The Effect of Regular Intake of Dry-Cured Ham Rich in Bioactive Peptides on Inflammation, Platelet and Monocyte Activation Markers in Humans
Abstract
:1. Introduction
2. Material and Methods
2.1. Ethics Statement
2.2. Study Design and Subjects
2.3. Meat Products
2.4. Blood Sampling and Biochemical Determinations
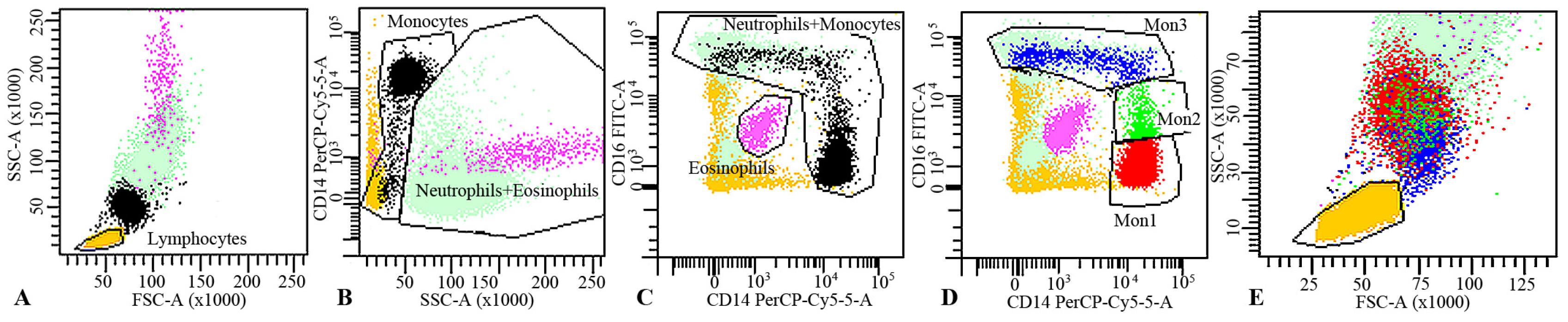
2.5. Flow Cytometry
2.6. Plasma Markers
2.7. Statistical Analyses
3. Results
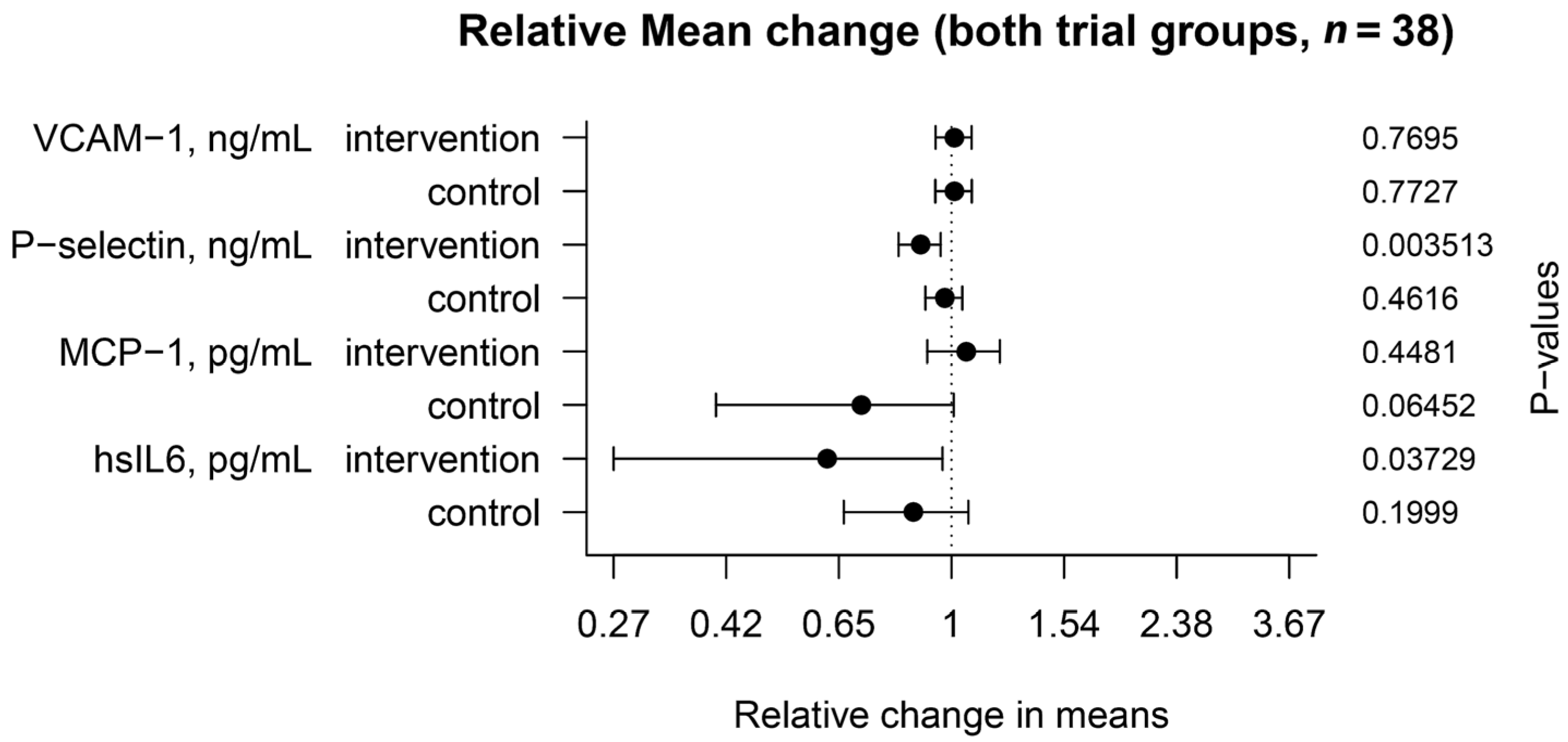
3.1. Plasmatic Markers
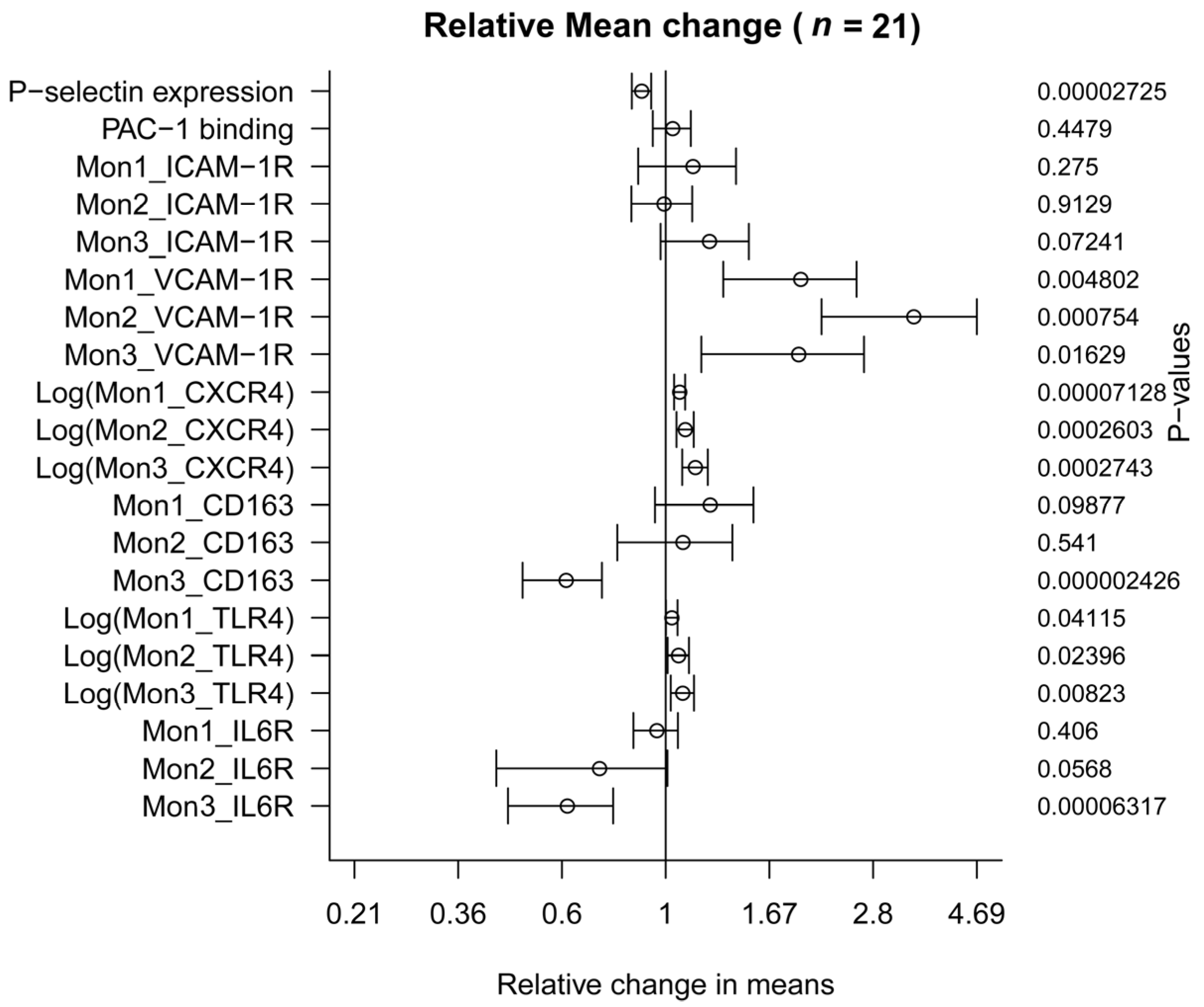
3.2. Effect on Platelet Activation
3.3. Effect on Monocytes Markers
4. Discussion
Limitations
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| ACE | Angiotensin I Converting Enzyme |
| ADP | Adenosine diphosphate |
| BMI | Body Mass Index |
| BP | Blood Pressure |
| CAD | Coronary artery Disease |
| CVD | Cardiovascular Disease |
| FSC/SSC | Forward-scattered light/Side-scattered light |
| hsIL6 | high-sensitivity Interleukin 6 |
| ICAM-1 | Intercellular Adhesion Molecule-1 |
| MCP-1 | Monocyte Chemoattractant Protein-1 |
| MIF | Mean Fluorescence Intensity |
| Mon | Monocyte |
| SDF1 | Stromal Derived Factor-1 |
| TLR4 | Toll-Like Receptor 4 |
| VCAM-1 | Vascular Cell Adhesion Molecule 1 |
| UCAM | Catholic University of Murcia |
References
- Eussen, S.R.; Verhagen, H.; Klungel, O.H.; Garssen, J.; van Loveren, H.; van Kranen, H.J.; Rompelberg, C.J. Functional foods and dietary supplements: Products at the interface between pharma and nutrition. Eur. J. Pharmacol. 2011, 668 (Suppl. S1), 2–9. [Google Scholar] [CrossRef] [PubMed]
- Aihara, K.; Osaka, M.; Yoshida, M. Oral administration of the milk casein-derived tripeptide Val-Pro-Pro attenuates high-fat diet-induced adipose tissue inflammation in mice. Br. J. Nutr. 2014, 112, 513–519. [Google Scholar] [CrossRef] [PubMed]
