Ambient Particulate Air Pollution and Daily Hospital Admissions in 31 Cities in Poland
Abstract
:1. Introduction
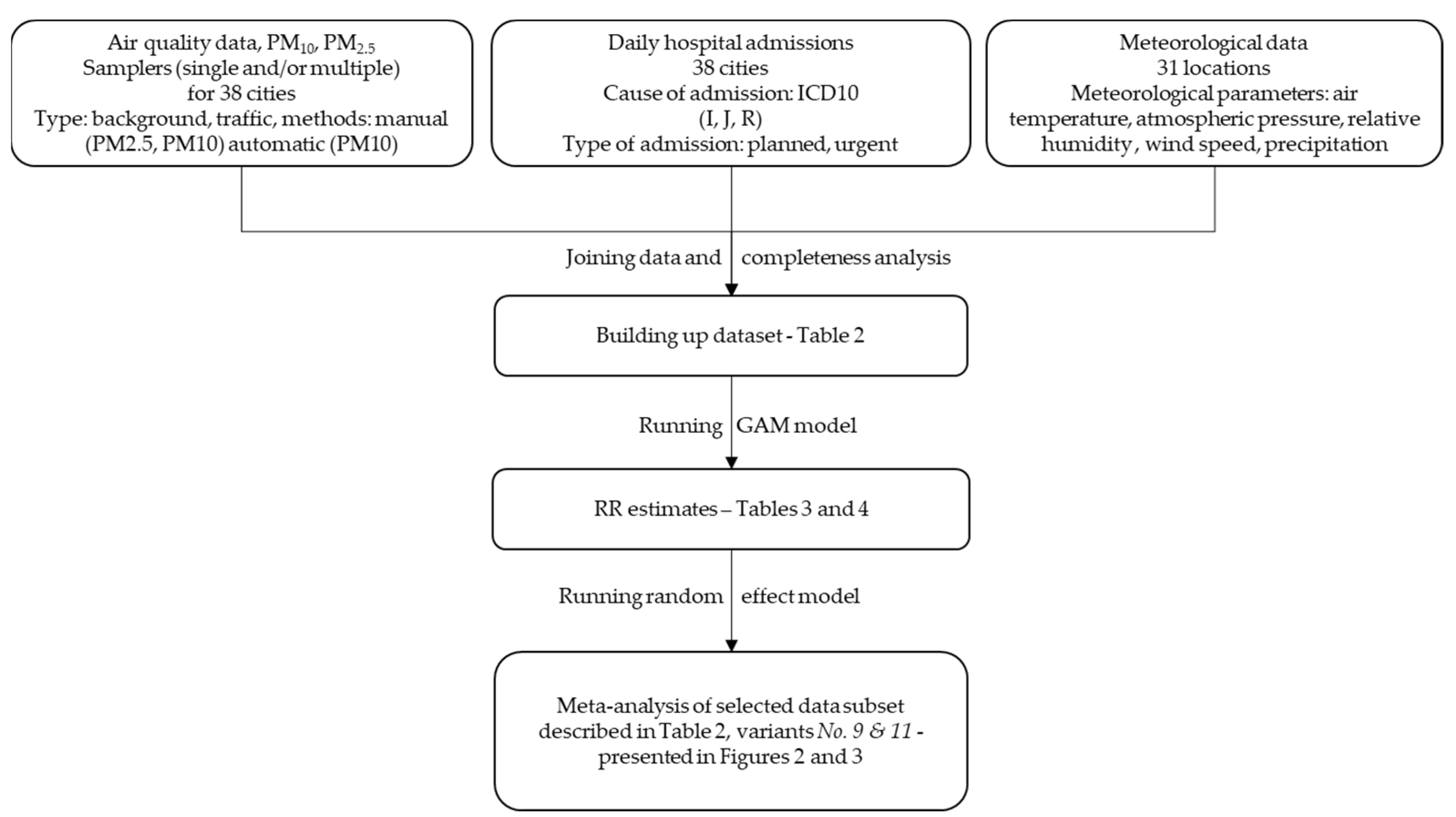
2. Material and Methods
2.1. Dataset
2.2. Statistical Model
2.3. Calculation Variants
3. Results
3.1. Meteorological Conditions and Air Quality in 31 Analyzed Cities in 2011–2017
3.2. Urgent Hospital Admissions Due to Cardiovascular and Respiratory Disorders
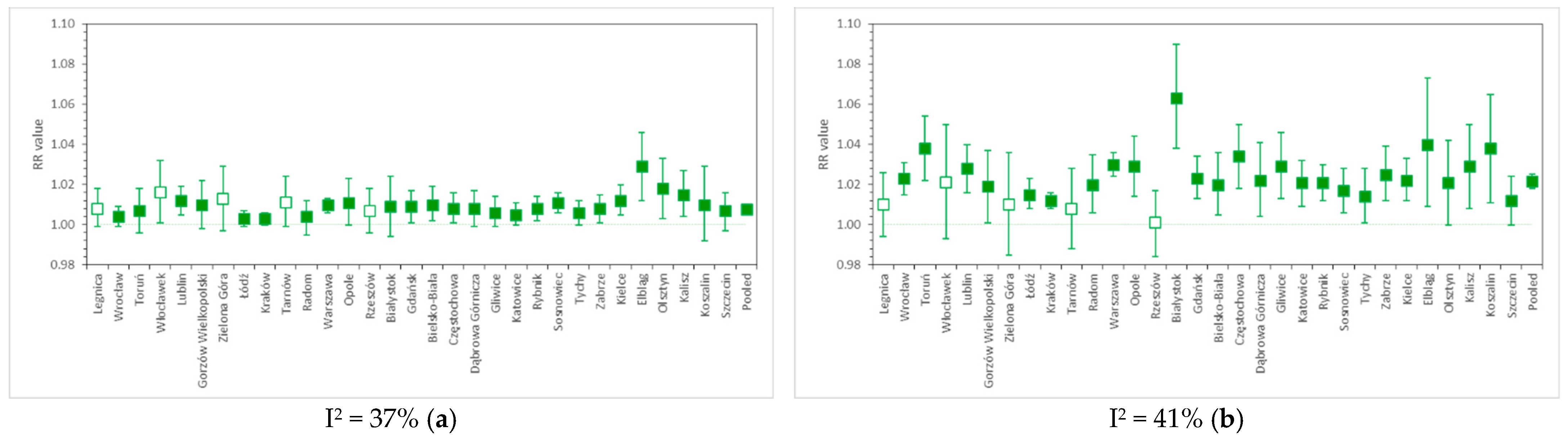
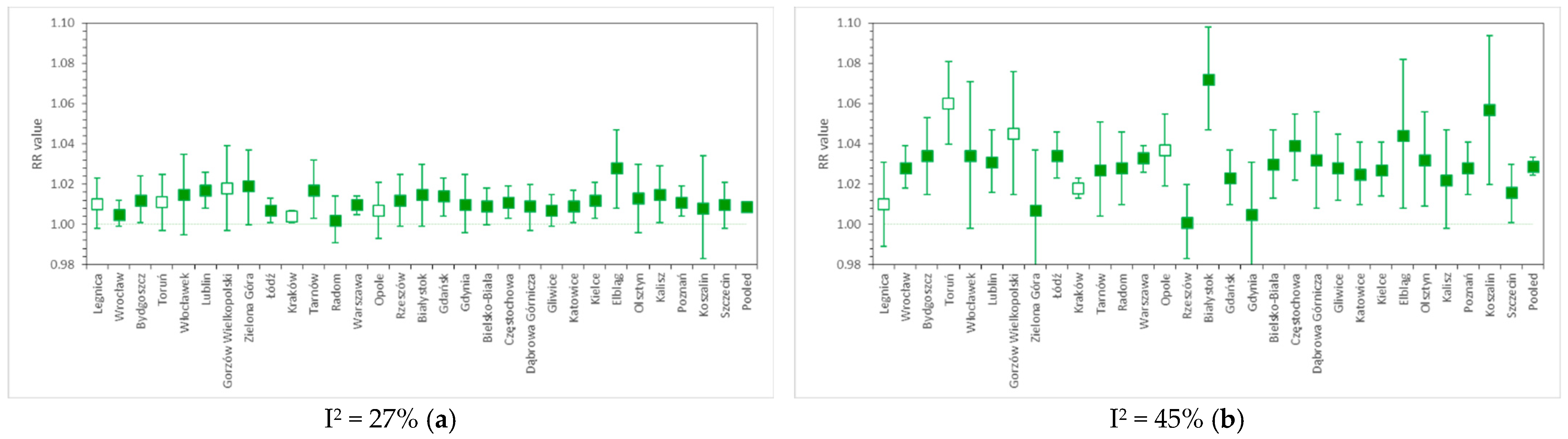
3.3. Pooled RR Results in Various Calculation Variants
4. Discussion
4.1. Influence of Input Data Modifications on the Results of Different Calculation Variants in Both Protocols
4.2. Relative Risk of Cardiovascular and Respiratory Hospital Admissions in 31 Polish Cities
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Brauer, M.; Brook, J.R.; Christidis, T.; Chu, Y.; Crouse, D.L.; Erickson, A.; Hystad, P.; Li, C.; Martin, R.V.; Meng, J.; et al. Mortality–Air Pollution Associations in Low-Exposure Environments (MAPLE), Phase 1; Research Reports; Health Effects Institute: Boston, MA, USA, 2019; p. 203. [Google Scholar]
- Dominici, F.; Schwartz, J.; Di, Q.; Braun, D.; Choirat, C.; Zanobetti, A. Assessing Adverse Health Effects of Long-Term Exposure to Low Levels of Ambient Air Pollution: Phase 1; Research Reports; Health Effects Institute: Boston, MA, USA, 2019; p. 200. [Google Scholar]
- Wolf, K.; Hoffmann, B.; Andersen, Z.J.; Atkinson, R.W.; Bauwelinck, M.; Bellander, T.; Brandt, J.; Brunekreef, B.; Cesaroni, G.; Chen, J.; et al. Long-term exposure to low-level ambient air pollution and incidence of stroke and coronary heart disease: A pooled analysis of six European cohorts within the ELAPSE project. Lancet Planet. Health 2021, 5, e620–e632. [Google Scholar] [CrossRef]
