Microsurgical Strategies in Post-Radiation and Revision Breast Reconstruction: Optimizing Outcomes in High-Risk Patients
Simple Summary
Abstract
1. Introduction
2. Patient Evaluation and Risk Stratification
3. Flap Selection and Design
3.1. Abdominal-Based Flaps: The Gold Standard
3.2. Alternative Donor Sites
3.3. Modifying Flaps for Irradiated Fields
3.4. Hybrid and Fat-Augmented Flaps
4. Intraoperative Techniques and Adjuncts
5. Recipient Vessel Strategies in the Vessel-Depleted Chest
5.1. The Internal Mammary System: First Line but Not Infallible
5.2. The Thoracodorsal System: Secondary but Valuable
5.3. Salvage Options in the Vessel-Depleted Chest
5.4. Vein Grafts and Arteriovenous (AV) Loops
5.5. Intraoperative Decision-Making and Contingency Planning
6. Revision-Specific Strategies
6.1. Management of IBBR Explantation and Re-Reconstruction
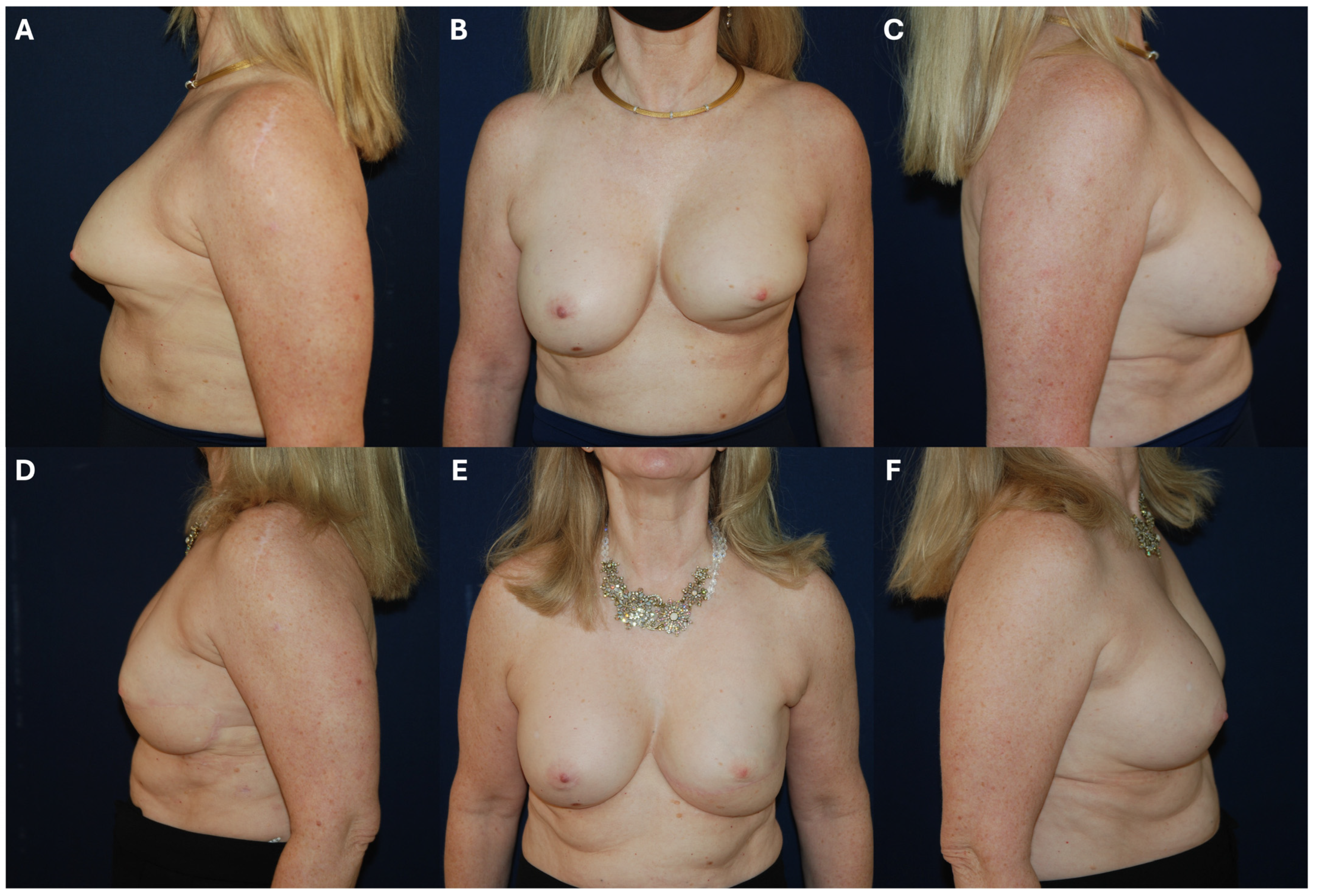
6.2. Management of Unsatisfactory Cosmetic Outcomes
7. Postoperative Management and Recovery
8. Outcomes
8.1. Flap Survival and Complication Rates
8.2. Aesthetic and Functional Outcomes
8.3. Evidence Gaps and Limitations
9. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Santosa, K.B.; Qi, J.; Kim, H.M.; Hamill, J.B.; Wilkins, E.G.; Pusic, A.L. Long-term Patient-Reported Outcomes in Postmastectomy Breast Reconstruction. JAMA Surg. 2018, 153, 891–899. [Google Scholar] [CrossRef]
- Lee, C.-H.; Cheng, M.-H.; Wu, C.-W.; Kuo, W.-L.; Yu, C.-C.; Huang, J.-J. Nipple-sparing Mastectomy and Immediate Breast Reconstruction After Recurrence From Previous Breast Conservation Therapy. Ann. Plast. Surg. 2019, 82, S95–S102. [Google Scholar] [CrossRef]
- Rubenstein, R.N.; Nelson, J.A.; Azoury, S.C.; Kim, M.B.; Boe, L.A.; Chu, J.J.; Stern, C.S.; Tadros, A.B.; Mehrara, B.J.; Matros, E.M. Disparity Reduction in U.S. Breast Reconstruction: An Analysis from 2005 to 2017 Using 3 Nationwide Data Sets. Plast. Reconstr. Surg. 2024, 154, 1065e–1075e. [Google Scholar] [CrossRef]
- Lisa, A.V.E.; Salgarello, M.; Huscher, A.; Corsi, F.; Piovani, D.; Rubbino, F.; Andreoletti, S.; Papa, G.; Klinger, F.; Tinterri, C.; et al. The Effect of Adjuvant Radiotherapy on One- and Two-Stage Prosthetic Breast Reconstruction and on Autologous Reconstruction: A Multicenter Italian Study among 18 Senonetwork Breast Centres. Breast J. 2023, 2023, 6688466. [Google Scholar] [CrossRef] [PubMed]
- Heiman, A.J.; Gabbireddy, S.R.; Kotamarti, V.S.; Ricci, J.A. A Meta-Analysis of Autologous Microsurgical Breast Reconstruction and Timing of Adjuvant Radiation Therapy. J. Reconstr. Microsurg. 2020, 37, 336–345. [Google Scholar] [CrossRef] [PubMed]
- Casella, D.; Calabrese, C.; Orzalesi, L.; Gaggelli, I.; Cecconi, L.; Santi, C.; Murgo, R.; Rinaldi, S.; Regolo, L.; Amanti, C.; et al. Current trends and outcomes of breast reconstruction following nipple-sparing mastectomy: Results from a national multicentric registry with 1006 cases over a 6-year period. Breast Cancer 2016, 24, 451–457. [Google Scholar] [CrossRef] [PubMed]
- Tran, N.V.; Chang, D.W.; Gupta, A.; Kroll, S.S.; Robb, G.L. Comparison of Immediate and Delayed Free TRAM Flap Breast Reconstruction in Patients Receiving Postmastectomy Radiation Therapy. Plast. Reconstr. Surg. 2001, 108, 78–82. [Google Scholar] [CrossRef]
- Roostaeian, J.; Yoon, A.P.; Ordon, S.; Gold, C.; Crisera, C.; Festekjian, J.; Da Lio, A.; Lipa, J.E. Impact of Prior Tissue Expander/Implant on Postmastectomy Free Flap Breast Reconstruction. Plast. Reconstr. Surg. 2016, 137, 1083–1091. [Google Scholar] [CrossRef]
