The Content of Antioxidant Compounds and VOCs in Sorghum Grain Grown in Central and Eastern Europe
Abstract
1. Introduction
2. Materials and Methods
2.1. Conducting the Field Experiment
2.2. Testing Methods
2.2.1. Analysis of VOCs
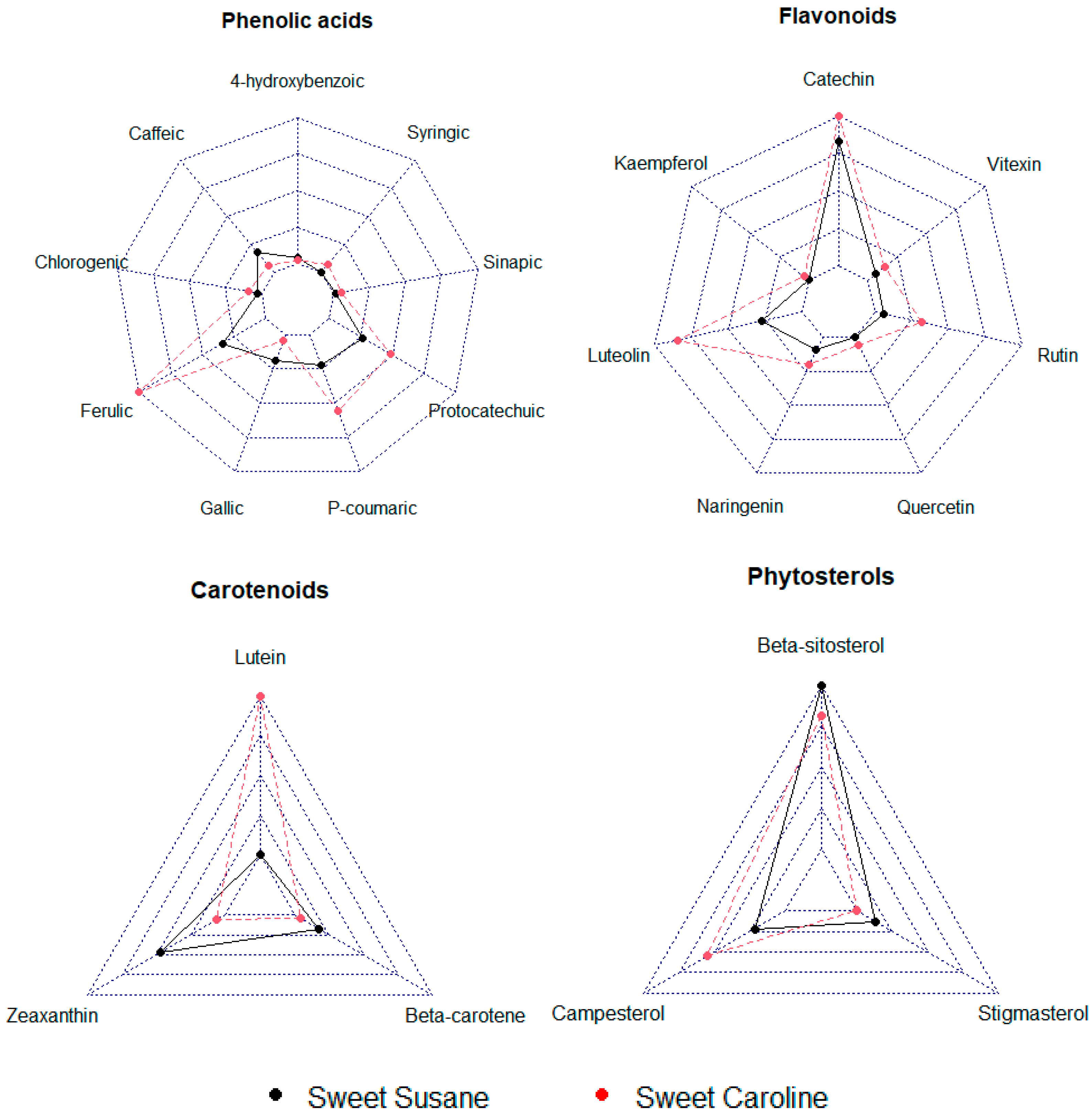
2.2.2. Analysis of Bound Phenolic Acids and Flavonoids
2.2.3. Determination of the Free Phenolic Acids (FPAs) and Antioxidant Activity (ABTS Method)
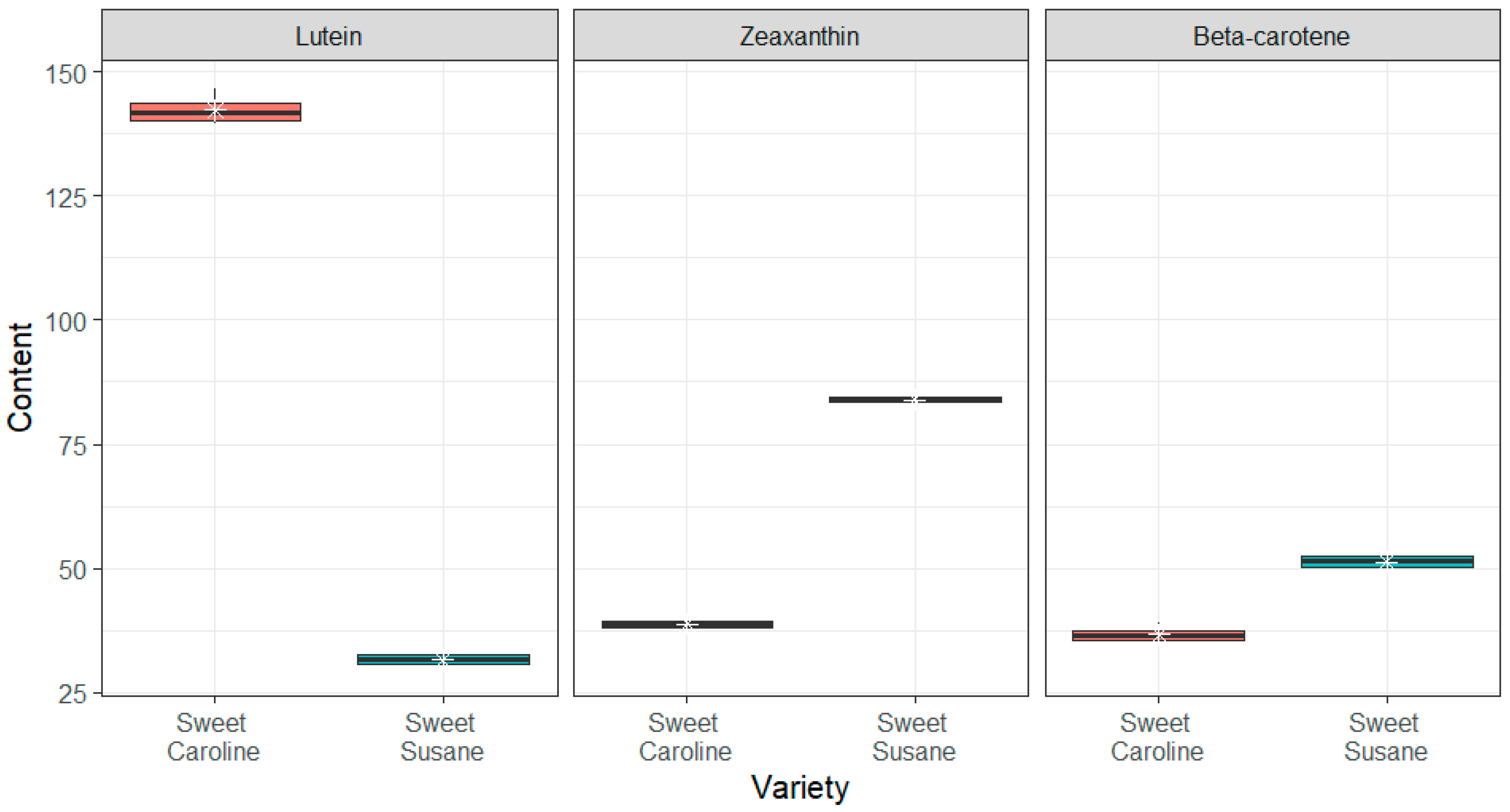
2.2.4. Analysis of Carotenoids
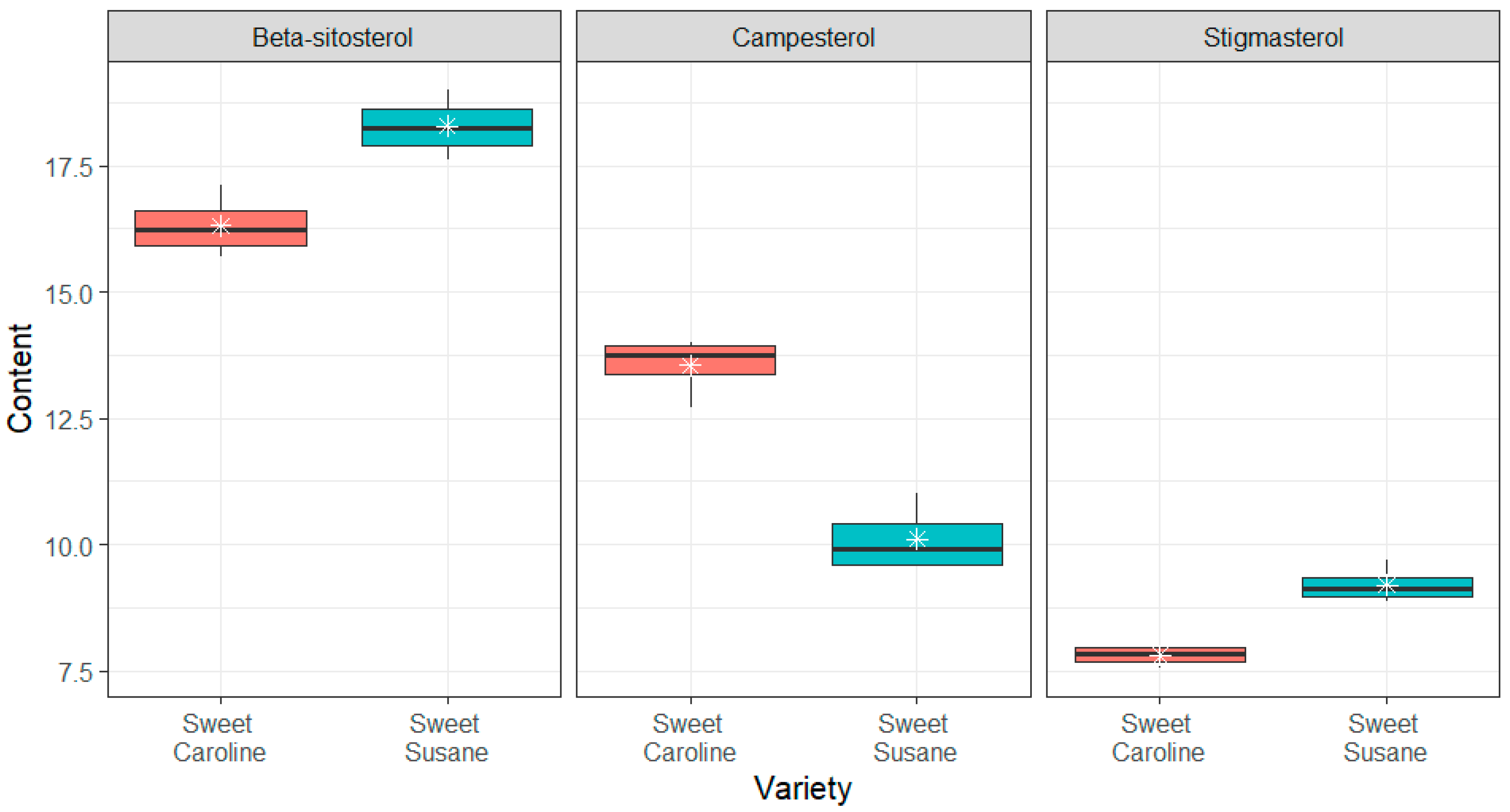
2.2.5. Analysis of Phytosterols
2.2.6. Elemental Analysis
2.2.7. Water Content Analysis
2.2.8. Fat Content Analysis [78]
2.2.9. Starch Content Analysis
2.3. Statistical Analysis
3. Results and Discussion
3.1. Weather Conditions during Cultivation
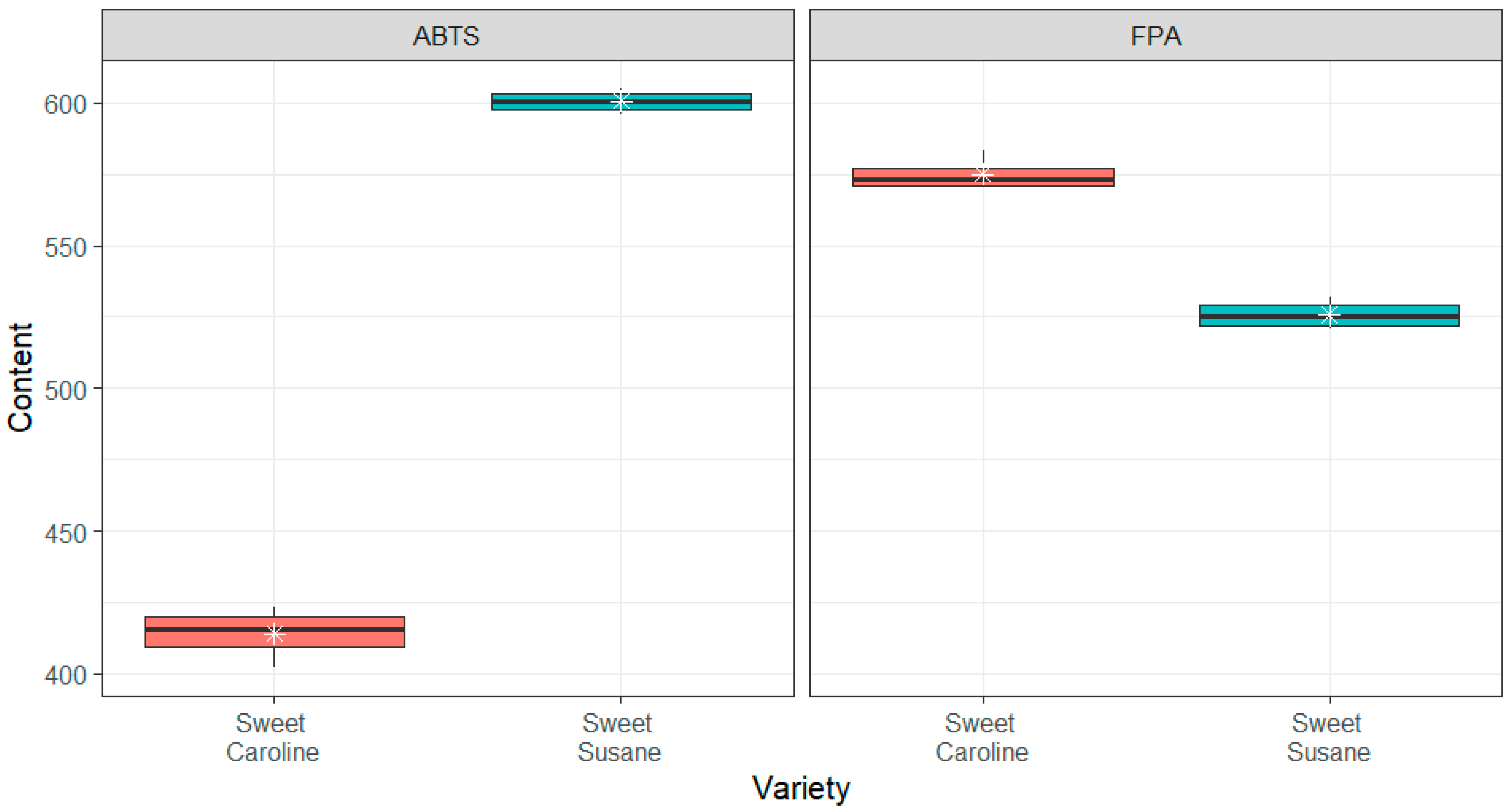
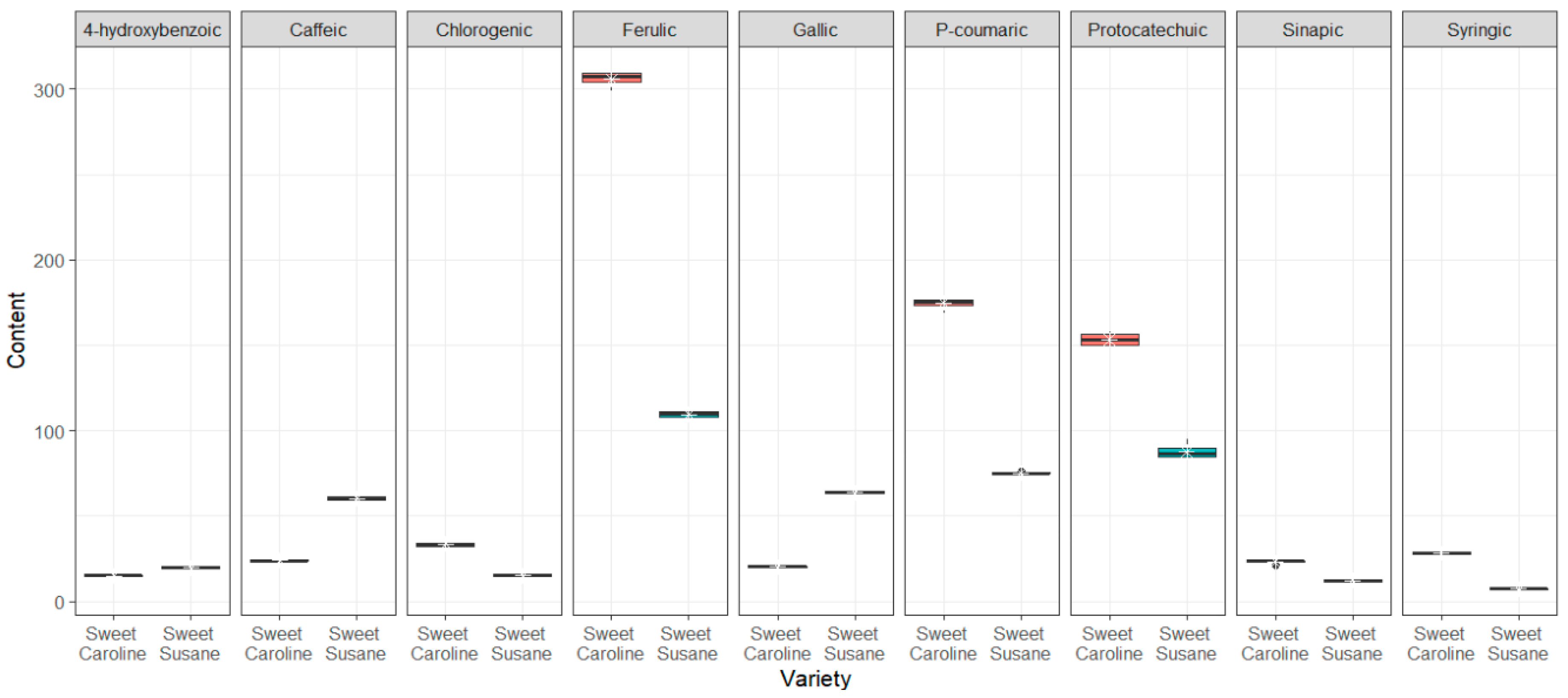
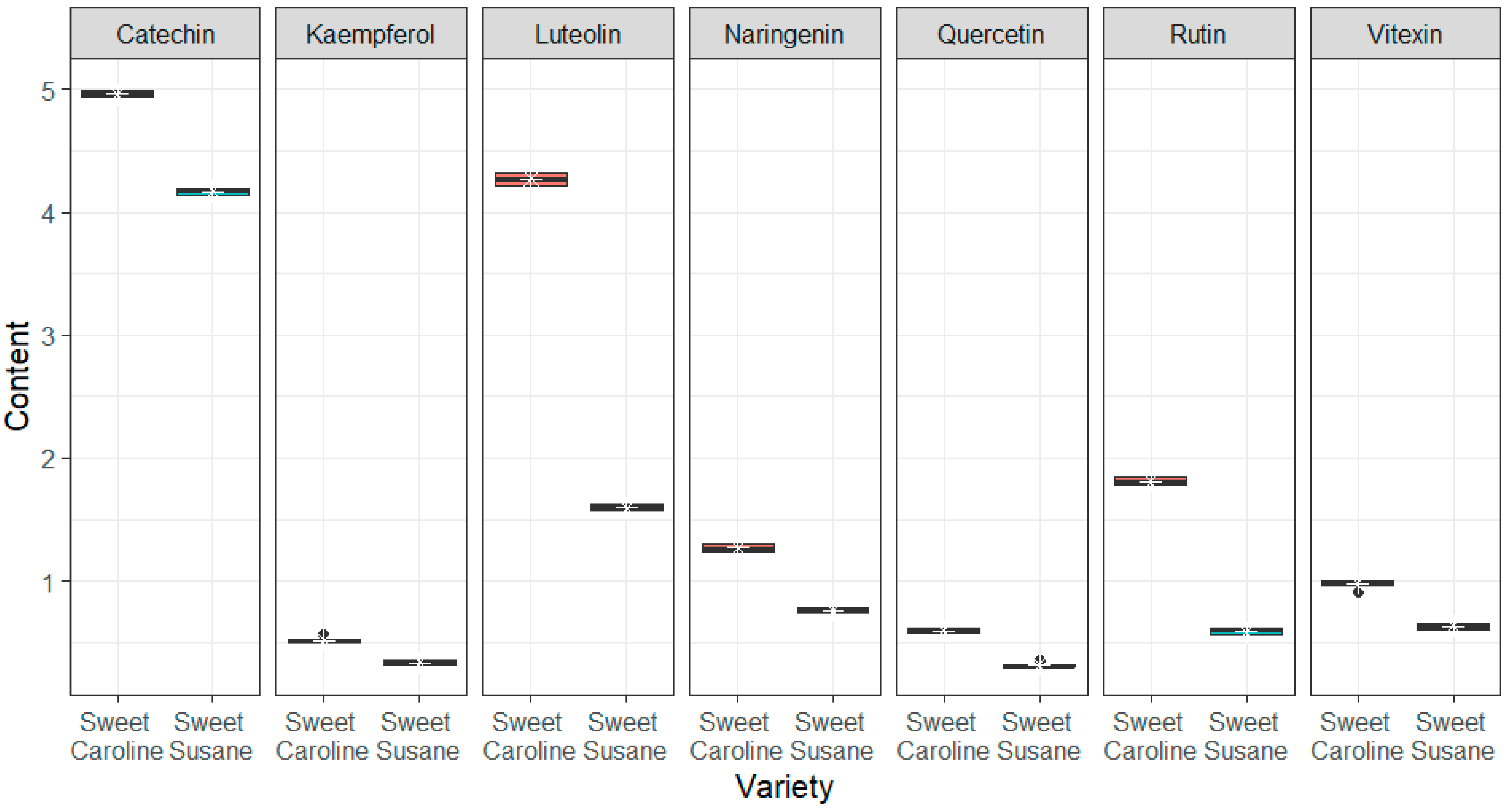
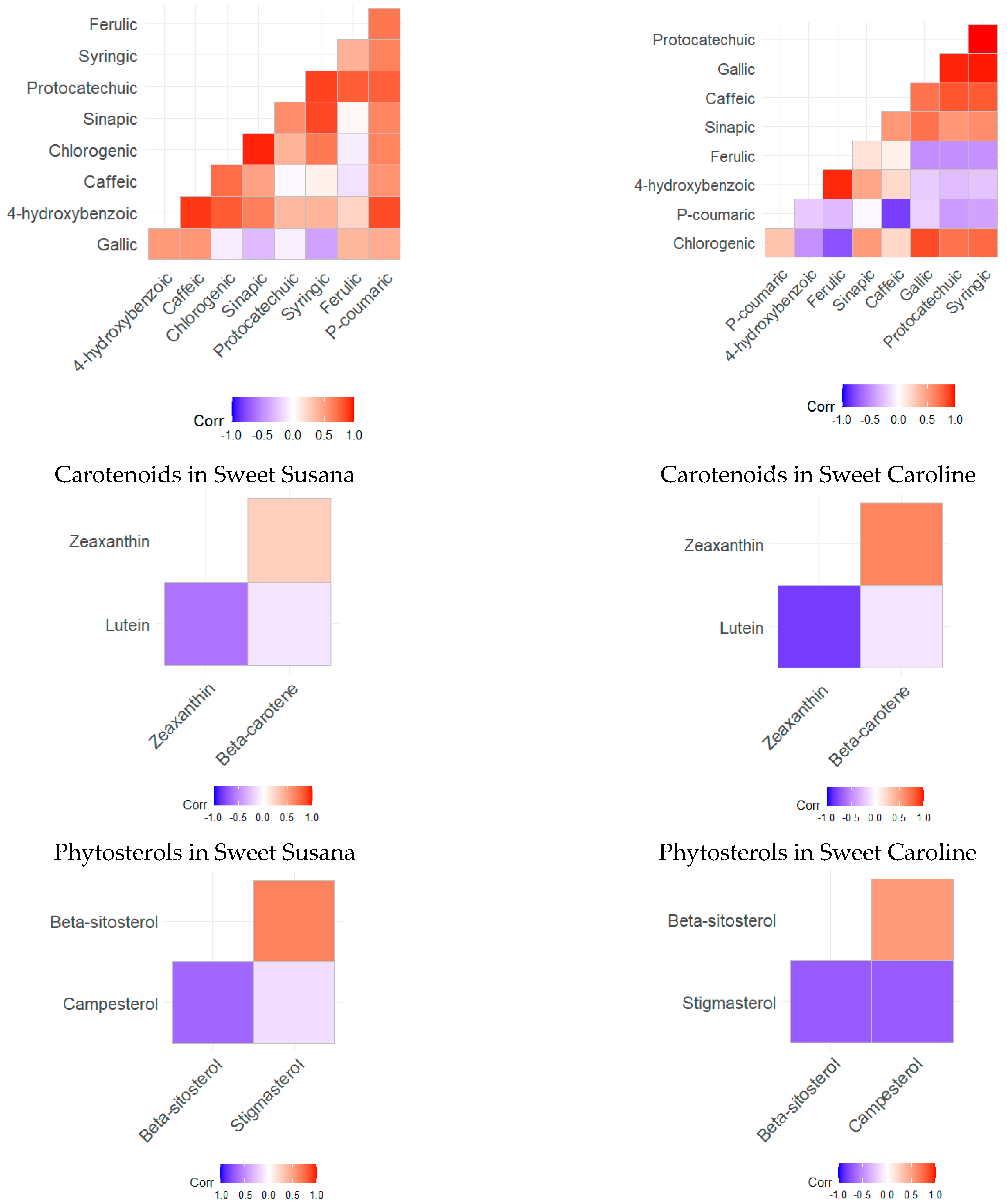
3.2. Bioactive Compounds
3.3. Content of Major and Trace Elements
3.4. Starch Content Analysis
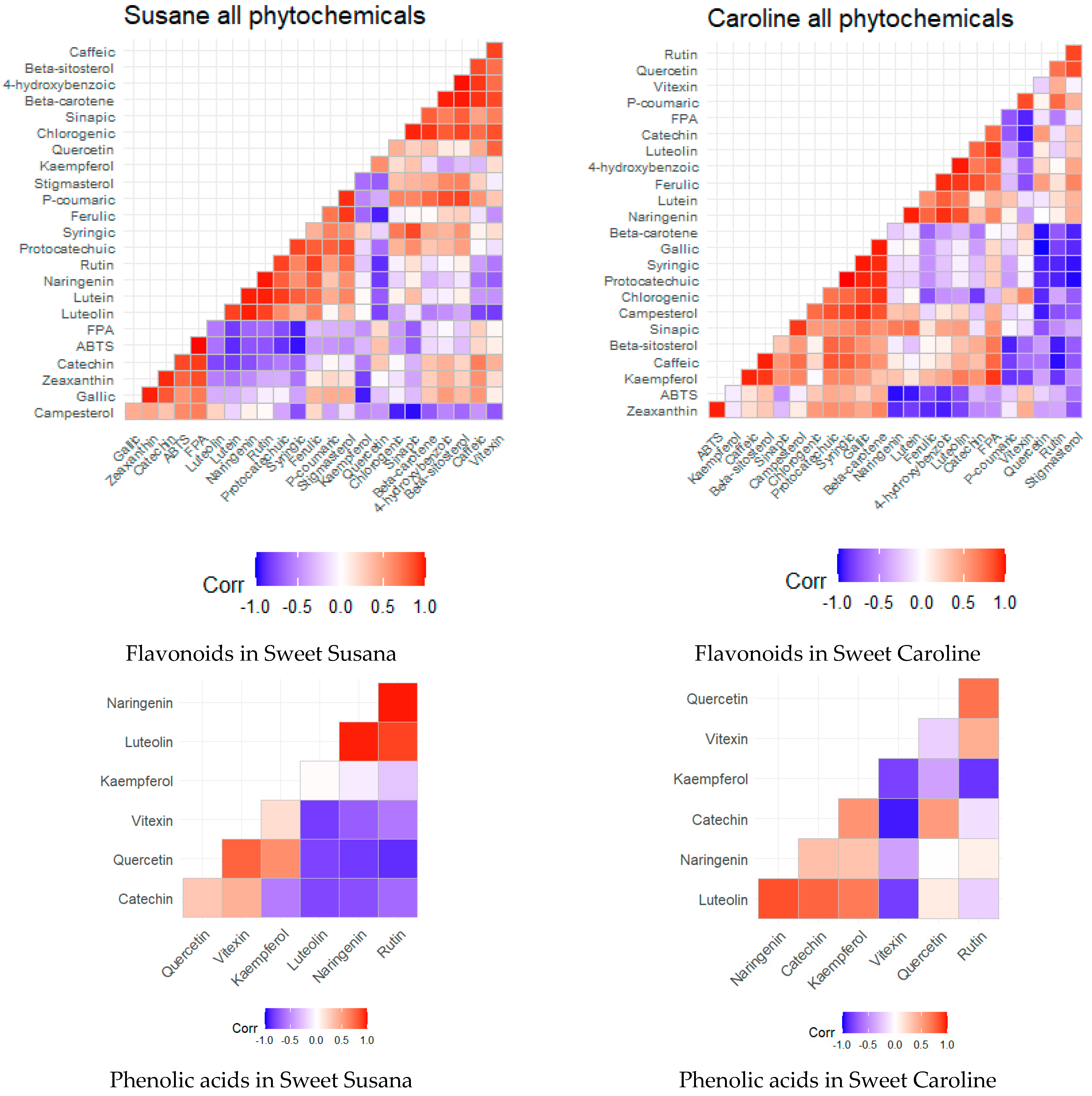
3.5. Statistical Analysis of the Results
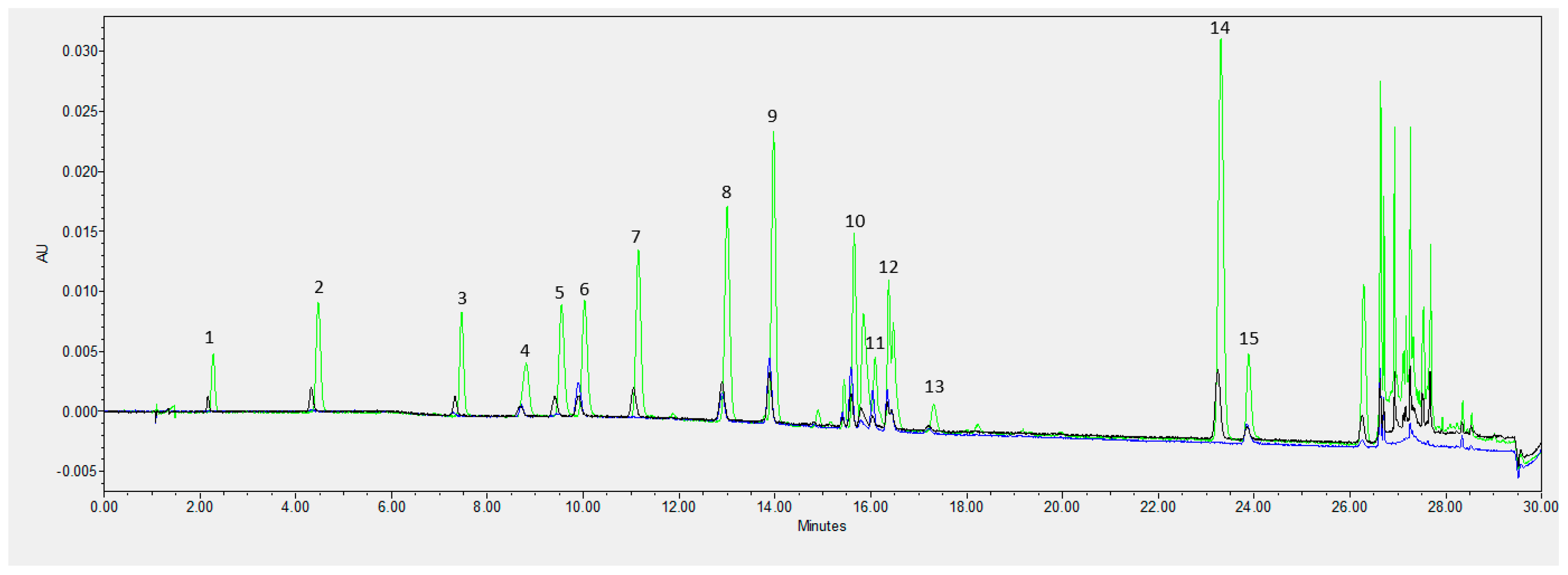
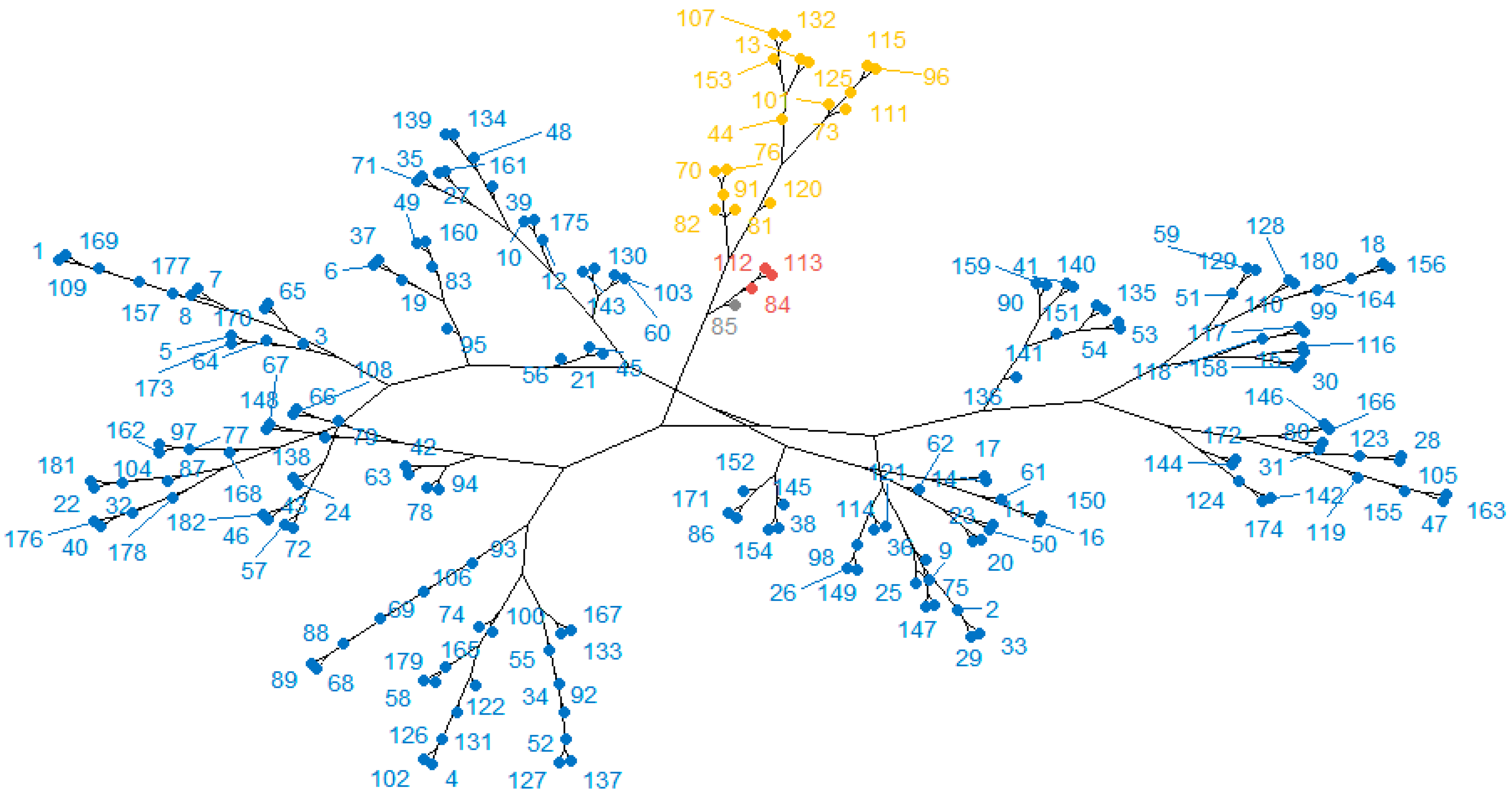
3.6. Organic Volatile Compounds
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Venkateswaran, K.; Elangovan, M.; Sivaraj, N. Origin, Domestication and Diffusion of Sorghum bicolor. In Woodhead Publishing Series in Food Science, Technology and Nutrition, Breeding Sorghum for Diverse End Uses; Aruna, C., Visarada, K.B.R.S., Venkatesh Bhat, B., Tonapi, V.A., Eds.; Woodhead Publishing: Sawston, UK, 2019; pp. 15–31. ISBN 9780081018798. [Google Scholar] [CrossRef]
- Hariprasanna, K.; Patil, J.V. Sorghum: Origin, Classification, Biology and Improvement. In Sorghum Molecular Breeding; Madhusudhana, R., Rajendrakumar, P., Patil, J., Eds.; Springer: New Delhi, India, 2015; pp. 3–20. [Google Scholar] [CrossRef]
- Hossain, M.S.; Islam, M.N.; Rahman, M.M.; Mostofa, M.G.; Khan, M.A.R. Sorghum: A prospective crop for climatic vulnerability, food and nutritional security. J. Agric. Food Res. 2022, 8, 100300. [Google Scholar] [CrossRef]
- Schaffasz, A.; Windpassinger, S.; Friedt, W.; Snowdon, R.; Wittkop, B. Sorghum as a Novel Crop for Central Europe: Using a Broad Diversity Set to Dissect Temperate-Adaptation. Agronomy 2019, 9, 535. [Google Scholar] [CrossRef]
- Popescu, A.; Dinu, T.A.; Stoian, E. Sorghum—An important cereal in the world, in the European Union and Romania. Sci. Pap. Ser. Manag. Econom. Eng. Agric. Rural Dev. 2018, 18, 271–284. [Google Scholar]
- Hacisalihoglu, G.; Armstrong, P.R. Flax and Sorghum: Multi-Element Contents and Nutritional Values within 210 Varieties and Potential Selection for Future Climates to Sustain Food Security. Plants 2022, 11, 451. [Google Scholar] [CrossRef]
- Khalid, W.; Ali, A.; Arshad, M.S.; Afzal, F.; Akram, R.; Siddeeg, A.; Kousar, S.; Rahim, M.A.; Aziz, A.; Maqbool, A.; et al. Nutrients and bioactive compounds of Sorghum bicolor L. used to prepare functional foods: A review on the efficacy against different chronic disorders. Int. J. Food Prop. 2022, 25, 1045–1062. [Google Scholar] [CrossRef]
- Paye, W.S.; Acharya, P.; Ghimire, R. Water productivity of forage sorghum in response to winter cover crops in semi-arid irrigated conditions. Field Crops Res. 2022, 283, 108552. [Google Scholar] [CrossRef]
- Bhattarai, B.; Singh, S.; West, C.P.; Ritchie, G.L.; Trostle, C.L. Water Depletion Pattern and Water Use Efficiency of Forage Sorghum, Pearl millet, and Corn Under Water Limiting Condition. Agric. Water Manag. 2020, 238, 106206. [Google Scholar] [CrossRef]
- Bhattarai, B.; Singh, S.; West, C.P.; Saini, R. Forage potential of pearl millet and forage sorghum alternatives to corn under the water-limiting conditions of the Texas high plains: A review. Crop Forage Turfgrass Manag. 2019, 5, 1–12. [Google Scholar] [CrossRef]
