The Effect of Breakfast Prior to Morning Exercise on Cognitive Performance, Mood and Appetite Later in the Day in Habitually Active Women
Abstract
:1. Introduction
2. Experimental Section
2.1. Pilot Study
2.1.1. Methods
Participants
Screening and Familiarisation
Experimental Procedures
2.1.2. Statistic
2.2. Main Study
2.2.1. Methods
Participants
Screening and Familiarisation
Cognitive Tasks
Visual Analogue Scales (VAS)
Preliminary Exercise Tests
Pre-Testing Procedures
Treatments/Test Meals
| kJ/Kcal | Carbohydrate (g) | Protein (g) | Fat (g) | Fibre (g) | |
|---|---|---|---|---|---|
| Breakfasts | |||||
| 20 g Special K cereal | 321/76 | 15 | 3 | 0.3 | 0.9 |
| 83 mL Semi-skimmed milk | 171/42 | 4 | 1.5 | 1.5 | <0.5 |
| Total | 492/118 | 19 | 4.5 | 1.8 | 0.9 |
| 40 g Special K cereal | 642/152 | 30 | 6 | 0.6 | 1.8 |
| 166 mL Semi-skimmed milk | 342/81 | 8 | 3 | 3 | <0.5 |
| Total | 984/236 | 38 | 9 | 3.6 | 1.8 |
| Lunch | |||||
| 125 g Penne Pasta | 1894/446 | 91 | 15 | 2 | 3 |
| 250 g Tomato and Herb pasta sauce | 540/128 | 20 | 4 | 3 | 4 |
| 15 g Olive Oil | 508/123 | <0.5 | <0.5 | 14 | <0.5 |
| 40 g Cheddar Cheese | 648/156 | <0.5 | 10 | 13 | <0.5 |
| Total per 430 g portion | 3544/938 | 111 | 29 | 32 | 7 |
| Total per 100 g | 824/218 | 26 | 7 | 7 | 1.5 |
Experimental procedures (Figure 1)
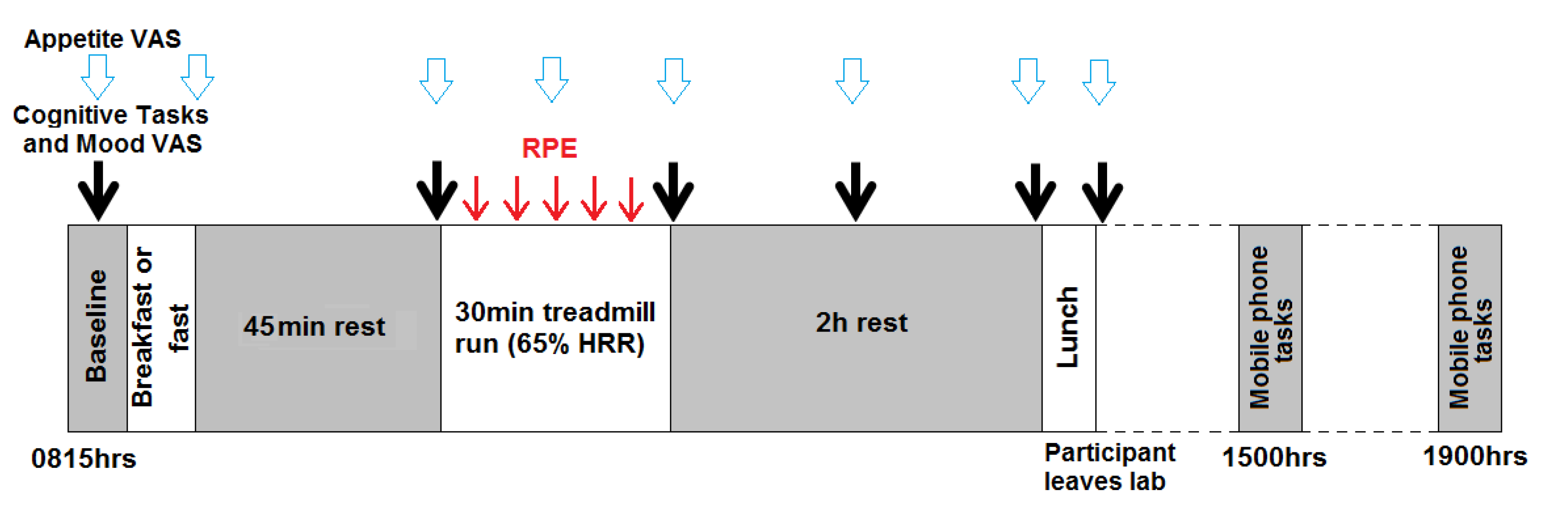
2.2.2. Statistical Analysis
3. Results
3.1. Pilot Study Results
3.2. Main Study Results
3.2.1. Cognitive Function
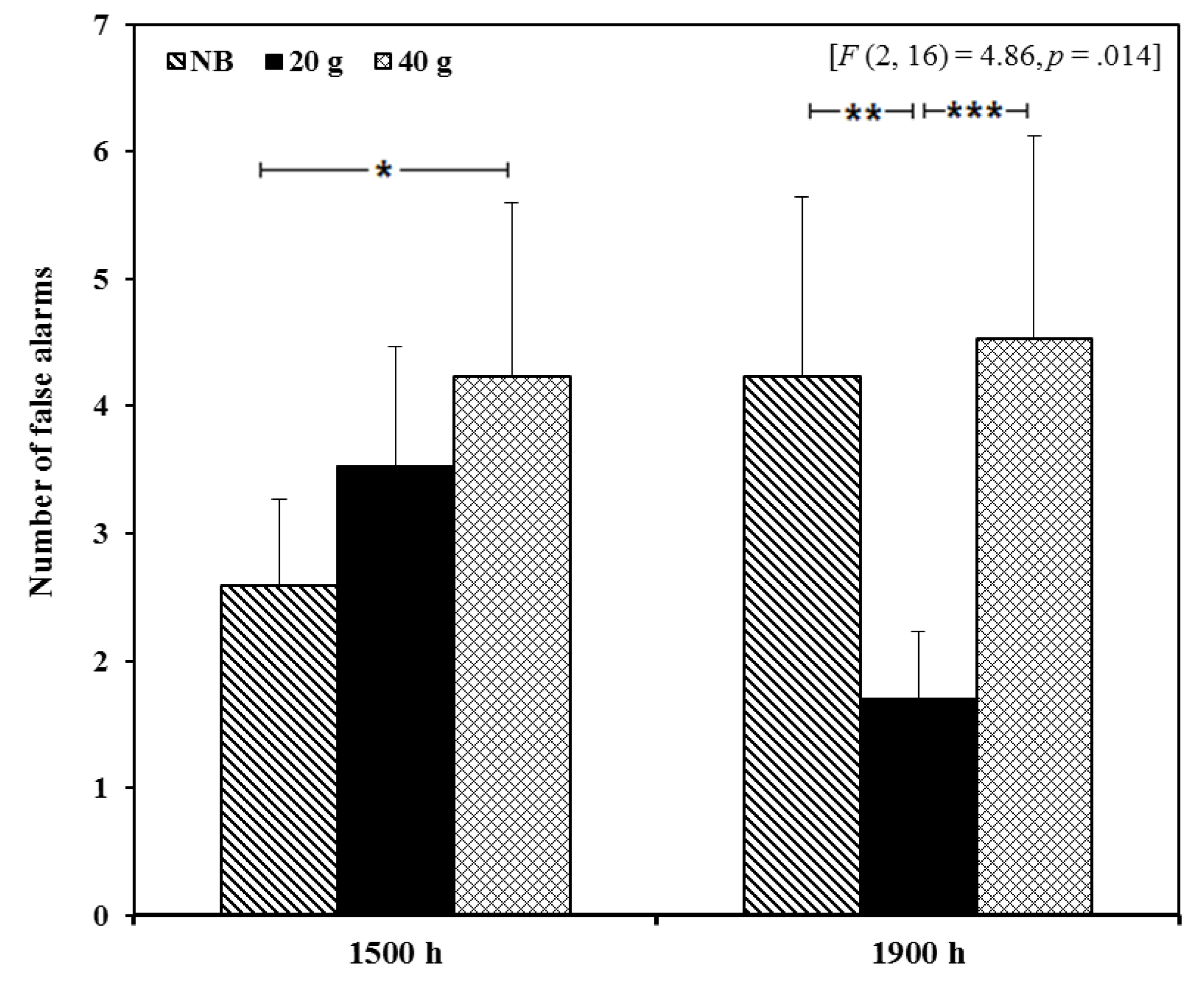
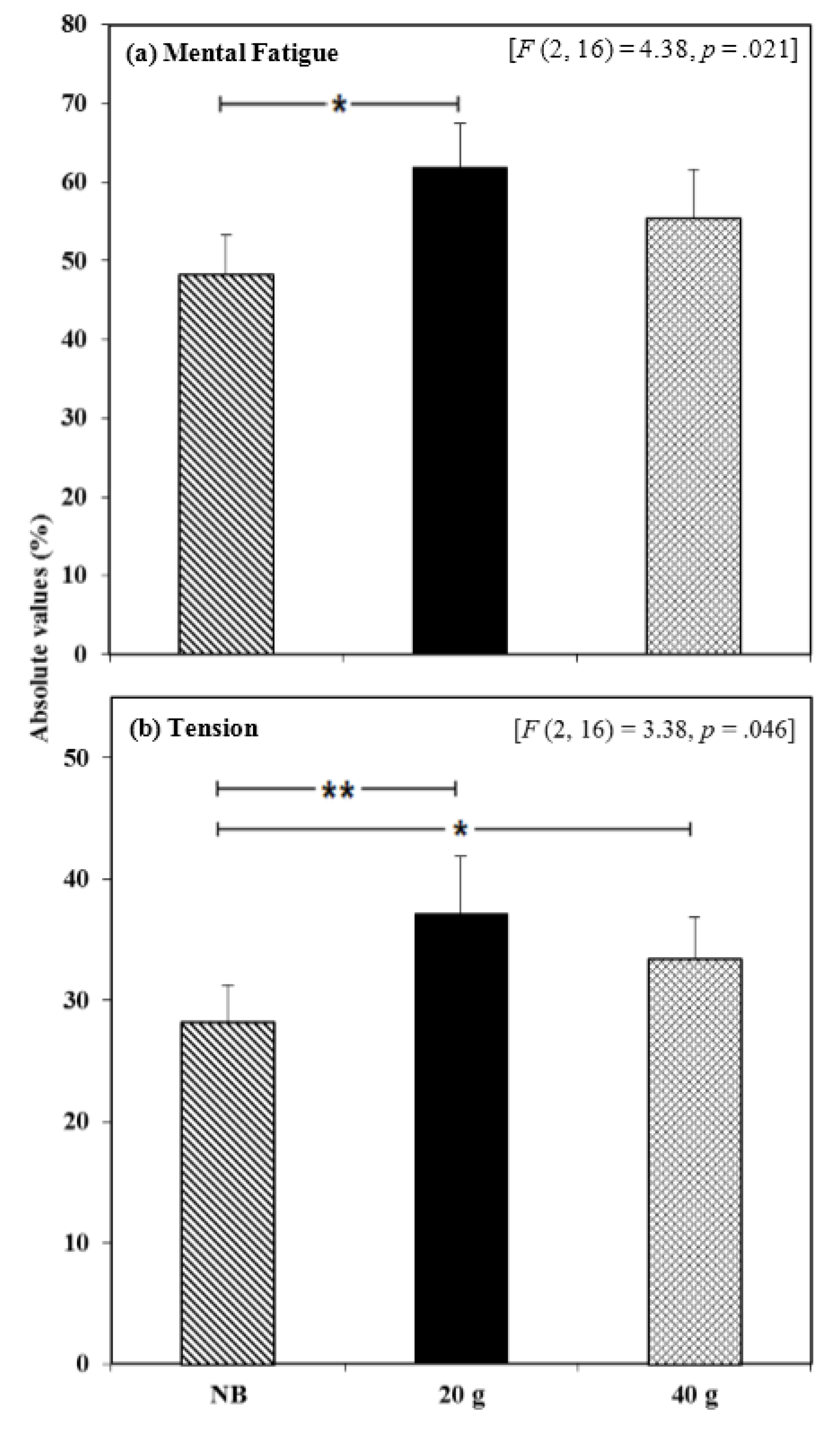
3.2.2. Mood and Physical State
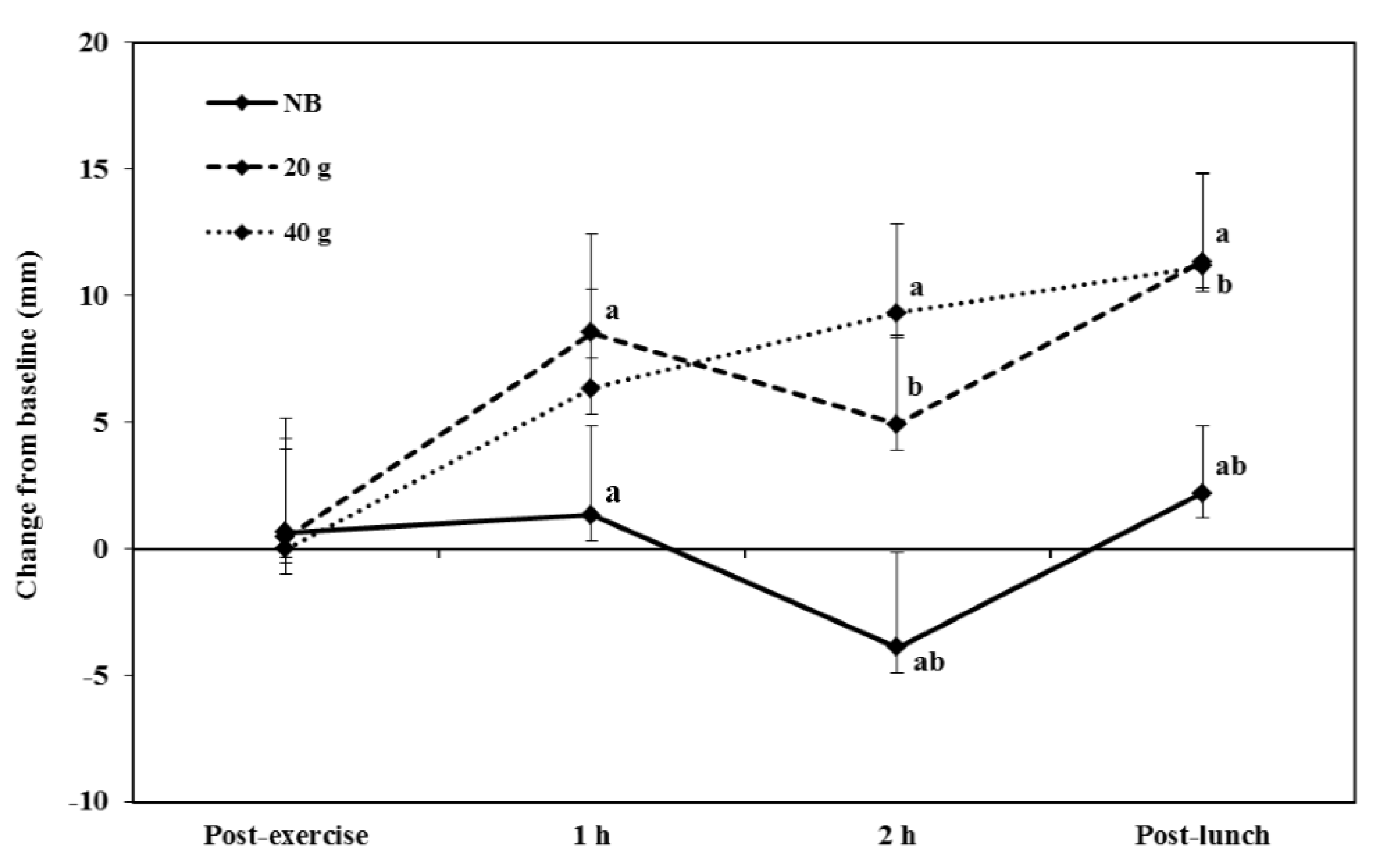
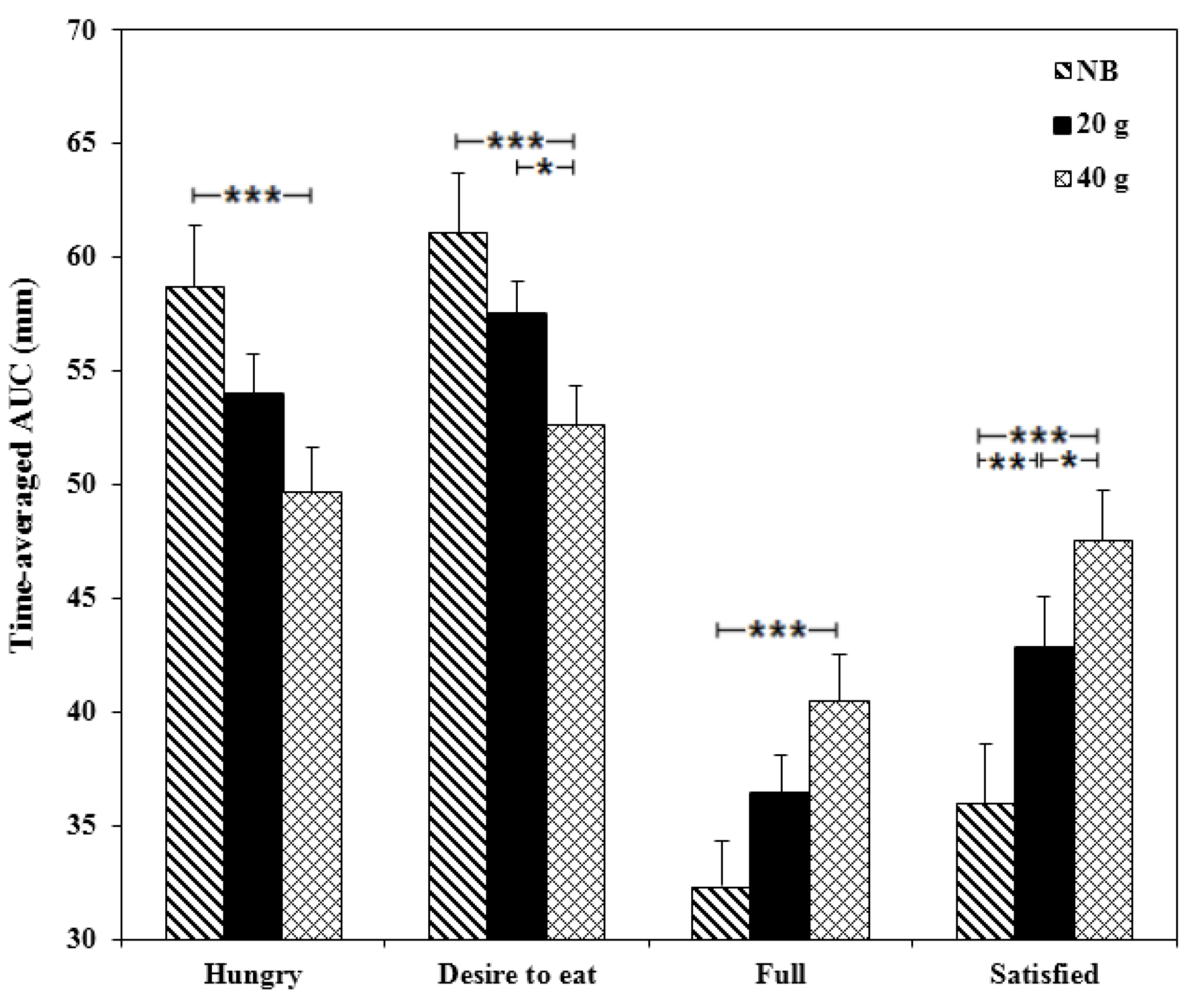
3.2.3. Appetite
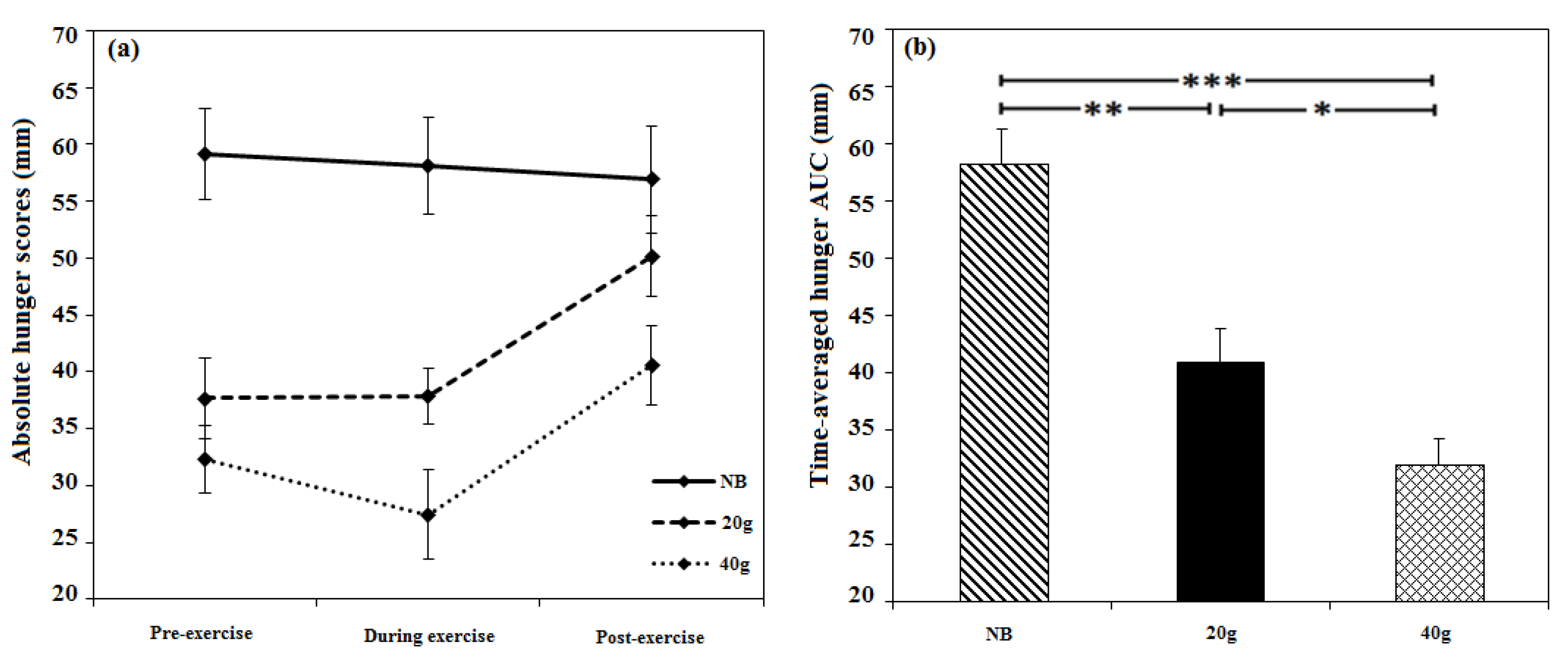
3.2.4. Energy Intake
3.2.5. Meal Liking
| Measure | Condition | Mean Value | ||
|---|---|---|---|---|
| Breakfast Liking (mm) | NB | 19.4 *, † | ± | 2.7 |
| 20 g | 68.9 | ± | 3.0 | |
| 40 g | 67.9 | ± | 3.5 | |
| Lunch Liking (mm) | NB | 74.5 | ± | 3.0 |
| 20 g | 76.5 | ± | 2.2 | |
| 40 g | 75.8 | ± | 2.4 | |
| Lunch Energy Intake (kcal) | NB | 763.2 | ± | 37.2 |