- Atkinson, R.W.; Kang, S.; Anderson, H.R.; Mills, I.C.; Walton, H.A. Epidemiological time series studies of PM2.5 and daily mortality and hospital admissions: A systematic review and meta-analysis. Thorax 2014, 69, 660–665. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kloog, I.; Ridgway, B.; Koutrakis, P.; Coull, B.A.; Schwartz, J.D. Long-and short-term exposure to PM2. 5 and mortality: Using novel exposure models. Epidemiology 2013, 24, 555. [Google Scholar] [CrossRef]
- Tian, Y.; Liu, H.; Wu, Y.; Si, Y.; Song, J.; Cao, Y.; Li, M.; Wu, Y.; Wang, X.; Chen, L.; et al. Association between ambient fine particulate pollution and hospital admissions for cause specific cardiovascular disease: Time series study in 184 major Chinese cities. BMJ 2019, 367, l6572. [Google Scholar] [CrossRef] [Green Version]
- Chen, C.; Zhao, B.; Weschler, C.J. Assessing the influence of indoor exposure to “outdoor ozone”on the relationship between ozone and short-term mortality in U.S. communities. Environ. Health Perspect. 2012, 120, 235–240. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hwang, S.H.; Choi, Y.; Paik, H.J.; Wee, W.R.; Kim, M.K.; Kim, D.H. Potential Importance of Ozone in the Association Between Outdoor Air Pollution and Dry Eye Disease in South Korea. JAMA Ophthalmol. 2016, 134, 503–510. [Google Scholar] [CrossRef] [Green Version]
- Goudarzi, G.; Geravandi, S.; Foruozandeh, H.; Babaei, A.A.; Alavi, N.; Niri, M.V.; Khodayar, M.J.; Salmanzadeh, S.; Mohammadi, M.J. Cardiovascular and respiratory mortality attributed to ground-level ozone in Ahvaz, Iran. Environ. Monit. Assess. 2015, 187, 1–9. [Google Scholar] [CrossRef]
