Effect of Low Dose of Fumonisins on Pig Health: Immune Status, Intestinal Microbiota and Sensitivity to Salmonella
Abstract
:1. Introduction
2. Results
2.1. Sphinganine/Sphingosine (Sa/So) Ratios in Pig Serum and Tissues
| Days post-fumonisin exposure | Parameter | Groups | Kidneys (nmol/L) | Liver (nmol/L) | Serum (nmol/L) |
|---|---|---|---|---|---|
| Day + 2 | Sa | F(−) | ND | ND | 15.36 ± 1.26 |
| F(+) | ND | ND | 16.56 ± 1.30 | ||
| So | F(−) | ND | ND | 76.67 ± 6.24 | |
| F(+) | ND | ND | 77.07 ± 6.22 | ||
| Sa/So | F(−) | ND | ND | 0.20 ± 0.01 | |
| F(+) | ND | ND | 0.22 ± 0.01 | ||
| Day + 9 | Sa | F(−) | 0.32 ± 0.02 a | 0.40 ± 0.03 a | 12.65 ± 1.77 |
| F(+) | 0.46 ± 0.03 b | 0.96 ± 0.11 b | 13.15 ± 0.57 | ||
| So | F(−) | 5.86 ± 0.39 | 3.32 ± 0.24 a | 76.45 ± 10.55 | |
| F(+) | 7.18 ± 0.68 | 5.30 ± 0.56 b | 54.91 ± 2.76 | ||
| Sa/So | F(−) | 0.05 ± 0.00 a | 0.12 ± 0.01 a | 0.17 ± 0.01 a | |
| F(+) | 0.07 ± 0.00 b | 0.18 ± 0.00 b | 0.24 ± 0.01 b | ||
| Day + 63 | Sa | F(−) | 0.33 ± 0.03 a | 0.31 ± 0.03 | 7.14 ± 0.52 a |
| F(+) | 0.72 ± 0.10 b | 0.47 ± 0.07 | 18.10 ± 1.75 b | ||
| So | F(−) | 6.88 ± 0.71 a | 2.94 ± 0.36 | 54.34 ± 3.46 | |
| F(+) | 9.28 ± 0.81 b | 3.05 ± 0.64 | 55.78 ± 4.90 | ||
| Sa/So | F(−) | 0.05 ± 0.00 a | 0.11 ± 0.00 a | 0.13 ± 0.00 a | |
| F(+) | 0.08 ± 0.00 b | 0.17 ± 0.01 b | 0.33 ± 0.02 b |
2.2. Growth Performance and Health Status
2.3. Serologic Status
| Groups Time | D + 2 | D + 7 | D + 14 | D + 21 | D + 28 | D + 35 | D + 42 | D + 49 |
|---|---|---|---|---|---|---|---|---|
| F(−)S(+) | 0/8 | 0/8 | 1/8 | 7/8 | 7/8b | 7/8 | 7/8 | 7/8 |
| F(+)S(+) | 0/8 | 0/8 | 2/8 | 4/8 | 2/8a | 3/8 | 3/8 | 4/8 |
2.4. Immune Status
2.4.1. Non-Specific Cellular Immune Response
2.4.2. Specific Immune Response
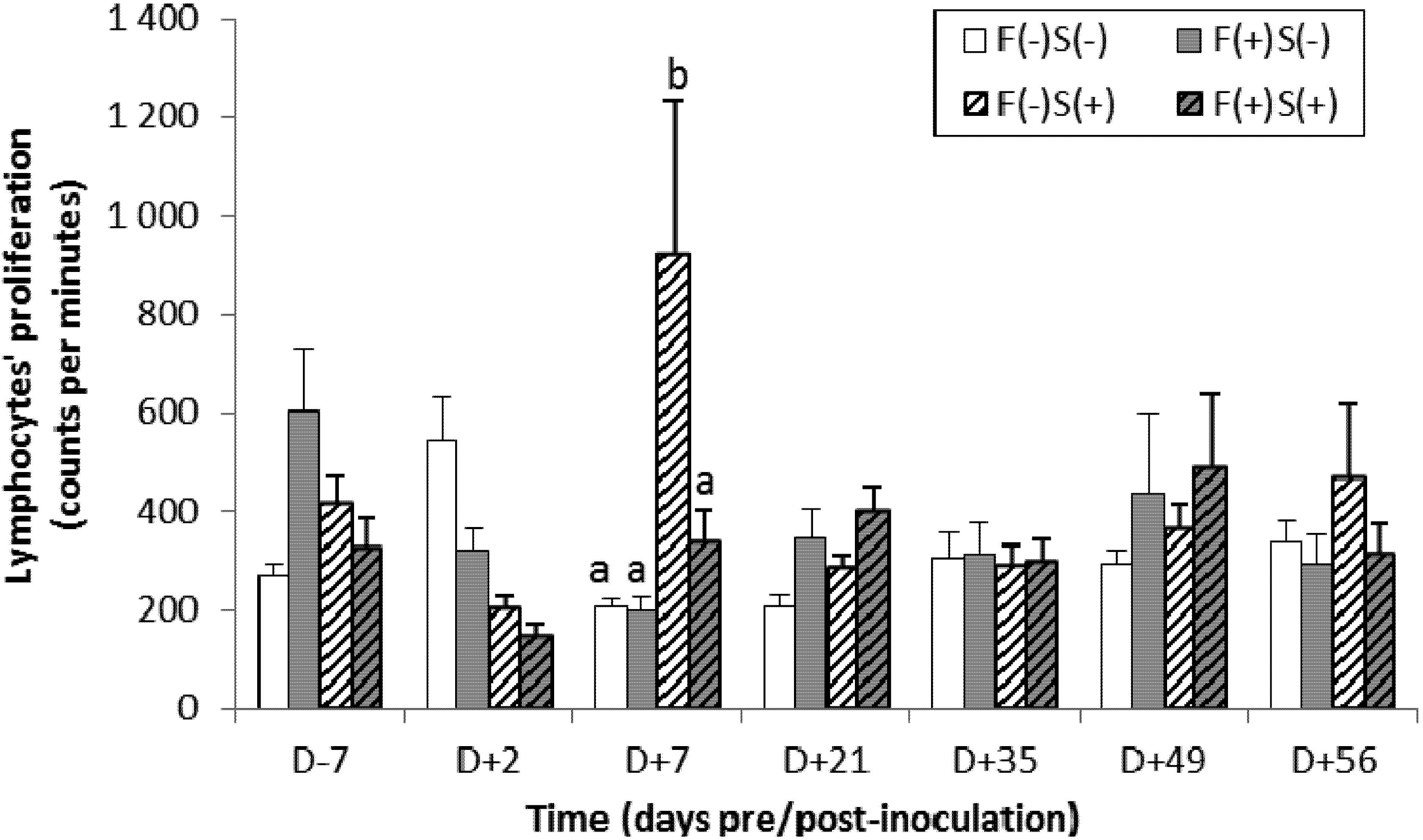
2.5. Bacteriological Status of the Animals
2.5.1. Changes over Time in the Percentage of Excretory Pigs and in the Intensity of Excretion