- Prazak, R. Prospects for Sorghum cultivation in Poland. Acta Agrobot. 2016, 69, 2. [Google Scholar] [CrossRef]
- Kanbar, A.; Flubacher, N.; Hermuth, J.; Kosová, K.; Horn, T.; Nick, P. Mining Sorghum Biodiversity—Potential of Dual-Purpose Hybrids for Bio-Economy. Diversity 2021, 13, 192. [Google Scholar] [CrossRef]
- Niu, H.; Ping, J.; Wang, Y.; Lv, X.; Li, H.; Zhang, F.; Chu, J.; Han, Y. Population genomic and genome-wide association analysis of lignin content in a global collection of 206 forage sorghum accessions. Mol. Breed. 2020, 40, 73. [Google Scholar] [CrossRef]
- Srinivasa Rao, P.; Ganesh Kumar, C.; Reddy, B.V.S. Sweet Sorghum: From Theory to Practice. In Characterization of Improved Sweet Sorghum Cultivars; Rao, P., Kumar, C., Eds.; Springer: New Delhi, India, 2013; pp. 1–15. [Google Scholar] [CrossRef]
- Przybylska-Balcerek, A.; Stuper-Szablewska, K. The effect of phenolic acids on living organisms. Indian J. Med. Res. Pharm. Sci. 2019, 9, 1–14. [Google Scholar]
- Przybylska-Balcerek, A.; Frankowski, J.; Stuper-Szablewska, K. Bioactive compounds in sorghum. Eur. Food Res. Technol. 2019, 245, 1075–1080. [Google Scholar] [CrossRef]
- Hermuth, J.; Kosová, K. Characterization of the first Czech sorghum variety Ruzrok tested in Czech Republic. Czech J. Genet. Plant Breed. 2017, 53, 37–44. [Google Scholar] [CrossRef]
- Pasteris, A.M.; Heiermann, M.; Theuerl, S.; Plogsties, V.; Jost, C.; Prochnow, A.; Herrmann, C. Multi-advantageous sorghum as feedstock for biogas production: A comparison between single-stage and two-stage anaerobic digestion systems. J. Clean. Prod. 2020, 358, 131985. [Google Scholar] [CrossRef]
- Grabovskyi, M.; Lozinskyi, M.; Grabovska, T.; Roubík, H. Green mass to biogas in Ukraine—Bioenergy potential of corn and sweet sorghum. Biomass Convers. Biorefin. 2021, 13, 3309–3317. [Google Scholar] [CrossRef]
- Bartzialis, D.; Giannoulis, K.D.; Skoufogianni, E.; Lavdis, A.; Zalaoras, G.; Charvalas, G.; Danalatos, N.G. Sorghum dry biomass yield for solid bio-fuel production affected by different N-fertilization rates. Agron. Res. 2020, 18, 1147–1153. [Google Scholar] [CrossRef]
- Stamenković, O.S.; Siliveru, K.; Veljković, V.B.; Banković-Ilić, I.B.; Tasić, M.B.; Ciampitti, I.A.; Đalović, I.G.; Mitrović, P.M.; Sikora, V.Š.; Prasad, P.V. Production of biofuels from sorghum. Renew. Sustain. Energy Rev. 2020, 124, 109769. [Google Scholar] [CrossRef]
- Velmurugan, B.; Narra, M.; Rudakiya, D.M.; Madamwar, D. Sweet sorghum: A potential resource for bioenergy production. In Refining Biomass Residues for Sustainable Energy and Bioproducts; Academic Press: Cambridge, MA, USA, 2020; pp. 215–242. [Google Scholar]
- Szambelan, K.; Nowak, J.; Frankowski, J.; Szwengiel, A.; Jeleń, H.; Burczyk, H. The comprehensive analysis of sorghum cultivated in Poland for energy purposes: Separate hydrolysis and fermentation and simultaneous saccharification and fermentation methods and their impact on bioethanol effectiveness and volatile by-products from the grain and the energy potential of sorghum straw. Bioresour. Technol. 2018, 250, 750–757. [Google Scholar] [CrossRef] [PubMed]
- Bhakar, A.; Singh, M.; Kumar, S.; Meena, R.K.; Meena, B.L.; Kumar, R.; Meena, V.K. Growth, productivity and profitability of fodder sorghum and cluster bean as influenced by mixed cropping and nutrient management. Legume Res. 2022, 44, 1308–1314. [Google Scholar] [CrossRef]
- Blümmel, M.; Zerbini, E.; Reddy, B.V.S.; Hash, C.T.; Bidinger, F.; Khan, A.A. Improving the production and utilization of sorghum and pearl millet as livestock feed: Progress towards dual-purpose genotypes. Field Crops Res. 2023, 84, 143–158. [Google Scholar] [CrossRef]
