Synergy Between Agroecological Practices and Arbuscular Mycorrhizal Fungi
Abstract
1. Introduction
2. Materials and Methods
3. Results
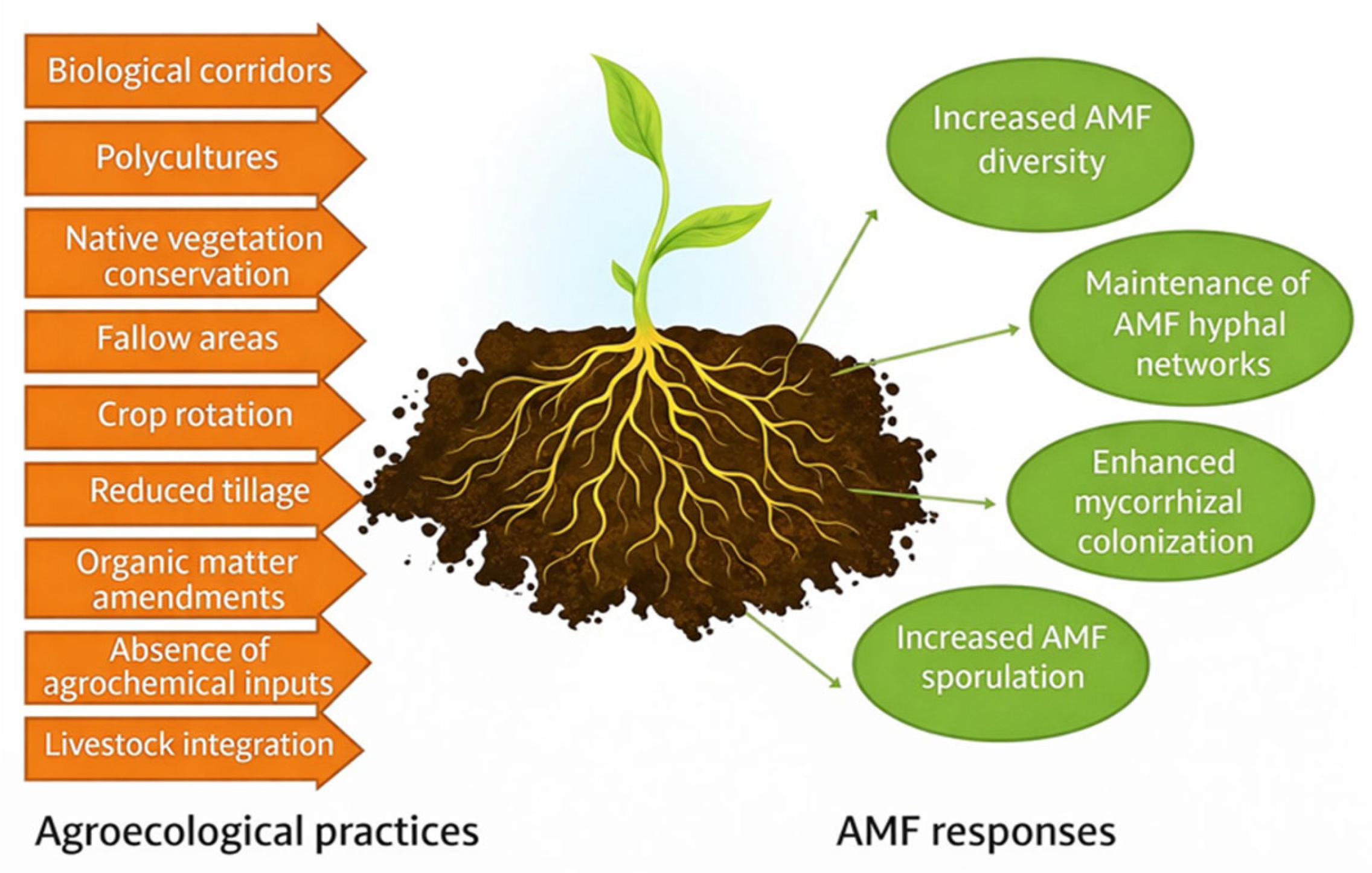
3.1. Agroecological Practices Benefits
3.1.1. Cover Crops
3.1.2. Crop Rotation
3.1.3. Tillage
3.1.4. Organic Matter Management
3.1.5. Crop Diversity
3.2. Impact of Agroecological Practices on Crop Resilience
3.2.1. Resilience to Pests and Diseases
3.2.2. Enhancing Plant Nutrition
3.2.3. Long-Term Sustainability
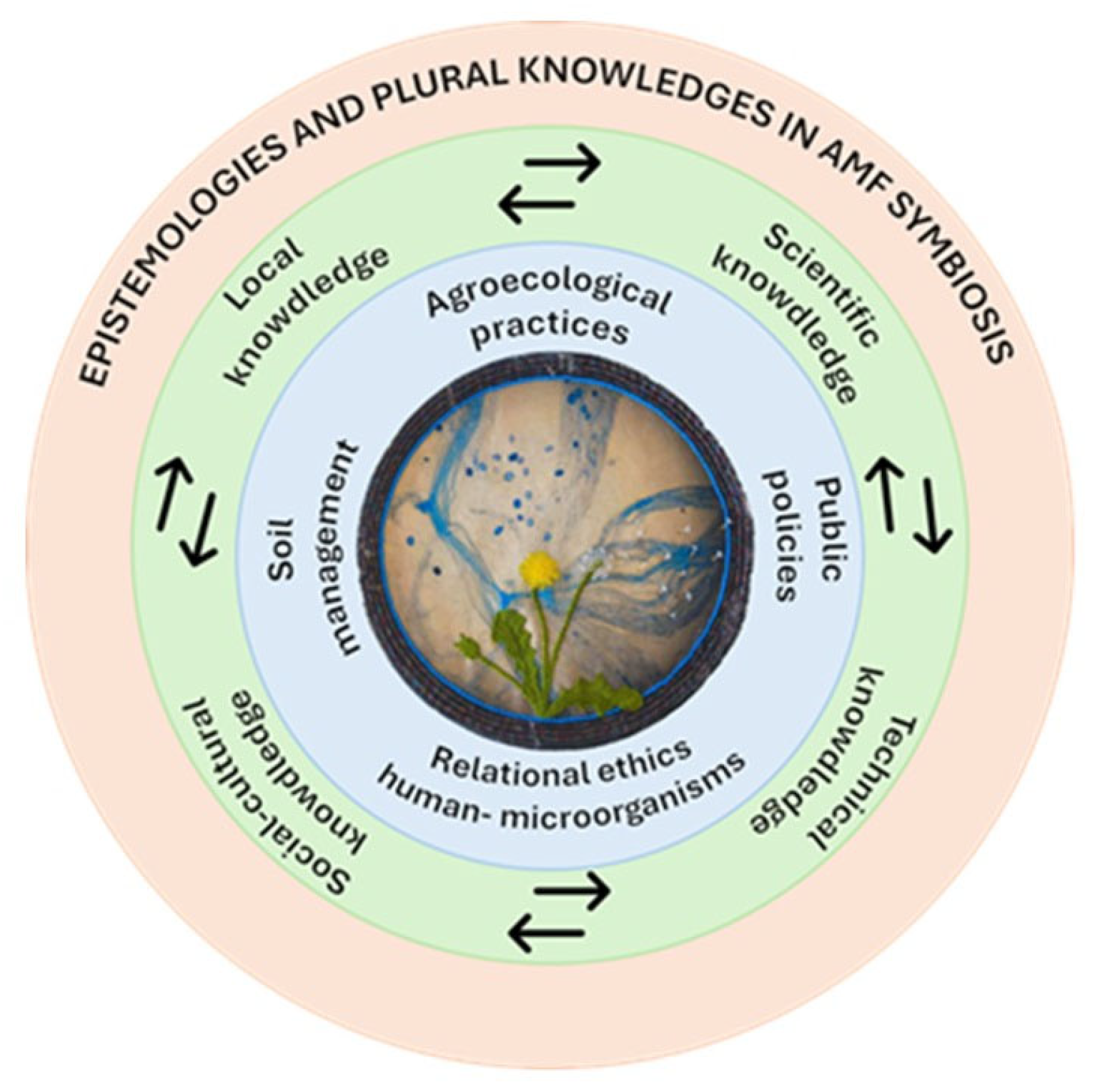
3.3. Beyond Functionality: AMF as a Sociocultural Web for Rethinking Living Soils
3.3.1. Soil as a Political Territory: Regulation, Power, and Epistemic Exclusion
3.3.2. Life-Based Economies and Microbial Capitalization: Economics Disputes Around the Soil
3.3.3. Situated Agroecology: Structural Conditions and Epistemic Disputes
3.3.4. Co-Habiting with the “Invisible Society”: Towards Relational Ethics with AMF
4. Discussion
| AMF 7 Species | Crops | Experimental Design and Soil Conditions | Results | Reported Benefits | Reference |
|---|---|---|---|---|---|
| Glomus intraradices 2 | Triticum aestivum | Field experiment evaluating the potential application of PGPR 1 (Biotol); no specific soil characteristics were reported. | Grain yield increased by 41% (Sakha 93) and 29% (Gemmeza 9); proline increased by 38.6%, salicylic acid by 192.6% (Sakha 93); proline by 37.5%, salicylic acid by 135.4% (Gemmeza 9); improvements in NPK, chlorophyll, and grain protein | Improved growth, yield, and salt stress tolerance. | [119] |
| Glomus clarum, Gigaspora margarita, Acaulospora sp. (mixed inoculum) | Coffea arabica cv. Obatã | Greenhouse pot experiment with AMF 7 inoculation and increasing Cu or Zn concentrations over 30 weeks. Sterilized Typic Hapludox soil (pH 5.5, P 28 mg/kg) was used, with Cu/Zn applied as sulfates and AMF 7 inoculum from the IAC collection. | Shoot biomass increased up to 20-fold; root colonization up to 44%; shoot phosphorus increased threefold; copper in leaves decreased by 70%. | Increased P and K uptake; decreased Cu in leaves; reduced oxidative stress; roots retained more metals; modified amino acid profiles. | [120] |
| Glomus intraradices 2 | Lactuca sativa | Greenhouse pot experiment under well-watered conditions without exogenous ABA 3 application; no specific soil characteristics reported. | Root colonization under drought increased to 70%; shoot biomass increased by 34%; root hydraulic conductivity improved by 520%; full recovery after drought. | Increased root hydraulic conductivity. | [121] |
| Glomus intraradices 2 (Ri), Gigaspora margarita (Gm) | Phaseolus vulgaris | Greenhouse cultivation using fine sandy loam soil, comparing wild-type mycorrhizal (myc+) plants with non-mycorrhizal mutants (myc−). | About 50% of the increase in stomatal conductance was due to mycorrhizal colonization. | Improved plant and soil water relations; root and soil colonization. | [12] |
| Rhizophagus intraradices, Glomus aggregatum, Glomus viscosum, Claroideoglomus etunicatum, Claroideoglomus claroideum (mixed inoculum) | Solanum lycopersicum | Field trial in an industrial tomato farm in Italy, testing seven treatments with AMF 7 and PGPR 1 strains (Pseudomonas sp. 19Fv1T and P. fluorescens C7), applied alone or in combination under reduced fertilization. The soil was clay loam (40% silt, 28% clay, 32% sand), with pH 8.2 and 1.5% organic matter, managed under drip irrigation. | Fruit weight increased by 35%, marketable fruits by 160%, dry matter by 100%, citric acid by 17.7%; nitrate decreased by 50%. | Enhanced root colonization, flowering, fruit size and quality; PGPR 1 increased sugars, reduced nitrate; synergistic effect on flavor balance. | [122] |
| Glomus irregulare | Olea europaea var. Haouzia | Greenhouse experiment with AMF 7 inoculation followed by inoculation with Verticillium dahliae to simulate biotic stress. Experimental conditions were not fully detailed. | Disease symptoms decreased compared to non-inoculated controls. | Likely induction of plant defense pathways; priming effect. | [123] |
| Rhizophagus aggregatus, R. intraradices, Claroideoglomus etunicatum, Endogone mosseae, Funneliformis caledonium, Gigaspora margarita (commercial mix– Mycoflor) | Capsicum annuum var. Roberta F1 | Three-year organic field trial (2016–2018) in southeastern Poland using a factorial design with and without AMF 7 and drip irrigation, applied at the transplant stage. Conducted on organic soil (pH 6.4–6.7) with moderate P, K, and Ca levels; rainfall ranged from 290 to 385 mm (May–September) and summer temperatures were 1.7–3.1 °C above the 1951–2010 average. | Marketable yield increased by 33.7%, total fruits by 16.8%; BER 4 incidence decreased; earliness and vegetative growth increased. | Improved water and nutrient uptake; earlier flowering and fruiting; decreased blossom-end rot. | [124] |
| Funneliformis mossea | Zea mays | Greenhouse root-bag experiment in a lead–zinc mining area in Yunnan, China, comparing AMF 7-inoculated and control plants grown for 60 days in three soil types: wasteland, farmland, and slopeland. Soils were autoclaved, with Cd concentrations of 25.3, 6.7, and 4.3 mg/kg, respectively, pH 6.2–6.8, and organic matter ranging from 3.1 to 34.0 g/kg. | Biomass increased by 1313% (farmland), 320% (slopeland), 10% (wasteland); cadmium uptake in shoots increased by 1250% and 170%. | Improved root architecture; increased organic acid exudation; decreased Cd trans-location; Cd speciation shifted toward oxidized forms. | [125] |
| Acaulospora mellea ZZ | Sorghum bicolor | Greenhouse experiment using acidic paddy soil contaminated with Cd, Pb, and Zn, applying a factorial design with or without AMF 7 and with or without bio-or sulfidized nano-zero valent iron (B-nZVI 6 or S-nZVI 6) at 50–1000 mg/kg. The soil had pH 5.0, contained 2.6 mg/kg Cd, 1796 mg/kg Pb, 1603 mg/kg Zn, and 25.8 g/kg organic matter; no fertilizers were added. Plants were grown in pots for 60 days. | Cadmium, lead, and zinc in roots decreased by 52%, 55%, and 33%; available Cd decreased by 40%; Zn uptake in roots increased. | Decreased heavy metal bio-availability; synergy with nZVI 6; reduced phytotoxicity and oxidative stress. | [126] |
| Rhizophagus fascic-ulatus, Rhizophagus aggregatum | Casuarina obesa | Greenhouse pot experiment over four months evaluating the effects of AMF 7 alone or co-inoculated with PGPR 1 (Pan-toea agglomerans, Bacillus sp.) under three NaCl salinity levels (0, 150, 300 mM). Plants were grown in sterilized sandy soil from Senegal with pH 7.78, 1.09% organic matter, 3.9 mg/kg total phosphorus, and electrical conductivity of 218.4 µS/cm, maintained at 30 °C. | Survival reached 100% with co-inoculation; biomass increased by 76.6%; chlorophyll increased by 51.6%. | Increased mycorrhization, nutrient uptake, salt exclusion, chlorophyll and proline levels; potential antioxidant and gene induction. | [127] |
| Rhizoglomus intraradices, Funneliformis mosseae, Claroideoglomus claroideum | Cicer arietinum | Greenhouse pot experiment using two chickpea genotypes (HC 3: tolerant, C 235: sensitive) exposed to 25 mg/kg of either As(V) or As(III), with or without AMF 7 inoculation. Plants were grown in a sterile 1:1 sand/loam mix with pH ~7.4, without fertilizer, and harvested 50–60 days after sowing. | Biomass increased by 48%; relative water content, chlorophyll, and NPK uptake increased; arsenic uptake decreased by 40%; root colonization increased by 50%. | Increased chlorophyll, relative water content, antioxidant activity and nutrient uptake; decreased arsenic and membrane damage. | [128] |