| Time (days pre-/post-inoculation) | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| D − 7 | D + 2 | D + 7 | D + 14 | D + 21 | D + 28 | D + 35 | D + 42 | D + 49 | ||
| Excretory pigs 1 | F(−)S(+) | 0 | 8/12 | 7/8 | 8/8 | 7/8 | 7/8 | 7/8 | 7/8 | 7/8 |
| F(+)S(+) | 0 | 11/12 | 5/8 | 4/8 | 4/8 | 4/8 | 4/8 | 4/8 | 4/8 | |
| Salmonella concentration 2 | F(−)S(+) | - | 2.7 ± 0.7 | 2.9 ± 0.3 | 2.7 ± 0.2 | 1.2 ± 0.3 | 1.1 ± 0.5 | 0.4 ± 0.5 | 0.7 ± 0.5 | 1.1 ± 0.6 |
| F(+)S(+) | - | 2.7 ± 0.2 | 2.9 ± 0.3 | 1.6 ± 0.6 | 1.2 ± 0.5 | 1.1 ± 0.5 | 0.8 ± 0.4 | 1.4 ± 0.8 | 1.0 ± 0.5 | |
2.5.2. Research and Counting of Salmonella typhimurium in Intestinal Contents and Tissues
| D + 2 (n = 4) | D + 56 (n = 8) | |||||||
|---|---|---|---|---|---|---|---|---|
| F(−)S(+) | F(+)S(+) | F(−)S(+) | F(+)S(+) | |||||
| Nb 1 | Conc. 2 | Nb 1 | Conc. 2 | Nb 1 | Conc. 2 | Nb 1 | Conc. 2 | |
| Liver, spleen, muscle | 0/4 | - | 0/4 | - | 0/8 | - | 0/8 | - |
| Mesenteric nodes | 2/4 | 1.3 ± 0.5 | 3/4 | 1.6 ± 0.4 | 0/8 | - | 2/8 | <0.1 |
| Ileum content | 2/4 | 1.7 ± 0.6 | 2/4 | 2.4 ± 0.6 | 3/8 | 2.1 ± 0.6 | 3/8 | 2.7 ± 0.6 |
| Colon content | 3/4 | 0.5 ± 0.3 | 3/4 | 2.0 ± 1.4 | 6/8 | 1.6 ± 0.4 | 3/8 | 2.4 ± 0.4 |
| Caecum content | 3/4 | <0.1 | 3/4 | 2.1 ± 1.9 | 7/8 | 2.1 ± 0.4 | 3/8 | 2.6 ± 0.7 |
2.5.3. Study of the Translocation of Salmonella into Organs
2.6. The Intestinal Microbiota of the Animals
2.6.1. Mesophylic Aerobic Counts
2.6.2. Faecal Microbiota SSCP-Profiles
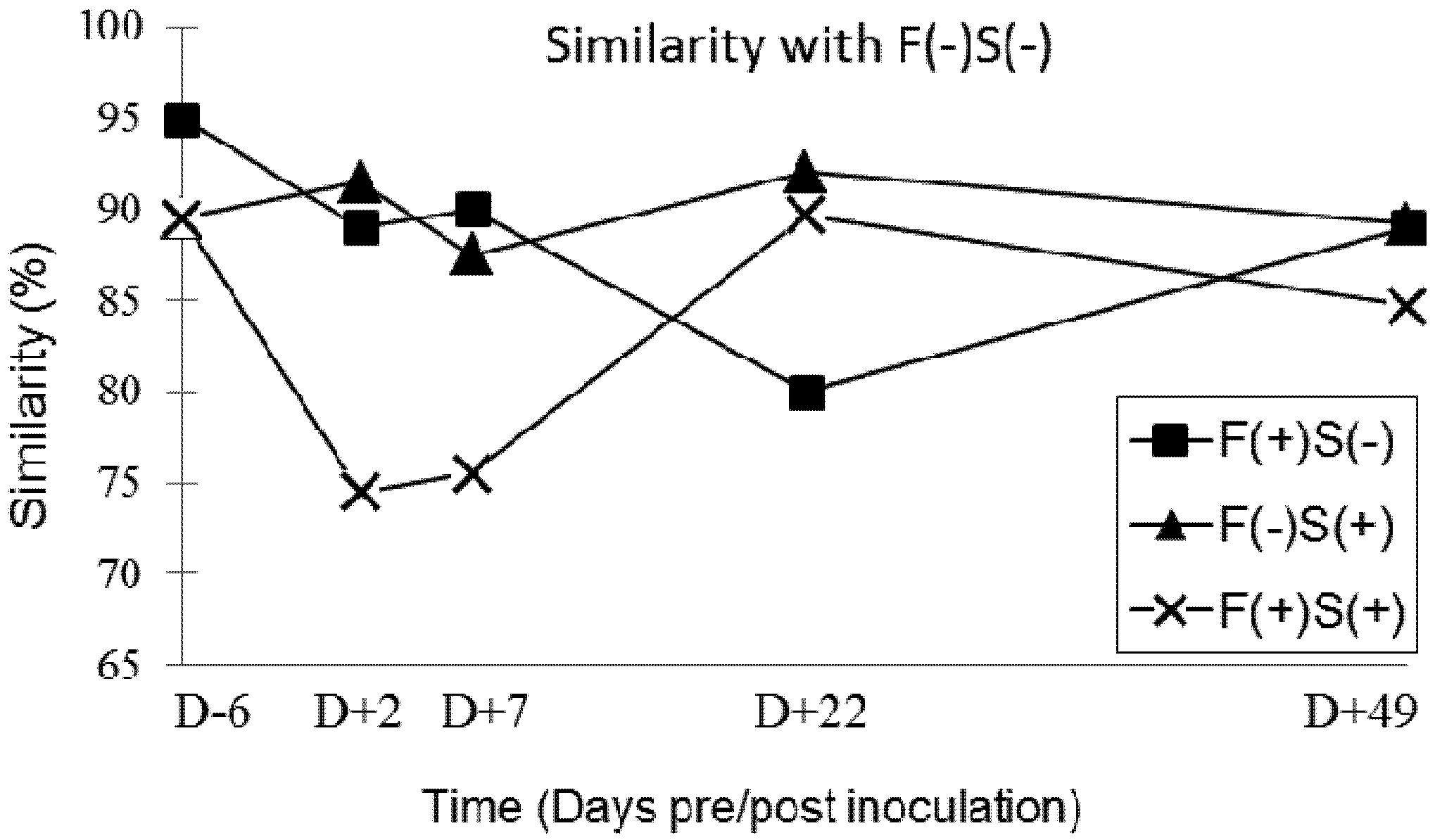
3. Discussion
3.1. Intoxication of Pigs by Fumonisins
3.2. Impact of FB1 on the Asymptomatic Carriage of Salmonella
3.3. The Impact of FB1 on Immune Status and Bacterial Infection
| Oswald et al. (2003) | Devriendt et al. (2009) | Our study | |
|---|---|---|---|
| Age of pigs at FB1 exposure | 3 weeks | 4 weeks | 7 weeks |
| Status of pigs | Conventional | Conventional | SPF |