- Szambelan, K.; Nowak, J.; Szwengiel, A.; Jeleń, H. Quantitative and qualitative analysis of volatile compounds in sorghum distillates obtained under various hydrolysis and fermentation conditions. Ind. Crops Prod. 2020, 155, 143–158. [Google Scholar] [CrossRef]
- Bean, S.R.; Wilson, J.D.; Moreau, R.A.; Galant, A.; Awika, J.M.; Kaufman, R.C.; Adrianos, S.L.; Ioerger, B.P. Structure and composition of the sorghum grain. Sorghum State Art Future Perspetives 2019, 58, 173–214. [Google Scholar] [CrossRef]
- Dykes, L.; Rooney, L.W. Sorghum and millet phenols and antioxidants. J. Cereal Sci. 2006, 44, 236–251. [Google Scholar] [CrossRef]
- Khalid, W.; Arshad, M.S.; Aslam, N.; Mukhtar, S.; Rahim, M.A.; Ranjha, M.M.A.N.; Noreen, S.; Afzal, M.F.; Aziz, A.; Awuchi, C.G. Food applications of sorghum derived kafirins potentially valuable in celiac disease. Inter. J. Food Prop. 2022, 25, 2348–2363. [Google Scholar] [CrossRef]
- Pontieri, P.; Mamone, G.; De Caro, S.; Tuinstra, M.R.; Roemer, R.; Okot, J.; De Vita, P.; Ficco, D.B.M.; Alifano, P.; Pignone, D.; et al. Sorghum, a Healthy and Gluten-Free Food for Celiac Patients as Demonstrated by Genome, Biochemical, and Immunochemical Analyses. J. Agric. Food Chem. 2013, 61, 2565–2571. [Google Scholar] [CrossRef]
- Ciacci, C.; Maiuri, L.; Caporaso, N.; Bucci, C.; Del Giudice, L.; Massardo, D.R.; Pontieri, P.; Di Fonzo, N.; Bean, S.R.; Ioerger, B.; et al. Celiac disease: In vitro and in vivo safety and palatability of wheat-free sorghum food products. Clin. Nutr. 2007, 26, 799–805. [Google Scholar] [CrossRef] [PubMed]
- Kaplan, M. Assessment of grain minerals of Turkish sorghum (Sorghum bicolor L.) landraces by GT biplot analysis. Qual. Assur. Saf. Crops Foods 2019, 11, 441–447. [Google Scholar] [CrossRef]
- Pontieri, P.; Troisi, J.; Di Fiore, R.; Di Maro, A.; Bean, S.R.; Tuinstra, M.R.; Roemer, E.; Boffa, A.; Del Giudice, A.; Pizzolante, G.; et al. Mineral contents in grains of seven food-grade sorghum hybrids grown in a Mediterranean environment. Aust. J. Crop Sci. 2014, 8, 1550–1559. [Google Scholar]
- Mawouma, S.; Condurache, N.N.; Turturică, M.; Constantin, O.E.; Croitoru, C.; Rapeanu, G. Chemical composition and antioxidant profile of sorghum (Sorghum bicolor (L.) Moench) and pearl millet (Pennisetum glaucum (L.) R. Br.) grains cultivated in the far-North region of Cameroon. Foods 2022, 11, 2026. [Google Scholar] [CrossRef]
- Xu, J.; Wang, W.; Zhao, Y. Phenolic compounds in whole grain sorghum and their health benefits. Foods 2021, 10, 1921. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, S.O.; Abdalla, A.W.H.; Inoue, T.; Ping, A.; Babiker, E.E. Nutritional quality of grains of sorghum cultivar grown under different levels of micronutrients fertilization. Food Chem. 2014, 159, 374–380. [Google Scholar] [CrossRef] [PubMed]
- Suha, O.A.; Abdalla, A.W.H.; Osman, M.A.; Inoue, T.; Ping, A.; Babiker, E.E. Changes in total and extractable macroelements of grains of sorghum cultivars grown under different levels of micronutrients. Int. J. Innov. Appl. Stud. 2013, 4, 649. [Google Scholar]
- Ofosu, F.K.; Elahi, F.; Daliri, E.B.M.; Yeon, S.J.; Ham, H.J.; Kim, J.H.; Sang-Ik Han, A.-I.; Oh, D.H. Flavonoids in decorticated sorghum grains exert antioxidant, antidiabetic and antiobesity activities. Molecules 2020, 25, 2854. [Google Scholar] [CrossRef] [PubMed]
- Dykes, L.; Peterson, G.C.; Rooney, W.L.; Rooney, L.W. Flavonoid composition of lemon-yellow sorghum genotypes. Food Chem. 2011, 128, 173–179. [Google Scholar] [CrossRef] [PubMed]
- Dykes, L.; Seitz, L.M.; Rooney, W.L.; Rooney, L.W. Flavonoid composition of red sorghum genotypes. Food Chem. 2009, 116, 313–317. [Google Scholar] [CrossRef]
- Speranza, S.; Knechtl, R.; Witlaczil, R.; Schönlechner, R. Reversed-phase HPLC characterization and quantification and antioxidant capacity of the phenolic acids and flavonoids extracted from eight varieties of sorghum grown in Austria. Front. Plant Sci. 2021, 12, 769151. [Google Scholar] [CrossRef]