- Polanco, T.O.; Shamsunder, M.G.; Parikh, R.P.M.; Chu, J.J.B.; McCarthy, C.M.; Tadros, A.; Matros, E.M.; Disa, J.J.; Mehrara, B.J.; Allen, R.J.; et al. Quality of Life in Breast Reconstruction Patients after Irradiation to Tissue Expander: A Propensity-Matched Preliminary Analysis. Plast. Reconstr. Surg. 2023, 152, 259–269. [Google Scholar] [CrossRef]
- Costanzo, D.; Klinger, M.; Lisa, A.; Maione, L.; Battistini, A.; Vinci, V. The evolution of autologous breast reconstruction. Breast J. 2020, 26, 2223–2225. [Google Scholar] [CrossRef]
- Nguyen, C.L.; Dayaratna, N.; Easwaralingam, N.; Seah, J.L.; Azimi, F.; Mak, C.; Pulitano, C.; Warrier, S.K. Developing an Indocyanine Green Angiography Protocol for Predicting Flap Necrosis During Breast Reconstruction. Surg. Innov. 2025, 32, 77–84. [Google Scholar] [CrossRef]
- Dalli, J.; Nguyen, C.; Jindal, A.; Epperlein, J.; Hardy, N.; Pulitano, C.; Warrier, S.; Cahill, R. A feasibility study assessing quantitative indocyanine green angiographic predictors of reconstructive complications following nipple-sparing mastectomy. JPRAS Open 2024, 40, 32–47. [Google Scholar] [CrossRef]
- Tange, F.; Verduijn, P.; Mulder, B.S.; van Capelle, L.; Koning, S.; Driessen, C.; Mureau, M.; Vahrmeijer, A.; van der Vorst, J. Near-infrared fluorescence angiography with indocyanine green for perfusion assessment of DIEP and msTRAM flaps: A Dutch multicenter randomized controlled trial. Contemp. Clin. Trials Commun. 2023, 33, 101128. [Google Scholar] [CrossRef] [PubMed]
- Chow, L.; Dziegielewski, P.; Chim, H. The Role of Computed Tomography Angiography in Perforator Flap Planning. Oral Maxillofac. Surg. Clin. N. Am. 2024, 36, 525–535. [Google Scholar] [CrossRef]
- Cowan, R.; Mann, G.; Salibian, A.A. Ultrasound in Microsurgery: Current Applications and New Frontiers. J. Clin. Med. 2024, 13, 3412. [Google Scholar] [CrossRef]
- Lese, I.; Biedermann, R.; Constantinescu, M.; Grobbelaar, A.O.; Olariu, R. Predicting risk factors that lead to free flap failure and vascular compromise: A single unit experience with 565 free tissue transfers. J. Plast. Reconstr. Aesthetic Surg. 2021, 74, 512–522. [Google Scholar] [CrossRef] [PubMed]
- Matkin, A.; Redwood, J.; Webb, C.; Temple-Oberle, C. Exploring breast surgeons’ reasons for women not undergoing immediate breast reconstruction. Breast 2022, 63, 37–45. [Google Scholar] [CrossRef] [PubMed]
- Elver, A.A.; Egan, K.G.; Cullom, M.E.; Nazir, N.; Johnson, B.M.; Limpiado, M.; Holding, J.; Lai, E.C.; Butterworth, J.A. A Paradigm Shift: Outcomes of Early Autologous Breast Reconstruction after Radiation Therapy. J. Reconstr. Microsurg. 2022, 39, 111–119. [Google Scholar] [CrossRef] [PubMed]
- Arnautovic, A.; Karinja, S.; Olafsson, S.; Carty, M.J.; Erdmann-Sager, J.; Caterson, S.A.; Broyles, J.M. Optimal Timing of Delayed Microvascular Breast Reconstruction after Radiation Therapy. J. Reconstr. Microsurg. 2022, 39, 165–170. [Google Scholar] [CrossRef]
- Heidekrueger, P.I.; Moellhoff, N.; Horch, R.E.; Lohmeyer, J.A.; Marx, M.; Heitmann, C.; Fansa, H.; Geenen, M.; Gabka, C.J.; Handstein, S.; et al. Overall Complication Rates of DIEP Flap Breast Reconstructions in Germany—A Multi-Center Analysis Based on the DGPRÄC Prospective National Online Registry for Microsurgical Breast Reconstructions. J. Clin. Med. 2021, 10, 1016. [Google Scholar] [CrossRef]
- Boyd, C.J.; Sorenson, T.J.; Hemal, K.; Karp, N.S. Maximizing volume in autologous breast reconstruction: Stacked/conjoined free flaps. Gland. Surg. 2023, 12, 687–695. [Google Scholar] [CrossRef]