| Estimated FB1 dietary concentration | 6.5 ppm FB1 | 13 ppm FB11.9 ppm FB22.2 ppm FB3 | 8.6 ppm FB13.2 ppm FB2 |
| FB1 presentation | Crude extract | Crude extract | Maize naturally contaminated |
| FB1 distribution | Gavage | Gavage | In feed |
| Age of pigs at inoculation | 4 weeks | 6 weeks | 8 weeks |
| Pathogens inoculated | E. coli (ExPEC strain) 1 × 109 CFU | E. coli (F4+ ETEC strain) 1010 CFU | S. typhimurium 5 × 104 CFU |
| FB1 as predisposing factor to disease | Yes | Yes | Questionable |
3.4. Faecal Microbiota Profiles in Groups of Pigs Exposed to Fumonisin and/or Salmonella
4. Experimental Section
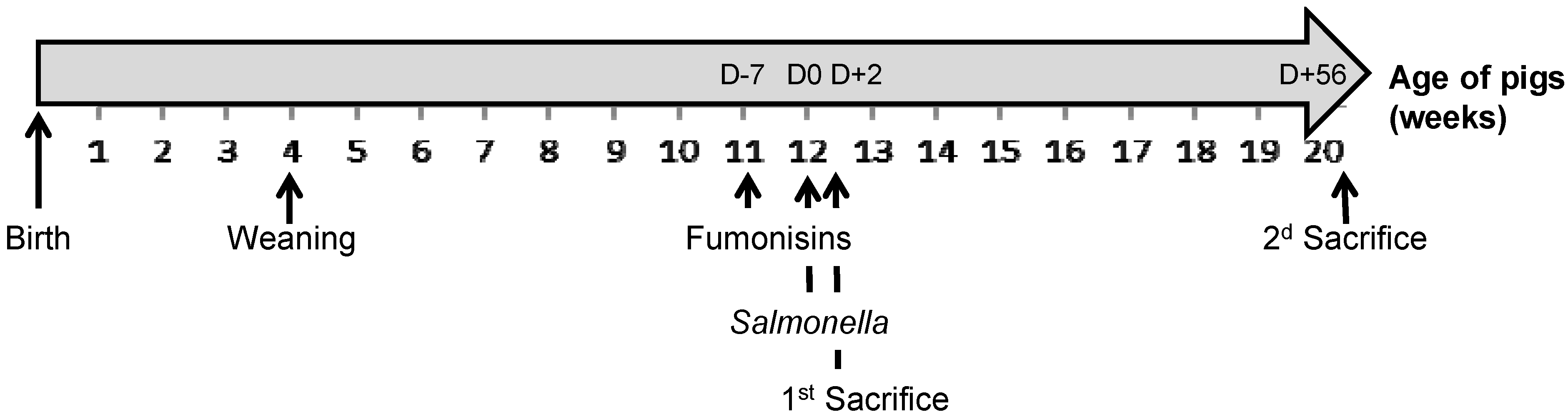
4.1. Animals and Housing
4.2. Feed
| Diets | Control F(−) | Contaminated F(+) |
|---|---|---|
| Ingredients (%) | ||
| Uncontaminated maize | 20.4 | 5.4 |
| Contaminated maize | 0 | 15 |
| Barley | 45 | 45 |
| Soya meal 48 | 16 | 16 |
| Dehydrated alfalfa | 10 | 10 |
| Oats | 4 | 4 |
| Calcium phosphorus | 2 | 2 |
| Vitamins, minerals, oligo-elements | 2.7 | 2.7 |
| Calculated composition 1 | ||
| Dry matter (g/kg) | 874 | |
| Whole Cellulose (g/kg) | 66 | |
| Fat (g/kg) | 24 | |
| Starch (g/kg) | 385 | |
| Net energy (MJ/kg) | 8.63 | |
| Gross Protein (g/kg) | 157 | |
| Ash (g/kg) | 68 | |
| Measured mycotoxin content (ppb) 2 | ||
| Fumonisin B1 (FB1) | ND (<10) | 8616 |
| Fumonisin B2 (FB2) | ND (<50) | 3205 |
| FB1 + FB2 | ND (<50) | 11,821 |
| Aflatoxin B1 | <0.1 | <0.1–0.24 |
| Ochratoxin 3 | ND (<0.5) | ND (<0.5) |
| Zearalenone | ND (<40) | ND (<40) |
| Trichothecenes type A 4 | ||
| Monoacetoxyscirpenol | ND (<40) | ND (<40) |
| Diacetoxyscirpenol | ND (<40) | ND (<40) |
| T-2 toxin | ND (<50) | ND (<50) |
| T-2 triol | ND (<40) | ND (<40) |
| T-2 tetraol | ND (<40) | ND (<40) |
| HT-2 toxin | ND (<40) | ND (<40) |
| Trichothecenes B 4 | ||
| Deoxynivalenol | 112–131 | 64–114 |
| Neosolaniol | ND (<40) | ND (<40) |