| 20 g | 786.2 | ± | 37.1 | |
| 40 g | 778.7 | ± | 38.8 | |
| Post-lunch Energy Intake (kcal; n = 22) | NB | 1000.8 | ± | 76.9 |
| 20 g | 1055.9 | ± | 105.6 | |
| 40 g | 1067.5 | ± | 114.5 | |
| Net Energy Intake (kcal; n = 22) | NB | 1780.14 | ± | 80.59 |
| 20 g | 1957.21 | ± | 114.64 | |
| 40 g | 2088.57 | ± | 132.30 | |
4. Discussion
4.1. Pilot Study
4.2. Main Study
5. Conclusions
Supplementary Files
Supplementary File 1Acknowledgments
Author Contributions
Conflicts of Interest
References
- Miles, L. Physical activity and health. Nutr. Bull. 2007, 32, 314–363. [Google Scholar] [CrossRef]
- Powell, K.E.; Paluch, A.E.; Blair, S.N. Physical activity for health: What kind? How much? How intense? On top of what? Annu. Rev. Public Health 2011, 32, 349–365. [Google Scholar] [CrossRef] [PubMed]
- Lambourne, K.; Tomporowski, P. The effect of exercise-induced arousal on cognitive task performance: A meta-regression analysis. Brain Res. 2010, 1314, 12–24. [Google Scholar] [CrossRef] [PubMed]
- Strohle, A. Physical activity, exercise, depression and anxiety disorders. J. Neural Transm. 2009, 116, 777–784. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.S. Physical activity and mental health. Hanyang Med. Rev. 2014, 34, 60–65. [Google Scholar] [CrossRef]
- Smith, A.P. Breakfast and mental health. Int. J. Food Sci. Nutr. 1998, 49, 397–402. [Google Scholar] [CrossRef] [PubMed]
- Barton, B.A.; Eldridge, A.L.; Thompson, D.; Affenito, S.G.; Striegel-Moore, R.H.; Franko, D.L.; Albertson, A.M.; Crockett, S.J. The Relationship of Breakfast and Cereal Consumption to Nutrient Intake and Body Mass Index: The National Heart, Lung, and Blood Institute Growth and Health Study. J. Am. Diet. Assoc. 2005, 105, 1383–1389. [Google Scholar] [CrossRef] [PubMed]
- Pereira, M.; Erickson, E.; McKee, P.; Schrankler, K.; Raatz, S.; Lytle, L.; Pellegrini, A.D. Breakfast Frequency and Quality May Affect Glycemia and Appetite in Adults and Children. J. Nutr. 2011, 141, 163–168. [Google Scholar] [CrossRef] [PubMed]