| Glomus intraradices 2 (BEG141), now Rhizophagus intraradices | Vitis berlandieri × Vitis riparia | Split-root greenhouse experiment using SO4 grapevine cuttings, inoculated with AMF 7 alone or co-/post-inoculated with the nematode Xiphinema index under controlled conditions. Plants were grown in a sterilized substrate composed of ter-ragreen® and clay–loam soil with defined physicochemical properties. | Gall number and nematode population decreased by 64% and 50%. | Activation of local and systemic defense genes in grapevine; AMF 7-induced priming. | [129] |
| Glomus mosseae | Glycine max, Lens culinaris | Greenhouse pot trial conducted over 12 weeks with five replicates, comparing AMF 7-inoculated plants to sievate controls under Zn and Ni applications at 0, 1, 3, and 5 g/kg. Plants were grown in sterile sand at 70% field capacity using quarter-strength Hoagland’s solution minus Zn and KH2SO4, under 25–29 °C and a 16 h photoperiod, without drainage. | Zinc and nickel uptake increased significantly; shoot biomass increased; deficiency symptoms decreased. | Increased Zn and Ni uptake; higher shoot biomass; reduced root biomass; AMF 7 remained effective under metal stress. | [130] |
| Glomus versiforme, Rhizophagus intraradices | Lonicera japonica | Greenhouse pot experiment with a randomized block design comparing AMF 7-inoculated and control plants under three cadmium treatments (0, 10, 20 μg/g) over four months. The substrate was autoclaved loamy soil with pH 6.85, 1.65% organic matter, and 52 μg/g available phosphorus. Cadmium was applied as CdCl2, and plants were grown at 28/22 °C (day/night) with 60% water holding capacity across five replicates. | Biomass increased by 625% (Ri) and 444% (Gv); shoot Cd decreased by 69% and 76%; phosphorus increased by 15%; antioxidant enzyme activities increased. | Increased biomass and P uptake; reduced Cd in shoots; enhanced antioxidant activity phytochelatin synthesis. | [131] |
| Glomus sp. | Lycopersicon escu-lentum var. PKM-1 | Greenhouse pot experiment testing AMF 7 and Fusarium oxysporum f.sp. lycopersici applied individually or in combination (pre-, post-, or simultaneous inoculation). Plants were grown in sterilized soil placed in 30 cm earthen pots, with the pathogen introduced using a 5% maize–sand medium. The experiment was conducted under greenhouse conditions at 25–30 °C. | Yield increased by 106%; disease incidence decreased by 75%; dry weight increased by 94%; NPK and chlorophyll increased. | Suppressed F. oxysporum; improved nutrient uptake, chlorophyll, biomass; defense induced by AMF 7. | [132] |
| Glomus sp. | Capsicum annuum | Greenhouse experiment assessing the effects of AMF 7 inoculation on plants challenged with Pythium aphanidermatum (damping-off pathogen), with GC-MS analysis of root and leaf tissues. Plants were grown in pots filled with sterile soil; no specific soil physicochemical properties were reported. GC-MS was performed after 72 h of methanol extraction. | Pathogen impact reduced and defense-related metabolites increased. | Biosynthesis of antifungal metabolites in leaves and roots; systemic resistance induced by AMF 7. | [133] |
| Glomus intraradices 2 | Lycopersicon escu-lentum cv. Platense | Greenhouse experiment with six treatments combining presence or absence of AMF 7 and Nacobbus aberrans, applied either at transplanting or later. Plants were grown in sterile sandy loam soil with pH 5.2, 2.58% organic matter, and 16.08 mg/kg phosphorus, under controlled conditions (24 °C, 10 h photoperiod) without fertilization. | Nematode population decreased by 58.3% with simultaneous inoculation and 63% with pre-inoculation. | Decreased gall formation and nematode reproduction; increased colonization under infestation. | [134] |
| Rhizophagus intraradices (BGC-BG09) | Lycium barbarum | Greenhouse pot experiment evaluating four treatments: control, AMF 7 only, Fusarium solani only, and AMF 7 + F. solani co-inoculation. Seedlings were grown in sterilized soil under controlled greenhouse conditions; specific soil properties were not reported. | Plant height increased by 24.8%; lignin by 141.6%, flavonoids by 44.6%; chitinase by 36%, glucanase by 58%; salicylic acid by 17.7%, jasmonic acid by 31.6%. | Activation of phenylpropanoid pathway; increased lignin, flavonoids, SA, JA, and defense proteins. | [86] |
| Funneliformis mosseae | Zea mays L. | Greenhouse pot experiment with four treatments, where drought stress was imposed after 7 days of growth. Plants were grown in 0.3 kg of sterilized soil per pot at 20–26 °C and 65–90% relative humidity. AMF 7 was applied at 10% of the substrate weight, and drought conditions were maintained at 35% field water capacity. | Biomass increased by 42.7%; chlorophyll by 13.4%, sugars by 56.2%, microbial biomass carbon by 71.8%, soil organic matter by 85.8%. | Increased root length and antioxidants; improved osmotic adjustment, soil nutrients, microbial biomass, and drought resilience. | [82] |
| Glomus intraradices 2 (BEG 141) | Zea mays (cv. Hongdan No. 897) | Greenhouse mesocosm experiment with a factorial design testing control, biochar (B), AMF 7 (M), and combined treatments (BM) under three cadmium levels (0, 3, 6 mg/kg) over 100 days. The substrate was loamy soil with pH 7.6, 1.26% organic matter, and 0.072 mg/kg Cd, placed in 5 kg pots without added fertilizers. Plants were maintained at 60–70% water holding capacity under natural light conditions. | Biomass increased by 79.1%; antioxidant enzyme activities increased; Cd in plant tissues decreased by up to 76%. | Decreased oxidative damage; improved root health and Cd stabilization; AMF 7 and biochar had additive effects. | [135] |
| Claroideoglomus etunicatum, Glomus versiforme, Funneliformis mosseae (alone and in combinations) | Astragalus adsur-gens | Greenhouse pot experiment with plants naturally infected by Erysiphe pisi (powdery mildew), assessing the effects of single, dual, and triple AMF 7 inoculations over a 12-week period. The substrate was a sterilized mix of 10% soil and 90% sand with pH 6.2, containing 6.6 mg/kg phosphorus, 120 mg/kg nitrogen, and 40 mg/kg potassium, supplemented with a modified Long Ashton solution lacking phosphorus. | Biomass increased by 55–125%; peroxidase activity increased; mildew severity increased. | Improved growth despite increased mildew severity; increased POD 10 activity; oxidative stress indicators elevated. | [136] |
| Acaulospora maa-rowe, Glomus lep-totichum (UAS-DAMF5, UAS- DAMF9), native and standard AMF 7 consortia | Saccharum offici-narum var. CO86032 | Field experiment in a Striga-infested sugarcane field in Karnataka, India, using a factorial design to evaluate AMF 7 inoculation combined with 0–100% of the recommended herbicide dose (RDH). Conducted on loamy soil with a high native Striga seedbank; atrazine was applied pre-emergence and 2,4-D post-emergence. No detailed soil physicochemical data were reported. | Striga emergence decreased by 96%; shoot height increased by 77%, tillers by 80%, cane girth by 123%. | Decreased Striga via strigolactone suppression; enhanced growth and nutrient uptake; native AMF 7 more effective. | [137] |
| Fourteen indigenous AMF 7 species (e.g., Glomus, Septoglomus, Funneliformis, Rhizophagus, Claroideoglomus) | Hordeum vulgare cvs. Atlante, Atomo and Con-certo | Two-year field cultivation study under Mediterranean climate conditions, with AMF 7 inoculation and no application of organic or chemical fertilizers, nor weed, pest, or pathogen control. Conducted on low-phosphorus soils: clay loam in 2020 and silty clay loam in 2021, within a rotation system that included clover and faba bean. | Grain yield increased by 64–134%; phosphorus in grain increased by 24–42%. | Increased colonization and arbuscules; activation of P and N pathways; stable genotype response. | [20] |
