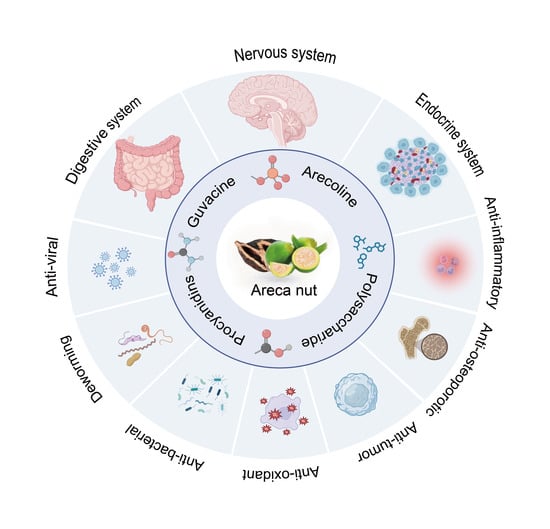
Bioactive Components of Areca Nut: An Overview of Their Positive Impacts Targeting Different Organs
Abstract
1. Introduction
2. Main Chemical Components of Areca Nut
2.1. Alkaloids
2.2. Polyphenols
2.2.1. Flavonoids
2.2.2. Tannins
2.3. Polysaccharides, Lipids, and Other Components
3. Functional Effects of Areca Nut Components
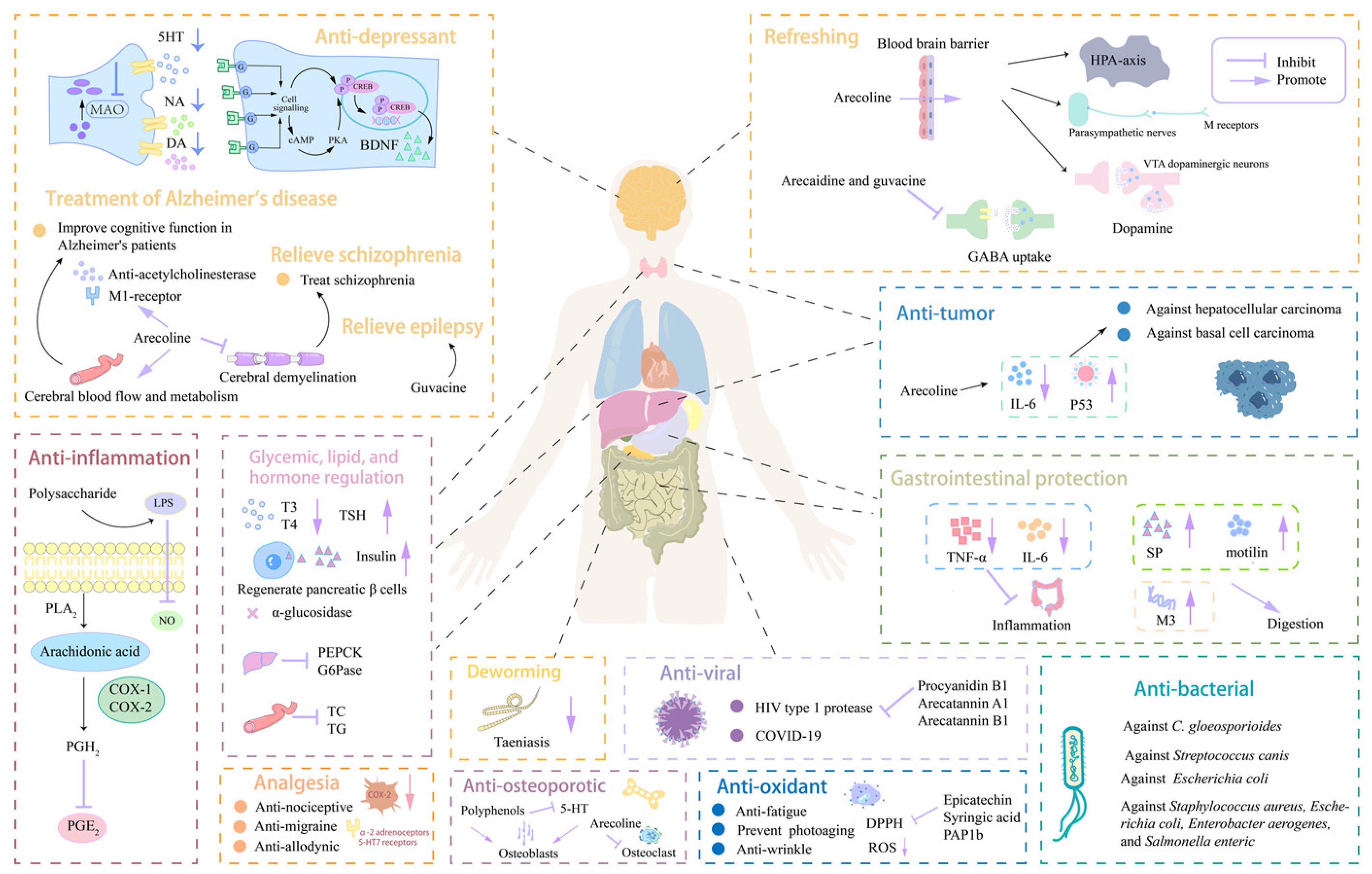
3.1. Effects on the Nervous System
3.1.1. Refreshing Functions
3.1.2. Anti-Depression Effects
3.1.3. Analgesic Efficacy
3.1.4. Treatment of Alzheimer’s Disease
3.1.5. Relieve Schizophrenia
3.1.6. Relieve Epilepsy
3.2. Effects on the Digestive System
3.3. Effects on the Endocrine System
3.3.1. Hypoglycemic Effects
3.3.2. Hypolipidemic Effects
3.3.3. Effects on Hormone Levels
3.4. Anti-Inflammatory Effects
3.5. Anti-Osteoporotic Effects
3.6. Anti-Tumor Efficacy
3.7. Anti-Oxidant Effects
3.8. Anti-Bacterial Efficacy
3.9. Deworming Efficacy
3.10. Anti-Viral Efficacy
4. The Potential Health Risks of Areca Nut
5. Conclusions and Future Prospects
Author Contributions
Funding
Conflicts of Interest
References
- Christenhusz, M.J.M.; Byng, J.W. The Number of Known Plants Species in the World and Its Annual Increase. Phytotaxa 2016, 261, 201–217. [Google Scholar] [CrossRef]
- Teerawichitchainan, B.; Knodel, J. Tradition and Change in Marriage Payments in Vietnam, 1963–2000. Asian Popul. Stud. 2012, 8, 151–172. [Google Scholar] [CrossRef] [PubMed]
- Zhang, P.; Chua, N.Q.E.; Dang, S.; Davis, A.; Chong, K.W.; Prime, S.S.; Cirillo, N. Molecular Mechanisms of Malignant Transformation of Oral Submucous Fibrosis by Different Betel Quid Constituents-Does Fibroblast Senescence Play a Role? Int. J. Mol. Sci. 2022, 23, 1637. [Google Scholar] [CrossRef] [PubMed]
- Wang, R.; Pan, F.; He, R.; Kuang, F.; Wang, L.; Lin, X. Arecanut (Areca catechu L.) Seed Extracts Extracted by Conventional and Eco-Friendly Solvents: Relation between Phytochemical Compositions and Biological Activities by Multivariate Analysis. J. Appl. Res. Med. Aromat. Plants 2021, 25, 100336. [Google Scholar] [CrossRef]
- Deshmukh, P.S.; Patil, P.G.; Shahare, P.U.; Bhanage, G.B.; Dhekale, J.S.; Dhande, K.G.; Aware, V.V. Effect of Mechanical and Chemical Treatments of Arecanut (Areca catechu L.) Fruit Husk on Husk and Its Fibre. Waste Manag. 2019, 95, 458–465. [Google Scholar] [CrossRef] [PubMed]
- Joo, Y.J.; Newcombe, D.; Nosa, V.; Bullen, C. Investigating Betel Nut Use, Antecedents and Consequences: A Review of Literature. Subst. Use Misuse 2020, 55, 1422–1442. [Google Scholar] [CrossRef]
- Mehrtash, H.; Duncan, K.; Parascandola, M.; David, A.; Gritz, E.R.; Gupta, P.C.; Mehrotra, R.; Nordin, A.S.A.; Pearlman, P.C.; Warnakulasuriya, S.; et al. Defining a Global Research and Policy Agenda for Betel Quid and Areca Nut. Lancet Oncol. 2017, 18, E767–E775. [Google Scholar] [CrossRef] [PubMed]
- Yang, B.; Chen, H.; Chen, W.; Chen, W.; Zhong, Q.; Zhang, M.; Pei, J. Edible Quality Analysis of Different Areca Nuts: Compositions, Texture Characteristics and Flavor Release Behaviors. Foods 2023, 12, 1749. [Google Scholar] [CrossRef]
- Zhang, X.; Li, C.; Liao, Q.; Reichart, P.A. Areca Chewing in Xiangtan, Hunan Province, China: Interviews with Chewers. J. Oral. Pathol. Med. 2008, 37, 423–429. [Google Scholar] [CrossRef]
- Xiong, X.; Hou, A.; Yi, S.; Guo, Y.; Zhao, Z.; Wu, Z.; Cheng, H.; Li, K.; Li, Z.; Ren, Y.; et al. Analysis of Oral Microorganism Diversity in Healthy Individuals before and after Chewing Areca Nuts Using PCR-Denatured Gradient Gel Electrophoresis. Anim. Nutr. 2018, 4, 294–299. [Google Scholar] [CrossRef]
- Ji, X.; Guo, J.; Pan, F.; Kuang, F.; Chen, H.; Guo, X.; Liu, Y. Structural Elucidation and Antioxidant Activities of a Neutral Polysaccharide From Arecanut (Areca catechu L.). Front. Nutr. 2022, 9, 853115. [Google Scholar] [CrossRef]
- Sun, M.; Yan, D.; Yang, X.; Xue, X.; Zhou, S.; Liang, S.; Wang, S.; Meng, J. Quality Assessment of Crude and Processed Arecae Semen Based on Colorimeter and HPLC Combined with Chemometrics Methods. J. Sep. Sci. 2017, 40, 2151–2160. [Google Scholar] [CrossRef]
- Kong, D.; Li, X.; Zhao, X.; Guo, M.; Pang, Z.; Zeng, L.; Yang, M. Domestic and International Research Progress of Edible and Medicinal Arecae Semen. China J. Chin. Mater. Medica 2021, 46, 1053–1059. [Google Scholar] [CrossRef]
- Xu, S.; Liu, Y.; Feng, J.; Zhan, Z.; Li, W. Herbal Textual Research on Arecae Semen in Famous Classical Formulas. Chin. J. Exp. Tradit. Med. Formulae 2022, 28, 167–175. [Google Scholar] [CrossRef]
- Celimuge, X. Research Progress in Chemical Composition, Pharmacology and Toxicity of Areca Semen and Prediction and Analysis of Its Quality Markers. Nat. Product. Res. Dev. 2023, 16, 1431. [Google Scholar]
- Zhao, Z.; Li, Y.; Zhou, L.; Zhou, X.; Xie, B.; Zhang, W.; Sun, J. Prevention and Treatment of COVID-19 Using Traditional Chinese Medicine: A Review. Phytomedicine 2021, 85, 153308. [Google Scholar] [CrossRef] [PubMed]
- Wang, G. Study on Key Issues Affecting the Internationalization of Quality Standards of Chinese Herbal Medicine Arecae Semen Decoction Pieces; Qinghai Normal University: Xining, China, 2022. [Google Scholar]
- Jam, N.; Hajimohammadi, R.; Gharbani, P.; Mehrizad, A. Evaluation of Antibacterial Activity of Aqueous, Ethanolic and Methanolic Extracts of Areca Nut Fruit on Selected Bacteria. Biomed. Res. Int. 2021, 2021, 6663399. [Google Scholar] [CrossRef] [PubMed]
- Khan, S.; Mehmood, M.H.; Ali, A.N.A.; Ahmed, F.S.; Dar, A.; Gilani, A.-H. Studies on Anti-Inflammatory and Analgesic Activities of Betel Nut in Rodents. J. Ethnopharmacol. 2011, 135, 654–661. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Huang, H.; Cui, Z.; Chu, J.; Du, G. UPLC-MS/MS and Network Pharmacology-Based Analysis of Bioactive Anti-Depression Compounds in Betel Nut. Drug Des. Dev. Ther. 2021, 15, 4827–4836. [Google Scholar] [CrossRef] [PubMed]
- Jing, Y.; Ma, Y.; Pan, F.; Kuang, F.; Zhang, D.; Guo, X.; Lanfang, W. Research Progress on Herbal Textual Research, Chemical Constituents and Pharmacological Effects of Areca catechu. Asia-Pac. Tradit. Med. 2022, 18, 232–239. [Google Scholar]
- Volgin, A.D.; Bashirzade, A.; Arnstislavskaya, T.G.; Yakovlev, O.A.; Demin, K.A.; Ho, Y.-J.; Wang, D.; Shevyrin, V.A.; Yan, D.; Tang, Z.; et al. DARK Classics in Chemical Neuroscience: Arecoline. ACS Chem. Neurosci. 2019, 10, 2176–2185. [Google Scholar] [CrossRef]
- Yuan, J.; Zhang, H.; Zhao, H.; Ren, H.; Zhai, H. Study on Dissociation and Chemical Structural Characteristics of Areca Nut Husk. Molecules 2023, 28, 1513. [Google Scholar] [CrossRef]
- Debnath, B.; Singh, W.S.; Das, M.; Goswami, S.; Singh, M.K.; Maiti, D.; Manna, K. Role of Plant Alkaloids on Human Health: A Review of Biological Activities. Mater. Today Chem. 2018, 9, 56–72. [Google Scholar] [CrossRef]