- Bjørndal, B.; Berge, C.; Ramsvik, M.S.; Svardal, A.; Bohov, P.; Skorve, J.; Berge, R.K. A fish protein hydrolysate alters fatty acid composition in liver and adipose tissue and increases plasma carnitine levels in a mouse model of chronic inflammation. Lipids Health Dis. 2013, 12, 143. [Google Scholar] [CrossRef] [PubMed]
- Girgih, A.T.; Alashi, A.M.; He, R.; Malomo, S.A.; Raj, P.; Netticadan, T.; Aluko, R.E. A novel hemp seed meal protein hydrolysate reduces oxidative stress factors in spontaneously hypertensive rats. Nutrients 2014, 6, 5652–5266. [Google Scholar] [CrossRef] [PubMed]
- Borissoff, J.I.; Spronk, H.M.; ten Cate, H. The hemostatic system as a modulator of atherosclerosis. N. Engl. J. Med. 2011, 364, 1746–1760. [Google Scholar] [PubMed]
- Passacquale, G.; Vamadevan, P.; Pereira, L.; Hamid, C.; Corrigall, V.; Ferro, A. Monocyte-platelet interaction induces a pro-inflammatory phenotype in circulating monocytes. PLoS ONE 2011, 6, e25595. [Google Scholar]
- Ed Rainger, G.; Chimen, M.; Harrison, M.J.; Yates, C.M.; Harrison, P.; Watson, S.P.; Lordkipanizdé, M.; Nash, G.B. The role of platelets in the recruitment of leukocytes during vascular disease. Platelets 2015, 26, 507–520. [Google Scholar] [PubMed]
- Chen, M.; Ye, X.; Ming, X.; Chen, Y.; Wang, Y.; Su, X.; Kong, Y. A Novel Direct Factor Xa Inhibitory Peptide with Anti-Platelet Aggregation Activity from Agkistrodon acutus Venom Hydrolysates. Sci. Rep. 2015, 5, 10846. [Google Scholar] [CrossRef]
- Majumder, K.; Chakrabarti, S.; Davidge, S.T.; Wu, J. Structure and activity study of egg protein ovotransferrin derived peptides (IRW and IQW) on endothelial inflammatory response and oxidative stress. J. Agric. Food Chem. 2013, 61, 2120–2129. [Google Scholar] [CrossRef] [PubMed]