- Haddock, N.T.; Teotia, S.S. Modern Approaches to Alternative Flap-Based Breast Reconstruction. Clin. Plast. Surg. 2023, 50, 325–335. [Google Scholar] [CrossRef]
- Salibian, A.A.; Bekisz, J.M.; Frey, J.D.; Nolan, I.T.; Kaoutzanis, C.; Yu, J.W.; Levine, J.P.; Karp, N.S.; Choi, M.; Thanik, V.D. Comparing outcomes between stacked/conjoined and non-stacked/conjoined abdominal microvascular unilateral breast reconstruction. Microsurgery 2020, 41, 240–249. [Google Scholar] [CrossRef]
- Ahmed, Z.; Ioannidi, L.M.; Ghali, S.F.; Hamilton, S.F.; Shtarbanov, P.; Mosahebi, A.F.; Nikkhah, D.F. A Single-center Comparison of Unipedicled and Bipedicled Diep Flap Early Outcomes in 98 Patients. Plast. Reconstr. Surg. Glob. Open 2023, 11, e5089. [Google Scholar] [CrossRef]
- Haddock, N.T.; Teotia, S.S.; Farr, D. Robotic Nipple-Sparing Mastectomy and Breast Reconstruction with Profunda Artery Perforator Flaps. Plast. Reconstr. Surg. 2025, 156, 337e–341e. [Google Scholar] [CrossRef] [PubMed]
- Jo, T.; Kim, E.K.; Eom, J.S.; Han, H.H. Comparison of transverse upper gracilis and profunda femoris artery perforator flaps for breast reconstruction: A systematic review. Microsurgery 2020, 40, 916–928. [Google Scholar] [CrossRef] [PubMed]
- Charlès, L.M.; Dabi, Y.; Mernier, T.; Lellouch, A.G.; Lantieri, L. Comparison of DIEP and PAP free flaps for breast reconstruction in the context of breast cancer: A retrospective study of 677 patients over 10 years. J. Plast. Reconstr. Aesthetic Surg. 2024, 101, 141–149. [Google Scholar] [CrossRef] [PubMed]
- Haddock, N.T.; Ercan, A.; Teotia, S.S. Bilateral Simultaneous Lumbar Artery Perforator Flaps in Breast Reconstruction: Perioperative Outcomes Addressing Safety and Feasibility. Plast. Reconstr. Surg. 2023, 153, 895e–901e. [Google Scholar] [CrossRef]
- Haddock, N.T.; Lakatta, A.C.B.; Steppe, C.B.; Teotia, S.S. DIEP Flap versus PAP Flap versus LAP Flap: A Propensity-Matched Analysis of Aesthetic Outcomes, Complications, and Satisfaction. Plast. Reconstr. Surg. 2024, 154, 41S–51S. [Google Scholar] [CrossRef]
- Opsomer, D.; Vyncke, T.; Ryx, M.; Van Landuyt, K.; Blondeel, P.; Stillaert, F. Donor Site Morbidity after Lumbar Artery Perforator Flap Breast Reconstruction. J. Reconstr. Microsurg. 2021, 38, 129–136. [Google Scholar] [CrossRef]
- Haddock, N.T.; Teotia, S.S. Lumbar Artery Perforator Flap: Initial Experience with Simultaneous Bilateral Flaps for Breast Reconstruction. Plast. Reconstr. Surg. Glob. Open 2020, 8, e2800. [Google Scholar] [CrossRef] [PubMed]
- Steele, T.N.; Teotia, S.S.; Haddock, N.T. Multi-Flap Microsurgical Autologous Breast Reconstruction. J. Clin. Med. 2024, 13, 5324. [Google Scholar] [CrossRef]
- Karir, A.; Stein, M.J.M.; Zhang, J.M. The Conjoined TUGPAP Flap for Breast Reconstruction: Systematic Review and Illustrative Anatomy. Plast. Reconstr. Surg. Glob. Open 2021, 9, e3512. [Google Scholar] [CrossRef]
- Andree, C.; Munder, B.I.; Seidenstuecker, K.; Richrath, P.; Behrendt, P.; Köppe, T.; Hagouan, M.; Audretsch, W.; Nestle-Kraemling, C.; Witzel, C. Skin-sparing mastectomy and immediate reconstruction with DIEP flap after breast-conserving therapy. Med. Sci. Monit. 2012, 18, CR716–CR720. [Google Scholar] [CrossRef]
- Dvali, L.T.M.; Dagum, A.B.M.; Pang, C.Y.; Kerluke, L.D.M.; Catton, P.M.; Pennock, P.D.; Mahoney, J.L.M. Effect of Radiation on Skin Expansion and Skin Flap Viability in Pigs. Plast. Reconstr. Surg. 2000, 106, 624–629. [Google Scholar] [CrossRef]