- Casciaro, B.; Calcaterra, A.; Cappiello, F.; Mori, M.; Loffredo, M.R.; Ghirga, F.; Mangoni, M.L.; Botta, B.; Quaglio, D. Nigritanine as a New Potential Antimicrobial Alkaloid for the Treatment of Staphylococcus Aureus-Induced Infections. Toxins 2019, 11, 511. [Google Scholar] [CrossRef]
- Liu, B.; Li, J.; Yi, R.; Mu, J.; Zhou, X.; Zhao, X. Preventive Effect of Alkaloids from Lotus Plumule on Acute Liver Injury in Mice. Foods 2019, 8, 36. [Google Scholar] [CrossRef] [PubMed]
- Peng, W.; Liu, Y.-J.; Wu, N.; Sun, T.; He, X.-Y.; Gao, Y.-X.; Wu, C.-J. Areca catechu L. (Arecaceae): A Review of Its Traditional Uses, Botany, Phytochemistry, Pharmacology and Toxicology. J. Ethnopharmacol. 2015, 164, 340–356. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.-J.; Peng, W.; Hu, M.-B.; Xu, M.; Wu, C.-J. The Pharmacology, Toxicology and Potential Applications of Arecoline: A Review. Pharm. Biol. 2016, 54, 2753–2760. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Zhang, H.; Wang, S.; Yuan, L.; Gruenhofer, P.; Schreiber, L.; Wan, Y. Tissue-Specific and Maturity-Dependent Distribution of Pyridine Alkaloids in Areca Triandra. J. Plant Res. 2019, 132, 531–540. [Google Scholar] [CrossRef]
- Zeng, Q.; Li, Z.; Yuan, L.; Zheng, J.; Zhong, H. Review on the Actuality and Prospect of Areca Alkaloids. Food Mach. 2006, 22, 158–161. [Google Scholar] [CrossRef]
- Papke, R.L.; Horenstein, N.A.; Stokes, C. Nicotinic Activity of Arecoline, the Psychoactive Element of “Betel Nuts”, Suggests a Basis for Habitual Use and AntiInflammatory Activity. PLoS ONE 2015, 10, e0140907. [Google Scholar] [CrossRef]
- Franke, A.A.; Mendez, A.J.; Lai, J.F.; Arat-Cabading, C.; Li, X.; Custer, L.J. Composition of Betel Specific Chemicals in Saliva during Betel Chewing for the Identification of Biomarkers. Food Chem. Toxicol. 2015, 80, 241–246. [Google Scholar] [CrossRef]
- Jain, V.; Garg, A.; Parascandola, M.; Chaturvedi, P.; Khariwala, S.S.; Stepanov, I. Analysis of Alkaloids in Areca Nut-Containing Products by Liquid Chromatography-Tandem Mass-Spectrometry. J. Agric. Food Chem. 2017, 65, 1977–1983. [Google Scholar] [CrossRef]
- Tang, S.-N.; Zhang, J.; Liu, D.; Liu, Z.-W.; Zhang, X.-Q.; Ye, W.-C. Three New Areca Alkaloids from the Nuts of Areca catechu. J. Asian Nat. Prod. Res. 2017, 19, 1155–1159. [Google Scholar] [CrossRef] [PubMed]
- Cao, M.; Yuan, H.; Daniyal, M.; Yu, H.; Xie, Q.; Liu, Y.; Li, B.; Jian, Y.; Peng, C.; Tan, D.; et al. Two New Alkaloids Isolated from Traditional Chinese Medicine Binglang the Fruit of Areca catechu. Fitoterapia 2019, 138, 104276. [Google Scholar] [CrossRef]
- Zhang, W.-M.; Huang, W.-Y.; Chen, W.-X.; Han, L.; Zhang, H.-D. Optimization of Extraction Conditions of Areca Seed Polyphenols and Evaluation of Their Antioxidant Activities. Molecules 2014, 19, 16416–16427. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Lu, J.; Li, J.; Li, P.; Zhao, M.; Xia, G. Optimization of Ultrasonic-Assisted Extraction of Polyphenol from Areca Nut (Areca catechu L.) Seeds Using Response Surface Methodology and Its Effects on Osteogenic Activity. Ultrason. Sonochem 2023, 98, 106511. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Lv, F.; Tai, J.; Wang, Z.; Fu, Q. Quantitative determination of total phenolics and tannin in areca nut and its products. Food Res. Dev. 2008, 29, 119–121. [Google Scholar]
- Sari, L.M. Antioxidant Activity of Areca Nut to Human Health: Effect on Oral Cancer Cell Lines and Immunomodulatory Activity. In Bioactive Compounds in Nutraceutical and Functional Food for Good Human Health; IntechOpen: London, UK, 2021; ISBN 978-1-83880-888-4. [Google Scholar]
- Li, A.; Chen, L.; Zhou, W.; Pan, J.; Gong, D.; Zhang, G. Effects of Baicalein and Chrysin on the Structure and Functional Properties of β-Lactoglobulin. Foods 2022, 11, 165. [Google Scholar] [CrossRef] [PubMed]
- Si, Y.; Liu, J.; Shen, H.; Zhang, C.; Wu, Y.; Huang, Y.; Gong, Z.; Xue, J.; Liu, T. Fisetin Decreases TET1 Activity and CCNY/CDK16 Promoter 5hmC Levels to Inhibit the Proliferation and Invasion of Renal Cancer Stem Cell. J. Cell Mol. Med. 2019, 23, 1095–1105. [Google Scholar] [CrossRef]
- Wu, Q.; Zhao, X.; Chen, C.; Zhang, Z.; Yu, F. Metabolite Profiling and Classification of Developing Styrax Tonkinensis Kernels. Metabolites 2020, 10, 21. [Google Scholar] [CrossRef]
- Xiao, P.; Liu, S.; Kuang, Y.; Jiang, Z.; Lin, Y.; Xie, Z.; Liu, E.-H. Network Pharmacology Analysis and Experimental Validation to Explore the Mechanism of Sea Buckthorn Flavonoids on Hyperlipidemia. J. Ethnopharmacol. 2021, 264, 113380. [Google Scholar] [CrossRef] [PubMed]
- Boateng, I.D. Polyprenols in Ginkgo Biloba; a Review of Their Chemistry (Synthesis of Polyprenols and Their Derivatives), Extraction, Purification, and Bioactivities. Food Chem. 2023, 418, 136006. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Li, X.; Kano, Y.; Yuan, D.; Qu, J. Oriental Traditional Herbal Medicine—Puerariae Flos: A Systematic Review. J. Ethnopharmacol. 2023, 306, 116089. [Google Scholar] [CrossRef] [PubMed]
- Badshah, S.L.; Faisal, S.; Muhammad, A.; Poulson, B.G.; Emwas, A.H.; Jaremko, M. Antiviral Activities of Flavonoids. Biomed. Pharmacother. 2021, 140, 111596. [Google Scholar] [CrossRef]
- Cui, L.; Liu, X.; Tian, Y.; Xie, C.; Li, Q.; Cui, H.; Sun, C. Flavonoids, Flavonoid Subclasses, and Esophageal Cancer Risk: A Meta-Analysis of Epidemiologic Studies. Nutrients 2016, 8, 350. [Google Scholar] [CrossRef]
- Hodgson, J.M.; Croft, K.D. Tea Flavonoids and Cardiovascular Health. Mol. Asp. Med. 2010, 31, 495–502. [Google Scholar] [CrossRef]
- Maleki, S.J.; Crespo, J.F.; Cabanillas, B. Anti-Inflammatory Effects of Flavonoids. Food Chem. 2019, 299, 125124. [Google Scholar] [CrossRef]
- Park, E.-J.; Pezzuto, J.M. Flavonoids in Cancer Prevention. Anti-Cancer Agents Med. Chem. 2012, 12, 836–851. [Google Scholar] [CrossRef]
- Yang, W.-Q.; Wang, H.-C.; Wang, W.-J.; Wang, Y.; Zhang, X.-Q.; Ye, W.-C. Chemical constituents from the fruits of Areca catechu. Zhong Yao Cai 2012, 35, 400–403. [Google Scholar]
- Ma, J.; Wang, R.; Du, X.; Zhao, A.; Wang, Z.; Zhang, H. Determination of Total Polyphenols and Catechins in Betel Nut Polyphenolextract. J. Pharm. Pract. 2022, 40, 243–247. [Google Scholar] [CrossRef]
- Yuan, M.; Ao, Y.; Yao, N.; Xie, J.; Zhang, D.; Zhang, J.; Zhang, X.; Ye, W. Two New Flavonoids from the Nuts of Areca catechu. Molecules 2019, 24, 2862. [Google Scholar] [CrossRef]
- Chen, X.; He, Y.; Deng, Y. Chemical Composition, Pharmacological, and Toxicological Effects of Betel Nut. Evid. Based Complement. Altern. Med. 2021, 2021, 1–7. [Google Scholar] [CrossRef]
- Sun, J.-T.; Liu, J.-H.; Jiang, X.-Q.; Luo, X.; Yuan, J.-D.; Zhang, Q.; Qi, X.-Y.; Lee, S.; Liu, Z.-H.; Jin, J.-X. Tannin Reduces the Incidence of Polyspermic Penetration in Porcine Oocytes. Antioxidants 2022, 11, 2027. [Google Scholar] [CrossRef]