- Beelen, R.; Raaschou-Nielsen, O.; Stafoggia, M.; Andersen, Z.J.; Weinmayr, G.; Hoffmann, B.; Wolf, K.; Samoli, E.; Fischer, P.; Nieuwenhuijsen, M.; et al. Effects of long-term exposure to air pollution on natural-cause mortality: An analysis of 22 European cohorts within the multicentre ESCAPE project. Lancet 2017, 383, 785–795. [Google Scholar] [CrossRef]
- Crouse, D.L.; Peters, P.A.; Hystad, P.; Brook, J.R.; van Donkelaar, A.; Martin, R.V.; Villeneuve, P.J.; Jerrett, M.; Goldberg, M.S.; Pope, C.A., 3rd; et al. Ambient PM2.5, O3, and NO2 Exposures and Associations with Mortality over 16 Years of Follow-Up in the Canadian Census Health and Environment Cohort (CanCHEC). Environ. Health Perspect. 2015, 123, 1180–1186. [Google Scholar] [CrossRef] [Green Version]
- Zanobetti, A.; Schwartz, J.; Dockery, D.W. Airborne particles are a risk factor for hospital admissions for heart and lung disease. Environ Health Perspect. 2000, 108, 1071–1077. [Google Scholar] [CrossRef] [PubMed]
- Dominici, F.; Peng, R.D.; Bell, M.L.; Pham, L.; McDermott, A.; Zeger, S.L.; Samet, J.M. Fine particulate air pollution and hospital admission for cardiovascular and respiratory diseases. JAMA 2006, 295, 1127–1134. [Google Scholar] [CrossRef] [Green Version]
- Sunyer, J.; Basagaña, X. Particles, and not gases, are associated with the risk of death in patients with chronic obstructive pulmonary disease. Int. J. Epidemiol. 2001, 30, 1138–1140. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, C.; Chen, R.; Sera, F.; Vicedo-Cabrera, A.M.; Guo, Y.; Tong, S.; Coelho, M.S.Z.S.; Saldiva, P.H.N.; Lavigne, E.; Matus, P.; et al. Ambient Particulate Air Pollution and Daily Mortality in 652 Cities. N. Engl. J. Med. 2019, 381, 705–715. [Google Scholar] [CrossRef]
- European Environment Agency. Air Quality in Europe—2019 Report, EEA 2019. p. 99. Available online: https://www.eea.europa.eu/publications/air-quality-in-europe-2019 (accessed on 8 November 2021).