- Demiri, E.C.; Dionyssiou, D.D.; Tsimponis, A.; Goula, C.-O.; Pavlidis, L.C.; Spyropoulou, G.-A. Outcomes of Fat-Augmented Latissimus Dorsi (FALD) Flap Versus Implant-Based Latissimus Dorsi Flap for Delayed Post-radiation Breast Reconstruction. Aesthetic Plast. Surg. 2018, 42, 692–701. [Google Scholar] [CrossRef]
- Kim, Y.H.; Lee, J.S.; Park, J.; Lee, J.; Park, H.Y.; Yang, J.D. Aesthetic outcomes and complications following post-mastectomy radiation therapy in patients undergoing immediate extended latissimus dorsi flap reconstruction and implant insertion. Gland. Surg. 2021, 10, 2095–2103. [Google Scholar] [CrossRef]
- Rigotti, G.; Marchi, A.; Galiè, M.; Baroni, G.; Benati, D.; Krampera, M.; Pasini, A.; Sbarbati, A. Clinical Treatment of Radiotherapy Tissue Damage by Lipoaspirate Transplant: A Healing Process Mediated by Adipose-Derived Adult Stem Cells. Plast. Reconstr. Surg. 2007, 119, 1409–1422. [Google Scholar] [CrossRef]
- Salgarello, M.; Visconti, G.; Farallo, E. Autologous Fat Graft in Radiated Tissue Prior to Alloplastic Reconstruction of the Breast: Report of Two Cases. Aesthetic Plast. Surg. 2009, 34, 5–10. [Google Scholar] [CrossRef] [PubMed]
- Salgarello, M.; Visconti, G.; Barone-Adesi, L. Fat Grafting and Breast Reconstruction with Implant. Plast. Reconstr. Surg. 2012, 129, 317–329. [Google Scholar] [CrossRef] [PubMed]
- Serra-Renom, J.M.; Muñoz-Olmo, J.L.; Serra-Mestre, J.M. Fat Grafting in Postmastectomy Breast Reconstruction with Expanders and Prostheses in Patients Who Have Received Radiotherapy: Formation of New Subcutaneous Tissue. Plast. Reconstr. Surg. 2010, 125, 12–18. [Google Scholar] [CrossRef]
- Panettiere, P.; Marchetti, L.; Accorsi, D. The Serial Free Fat Transfer in Irradiated Prosthetic Breast Reconstructions. Aesthetic Plast. Surg. 2009, 33, 695–700. [Google Scholar] [CrossRef]
- Fodor, J.; Gulyás, G.; Polgár, C.; Major, T.; Kásler, M. Radiotherapy and breast reconstruction: The issue of compatibility. Orvosi Hetil. 2003, 144, 549–555. [Google Scholar]
- Al Qurashi, A.A.; Mardan, Q.N.M.S.; Alzahrani, I.A.; AlAlwan, A.Q.; Bafail, A.; Adeen, A.M.A.; Albahrani, A.; Aledwani, B.N.; Halawani, I.R.; AlBattal, N.Z.; et al. Efficacy of Exclusive Fat Grafting for Breast Reconstruction: An Updated Systematic Review and Meta-analysis. Aesthetic Plast. Surg. 2024, 48, 4979–4985. [Google Scholar] [CrossRef]
- Beque, M.; Aubry, G.; Bichet, J.-C.; Uzan, C.; Canlorbe, G. Comparison of Breast Reconstruction Strategies Using Exclusive Lipofilling or Prosthesis After Total Mastectomy for Breast Cancer: Satisfaction and Morbidity After More Than 2 Years. Aesthetic Surg. J. 2025, sjaf174. [Google Scholar] [CrossRef]
- Giannakaki, A.-G.; Papachatzopoulou, E.; Papapanagiotou, I.; Koura, S.; Baroutis, D.; Marinopoulos, S.; Daskalakis, G.; Dimitrakakis, C. The Role of Exclusive Autologous Lipotransfer in Non-Irradiated Breasts After Mastectomy. J. Clin. Med. 2025, 14, 4468. [Google Scholar] [CrossRef] [PubMed]
- Hembd, A.S.; Yan, J.; Zhu, H.; Haddock, N.T.; Teotia, S.S. Intraoperative Assessment of DIEP Flap Breast Reconstruction Using Indocyanine Green Angiography: Reduction of Fat Necrosis, Resection Volumes, and Postoperative Surveillance. Plast. Reconstr. Surg. 2020, 146, 1e–10e. [Google Scholar] [CrossRef]
- Chun, L.; Fu, Z.; Zheng, Y.; Lin, H.; Hu, Y.; Li, D. The impact of indocyanine green fluorescence imaging on skin flap necrosis: A systematic review and meta-analysis. BMC Surg. 2025, 25, 311. [Google Scholar] [CrossRef]