- Ghasemzadeh, A.; Ghasemzadeh, N. Flavonoids and phenolic acids: Role and biochemical activity in plants and human. J. Med. Plants Res. 2011, 5, 6697–6703. [Google Scholar] [CrossRef]
- Svensson, L.; Sekwati-Monang, B.; Lutz, D.L.; Schieber, A.; Ganzle, M.G. Phenolic acids and flavonoids in nonfermented and fermented red sorghum (Sorghum bicolor (L.) Moench). J. Agric. Food Chem. 2010, 58, 9214–9220. [Google Scholar] [CrossRef]
- Sathasivam, R.; Radhakrishnan, R.; Kim, J.K.; Park, S.U. An update on biosynthesis and regulation of carotenoids in plants. S. Afr. J. Bot. 2021, 140, 290–302. [Google Scholar] [CrossRef]
- de Morais Cardoso, L.; Pinheiro, S.S.; da Silva, L.L.; de Menezes, C.B.; de Carvalho, C.W.P.; Tardin, F.D.; Queiroz, V.A.V.; Martino, H.S.D.; Pinheiro-Sant’Ana, H.M. Tocochromanols and carotenoids in sorghum (Sorghum bicolor L.): Diversity and stability to the heat treatment. Food Chem. 2015, 172, 900–908. [Google Scholar] [CrossRef] [PubMed]
- Szambelan, K.; Szwengiel, A.; Nowak, J.; Jeleń, H.; Frankowski, J. Low-waste technology for the production of bioethanol from sorghum grain: Comparison of Zymomonas mobilis and Saccharomyces cerevisiae in fermentation with stillage reusing. J. Clean. Product. 2022, 352, 131607. [Google Scholar] [CrossRef]
- Duodu, K.G.; Awika, J.M. Phytochemical-related health-promoting attributes of sorghum and millets. In Sorghum and Millets, 2nd ed.; Taylor, J.R.N., Duodu, K.G., Eds.; AACC International Press: St. Paul, MN, USA, 2019; pp. 225–258. [Google Scholar]
- Moreau, R.A.; Nyström, L.; Whitaker, B.D.; Winkler-Moser, J.K.; Baer, D.J.; Gebauer, S.K.; Hicks, K.B. Phytosterols and their derivatives: Structural diversity, distribution, metabolism, analysis, and health-promoting uses. Prog. Lipid Res. 2018, 70, 35–61. [Google Scholar] [CrossRef] [PubMed]
- Uddin, M.S.; Sarker, M.Z.I.; Ferdosh, S.; Akanda, M.J.H.; Easmin, M.S.; Bt Shamsudin, S.H.; Yunus, K.B. Phytosterols and their extraction from various plant matrices using supercritical carbon dioxide: A review. J. Sci. Food Agric. 2015, 95, 1385–1394. [Google Scholar] [CrossRef] [PubMed]
- Stuper-Szablewska, K.; Przybylska, A.; Kurasiak-Popowska, D.; Perkowski, J. Ferulic acid. Properties, determination and application in the cosmetics industry. Przem. Chem. 2017, 96, 2070–2076. [Google Scholar]
- Li, Z.; Zhao, X.; Zhang, X.; Liu, H. The Effects of Processing on Bioactive Compounds and Biological Activities of Sorghum Grains. Molecules 2022, 27, 3246. [Google Scholar] [CrossRef]
- Mrid, R.B.; Bouargalne, Y.; El Omari, R.; Nhiri, M. New insights into the therapeutic effects of phenolic acids from sorghum seeds. J. Rep. Pharm. Sci. 2019, 8, 91. [Google Scholar] [CrossRef]
- Vanamala, J.K.; Massey, A.R.; Pinnamaneni, S.R.; Reddivari, L.; Reardon, K.F. Grain and sweet sorghum (Sorghum bicolor L. Moench) serves as a novel source of bioactive compounds for human health. Crit. Rev. Food Sci. Nutr. 2018, 58, 2867–2881. [Google Scholar] [CrossRef]
- de Morais Cardoso, L.; Pinheiro, S.S.; Martino, H.S.D.; Pinheiro-Sant’Ana, H.M. Sorghum (Sorghum bicolor L.): Nutrients, bioactive compounds, and potential impact on human health. Crit. Rev. Food Sci. Nutr. 2017, 57, 372–390. [Google Scholar] [CrossRef]
- Atkinson, R.; Arey, J. Atmospheric degradation of volatile organic compounds. Chem. Rev. 2003, 103, 4605–4638. [Google Scholar] [CrossRef]
- Gallagher, M.; Wysocki, C.J.; Leyden, J.J.; Spielman, A.I.; Sun, X.; Preti, G. Analyses of volatile organic compounds from human skin. Brit. J. Dermatol. 2008, 159, 780–791. [Google Scholar] [CrossRef] [PubMed]