- Air Pollutant Measurement Database in Poland/Główny Inspektorat Środowiska (Chief Inspectorate of Environmental Protection). Available online: https://powietrze.gios.gov.pl/pjp/archives (accessed on 13 September 2021).
- Bhaskaran, K.; Gasparrini, A.; Hajat, S.; Smeeth, L.; Armstrong, B. Time series regression studies in environmental epidemiology. Int. J. Epidemiol. 2013, 42, 1187–1195. [Google Scholar] [CrossRef] [PubMed]
- Available online: https://danepubliczne.imgw.pl/ (accessed on 13 September 2021).
- Available online: https://icd.who.int/browse10/2019/en (accessed on 13 September 2021).
- Hastie, T.; Tibshirani, R. Generalized additive models. Stat. Sci. 1986, 1, 297–318. [Google Scholar] [CrossRef]
- Katsouyanni, K.; Zmirou, D.; Spix, C.; Sunyer, J.; Schouten, J.P.; Pönkä, A.; Anderson, H.R.; Le Moullec, Y.; Wojtyniak, B.; Vigotti, M.A.; et al. Short-term effects of air pollution on health: A European approach using epidemiological time-series data. The APHEA project: Background, objectives, design. Eur. Respir. J. 1995, 8, 1030–1038. [Google Scholar]
- Katsouyanni, K.; Schwartz, J.; Spix, C.; Touloumi, G.; Zmirou, D.; Zanobetti, A.; Wojtyniak, B.; Vonk, J.M.; Tobias, A.; Pönkä, A.; et al. Short term effects of air pollution on health: A European approach using epidemiologic time series data: The APHEA protocol. J. Epidemiol. Community Health 1996, 50 (Suppl. 1), S12–S18. [Google Scholar] [CrossRef] [Green Version]
- Samet, J.M.; Zeger, S.L.; Dominici, F.; Curriero, F.C.; Coursac, I.; Dockery, D.W.; Schwartz, J.; Zanobetti, A. The National Morbidity, Mortality, and Air Pollution Study Part II: Morbidity and Mortality from Air Pollution in the United States; Research Report 94; Health Effects Institute: Cambridge, MA, USA, 2000. [Google Scholar]
- Wojtyniak, B.; Goryński, P. (Eds.) Health Status of Polish Population and Its Determinants; National Institute of Public Health—National Institute of Hygiene: Warsaw, Poland, 2018; p. 492. [Google Scholar]
- Bayer-Oglesby, L.; Schindler, C.; Hazenkamp-von Arx, M.E.; Braun-Fahrländer, C.; Keidel, D.; Rapp, R.; Künzli, N.; Braendli, O.; Burdet, L.; Sally, L.L.J.; et al. Living near main streets and respiratory symptoms in adults: The Swiss Cohort Study on Air Pollution and Lung Diseases in Adults. Am. J. Epidemiol. 2006, 164, 1190–1198. [Google Scholar] [CrossRef] [Green Version]
- McConnell, R.; Islam, T.; Shankardass, K.; Jerrett, M.; Lurmann, F.; Gilliland, F.; Gauderman, J.; Avol, E.; Künzli, N.; Yao, L.; et al. Childhood incident asthma and traffic-related air pollution at home and school. Environ. Health Perspect. 2010, 118, 1021–1026. [Google Scholar] [CrossRef] [Green Version]
- Badyda, A.J.; Dabrowiecki, P.; Lubinski, W.; Czechowski, P.O.; Majewski, G.; Chcialowski, A.; Kraszewski, A. Influence of traffic-related air pollutants on lung function. Adv. Exp. Med. Biol. 2013, 788, 229–235. [Google Scholar] [CrossRef]
- Cakmak, S.; Dales, R.E.; Vida, C.B. Components of particulate air pollution and mortality in Chile. Int. J. Occup. Environ. Health. 2009, 15, 152–158. [Google Scholar] [CrossRef]
- Sarnat, J.A.; Marmur, A.; Klein, M.; Kim, E.; Russell, A.G.; Sarnat, S.E.; Mulholland, J.A.; Hopke, P.K.; Tolbert, P.E. Fine particle sources and cardiorespiratory morbidity: An application of chemical mass balance and factor analytical source-apportionment methods. Environ. Health Perspect. 2008, 116, 459–466. [Google Scholar] [CrossRef]