- Das, A.K.; Islam, M.N.; Faruk, M.O.; Ashaduzzaman, M.; Dungani, R. Review on Tannins: Extraction Processes, Applications and Possibilities. S. Afr. J. Bot. 2020, 135, 58–70. [Google Scholar] [CrossRef]
- Ojo, M.A. Tannins in Foods: Nutritional Implications and Processing Effects of Hydrothermal Techniques on Underutilized Hard-to-Cook Legume Seeds-A Review. Prev. Nutr. Food Sci. 2022, 27, 14–19. [Google Scholar] [CrossRef] [PubMed]
- Soares, S.; Brandão, E.; Guerreiro, C.; Soares, S.; Mateus, N.; de Freitas, V. Tannins in Food: Insights into the Molecular Perception of Astringency and Bitter Taste. Molecules 2020, 25, 2590. [Google Scholar] [CrossRef]
- Bhandare, A.; Kshirsagar, A.; Vyawahare, N.; Sharma, P.; Mohite, R. Evaluation of Anti-Migraine Potential of Areca catechu to Prevent Nitroglycerin-Induced Delayed Inflammation in Rat Meninges: Possible Involvement of NOS Inhibition. J. Ethnopharmacol. 2011, 136, 267–270. [Google Scholar] [CrossRef]
- Wadhwa, M.; Sidhu, P.K.; Bakshi, M.P.S. Herbal Feed Additives Containing Tannins: Impact on in Vitro Fermentation and Methane Mitigation from Total Mixed Ration. Turk. J. Vet. Anim. Sci. 2020, 44, 47–58. [Google Scholar] [CrossRef]
- Amidžić Klarić, D.; Klarić, I.; Mornar, A.; Velić, N.; Velić, D. Assessment of Bioactive Phenolic Compounds and Antioxidant Activity of Blackberry Wines. Foods 2020, 9, 1623. [Google Scholar] [CrossRef] [PubMed]
- Chavan, Y.V.; Singhal, R.S. Separation of Polyphenols and Arecoline from Areca Nut (Areca catechu L.) by Solvent Extraction, Its Antioxidant Activity, and Identification of Polyphenols: Separation of Polyphenols and Arecoline from Areca Nut. J. Sci. Food Agric. 2013, 93, 2580–2589. [Google Scholar] [CrossRef]
- Ansari, A.; Mahmood, T.; Bagga, P.; Ahsan, F.; Shamim, A.; Ahmad, S.; Shariq, M.; Parveen, S. Areca catechu: A Phytopharmacological Legwork. Food Front. 2021, 2, 163–183. [Google Scholar] [CrossRef]
- Chen, J.; Sun, A.; Gao, X.; Pan, N. Process Optimization for Ultrasonic-Assisted Solvent Extraction of Proanthocyanidins from Areca catechu L. Fruit by Response Surface Analysis. Food Sci. 2011, 32, 82–86. [Google Scholar]
- Tong, Z.; He, W.; Fan, X.; Guo, A. Biological Function of Plant Tannin and Its Application in Animal Health. Front. Vet. Sci. 2022, 8, 803657. [Google Scholar] [CrossRef] [PubMed]
- Yin, M.; Pan, F.; Guo, J.; Ji, X.; Liu, Y. Research into Chemical Constituents and Pharmacological Activities in Areca catechu L. Food Res. Dev. 2021, 42, 219–224. [Google Scholar]
- He, X.; Li, Y.; Zhang, C.; Hu, X. Arborinol Methyl Ether from Areca catechu L. Acta Crystallogr. Sect. E.-Struct. Rep. Online 2010, 66, O2273-U38. [Google Scholar] [CrossRef]
- Zote, L.; Lalrammawia, K.; Buragohain, A.; Lalrinhlupuii; Kakki, B.; Lalmuanpuii, R.; Pachuau, Z.; Vanlalhruaia, J.; Muthukumaran, R.B.; Kumar, N.S.; et al. Macro-, Micro-, and Trace Element Distributions in Areca Nut, Husk, and Soil of Northeast India. Environ. Monit. Assess. 2021, 193, 65. [Google Scholar] [CrossRef] [PubMed]
- Abbas, G.; Naqvi, S.; Erum, S.; Ahmed, S.; Atta-ur-Rahman; Dar, A. Potential Antidepressant Activity of Areca catechu Nut via Elevation of Serotonin and Noradrenaline in the Hippocampus of Rats. Phytother. Res. 2013, 27, 39–45. [Google Scholar] [CrossRef] [PubMed]
- Dar, A.; Khatoon, S.; Rahman, G. Atta-Ur-Rahman Anti-Depressant Activities of Areca catechu Fruit Extract. Phytomedicine 1997, 4, 41–45. [Google Scholar] [CrossRef]
- Yuan, L.; Li, Z.; Zheng, J. Effect of Areca Nut Extracts on the Hyperlipaemia-Moderating Function in Rats. Food Sci. Technol. 2009, 34, 188–192. [Google Scholar]
- Lee, H.G.; Song, J.A.; Han, D.S.; Woo, K.W.; Yoon, M.H. Antiallodynic Effects of Intrathecal Areca Nut for Spinal Nerve-Ligated and Chemotherapy-Induced Neuropathic Pain in Rats. Pharmacology 2018, 102, 332–338. [Google Scholar] [CrossRef]
- Park, Y.B.; Jeon, S.-M.; Byun, S.-J.; Kim, H.-S.; Choi, M.-S. Absorption of Intestinal Free Cholesterol Is Lowered by Supplementation of Areca catechu L. Extract in Rats. Life Sci. 2002, 70, 1849–1859. [Google Scholar] [CrossRef] [PubMed]
- Dhanraj, M.; Veerakumari, L. Effect of ethanol extract of Areca catechu on fumarate reductase and succinate dehydrogenase of Cotylophoron cotylophorum. Int. J. Res. Dev. Pharm. Life Sci. 2016, 5, 2117–2123. [Google Scholar]
- Wang, D.; Zhou, L.; Li, W.; Zhou, H.; Hou, G. Anticoccidial Effects of Areca Nut (Areca catechu L.) Extract on Broiler Chicks Experimentally Infected with Eimeria Tenella. Exp. Parasitol. 2018, 184, 16–21. [Google Scholar] [CrossRef] [PubMed]
- Wei, P.-L.; Hung, C.-S.; Lu, H.-H.; Batzorig, U.; Huang, C.-Y.; Chang, Y.-J. Areca Nut Extract (ANE) Inhibits the Progression of Hepatocellular Carcinoma Cells via Activation of ROS Production and Activation of Autophagy. Int. J. Med. Sci. 2021, 18, 3452–3462. [Google Scholar] [CrossRef]
- Li, M.; Lin, M.; Jin, S.; Li, W.; Zheng, T.; Qu, S. Effects of Pericarpium Arecae on Contraction of Lsolated Circular Muscle Strips of Gastric Body in Guinea Pigs. Acad. J. Shanghai Univ. Tradit. Chin. Med. 2008, 2, 46–47. [Google Scholar] [CrossRef]
- Zhang, S.; Yang, P.; Li, X.; Wang, X.; Song, J.; Peng, W.; Wu, C. Comparative Researches of Semen Arecae and Charred Semen Arecae on Gastrointestinal Motility, Motilin, Substance P, and CCK in Chronically Stressed Rats. Evid.-Based Complement. Altern. Med. 2017, 2017, 1273561. [Google Scholar] [CrossRef] [PubMed]
- Badar, S.N.; Iqbal, Z.; Sajid, M.S.; Rizwan, H.M.; Shareef, M.; Malik, M.A.; Khan, M.N. Comparative Anthelmintic Efficacy of Arundo Donax, Areca catechu, and Ferula Assa-Foetida against Haemonchus Contortus. Rev. Bras. Parasitol. Vet. 2021, 30, e001221. [Google Scholar] [CrossRef]
- Zhao, L.; Li, Y.; Yang, S.; Zhang, P.; Wang, J. Anti-Nociceptive Effect of Total Alkaloids Isolated from the Seeds of Areca catechu L (Arecaceae) in Mice. Trop. J. Pharm. Res. 2017, 16, 363–369. [Google Scholar] [CrossRef][Green Version]
- Serikuly, N.; Alpyshov, E.T.; Wang, D.; Wang, J.; Yang, L.; Hu, G.; Yan, D.; Demin, K.A.; Kolesnikova, T.O.; Galstyan, D.; et al. Effects of Acute and Chronic Arecoline in Adult Zebrafish: Anxiolytic-like Activity, Elevated Brain Monoamines and the Potential Role of Microglia. Progress. Neuro-Psychopharmacol. Biol. Psychiatry 2021, 104, 109977. [Google Scholar] [CrossRef]
- Hernandez, B.Y.; Zhu, X.; Sotto, P.; Paulino, Y. Oral Exposure to Environmental Cyanobacteria Toxins: Implications for Cancer Risk. Environ. Int. 2021, 148, 106381. [Google Scholar] [CrossRef]
- Siregar, P.; Audira, G.; Feng, L.-Y.; Lee, J.-H.; Santoso, F.; Yu, W.-H.; Lai, Y.-H.; Li, J.-H.; Lin, Y.-T.; Chen, J.-R.; et al. Pharmaceutical Assessment Suggests Locomotion Hyperactivity in Zebrafish Triggered by Arecoline Might Be Associated with Multiple Muscarinic Acetylcholine Receptors Activation. Toxins 2021, 13, 259. [Google Scholar] [CrossRef]