- Duggal, C.S.; Madni, T.; Losken, A. An Outcome Analysis of Intraoperative Angiography for Postmastectomy Breast Reconstruction. Aesthetic Surg. J. 2014, 34, 61–65. [Google Scholar] [CrossRef] [PubMed]
- Diep, G.K.; Hui, J.Y.C.; Marmor, S.; Cunningham, B.L.; Choudry, U.; Portschy, P.R.; Tuttle, T.M. Postmastectomy Reconstruction Outcomes After Intraoperative Evaluation with Indocyanine Green Angiography Versus Clinical Assessment. Ann. Surg. Oncol. 2016, 23, 4080–4085. [Google Scholar] [CrossRef] [PubMed]
- Pruimboom, T.; Schols, R.M.; Van Kuijk, S.M.; Van der Hulst, R.R.; Qiu, S.S. Indocyanine green angiography for preventing postoperative mastectomy skin flap necrosis in immediate breast reconstruction. Cochrane Database Syst. Rev. 2020, 2020, CD013280. [Google Scholar] [CrossRef]
- Kleiss, S.F.; Michi, M.; Schuurman, S.N.; de Vries, J.-P.P.; Werker, P.M.; de Jongh, S.J. Tissue perfusion in DIEP flaps using Indocyanine Green Fluorescence Angiography, Hyperspectral imaging, and Thermal imaging. JPRAS Open 2024, 41, 61–74. [Google Scholar] [CrossRef] [PubMed]
- Nahabedian, M. The Internal Mammary Artery and Vein as Recipient Vessels for Microvascular Breast Reconstruction. Ann. Plast. Surg. 2012, 68, 537–538. [Google Scholar] [CrossRef] [PubMed]
- Bouaoud, J.; Honart, J.-F.; Bennis, Y.; Leymarie, N. How to manage calcified vessels for head and neck microsurgical reconstruction. J. Stomatol. Oral Maxillofac. Surg. 2020, 121, 439–441. [Google Scholar] [CrossRef]
- Lemdani, M.S.; Crystal, D.T.; Ewing, J.N.; Gala, Z.; Amro, C.; Azoury, S.C.; Broach, R.B.; Rhemtulla, I.A.; Serletti, J.M. Reevaluation of Recipient Vessel Selection in Breast Free Flap Reconstruction. Microsurgery 2024, 44, e31222. [Google Scholar] [CrossRef] [PubMed]
- Banwell, M.; Trotter, D.; Ramakrishnan, V. The Thoracodorsal Artery and Vein as Recipient Vessels for Microsurgical Breast Reconstruction. Ann. Plast. Surg. 2012, 68, 542–543. [Google Scholar] [CrossRef]
- Teotia, S.S.; Cho, M.-J.; Haddock, N.T. Salvaging Breast Reconstruction: Profunda Artery Perforator Flaps Using Thoracodorsal Vessels. Plast. Reconstr. Surg. Glob. Open 2018, 6, e1837. [Google Scholar] [CrossRef]
- Yamamoto, T.; Kageyama, T.; Sakai, H.; Fuse, Y.; Tsukuura, R.; Yamamoto, N. Thoracoacromial artery and vein as main recipient vessels in deep inferior epigastric artery perforator (DIEP) flap transfer for breast reconstruction. J. Surg. Oncol. 2021, 123, 1232–1237. [Google Scholar] [CrossRef]
- Changchien, C.-H.; Fang, C.-L.; Hsu, C.-H.; Yang, H.-Y.; Lin, Y.-L. Creating a context for recipient vessel selection in deep inferior epigastric perforator flap breast reconstruction. J. Plast. Reconstr. Aesthetic Surg. 2023, 84, 618–625. [Google Scholar] [CrossRef]
- Chen, C.-K.; Tai, H.-C.; Chien, H.-F.; Chen, Y.-B.T. Various Modifications to Internal Mammary Vessel Anastomosis in Breast Reconstruction with Deep Inferior Epigastric Perforator Flap. J. Reconstr. Microsurg. 2010, 26, 219–223. [Google Scholar] [CrossRef]
- Follmar, K.E.; Prucz, R.B.; Manahan, M.A.; Magarakis, M.; Rad, A.N.; Rosson, G.D. Internal Mammary Intercostal Perforators instead of the True Internal Mammary Vessels as the Recipient Vessels for Breast Reconstruction. Plast. Reconstr. Surg. 2011, 127, 34–40. [Google Scholar] [CrossRef] [PubMed]
- Kapila, A.K.; Wakure, A.; Morgan, M.; Belgaumwala, T.; Ramakrishnan, V. Characteristics and outcomes of primary interposition vascular grafts in free flap breast reconstruction. J. Plast. Reconstr. Aesthetic Surg. 2020, 73, 2142–2149. [Google Scholar] [CrossRef]