- Kelly, F.J.; Fussell, J.C. Size, source and chemical composition as determinants of toxicity attributable to ambient particulate matter. Atmos. Environ. 2012, 60, 504–526. [Google Scholar] [CrossRef]
- Shah, A.S.; Langrish, J.P.; Nair, H.; McAllister, D.A.; Hunter, A.L.; Donaldson, K.; Newby, D.E.; Mills, N.L. Global association of air pollution and heart failure: A systematic review and meta-analysis. Lancet 2013, 382, 1039–1048. [Google Scholar] [CrossRef] [Green Version]
- Maciejewska, K. Short-term impact of PM2.5, PM10, and PMc on mortality and morbidity in the agglomeration of Warsaw, Poland. Air Qual. Atmos. Health 2020, 13, 659–672. [Google Scholar] [CrossRef]
- Yee, J.; Cho, Y.A.; Yoo, H.J.; Yun, H.; Gwak, H.S. Short-term exposure to air pollution and hospital admission for pneumonia: A systematic review and meta-analysis. Environ. Health. 2021, 20, 6. [Google Scholar] [CrossRef] [PubMed]
- WHO Health Risks of air Pollution in Europe—HRAPIE Project. Recommendations for Concentration–Response Functions for Cost–Benefit Analysis of Particulate Matter, Ozone and Nitrogen Dioxide; World Health Organization; Regional Office for Europe, European Centre for Environment and Health: Bonn, Germany, 2013. [Google Scholar]
- DeVries, R.; Kriebel, D.; Sama, S. Outdoor Air Pollution and COPD-Related Emergency Department Visits, Hospital Admissions, and Mortality: A Meta-Analysis. J. Chronic Obstr. Pulm. Dis. 2017, 14, 113–121. [Google Scholar] [CrossRef] [PubMed]
- Kuźma, Ł.; Pogorzelski, S.; Struniawski, K.; Bachórzewska-Gajewska, H.; Dobrzycki, S. Exposure to air pollution-a trigger for myocardial infarction? A nine-year study in Bialystok-the capital of the Green Lungs of Poland (BIA-ACS registry). Int. J. Hyg. Environ. Health 2020, 229, 113578. [Google Scholar] [CrossRef]
- Kuźma, Ł.; Pogorzelski, S.; Struniawski, K.; Dobrzycki, S.; Bachórzewska-Gajewska, H. Effect of air pollution on the number of hospital admissions for acute coronary syndrome in elderly patients. Pol. Arch. Intern. Med. 2020, 130, 38–46. [Google Scholar] [CrossRef] [Green Version]
- Konduracka, E.; Niewiara, Ł.; Guzik, B.; Kotynia, M.; Szolc, P.; Gajos, G.; Nessler, J.; Podolec, P.; Żmudka, K. Effect of short-term fluctuations in outdoor air pollution on the number of hospital admissions due to acute myocardial infarction among inhabitants of Kraków, Poland. Pol. Arch. Intern. Med. 2019, 129, 88–96. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Niewiadomska, E.; Kowalska, M.; Niewiadomski, A.; Skrzypek, M.; Kowalski, M.A. Assessment of Risk Hospitalization due to Acute Respiratory Incidents Related to Ozone Exposure in Silesian Voivodeship (Poland). Int. J. Environ. Res. Public Health 2020, 17, 3591. [Google Scholar] [CrossRef] [PubMed]
- Wrotek, A.; Badyda, A.; Czechowski, P.O.; Owczarek, T.; Dąbrowiecki, P.; Jackowska, T. Air Pollutants’ Concentrations Are Associated with Increased Number of RSV Hospitalizations in Polish Children. J. Clin. Med. 2021, 10, 3224. [Google Scholar] [CrossRef] [PubMed]
- Kuźma, Ł.; Wańha, W.; Kralisz, P.; Kazmierski, M.; Bachórzewska-Gajewska, H.; Wojakowski, W.; Dobrzycki, S. Impact of short-term air pollution exposure on acute coronary syndrome in two cohorts of industrial and non-industrial areas: A time series regression with 6,000,000 person-years of follow-up (ACS—Air Pollution Study). Environ. Res. 2021, 19, 111154. [Google Scholar] [CrossRef]