| 3 acetyl-deoxynivalenol | ND (<40) | ND (<40) |
| 15 acetyl-deoxynivalenol | ND (<40) | ND (<40) |
| Fusarenon X | ND (<40) | ND (<40) |
| Nivalenol | ND (<50) | ND (<50) |
4.3. Experimental Design
4.4. Animal Growth Performance and Sample Collection
4.5. Evaluation of Fumonisin Exposure: The Sa/So Ratio
4.6. Specific Serology for Salmonella typhimurium
4.6.1. Qualitative Method
4.6.2. Quantitative Method
4.7. Lymphocyte Proliferation Following Stimulation with a Mitogenic Agent or Specific Antigen
4.8. Salmonella Detection and Count in Faeces and Tissues
4.9. Aerobic Mesophilic Bacteria (AMB) Numeration in Faeces
4.10. Salmonella Immunohistochemical Analysis of the Peyer’s Patches
4.11. Capillary Single-Stranded Conformation Polymorphism (CE-SSCP) Analysis
4.11.1. DNA Extraction/Amplification
4.11.2. PCR Amplification
4.11.3. CE-SSCP Electrophoresis
4.12. Statistical Analysis
5. Conclusions
Acknowledgements
Conflicts of interest
References
- The Commission of the European Communities. Commission Regulation (EC) No 1881/2006 of 19 December 2006 setting maximum levels for certain contaminants in foodstuffs. Off. J. Eur. Union 2006, L364/5, 5–24.
- The Commission of the European Communities. Commission Recommendation of 17 August 2006 on the prevention and reduction of Fusarium toxins in cereals and cereal products. Off. J. Eur. Union 2006, L234/35, 35–40.
- Haschek, W.M.; Gumprecht, L.A.; Smith, G.; Tumbleson, M.E.; Constable, P.D. Fumonisin toxicosis in swine: An overview of porcine pulmonary edema and current perspectives. Environ. Health Perspect. 2001, 109, 251–257. [Google Scholar]
- Pettersson, H. Mycotoxin Contamination of Animal Feed. In Animal Feed Contamination: Effects on Livestock and Food Safety; Fink-Gremmels, J., Ed.; Woodhead Publishing Series in Food Science, Technology and Nutrition: Cambridge, UK, 2012; pp. 233–285. [Google Scholar]