- Qin, L.Q.; Xu, J.Y.; Dong, J.Y.; Zhao, Y.; van Bladeren, P.; Zhang, W. Lactotripeptides intake and blood pressure management: A meta-analysis of randomised controlled clinical trials. Nutr. Metab. Cardiovasc. Dis. 2013, 23, 395–402. [Google Scholar] [CrossRef] [PubMed]
- Bähr, M.; Fechner, A.; Kiehntopf, M.; Jahreis, G. Consuming a mixed diet enriched with lupin protein beneficially affects plasma lipids in hypercholesterolemic subjects: A randomized controlled trial. Clin. Nutr. 2015, 34, 7–14. [Google Scholar] [CrossRef] [PubMed]
- Majumder, K.; Wu, J. Molecular targets of antihypertensive peptides: Understanding the mechanisms of action based on the pathophysiology of hypertension. Int. J. Mol. Sci. 2014, 16, 256–283. [Google Scholar] [CrossRef] [PubMed]
- Ryan, J.T.; Ross, R.P.; Bolton, D.; Fitzgerald, G.F.; Stanton, C. Bioactive peptides from muscle sources: Meat and fish. Nutrients 2011, 3, 765–791. [Google Scholar] [CrossRef] [PubMed]
- Escudero, E.; Mora, L.; Fraser, P.D.; Aristoy, M.C.; Arihara, K.; Toldrá, F. Purification and Identification of antihypertensive peptides in Spanish dry-cured ham. J. Proteom. 2013, 78, 499–507. [Google Scholar] [CrossRef] [PubMed]
- Mora, L.; Gallego, M.; Escudero, E.; Reig, M.; Aristoy, M.C.; Toldrá, F. Small peptides hydrolysis in dry-cured meats. Int. J. Food Microbiol. 2015, 212, 9–15. [Google Scholar] [CrossRef] [PubMed]
- Escudero, E.; Aristoy, M.C.; Nishimura, H.; Arihara, K.; Toldrá, F. Antihypertensive effect and antioxidant activity of peptide fractions extracted from Spanish dry-cured ham. Meat Sci. 2012, 91, 306–311. [Google Scholar] [CrossRef] [PubMed]
- Escudero, E.; Mora, L.; Toldrá, F. Stability of ACE inhibitory ham peptides against heat treatment and in vitro digestion. Food Chem. 2014, 161, 305–311. [Google Scholar] [CrossRef] [PubMed]
- Jiménez-Colmenero, F.; Ventanas, J.; Toldrá, F. Nutritional composition of dry-cured ham and its role in a healthy diet. Meat Sci. 2010, 84, 585–593. [Google Scholar] [CrossRef] [PubMed]