| Rhizophagus intraradices, Funneliformis mosseae (individual and combined) | Solanum lycopersicum cv. Platense | Greenhouse pot trial with tomato seedlings pre-inoculated with AMF 7 for 45 days, then challenged with 300 s-stage juveniles (J2) of Nacobbus aberrans. Plants were analyzed at 4, 8, and 12 days post-inoculation; soil was a sterile 3:1 mix of soil and sand, with pH 6.6, 4.06% organic matter, 0.22% nitrogen, and 116.7 ppm phosphorus. | Nematode penetration decreased by 20–27%. | Reduced nematode penetration; induced resistance and altered root exudates. | [107] |
| Funneliformis mosseae, Gigaspora gi-gantea, Septoglomus constrictum, Scutellospora pellucida | Solanum tuberosum cvs. Agria and Innovator | Field trial conducted in the Netherlands using sandy soil to evaluate four AMF 7 strains versus a control across four replicates, with NPK fertilization. Assessment included litterbag, NIRS, and SIR techniques. The soil had 39% sand, 1% organic matter, pH 7.2, and CEC of 154 cmolc/kg; 200 kg/ha nitrogen was applied, no phosphorus was added, and plant density was 5555 plants/ha during the 2019 season. | Agria yield increased by 5.6–8%; Innovator yield decreased by 5.6%, tubers by 11.8%. | Increased soil respiration, AMF 7-specific activity and tuber production in cultivar Agria. | [138] |
| Glomus mosseae, Glomus intraradices 2; Native mix (Glomus, Acaulospora, Scutellospora, etc.) | Trifolium alexandrinum cv. Tigri; Zea mays cv. Eleo-nora | Field trials conducted under Mediterranean low-input rotation (from Trifolium alexandrinum to Zea mays), testing single, mixed exotic, and native AMF 7 inoculations. The soil was sandy loam with pH 8.4 and 1.5% organic matter, under a Mediterranean climate, with no fertilizer applied and low native mycorrhizal potential. | Shoot biomass increased by 49–99%; seed yield by 100–134%; shoot phosphorus by 78–129%; maize grain yield by 70%. | Enhanced N and P uptake; synergy with rhizobia; long-term persistence of AMF 7 strains. | [22] |
| Glomus mosseae and Glomus intraradices 2 (Rhizophagus intraradices) | Lycopersicon escu-lentum cv. Ear-lymech | Split-root greenhouse experiment using a sterilized 9:1 sand–soil mix, where one half of the root system was inoculated with AMF 7 and the other with Phytophthora parasitica, under controlled environmental conditions. Plants were grown in a growth chamber with a low-phosphorus nutrient regime based on one-quarter strength Hoagland’s solution. | Strong disease reduction with G. mosseae; partial protection with G. intraradices 2. | Local and systemic resistance induced; lytic enzymes active in non-colonized roots. | [76] |
| Multiple genera: Acaulospora, Glomus, Ambispora, Archaeospora, Den-tiscutata, Gigaspora, Paraglomus, Rhizophagus, Scutellospora | Coffea arabica | Field study in Minas Gerais, Brazil, comparing agroecological and conventional coffee systems with native forest, with seasonal sampling during flowering, grain filling, and harvest. Conducted on acidic oxisols (pH ~4.3–5.2), low fertility, ~1040 m altitude; agroecological farms incorporated leguminous cover crops and low-input fertilization. | Shannon diversity index was 11% higher under agroecological management; 96.3% of agroecological samples clustered with forest vs. 44.4% of conventional samples. | Increased AMF 7 diversity and OTU 9 richness; greater similarity to forest; agroecological practices promoted diverse AMF 7 communities. | [139] |
| Rhizophagus irregularis (EEZ 58) | Lactuca sativa, Solanum lycopersicum | Greenhouse pot experiment using a 2:2:1 mix of loamy soil, sand, and vermiculite to evaluate plant responses to three irrigation regimes: well-watered (100% field capacity), moderate drought (75%), and severe drought (55%) over 8 weeks. The loamy soil, sourced from Dúrcal, Spain, had pH 8.2, 1.8% organic matter, 2.5 g/kg nitrogen, 6.2 mg/kg phosphorus, and 13.2 g/kg potassium. The substrate was sterilized by steaming, and plants were grown at 19–25 °C with a 16/8 h photoperiod and 50–60% relative humidity. | Lettuce biomass increased by 60% (well-watered), 39% (moderate drought), 26% (severe drought); photosystem II efficiency increased by 16%; ABA 3 and SL 8 increased. | Increased ABA 3, SL 8s, and related gene expression; improved growth and photosynthesis under drought. | [79] |
| Glomus mosseae, Glomus intraradices 2 | Helianthus annuus | Greenhouse pot trial using chrome mine tailing soil to assess the effects of AMF 7 inoculation and sewage sludge application (20 or 30 g/kg) over a 3-month period. The soil had pH 7.9–8.5, bulk density of 1.7 g/cm3, and high levels of Cr, Fe, and Al. Sewage sludge, sourced from a wastewater treatment plant, was rich in nitrogen, phosphorus, and zinc. Plants were grown under greenhouse conditions at 23–30 °C. | Chromium uptake increased by 225%, copper by 270%, zinc by 260%, manganese by 108%; shoot biomass increased by 124%. | Enhanced metal uptake and biomass; sludge improved growth but reduced colonization at high doses. | [140] |
| Rhizophagus intraradices | Glycine max | Greenhouse pot experiment with a factorial design testing the effects of AMF 7 inoculation, Macrophomina phaseolina infection, and nitrogen fertilization (0 and 92 kg/ha urea). The substrate was a sterilized mix of soil, sand, and perlite (7:3:2) based on a Typic Argiudoll with pH 6.9, 17.4 g/kg organic matter, and 34.7 mg/kg available phosphorus. Plants were grown under temperatures ranging from 25 to 37 °C. | Disease severity decreased by 49% with AMF 7 + nitrogen; shoot biomass increased by ~40%; pod number and chlorophyll increased. | Decreased pathogen load and root rot severity; increased biomass, chlorophyll and pod number. | [141] |
| Funneliformis mosseae | Sorghum bicolor cv. Hunnigreen | Greenhouse microcosm experiment using a dual-compartment design to study AMF 7 inoculation versus control under progressive drought stress starting at week 10, focusing on common mycorrhizal network (CMN 5) dynamics. The substrate was sterilized loessial sandy soil with pH 7.7, 7.9 g/kg organic matter, 0.97 g/kg nitrogen, 3.05 mg/kg available phosphorus, and 62.7 mg/kg potassium, with 2.5 L of soil per compartment. | Biomass increased by 70%; specific leaf area by 47%; lifespan increased by up to 70%; arbuscule integrity maintained longer. | Increased shoot/root biomass and lifespan; better survival linked to early AMF 7 connection. | [142] |
| Glomus microaggregatum, Funneliformis geosporum, Claroideoglomus etunicatum, Funneliformis mosseae, Rhizophagus intraradices, Glomus claroideum (commercial mix) | Zea mays, Triticum aestivum | Field trials conducted over two seasons in Peshawar, Pakistan, testing 10 treatment combinations of AMF 7, Bacillus sp. PIS7, and rock phosphate (RP). The soil was calcareous silty clay with pH 7.83, 0.9% organic matter, low available phosphorus (2.8 mg/kg), 15.3% lime, and electrical conductivity of 0.18 dS/m, under a semi-arid climate with temperatures ranging from 30 to 37 °C. | Maize yield increased by 103%, phosphorus uptake by 6×; wheat yield increased by 80%, phosphorus by 12×. | Higher colonization, AMF 7 spores, P solubilization, organic matter; long-term soil improvement. | [143] |
| Funneliformis mosseae (isolate BGC YN05) | Zelkova serrata | Greenhouse pot experiment exposing 96 seedlings to four NaCl concentrations (0, 50, 100, 150 mM) with or without AMF 7 inoculation over 12 weeks. The substrate was an autoclaved 1:1:1 mix of soil, sand, and vermiculite (pH 7.15), containing 0.03% nitrogen, 570 mg/kg phosphorus, and 15.18 g/kg potassium. Plants were grown at 18–35 °C with 40–80% relative humidity | Leaf biomass increased by 20%, root biomass by 14%; photosynthesis, chlorophyll, and nutrient content increased; oxidative stress reduced. | Alleviated osmotic stress, reduced oxidative stress, improved ion balance and nutrient uptake. | [144] |