- Bouhet, S.; Oswald, I.P. The intestine as a possible target for fumonisin toxicity. Mol. Nutr. Food Res. 2007, 51, 925–931. [Google Scholar] [CrossRef]
- Lessard, M.; Boudry, G.; Seve, B.; Oswald, I.P.; Lalles, J.P. Intestinal physiology and peptidase activity in male pigs are modulated by consumption of corn culture extracts containing fumonisins. J. Nutr. 2009, 139, 1303–1307. [Google Scholar] [CrossRef]
- Lalles, J.P.; Lessard, M.; Boudry, G. Intestinal barrier function is modulated by short-term exposure to fumonisin B-1 in Ussing chambers. Vet. Res. Commun. 2009, 33, 1039–1043. [Google Scholar] [CrossRef]
- Bracarense, A.P.; Lucioli, J.; Grenier, B.; Drociunas Pacheco, G.; Moll, W.D.; Schatzmayr, G.; Oswald, I.P. Chronic ingestion of deoxynivalenol and fumonisin, alone or in interaction, induces morphological and immunological changes in the intestine of piglets. Br. J. Nutr. 2012, 107, 1776–1786. [Google Scholar] [CrossRef]
- Riley, R.T.; An, N.H.; Showker, J.L.; Yoo, H.S.; Norred, W.P.; Chamberlain, W.J.; Wang, E.; Merrill, A.H., Jr.; Motelin, G.; Beasley, V.R.; et al. Alteration of tissue and serum sphinganine to sphingosine ratio: An early biomarker of exposure to fumonisin-containing feeds in pigs. Toxicol. Appl. Pharmacol. 1993, 118, 105–112. [Google Scholar] [CrossRef]
- Zomborszky-Kovacs, M.; Vetesi, F.; Horn, P.; Repa, I.; Kovacs, F. Effects of prolonged exposure to low-dose fumonisin B-1 in pigs. J. Vet. Med. Ser. B 2002, 49, 197–201. [Google Scholar] [CrossRef]
- Wang, E.; Norred, W.P.; Bacon, C.W.; Riley, R.T.; Merrill, A.H. Inhibition of sphingolipid biosynthesis by fumonisins—Implications for diseases associated with fusarium-moniliforme. J. Biol. Chem. 1991, 266, 14486–14490. [Google Scholar]
- AFSSA, Evaluation des Risques Liés à la Présence de Mycotoxines Dans les Chaines Alimentaires Humaine et Animale. AFSSA: Maisons-Alfort, France, 2009; 339.
