1. Introduction
The way a cell senses and responds to a physical force can vary based on cell type or tissue microenvironment, potentially altering downstream events such as migration, homeostasis, differentiation, proliferation, or tumorigenesis [
1,
2]. An important element in the force transmission pathway is the nucleus, the largest and stiffest cell organelle [
3]. The nucleus has been characterized as a mechanosensor of the cell and transmits external forces emanating from the cellular microenvironment to the nuclear envelope housing the chromatin [
4,
5,
6]. Mechanical forces propagate from the extracellular matrix (ECM) to mechanosensitive focal adhesions and cytoskeletal structures. Cytoskeletal structures are then connected to the nuclear lamina, below the nuclear envelope, through the linker of nucleoskeleton and cytoskeleton (LINC) complex [
5]. Additionally, direct force application to the nucleus can induce chromatin stretching and transcriptional upregulation of a reporter transgene without involvement of the nucleo-cytoskeleton network [
7]. Isolated and intracellular nuclei can deform anisotropically in response to both active and passive externally applied forces. Active forces include atomic force microscopy (AFM) [
8], biaxial stretching devices [
9], and magnetic tweezers [
10], while passive forces include glass microtubes [
11], three-dimensional (3D) hydrogels [
12], and microfluidic devices [
13,
14,
15,
16,
17]. Using AFM and micropipette nuclear aspiration, the nucleus can physically deform with as little as a few nanonewtons of force [
8,
18]. Specifically, differentiated and undifferentiated nuclei alike preferentially deform along their minor (short) axis in reaction to an applied force [
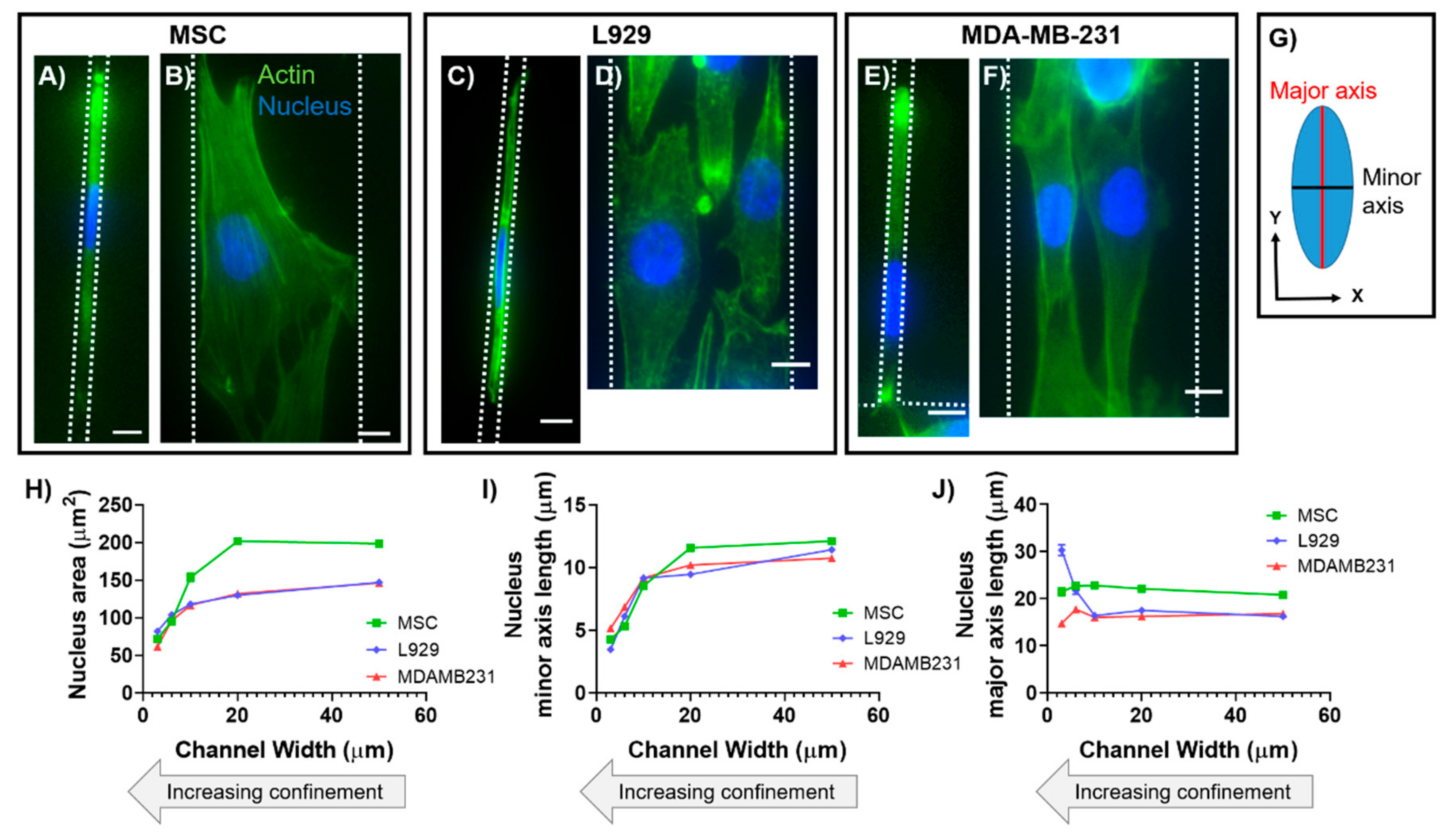
8,
9,
19]. We have previously shown that the nuclear major (long) axis in human mesenchymal stem cells (MSCs) maintains a constant length with increasing degrees of lateral confinement [
13]. In contrast, we have also shown that nuclear major (long) axis in mouse sarcoma (Ab3) cells increases in length with increasing degrees of lateral confinement [
14]. While anisotropic deformation has been found to be inherent to nuclei of many different cell types, nuclear axis lengths in three dimensions have not been quantified in confinement.