| Funneliformis mosseae | Phragmites austra-lis | Greenhouse pot experiment evaluating plant response to three water regimes (50%, 70%, and 100% field capacity) and four levels of TiO2 nanoparticles (0, 100, 200, 500 mg/kg) over a 60-day period. The substrate was a sterilized 1:1 vermiculite/sand mix, pH ~7, with no added fertilizers. TiO2NPs (60 nm anatase) were applied, and plants were grown at 25 °C under a controlled light/dark cycle. | Biomass increased by 65%; relative water content by 8%; nitrogen by 1.3×, phosphorus by 1.1×; titanium uptake and root retention increased. | Increased nutrient uptake, chlorophyll, antioxidants, and stress resilience; Ti retained in roots. | [145] |
| Glomus aggregatum, G. intraradices 2, G. elunicatum, G. versiforme (1:1:1:1 mix) | Medicago sativa | Greenhouse pot experiment with a factorial design testing the effects of AMF 7, biochar (3%), and cadmium (20 mg/kg) over a 60-day period. The substrate was a sand/soil mix (1:2) with pH 4.38, Cd added as CdCl2, and rice straw-derived biochar (450–550 °C). Plants were grown under 20–35 °C and maintained at 60% water holding capacity. | Shoot biomass increased with AMF 7 and biochar; shoot cadmium decreased by up to 80%; nitrogen and phosphorus uptake increased with AMF 7; potassium and calcium increased with biochar. | Increased N, P, K, Ca uptake; decreased Cd in shoots through hyphal retention and pH shift; biochar more effective on Cd. | [146] |
| Claroideoglomus etunicatum and Rhizophagus intraradices | Zea mays | Greenhouse pot experiment using sterilized soil obtained from an agricultural field; no additional soil properties reported. | Biomass increased by 222%; molybdenum in roots increased by 80%; trans-location factor reduced to 0.09; net photosynthesis, chlorophyll a, carotenoids, proline, salicylic acid, and nutrient uptake increased significantly. | Decreased trans-location of heavy metals such as arsenic and molybdenum to shoots. | [147] |
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ABA | Abscisic Acid |
| AMF | Arbuscular Mycorrhizal Fungi |
| APX | Ascorbate Peroxidase |
| Ca | Calcium |
| Cd2+ | Cadmium |
| Cu | Copper |
| Fe | Iron |
| Gs | Stomatal Conductance |
| K | Potassium |
| LeNCED1 | 9-cis-epoxycarotenoid dioxygenase 1 |
| MDA | Malondialdehyde |
| Mg | Magnesium |
| N | Nitrogen |
| Na+ | Sodium |
| NPR1 | Nonexpressor of Pathogenesis-Related Genes 1 |
| P | Phosphorus |
| POD | Peroxidase |
| Pn | Net Photosynthesis |
| PR | Pathogenesis-Related Proteins |
| PR-1 | Pathogenesis-Related Protein 1 |
| PR-2 | Pathogenesis-Related Protein 2 |
| PR-5 | Pathogenesis-Related Protein 5 |
| SOD | Superoxide Dismutase |
| SA | Salicylic Acid |
| SLs | Strigolactones |
| SlCCD7 | Carotenoid Cleavage Dioxygenase 7 |
| STS | Science and Technology Studies |
| UNAM-ENES | Universidad Nacional Autónoma de México–Escuela Nacional de Estudios Superiores |
| Tr | Transpiration |
| Zn | Zinc |
| WRKY | WRKY Transcription Factors |
References
- Philippot, L.; Raaijmakers, J.M.; Lemanceau, P.; Van Der Putten, W.H. Going back to the roots: The microbial ecology of the rhizosphere. Nat. Rev. Microbiol. 2013, 11, 789–799. [Google Scholar] [CrossRef] [PubMed]
- Mendes, R.; Garbeva, P.; Raaijmakers, J.M. The rhizosphere microbiome: Significance of plant-beneficial, plant-pathogenic, and human-pathogenic microorganisms. FEMS Microbiol. Rev. 2013, 37, 634–663. [Google Scholar] [CrossRef] [PubMed]
- Bardgett, R.D.; Van Der Putten, W.H. Belowground biodiversity and ecosystem functioning. Nature 2014, 515, 505–511. [Google Scholar] [CrossRef] [PubMed]
- Thirkell, T.; Charters, M.; Elliott, A.; Sait, S.; Field, K. Are mycorrhizal fungi our sustainable saviours? Considerations for achieving food security. J. Ecol. 2017, 105, 921–929. [Google Scholar] [CrossRef]
- Smith, S.E.; Read, D.J. Mycorrhizal Symbiosis, 3rd ed.; Academic Press: London, UK, 2008. [Google Scholar]
- Brundrett, M.; Tedersoo, L. Evolutionary history of mycorrhizal symbioses and global host plant diversity. New Phytol. 2018, 220, 1108–1115. [Google Scholar] [CrossRef]
- Parniske, M. Arbuscular mycorrhiza: The mother of plant root endosymbioses. Nat. Rev. Microbiol. 2008, 6, 763–775. [Google Scholar] [CrossRef]
- Rich, M.K.; Nouri, E.; Courty, P.E.; Reinhardt, D. Diet of arbuscular mycorrhizal fungi: Bread and butter? Trends Plant Sci. 2017, 22, 652–660. [Google Scholar] [CrossRef]
- Azcón-Aguilar, C.; Barea, J. Arbuscular mycorrhizas and biological control of soil-borne plant pathogens: An overview of the mechanisms involved. Mycorrhiza 1997, 6, 457–464. [Google Scholar] [CrossRef]
- Smith, S.E.; Smith, F.A. Fresh perspectives on the roles of arbuscular mycorrhizal fungi in plant nutrition and growth. Mycologia 2012, 104, 1–13. [Google Scholar] [CrossRef]
- Cavagnaro, T.; Bender, S.; Asghari, H.; Van Der Heijden, M. The role of arbuscular mycorrhizas in reducing soil nutrient loss. Trends Plant Sci. 2015, 20, 283–290. [Google Scholar] [CrossRef]
- Augé, R.M. Arbuscular mycorrhizae and soil/plant water relations. Can. J. Soil Sci. 2004, 84, 373–381. [Google Scholar] [CrossRef]
- Cameron, D.D.; Neal, A.L.; Van Wees, S.C.M.; Ton, J. Mycorrhiza-induced resistance: More than the sum of its parts? Trends Plant Sci. 2013, 18, 539–545. [Google Scholar] [CrossRef] [PubMed]
- Aguilera, P.; Ortiz, N.; Becerra, N.; Turrini, A.; Gaínza-Cortés, F.; Silva-Flores, P.; Aguilar-Paredes, A.; Romero, J.K.; Jorquera-Fontena, E.; Mora, M.L.; et al. Application of arbuscular mycorrhizal fungi in vineyards: Water and biotic stress under a climate change scenario—A new challenge for Chilean grapevine crops. Front. Microbiol. 2022, 13, 826571. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.; Arato, M.; Borghi, L.; Nouri, E.; Reinhardt, D. Beneficial services of arbuscular mycorrhizal fungi—From ecology to application. Front. Plant Sci. 2018, 9, 1–14. [Google Scholar] [CrossRef]
- Genre, A.; Lanfranco, L.; Perotto, S.; Bonfante, P. Unique and common traits in mycorrhizal symbioses. Nat. Rev. Microbiol. 2020, 18, 649–660. [Google Scholar] [CrossRef]
- van Der Heijden, M.G.A.; Martin, F.M.; Selosse, M.A.; Sanders, I.R. Mycorrhizal ecology and evolution: The past, the present, and the future. New Phytol. 2015, 205, 1406–1423. [Google Scholar] [CrossRef]
- Gianinazzi, S.; Gollotte, A.; Binet, M.N.; Van Tuinen, D.; Redecker, D.; Wipf, D. Agroecology: The key role of arbuscular mycorrhizas in ecosystem services. Mycorrhiza 2010, 20, 519–530. [Google Scholar] [CrossRef]
- Turrini, A.; Avio, L.; Giovannetti, M.; Agnolucci, M. Functional complementarity of arbuscular mycorrhizal fungi and associated microbiota: The challenge of translational research. Front. Plant Sci. 2018, 9, 10–13. [Google Scholar] [CrossRef]
- Marrassini, V.; Ercoli, L.; Aguilar-Paredes, A.V.; Pellegrino, E. Positive response to inoculation with indigenous arbuscular mycorrhizal fungi as modulated by barley genotype. Agron. Sustain. Dev. 2025, 45, 21. [Google Scholar] [CrossRef]
- Berruti, A.; Lumini, E.; Balestrini, R.; Bianciotto, V. Arbuscular mycorrhizal fungi as natural biominiers: Learning from past successes. Front. Microbiol. 2016, 6, 1559. [Google Scholar] [CrossRef]
- Pellegrino, E.; Bedini, S.; Avio, L.; Bonari, E.; Giovannetti, M. Field inoculation effectiveness of native and exotic arbuscular mycorrhizal fungi in a Mediterranean agricultural soil. Soil Biol. Biochem. 2011, 43, 367–376. [Google Scholar] [CrossRef]
- Rouphael, Y.; Franken, P.; Schneider, C.; Schwarz, D.; Giovannetti, M.; Agnolucci, M.; Di Pascale, S.; Bonini, P.; Colla, G. Arbuscular mycorrhizal fungi act as biostimulants in horticultural crops. Sci. Hortic. 2015, 196, 91–108. [Google Scholar] [CrossRef]
- Verbruggen, E.; Toby Kiers, E.; Bakelaar, P.N.; Röling, W.F. Mycorrhizal fungal establishment in agricultural soils: Factors determining inoculation success. New Phytol. 2012, 196, 1070–1079. [Google Scholar] [CrossRef] [PubMed]