- Gallego, M.; Mora, L.; Toldrá, F. Peptidomics as a tool for quality control in dry-cured ham processing. J. Proteom. 2016, 147, 98–107. [Google Scholar] [CrossRef] [PubMed]
- Montoro-Garcia, S.; Shantsila, E.; Hernandez-Romero, D.; Jover, E.; Valdes, M.; Marin, F.; Lip, G.Y. Small-size platelet microparticles trigger platelet and monocyte functionality and modulate thrombogenesis via P-selectin. Br. J. Haematol. 2014, 166, 571–580. [Google Scholar] [CrossRef] [PubMed]
- Shantsila, E.; Tapp, L.D.; Wrigley, B.J.; Pamukcu, B.; Apostolakis, S.; Montoro-Garcia, S.; Lip, G.Y. Monocyte subsets in coronary artery disease and their associations with markers of inflammation and fibrinolysis. Atherosclerosis 2014, 234, 4–10. [Google Scholar] [CrossRef] [PubMed]
- Micha, R.; Michas, G.; Mozaffarian, D. Unprocessed red and processed meats and risk of coronary artery disease and type 2 diabetes—An updated review of the evidence. Curr. Atheroscler. Rep. 2012, 14, 515–524. [Google Scholar] [CrossRef] [PubMed]
- Gallego, M.; Aristoy, M.C.; Toldrá, F. Dipeptidyl peptidase IV inhibitory peptides generated in Spanish dry-cured ham. Meat Sci. 2014, 96, 757–761. [Google Scholar] [CrossRef] [PubMed]
- Escudero, E.; Mora, L.; Fraser, P.D.; Aristoy, M.C.; Toldrá, F. Identification of novel antioxidant peptides generated in Spanish dry-cured ham. Food Chem. 2013, 138, 1282–1288. [Google Scholar] [CrossRef] [PubMed]
- Blann, A.D.; Nadar, S.K.; Lip, G.Y. The adhesion molecule P-selectin and cardiovascular disease. Eur. Heart J. 2003, 24, 2166–2179. [Google Scholar] [CrossRef] [PubMed]
- Kling, D.; Stucki, C.; Kronenberg, S.; Tuerck, D.; Rhéaume, E.; Tardif, J.C.; Gaudreault, J.; Schmit, C. Pharmacological control of platelet-leukocyte interactions by the human anti-P-selectin antibody inclacumab—Preclinical and clinical studies. Thromb. Res. 2013, 131, 401–410. [Google Scholar] [CrossRef] [PubMed]