Physical confinement is experienced by cells in vivo in many contexts. For example, cells may be confined within channels between connective tissue and the basement membrane of muscle, nerve, or epithelium [
20,
21], during cell intravasation or extravasation [
22], in a tumor microenvironment [
23], or in interstitial tissue [
24]. Indeed, the migration modes of several cell types are altered by the degree of confinement induced [
20,
24,
25,
26,
27]. Although a vast array of cell types experience mechanical confinement, the components involved in mechanotransduction can differ among cell types. Cytoskeleton prestress, the composition of the LINC complex, and the organization of the nucleoskeleton can differ based on cell type [
10]. Specifically, increased expression of lamin A has been shown to correlate with increased nuclear, cell, and tissue stiffness [
28]. Furthermore, the sensitivity of a particular cell type to nuclear anisotropy is influenced by lamin A expression as well as chromatin organization, and lamin A over-expression results in an increase in strain along the nuclear minor axis in response to an applied force [
8]. Additionally, the deformation of the nuclear major and minor axes can be influenced by microtubule and actin structures. In short-term biaxial stretching experiments with epithelial monolayers, it was discovered that microtubules resist deformation along the major axis, while actin resists deformation along the minor axis [
9]. Microtubule and actin networks have also been known to regulate cell volume, which can dynamically alter nuclear morphology [
29]. In micro-engineered 3D environments, the ratio of cellular to nuclear volume is conserved, but the specific volumes of the nucleus and cell are dependent on cell type [
30]. It remains unknown whether cytoskeletal proteins influence nuclear deformability and nuclear volume in confined migration, and whether their role differs in varying cell types.
In this work, we characterized the 3D morphology of nuclei within two cell types in response to physical confinement. Cell types were chosen for experimentation due to their differences in tissue origin, organism origin, disease pathology, and level of differentiation potency. To recapitulate and control the degree of confinement experienced by cells in vitro, we employed polydimethylsiloxane (PDMS)-based microchannels that were 30, 60, 100, 200, or 500 μm
2 in cross-sectional area, which was previously designed and characterized [
16,
17]. Nuclear deformation in the XYZ planes, as well as nuclear area and volume, were quantified using immunofluorescence and ImageJ software. The effects of myosin II inhibition and microtubule depolymerization on nuclear deformation were additionally investigated. Our results suggest that cell type-dependent phenotypes may possess a larger role in governing nuclear deformation than the force of confinement alone, and that actin and microtubules may not independently modulate nuclear anisotropy in confined cell migration.
2. Materials and Methods
2.1. Cell Culture and Reagents
Bone marrow-derived human MSCs (donor: 20-year-old female) were purchased (RoosterBio Inc., Frederick, MD, USA) and experimentally used up until passage 5, which is when the population reaches a doubling level of 20. Human breast adenocarcinoma highly metastatic cells (MDA-MB-231 cells; American Type Culture Collection, Manassas, VA, USA) were used up to a passage of 30 after purchase. Finally, adult mouse fibroblasts (L929 cells) were gifted from Dr. John Fisher (Fischell Department of Bioengineering at the University of Maryland, College Park, MD, USA) and were used up to a passage of 10.
All cell types were cultured in either T-25 or T-75 polystyrene, plasma-treated flasks (VWR, Radnor, PA) and grown in medium comprised of 89% Dulbecco’s Modified Eagle’s Medium with high glucose (DMEM; ThermoFisher Scientific, Waltham, MA, USA), 10% heat-inactivated fetal bovine serum (FBS; ThermoFisher Scientific, Waltham, MA, USA), and 1% penicillin/streptomycin 1000 U/mL (P/S; ThermoFisher Scientific, Waltham, MA, USA). All cells were cultured at 37 °C, with 50% humidity and 5% CO2:95% air, and passaged at or below 90% confluency. Cells to be passaged were first washed in phosphate buffered saline (PBS; VWR, Radnor, PA) and detached from the flasks with 0.25% Trypsin-EDTA (ThermoFisher Scientific, Waltham, MA, USA), except for MSCs, which were treated with TrypLE Express Enzyme (ThermoFisher Scientific, Waltham, MA, USA).