- Calogero, A.E.; Kamilaris, T.C.; Gomez, M.T.; Johnson, E.O.; Tartaglia, M.E.; Gold, P.W.; Chrousos, G.P. The Muscarinic Cholinergic Agonist Arecoline Stimulates the Rat Hypothalamic-Pituitary-Adrenal Axis through a Centrally-Mediated Corticotropin-Releasing Hormone-Dependent Mechanism. Endocrinology 1989, 125, 2445–2453. [Google Scholar] [CrossRef]
- Lan, Q.; Guan, P.; Huang, C.; Huang, S.; Zhou, P.; Zhang, C. Arecoline Induces an Excitatory Response in Ventral Tegmental Area Dopaminergic Neurons in Anesthetized Rats. Front. Pharmacol. 2022, 13, 872212. [Google Scholar] [CrossRef]
- Machova, M.; Bajer, T.; Silha, D.; Ventura, K.; Bajerova, P. Volatiles Composition and Antimicrobial Activities of Areca Nut Extracts Obtained by Simultaneous Distillation-Extraction and Headspace Solid-Phase Microextraction. Molecules 2021, 26, 7422. [Google Scholar] [CrossRef] [PubMed]
- Xu, Z.; Adilijiang, A.; Wang, W.; You, P.; Lin, D.; Li, X.; He, J. Arecoline Attenuates Memory Impairment and Demyelination in a Cuprizone-Induced Mouse Model of Schizophrenia. Neuroreport 2019, 30, 134–138. [Google Scholar] [CrossRef]
- Maiese, K.; Holloway, H.; Larson, D.; Soncrant, T. Effect of Acute and Chronic Arecoline Treatment on Cerebral Metabolism and Blood-Flow in the Conscious Rat. Brain Res. 1994, 641, 65–75. [Google Scholar] [CrossRef]
- Dasgupta, R.; Chatterji, U.; Nag, T.C.; Chaudhuri-Sengupta, S.; Nag, D.; Maiti, B.R. Ultrastructural and Hormonal Modulations of the Thyroid Gland Following Arecoline Treatment in Albino Mice. Mol. Cell. Endocrinol. 2010, 319, 1–7. [Google Scholar] [CrossRef]
- Huang, L.-W.; Hsieh, B.-S.; Cheng, H.-L.; Hu, Y.-C.; Chang, W.-T.; Chang, K.-L. Arecoline Decreases Interleukin-6 Production and Induces Apoptosis and Cell Cycle Arrest in Human Basal Cell Carcinoma Cells. Toxicol. Appl. Pharmacol. 2012, 258, 199–207. [Google Scholar] [CrossRef] [PubMed]
- Shih, L.-J.; Wang, J.-Y.; Jheng, J.-Y.; Siao, A.-C.; Lin, Y.-Y.; Tsuei, Y.-W.; Kuo, Y.-C.; Chuu, C.-P.; Kao, Y.-H. Betel Nut Arecoline Induces Different Phases of Growth Arrest between Normal and Cancerous Prostate Cells through the Reactive Oxygen Species Pathway. Int. J. Mol. Sci. 2020, 21, 9219. [Google Scholar] [CrossRef] [PubMed]
- Yao, N.; Feng, L.; Jiang, W.; Wu, P.; Ren, H.; Shi, H.; Tang, L.; Li, S.; Wu, C.; Li, H.; et al. An Emerging Role of Arecoline on Growth Performance, Intestinal Digestion and Absorption Capacities and Intestinal Structural Integrity of Adult Grass Carp (Ctenopharyngodon idella). Anim. Nutr. 2023, 15, 173–186. [Google Scholar] [CrossRef]
- Liu, F.-L.; Chen, C.-L.; Lai, C.-C.; Lee, C.-C.; Chang, D.-M. Arecoline Suppresses RANKL-Induced Osteoclast Differentiation in Vitro and Attenuates LPS-Induced Bone Loss in Vivo. Phytomedicine 2020, 69, 153195. [Google Scholar] [CrossRef] [PubMed]
- Zhang, P.; Sari, E.F.; McCullough, M.J.; Cirillo, N. Metabolomic Profile of Indonesian Betel Quids. Biomolecules 2022, 12, 1469. [Google Scholar] [CrossRef] [PubMed]
- Song, F.; Zhang, Y.; Guo, Y.; Li, R.; Chen, H.; Tang, M. Inhibitory Effects of Areca Nut Extract on the Activity of α-Glucosidase. Food Res. Dev. 2019, 40, 79–83. [Google Scholar]
- Meng, K.; Mei, F.; Zhu, L.; Xiang, Q.; Quan, Z.; Pan, F.; Xia, G.; Shen, X.; Yun, Y.; Zhang, C.; et al. Arecanut (Areca catechu L.) Seed Polyphenol Improves Osteoporosis via Gut-Serotonin Mediated Wnt/Beta-Catenin Pathway in Ovariectomized Rats. J. Funct. Food. 2021, 84, 104598. [Google Scholar] [CrossRef]
- Tang, M.; Chen, H.; Wang, H.; Zhao, S.; Qi, J. Anti-Fatigue Effects of Polyphenols Extracted from Areca catechu L. Husk and Determination of the Main Components by High Performance Capillary Electrophoresis. Bangladesh J. Bot. 2016, 45, 783–790. [Google Scholar]
- Yi, S.; Zou, L.; Li, Z.; Sakao, K.; Wang, Y.; Hou, D.-X. In Vitro Antioxidant Activity of Areca Nut Polyphenol Extracts on RAW264.7 Cells. Foods 2022, 11, 3607. [Google Scholar] [CrossRef]
- Kusumoto, I.; Nakabayashi, T.; Kida, H.; Miyashiro, H.; Hattori, M.; Namba, T.; Shimotohno, K. Screening of Various Plant-Extracts Used in Ayurvedic Medicine for Inhibitory Effects on Human-Immunodeficiency-Virus Type-1 (Hiv-1) Protease. Phytother. Res. 1995, 9, 180–184. [Google Scholar] [CrossRef]
- Wang, C.-C.; Lin, Y.-R.; Liao, M.-H.; Jan, T.-R. Oral Supplementation with Areca-Derived Polyphenols Attenuates Food Allergic Responses in Ovalbumin-Sensitized Mice. BMC Complement. Altern. Med. 2013, 13, 154. [Google Scholar] [CrossRef]
- Huang, P.-L.; Chi, C.-W.; Liu, T.-Y. Areca Nut Procyanidins Ameliorate Streptozocin-Induced Hyperglycemia by Regulating Gluconeogenesis. Food Chem. Toxicol. 2013, 55, 137–143. [Google Scholar] [CrossRef]
- Weng, C.-L.; Chen, C.-C.; Tsou, H.-H.; Liu, T.-Y.; Wang, H.-T. Areca Nut Procyanidins Prevent Ultraviolet Light B-Induced Photoaging via Suppression of Cyclooxygenase-2 and Matrix Metalloproteinases in Mouse Skin. Drug Chem. Toxicol. 2022, 45, 353–359. [Google Scholar] [CrossRef]
- Yu, X.; Tang, M.; Luo, J.; Song, F.; Zhu, T.; Chen, H.; Zhao, S. Inhibitory Effects of Areca Nut Extract on the Activity of Angiotensin Converting Enzyme. Chin. J. Trop. Crops 2023, 14, 78–83. [Google Scholar]
- Li, Y. The Study on the Chemical Constituents of Antidepressant Activity of Areca catechu L.; Guangzhou University of Chinese Medicine: Guangdong, China, 2011. [Google Scholar]
- Tang, M.; Chen, H.; Li, R. Optimization of Ultrasound-Assisted Extraction of Areca Nut Polysaccharides Based on Response Surface Methodology and Anti-Inflammatory Activity. Anhui Agric. Sci. Bull. 2019, 25, 21–24+74. [Google Scholar] [CrossRef]
- Mateen, F.J.; Carone, M.; Tshering, L.; Dorji, C.; Dema, U.; Grundy, S.J.; Pokhrel, D.R.; Nirola, D.K. Areca catechu (Palm Tree) Nut Chewing and Seizures: An Observational Study. Epilepsy Behav. 2017, 74, 76–80. [Google Scholar] [CrossRef] [PubMed]
- Osborne, P.G.; Pasupuleti, R.R.; Wu, M.-T.; Lee, C.-H.; Ponnusamy, V.K. LC-MS/MS Measurement of Alkaloids in Alkaline Extracts of Areca Nut Preparations and Their Physiological Effects. Process Biochem. 2022, 118, 65–73. [Google Scholar] [CrossRef]
- Yang, T.-Y.; Lin, H.-R. Taking Actions to Quit Chewing Betel Nuts and Starting a New Life: Taxi Drivers’ Successful Experiences of Quitting Betel Nut Chewing. J. Clin. Nurs. 2017, 26, 1031–1041. [Google Scholar] [CrossRef] [PubMed]
- Alavian, K.N.; Scholz, C.; Simon, H.H. Transcriptional Regulation of Mesencephalic Dopaminergic Neurons: The Full Circle of Life and Death. Mov. Disord. 2008, 23, 319–328. [Google Scholar] [CrossRef]
- Huang, Y.; Liu, Z.; Cao, B.-B.; Qiu, Y.-H.; Peng, Y.-P. Treg Cells Protect Dopaminergic Neurons against MPP+ Neurotoxicity via CD47-SIRPA Interaction. Cell Physiol. Biochem. 2017, 41, 1240–1254. [Google Scholar] [CrossRef]