- Oswald, I.P.; Desautels, C.; Laffitte, J.; Fournout, S.; Peres, S.Y.; Odin, M.; le Bars, P.; le Bars, J.; Fairbrother, J.M. Mycotoxin fumonisin B-1 increases intestinal colonization by pathogenic Escherichia coli in pigs. Appl. Environ. Microbiol. 2003, 69, 5870–5874. [Google Scholar] [CrossRef]
- Devriendt, B.; Gallois, M.; Verdonck, F.; Wache, Y.; Bimczok, D.; Oswald, I.P.; Goddeeris, B.M.; Cox, E. The food contaminant fumonisin B(1) reduces the maturation of porcine CD11R1(+) intestinal antigen presenting cells and antigen-specific immune responses, leading to a prolonged intestinal ETEC infection. Vet. Res. 2009, 40, 40. [Google Scholar] [CrossRef]
- Deshmukh, S.; Asrani, R.K.; Jindal, N.; Ledoux, D.R.; Rottinghaus, G.E.; Sharma, M.; Singh, S.P. Effects of Fusarium moniliforme culture material containing known levels of fumonisin B1 on progress of Salmonella Gallinarum infection in Japanese quail: Clinical signs and hematologic studies. Avian Dis. 2005, 49, 274–280. [Google Scholar] [CrossRef]
- The Commission of the European Communities. Regulation (EC) No 2160/2003 of the European Parliament and of the Council of 17 November 2003 on the control of salmonella and other specified food-borne zoonotic agents. Off. J. Eur. Union 2003, L325/1, 1–15.
- D’Aoust, J.Y. Salmonella and the international food trade. Int. J. Food Microbiol. 1994, 24, 11–31. [Google Scholar] [CrossRef]
- Patten, R.C. Salmonellosis. Am. Fam. Physician 1981, 23, 113–117. [Google Scholar]
- Berends, B.R.; VanKnapen, F.; Snijders, J.M.A.; Mossel, D.A.A. Identification and quantification of risk factors regarding Salmonella spp. On pork carcasses. Int. J. Food Microbiol. 1997, 36, 199–206. [Google Scholar] [CrossRef]
- Steinbach, G.; Kroell, U. Salmonella infections in swine herds—Epidemiology and importance for human illnesses. Deutsch. Tierarztl. Wochenschr. 1999, 106, 282–288. [Google Scholar]
- Mikkelsen, L.L.; Naughton, P.J.; Hedemann, M.S.; Jensen, B.B. Effects of physical properties of feed on microbial ecology and survival of Salmonella enterica serovar Typhimurium in the pig gastrointestinal tract. Appl. Environ. Microbiol. 2004, 70, 3485–3492. [Google Scholar] [CrossRef]
- Hopwood, D.E.; Pethick, D.W.; Hampson, D.J. Increasing the viscosity of the intestinal contents stimulates proliferation of enterotoxigenic Escherichia coli and Brachyspira pilosicoli in weaner pigs. Br. J. Nutr. 2002, 88, 523–532. [Google Scholar] [CrossRef]
- Becker, B.; Bresch, H.; Schillinger, U.; Thiel, P.G. The effect of fumonisin B-1 on the growth of bacteria. World J. Microbiol. Biotechnol. 1997, 13, 539–543. [Google Scholar] [CrossRef]
- Tenk, I.; Fodor, E.; Szathmary, C. The effect of pure fusarium toxin-t-2, toxin-f-2 and toxin-das on the microflora of the gut and on plasma glucocorticoid levels in rat and swine. Zentralblatt Bakteriol. Mikrobiol. Hyg. Ser. A 1982, 252, 384–393. [Google Scholar]
- Verbrugghe, E.; Vandenbroucke, V.; Dhaenens, M.; Shearer, N.; Goossens, J.; de Saeger, S.; Eeckhout, M.; D’Herde, K.; Thompson, A.; Deforce, D.; et al. T-2 toxin induced Salmonella typhimurium intoxication results in decreased Salmonella numbers in the cecum contents of pigs, despite marked effects on Salmonella-host cell interactions. Vet. Res. 2012, 43, 22. [Google Scholar] [CrossRef]
- Wache, Y.J.; Valat, C.; Postollec, G.; Bougeard, S.; Burel, C.; Oswald, I.P.; Fravalo, P. Impact of deoxynivalenol on the intestinal microflora of pigs. Int. J. Mol. Sci. 2009, 10, 1–17. [Google Scholar]
- Cariolet, R.; Le Diguerher, G.; Ecobichon, P.; Julou, P.; Jolly, J.-P.; Madec, F. Production of Long Term, Low-Cost Specific Pathogen Free Pigs. In Proceedings of Animal Production in Europe: The Way Forward in a Changing World, International Society for Animal Hygiene: Saint Malo, France, 11—13 October 2004; p. 149.