- Balestrini, R.; Salvioli, A.; Dal Molin, A.; Novero, M.; Gabelli, G.; Paparelli, E.; Marroni, F.; Bonfante, P. Impact of an arbuscular mycorrhizal fungus versus a mixed microbial inoculum on transcriptome reprogramming of grapevine roots. Mycorrhiza 2017, 27, 417–430. [Google Scholar] [CrossRef]
- Hart, M.M.; Antunes, P.M.; Chaudhary, V.B.; Abbott, L.K. Fungal inoculants in the field: Is the reward greater than the risk? Funct. Ecol. 2018, 32, 126–135. [Google Scholar] [CrossRef]
- Crossay, T.; Majorel, C.; Redecker, D.; Gensous, S.; Medevielle, V.; Durrieu, G.; Cavaloc, Y.; Amir, H. Is a mixture of arbuscular mycorrhizal fungi better for plant growth than single-species inoculants? Mycorrhiza 2019, 29, 325–339. [Google Scholar] [CrossRef]
- Stavi, I.; Lal, R. Achieving zero net land degradation: Challenges and opportunities. J. Arid Environ. 2015, 112, 44–51. [Google Scholar] [CrossRef]
- Hartman, K.; Van Der Heijden, M.G.A.; Wittwer, R.A.; Banerjee, S.; Walser, J.C.; Schlaeppi, K. Cropping practices manipulate abundance patterns of root and soil microbiome members paving the way to smart farming. Microbiome 2018, 6, 14. [Google Scholar] [CrossRef]
- Springmann, M.; Clark, M.; Mason-D’Croz, D.; Wiebe, K.; Bodirsky, B.L.; Lassaletta, L.; De Vries, W.; Vermeulen, S.J.; Herrero, M.; Carlson, K.M.; et al. Options for keeping the food system within environmental limits. Nature 2018, 562, 519–525. [Google Scholar] [CrossRef]
- Ciccolini, V.; Ercoli, L.; Davison, J.; Vasar, M.; Öpik, M.; Pellegrino, E. Land-use intensity and host plant simultaneously shape arbuscular mycorrhizal fungal communities in a Mediterranean drained peatland. FEMS Microbiol. Ecol. 2016, 92, fiw186. [Google Scholar] [CrossRef]
- Giovannini, L.; Palla, M.; Agnolucci, M.; Avio, L.; Sbrana, C.; Turrini, A.; Giovannetti, M. Arbuscular mycorrhizal fungi and associated microbiota as plant biostimulants: Research strategies for selecting the best-performing inocula. Agronomy 2020, 10, 1. [Google Scholar] [CrossRef]
- Delgado-Baquerizo, M.; Reich, P.B.; Trivedi, C.; Eldridge, D.J.; Abades, S.; Alfaro, F.D.; Singh, B.K. Multiple elements of soil biodiversity drive ecosystem functions across biomes. Nat. Ecol. Evol. 2020, 4, 210–220. [Google Scholar] [CrossRef] [PubMed]
- Oehl, F.; Laczko, E.; Oberholzer, H.; Jansa, J.; Egli, S. Diversity and biogeography of arbuscular mycorrhizal fungi in agricultural soils. Biol. Fertil. Soils 2017, 53, 777–797. [Google Scholar] [CrossRef]
- Verbruggen, E.; Kiers, E.T. Evolutionary ecology of mycorrhizal functional diversity in agricultural systems. Evol. Appl. 2010, 3, 547–560. [Google Scholar] [CrossRef] [PubMed]
- Verbruggen, E.; Röling, W.F.M.; Gamper, H.A.; Kowalchuk, G.A.; Verhoef, H.A.; Van Der Heijden, M.G.A. Positive effects of organic farming on below-ground mutualists: Large-scale comparison of mycorrhizal fungal communities in agricultural soils. New Phytol. 2010, 186, 968–979. [Google Scholar] [CrossRef]
- Bedini, S.; Avio, L.; Sbrana, C.; Turrini, A.; Migliorini, P.; Vazzana, C.; Giovannetti, M. Mycorrhizal activity and diversity in a long-term organic Mediterranean agroecosystem. Biol. Fertil. Soils 2013, 49, 781–790. [Google Scholar] [CrossRef]
- Säle, V.; Aguilera, P.; Laczko, E.; Mäder, P.; Berner, A.; Zihlmann, U.; Van Der Heijden, M.; Oehl, F. Impact of conservation tillage and organic farming on the diversity of arbuscular mycorrhizal fungi. Soil Biol. Biochem. 2015, 84, 38–52. [Google Scholar] [CrossRef]
- Aguilar-Paredes, A.; Valdés, G.; Nuti, M. Ecosystem functions of microbial consortia in sustainable agriculture. Agronomy 2020, 10, 1902. [Google Scholar] [CrossRef]
- Altieri, M.; Farrell, J.; Hecht, S.; Liebman, M.; Magdoff, F.; Murphy, B.; Norgaard, R.; Sikor, T. Agroecology; CRC Press: Boca Raton, FL, USA, 2018. [Google Scholar]
- Bender, S.F.; Wagg, C.; Van Der Heijden, M.G. An underground revolution: Biodiversity and soil ecological engineering for agricultural sustainability. Trends Ecol. Evol. 2016, 29, 600–607. [Google Scholar] [CrossRef]
- Eyhorn, F.; Muller, A.; Reganold, J.P.; Frison, E.; Herren, H.R.; Seufert, V.; Smith, P. Sustainability in global agriculture driven by organic farming. Nat. Sustain. 2019, 2, 253–255. [Google Scholar] [CrossRef]
- Isbell, F.; Craven, D.; Connolly, J.; Loreau, M.; Schmid, B.; Beierkuhnlein, C.; Bezemer, T.; Kreyling, J.; Lanta, V.; Manning, P.; et al. Biodiversity increases the resistance of ecosystem productivity to climate extremes. Nature 2015, 526, 574–577. [Google Scholar] [CrossRef]
- Wall, D.H.; Nielsen, U.N.; Six, J. Soil biodiversity and human health. Nature 2015, 528, 69–76. [Google Scholar] [CrossRef] [PubMed]
- Ebbisa, A. Arbuscular mycorrhizal fungi in optimizing nutrient bioavailability and reducing agrochemicals for maintaining sustainable agroecosystems. In Arbuscular Mycorrhizal Fungi in Agriculture—New Insights; IntechOpen: London, UK, 2022. [Google Scholar]
- Aguilar-Paredes, A.; Turrini, A.; Avio, L.; Stuardo, C.; Velásquez, A.; Becerra, J.; Giovannetti, M.; Seeger, M. Agricultural management influences the diversity of arbuscular mycorrhizal fungi in vineyards from Chilean Mediterranean ecosystems. J. Soil Sci. Plant Nutr. 2024, 24, 6099–6112. [Google Scholar] [CrossRef]
- Silva-Flores, P.; Argüelles-Moyao, A.; Aguilar-Paredes, A.; Simões-Calaça, F.J.; Duchicela, J.; Fernández, N.; Furtado, A.N.M.; Guerra-Sierra, B.; Lovera, M.; Marín, C.; et al. Mycorrhizal science outreach: Scope of action and available resources in the face of global change. Plants People Planet 2021, 3, 506–522. [Google Scholar] [CrossRef]
- Puig De La Bellacasa, M. The Spirit of Soil: For a More-Than-Human Community; Tercero Incluido Publishing: Madrid, Spain, 2023; p. 169. [Google Scholar]
- Tsing, A.L. The Mushroom at the End of the World: On the Possibility of Life in Capitalist Ruins; Caja Negra: Buenos Aires, Argentina, 2015; p. 448. [Google Scholar]
- Lyons, K. Vital Decomposition: Soils, Forests, and Life Proposals; Universidad del Rosario Press: Bogotá, Colombia, 2021; p. 247. [Google Scholar]
- Blanco-Canqui, H.; Shaver, T.; Lindquist, J.; Shapiro, C.; Elmore, R.; Francis, C.; Hergert, G. Cover crops and ecosystem services: Insights from studies in temperate soils. Agron. J. 2015, 107, 2449–2474. [Google Scholar] [CrossRef]
- Winter, S.; Bauer, T.; Strauss, P.; Kratschmer, S.; Zaller, J.G.; Batáry, P. Effects of vegetation management intensity on biodiversity and ecosystem services in vineyards: A meta-analysis. J. Appl. Ecol. 2018, 55, 2484–2495. [Google Scholar] [CrossRef]
- García, L.; Celette, F.; Gary, C.; Ripoche, A.; Valdés-Gómez, H.; Metay, A. Management of service crops for the provision of ecosystem services in vineyards: A review. Agric. Ecosyst. Environ. 2018, 251, 158–170. [Google Scholar] [CrossRef]
- Capó-Bauçà, S.; Marqués, A.; Llopis-Vidal, N.; Bota, J.; Baraza, E. Long-term establishment of natural green cover provides agroecosystem services by improving soil quality in a Mediterranean vineyard. Ecol. Eng. 2019, 127, 285–291. [Google Scholar] [CrossRef]
- Vukicevich, E.; Lowery, T.; Bowen, P.; Úrbez-Torres, J.R.; Hart, M.M. Cover crops to increase soil microbial diversity and mitigate decline in perennial agriculture: A review. Agron. Sustain. Dev. 2016, 36, 287–299. [Google Scholar] [CrossRef]
- Quintarelli, V.; Radicetti, E.; Allevato, E.; Stazi, S.R.; Haider, G.; Abideen, Z.; Bibi, S.; Jamal, A.; Mancinelli, R. Cover Crops for Sustainable Cropping Systems: A Review. Agriculture 2022, 12, 2076. [Google Scholar] [CrossRef]
- Williams, A.; Birt, H.W.G.; Raghavendra, A.; Dennis, P.G. Cropping system diversification influences soil microbial diversity in subtropical dryland farming systems. Microb. Ecol. 2023, 85, 1473–1484. [Google Scholar] [CrossRef] [PubMed]
- Fall, A.F.; Nakabonge, G.; Ssekandi, J.; Founoune-Mboup, H.; Apori, S.O.; Ndiaye, A.; Badji, A.; Ngom, K. Roles of arbuscular mycorrhizal fungi on soil fertility: Contribution to the improvement of physical, chemical, and biological properties of the soil. Front. Fungal Biol. 2022, 3, 723892. [Google Scholar] [CrossRef] [PubMed]