- Konić-Ristić, A.; Srdić-Rajić, T.; Kardum, N.; Aleksić-Veličković, V.; Kroon, P.A.; Hollands, W.J.; Needs, P.W.; Boyko, N.; Hayran, O.; Jorjadze, M.; et al. Effects of bioactive-rich extracts of pomegranate, persimmon, nettle, dill, kale and Sideritis and isolated bioactives on arachidonic acid induced markers of platelet activation and aggregation. J. Sci. Food. Agric. 2013, 93, 3581–3587. [Google Scholar] [CrossRef] [PubMed]
- Shantsila, E.; Wrigley, B.; Tapp, L.; Apostolakis, S.; Montoro-Garcia, S.; Drayson, M.T.; Lip, G.Y. Immunophenotypic characterization of human monocyte subsets: Possible implications for cardiovascular disease pathophysiology. J. Thromb. Haemost. 2011, 9, 1056–1066. [Google Scholar] [CrossRef] [PubMed]
- Canton, J.; Neculai, D.; Grinstein, S. Scavenger receptors in homeostasis and immunity. Nat. Rev. Immunol. 2013, 13, 621–634. [Google Scholar] [CrossRef] [PubMed]
| Active Biopeptides | Dry-Cured Ham | Cooked Ham |
|---|---|---|
| KAAAAP | Identified | N.D. |
| AAPLAP | Identified | N.D. |
| KPVAAP | Identified | N.D. |
| VPPAK | Identified | N.D. |
| KPGRP | Identified | N.D. |
| IAGRP | N.D. | N.D. |
| PAAPPK | Identified | N.D. |
| TGLKP | N.D. | N.D. |
| AAATP | Identified | N.D. |
| KAAAATP | N.D. | N.D. |
| n = 38 | Volunteers |
|---|---|
| Age | 44.3 ± 5.3 |
| Gender (male) | 82% |
| BMI, (kg/m2) | 27.02 ± 3.9 |
| Fat content, % | 25.08 ± 7.3 |
| Systolic BP, mmHg | 137.4 ± 10.6 |
| Diastolic BP, mmHg | 80.0 ± 5.8 |
| n = 38 | Period of Intervention | Mean Difference | Confidence Interval | p Value | |
|---|---|---|---|---|---|
| Low 95% | Upp 95% | ||||
| Plasmatic markers, ELISA | |||||
| P-selectin, ng/mL | (1) Dry-cured | −5.00 | −8.19 | −1.80 | 0.003513 |