- Solfrizzo, M.; Avantaggiato, G.; Visconti, A. Rapid method to determine sphinganine/sphingosine in human and animal urine as a biomarker for fumonisin exposure. J. Chromatogr. B 1997, 692, 87–93. [Google Scholar] [CrossRef]
- Dilkin, P.; Direito, G.; Simas, M.M.S.; Mallmann, C.A.; Correa, B. Toxicokinetics and toxicological effects of single oral dose of fumonisin B1 containing Fusarium verticillioides culture material in weaned piglets. Chem. Biol. Interact. 2010, 185, 157–162. [Google Scholar] [CrossRef]
- Zomborszky, M.K.; Vetesi, F.; Repa, I.; Kovacs, F.; Bata, A.; Horn, P.; Toth, A.; Romvari, R. Experiment to determine limits of tolerance for fumonisin B1 in weaned piglets. J. Vet. Med. Ser. B 2000, 47, 277–286. [Google Scholar]
- Rotter, B.A.; Thompson, B.K.; Prelusky, D.B.; Trenholm, H.L.; Stewart, B.; Miller, J.D.; Savard, M.E. Response of growing swine to dietary exposure to pure fumonisin B1 during an eight-week period: Growth and clinical parameters. Nat. Toxins 1996, 4, 42–50. [Google Scholar] [CrossRef]
- Dilkin, P.; Zorzete, P.; Mallmann, C.A.; Gomes, J.D.F.; Utiyama, C.E.; Oetting, L.L.; Correa, B. Toxicological effects of chronic low doses of aflatoxin B-1 and fumonisin B-1-containing Fusarium mondiforme culture material in weaned piglets. Food Chem. Toxicol. 2003, 41, 1345–1353. [Google Scholar] [CrossRef]
- Grenier, B.; Loureiro-Bracarense, A.P.; Lucioli, J.; Pacheco, G.D.; Cossalter, A.M.; Moll, W.D.; Schatzmayr, G.; Oswald, I.P. Individual and combined effects of subclinical doses of deoxynivalenol and fumonisins in piglets. Mol. Nutr. Food Res. 2011, 55, 761–771. [Google Scholar] [CrossRef]
- Fravalo, P.; Cariolet, R.; Proux, K.; Salvat, G. Le portage asymptomatique de Salmonella enterica par les porcs: Résultats issus de la constitution d’un modèle en conditions expérimentales. J. Rech. Porcine 2003, 35, 393–400. [Google Scholar]
- Oswald, I.P.; Coméra, C. Immunotoxicity of mycotoxins. Rev. Méd. Vét. 1998, 149, 585–590. [Google Scholar]
- Halloy, D.J.; Gustin, P.G.; Bouhet, S.; Oswald, I.P. Oral exposure to culture material extract containing fumonisins predisposes swine to the development of pneumonitis caused by Pasteurellamultocida. Toxicology 2005, 213, 34–44. [Google Scholar] [CrossRef]
- Posa, R.; Donko, T.; Bogner, P.; Kovacs, M.; Repa, I.; Magyar, T. Interaction of Bordetella bronchiseptica, Pasteurella multocida, and fumonisin B1 in the porcine respiratory tract as studied by computed tomography. Can. J. Vet. Res. Rev. Canadienne Rech. Vet. 2011, 75, 176–182. [Google Scholar]
- Bouhet, S.; Hourcade, E.; Loiseau, N.; Fikry, A.; Martinez, S.; Roselli, M.; Galtier, P.; Mengheri, E.; Oswald, I.P. The mycotoxin fumonisin B1 alters the proliferation and the barrier function of porcine intestinal epithelial cells. Toxicol. Sci. 2004, 77, 165–171. [Google Scholar]
- Loiseau, N.; Debrauwer, L.; Sambou, T.; Bouhet, S.; Miller, J.D.; Martin, P.G.; Viadere, J.L.; Pinton, P.; Puel, O.; Pineau, T.; et al. Fumonisin B1 exposure and its selective effect on porcine jejunal segment: Sphingolipids, glycolipids and trans-epithelial passage disturbance. Biochem. Pharmacol. 2007, 74, 144–152. [Google Scholar]
- Bouhet, S.; le Dorze, E.; Peres, S.; Fairbrother, J.M.; Oswald, I.P. Mycotoxin fumonisin B1 selectively down-regulates the basal IL-8 expression in pig intestine: In vivo and in vitro studies. Food Chem. Toxicol. 2006, 44, 1768–1773. [Google Scholar] [CrossRef]