- Silliman, K.; Rodas-Fortier, K.; Neyman, M. A Survey of Dietary and Exercise Habits and Perceived Barriers to Following a Healthy Lifestyle in a College Population. Calif. J. Health Promot. 2004, 2, 10–19. [Google Scholar]
- Ozdogan, Y.; Ozcelik, A.O.; Surucuoglu, M.S. The Breakfast Habits of Female University Students. Pak. J. Nutr. 2010, 9, 882–886. [Google Scholar] [CrossRef]
- Hill, K.M.; Whitehead, J.R.; Goodwin, J.K. Pre-workout carbohydrate supplementation does not affect measures of self-assesed vitality and affect in college swimmers. J. Sports Sci. Med. 2011, 10, 478–482. [Google Scholar]
- Dundes, L. Fleshing out the role of skipping breakfast in weight control. Suppl. AgroFood Ind. Hi-Tech 2008, 19, 26–27. [Google Scholar]
- Gonzalez, J.T.; Veasey, R.C.; Rumbold, P.L.S.; Stevenson, E.J. Breakfast and exercise contingently affect postprandial metabolism and energy balance in physically active males. Br. J. Nutr. 2013, 110, 721–732. [Google Scholar] [CrossRef] [PubMed]
- Backhouse, S.H.; Williams, C.; Stevenson, E.; Nute, M. Effects of the glycemic index of breakfast on metabolic responses to brisk walking in females. Eur. J. Clin. Nutr. 2007, 61, 590–596. [Google Scholar] [CrossRef] [PubMed]
- Veasey, R.C.; Gonzalez, J.T.; Kennedy, D.O.; Haskell, C.F.; Stevenson, E.J. Breakfast consumption and exercise interact to affect cognitive performance and mood later in the day. A randomized controlled trial. Appetite 2013, 68, 38–44. [Google Scholar] [CrossRef] [PubMed]
- Paul, G.L.; Rokusek, J.T.; Dykstra, G.L.; Boileau, R.A.; Layman, D.K. Preexercise meal composition alters plasma large neutral amino acid responses during exercise and recovery. Am. J. Clin. Nutr. 1996, 64, 778–786. [Google Scholar] [PubMed]
- Hargreaves, M.; Hawley, J.A.; Jeukendrup, A. Pre-exercise carbohydrate and fat ingestion: Effects on metabolism and performance. J. Sports Sci. 2004, 22, 31–38. [Google Scholar] [CrossRef] [PubMed]
- Reeves, S.; Halsey, L.G.; McMeel, Y.; Huber, J.W. Breakfast habits, beliefs and measures of health and wellbeing in a nationally representative UK sample. Appetite 2013, 60, 51–57. [Google Scholar] [CrossRef] [PubMed]