- Zhao, H.; Ma, H.; Ha, X.; Zheng, P.; Li, X.; Zhang, M.; Dong, J.; Yang, Y. Salidroside Induces Rat Mesenchymal Stem Cells to Differentiate into Dopaminergic Neurons. Cell Biol. Int. 2014, 38, 462–471. [Google Scholar] [CrossRef]
- Chu, N.S. Effects of Betel Chewing on the Central and Autonomic Nervous Systems. J. Biomed. Sci. 2001, 8, 229–236. [Google Scholar] [CrossRef]
- An, J.; Shi, Y.; Fang, J.; Hu, Y.; Liu, Y. Multichannel Ratiometric Fluorescence Sensor Arrays for Rapid Visual Monitoring of Epinephrine, Norepinephrine, and Levodopa. Chem. Eng. J. 2021, 425, 130595. [Google Scholar] [CrossRef]
- Bai, J.; Ao, M.; Xing, A.; Yu, L.; Tong, H.; Bao, W.; Wang, Y. Areca Thirteen Pill Improves Depression in Rat by Modulation of the Chemokine/Chemokine Receptor Axis. Mol. Neurobiol. 2023, 1–15. [Google Scholar] [CrossRef]
- Cai, M.; Yang, Z.; Huang, X.; Li, J.; Bao, W.; Cui, J.W.; Ma, L.Q.; Tong, H.Y. Mongolian Medicine Areca Thirteen Pill (GY-13) Improved Depressive Syndrome via Upregulating cAMP/PKA/CREB/BDNF Signaling Pathway. J. Ethnopharmacol. 2022, 293, 115310. [Google Scholar] [CrossRef]
- Yao, C.; Zhang, Y.; Sun, X.; Pei, H.; Wei, S.; Wang, M.; Chang, Q.; Liu, X.; Jiang, N. Areca Catechu L. Ameliorates Chronic Unpredictable Mild Stress-Induced Depression Behavior in Rats by the Promotion of the BDNF Signaling Pathway. Biomed Pharmacother 2023, 164, 114459. [Google Scholar] [CrossRef]
- Dobrek, L.; Głowacka, K. Depression and Its Phytopharmacotherapy—A Narrative Review. Int. J. Mol. Sci. 2023, 24, 4772. [Google Scholar] [CrossRef]
- Lamanna, C.; Sinicropi, M.S.; Pietrangeli, P.; Corbo, F.; Franchini, C.; Mondovi, B.; Perrone, M.G.; Scilimati, A. Synthesis and Biological Evaluation of 3-Alkyloxazolidin-2-Ones as Reversible MAO Inhibitors. Arkivoc 2004, 5, 118–130. [Google Scholar] [CrossRef]
- Martins, J.; Brijesh, S. Phytochemistry and Pharmacology of Anti-Depressant Medicinal Plants: A Review. Biomed. Pharmacother. 2018, 104, 343–365. [Google Scholar] [CrossRef] [PubMed]
- Franklin, M.; Cowen, P.J. Researching the Antidepressant Actions of Hypericum Perforatum (St. John’s Wort) in Animals and Man. Pharmacopsychiatry 2001, 34 (Suppl. 1), S29–S37. [Google Scholar] [CrossRef] [PubMed]
- Li, X.-X.; Yan, Y.; Zhang, J.; Ding, K.; Xia, C.-Y.; Pan, X.-G.; Shi, Y.-J.; Xu, J.-K.; He, J.; Zhang, W.-K. Hyperforin: A Natural Lead Compound with Multiple Pharmacological Activities. Phytochemistry 2023, 206, 113526. [Google Scholar] [CrossRef] [PubMed]
- Jost, W.H. A Critical Appraisal of MAO-B Inhibitors in the Treatment of Parkinson’s Disease. J. Neural Transm. 2022, 129, 723–736. [Google Scholar] [CrossRef] [PubMed]
- Nam, M.-H.; Park, M.; Park, H.; Kim, Y.; Yoon, S.; Sawant, V.S.; Cho, J.W.; Park, J.-H.; Park, K.D.; Min, S.-J.; et al. Indole-Substituted Benzothiazoles and Benzoxazoles as Selective and Reversible MAO-B Inhibitors for Treatment of Parkinson’s Disease. ACS Chem. Neurosci. 2017, 8, 1519–1529. [Google Scholar] [CrossRef] [PubMed]
- Bolay, H.; Reuter, U.; Dunn, A.K.; Huang, Z.; Boas, D.A.; Moskowitz, M.A. Intrinsic Brain Activity Triggers Trigeminal Meningeal Afferents in a Migraine Model. Nat. Med. 2002, 8, 136–142. [Google Scholar] [CrossRef] [PubMed]
- Reuter, U.; Bolay, H.; Jansen-Olesen, I.; Chiarugi, A.; del Rio, M.S.; Letourneau, R.; Theoharides, T.C.; Waeber, C.; Moskowitz, M.A. Delayed Inflammation in Rat Meninges: Implications for Migraine Pathophysiology. Brain 2001, 124, 2490–2502. [Google Scholar] [CrossRef] [PubMed]
- Bannister, K.; Dickenson, A.H. What Do Monoamines Do in Pain Modulation? Curr. Opin. Support. Palliat. Care 2016, 10, 143–148. [Google Scholar] [CrossRef] [PubMed]
- Zhang, P.; Xu, S.; Zhu, Z.; Xu, J. Multi-Target Design Strategies for the Improved Treatment of Alzheimer’s Disease. Eur. J. Med. Chem. 2019, 176, 228–247. [Google Scholar] [CrossRef] [PubMed]
- Cornec, A.-S.; Monti, L.; Kovalevich, J.; Makani, V.; James, M.J.; Vijayendran, K.G.; Oukoloff, K.; Yao, Y.; Lee, V.M.-Y.; Trojanowski, J.Q.; et al. Multitargeted Imidazoles: Potential Therapeutic Leads for Alzheimer’s and Other Neurodegenerative Diseases. J. Med. Chem. 2017, 60, 5120–5145. [Google Scholar] [CrossRef] [PubMed]
- Felder, C.C.; Goldsmith, P.J.; Jackson, K.; Sanger, H.E.; Evans, D.A.; Mogg, A.J.; Broad, L.M. Current Status of Muscarinic M1 and M4 Receptors as Drug Targets for Neurodegenerative Diseases. Neuropharmacology 2018, 136, 449–458. [Google Scholar] [CrossRef] [PubMed]
- Brown, A.J.H.; Bradley, S.J.; Marshall, F.H.; Brown, G.A.; Bennett, K.A.; Brown, J.; Cansfield, J.E.; Cross, D.M.; de Graaf, C.; Hudson, B.D.; et al. From Structure to Clinic: Design of a Muscarinic M1 Receptor Agonist with the Potential to Treat Alzheimer’s Disease. Cell 2021, 184, 5886–5901.e22. [Google Scholar] [CrossRef]
- Gilani, A.H.; Ghayur, M.N.; Saify, Z.S.; Ahmed, S.P.; Choudhary, M.I.; Khalid, A. Presence of Cholinomimetic and Acetylcholinesterase Inhibitory Constituents in Betel Nut. Life Sci. 2004, 75, 2377–2389. [Google Scholar] [CrossRef]
- Pradeep, S.; Prabhuswaminath, S.C.; Reddy, P.; Srinivasa, S.M.; Shati, A.A.; Alfaifi, M.Y.; Eldin I Elbehairi, S.; Achar, R.R.; Silina, E.; Stupin, V.; et al. Anticholinesterase Activity of Areca catechu: In Vitro and in Silico Green Synthesis Approach in Search for Therapeutic Agents against Alzheimer’s Disease. Front. Pharmacol. 2022, 13, 1044248. [Google Scholar] [CrossRef]
- Vrabec, R.; Blunden, G.; Cahlíková, L. Natural Alkaloids as Multi-Target Compounds towards Factors Implicated in Alzheimer’s Disease. Int. J. Mol. Sci. 2023, 24, 4399. [Google Scholar] [CrossRef]
- Wong, K.H.; Riaz, M.K.; Xie, Y.; Zhang, X.; Liu, Q.; Chen, H.; Bian, Z.; Chen, X.; Lu, A.; Yang, Z. Review of Current Strategies for Delivering Alzheimer’s Disease Drugs across the Blood-Brain Barrier. Int. J. Mol. Sci. 2019, 20, 381. [Google Scholar] [CrossRef] [PubMed]
- Scheltens, P.; Strooper, B.D.; Kivipelto, M.; Holstege, H.; Chételat, G.; Teunissen, C.E.; Cummings, J.; van der Flier, W.M. Alzheimer’s Disease. Lancet 2021, 397, 1577–1590. [Google Scholar] [CrossRef] [PubMed]
- Sullivan, R.J.; Andres, S.; Otto, C.; Miles, W.; Kydd, R. The Effects of an Indigenous Muscarinic Drug, Betel Nut (Areca catechu), on the Symptoms of Schizophrenia: A Longitudinal Study in Palau, Micronesia. Am. J. Psychiatry 2007, 164, 670–673. [Google Scholar] [CrossRef]
- Xu, M.-Y.; Wong, A.H.C. GABAergic Inhibitory Neurons as Therapeutic Targets for Cognitive Impairment in Schizophrenia. Acta Pharmacol. Sin. 2018, 39, 733–753. [Google Scholar] [CrossRef]
- Zafar, S.; Jabeen, I. Structure, Function, and Modulation of γ-Aminobutyric Acid Transporter 1 (GAT1) in Neurological Disorders: A Pharmacoinformatic Prospective. Front. Chem. 2018, 6, 360060. [Google Scholar] [CrossRef] [PubMed]