- Otsuka, W.; Karakawa, R.; Yoshimatsu, H.; Yano, T. Breast reconstruction using a superficial inferior epigastric artery flap with pedicle elongation via an arteriovenous loop: A case report. Microsurgery 2024, 44, e31183. [Google Scholar] [CrossRef]
- Flores, J.I.; Rad, A.N.; Shridharani, S.M.; Stapleton, S.M.; Rosson, G.D. Saphenous vein grafts for perforator flap salvage in autologous breast reconstruction. Microsurgery 2008, 29, 236–239. [Google Scholar] [CrossRef]
- Chang, E.I.; Fearmonti, R.M.; Chang, D.W.; Butler, C.E. Cephalic Vein Transposition versus Vein Grafts for Venous Outflow in Free-flap Breast Reconstruction. Plast. Reconstr. Surg. Glob. Open 2014, 2, e141. [Google Scholar] [CrossRef] [PubMed]
- Silhol, T.; Suffee, T.; Hivelin, M.; Lantieri, L. Déroutage de la veine céphalique dans la reconstruction mammaire par lambeaux libres: Note technique. Ann. De Chir. Plast. Esthet. 2018, 63, 75–80. [Google Scholar] [CrossRef] [PubMed]
- Langdell, H.C.; Shammas, R.L.; Atia, A.; Chang, E.I.; Matros, E.M.; Phillips, B.T.M. Vein Grafts in Free Flap Reconstruction: Review of Indications and Institutional Pearls. Plast. Reconstr. Surg. 2022, 149, 742–749. [Google Scholar] [CrossRef]
- Haque, S.; Kanapathy, M.; Bollen, E.; Mosahebi, A.; Younis, I. Patient-reported outcome and cost implication of acute salvage of infected implant-based breast reconstruction with negative pressure wound therapy with Instillation (NPWTi) compared to standard care. J. Plast. Reconstr. Aesthetic Surg. 2021, 74, 3300–3306. [Google Scholar] [CrossRef]
- Gowda, M.S.; Jafferbhoy, S.; Marla, S.; Narayanan, S.; Soumian, S. A Simple Technique Using Peri-Prosthetic Irrigation Improves Implant Salvage Rates in Immediate Implant-Based Breast Reconstruction. Medicina 2023, 59, 2039. [Google Scholar] [CrossRef]
- Ahmed, S.; Hulsman, L.B.; Imeokparia, F.; Ludwig, K.; Fisher, C.M.; Bamba, R.; Danforth, R.; VonDerHaar, R.J.; Lester, M.E.; Hassanein, A.H.M. Implant-based Breast Reconstruction Salvage with Negative Pressure Wound Therapy with Instillation: An Evaluation of Outcomes. Plast. Reconstr. Surg. Glob. Open 2024, 12, e6116. [Google Scholar] [CrossRef]
- De Pellegrin, L.; Zucal, I.; Treglia, G.; Parodi, C.; Schweizer, R.; De Monti, M.; Harder, Y. Salvage of the Mastectomy Pocket in Infected Implant-Based Breast Reconstruction Using Negative-Pressure Wound Therapy with Instillation and Dwell: A Systematic Review and Meta-Analysis. J. Clin. Med. 2025, 14, 2730. [Google Scholar] [CrossRef]
- Coriddi, M.; Shenaq, D.; Kenworthy, E.; Mbabuike, J.; Nelson, J.; Pusic, A.; Mehrara, B.; Disa, J.J. Autologous Breast Reconstruction after Failed Implant-Based Reconstruction: Evaluation of Surgical and Patient-Reported Outcomes and Quality of Life. Plast. Reconstr. Surg. 2019, 143, 373–379. [Google Scholar] [CrossRef]
- Bitoiu, B.; Schlagintweit, S.; Zhang, Z.; Bovill, E.; Isaac, K.; Macadam, S. Conversion from Alloplastic to Autologous Breast Reconstruction: What Are the Inciting Factors? Plast. Surg. 2022, 32, 213–219. [Google Scholar] [CrossRef] [PubMed]
- Bigdeli, A.K.; Tee, J.W.; Vollbach, F.H.; Diehm, Y.F.; Falkner, F.; Strübing, F.; Mahrhofer, M.; Gazyakan, E.; Kneser, U.; Siegwart, L.C. Microsurgical breast reconstruction—A salvage option for failed implant-based breast reconstruction. Breast 2025, 82, 104480. [Google Scholar] [CrossRef]