- Spring, B.; Maller, O.; Wurtman, J.; Digman, L.; Cozolino, L. Effects of protein and carbohydrate meals on mood and performance: Interactions with sex and age. J. Psychiatr. Res. 1982, 17, 155–167. [Google Scholar] [CrossRef]
- Hagobian, T.A.; Sharoff, C.G.; Stephens, B.R.; Wade, G.N.; Silva, J.E.; Chipkin, S.R.; Braun, B. Effects of exercise on energy-regulating hormones and appetite in men and women. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2009, 296, R233–R242. [Google Scholar] [CrossRef] [PubMed]
- Stein, A.D.; Rivera, J.M.; Pivarnik, J.M. Measuring Energy Expenditure in Habitually Active and Sedentary Pregnant Women. Med. Sci. Sports Exerc. 2003, 35, 1441–1446. [Google Scholar] [CrossRef] [PubMed]
- Rogers, P.J.; Martin, J.; Smith, C.; Heatherley, S.V.; Smit, H.J. Absence of reinforcing, mood and psychomotor performance effects of caffeine in habitual non-consumers of caffeine. Psychopharmacology 2003, 167, 54–62. [Google Scholar] [PubMed]
- Stunkard, A.J.; Messick, S. The three factor eating questionnaire to measure dietary restraint, disinhibition and hunger. J. Psychosom. Res. 1985, 29, 71–81. [Google Scholar] [CrossRef]
- Kennedy, D.; Veasey, R.; Watson, A.; Dodd, F.; Jones, E.; Maggini, S.; Haskell, C.F. Effects of high-dose B vitamin complex with vitamin C and minerals on subjective mood and performance in healthy males. Psychopharmacology 2010, 211, 55–68. [Google Scholar] [CrossRef] [PubMed]
- Kennedy, D.O.; Dodd, F.L.; Robertson, B.C.; Okello, E.J.; Reay, J.L.; Scholey, A.B.; Haskell, C.F. Monoterpenoid extract of sage (Salvia lavandulaefolia) with cholinesterase inhibiting properties improves cognitive performance and mood in healthy adults. J. Psychopharmacol. (Oxf.) 2011, 25, 1088–1100. [Google Scholar] [CrossRef] [PubMed]
- Wightman, E.L.; Haskell, C.F.; Forster, J.S.; Veasey, R.C.; Kennedy, D.O. Epigallocatechin gallate, cerebral blood flow parameters, cognitive performance and mood in healthy humans: A double-blind, placebo-controlled, crossover investigation. Hum. Psychopharmacol. Clin. Exp. 2012, 27, 177–186. [Google Scholar] [CrossRef] [PubMed]