- Pavia, M.R.; Lobbestael, S.J.; Nugiel, D.; Mayhugh, D.R.; Gregor, V.E.; Taylor, C.P.; Schwarz, R.D.; Brahce, L.; Vartanian, M.G. Structure-Activity Studies on Benzhydrol-Containing Nipecotic Acid and Guvacine Derivatives as Potent, Orally-Active Inhibitors of GABA Uptake. J. Med. Chem. 1992, 35, 4238–4248. [Google Scholar] [CrossRef]
- Luan, J.; Tao, X. Study on the Biological Activity of Areca catechu. Anhui Agric. Sci. Bull. 2018, 24, 8–10+12. [Google Scholar] [CrossRef]
- Wang, G.; Hu, B. Research Progress of Arecoline. J. Clin. Pathol. Res. 2010, 30, 171–175. [Google Scholar]
- Hou, Q.; Fu, L.; Liang, H.; Zhou, Y.; Yang, X.; Zhao, X. Determination of Four Alkaloids Components in Arecae Semen, Arecae Semen Tostum and Arecae Pericarpium by LC-PDA. Chin. J. Mod. Appl. Pharm. 2023, 40, 443–447. [Google Scholar] [CrossRef]
- Liu, Y.; Peng, W.; Hu, M.; Yan, D.; Xu, M.; He, X.; Wu, C. The Substance Basis Research of Stir-Baking to Dark Brown Could Enhance the Promoting Effects of Areca Nut on Gastrointestinal Motility. J. Food Process Preserv. 2017, 41, e13103. [Google Scholar] [CrossRef]
- Zhong, Q.; Chun, R.; Feng, Q.; Chang, W.; Ling, F. Effect of Piperine on Arecoline Induced Contraction of Isolated Small Intestinal Smooth Muscle from Rabbits. World Chin. J. Dig. 2019, 27, 20–28. [Google Scholar]
- Jing, X.; Zhang, Y.; Gu, Y.; Fei, J. Clinical Effect of Acupoint Application Therapy of Modified Wuzhuyu Binglang Tang on Chronic Non-Atrophic Gastritis. Chin. J. Exp. Tradit. Med. Formulae 2019, 25, 104–109. [Google Scholar] [CrossRef]
- Kamalumpundi, V.; Shams, E.; Tucker, C.; Cheng, L.; Peterson, J.; Thangavel, S.; Ofori, O.; Correia, M. Mechanisms and Pharmacotherapy of Hypertension Associated with Type 2 Diabetes. Biochem. Pharmacol. 2022, 206, 115304. [Google Scholar] [CrossRef]
- Chung, F.-M.; Shieh, T.-Y.; Yang, Y.-H.; Chang, D.-M.; Shin, S.-J.; Tsai, J.C.-R.; Chen, T.H.; Tai, T.-Y.; Lee, Y.-J. The Role of Angiotensin-Converting Enzyme Gene Insertion/Deletion Polymorphism for Blood Pressure Regulation in Areca Nut Chewers. Transl. Res. 2007, 150, 58–65. [Google Scholar] [CrossRef]
- Serina, J.J.C.; Castilho, P.C.M.F. Using Polyphenols as a Relevant Therapy to Diabetes and Its Complications, a Review. Crit. Rev. Food Sci. Nutr. 2022, 62, 8355–8387. [Google Scholar] [CrossRef]
- Alotaibi, B.S.; Ijaz, M.; Buabeid, M.; Kharaba, Z.J.; Yaseen, H.S.; Murtaza, G. Therapeutic Effects and Safe Uses of Plant-Derived Polyphenolic Compounds in Cardiovascular Diseases: A Review. Drug Des. Devel Ther. 2021, 15, 4713–4732. [Google Scholar] [CrossRef] [PubMed]
- Flory, J.; Lipska, K. Metformin in 2019. JAMA 2019, 321, 1926–1927. [Google Scholar] [CrossRef]
- Luo, J.; Yang, H.; Song, B.-L. Mechanisms and Regulation of Cholesterol Homeostasis. Nat. Rev. Mol. Cell Biol. 2020, 21, 225–245. [Google Scholar] [CrossRef]
- Wang, J.-Q.; Li, L.-L.; Hu, A.; Deng, G.; Wei, J.; Li, Y.-F.; Liu, Y.-B.; Lu, X.-Y.; Qiu, Z.-P.; Shi, X.-J.; et al. Inhibition of ASGR1 Decreases Lipid Levels by Promoting Cholesterol Excretion. Nature 2022, 608, 413–420. [Google Scholar] [CrossRef]
- Huang, H.; Liu, P.; Ma, D.; Zhang, H.; Xu, H.; Zhou, J.; Zhao, H.; Zhao, T.; Li, C. Triiodothyronine Attenuates Neurocognitive Dysfunction Induced by Sevoflurane in the Developing Brain of Neonatal Rats. J. Affect. Disord. 2022, 297, 455–462. [Google Scholar] [CrossRef]
- van der Spek, A.H.; Surovtseva, O.V.; Jim, K.K.; van Oudenaren, A.; Brouwer, M.C.; Vandenbroucke-Grauls, C.M.J.E.; Leenen, P.J.M.; van de Beek, D.; Hernandez, A.; Fliers, E.; et al. Regulation of Intracellular Triiodothyronine Is Essential for Optimal Macrophage Function. Endocrinology 2018, 159, 2241–2252. [Google Scholar] [CrossRef]
- Yang, M.; Wang, D.; Gan, S.; Wang, B.; Yu, L.; Xie, Y.; Fan, L.; Ma, J.; Chen, W. Triiodothyronine Ameliorates Silica-Induced Pulmonary Inflammation and Fibrosis in Mice. Sci. Total Environ. 2021, 790, 148041. [Google Scholar] [CrossRef] [PubMed]
- Pan, J.; Zhou, L.; Zhang, C.; Xu, Q.; Sun, Y. Targeting Protein Phosphatases for the Treatment of Inflammation-Related Diseases: From Signaling to Therapy. Signal Transduct. Target. Ther. 2022, 7, 177. [Google Scholar] [CrossRef]
- Dalbeth, N.; Choi, H.K.; Joosten, L.A.B.; Khanna, P.P.; Matsuo, H.; Perez-Ruiz, F.; Stamp, L.K. Gout. Nat. Rev. Dis. Primers 2019, 5, 69. [Google Scholar] [CrossRef] [PubMed]
- Iovoli, A.J.; Hermann, G.M.; Ma, S.J.; Platek, A.J.; Farrugia, M.K.; Yau, E.; Wooten, K.E.; Arshad, H.; Gupta, V.; Kuriakose, M.A.; et al. Association of Nonsteroidal Anti-Inflammatory Drug Use With Survival in Patients With Squamous Cell Carcinoma of the Head and Neck Treated With Chemoradiation Therapy. JAMA Netw. Open 2020, 3, e207199. [Google Scholar] [CrossRef]
- Parolini, M. Toxicity of the Non-Steroidal Anti-Inflammatory Drugs (NSAIDs) Acetylsalicylic Acid, Paracetamol, Diclofenac, Ibuprofen and Naproxen towards Freshwater Invertebrates: A Review. Sci. Total Environ. 2020, 740, 140043. [Google Scholar] [CrossRef]
- Zhang, J.; Fu, Y.; Yang, P.; Liu, X.; Li, Y.; Gu, Z. ROS Scavenging Biopolymers for Anti-Inflammatory Diseases: Classification and Formulation. Adv. Mater. Interfaces 2020, 7, 2000632. [Google Scholar] [CrossRef]
- Li, Q.; Liu, Y.; Dai, X.; Jiang, W.; Zhao, H. Nanozymes Regulate Redox Homeostasis in ROS-Related Inflammation. Front. Chem. 2021, 9, 740607. [Google Scholar] [CrossRef]
- Lu, Y.-H.; Geng, J.-H.; Wu, D.-W.; Chen, S.-C.; Hung, C.-H.; Kuo, C.-H. Betel Nut Chewing Decreased Calcaneus Ultrasound T-Score in a Large Taiwanese Population Follow-Up Study. Nutrients 2021, 13, 3655. [Google Scholar] [CrossRef]
- Willetts, S.; Foley, D.W. True or False? Challenges and Recent Highlights in the Development of Aspirin Prodrugs. Eur. J. Med. Chem. 2020, 192, 112200. [Google Scholar] [CrossRef]
- Zhou, B.; Lin, W.; Long, Y.; Yang, Y.; Zhang, H.; Wu, K.; Chu, Q. Notch Signaling Pathway: Architecture, Disease, and Therapeutics. Sig. Transduct. Target. Ther. 2022, 7, 1–33. [Google Scholar] [CrossRef] [PubMed]
- Chen, Q.; Li, Q.; Liang, Y.; Zu, M.; Chen, N.; Canup, B.S.B.; Luo, L.; Wang, C.; Zeng, L.; Xiao, B. Natural Exosome-like Nanovesicles from Edible Tea Flowers Suppress Metastatic Breast Cancer via ROS Generation and Microbiota Modulation. Acta Pharm. Sin. B 2022, 12, 907–923. [Google Scholar] [CrossRef]
- Kopustinskiene, D.M.; Jakstas, V.; Savickas, A.; Bernatoniene, J. Flavonoids as Anticancer Agents. Nutrients 2020, 12, 457. [Google Scholar] [CrossRef]
- Kastenhuber, E.; Lowe, S. Putting P53 in Context. Cell 2017, 170, 1062–1078. [Google Scholar] [CrossRef]
- Hu, J.; Cao, J.; Topatana, W.; Juengpanich, S.; Li, S.; Zhang, B.; Shen, J.; Cai, L.; Cai, X.; Chen, M. Targeting Mutant P53 for Cancer Therapy: Direct and Indirect Strategies. J. Hematol. Oncol. 2021, 14, 157. [Google Scholar] [CrossRef]