- Slama, A.; Śliwczyński, A.; Woźnica-Pyzikiewicz, J.; Zdrolik, M.; Wiśnicki, B.; Kubajek, J.; Turżańska-Wieczorek, O.; Studnicki, M.; Wierzba, W.; Franek, E. The short-term effects of air pollution on respiratory disease hospitalizations in 5 cities in Poland: Comparison of time-series and case-crossover analyses. Environ. Sci. Pollut. Res. Int. 2020, 27, 24582–24590. [Google Scholar] [CrossRef]
- Buszman, P.E.; Derbisz, K.; Kwasiborski, P.; Chrząszcz, P.; Mularska, M.; Baron, D.; Sobieszek, A.; Mendyk, A.; Skoczylas, P.; Cisowski, M.; et al. Impact of air pollution on hospital patients admitted with ST- and non-ST-segment elevation myocardial infarction in heavily polluted cities within the European Union. Cardiol. J. 2020, 27, 541–547. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Slama, A.; Śliwczyński, A.; Woźnica, J.; Zdrolik, M.; Wiśnicki, B.; Kubajek, J.; Turżańska-Wieczorek, O.; Gozdowski, D.; Wierzba, W.; Franek, E. Impact of air pollution on hospital admissions with a focus on respiratory diseases: A time-series multi-city analysis. Environ. Sci. Pollut. Res. Int. 2019, 26, 16998–17009. [Google Scholar] [CrossRef] [Green Version]
- Balun, M.; Białoskórska, U.; Bruszewski, H.; Burzyński, J.; Błaszczyk, J.; Cenowski, M.; Degórska, A.; Dyduch, B.; Greger, J.; Hławiczka, S.; et al. Analiza Stanu Zanieczyszczenia Powietrza Pyłem PM10 i PM2,5 z Uwzględnieniem Składu Chemicznego Pyłu Oraz Wpływu źródeł Naturalnych—Raport Końcowy; Report of the Chief Inspectorate of Environmental Protection; Instytut Podstaw Inżynierii Środowiska PAN; Instytut Ekologii Terenów Uprzemysłowionych; Instytut Meteorologii i Gospodarki Wodnej PIB; Instytut Ochrony Środowiska PIB: Zabrze, Poland, 2011; p. 297. (In Polish) [Google Scholar]
| Pollutant | Limit Value | Share of Monitoring Stations with Exceedances | Q1 [µg/m3] | Q3 [µg/m3] | Mean [µg/m3] |
|---|---|---|---|---|---|
| PM10 | 50 µg/m3, not to be exceeded daily more than 35 times per year | 31% | 13.3 a | 45.0 a | 29.8 a |
| PM10 | 40 µg/m3, annual mean | 3% | 22.5 | 29.8 | 26.6 |
| PM2.5 | 20 µg/m3 annual mean | 40% | 16.0 | 21.4 | 19.1 |
| Input Data | Statistical Model | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| No. | PMX | Type of Monitoring Stations | Measurement Mode | Meteo Var. | ICD-10 Codes | Patient Age Groups | Lags | Method of Choosing the Number of df | Spline Type | |
| 1 | PM10 | UB, traf, ind | manual | T, RH, p, WS, R | I00-I99, J00-J99, R00-R09 | all ages together | 0, 1, 5, 0–1, 0–5 | min PACF | penalized cubic splines | APHEA protocol |
| 2 | PM10 | UB | manual | I00-I99, J00-J99, R00-R09 | all ages together | 0, 1, 5, 0–1, 0–5 | ||||
| 3 | PM10 | UB | manual + automatic | I00-I99, J00-J99, R00-R09 | all ages together | 0, 1, 5, 0–1, 0–5 | ||||
| 4 | PM10 | UB, traf, ind | manual + automatic | I00-I99, J00-J99, R00-R09 | all ages together | 0, 0–1, 0–5 | ||||
| 5 | PM10 | UB | manual | I00-I99, J00-J99, R00-R09 | 0–14, 15–64, >65 | 0, 0–1, 0–5 | ||||
| 6 | PM10 | UB | manual + automatic | I00-I99, J00-J99, R00-R09 | 0–14, 15–64, >65 | 0, 0–1, 0–5 | ||||
| 7 | PM10 | UB | manual | I20-I25, I46-I52, I63-I69, J00-J47, R00, R05-R07 | all ages together | 0, 0–1, 0–5 | ||||
| 8 | PM2.5 | UB | manual | I00-I99, J00-J99, R00-R09 | all ages together | 0, 0–1, 0–5 | ||||