- Holmes, W.J.; Quinn, M.; Emam, A.T.; Ali, S.R.; Prousskaia, E.; Wilson, S.M. Salvage of the failed implant-based breast reconstruction using the Deep Inferior Epigastric Perforator Flap: A single centre experience with tertiary breast reconstruction. J. Plast. Reconstr. Aesthetic Surg. 2019, 72, 1075–1083. [Google Scholar] [CrossRef]
- Karp, N.; Sorenson, T.J.; Boyd, C.J.M.M.; Hemal, K.; Lin, A.; Robinson, I.S.; Choi, M. The GalaFLEX “Empanada” for Direct-to-Implant Prepectoral Breast Reconstruction. Plast. Reconstr. Surg. 2024, 155, 488e–491e. [Google Scholar] [CrossRef]
- Sorenson, T.J.; Boyd, C.J.; Hemal, K.; Park, J.J.; Amro, C.; Vernice, N.; Lakatta, A.; Cohen, O.; Choi, M.; Karp, N. Outcome of Prepectoral Direct-to-Implant Breast Reconstruction With the Poly-4-hydroxybutyrate Wrap. Plast. Reconstr. Surg. Glob. Open 2025, 13, e7268. [Google Scholar] [CrossRef] [PubMed]
- Rich, M.D.; Sorenson, T.J.; Lamba, A.; Hillard, C.; Mahajan, A. Routine Preoperative Nutritional Optimization Not Required in Patients Undergoing Breast Reconstruction. Breast Care 2022, 17, 495–500. [Google Scholar] [CrossRef] [PubMed]
- Chi, D.; Chen, A.D.; Ha, A.Y.; Yaeger, L.H.M.; Lee, B.T.M. Comparative Effectiveness of Transversus Abdominis Plane Blocks in Abdominally Based Autologous Breast Reconstruction. Ann. Plast. Surg. 2020, 85, e76–e83. [Google Scholar] [CrossRef]
- Park, R.H.; Chou, J.; DeVito, R.G.; Elmer, A.; Hollenbeck, S.T.; Campbell, C.A.; Stranix, J.T. Effectiveness of Liposomal Bupivacaine Transversus Abdominis Plane Block in DIEP Flap Breast Reconstruction: A Randomized Controlled Trial. Plast. Reconstr. Surg. 2024, 154, 52S–59S. [Google Scholar] [CrossRef]
- Foppiani, J.; Lee, T.C.; Alvarez, A.H.; Escobar-Domingo, M.J.; Taritsa, I.C.; Lee, D.; Schuster, K.; Wood, S.; Utz, B.; Bai, C.; et al. Beyond Surgery: Psychological Well-Being’s Role in Breast Reconstruction Outcomes. J. Surg. Res. 2024, 305, 26–35. [Google Scholar] [CrossRef]
- Prantl, L.; Moellhoff, N.; von Fritschen, U.; Giunta, R.; Germann, G.; Kehrer, A.; Thiha, A.; Ehrl, D.; Zeman, F.M.; Broer, P.N.; et al. Effect of Radiation Therapy on Microsurgical Deep Inferior Epigastric Perforator Flap Breast Reconstructions. Ann. Plast. Surg. 2020, 86, 627–631. [Google Scholar] [CrossRef]
- Ly, C.L.; Khetpal, S.; Heller, D.R.; Higgins, S.A.; Killelea, B.; Alperovich, M.; Avraham, T. Comparing complications in irradiated and non-irradiated free-flaps in patients with bilateral immediate breast reconstruction and unilateral post-mastectomy radiotherapy. Microsurgery 2021, 41, 615–621. [Google Scholar] [CrossRef]
- Clemens, M.W.; Kronowitz, S.J. Current perspectives on radiation therapy in autologous and prosthetic breast reconstruction. Gland Surg. 2015, 4, 222–231. [Google Scholar] [CrossRef]
- Rogers, N.E.; Allen, R.J. Radiation Effects on Breast Reconstruction with the Deep Inferior Epigastric Perforator Flap. Plast. Reconstr. Surg. 2002, 109, 1919–1924. [Google Scholar] [CrossRef] [PubMed]
- Song, J.H.; Kim, Y.S.; Jung, B.K.; Lee, D.W.; Song, S.Y.; Roh, T.S.; Lew, D.H. Salvage of Infected Breast Implants. Arch. Plast. Surg. 2017, 44, 516–522. [Google Scholar] [CrossRef] [PubMed]
- Francis, S.D.M.; Thawanyarat, K.B.; Johnstone, T.M.B.; Yesantharao, P.S.; Kim, T.S.B.; Rowley, M.A.B.; Sheckter, C.C.; Nazerali, R.S.M. How Postoperative Infection Affects Reoperations after Implant-based Breast Reconstruction: A National Claims Analysis of Abandonment of Reconstruction. Plast. Reconstr. Surg. Glob. Open 2023, 11, e5040. [Google Scholar] [CrossRef]