- McDaniel, M.D.; Tiemann, L.K.; Grandy, A.S. Does agricultural crop diversity enhance soil microbial biomass and organic matter dynamics? A meta-analysis. Ecol. Appl. 2014, 24, 560–570. [Google Scholar] [CrossRef] [PubMed]
- Köhl, L.; Oehl, F.; Van Der Heijden, M.G.A. Agricultural practices indirectly influence plant productivity and ecosystem services through effects on soil biota. Ecol. Appl. 2014, 24, 1842–1853. [Google Scholar] [CrossRef]
- Lehman, R.M.; Cambardella, C.A.; Stott, D.E.; Acosta-Martinez, V.; Manter, D.K.; Buyer, J.S.; Maul, J.E.; Smith, J.L.; Collins, H.P.; Halvorson, J.J.; et al. Understanding and enhancing soil biological health: The solution for reversing soil degradation. Sustainability 2015, 7, 988–1027. [Google Scholar] [CrossRef]
- Patanita, M.; Campos, M.D.; Félix, M.D.R.; Carvalho, M.; Brito, I. Effect of tillage system and cover crop on maize mycorrhization and presence of Magnaporthiopsis maydis. Biology 2020, 9, 46. [Google Scholar] [CrossRef]
- Orrù, L.; Canfora, L.; Trinchera, A.; Migliore, M.; Pennelli, B.; Marcucci, A.; Farina, R.; Pinzari, F. How tillage and crop rotation change the distribution pattern of fungi. Front. Microbiol. 2021, 12, 634325. [Google Scholar] [CrossRef]
- Rebouh, N.Y.; Khugaev, C.V.; Utkina, A.O.; Isaev, K.V.; Mohamed, E.S.; Kucher, D.E. Contribution of eco-friendly agricultural practices in improving and stabilizing wheat crop yield: A review. Agronomy 2023, 13, 2400. [Google Scholar] [CrossRef]
- Chaudhary, A.; Poudyal, S.; Kaundal, A. Role of arbuscular mycorrhizal fungi in maintaining sustainable agroecosystems. Appl. Microbiol. 2025, 5, 6. [Google Scholar] [CrossRef]
- Nyamwange, M.M.; Njeru, E.M.; Mucheru-Muna, M.; Ngetich, F. Soil management practices affect arbuscular mycorrhizal fungi propagules, root colonization, and growth of rainfed maize. AIMS Agric. Food 2018, 3, 120–133. [Google Scholar] [CrossRef]
- Tian, L.; Wang, T.; Cui, S.; Li, Y.; Gui, W.; Yang, F.; Chen, J.; Dong, R.; Gu, X.; Zhao, X.; et al. Diversified cover crops and no-till enhanced soil total nitrogen and arbuscular mycorrhizal fungi diversity: A case study from the karst area of southwest China. Agriculture 2024, 14, 1103. [Google Scholar] [CrossRef]
- Tamburini, G.; Bommarco, R.; Wanger, T.C.; Kremen, C.; Van Der Heijden, M.G.A.; Liebman, M.; Hallin, S. Agricultural diversification promotes multiple ecosystem services without compromising yield. Sci. Adv. 2020, 6, eaba1715. [Google Scholar] [CrossRef] [PubMed]
- Tiemann, L.K.; Grandy, A.S.; Atkinson, E.E.; Marin-Spiotta, E.; McDaniel, M.D. Crop rotational diversity enhances belowground communities and functions in an agroecosystem. Ecol. Lett. 2015, 18, 761–771. [Google Scholar] [CrossRef] [PubMed]
- Ghimire, R.; Norton, J.B.; Stahl, P.D.; Norton, U. Soil microbial substrate properties and microbial community responses under irrigated organic and reduced tillage crop and forage systems. PLoS ONE 2014, 9, e103901. [Google Scholar] [CrossRef]
- Kaur, M.; Faizan, M.; Alam, P. Farmers’ suicide and stress resilience in agriculture through microbial interactions: A perspective analysis. Discov. Plants 2025, 2, 42. [Google Scholar] [CrossRef]
- Kumawat, K.C.; Sharma, B.; Nagpal, S.; Kumar, A.; Tiwari, S.; Nair, R.M. Plant growth-promoting rhizobacteria: Salt stress alleviators to improve crop productivity for sustainable agriculture development. Front. Plant Sci. 2023, 13, 1101862. [Google Scholar] [CrossRef]
- Pozo, M.J.; Azcón-Aguilar, C. Unraveling mycorrhiza-induced resistance. Curr. Opin. Plant Biol. 2007, 10, 393–398. [Google Scholar] [CrossRef]
- Medina, A.; Azcón, R. Effectiveness of arbuscular mycorrhizal fungi and organic amendments to improve soil quality and plant performance under stress conditions. J. Soil Sci. Plant Nutr. 2010, 10, 354–372. [Google Scholar] [CrossRef]
- Gebremeskel, K.; Birhane, E.; Habtu, S.; Haile, M.; Chanyalew, S.; Tadele, Z.; Assefa, K. Arbuscular mycorrhizal fungi improve morphological and yield performance of Eragrostis tef genotypes in Tigray, Ethiopia. Sci. Rep. 2024, 14, 29716. [Google Scholar] [CrossRef]
- Pozo, M.J.; Cordier, C.; Dumas-Gaudot, E.; Gianinazzi, S.; Barea, J.M.; Azcón-Aguilar, C. Localized versus systemic effect of arbuscular mycorrhizal fungi on defense responses to Phytophthora infection in tomato plants. J. Exp. Bot. 2002, 53, 525–534. [Google Scholar] [CrossRef]
- Pozo, M.J.; Jung, S.C.; Martínez-Medina, A.; López-Ráez, J.A.; Azcón-Aguilar, C.; Barea, J.M. Root allies: Arbuscular mycorrhizal fungi help plants to cope with biotic stresses. In Symbiotic Endophytes; Aroca, R., Ed.; Springer: Berlin/Heidelberg, Germany, 2013; pp. 289–306. [Google Scholar]
- Coşkun, F.; Alptekin, Y.; Demir, S. Effects of arbuscular mycorrhizal fungi and salicylic acid on plant growth and antioxidative enzyme activity against wilt disease in pepper. Eur. J. Plant Pathol. 2023, 165, 163–177. [Google Scholar] [CrossRef]
- Ruiz-Lozano, J.M.; Aroca, R.; Zamarreño, Á.M.; Molina, S.; Andreo-Jiménez, B.; Porcel, R.; García-Mina, J.M.; Ruyter-Spira, C.; López-Ráez, J.A. Arbuscular mycorrhizal symbiosis induces strigolactone biosynthesis under drought and improves drought tolerance in lettuce and tomato. Plant Cell Environ. 2016, 39, 441–452. [Google Scholar] [CrossRef] [PubMed]
- Guo, S.; Xia, L.; Xia, D.; Li, M.; Xu, W.; Liu, L. Enhancing plant resilience: Arbuscular mycorrhizal fungi’s role in alleviating drought stress in vegetation concrete. Front. Plant Sci. 2024, 15, 1401050. [Google Scholar] [CrossRef] [PubMed]
- Camuy-Velez, L.; Chakraborty, D.; Young, A.; Paudel, S.; Elvers, R.; Vanderhyde, M.; Walter, K.; Herzog, C.; Banerjee, S. Context-dependent contributions of arbuscular mycorrhizal fungi to host performance under global change factors. Soil Biol. Biochem. 2025, 204, 109707. [Google Scholar] [CrossRef]
- Li, J.; Zhou, L.; Chen, G.; Yao, M.; Liu, Z.; Li, X.; Yang, X.; Yang, Y.; Cai, D.; Tuerxun, Z.; et al. Arbuscular mycorrhizal fungi enhance drought resistance and alter microbial communities in maize rhizosphere soil. Environ. Technol. Innov. 2025, 37, 103947. [Google Scholar] [CrossRef]
- Nie, W.; He, Q.; Guo, H.; Zhang, W.; Ma, L.; Li, J.; Wen, D. Arbuscular mycorrhizal fungi boost crop resilience to environmental stresses. Microorganisms 2024, 12, 2448. [Google Scholar] [CrossRef]
- Ahmed, N.; Li, J.; Li, Y.; Deng, L.; Chachar, M.; Hayat, F.; Raza, A.; Umrani, J.H.; Gong, L.; Tu, P. Symbiotic synergy: How arbuscular mycorrhizal fungi enhance nutrient uptake, stress tolerance, and soil health through molecular mechanisms and hormonal regulation. IMA Fungus 2025, 16, e144989. [Google Scholar] [CrossRef]
- Wahab, A.; Muhammad, M.; Munir, A.; Abdi, G.; Zaman, W.; Ayaz, A.; Khizar, C.; Reddy, S.P.P. Role of arbuscular mycorrhizal fungi in regulating growth, enhancing productivity, and potentially influencing ecosystems under abiotic and biotic stresses. Plants 2023, 12, 3102. [Google Scholar] [CrossRef]
- Li, N.; Chen, W.; Wang, B.; Zhang, C.; Wang, Y.; Li, R.; Yan, Y.; He, J. Arbuscular mycorrhizal fungi improve the disease resistance of Lycium barbarum to root rot by activating phenylpropanoid metabolism. Front. Plant Sci. 2024, 15, 1459651. [Google Scholar]
- Puig De La Bellacasa, M.P. Matters of Care: Speculative Ethics in More-than-Human Worlds; University of Minnesota Press: Minneapolis, MN, USA, 2017; p. 280. [Google Scholar]
- Ureta, S.; Oteagui, Á.; Llona, M. The SIRSD-S or the risks of a productivist problematization of degraded soils. Rev. Geogr. Norte Gd. 2024, 88, 1–23. [Google Scholar] [CrossRef]
- Auyero, J. The Gray Zone: Collective Violence and Party Politics in Contemporary Argentina; Siglo XXI Publishers: Buenos Aires, Argentina, 2007; p. 234. [Google Scholar]