- Abbas, F.; O’Neill Rothenberg, D.; Zhou, Y.; Ke, Y.; Wang, H.C. Volatile organic compounds as mediators of plant communication and adaptation to climate change. Physiol. Plant. 2022, 174, e13840. [Google Scholar] [CrossRef] [PubMed]
- Fincheira, P.; Quiroz, A.; Tortella, G.; Diez, M.C.; Rubilar, O. Current advances in plant-microbe communication via volatile organic compounds as an innovative strategy to improve plant growth. Microbiol. Res. 2021, 247, 126726. [Google Scholar] [CrossRef] [PubMed]
- Holopainen, J.K.; Blande, J.D. Molecular plant volatile communication. Adv. Exp. Med. Biol. 2012, 739, 17–31. [Google Scholar] [PubMed]
- Bao, X.; Zhou, W.; Xu, L.; Zheng, Z. A meta-analysis on plant volatile organic compound emissions of different plant species and responses to environmental stress. Environ. Pollut. 2022, 318, 120886. [Google Scholar] [CrossRef]
- Manco, A.; Gasbarra, D.; Famulari, D.; Di Tommasi, P.; Brilli, F.; Arena, C.; Gioli, B.; Magliulo, E.; Vitale, L. Biogenic Volatile Organic Compounds (BVOCs) exchanges over Sorghum bicolor L. during a whole growing season in the Southern Europe. In Proceedings of the 2019 IEEE International Workshop on Metrology for Agriculture and Forestry (MetroAgriFor), Prtici, Italy, 24–26 October 2019; pp. 72–76. [Google Scholar]
- Spinelli, F.; Cellini, A.; Marchetti, L.; Mudigere, K.; Piovene, C. Emission and function of volatile organic compounds in response to abiotic stress. In Abiotic Stress in Plants—Mechanisms and Adaptations; Shanker, A., Venkateswarlu, B., Eds.; InTech: Rjieka, Croatia, 2011; pp. 367–394. [Google Scholar]
- Loreto, F.; Barta, C.; Brilli, F.; Nogues, I. On the induction of volatile organic compound emissions by plants as consequence of wounding or fluctuations of light and temperature. Plant Cell Environ. 2006, 29, 1820–1828. [Google Scholar] [CrossRef] [PubMed]
- Vivaldo, G.; Masi, E.; Taiti, C.; Caldarelli, G.; Mancuso, S. The network of plants volatile organic compounds. Sci. Rep. 2017, 7, 11050. [Google Scholar] [CrossRef]
- Karl, T.; Harren, F.; Warneke, C.; De Gouw, J.; Grayless, C.; Fall, R. Senescing grass crops as regional sources of reactive volatile organic compounds. J. Geophys. Res. Atmos. 2005, 110, D15. [Google Scholar] [CrossRef]
- Catola, S.; Centritto, M.; Cascone, P.; Ranieri, A.; Loreto, F.; Calamai, L.; Balestrini, R.; Guerrieri, E. Effects of single or combined water deficit and aphid attack on tomato volatile organic compound (VOC) emission and plant-plant communication. Environ. Exp. Bot. 2018, 153, 54–62. [Google Scholar] [CrossRef]
- Filella, I.; Wilkinson, M.J.; Llusia, J.; Hewitt, C.N.; Peñuelas, J. Volatile organic compounds emissions in Norway spruce (Picea abies) in response to temperature changes. Physiol. Plant. 2007, 130, 58–66. [Google Scholar] [CrossRef]
- Owen, S.M.; Boissard, C.; Hewitt, C.N. Volatile organic compounds (VOCs) emitted from 40 Mediterranean plant species: VOC speciation and extrapolation to habitat scale. Atmos. Environ. 2001, 35, 5393–5409. [Google Scholar] [CrossRef]
- Liu, Y.; Song, M.; Liu, X.; Zhang, Y.; Hui, L.; Kong, L.; Zhang, Y.; Zhang, C.; Qu, Y.; An, J.; et al. Characterization and sources of volatile organic compounds (VOCs) and their related changes during ozone pollution days in 2016 in Beijing, China. Environ. Pollut. 2020, 257, 113599. [Google Scholar] [CrossRef] [PubMed]
- Sadatshojaie, A.; Rahimpour, M.R. CO2 emission and air pollution (volatile organic compounds, etc.)—Related problems causing climate change. In Current Trends and Future Developments on (Bio-) Membranes; Figoli, A., Li, Y., Basile, A., Eds.; Elsevier: Amsterdam, The Netherland, 2020; pp. 1–30. [Google Scholar] [CrossRef]
- Frankowski, J.; Przybylska-Balcerek, A.; Stuper-Szablewska, K. Concentration of Pro-Health Compound of Sorghum Grain-Based Foods. Foods 2022, 11, 216. [Google Scholar] [CrossRef]