| (2) Control | −1.31 | −4.60 | 2.10 | 0.461629 | |
| (1)–(2) | −3.69 | −7.36 | 0.02 | 0.062522 | |
| VCAM-1, ng/mL | (1) Dry-cured | 3.23 | −17.97 | 24.42 | 0.769504 |
| (2) Control | 3.41 | −19.33 | 26.15 | 0.772709 | |
| (1)–(2) | −0.19 | −25.61 | 25.24 | 0.988745 | |
| MCP-1, pg/mL | (1) Dry-cured | 5.45 | −8.35 | 19.25 | 0.448144 |
| (2) Control | −14.39 | −29.20 | 0.42 | 0.064524 | |
| (1)–(2) | 19.84 | 3.28 | 36.40 | 0.023489 | |
| hs-IL6, pg/mL | (1) Dry-cured | −0.60 | −1.15 | −0.05 | 0.037294 |
| (2) Control | −0.39 | −0.96 | 0.19 | 0.199947 | |
| (1)–(2) | −0.52 | −0.77 | −0.04 | 0.049448 | |
| n = 21 | Baseline (before Dry-Cured Ham) | Mean Difference | Confidence Interval | p Value | |
|---|---|---|---|---|---|
| Low 95% | Upp 95% | ||||
| Platelet activation markers | |||||
| P-selectin expression, MFI | 492.67 | −55.24 | −76.32 | −34.16 | 0.0000272 |
| PAC-1 binding, MFI | 426.12 | 15.18 | −26.33 | 56.68 | 0.447 |
| Monocyte activation markers (n = 21) | |||||
| Mon1_ICAM-1R, MFI | 3799.22 | 552.94 | −484.15 | 1590.03 | 0.275 |
| Mon2_ICAM-1R, MFI | 6325.33 | −49.33 | −989.93 | 891.26 | 0.912 |
| Mon3_ICAM-1R, MFI | 4579.78 | 1114.66 | −113.87 | 2343.20 | 0.072 |
| Mon1_VCAM-1R, MFI | 672.70 | 643.25 | 222.89 | 1063.61 | 0.00480 |
| Mon2_VCAM-1R, MFI | 1243.75 | 3022.30 | 1453.88 | 4590.72 | 0.00075 |
| Mon3_VCAM-1R, MFI | 2048.55 | 1917.80 | 397.35 | 3438.25 | 0.01629 |
| Mon1_CXCR4, MFI | 1152.70 | 1019.90 | 545.43 | 1494.38 | 0.0000713 * |
| Mon2_CXCR4, MFI | 2415.00 | 4244.17 | 1313.45 | 7174.89 | 0.0002603 * |
| Mon3_CXCR4, MFI | 2215.55 | 9601.76 | 1498.90 | 17,704.61 | 0.0002743 * |
| Mon1_CD163, MFI | 1376.20 | 340.70 | −70.88 | 752.28 | 0.098 |
| Mon2_CD163, MFI | 1838.90 | 164.94 | −392.83 | 722.70 | 0.540 |
| Mon3_CD163, MFI | 2606.90 | −1016.48 | −1325.88 | −707.08 | 0.0000024 |