2.2. Microfluidic Device Manufacturing and Cell Seeding
Polydimethylsiloxane (PDMS) microfluidic devices were prepared according to previously described protocols [
13,
16,
17]. The devices contained either a variety of channel widths or a repeating, single channel width. All photolithography procedures were carried out in the University of Maryland Nanocenter FabLab. In summary, separate masks were designed in AutoCAD (AutoDesk, San Rafael, CA, USA) for the channels (first feature) and the seeding and collection reservoirs (second feature). A 4-inch diameter silicon wafer (University Wafer, Boston, MA, USA) was spin coated with a 10-μm thick layer of SU-8-3010 negative photoresist (MicroChem, Westborough, MA, USA), and the mask representing the channels was placed over the coated wafer using the EVG620 mask aligner (EVG Group, Albany, NY, USA). The layers were then selectively exposed to UV to complete crosslinking of the SU-8-3010 in the areas dictated by the mask. Remaining SU-8-3010 was then dissolved using SU-8 developer (MicroChem). To create the second feature, a layer of SU-8 3025 negative photoresist (MicroChem, Westborough, MA, USA) was spin coated onto the wafer. Then, the EVG620 mask aligner was used to place the mask of the seeding and collection reservoirs over the coated wafer. The system was once again selectively exposed to UV, and the extra SU-8-3025 was removed. The completed wafers were silanized using 97% tridecafluoro-1,1,2,2, tetrahydrooctyl-1-trichlorosilane (UCT Inc., Bristol, PA, USA) overnight in a vacuum desiccator. The finished silicon masters contained the mold for manufacturing single- or multi-microchannel devices, defined by the widths of their channels, which included 50, 20, 10, 6, and 3 μm. Regardless of channel width, all channels were 10 μm in height and 200 μm in length.
PDMS devices were fabricated from the silicon masters. PDMS (Krayden, Denver, CO, USA) was weighed and mixed at a 10:1 base-to-curing agent ratio, poured onto a silicon master, degassed in a vacuum desiccator for approximately 30 min, and baked at 80 °C for at least 1 hour. Devices were cut out of the silicon master, and holes were punched into the PDMS to create cell and media inlets and outlets. Microchannels and 25 × 75 mm #1 glass coverslips (Electron Microscopy Sciences, Hatfield, PA, USA) were washed with ethanol and reverse osmosis or MilliQ water, dried for 5 min at 80 °C, then plasma treated in plasma cleaner (Harrick Plasma, Ithaca, NY, USA) for 2.5 min. The devices were bonded to the glass coverslips by applying pressure for 3 to 5 min, then UV sterilized for 10 min. Finally, devices were coated with 20 μg/mL collagen I (Sigma Aldrich, St. Louis, MO, USA) for 1 hour at 37 °C. After incubation, collagen was removed, devices were washed twice with PBS, and cells were immediately seeded into the cell inlet.
During the collagen I coating incubation time period, cells were prepared for device seeding. Cells were removed from the incubator, washed once with PBS, and detached with the addition of trypsin or TrypLE for 5 min. FBS-containing media was then added, and the cells were centrifuged at 1000 RPM for 5 min. Cells were resuspended in FBS-free media, counted, centrifuged again, and resuspended again in FBS-free media to yield 100,000 cells per 25 μl. Twenty-five microliters of cell suspension was pipetted into the cell inlets of the devices and incubated. After 5 min, excess cell suspension was removed, 50 μl of FBS-free media was added to the cell inlet and bottom two media inlets, and 50 μl of FBS-containing media was added to the topmost inlet to create a chemoattractant gradient. For studies including blebbistatin (50 μM, Sigma Aldrich, St. Louis, MO, USA) or nocodazole (10 μM, Sigma Aldrich, St. Louis, MO, USA), the inhibitor was added to both FBS-free and FBS-containing media, such that it was in equal concentrations throughout the device. Dimethylsulfoxide (Sigma Aldrich, St. Louis, MO, USA) served as the vehicle control in all experiments. Cells were incubated overnight to allow for migration through the channels. All microchannel devices containing cells were cultured at 37 °C, 50% humidity, and 5% CO2: 95% air.
2.3. Immunofluorescence
Following an overnight incubation period, cells and media were removed from the inlets and outlets. In all future steps listed below, when a reagent was added, it is implied that the same volume was added to all wells. Cells were washed once with PBS and immediately fixed with 3.7% formaldehyde (Fisher Scientific, Fair Lawn, NJ, USA). After a 10-min incubation at room temperature, cells were washed thrice with PBS. Cells were permeabilized by the addition of 0.5% Triton-X 100 (Sigma Aldrich, St. Louis, MO, USA) for 5 min, followed by three PBS washes. Nonspecific binding was then blocked with 2.5% goat serum (Abcam, Cambridge, MA, USA) for 1 hour at room temperature. Primary antibody diluted in PBS and 1% goat serum was added, and samples were incubated overnight at 4 °C. Primary antibody used was mouse anti-α-tubulin (ThermoFisher Scientific, Waltham, MA, USA, #A11126, 1:100 or 1:200). The next day, cells were washed twice or thrice in PBS, incubated in 2.5% goat serum for at least 1 hour and incubated with AlexaFluor 488 Phalloidin (ThermoFisher Scientific, Waltham, MA, USA, 1:500), Hoechst (ThermoFisher Scientific, Waltham, MA, USA, 1:500 or 1:2500), and a fluorescently labeled secondary antibody for 1 hour. Secondary antibody used was AlexaFluor 568 goat anti-mouse (ThermoFisher Scientific, Waltham, MA, USA, #A11004, 1:200). Cells were washed twice or thrice in PBS and imaged immediately or within a few days when Fluoromount-G (ThermoFisher Scientific, Waltham, MA, USA) was added for preservation.