- Przybylska-Balcerek, A.; Frankowski, J.; Stuper-Szablewska, K. The influence of weather conditions on bioactive compound content in sorghum grain. Eur. Food Res. Technol. 2020, 246, 13–22. [Google Scholar] [CrossRef]
- Suchowilska, E.; Wiwart, M.; Przybylska-Balcerek, A.; Stuper-Szablewska, K. The profile of bioactive compounds in the grain of various × Tritordeum genotypes. J. Cereal Sci. 2021, 102, 103352. [Google Scholar] [CrossRef]
- Kurasiak-Popowska, D.; Graczyk, M.; Przybylska-Balcerek, A.; Stuper-Szablewska, K.; Szwajkowska-Michałek, L. An Analysis of Variability in the Content of Phenolic Acids and Flavonoids in Camelina Seeds Depending on Weather Conditions, Functional Form, and Genotypes. Molecules 2022, 27, 3364. [Google Scholar] [CrossRef]
- Frankowski, J.; Przybylska-Balcerek, A.; Graczyk, M.; Niedziela, G.; Sieracka, D.; Stuper-Szablewska, K. The Effect of Mineral Fertilization on the Content of Bioactive Compounds in Hemp Seeds and Oil. Molecules 2023, 28, 4870. [Google Scholar] [CrossRef]
- Ostrowska-Kołodziejczak, A.; Stuper-Szablewska, K.; Kulik, T.; Buśko, M.; Rissmann, I.; Wiwart, M.; Perkowski, J. Concentration of fungal metabolites, phenolic acids and metals in mixtures of cereals grown in organic and conventional farms. J. Anim. Feed Sci. 2016, 25, 74–81. [Google Scholar] [CrossRef]
- PN-EN ISO712:2012; Determination of Humidity Using the Drying Method. International Organization for Standardization: Vernier, Switzerland, 2012.
- PN-EN ISO 11085:2015-10; Determination of Fat Content. International Organization for Standardization: Vernier, Switzerland, 2015.
- BS EN ISO 10520:1998; Native Starch. Determination of Starch Content. Ewers Polarimetric Method. International Organization for Standardization: Vernier, Switzerland, 1998; ISBN 0580303950.
- Xiong, Y.; Zhang, P.; Luo, J.; Johnson, S.; Fang, Z. Effect of Processing on the Phenolic Contents, Antioxidant Activity and Volatile Compounds of Sorghum Grain Tea. J. Cereal Sci. 2019, 85, 6–14. [Google Scholar] [CrossRef]
- Przybylska-Balcerek, A.; Góral, T.; Kurasiak-Popowska, D.; Stuper-Szablewska, K. Phenolic acids in various genotypes of cereals grown in Poland in the years 2017–2018. Acad. J. Med. Plants 2019, 7, 092–099. [Google Scholar]
- Khoddami, A.; Truong, H.H.; Liu, S.Y.; Roberts, T.H.; Selle, P.H. Concentrations of specific phenolic compounds in six red sorghums influence nutrient utilisation in broiler chickens. Anim. Feed. Sci. Technol. 2015, 210, 190–199. [Google Scholar] [CrossRef]
- Adom, K.K.; Liu, R.H. Antioxidant Activity of Grains. J. Agric. Food Chem. 2002, 50, 6182–6187. [Google Scholar] [CrossRef] [PubMed]
- Ou, S.; Kwok, K.C. Ferulic acid: Pharmaceutical functions, preparation and applications in foods. J. Sci. Food Agric. 2004, 84, 1261–1269. [Google Scholar] [CrossRef]
- Carbonneau, M.A.; Cisse, M.; Mora-Soumille, N.; Dairi, S.; Rosa, M.; Michel, F.; Dangles, O. Antioxidant properties of 3-deoxyanthocyanidins and polyphenolic extracts from Côte d’Ivoire’s red and white sorghums assessed by ORAC and in vitro LDL oxidisability tests. Food Chem. 2014, 145, 701–709. [Google Scholar] [CrossRef] [PubMed]
- Carr, T.P.; Weller, C.L.; Schlegel, V.L.; Cuppett, S.L.; Guderian, D.M., Jr.; Johnson, K.R. Grain sorghum lipid extract reduces cholesterol absorption and plasma non-HDL cholesterol concentration in hamsters. J. Nutr. 2005, 135, 2236–2240. [Google Scholar] [CrossRef]
- Heupel, R.C.; Sauvaire, Y.; Le, P.H.; Parish, E.J.; Nes, W.D. Sterol composition and biosynthesis in sorghum: Importance to developmental regulation. Lipids 1986, 21, 69–75. [Google Scholar] [CrossRef]
- Lee, Y.-S. The Contents of Phytosterols, Squalene, and Vitamin E and the Composition of Fatty Acids of Korean Landrace Setaria italica and Sorghum bicolar Seeds. Korean J. Plant Resour. 2013, 26, 663–672. [Google Scholar]