| Mon1_TLR4, MFI | 1050.57 | 261.05 | −44.91 | 567.00 | 0.0411524 * |
| Mon2_TLR4, MFI | 1915.72 | 2061.10 | 52.87 | 4069.33 | 0.0239622 * |
| Mon3_TLR4, MFI | 1611.00 | 3266.19 | 468.86 | 6063.52 | 0.0082297 * |
| Mon1_IL6R, MFI | 576.45 | −24.60 | −85.34 | 36.14 | 0.406 |
| Mon2_IL6R, MFI | 1186.40 | −332.05 | −674.78 | 10.68 | 0.0568 |
| Mon3_IL6R, MFI | 3820.70 | −1475.70 | −2074.37 | −877.03 | 0.0000632 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Martínez-Sánchez, S.M.; Minguela, A.; Prieto-Merino, D.; Zafrilla-Rentero, M.P.; Abellán-Alemán, J.; Montoro-García, S. The Effect of Regular Intake of Dry-Cured Ham Rich in Bioactive Peptides on Inflammation, Platelet and Monocyte Activation Markers in Humans. Nutrients 2017, 9, 321. https://doi.org/10.3390/nu9040321
Martínez-Sánchez SM, Minguela A, Prieto-Merino D, Zafrilla-Rentero MP, Abellán-Alemán J, Montoro-García S. The Effect of Regular Intake of Dry-Cured Ham Rich in Bioactive Peptides on Inflammation, Platelet and Monocyte Activation Markers in Humans. Nutrients. 2017; 9(4):321. https://doi.org/10.3390/nu9040321
Chicago/Turabian StyleMartínez-Sánchez, Sara María, Alfredo Minguela, David Prieto-Merino, María Pilar Zafrilla-Rentero, José Abellán-Alemán, and Silvia Montoro-García. 2017. "The Effect of Regular Intake of Dry-Cured Ham Rich in Bioactive Peptides on Inflammation, Platelet and Monocyte Activation Markers in Humans" Nutrients 9, no. 4: 321. https://doi.org/10.3390/nu9040321
APA StyleMartínez-Sánchez, S. M., Minguela, A., Prieto-Merino, D., Zafrilla-Rentero, M. P., Abellán-Alemán, J., & Montoro-García, S. (2017). The Effect of Regular Intake of Dry-Cured Ham Rich in Bioactive Peptides on Inflammation, Platelet and Monocyte Activation Markers in Humans. Nutrients, 9(4), 321. https://doi.org/10.3390/nu9040321