- Hassin, O.; Oren, M. Drugging P53 in Cancer: One Protein, Many Targets. Nat. Rev. Drug Discov. 2023, 22, 127–144. [Google Scholar] [CrossRef]
- Fang, D.; Hu, H.; Zhao, K.; Xu, A.; Yu, C.; Zhu, Y.; Yu, N.; Yao, B.; Tang, S.; Wu, X.; et al. MLF2 Negatively Regulates P53 and Promotes Colorectal Carcinogenesis. Adv. Sci. 2023, 10, e2303336. [Google Scholar] [CrossRef]
- Marvalim, C.; Datta, A.; Lee, S.C. Role of P53 in Breast Cancer Progression: An Insight into P53 Targeted Therapy. Theranostics 2023, 13, 1421–1442. [Google Scholar] [CrossRef] [PubMed]
- Tiwari, B.; Jones, A.E.; Abrams, J.M. Transposons, P53 and Genome Security. Trends Genet. 2018, 34, 846–855. [Google Scholar] [CrossRef]
- Yang, X.; Wang, Z.; Zandkarimi, F.; Liu, Y.; Duan, S.; Li, Z.; Kon, N.; Zhang, Z.; Jiang, X.; Stockwell, B.R.; et al. Regulation of VKORC1L1 Is Critical for P53-Mediated Tumor Suppression through Vitamin K Metabolism. Cell Metab. 2023, 35, 1474–1490.e8. [Google Scholar] [CrossRef]
- Klimovich, B.; Meyer, L.; Merle, N.; Neumann, M.; König, A.M.; Ananikidis, N.; Keber, C.U.; Elmshäuser, S.; Timofeev, O.; Stiewe, T. Partial P53 Reactivation Is Sufficient to Induce Cancer Regression. J. Exp. Clin. Cancer Res. 2022, 41, 80. [Google Scholar] [CrossRef]
- Wang, M.; Attardi, L.D. A Balancing Act: P53 Activity from Tumor Suppression to Pathology and Therapeutic Implications. Annu. Rev. Pathol. 2022, 17, 205–226. [Google Scholar] [CrossRef]
- Huang, J. Current Developments of Targeting the P53 Signaling Pathway for Cancer Treatment. Pharmacol. Ther. 2021, 220, 107720. [Google Scholar] [CrossRef]
- Yan, W.; Zhang, T.; Li, S.; Wang, Y.; Zhu, L.; Cao, Y.; Lai, X.; Huang, H. Oxidative Stress and Endoplasmic Reticulum Stress Contributes to Arecoline and Its Secondary Metabolites-Induced Dyskinesia in Zebrafish Embryos. Int. J. Mol. Sci. 2023, 24, 6327. [Google Scholar] [CrossRef] [PubMed]
- Im, K.H.; Baek, S.A.; Choi, J.; Lee, T.S. Antioxidant, Anti-Melanogenic and Anti-Wrinkle Effects of Phellinus vaninii. Mycobiology 2019, 47, 494–505. [Google Scholar] [CrossRef] [PubMed]
- Byun, N.; Heo, M.-R.; Yim, S.-H. Correlation of Anti-Wrinkling and Free Radical Antioxidant Activities of Areca Nut with Phenolic and Flavonoid Contents. Food Sci. Technol. 2021, 41, 1041–1049. [Google Scholar] [CrossRef]
- Wieland, L.S.; Moffet, I.; Shade, S.; Emadi, A.; Knott, C.; Gorman, E.F.; D’Adamo, C. Risks and Benefits of Antioxidant Dietary Supplement Use during Cancer Treatment: Protocol for a Scoping Review. BMJ Open 2021, 11, e047200. [Google Scholar] [CrossRef] [PubMed]
- Luo, M.; Zhou, L.; Huang, Z.; Li, B.; Nice, E.C.; Xu, J.; Huang, C. Antioxidant Therapy in Cancer: Rationale and Progress. Antioxidants 2022, 11, 1128. [Google Scholar] [CrossRef] [PubMed]
- Yenjit, P.; Issarakraisila, M.; Intana, W.; Chantrapromma, K. Fungicidal Activity of Compounds Extracted from the Pericarp of Areca catechu against Colletotrichum Gloeosporioides in Vitro and in Mango Fruit. Postharvest Biol. Technol. 2010, 55, 129–132. [Google Scholar] [CrossRef]
- Choi, J.S.; Jung, H.C.; Baek, Y.J.; Kim, B.Y.; Lee, M.W.; Kim, H.D.; Kim, S.W. Antibacterial Activity of Green-Synthesized Silver Nanoparticles Using Areca catechu Extract against Antibiotic-Resistant Bacteria. Nanomaterials 2021, 11, 205. [Google Scholar] [CrossRef] [PubMed]
- Jam, N.; Hajimohammadi, R.; Gharbani, P.; Mehrizad, A. Antibacterial Activity of Punica granatum L. and Areca Nut (P.A) Combined Extracts against Some Food Born Pathogenic Bacteria. Saudi J. Biol. Sci. 2022, 29, 1730–1736. [Google Scholar] [CrossRef] [PubMed]
- Luu, L.; Bettridge, J.; Christley, R.M.; Melese, K.; Blake, D.; Dessie, T.; Wigley, P.; Desta, T.T.; Hanotte, O.; Kaiser, P.; et al. Prevalence and Molecular Characterisation of Eimeria Species in Ethiopian Village Chickens. BMC Vet. Res. 2013, 9, 208. [Google Scholar] [CrossRef] [PubMed]
- Spolski, R.; Li, P.; Leonard, W.J. Biology and Regulation of IL-2: From Molecular Mechanisms to Human Therapy. Nat. Rev. Immunol. 2018, 18, 648–659. [Google Scholar] [CrossRef] [PubMed]
- Tchitchek, N.; Nguekap Tchoumba, O.; Pires, G.; Dandou, S.; Campagne, J.; Churlaud, G.; Fourcade, G.; Hoffmann, T.W.; Strozzi, F.; Gaal, C.; et al. Low-Dose IL-2 Shapes a Tolerogenic Gut Microbiota That Improves Autoimmunity and Gut Inflammation. JCI Insight 2022, 7, e159406. [Google Scholar] [CrossRef] [PubMed]
- Molan, A.L.; Waghorn, G.C.; Min, B.R.; McNabb, W.C. The Effect of Condensed Tannins from Seven Herbages on Trichostrongylus colubriformis Larval Migration in Vitro. Folia Parasitol. 2000, 47, 39–44. [Google Scholar] [CrossRef] [PubMed]
- Saeed, M.A.; Beveridge, I.; Abbas, G.; Beasley, A.; Bauquier, J.; Wilkes, E.; Jacobson, C.; Hughes, K.J.; El-Hage, C.; O’Handley, R.; et al. Systematic Review of Gastrointestinal Nematodes of Horses from Australia. Parasites Vectors 2019, 12, 188. [Google Scholar] [CrossRef] [PubMed]
- Derosa, G.; Maffioli, P.; D’Angelo, A.; Di Pierro, F. A Role for Quercetin in Coronavirus Disease 2019 (COVID-19). Phytother. Res. 2021, 35, 1230–1236. [Google Scholar] [CrossRef] [PubMed]
- Lazos, J.P.; Piemonte, E.D.; Lanfranchi, H.E.; Brunotto, M.N. Characterization of Chronic Mechanical Irritation in Oral Cancer. Int. J. Dent. 2017, 2017, 6784526. [Google Scholar] [CrossRef]
- Piemonte, E.; Lazos, J.; Belardinelli, P.; Secchi, D.; Brunotto, M.; Lanfranchi-Tizeira, H. Oral Cancer Associated with Chronic Mechanical Irritation of the Oral Mucosa. Med. Oral Patol. Oral Cir. Bucal 2018, 23, e151–e160. [Google Scholar] [CrossRef]
- Senevirathna, K.; Pradeep, R.; Jayasinghe, Y.A.; Jayawickrama, S.M.; Illeperuma, R.; Warnakulasuriya, S.; Jayasinghe, R.D. Carcinogenic Effects of Areca Nut and Its Metabolites: A Review of the Experimental Evidence. Clin. Pract. 2023, 13, 326–346. [Google Scholar] [CrossRef]
- Chang, Y.-C.; Hu, C.-C.; Lii, C.-K.; Tai, K.-W.; Yang, S.-H.; Chou, M.-Y. Cytotoxicity and Arecoline Mechanisms in Human Gingival Fibroblasts in Vitro. Clin. Oral Investig. 2001, 5, 51–56. [Google Scholar] [CrossRef] [PubMed]
- Xu, M.; Su, S.; Jiang, S.; Li, W.; Zhang, Z.; Zhang, J.; Hu, X. Short-Term Arecoline Exposure Affected the Systemic Health State of Mice, in Which Gut Microbes Played an Important Role. Ecotoxicol. Environ. Saf. 2023, 259, 115055. [Google Scholar] [CrossRef] [PubMed]
- Ko, A.M.-S.; Tu, H.-P.; Ko, Y.-C. Systematic Review of Roles of Arecoline and Arecoline N-Oxide in Oral Cancer and Strategies to Block Carcinogenesis. Cells 2023, 12, 1208. [Google Scholar] [CrossRef] [PubMed]

| Extracts/Ingredients | Functions | Model | Mechanism | Dose | Reference | |
|---|---|---|---|---|---|---|