- Naciones Unidas México. Mexico Advances in Building a Regulatory Framework for Bioinputs; United Nations Mexico Office: Mexico City, Mexico, 2024. [Google Scholar]
- Biblioteca del Congreso Nacional de Chile. Law No. 21.349: Establishes Regulations on the Composition, Labeling, and Marketing of Fertilizers and Biostimulants; Biblioteca del Congreso Nacional de Chile: Santiago, Chile, 2021. [Google Scholar]
- Instituto Interamericano de Cooperación para la Agricultura (IICA); Instituto Nacional de Tecnología Agropecuaria (INTA); Empresa Brasileira de Pesquisa Agropecuaria (EMBRAPA); Instituto de Investigaciones Agropecuarias (INIA); Instituto Paraguayo de Tecnología Agraria (IPTA); Programa Cooperativo para el Desarrollo Tecnológico Agroalimentario y Agroindustrial del Cono Sur (PROCISUR). Bioinputs in the Southern Cone: Innovation from the INIAs of PROCISUR Toward Sustainable Agriculture; PROCISUR: Montevideo, Uruguay, 2022. [Google Scholar]
- Van Der Ploeg, J.D. The New Peasantries: Struggles for Autonomy and Sustainability in an Era of Empire and Globalization; Earthscan: London, UK, 2008; p. 356. [Google Scholar]
- Engel-Di Mauro, S. Ecology, Soils, and the Left: An Eco-Social Approach; Palgrave Macmillan: New York, NY, USA, 2014; p. 226. [Google Scholar]
- Gómez-Perazzoli, A.; Gazzano, M.I.; Dieguez-Cameroni, F. Agroecological family farming in Uruguay: Innovative and counter-hegemonic contributions to sustainability. Agrociencia Urug. 2024, 28, e1140. [Google Scholar]
- Norton, D.A. Native Biodiversity and Regenerative Agriculture in New Zealand; Manaaki Whenua Press: Lincoln, New Zealand, 2021; p. 84. [Google Scholar]
- García, A.; Varón, D. The Atrato River ruling: A step beyond the Green Constitution. In The Environmental Court: Environmental Justice in the Jurisprudence of the Constitutional Court of Colombia; Heinrich Böll Foundation: Bogotá, Colombia, 2019; pp. 297–318. [Google Scholar]
- Cagüeñas, D.; Galindo Orrego, M.I.; Rasmussen, S. The Atrato River and its guardians: Ecopolitical imagination to weave new rights. Rev. Colomb. Antropol. 2020, 56, 169–196. [Google Scholar] [CrossRef]
- Altieri, M.A.; Rosset, P.M. Agroecology: Science and Politics; Icaria Publishing: Barcelona, Spain, 2018; p. 208. [Google Scholar]
- Aguilar, E. Agroecology and Social Organization; Ítaca Publishing House: Mexico City, Mexico, 2022; p. 250. [Google Scholar]
- Oficina Española de Patentes y Marcas (OEPM). Biodiversity in Biotechnological Inventions: Patent Analysis and Its Relation to Genetic Resources and Traditional Knowledge; FAO Working Document; OEPM: Madrid, Spain, 2012. [Google Scholar]
- Nuti, M. The Invisibles in Agriculture; Piedra Molle Editions: Limache, Chile, 2021; p. 97. [Google Scholar]
- Kazic, D. When Plants Do What They Want; Cactus Publishing: Buenos Aires, Argentina, 2024; p. 414. [Google Scholar]
- Clavijo Ponce, N.; Pérez Martínez, M. Andean tubers and local agricultural knowledge in rural communities of Ecuador and Colombia. Cuad. Desarro. Rural. 2014, 11, 149–166. [Google Scholar]
- Koziol, L.; McKenna, T.P.; Bever, J.D. Meta-analysis reveals globally sourced commercial mycorrhizal inoculants fall short. New Phytol. 2025, 246, 821–827. [Google Scholar] [CrossRef] [PubMed]
- Basiru, S.; Mwanza, H.P.; Hijri, M. Analysis of arbuscular mycorrhizal fungal inoculant benchmarks. Microorganisms 2020, 9, 81. [Google Scholar] [CrossRef]
- Marro, N.; Lax, P.; Doucet, M.E.; Cabello, M.; Becerra, A. Mycorrhizas reduce tomato root penetration by the false root-knot nematode Nacobbus aberrans. Appl. Soil Ecol. 2017, 124, 90–96. [Google Scholar] [CrossRef]
- Wu, Y.; Chen, C.; Wang, G. Inoculation with arbuscular mycorrhizal fungi improves plant biomass and nitrogen and phosphorus nutrients: A meta-analysis. BMC Plant Biol. 2024, 24, 960. [Google Scholar] [CrossRef]
- Shiva, V.; Shiva, K. Unity Versus the 1%: Breaking Illusions, Sowing Freedom; LOM Ediciones: Santiago, Chile, 2020; p. 169. [Google Scholar]
- Altieri, M.A.; Toledo, V.M. The agroecological revolution in Latin America: Rescuing nature, ensuring food sovereignty and empowering peasants. J. Peasant Stud. 2011, 38, 587–612. [Google Scholar] [CrossRef]
- Puleo, A. Ecofeminist Keys: For Rebels Who Love the Earth and Animals; Plaza y Valdés Publishers: Madrid, Spain, 2021; p. 163. [Google Scholar]
- De Sousa Santos, B. Justice Between Knowledges: Southern Epistemologies Against Epistemicide; Morata Publishing: Madrid, Spain, 2018; p. 345. [Google Scholar]
- Viveiros De Castro, E. The Jaguar’s Gaze: Introduction to Amerindian Perspectivism; Tinta Limón Ediciones: Buenos Aires, Argentina, 2013; p. 287. [Google Scholar]
- Eschenhagen, M.L. Three axes of epistemological dialogue to approach an interpretation of the human–nature relationship. Rev. Austral Cienc. Soc. 2017, 32, 185–205. [Google Scholar] [CrossRef]
- Haraway, D.J. Staying with the Trouble: Making Kin in the Chthulucene; Consonni Publishing: Buenos Aires, Argentina, 2022; p. 365. [Google Scholar]
- Salazar, J.; Granjou, C.; Kearnes, M.; Krzywoszynska, A.; Tironi, M. Thinking with Soils: Material Politics and Social Theory; Bloomsbury Academic: London, UK, 2020; p. 217. [Google Scholar]
- Carrillo-Saucedo, S.; Gutiérrez-Castañeda, D.; Basurto-Lucio, I.; Jiménez, K.; Castro, C. Between Roots, Beings, and Mycelia: Weaving, We Know Ourselves; Agroforestry University Garden Project, UNAM-ENES Morelia: Mexico City, Mexico, 2023. [Google Scholar]
- Plumwood, V. Environmental Culture: The Ecological Crisis of Reason; Routledge: London, UK, 2002; p. 291. [Google Scholar]
- Aboul-Nasr, A.; Al-Fayoumy, M.E.; Aboul-Magd, M.; Elhabbab, A. Potential application of Glomus intraradices (AMF) and different isolates of PGPR (Biotol) to enhance yield and quality of wheat grown in calcareous soil under different salinity levels. J. Adv. Agric. Res. 2016, 21, 150–177. [Google Scholar] [CrossRef]
- Andrade, S.A.L.; Silveira, A.P.D.; Mazzafera, P. Arbuscular mycorrhiza alters metal uptake and physiological response of Coffea arabica seedlings to increasing Zn and Cu concentrations in soil. Sci. Total Environ. 2010, 408, 5381–5391. [Google Scholar] [CrossRef]
- Aroca, R.; Vernieri, P.; Ruiz-Lozano, J.M. Mycorrhizal and non-mycorrhizal tomato plants show different responses to exogenous ABA during drought stress. J. Plant Growth Regul. 2008, 27, 131–137. [Google Scholar]
- Bona, E.; Cantamessa, S.; Massa, N.; Manassero, P.; Marsano, F.; Copetta, A.; Lingua, G.; D’Agostino, G.; Gamalero, E.; Berta, G. Arbuscular mycorrhizal fungi and plant growth-promoting pseudomonads improve yield, quality, and nutritional value of tomato: A field study. Mycorrhiza 2016, 26, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Boutaj, H.; Meddich, A.; Wahbi, S.; Moukhli, A.; El Alaoui-Talibi, Z.; Douira, A.; Filali-Maltouf, A.; El Modafar, C. Effect of arbuscular mycorrhizal fungi on Verticillium wilt development in olive trees caused by Verticillium dahliae. Res. J. Biotechnol. 2019, 14, 79–88. [Google Scholar]
- Buczkowska, H.; Sałata, A. Inoculation with arbuscular mycorrhizal fungi (AMF) and plant irrigation as yield-forming factors in organic sweet pepper (Capsicum annuum L.) cultivation. Acta Sci. Pol. Hortorum Cultus 2020, 19, 125–138. [Google Scholar] [CrossRef]
- Chen, J.; Guo, J.; Li, Z.; Liang, X.; You, Y.; Li, M.; He, Y.; Zhan, F. Effects of an arbuscular mycorrhizal fungus on growth and cadmium uptake in maize grown on polluted soils in a lead–zinc mining area. Toxics 2022, 10, 359. [Google Scholar] [CrossRef]
- Cheng, P.; Zhang, S.; Wang, Q.; Feng, X.; Zhang, S.; Sun, Y.; Wang, F. Contribution of nano zero-valent iron and arbuscular mycorrhizal fungi to phytoremediation of heavy metal-contaminated soil. Nanomaterials 2021, 11, 1264. [Google Scholar] [CrossRef]