2.4. Imaging
Widefield or confocal microscopy was used to extrapolate nuclear dimensions. MSC, MDA-MB-231, and L929 images for 2D measurement were acquired on an Olympus IX83 microscope (Olympus, Tokyo, Japan) using a 40× or 60× oil immersion objective. All z-stack images were taken using a PerkinElmer UltraVIEW Vox confocal spinning disk microscope (PerkinElmer, Waltham, MA) with a 100x oil immersion objective. Z-step size was 0.1 or 0.2 μm, and the boundaries of the stack were defined as the planes in which no staining could be observed. Use of the PerkinElmer confocal microscope was performed courtesy of the University of Maryland imaging core. In all experiments, fluorescence intensity and sensitivity were adjusted manually to optimize visualization.
2.5. Data Analysis in 2D
All data analysis for calculating 2D nuclear major axis length, minor axis length, and area was completed in ImageJ (U. S. National Institutes of Health, Bethesda, Maryland, USA) using the built-in morphology measurements. Widefield images were used for analysis. Nuclei were manually traced and measured, then dimensions originally output as pixels were converted into microns for analysis. Specifically, to calculate nuclear major and minor axis lengths, ImageJ fitted the traced nuclei to ellipses.
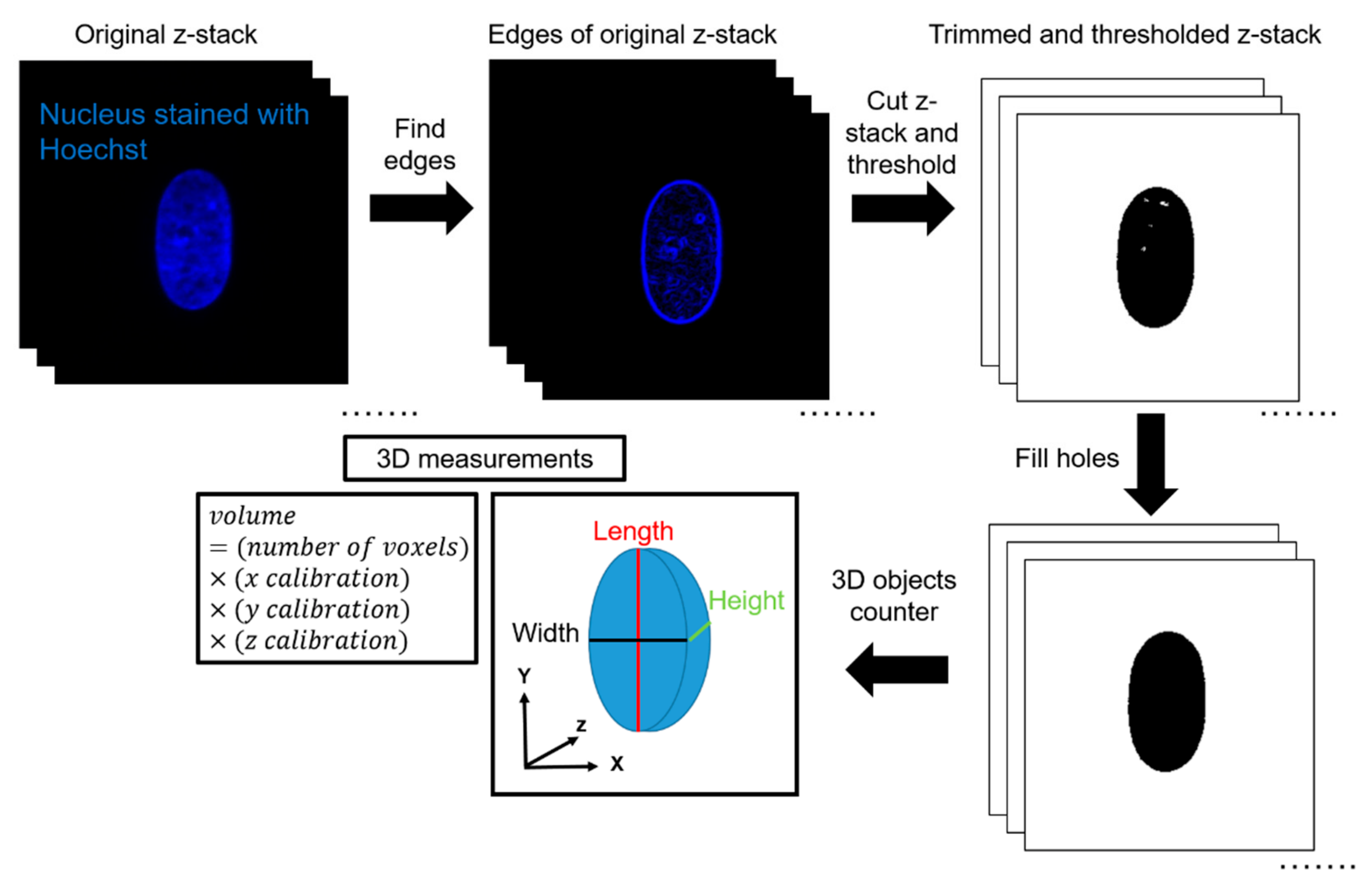
2.6. Data Analysis in 3D
ImageJ was used to quantify volume from the acquired z-stacks. First, the edges of the z-stacks were cut according to the built-in ImageJ feature ‘Find Edges’. This was completed to minimize the effects of background fluorescence during thresholding. The top and bottom planes were defined as the first and last image plane, respectively, in which an edge of the nucleus was visible. Next, IsoData thresholding was performed by ImageJ. IsoData threshold can be expressed mathematically as . This automated thresholding method was chosen to exclude any bias in the analysis, and for its ability to threshold consistently for nuclei in all channel widths. Thresholding was calculated based on the middle plane of the stack and applied to all other planes in the stack. After thresholding, the ImageJ feature ‘Fill Holes’was used to correct any large holes in the interior of the nuclei caused by excess thresholding or low fluorescence signal. The ImageJ feature ‘3D Object Counter’ was then used to automatically calculate the volume based on the number of voxels of the object. 3D Object Counter also yielded the length, width, and height of the object bounding box. This methodology was validated by seeding 10-μm, yellow-green fluorescent polystyrene microspheres (ThermoFisher Scientific, Waltham, MA, USA) inside the channels and completing a similar imaging protocol to experimental conditions. In a 50-μm wide channel and a z-step size of 0.15 μm, the volume calculated of the microsphere with the described methodology was 570 μm3, in comparison with the expected volume of 524 μm3, which yields an error percentage of 8.8%.