- Hoyland, A.; Lawton, C.L.; Dye, L. Acute effects of macronutrient manipulations on cognitive test performance in healthy young adults: A systematic research review. Neurosci. Biobehav. Rev. 2008, 32, 72–85. [Google Scholar] [CrossRef] [PubMed]
- Kennedy, D.O.; Veasey, R.C.; Watson, A.W.; Dodd, F.L.; Jones, E.K.; Tiplady, B.; Haskell, C.F. Vitamins and psychological functioning: A mobile phone assessment of the effects of a B vitamin complex, vitamin C and minerals on cognitive performance and subjective mood and energy. Hum. Psychopharmacol. Clin. Exp. 2011, 26, 338–347. [Google Scholar] [CrossRef] [PubMed]
- Stensel, D. Exercise, Appetite and Appetite-Regulating Hormones: Implications for Food Intake and Weight Control. Ann. Nutr. Metab. 2010, 57, 36–42. [Google Scholar] [CrossRef] [PubMed]
- Williams, C.; Nute, M.G.; Broadbank, L.; Vinall, S. Influence of fluid intake on endurance running performance. A comparison between water, glucose and fructose solutions. Eur. J. Appl. Physiol. 1990, 60, 112–119. [Google Scholar]
- American College of Sports Medicine. ACSM’s Resource Manual for Guidelines for Exercise Testing and Prescription; Swain, D.P., Brawner, C.A., Eds.; Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2012. [Google Scholar]
- Hung, T.M.; Tsai, C.L.; Chen, F.T.; Wang, C.C.; Chang, Y.K. The Immediate and Sustained Effects of Acute Exercise on Planning Aspect of Executive Function. Psychol. Sport Exerc. 2013, 14, 728–736. [Google Scholar] [CrossRef]
- Borg, G.A. Perceived exertion: a note on “history” and methods. Med. Sci. Sports 1973, 5, 90–93. [Google Scholar] [CrossRef] [PubMed]
- Lamport, D.J.; Hoyle, E.; Lawton, C.L.; Mansfield, M.W.; Dye, L. Evidence for a second meal cognitive effect: Glycaemic responses to high and low glycaemic index evening meals are associated with cognition the following morning. Nutr. Neurosci. 2011, 14, 66–71. [Google Scholar] [CrossRef] [PubMed]