- Singh, V.; Moreau, R.A.; Hicks, K.B. Yield and phytosterol composition of oil extracted from grain sorghum and its wet-milled fractions. Cereal Chem. 2003, 80, 126–129. [Google Scholar] [CrossRef]
- Ryan, E.; Galvin, K.; O’Connor, T.P.; Maguire, A.R.; O’Brien, N.M. Phytosterol, squalene, tocopherol content and fatty acid profile of selected seeds, grains, and legumes. Plant Foods Hum. Nutr. 2007, 62, 85–91. [Google Scholar] [CrossRef]
- Paiva, C.L.; Queiroz, V.A.V.; Simeone, M.L.F.; Schaffert, R.E.; de Oliveira, A.C.; da Silva, C.S. Mineral content of sorghum genotypes and the influence of water stress. Food Chem. 2017, 214, 400–405. [Google Scholar] [CrossRef]
- Motlhaodi, T.; Bryngelsson, T.; Chite, S.; Fatih, M.; Ortiz, R.; Geleta, M. Nutritional variation in sorghum [Sorghum bicolor (L.) Moench] accessions from southern Africa revealed by protein and mineral composition. J. Cereal Sci. 2018, 83, 123–129. [Google Scholar] [CrossRef]
- Ikem, A.; Odumosu, O.P.; Udousoro, I. Elemental composition of cereal grains and the contribution to the dietar intake in the Nigerian population. J. Food Compos. Anal. 2023, 118, 105207. [Google Scholar] [CrossRef]
- Soetan, K.O.; Olaiya, C.O.; Oyewole, O.E. The importance of mineral elements for humans, domestic animals and plants: A review. Afr. J. Food Sci. 2010, 4, 200–222. [Google Scholar]
- Victora, C.G.; Christian, P.; Vidaletti, L.P.; Gatica-Domínguez, G.; Menon, P.; Black, R.E. Revisiting maternal and child undernutrition in low-income and middle-income countries: Variable progress towards an unfinished agenda. Lancet 2021, 397, 1388–1399. [Google Scholar] [CrossRef]
- Zlotkin, S.; Dewey, K.G. Perspective: Putting the youngest among us into the nutrition “call for action” for food fortification strategies. Am. J. Clin. Nutr. 2021, 114, 1257–1260. [Google Scholar] [CrossRef]
- Madhusudhan, R.; Hariprasanna, K.; Aruna, C.; Sajjanar, G.M.; Hanamaratti, N.G.; Sameera, S.; Tonapi, V.A. Genetic variability, G × E interaction and stability for iron and zinc content in sorghum grains in advanced breeding lines. J. Cereal Sci. 2023, 110, 103653. [Google Scholar] [CrossRef]
- Jaćimović, S.; Kiprovski, B.; Ristivojević, P.; Dimić, D.; Nakarada, Đ.; Dojčinović, B.; Sikora, V.; Teslić, N.; Pantelić, N.Đ. Chemical Composition, Antioxidant Potential, and Nutritional Evaluation of Cultivated Sorghum Grains: A Combined Experimental, Theoretical, and Multivariate Analysis. Antioxidants 2023, 12, 1485. [Google Scholar] [CrossRef]
- Pontieri, P.; Troisi, J.; Calcagnile, M.; Bean, S.R.; Tilley, M.; Aramouni, F.; Boffa, A.; Pepe, G.; Campiglia, P.; Del Giudice, F.; et al. Chemical Composition, Fatty Acid and Mineral Content of Food-Grade White, Red and Black Sorghum Varieties Grown in the Mediterranean Environment. Foods 2022, 11, 436. [Google Scholar] [CrossRef] [PubMed]
- Dicko, M.H.; Gruppen, H.; Zouzouho, O.C.; Traore, A.S.; van Berkel, W.J.H.; Voragen, A.G.J. Effect of germination on the activities of amylases and phenolic enzymes in sorghum varieties grouped according to food end—Use properties. J. Sci. Food Agric. 2006, 86, 953–963. [Google Scholar] [CrossRef]
- Stachowiak, B.; Nowak, J.; Szambelan, K.; Bajon, A. Sorghum—Agricultural and industrial potential. Agric. Adv. Issues 2023, 110, 49–61. [Google Scholar]
| Month | Temperature (°C) | Precipitation (mm) | ||
|---|---|---|---|---|
| Average Monthly (2022) | Long-Term Average (1960–2020) | Average Monthly (2022) | Long-Term Average (1960–2020) | |
| May | 14.7 | 12.7 | 43.0 | 61.0 |
| June | 19.7 | 15.6 | 73.5 | 53.0 |
| July | 19.4 | 17.4 | 48.5 | 78.0 |
| August | 21.0 | 16.2 | 28.5 | 60.0 |
| September | 12.9 | 12.6 | 85.0 | 49.0 |
| October | 12.6 | 8.1 | 31.0 | 41.0 |
| Phenological Development | Date |
|---|---|
| Sowing | 16 May 2022 |