2.7. Statistical Analysis
To compare nuclear dimensions in different channel widths, a one-way ANOVA with Holm-Sidak’s multiple comparisons test, in the case of normally distributed data, or Kruskal–Wallis test with a Dunn’s post hoc test, in the case of non-normally distributed data, was completed. Data were pooled from at least three independent trials, except in 2D data for MSC and MDA-MB-231, which were pooled from two independent trials. A significance level of 0.05 was used. Error bars report the standard error of the mean. Select graphs were unable to fit all comparisons, in which case we refer readers to the
Figure 1H and
supplementary tables for full statistical analysis.
4. Discussion
In this study, we investigated how the passive force of confinement in microchannels affected nuclear deformation of various cell types. In 2D, nuclei of MSCs and MDA-MB-231 cells maintained a fairly constant major axis length when the minor axis was forcibly decreased by confining microchannel walls. In contrast, nuclei of L929 cells increased their major axis length as the minor axis was forcibly decreased by microchannels, which is similar to what we have previously shown in mouse sarcoma cells [
14]. It has also been reported in literature that the nuclei of several cell lines deform anisotropically in response to an applied force, with the minor axis exhibiting higher strain than the major axis [
8]. These results were demonstrated to be both intrinsic to the nucleus and controlled by the cytoskeleton, and it was postulated that microtubules resist nuclear deformation along the major axis, while actin resists deformation along the minor axis [
8].
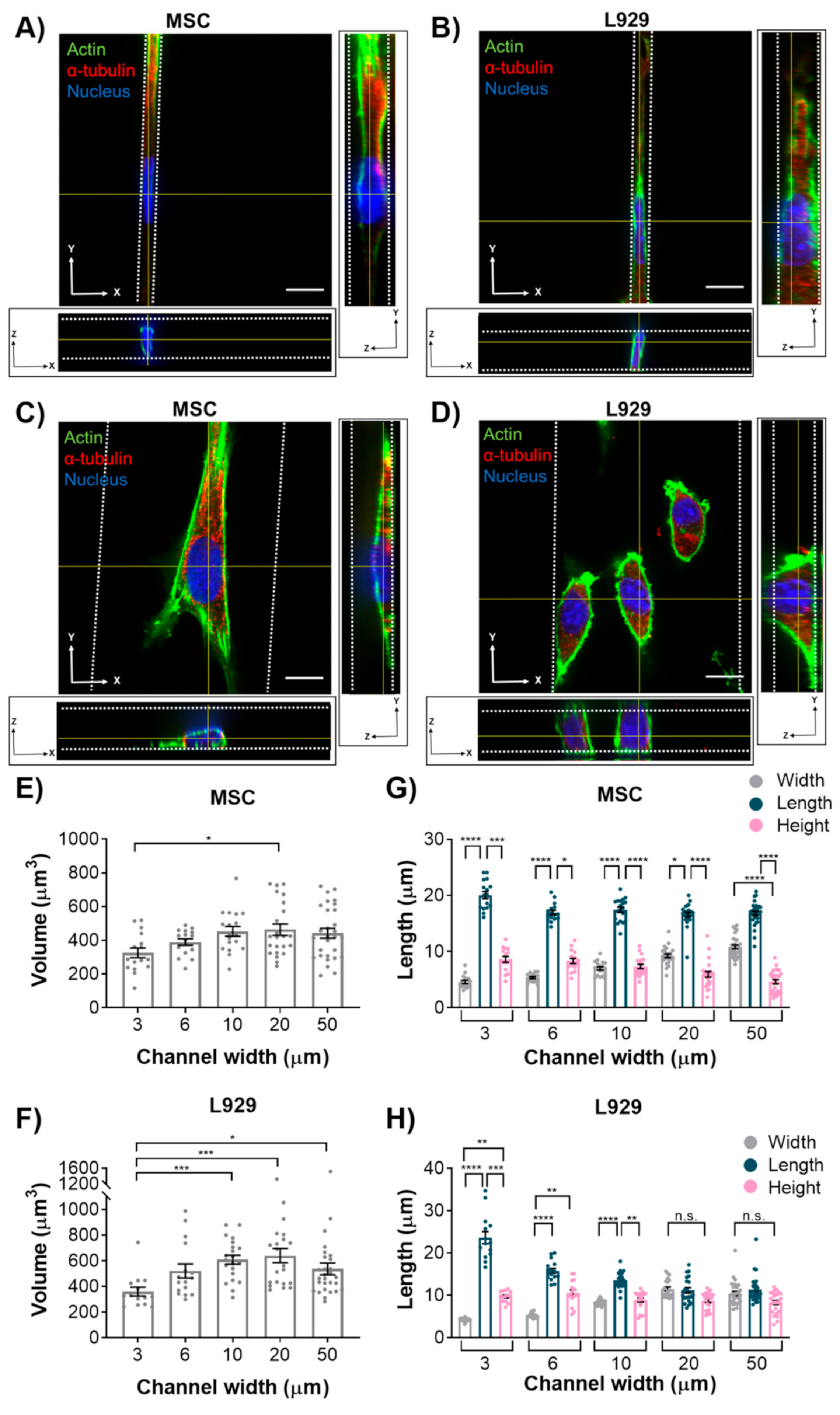
We initially hypothesized that nuclear volume would not change with increasing confinement, leading us to a second hypothesis that the height of the MSC nuclei would increase with increasing confinement, whereas the height of L929 nuclei would remain constant. However, interestingly, nuclei of MSCs and L929 cells decreased in volume when they experienced the highest degree of confinement. This result is consistent with other studies that have reported changes in nuclear volume in response to mechanical microenvironments. For example, nuclear volume was altered by nanotopography, potentially mediated by focal adhesion arrangement [