- Diagne, N.; Ndour, M.; Djighaly, P.I.; Ngom, D.; Ndong, G.; Svistoonoff, S.; Cherif-Silini, H. Effect of plant growth-promoting rhizobacteria and arbuscular mycorrhizal fungi on salt stress tolerance of Casuarina obesa. Front. Sustain. Food Syst. 2020, 4, 601004. [Google Scholar] [CrossRef]
- Garg, N.; Cheema, D.S. Comparative effectiveness of arbuscular mycorrhizal fungi in alleviating arsenic stress in chickpea (Cicer arietinum L.): Role in growth, productivity, nutrient acquisition, and soil biological functioning. Environ. Pollut. 2021, 287, 117338. [Google Scholar]
- Hao, Z.; Fayolle, L.; Van Tuinen, D.; Chatagnier, O.; Li, X.; Gianinazzi, S.; Gianinazzi-Pearson, V. Local and systemic mycorrhiza-induced protection against the ectoparasitic nematode Xiphinema index involves priming of defense gene responses in grapevine. J. Exp. Bot. 2012, 63, 3657–3672. [Google Scholar] [CrossRef]
- Jamal, A.; Ayub, N.; Usman, M.; Khan, A.G. Arbuscular mycorrhizal fungi enhance zinc and nickel uptake from contaminated soil by soybean and lentil. Int. J. Phytoremediation 2002, 4, 205–221. [Google Scholar] [CrossRef]
- Jiang, Q.Y.; Zhuo, F.; Long, S.H.; Zhao, H.D.; Yang, D.J.; Ye, Z.H.; Li, S.S.; Jing, Y.X. Can arbuscular mycorrhizal fungi reduce cadmium uptake and alleviate Cd toxicity of Lonicera japonica grown in Cd-added soils? Sci. Rep. 2016, 6, 21805. [Google Scholar] [CrossRef] [PubMed]
- Kumari, S.M.P.; Prabina, J. Protection of tomato (Lycopersicon esculentum) from wilt pathogen Fusarium oxysporum f. sp. lycopersici by arbuscular mycorrhizal fungi Glomus sp. Int. J. Curr. Microbiol. Appl. Sci. 2019, 8, 1368–1378. [Google Scholar] [CrossRef]
- Kumari, S.M.P.; Srimeena, N. Arbuscular mycorrhizal fungi-induced defense factors against damping-off disease pathogen Pythium aphanidermatum in chili (Capsicum annuum). Int. J. Curr. Microbiol. Appl. Sci. 2019, 8, 2243–2248. [Google Scholar] [CrossRef]
- Lax, P.; Becerra, A.G.; Soteras, F.; Cabello, M.; Doucet, M.E. Effect of the arbuscular mycorrhizal fungus Glomus intraradices on the false root-knot nematode Nacobbus aberrans in tomato plants. Biol. Fertil. Soils 2011, 47, 591–597. [Google Scholar] [CrossRef]
- Liu, L.; Li, J.; Yue, F.; Yan, X.; Wang, F.; Bloszies, S.; Wang, Y. Effects of arbuscular mycorrhizal inoculation and biochar amendment on maize growth, cadmium uptake, and soil cadmium speciation in Cd-contaminated soil. Chemosphere 2018, 194, 495–503. [Google Scholar] [CrossRef]
- Liu, Y.; Feng, X.; Gao, P.; Li, Y.; Christensen, M.J.; Duan, T. Arbuscular mycorrhizal fungi increased the susceptibility of Astragalus adsurgens to powdery mildew caused by Erysiphe pisi. Mycology 2018, 9, 223–232. [Google Scholar] [CrossRef]
- Manjunatha, H.P.; Nirmalnath, P.J.; Chandranath, H.T.; Ammanna, S.; Jagadeesh, K.S. Field evaluation of native arbuscular mycorrhizal fungi in the management of Striga in sugarcane (Saccharum officinarum L.). J. Pharmacogn. Phytochem. 2018, 7, 2496–2500. [Google Scholar]
- Masoero, G.; Volpato, S.; Giovannetti, G.; Nuti, M. Arbuscular mycorrhizal biofertilizer sources in the potato (Solanum tuberosum) plant show cultivar interactions on yield and litter-bag spectral features. J. Agron. Res. 2020, 2, 9–17. [Google Scholar]
- Prates Júnior, P.; Moreira, B.C.; Da Silva, M.C.S.; Veloso, T.G.R.; Stürmer, S.L.; Fernandes, R.B.A.; Mendonça, E.S.; Kasuya, M.C.M. Agroecological coffee management increases arbuscular mycorrhizal fungi diversity. PLoS ONE 2019, 14, e0209093. [Google Scholar] [CrossRef]
- Sayın, F.E.; Khalvati, M.A.; Erdinçler, A. Effects of Sewage Sludge Application and Arbuscular Mycorrhizal Fungi (G. mosseae and G. intraradices) Interactions on the Heavy Metal Phytoremediation in Chrome Mine Tailings. 2019. Available online: https://www.researchgate.net/profile/F-Ece-Sayin/publication/370265221_Effects_of_Sewage_Sludge_Application_and_Arbuscular_Mycorrhizal_Fungi_G_mosseae_and_G_intraradices_Interactions_on_the_Heavy_Metal_Phytoremediation_in_Chrome_Mine_Tailings/links/6448d (accessed on 21 November 2025).
- Spagnoletti, F.N.; Cornero, M.; Chiocchio, V.; Lavado, R.S.; Roberts, I.N. Arbuscular mycorrhiza protects soybean plants against Macrophomina phaseolina even under nitrogen fertilization. Eur. J. Plant Pathol. 2020, 156, 1097–1113. [Google Scholar] [CrossRef]
- Sun, X.; Shi, J.; Ding, G. Combined effects of arbuscular mycorrhiza and drought stress on plant growth and mortality of forage sorghum. Appl. Soil Ecol. 2017, 119, 384–391. [Google Scholar] [CrossRef]
- Wahid, F.; Fahad, S.; Danish, S.; Adnan, M.; Yue, Z.; Saud, S.; Siddiqui, M.H.; Brtnicky, M.; Hammerschmiedt, T.; Datta, R. Sustainable management with mycorrhizae and phosphate-solubilizing bacteria for enhanced phosphorus uptake in calcareous soils. Agriculture 2020, 10, 334. [Google Scholar] [CrossRef]
- Wang, J.; Zhai, L.; Ma, J.; Zhang, J.; Wang, G.G.; Liu, X.; Zhang, S.; Song, J.; Wu, Y. Comparative physiological mechanisms of arbuscular mycorrhizal fungi in mitigating salt-induced adverse effects on leaves and roots of Zelkova serrata. Mycorrhiza 2020, 30, 583–597. [Google Scholar] [CrossRef]
- Xu, Z.; Wu, Y.; Xiao, Z.; Ban, Y.; Belvett, N. Positive effects of Funneliformis mosseae inoculation on reed seedlings under water and TiO2 nanoparticle stresses. World J. Microbiol. Biotechnol. 2019, 35, 81. [Google Scholar] [CrossRef]
- Zhang, F.; Liu, M.; Li, Y.; Che, Y.; Xiao, Y. Effects of arbuscular mycorrhizal fungi, biochar, and cadmium on the yield and element uptake of Medicago sativa. Sci. Total Environ. 2019, 655, 1150–1158. [Google Scholar] [CrossRef]
- Zhang, M.; Shi, Z.; Lu, S.; Wang, F. AMF inoculation alleviates molybdenum toxicity to maize by protecting leaf performance. J. Fungi 2023, 9, 479. [Google Scholar] [CrossRef]
- Rodríguez, A.; Sanders, I.R. The role of community and population ecology in applying mycorrhizal fungi for improved food security. ISME J. 2015, 9, 1053–1061. [Google Scholar] [CrossRef]
- Sánchez De Prager, M.; Posada, R.; Velásquez, D.; Narváez, M. Basic Methodologies for Working with Arbuscular Mycorrhiza and Arbuscular Mycorrhizal Fungi; National University of Colombia Press: Palmira, Colombia, 2010. [Google Scholar]
- Cué, J.L.; Torres, A. Arbuscular mycorrhizae: Their value in the framework of agroecology. Rev. Alfa 2019, 3, 143–150. [Google Scholar]
- Castillo, C.G.; Rubio, R.; Rouanet, L.; Borie, F. Early effects of tillage and crop rotation on arbuscular mycorrhizal fungal propagules in an Ultisol. Biol. Fertil. Soils 2006, 43, 83–92. [Google Scholar] [CrossRef]
- García De León, D.; Moora, M.; Öpik, M.; Neuenkamp, L.; Gerz, M.; Jairus, T.; Vasar, M. Long-term cover cropping with clover increases arbuscular mycorrhizal fungal richness and diversity. Agric. Ecosyst. Environ. 2020, 290, 106765. [Google Scholar]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Aguilar-Paredes, A.; Valdés, G.; Aguilar-Paredes, A.; Muñoz-Arbelaez, M.; Carrillo-Saucedo, M.; Nuti, M. Synergy Between Agroecological Practices and Arbuscular Mycorrhizal Fungi. Agronomy 2026, 16, 103. https://doi.org/10.3390/agronomy16010103
Aguilar-Paredes A, Valdés G, Aguilar-Paredes A, Muñoz-Arbelaez M, Carrillo-Saucedo M, Nuti M. Synergy Between Agroecological Practices and Arbuscular Mycorrhizal Fungi. Agronomy. 2026; 16(1):103. https://doi.org/10.3390/agronomy16010103
Chicago/Turabian StyleAguilar-Paredes, Ana, Gabriela Valdés, Andrea Aguilar-Paredes, María Muñoz-Arbelaez, Margarita Carrillo-Saucedo, and Marco Nuti. 2026. "Synergy Between Agroecological Practices and Arbuscular Mycorrhizal Fungi" Agronomy 16, no. 1: 103. https://doi.org/10.3390/agronomy16010103
APA StyleAguilar-Paredes, A., Valdés, G., Aguilar-Paredes, A., Muñoz-Arbelaez, M., Carrillo-Saucedo, M., & Nuti, M. (2026). Synergy Between Agroecological Practices and Arbuscular Mycorrhizal Fungi. Agronomy, 16(1), 103. https://doi.org/10.3390/agronomy16010103