- Kim, M.B.; Vingan, P.B.; Boe, L.A.; Mehrara, B.J.; Stern, C.S.; Allen, R.J.; Nelson, J.A. Satisfaction with Breasts following Autologous Reconstruction: Assessing Associated Factors and the Impact of Revisions. Plast. Reconstr. Surg. 2024, 155, 235–244. [Google Scholar] [CrossRef] [PubMed]
- Zong, A.M.; Leibl, K.E.; Weichman, K.E. Effects of Elective Revision after Breast Reconstruction on Patient-Reported Outcomes. J. Reconstr. Microsurg. 2024, 41, 100–112. [Google Scholar] [CrossRef]
- Shiraishi, M.; Sowa, Y.; Tsuge, I.; Kodama, T.; Inafuku, N.; Morimoto, N. Long-Term Patient Satisfaction and Quality of Life Following Breast Reconstruction Using the BREAST-Q: A Prospective Cohort Study. Front. Oncol. 2022, 12, 815498. [Google Scholar] [CrossRef]
- Pusic, A.L.; Matros, E.; Fine, N.; Buchel, E.; Gordillo, G.M.; Hamill, J.B.; Kim, H.M.; Qi, J.; Albornoz, C.; Klassen, A.F.; et al. Patient-Reported Outcomes 1 Year After Immediate Breast Reconstruction: Results of the Mastectomy Reconstruction Outcomes Consortium Study. J. Clin. Oncol. 2017, 35, 2499–2506. [Google Scholar] [CrossRef] [PubMed]
- Toyserkani, N.M.; Jørgensen, M.G.; Tabatabaeifar, S.; Damsgaard, T.; Sørensen, J.A. Autologous versus implant-based breast reconstruction: A systematic review and meta-analysis of Breast-Q patient-reported outcomes. J. Plast. Reconstr. Aesthetic Surg. 2020, 73, 278–285. [Google Scholar] [CrossRef]
- Sadok, N.; Refaee, M.S.; Eltahir, Y.; de Bock, G.H.; van Veen, M.M.; Werker, P.M.N. Quality of Life 9 to 13 Years after Autologous or Alloplastic Breast Reconstruction: Which Breast Remains Best? Plast. Reconstr. Surg. 2022, 151, 467–476. [Google Scholar] [CrossRef] [PubMed]
- von Glinski, M.; Holler, N.; Kümmel, S.; Reinisch, M.; Wallner, C.; Wagner, J.M.; Dadras, M.; Sogorski, A.; Lehnhardt, M.; Behr, B. Autologous Reconstruction After Failed Implant-Based Breast Reconstruction. Ann. Plast. Surg. 2023, 91, 42–47. [Google Scholar] [CrossRef] [PubMed]

| Recipient Vessel | Advantages | Disadvantages | Ideal Scenarios |
|---|---|---|---|
| Internal Mammary Vessels (IMV) |
|
|
|
| Thoracodorsal Vessels (TDV) |
|
|
|
| Circumflex Scapular Vessels |
|
|
|
| Cephalic Vein (for venous outflow) |
|
|
|
| Transverse Cervical Vessels |
|
|
|
| Contralateral IMV (Cross-chest) |
|
|
|
| AV Loop Graft (saphenous vein graft to axillary or subclavian) |
|
|
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sorenson, T.J.; Boyd, C.J.; Cohen, O.; Choi, M.; Karp, N. Microsurgical Strategies in Post-Radiation and Revision Breast Reconstruction: Optimizing Outcomes in High-Risk Patients. Cancers 2025, 17, 3831. https://doi.org/10.3390/cancers17233831
Sorenson TJ, Boyd CJ, Cohen O, Choi M, Karp N. Microsurgical Strategies in Post-Radiation and Revision Breast Reconstruction: Optimizing Outcomes in High-Risk Patients. Cancers. 2025; 17(23):3831. https://doi.org/10.3390/cancers17233831
Chicago/Turabian StyleSorenson, Thomas J., Carter J. Boyd, Oriana Cohen, Mihye Choi, and Nolan Karp. 2025. "Microsurgical Strategies in Post-Radiation and Revision Breast Reconstruction: Optimizing Outcomes in High-Risk Patients" Cancers 17, no. 23: 3831. https://doi.org/10.3390/cancers17233831
APA StyleSorenson, T. J., Boyd, C. J., Cohen, O., Choi, M., & Karp, N. (2025). Microsurgical Strategies in Post-Radiation and Revision Breast Reconstruction: Optimizing Outcomes in High-Risk Patients. Cancers, 17(23), 3831. https://doi.org/10.3390/cancers17233831