| Areca nut extract | Aqueous fraction | Anti-inflammatory | Mice and rats | ↓ Arachidonic acid metabolism, ↑ degradation and/or inactivation of PGE2 | 100 mg/kg (p.o.) | [19] |
| Aqueous fraction | Anti-depressant | Rats | ↑ Secretion of serotonin | 10 mg/kg (i.p.) | [69] | |
| Hexane fraction | Anti-depressant | Mice and rats | ↓ MAOs | 2.5 mg/kg (i.p.) | [70] | |
| 70% aqueous methanol extract | Hypolipidemic | Rats | ↓ pCEase and pancrelipase | 10 mg/kg (i.g.) | [71] | |
| 80% (v/v) aqueous ethanol extract | Anti-allodynic | Rats | ↑ α-2 adrenoceptors and 5-HT7 receptors | 300 µg (intrathecal administration) | [72] | |
| 90% aqueous methanol extract | Hypolipidemic | Rats | ↓ ACAT and pCEase activity | 0.5% (p.o.) | [73] | |
| Methanol extract | Anti-bacterial | In vitro | ↑ Destroying the cell wall | [18] | ||
| Ethylene extract | Deworming | In vitro | ↓ Carbohydrate metabolism of the parasite | [74] | ||
| Areca nut extract | Deworming | Chicks | ↓ The causative agent Eimeria tenella | 100 mg/kg (p.o.) | [75] | |
| Anti-tumor | Mice | ↑ ROS, autophagy, and lysosomal formation | 20 mg/kg (i.p.) | [76] | ||
| Pericarpium arecae extract | The improvement of intestinal peristalsis | Guinea pigs | ↑ Cholinergic M3 receptors | 1 g/mL concentrated stock solution of crude drug (3% concentration) | [77] | |
| Semen Arecae and Charred Semen Arecae extract | Gastrointestinal protection | Rts | ↓ Cholecystokinin mRNA expressions, ↑ serum levels of substance P and motilin | 3 g/kg (p.o.) | [78] | |
| Areca nuts powder extracted with supercritical carbon dioxide | Deworming | Chicks | ↓ Eimeria tenella | 100 mg/kg (p.o.) | [75] | |
| Ethylene extract | Deworming | In vitro | ↓ Carbohydrate metabolism | 0.5 mg/mL | [74] | |
| Crude aqueous-methanol extract | Deworming | Sheep | ↓ Hatching of parasite eggs, the development of the infected larval stage, and the physiological functions of the parasite | 0.33 g/kg (p.o.) | [79] | |
| Alkaloids | Total alkaloids | Analgesia | Mice | ↓ COX-2 expression | 100 mg/kg (i.g.) | [80] |
| Arecoline | Anxiolytic | Zebrafish | ↑ Norepinephrine, ↑ Serotonin | 10 mg/mL | [81] | |
| Refreshing | ↑ Parasympathetic nerves | [82] | ||||
| Zebrafish | ↑ Central M receptors | 0.01 ppm | [83] | |||
| Rats | ↑ HPA axis | 0.2 mg/kg (i.p.) | [84] | |||
| Rats | ↑ VTA dopaminergic neurons | 0.2 mg/kg (i.v.) | [85] | |||
| Anti-bacterial | In vitro | ↓ Bacillus subtilis, Enterococcus faecalis, Escherichia coli, Pseudomonas aeruginosa, Staphylococcus aureus, Streptococcus | [86] | |||
| Relieve schizophrenia | Mice | ↓ Cerebral demyelination | 2.5 mg/kg/day (p.o.) | [87] | ||
| Relieve Alzheimer’s disease | Rats | ↑ Cerebral blood flow and metabolism | 2 mg/kg (p.o.) | [88] | ||
| Hormone levels regulation | Mice | ↑ Muscarinic cholinergic receptors | 10 mg/kg (i.p.) | [89] | ||
| Anti-tumor | BCC-1/KMC and HaCaT | ↓ IL-6, ↑ P53 and apoptosis | 30 μg/mL | [90] | ||
| In vitro | Alters the cell cycle, ↓ cell viability, ↓ cancerous prostate cells | 0.4 mM | [91] | |||
| Gastrointestinal protection | Grass carp | ↑ M3, ↑ Keap1a/Nrf2 signaling pathway, ↓ RhoA/ROCK signaling pathway | 1 mg/kg (p.o.) | [92] | ||
| Anti-osteoporotic | Mice | ↑ Osteoblast differentiation, ↓ osteoclastogenesis | 5 mg/kg (p.o.) | [93] | ||
| Arecaidine | Refreshing | In vitro | ↓ GABA uptake | [94] | ||
| Guvacine | ||||||
| Phenols | Total phenols | Hypoglycemic | In vitro | ↓ α-glucosidase activity | IC50 1.50 ± 0.31 μg/mL | [95] |
| Anti-osteoporotic | Rats | ↓ 5-HT, ↑ bone resorption and formation | 400 mg/kg (i.g.) | [96] | ||
| In vitro | ↑ Proliferation, differentiation, and mineralization of osteoblasts | 25 µg/mL | [37] | |||
| Anti-fatigue | Mice | ↑ Lactate dehydrogenase, superoxide dismutase, and catalase activities, ↓ malondialdehyde content | 20 mg/kg (i.g.) | [97] | ||
| Anti-oxidant | RAW264.7 cells | ↓ MAPK pathway, ↑ Nrf2/HO-1 anti-oxidant pathways | 40 µg/mL | [98] | ||
| Anti-viral | In vitro | ↓ HIV type 1 protease | 0.2 mg/mL | [99] | ||
| Procyanidins | Analgesia | Mice and rats | ↓ Plasma protein extravasation and inflammation in the dura | 250 mg/kg (p.o.) | [58] | |
| Gastrointestinal protection | Mice | ↓ Th2 responses, ↑ induction function of myeloid-derived suppressor cells | 0.05% in water | [100] | ||
| Hypoglycemic | Mice | ↓ G6Pase and PEPCK | 10 mg/kg/day (i.g.) | [101] | ||
| Anti-photoaging | Mice | ↓ Cyclooxygenase-2, matrix metalloproteinase, and oxidative stress | 10 mg/kg (p.o.) | [102] | ||
| Anti-hypertensive | In vitro | ↓ Angiotensin-converting enzyme | IC50 1.51 ± 0.65 mg/mL | [103] | ||
| Anti-viral | In vitro | ↓ HIV type 1 protease | 0.2 mg/mL | [99] | ||
| Isorhamnetin | Anti-depressant | Mice | ↓ MAOs | 0.2 mL/10 g (i.g., concentration of 2.5 mg/mL) | [104] | |
| Chrysoeriol Luteolin Chrysophanol | ||||||
| Syringic acid | Anti-oxidant | In vitro | ↓ DPPH radical scavenging activity, hydroxyl radical scavenging activity, and reducing power | EC50 0.409 mg/mL and 0.188 mg/mL, respectively | [36] | |
| Epicatechin | ||||||
| Anti-hypertensive | In vitro | ↓ Angiotensin-converting enzyme | IC50 1.51 ± 0.65 mg/mL | [103] | ||
| Catechin | ||||||
| Arecatannin | Anti-viral | In vitro | ↓ HIV type 1 protease | 0.2 mg/mL | [99] | |
| Triterpenes and Steroids | Fernenol | Hypoglycemic | In vitro | ↓ α-glucosidase activity | IC50 1.50 ± 0.31 μg/mL | [95] |
| Arundoin | EC50 47.5 mg/L | |||||
| The mixture of stigmasterol and β-sitosterol | EC50 56.7 mg/L | |||||
| Polysaccharide | Anti-inflammation | Raw264.7 cell | ↓ NO production | IC50 85.64 mg/mL | [105] | |
| Anti-oxidant | In vitro | ↓ DPPH/hydroxyl radicals | 2 mg/mL | [11] | ||
| Fatty acids | Anti-bacterial | In vitro | ↓ Bacillus subtilis, Enterococcus faecalis, Escherichia coli, Pseudomonas aeruginosa, Staphylococcus aureus, Streptococcus | [86] | ||
| Areca fruit | Relieve epilepsy | Human | ↑ Muscarinic receptor | Three nuts/day | [106] | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sun, H.; Yu, W.; Li, H.; Hu, X.; Wang, X. Bioactive Components of Areca Nut: An Overview of Their Positive Impacts Targeting Different Organs. Nutrients 2024, 16, 695. https://doi.org/10.3390/nu16050695
Sun H, Yu W, Li H, Hu X, Wang X. Bioactive Components of Areca Nut: An Overview of Their Positive Impacts Targeting Different Organs. Nutrients. 2024; 16(5):695. https://doi.org/10.3390/nu16050695
Chicago/Turabian StyleSun, Huihui, Wenzhen Yu, Hu Li, Xiaosong Hu, and Xiaofei Wang. 2024. "Bioactive Components of Areca Nut: An Overview of Their Positive Impacts Targeting Different Organs" Nutrients 16, no. 5: 695. https://doi.org/10.3390/nu16050695
APA StyleSun, H., Yu, W., Li, H., Hu, X., & Wang, X. (2024). Bioactive Components of Areca Nut: An Overview of Their Positive Impacts Targeting Different Organs. Nutrients, 16(5), 695. https://doi.org/10.3390/nu16050695