- Cohen, J. Statistical Power Analysis for the Behavioral Sciences, 2nd ed; Lawrence Erlbaum Associates: New Jersey, NJ, USA, 1988. [Google Scholar]
- Faul, F.; Erdfelder, E.; Lang, A.G.; Buchner, A. G * Power 3: A flexible statistical power analysis program for the social, behavioral, and biomedical sciences. Behav. Res. Methods 2007, 39, 175–191. [Google Scholar] [CrossRef] [PubMed]
- Drapeau, V.; Blundell, J.; Therrien, F.; Lawton, C.; Richard, D.; Tremblay, A. Appetite sensations as a marker of overall intake. Br. J. Nutr. 2005, 93, 273–280. [Google Scholar] [CrossRef] [PubMed]
- Livingstone, M.B.E.; Black, A.E. Markers of the Validity of Reported Energy Intake. J. Nutr. 2003, 133, 895S–920S. [Google Scholar] [PubMed]
- Le Noury, J.; Lawton, C.; Blundell, J. Food choice and hedonic responses: Difference between overweight and lean high fat phenotypes. Int. J. Obes. 2002, 26, S125. [Google Scholar]
- Lloyd, H.M.; Rogers, P.J.; Hedderley, D.I.; Walker, A.F. Acute Effects on Mood and Cognitive Performance of Breakfasts Differing in Fat and Carbohydrate Content. Appetite 1996, 27, 151–164. [Google Scholar] [CrossRef] [PubMed]
- Ide, K.; Horn, A.; Secher, N.H. Cerebral metabolic response to submaximal exercise. J. Appl. Physiol. 1999, 87, 1604–1608. [Google Scholar] [PubMed]
- Backhouse, S.H.; Bishop, N.C.; Biddle, S.J.H.; Williams, C. Effect of carbohydrate and prolonged exercise on affect and perceived exertion. Med. Sci. Sports Exerc. 2005, 37, 1768–1773. [Google Scholar] [CrossRef] [PubMed]
- Foster, J.; Smith, M.; Woodman, M.; Zombor, R.; Ashton, J. Impact of a wholegrain breakfast cereal meal on blood glucose level, mood and affect. Food Aust. 2007, 59, 593–596. [Google Scholar]
- Nabb, S.; Benton, D. The influence on cognition of the interaction between the macro-nutrient content of breakfast and glucose tolerance. Physiol. Behav. 2006, 87, 16–23. [Google Scholar] [CrossRef] [PubMed]
- Zilberter, T.; Zilberter, E.Y. Breakfast and cognition: Sixteen effects in nine populations, no single recipe. Front. Hum. Neurosci. 2013, 7. [Google Scholar] [CrossRef] [PubMed]