- Taranu, I.; Marin, D.E.; Bouhet, S.; Pascale, F.; Bailly, J.D.; Miller, J.D.; Pinton, P.; Oswald, I.P. Mycotoxin fumonisin B1 alters the cytokine profile and decreases the vaccinal antibody titer in pigs. Toxicol. Sci. 2005, 84, 301–307. [Google Scholar] [CrossRef]
- Marteau, P. Factor Controlling the Bacterial Microflora. In Gut Microflora: Digestive Physiology and Pathology; Rambaud, J.C., Buts, J.P., Cortier, G., Flourié, B., Eds.; John Libbey Eurotext, DL: Paris, France, 2006; pp. 37–55. [Google Scholar]
- Proux, K.; Houdayer, C.; Humbert, F.; Cariolet, R.; Rose, V.; Eveno, E.; Madec, F. Development of a complete ELISA using Salmonella lipopolysaccharides of various serogronps allowing to detect all infected pigs. Vet. Res. 2000, 31, 481–490. [Google Scholar] [CrossRef]
- Pinton, P.; Accensi, F.; Beauchamp, E.; Cossalter, A.M.; Callu, P.; Grosjean, F.; Oswald, I.P. Ingestion of deoxynivalenol (DON) contaminated feed alters the pig vaccinal immune responses. Toxicol. Lett. 2008, 177, 215–222. [Google Scholar] [CrossRef]
- Fravalo, P.; Hascoet, Y.; Le Fellic, M.; Queguiner, S.; Petton, J.; Salvat, G. Convenient method for rapid and quantitative assessment of Salmonella enterica contamination: The mini-MSRV MPN technique. J. Rapid Methods Autom. Microbiol. 2003, 11, 81–88. [Google Scholar]
- McOrist, A.L.; Jackson, M.; Bird, A.R. A comparison of five methods for extraction of bacterial DNA from human faecal samples. J. Microbiol. Methods 2002, 50, 131–139. [Google Scholar]
- Delbes, C.; Moletta, R.; Godon, J.J. Bacterial and archaeal 16S rDNA and 16S rRNA dynamics during an acetate crisis in an anaerobic digestor ecosystem. FEMS Microbiol. Ecol. 2001, 35, 19–26. [Google Scholar] [CrossRef]
- Legendre, P.; Legendre, L. Numerical Ecology; Elsevier: Amsterdan, The Netherlands, 1998; Volume 20. [Google Scholar]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Burel, C.; Tanguy, M.; Guerre, P.; Boilletot, E.; Cariolet, R.; Queguiner, M.; Postollec, G.; Pinton, P.; Salvat, G.; Oswald, I.P.; et al. Effect of Low Dose of Fumonisins on Pig Health: Immune Status, Intestinal Microbiota and Sensitivity to Salmonella. Toxins 2013, 5, 841-864. https://doi.org/10.3390/toxins5040841
Burel C, Tanguy M, Guerre P, Boilletot E, Cariolet R, Queguiner M, Postollec G, Pinton P, Salvat G, Oswald IP, et al. Effect of Low Dose of Fumonisins on Pig Health: Immune Status, Intestinal Microbiota and Sensitivity to Salmonella. Toxins. 2013; 5(4):841-864. https://doi.org/10.3390/toxins5040841
Chicago/Turabian StyleBurel, Christine, Mael Tanguy, Philippe Guerre, Eric Boilletot, Roland Cariolet, Marilyne Queguiner, Gilbert Postollec, Philippe Pinton, Gilles Salvat, Isabelle P. Oswald, and et al. 2013. "Effect of Low Dose of Fumonisins on Pig Health: Immune Status, Intestinal Microbiota and Sensitivity to Salmonella" Toxins 5, no. 4: 841-864. https://doi.org/10.3390/toxins5040841
APA StyleBurel, C., Tanguy, M., Guerre, P., Boilletot, E., Cariolet, R., Queguiner, M., Postollec, G., Pinton, P., Salvat, G., Oswald, I. P., & Fravalo, P. (2013). Effect of Low Dose of Fumonisins on Pig Health: Immune Status, Intestinal Microbiota and Sensitivity to Salmonella. Toxins, 5(4), 841-864. https://doi.org/10.3390/toxins5040841