| 9 | PM10 | UB | manual | T, RH | I00-I99, J00-J99, R00-R09 | all ages together | 0–1 | fixed | natural splines | NMMAPS protocol |
| 10 | PM10 | UB | manual | I00-I99, J00-J99, R00-R10 | all ages together | 0–1 | penalized cubic splines | |||
| 11 | PM2.5 | UB | manual | I00-I99, J00-J99, R00-R11 | all ages together | 0–1 | natural splines | |||
| 12 | PM2.5 | UB | manual | I00-I99, J00-J99, R00-R12 | all ages together | 0–1 | penalized cubic splines | |||
| CV Disorders | RS Disorders | |||||
|---|---|---|---|---|---|---|
| City | All Year | Heating Season | Non-Heating Season | All Year | Heating Season | Non-Heating Season |
| Bialystok | 0.42 | 0.42 | 0.41 | 0.16 | 0.18 | 0.14 |
| Bielsko-Biala | 0.55 | 0.56 | 0.54 | 0.15 | 0.17 | 0.13 |
| Czestochowa | 0.75 | 0.76 | 0.73 | 0.18 | 0.21 | 0.15 |
| Dabrowa Gornicza | 0.68 | 0.70 | 0.67 | 0.16 | 0.18 | 0.14 |
| Elblag | 0.65 | 0.67 | 0.62 | 0.18 | 0.21 | 0.16 |
| Gdansk | 0.72 | 0.74 | 0.70 | 0.39 | 0.43 | 0.34 |
| Gliwice | 0.66 | 0.67 | 0.64 | 0.14 | 0.16 | 0.13 |
| Gorzow Wielkopolski | 0.64 | 0.67 | 0.61 | 0.31 | 0.34 | 0.27 |
| Kalisz | 0.71 | 0.74 | 0.67 | 0.23 | 0.25 | 0.20 |
| Katowice | 0.71 | 0.73 | 0.69 | 0.17 | 0.19 | 0.14 |
| Kielce | 0.87 | 0.89 | 0.84 | 0.41 | 0.45 | 0.38 |
| Koszalin | 0.77 | 0.80 | 0.74 | 0.39 | 0.43 | 0.35 |
| Kraków | 0.74 | 0.75 | 0.73 | 0.34 | 0.38 | 0.29 |
| Legnica | 0.82 | 0.84 | 0.81 | 0.28 | 0.31 | 0.25 |
| Lublin | 0.92 | 0.96 | 0.89 | 0.31 | 0.35 | 0.27 |
| Lodz | 0.80 | 0.82 | 0.78 | 0.19 | 0.21 | 0.16 |
| Olsztyn | 0.62 | 0.64 | 0.59 | 0.32 | 0.36 | 0.29 |
| Opole | 0.68 | 0.71 | 0.65 | 0.40 | 0.47 | 0.34 |
| Radom | 0.77 | 0.79 | 0.74 | 0.29 | 0.33 | 0.25 |
| Rybnik | 0.65 | 0.67 | 0.62 | 0.26 | 0.30 | 0.22 |
| Rzeszów | 0.58 | 0.60 | 0.55 | 0.26 | 0.29 | 0.23 |
| Sosnowiec | 0.68 | 0.69 | 0.66 | 0.13 | 0.14 | 0.11 |
| Szczecin | 0.64 | 0.66 | 0.61 | 0.38 | 0.42 | 0.33 |
| Tarnow | 0.70 | 0.72 | 0.68 | 0.27 | 0.30 | 0.23 |
| Torun | 0.62 | 0.64 | 0.61 | 0.30 | 0.34 | 0.26 |
| Tychy | 0.65 | 0.67 | 0.64 | 0.13 | 0.14 | 0.11 |
| Warszawa | 0.66 | 0.67 | 0.64 | 0.25 | 0.29 | 0.22 |
| Wloclawek | 0.64 | 0.65 | 0.62 | 0.19 | 0.22 | 0.16 |
| Wroclaw | 0.64 | 0.66 | 0.63 | 0.23 | 0.26 | 0.20 |
| Zabrze | 0.46 | 0.46 | 0.45 | 0.13 | 0.14 | 0.11 |
| Zielona Gora | 0.63 | 0.65 | 0.60 | 0.24 | 0.27 | 0.22 |
| MEAN | 0.68 | 0.70 | 0.66 | 0.25 | 0.28 | 0.22 |
| No. | Age Group | RR Lag 0 | C.I. | RR Lag 1 | C.I. | RR Lag 5 | C.I. | RR Lag 0_1 | C.I. | RR Lag 0_5 | C.I. | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | all | 1.006 | 1.005–1.007 | 1.002 | 1.001–1.004 | 1.001 | 1.001–1.002 | 1.003 | 1.002–1.004 | 1.002 | 1.001–1.002 | APHEA protocol |
| 2 | 1.006 | 1.004–1.007 | 1.002 | 1.001–1.003 | 1.001 | 1.000–1.002 | 1.003 | 1.002–1.003 | 1.002 | 1.001–1.002 | ||
| 3 | 1.006 | 1.004–1.007 | 1.003 | 1.002–1.004 | 1.002 | 1.001–1.003 | 1.003 | 1.002–1.004 | 1.002 | 1.001–1.002 | ||
| 4 | 1.006 | 1.005–1.007 | - | - | - | - | 1.003 | 1.002–1.004 | 1.002 | 1.002–1.003 | ||
| 5 | 0–14 | 1.013 | 1.002–1.024 | - | - | - | - | 1.011 | 1.001–1.021 | 1.011 | 1.004–1.019 | |
| 15–64 | 1.006 | 1.004–1.008 | - | - | - | - | 1.003 | 1.001–1.004 | 1.001 | 1.000–1.003 | ||
| >65 | 1.006 | 1.004–1.007 | - | - | - | - | 1.003 | 1.002–1.004 | 1.001 | 1.001–1.002 | ||
| 6 | 0–14 | 1.013 | 1.004–1.023 | - | - | - | - | 1.009 | 1.001–1.018 | 1.010 | 1.003–1.017 | |