| Sprout | 24 May 2022 |
| One pair of leaves | 28 May 2022 |
| Two pairs of leaves | 1 June 2022 |
| Three pairs of leaves | 6 June 2022 |
| Four pairs of leaves | 10 June 2022 |
| Height—0.15 m | 12 June 2022 |
| Height—0.30 m | 23 June 2022 |
| Flowering ‘Sweet Caroline’ | 17 July 2022 |
| Flowering ‘Sweet Susana’ | 19 July 2022 |
| Height 0.8 m | 19 July 2022 |
| Height 1.0 m | 25 July 2022 |
| Harvesting ‘Sweet Caroline’ and ‘Sweet Susana’ | 26 October 2022 |
| Content of Major and Trace Elements (mg/100 g) | Content of Fat (g/100 g) | Content of Water (%) | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Ca | Fe | Mn | Mg | P | Cu | Na | Zn | |||
| Sweet Susana | 19.54 b | 4.55 a | 2.55 a | 225 b | 425 a | 20.75 a | 3.28 a | 4.28 a | 0.92 a | 11.3 a |
| Sweet Caroline | 17.52 a | 4.28 a | 2.67 a | 211 a | 485 b | 20.49 a | 3.41 a | 4.33 a | 0.91 a | 12.3 b |
| Sorghum Variety | VOC | TR | IR | Number on Dendrogram (Figure 6) and Clusters | |
|---|---|---|---|---|---|
| Sweet Caroline | Nonanoic acid | 12.03 | 1269 | 36 | D |
| 2,4-Di-tert-butylphenol | 15.29 | 1518 | 85 | A | |
| trans-Calamenene | 15.54 | 1545 | 96 | C | |
| Actinidiolide, dihydro- | 15.72 | 1564 | 101 | C | |
| Spathulenol | 16.07 | 1602 | 112 | B | |
| Caryophyllene oxide | 16.14 | 1613 | 113 | B | |
| Humulene-1,2-epoxide | 16.34 | 1644 | 120 | C | |
| Humulenol-II | 16.49 | 1667 | 125 | D | |
| Azulene, 1,4-dimethyl-7-(1-methylethyl)- | 16.7 | 1698 | 132 | D | |
| Oplopanone | 17.07 | 1770 | 153 | D | |
| Tricyclo [5.4.3.0(1,8)]tetradecan-6-one, 4-ethenyl-3-hydroxy-2,4,7,14-tetramethyl | 18.03 | 179 | D | ||
| Sweet Susana | 2-Pinen-4-one | 11.42 | 1225 | 30 | D |
| Nonanoic acid | 12.03 | 1268 | 36 | D | |
| Copaene | 13.31 | 1360 | 55 | D | |
| β-Elemen | 13.89 | 1402 | 70 | C | |
| Cyclobuta [1,2:3,4]dicyclooctene, hexadecahydro | 14.12 | 1421 | 73 | C | |
| Caryophyllene | 14.38 | 1442 | 74 | D | |
| Humulene | 14.84 | 1478 | 76 | C | |
| 2,4-Di-tert-butylphenol | 15.29 | 1518 | 85 | A | |
| Cadine-3,9-diene | 15.49 | 1539 | 94 | D | |
| Actinidiolide, dihydro- | 15.73 | 1565 | 101 | C | |
| Spathulenol | 16.08 | 1603 | 112 | B | |
| Caryophyllene oxide | 16.14 | 1613 | 113 | B | |
| Humulene-1,2-epoxide | 16.34 | 1644 | 120 | C | |
| Azulene, 1,4-dimethyl-7-(1-methylethyl)- | 16.7 | 1698 | 132 | D | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Przybylska-Balcerek, A.; Frankowski, J.; Sieracka, D.; Sázavská, T.; Wacławek, S.; Raczak, B.K.; Szwajkowska-Michałek, L.; Buśko, M.; Graczyk, M.; Niedziela, G.; et al. The Content of Antioxidant Compounds and VOCs in Sorghum Grain Grown in Central and Eastern Europe. Agronomy 2024, 14, 217. https://doi.org/10.3390/agronomy14010217
Przybylska-Balcerek A, Frankowski J, Sieracka D, Sázavská T, Wacławek S, Raczak BK, Szwajkowska-Michałek L, Buśko M, Graczyk M, Niedziela G, et al. The Content of Antioxidant Compounds and VOCs in Sorghum Grain Grown in Central and Eastern Europe. Agronomy. 2024; 14(1):217. https://doi.org/10.3390/agronomy14010217
Chicago/Turabian StylePrzybylska-Balcerek, Anna, Jakub Frankowski, Dominika Sieracka, Tereza Sázavská, Stanisław Wacławek, Barbara Klaudia Raczak, Lidia Szwajkowska-Michałek, Maciej Buśko, Małgorzata Graczyk, Grażyna Niedziela, and et al. 2024. "The Content of Antioxidant Compounds and VOCs in Sorghum Grain Grown in Central and Eastern Europe" Agronomy 14, no. 1: 217. https://doi.org/10.3390/agronomy14010217
APA StylePrzybylska-Balcerek, A., Frankowski, J., Sieracka, D., Sázavská, T., Wacławek, S., Raczak, B. K., Szwajkowska-Michałek, L., Buśko, M., Graczyk, M., Niedziela, G., & Stuper-Szablewska, K. (2024). The Content of Antioxidant Compounds and VOCs in Sorghum Grain Grown in Central and Eastern Europe. Agronomy, 14(1), 217. https://doi.org/10.3390/agronomy14010217