- Astbury, N.M.; Taylor, M.A.; Macdonald, I.A. Breakfast consumption affects appetite, energy intake, and the metabolic and endocrine responses to foods consumed later in the day in male habitual breakfast eaters. J. Nutr. 2011, 141, 1381–1389. [Google Scholar] [CrossRef] [PubMed]
- Kral, T.V.E.; Heo, M.; Whiteford, L.M.; Faith, M.S. Effects on Cognitive Performance of Eating Compared With Omitting Breakfast in Elementary Schoolchildren. J. Dev. Behav. Pediatr. 2012, 33, 9–16. [Google Scholar] [CrossRef] [PubMed]
- Farshchi, H.R.; Taylor, M.A.; Macdonald, I.A. Deleterious effects of omitting breakfast on insulin sensitivity and fasting lipid profiles in healthy lean women. Am. J. Clin. Nutr. 2005, 81, 388–396. [Google Scholar] [PubMed]
- King, J.A.; Miyashita, M.; Wasse, L.K.; Stensel, D.J. Influence of prolonged treadmill running on appetite, energy intake and circulating concentrations of acylated ghrelin. Appetite 2010, 54, 492–498. [Google Scholar] [CrossRef] [PubMed]
- Martins, C.; Morgan, L.M.; Bloom, S.R.; Robertson, M.D. Effects of exercise on gut peptides, energy intake and appetite. J. Endocrinol. 2007, 193, 251–258. [Google Scholar] [CrossRef] [PubMed]
- Conner, M.; Johnson, C.; Grogan, S. Gender, sexuality, body image and eating behaviours. J. Health Psychol. 2004, 9, 505–515. [Google Scholar] [CrossRef] [PubMed]
- Bellisle, F.; Dalix, A.; Airinei, G.; Hercberg, S.; Péneau, S. Influence of dietary restraint and environmental factors on meal size in normal-weight women. A laboratory study. Appetite 2009, 53, 309–313. [Google Scholar] [CrossRef] [PubMed]
- Klesges, R.C.; Klem, M.L; Epkins, C.C.; Klesges, L.M. A longitudinal evaluation of dietary restraint and its relationship to changes in body weight. Addict. Behav. 1991, 16, 363–368. [Google Scholar] [CrossRef]
- Stubbs, R.J. Appetite, feeding behaviour and energy balance in human subjects. In Proceedings-Nutrition Society of London; Cambridge University Press: Cambridge, UK, 1998. [Google Scholar]
- Schoeller, D.A. Limitations in the assessment of dietary energy intake by self-report. Metabolism 1995, 44, 18–22. [Google Scholar] [CrossRef]