| 15–64 | 1.006 | 1.005–1.008 | - | - | - | - | 1.003 | 1.002–1.004 | 1.002 | 1.001–1.003 | ||
| >65 | 1.006 | 1.005–1.007 | - | - | - | - | 1.003 | 1.002–1.004 | 1.002 | 1.001–1.002 | ||
| 7 | all | 1.006 | 1.004–1.007 | - | - | - | - | 1.003 | 1.002–1.004 | 1.001 | 1.001–1.002 | |
| 8 | all | 1.005 | 1.003–1.006 | - | - | - | - | 1.002 | 1.001–1.003 | 1.002 | 1.001–1.002 | |
| 9 | all | - | - | - | - | - | - | 1.0077 | 1.0062–1.0092 | - | - | NMMAPS protocol |
| 10 | - | - | - | - | - | - | 1.0078 | 1.0062–1.0093 | - | - | ||
| 11 | - | - | - | - | - | - | 1.0088 | 1.0072–1.0103 | - | - | ||
| 12 | - | - | - | - | - | - | 1.0087 | 1.0071–1.0103 | - | - |
| No. | Age Group | RR Lag 0 | C.I. | RR Lag 1 | C.I. | RR Lag 5 | C.I. | RR Lag 0_1 | C.I. | RR Lag 0_5 | C.I. | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | all | 1.008 | 1.006–1.010 | 1.005 | 1.003–1.008 | 1.005 | 1.003–1.006 | 1.004 | 1.002–1.006 | 1.003 | 1.001–1.004 | APHEA protocol |
| 2 | 1.008 | 1.005–1.010 | 1.005 | 1.003–1.007 | 1.004 | 1.003–1.006 | 1.004 | 1.002–1.005 | 1.003 | 1.001–1.004 | ||
| 3 | 1.007 | 1.005–1.009 | 1.005 | 1.003–1.006 | 1.005 | 1.003–1.006 | 1.003 | 1.002–1.004 | 1.003 | 1.002–1.004 | ||
| 4 | 1.007 | 1.005–1.009 | - | - | - | - | 1.003 | 1.002–1.004 | 1.002 | 1.001–1.002 | ||
| 5 | 0–14 | 1.008 | 1.004–1.012 | - | - | - | - | 1.004 | 1.000–1.008 | 1.001 | 0.998–1.004 | |
| 15–64 | 1.008 | 1.005–1.011 | - | - | - | - | 1.004 | 1.002–1.006 | 1.003 | 1.001–1.004 | ||
| >65 | 1.010 | 1.007–1.014 | - | - | - | - | 1.005 | 1.003–1.007 | 1.004 | 1.002–1.006 | ||
| 6 | 0–14 | 1.007 | 1.003–1.011 | - | - | - | - | 1.003 | 1.000–1.006 | 1.001 | 0.998–1.003 | |
| 15–64 | 1.008 | 1.005–1.010 | - | - | - | - | 1.004 | 1.002–1.005 | 1.003 | 1.002–1.004 | ||
| >65 | 1.010 | 1.007–1.013 | - | - | - | - | 1.005 | 1.004–1.007 | 1.004 | 1.003–1.006 | ||
| 7 | all | 1.008 | 1.005–1.010 | - | - | - | - | 1.004 | 1.002–1.006 | 1.003 | 1.001–1.004 | |
| 8 | all | 1.007 | 1.004–1.010 | - | - | - | - | 1.003 | 1.002–1.005 | 1.003 | 1.001–1.004 | |
| 9 | all | - | - | - | - | - | - | 1.0218 | 1.0182–1.0253 | - | - | NMMAPS protocol |
| 10 | - | - | - | - | - | - | 1.0202 | 1.0168–1.0236 | - | - | ||
| 11 | - | - | - | - | - | - | 1.0289 | 1.0244–1.0335 | - | - | ||
| 12 | - | - | - | - | - | - | 1.0263 | 1.0219–1.0307 | - | - |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Adamkiewicz, Ł.; Maciejewska, K.; Rabczenko, D.; Drzeniecka-Osiadacz, A. Ambient Particulate Air Pollution and Daily Hospital Admissions in 31 Cities in Poland. Atmosphere 2022, 13, 345. https://doi.org/10.3390/atmos13020345
Adamkiewicz Ł, Maciejewska K, Rabczenko D, Drzeniecka-Osiadacz A. Ambient Particulate Air Pollution and Daily Hospital Admissions in 31 Cities in Poland. Atmosphere. 2022; 13(2):345. https://doi.org/10.3390/atmos13020345
Chicago/Turabian StyleAdamkiewicz, Łukasz, Katarzyna Maciejewska, Daniel Rabczenko, and Anetta Drzeniecka-Osiadacz. 2022. "Ambient Particulate Air Pollution and Daily Hospital Admissions in 31 Cities in Poland" Atmosphere 13, no. 2: 345. https://doi.org/10.3390/atmos13020345
APA StyleAdamkiewicz, Ł., Maciejewska, K., Rabczenko, D., & Drzeniecka-Osiadacz, A. (2022). Ambient Particulate Air Pollution and Daily Hospital Admissions in 31 Cities in Poland. Atmosphere, 13(2), 345. https://doi.org/10.3390/atmos13020345