31]. When confined to microwells, nuclear volume decreased with decreasing microwell volume [
32]. Additionally, we observed a large range in the values of nucleus volume, which were on par with what others have reported [
12,
29]. When MSCs were confined to microwells in a previous study, their nuclear volume was less than observed here [
32]; however, the microwells in that study were approximately 10 kPa in elastic modulus, whereas the microchannels herein were approximately 1 MPa in elastic modulus. This suggests that there may be a complex interplay between physical confinement and matrix stiffness in regulating nuclear volume.
MSC nuclei were anisotropic in all channel widths, while L929 cell nuclei were anisotropic in the narrowest channels and isotropic in the widest channels. MSCs exhibited more defined actin stress fibers than L929 cells in wide channels. Given the flattened morphology of MSCs in wide channels, we hypothesize that actin stress fibers compress the MSC nucleus in wide channels, thereby reducing nuclear height and increasing nuclear width with decreasing confinement. MSCs increased nuclear height in response to increasing confinement, as nuclear width was forced to decrease. Interestingly, the MSC nuclear length remained constant in all channel widths. Conversely, L929 cells maintained the same nuclear height in all channel widths. We suggest that this was due to minimal stress fiber formation and minimal cytoskeletal tension in L929 cells, which were more rounded. To compensate, L929 cells increased nuclear length in response to increasing confinement, as nuclear width was forced to decrease. It is interesting to note that L929 cells and MSCs have different origins—from mouse adipose and human bone marrow, respectively. It was previously observed that nuclear stiffness scales with tissue stiffness due to differences in lamin A content [
28]. Although adipose and bone marrow are both soft tissues, MSCs have higher lamin A content than cells derived from fat or lung tissue [
28]. Chromatin condensation may also affect nuclear deformation in confinement. It has been shown that chromatin decondensation leads to a more-isotropic deformation of fibroblast nuclei [
8]. Additionally, it has been shown that polarized, elongated cells (similar to MSCs in wide channels) generate higher stress on the nucleus, have higher lamin A/C levels, and have less dynamic heterochromatin foci [
33]. Conversely, isotropic cells contain a rounder nucleus, lower lamin A/C levels, and more dynamic heterochromatin foci [
33]. These chromatin dynamics may ultimately influence nuclear deformation.