- Palmblad, M.; Tiplady, B. Electronic diaries and questionnaires: Designing user interfaces that are easy for all patients to use. Qual. Life Res. 2004, 13, 1199–1207. [Google Scholar] [CrossRef] [PubMed]
- Timlin, M.T.; Pereira, M.A. Breakfast frequency and quality in the etiology of adult obesity and chronic diseases. Nutr. Rev. 2007, 65, 268–281. [Google Scholar] [CrossRef] [PubMed]
- Cooper, S.B.; Bandelow, S.; Nute, M.L.; Morris, J.G.; Nevill, M.E. Breakfast glycaemic index and exercise: Combined effects on adolescents’ cognition. Physiol. Behav. 2015, 139, 104–111. [Google Scholar] [CrossRef] [PubMed]
- Duckworth, L.C.; Backhouse, S.H.; Stevenson, E.J. The effect of galactose ingestion on affect and perceived exertion in recreationally active females. Appetite 2013, 71, 252–258. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Veasey, R.C.; Haskell-Ramsay, C.F.; Kennedy, D.O.; Tiplady, B.; Stevenson, E.J. The Effect of Breakfast Prior to Morning Exercise on Cognitive Performance, Mood and Appetite Later in the Day in Habitually Active Women. Nutrients 2015, 7, 5712-5732. https://doi.org/10.3390/nu7075250
Veasey RC, Haskell-Ramsay CF, Kennedy DO, Tiplady B, Stevenson EJ. The Effect of Breakfast Prior to Morning Exercise on Cognitive Performance, Mood and Appetite Later in the Day in Habitually Active Women. Nutrients. 2015; 7(7):5712-5732. https://doi.org/10.3390/nu7075250
Chicago/Turabian StyleVeasey, Rachel C., Crystal F. Haskell-Ramsay, David O. Kennedy, Brian Tiplady, and Emma J. Stevenson. 2015. "The Effect of Breakfast Prior to Morning Exercise on Cognitive Performance, Mood and Appetite Later in the Day in Habitually Active Women" Nutrients 7, no. 7: 5712-5732. https://doi.org/10.3390/nu7075250
APA StyleVeasey, R. C., Haskell-Ramsay, C. F., Kennedy, D. O., Tiplady, B., & Stevenson, E. J. (2015). The Effect of Breakfast Prior to Morning Exercise on Cognitive Performance, Mood and Appetite Later in the Day in Habitually Active Women. Nutrients, 7(7), 5712-5732. https://doi.org/10.3390/nu7075250





