 20:0 | 0.49 ± 0.06 | 1.56 ± 0.77 | 0.41 ± 0.04 | 0.34 ± 0.01 | |||
| 21:0 | 0.10 ± 0.03 | 0.18 ± 0.10 | 0.16 ± 0.01 | 0.09 ± 0.06 | |||
| 22:0 | 0.53 ± 0.05 | 0.27 ± 0.15 | 1.33 ± 0.05 | 0.19 ± 0.06 | |||
| 24:0 | 2.54 ± 0.19 | 2.63 ± 0.23 | 1.38 ± 0.23 | 1.84 ± 0.11 | ** | ||
| 16:1n7 | 0.16 ± 0.11 | 0.80 ± 0.18 | 0.16 ± 0.14 | 0.30 ± 0.13 | *** | ||
| 17:1 | 0.95 ± 0.44 | 1.25 ± 0.28 | 0.63 ± 0.40 | 1.40 ± 0.55 | |||
| 18:1n9 | 22.05 ± 1.59 | 24.28 ± 1.24 | 19.72 ± 1.13 | 18.73 ± 0.88 | *** | ||
| 18:1n7 | 0.40 ± 0.20 | 1.03 ± 0.05 | 0.67 ± 0.18 | 0.24 ± 0.17 | ** | ||
| 20:1n9 | 2.83 ± 0.12 | 2.83 ± 0.18 | 3.41 ± 0.46 | 2.76 ± 0.13 | |||
| 22:1n9 | 0.16 ± 0.03 | 0.15 ± 0.04 | 0.26 ± 0.03 | 0.15 ± 0.02 | |||
| 18:2n6 | 12.57 ± 0.86 | 12.94 ± 0.01 | 11.55 ± 0.62 | 9.80 ± 0.59 | ** | ||
| 20:2n6 | 5.98 ± 0.56 | 4.92 ± 0.32 | 6.06 ± 0.53 | 6.53 ± 0.51 | * | * | |
| 22:2n6 | 1.08 ± 0.56 | 2.76 ± 0.89 | 1.62 ± 0.63 | 0.24 ± 0.12 | * | ||
| 18:3n6 | 0.37 ± 0.09 | 0.08 ± 0.08 | 0.39 ± 0.11 | 0.27 ± 0.10 | * | * | |
| 18:3n4 | 0.28 ± 0.07 | 0.22 ± 0.02 | 0.44 ± 0.04 | 0.31 ± 0.07 | * | ||
| 18:3n3 | 0.82 ± 0.02 | 0.69 ± 0.00 | 0.80 ± 0.02 | 0.64 ± 0.03 | ** | ||
| 20:3n6 | 1.91 ± 0.35 | 0.95 ± 0.04 | 1.65 ± 0.12 | 1.65 ± 0.03 | ** | ** | |
| 20:3n3 | 0.67 ± 0.15 | 0.50 ± 0.05 | 0.53 ± 0.18 | 0.33 ± 0.13 | |||
| 18:4n3 | 0.13 ± 0.02 | 0.11 ± 0.32 | 0.12 ± 0.01 | 0.08 ± 0.00 | * | ||
| 20:4n6 | 10.00 ± 1.52 | 6.60 ± 0.06 | 12.20 ± 1.01 | 13.07 ± 0.04 | *** | ** | |
| 20:4n3 | 0.33 ± 0.07 | 0.23 ± 0.00 | 0.36 ± 0.12 | 0.11 ± 0.01 | ** | ||
| 20:5n3 | 4.90 ± 0.25 | 4.43 ± 0.30 | 4.79 ± 0.32 | 4.53 ± 0.38 | |||
| 22:5n3 | 0.34 ± 0.15 | 0.35 ± 0.13 | 0.44 ± 0.19 | 0.46 ± 0.22 | |||
| Others * | 0.43 ± 0.05 | 0.22 ± 0.25 | 0.22 ± 0.02 | 0.25 ± 0.15 | |||
| U/S Ratio | 2.00 ± 0.13 | 1.92 ± 0.15 | 2.00 ± 0.18 | 1.65 ± 0.11 | |||
| n-9 UFA | 25.06 ± 1.64 | 27.34 ± 1.38 | 23.48 ± 0.81 | 21.67 ± 0.84 | *** | ||
| n-6 UFA | 31.89 ± 1.66 | 28.25 ± 1.12 | 33.47 ± 1.84 | 31.57 ± 1.61 | * | ||
| n-3 UFA | 7.28 ± 0.34 | 6.36 ± 0.41 | 7.06 ± 0.64 | 6.18 ± 0.49 | |||
| SFA | 33.66 ± 1.35 | 34.64 ± 1.77 | 33.97 ± 2.03 | 36.56 ± 2.14 | |||
| MUFA | 26.64 ± 1.84 | 30.46 ± 1.64 | 24.99 ± 1.15 | 23.82 ± 0.73 | *** | * | |
| PUFA | 39.71 ± 1.99 | 34.90 ± 1.44 | 41.04 ± 2.41 | 39.62 ± 2.27 | |||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fadhlaoui, M.; Lavoie, I. Effects of Temperature and Glyphosate on Fatty Acid Composition, Antioxidant Capacity, and Lipid Peroxidation in the Gastropod Lymneae sp. Water 2021, 13, 1039. https://doi.org/10.3390/w13081039
Fadhlaoui M, Lavoie I. Effects of Temperature and Glyphosate on Fatty Acid Composition, Antioxidant Capacity, and Lipid Peroxidation in the Gastropod Lymneae sp. Water. 2021; 13(8):1039. https://doi.org/10.3390/w13081039
Chicago/Turabian StyleFadhlaoui, Mariem, and Isabelle Lavoie. 2021. "Effects of Temperature and Glyphosate on Fatty Acid Composition, Antioxidant Capacity, and Lipid Peroxidation in the Gastropod Lymneae sp." Water 13, no. 8: 1039. https://doi.org/10.3390/w13081039