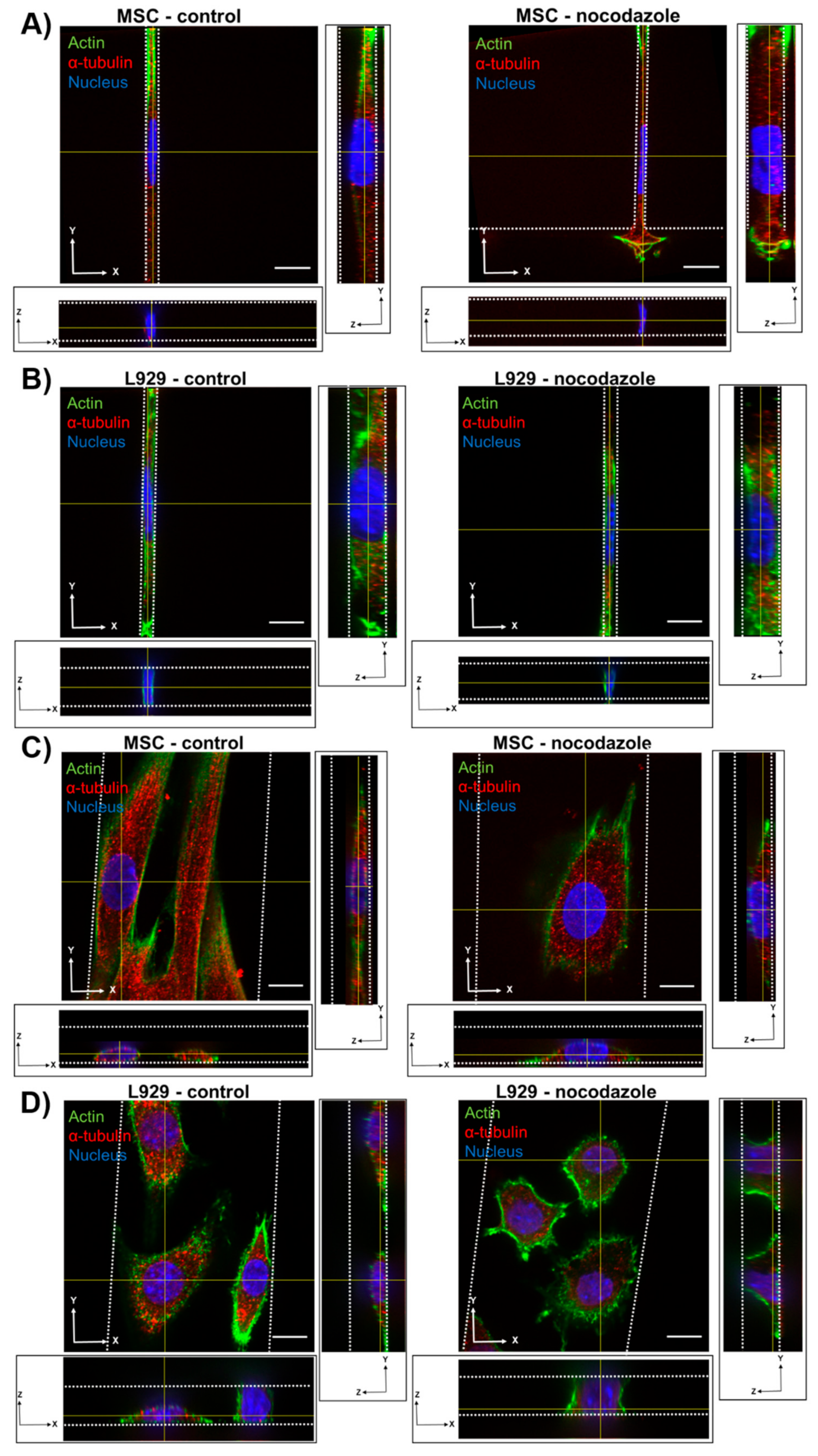
We observed the inhibition of microtubule polymerization via nocodazole increased MSC nuclear volume within wider 10- and 50-μm channels. Nocodazole has been shown to increase nuclear volume and increase major axis strain in 2D [
8,
29]. In two dimensions, microtubules seem to exert a force on the nucleus, as evidenced by a crescent-shaped nucleus around the microtubule organizing center (MTOC) when lamin A is knocked out [
33], and by decreases in nuclear envelope fluctuations after nocodazole treatment [
34]. Furthermore, inhibition of microtubule polymerization in the long term decreases elasticity of fibroblasts in 2D [
35]. Similarly, we showed previously that nocodazole did not affect MSC migration speed in confinement, but reduced cell speed in wide channels [
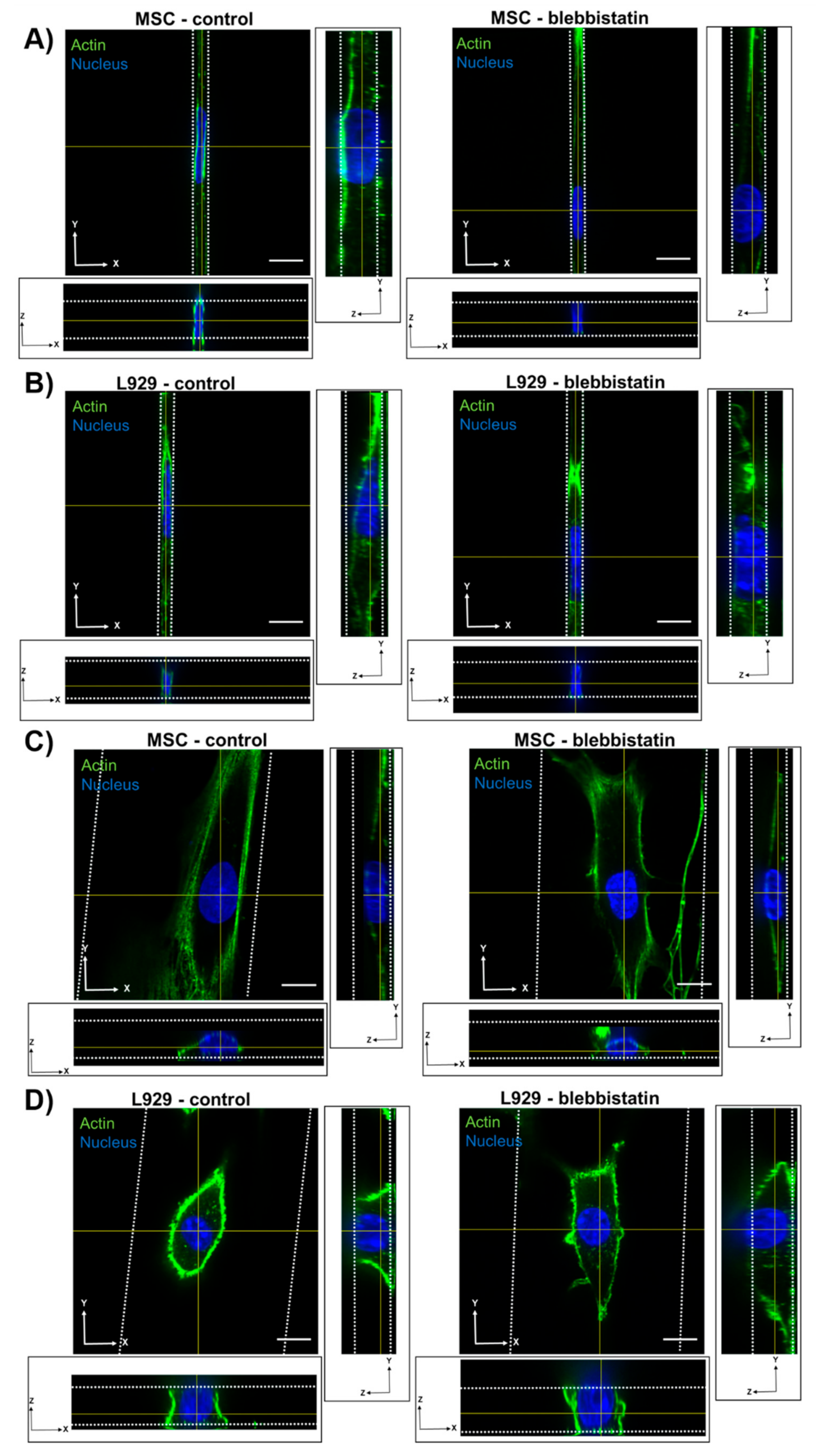
13]. It is possible that microtubule polymerization has a reduced role in regulating nuclear morphology in confinement compared with 2D microenvironments. Nocodazole did not, however, affect the nuclear volume or dimensions of L929 cells in any channel width. The microtubule network in L929 cells already appeared quite diffuse in all channel widths before treatment, suggesting that microtubules in these cells contribute little to maintaining nuclear morphology even in 2D microenvironments.
We also observed that inhibition of myosin II contractility decreased the nuclear volume of MSCs in 20-μm wide channels. Blebbistatin is often used to decrease the stiffness of cortical actin [
36], and may have reduced the force on the nucleus exerted by the actin network. Inhibition of myosin II by blebbistatin has been shown to decrease the force on the nucleus and increase the levels of phosphorylated lamin A/C, which is typically associated with lamin A/C turnover and rounding nuclei [
37]. Untreated L929 cells did not appear to have many actin stress fibers in any channel width, and hence they may have a low-contractile basal state. Therefore, the addition of blebbistatin did not seem to alter the cytoskeletal structure of L929 cells. In reporting these results, we also note an inherent limitation in our methods: Confocal microscopy is a costly and time-consuming technique, so it is difficult to obtain a significantly large sample size for the quantification of nuclear 3D volume and dimensions. Future studies could work towards developing a high-throughput method of confining and imaging cells, which could enable larger sample sizes to support our conclusions.
It does not appear that the external effectors of cell contractility or microtubule polymerization are key determinants of nuclear dimensions in confinement, which leaves the question of whether internal nuclear effectors control nuclear volume in confinement. Future studies may investigate the effects of chromatin decondensation or lamina disruption on nuclear volume, as these have both been demonstrated to decrease or increase, respectively, the anisotropic nucleus deformation in response to force [
8]. Additionally, the LINC complex has been demonstrated to control the homeostatic position of the nucleus [
38]. It is possible that the LINC complex may have a role in nuclear volume regulation or nuclear deformation in response to force. Internal nuclear effectors ought to be investigated for their role in nuclear deformation in three dimensions.